Abstract
Histologic grade is an important determinant in clinical outcome of human osteosarcoma (OS). In this study, the histologic characteristics of primary and metastatic canine OS were evaluated using a new classification system. Histologic characteristics were classified in 166 primary and 34 metastatic canine OS. Prognostic variables for clinical outcome were determined using multivariate analysis. Most OS were histologically characterized by severe to extreme cellular pleomorphism, a variable number of mitoses, small to moderate amounts of matrix, a high percentage of tumor cells, and minimal to moderate amounts of necrosis. Tumor invasion into vessels was present in 117/152 (71%) tumors, and 12/50 (24%) of the regional lymph nodes had evidence of metastasis. Classification of the 166 tumors resulted in seven (4%) grade I, 34 (21%) grade II, and 125 (75%) grade III OS. In the multivariate analysis, histologic grade III OS and elevated pretreatment plasma alkaline phosphatase (AP) levels were independent predictors of clinical outcome. Dogs with high-grade tumors and elevated AP should be carefully evaluated for the presence of metastatic disease before starting adjunctive therapy protocols.
Canine osteosarcoma (OS) is the most common bone tumor in dogs and is characterized by locally aggressive and highly metastatic behavior.8 Primary control of the tumor, consisting of amputation of the affected limb or resection of the tumor, is associated with prolonged survival when adjunctive chemotherapy is administrated.7,35 The estimated 1-year survival rate for dogs having an amputation as their only treatment is 11.5%,35 but this rate increases to approximately 50% after appropriate adjunctive chemotherapy protocols.5,6,27
By definition, OS is a malignant spindle-cell tumor characterized by the formation of osteoid or immature bone or both by the tumor cells.17,25,26 Canine OS is divided into several morphologic subclasses: osteoblastic, fibroblastic, chondroblastic, and teleangectatic. The histologic features of OS, however, vary greatly from tumor to tumor and within tumors themselves. Multiple types of well- and undifferentiated cells and matrix are often observed in a single tumor,16,25,26 which may be why subclassification of OS has not yielded significant differences in the prognosis of human and canine OS.18,40
In human OS, histologic grading of the tumor, in contrast to subclassification, serves as a good indicator for prognosis.18 Histologic tumor grade may also be important in canine OS and could be a criterion for the application of surgery and adjuvant chemotherapy. Histologic tumor grading was associated with increased survival times (ST) and disease-free intervals (DFI) in various other canine sarcomas, such as synovial cell sarcoma,39 multilobular osteochondrosarcoma,10 and hemangiosarcoma.28 Histologic grading, however, has not been widely used as a prognosticator in canine OS and has failed to be a significant indicator for decreased survival in flat and irregular bones, including the mandible.19,36 However, previous studies have included a relatively small number of dogs and a subgroup of OS (mandibular) that seemed to have a distinctly better clinical outcome than does OS of other locations.36
In the present study, the importance of histologic grading as a significant indicator of the clinical outcome of canine OS was evaluated. Because a grading system to evaluate canine OS was not previously available, such a grading system was developed and tested on 166 dogs with OS.
Materials and Methods
Animals
All dogs with OS that were presented between 1985 and 1999 at the Utrecht University Department of Veterinary Pathology were eligible to enter the study if historic paraffin-embedded material or formalin- or ethanol-fixed material was available at the time of evaluation.
Clinical data
Clinical data evaluated included breed, sex, age (years), weight (kg), affected bone, location of tumor (left, right, or middle), procurement of a fine-needle aspiration biopsy (FNAB), plasma alkaline phosphatase (AP) level corrected for the steroid-induced fraction (normal range, 40–120 U/liter),38 presence of metastases at time of diagnosis, and type of surgical and medical therapy. Marginal resection was defined as surgical removal of the tumor but microscopic tumor disease at the margin of the excised specimen. If the margins were free of tumor, the tumor was categorized as totally resected. Breeds were categorized as small (<10 kg), medium (11–20 kg), large (20–40 kg), and giant (>40 kg).26
Biopsy specimens
Tumor tissue samples were collected at surgery or necropsy immediately after euthanasia. Biopsy specimens were obtained using a Michele trephine or by incision of the tumor. Soft, incisable biopsy specimens were fixed in neutral buffered 10% formalin and processed for routine histologic examination. Hard specimens were fixed in 70% ethanol, embedded in methylmethacrylate, and cut on a model K microtome (R. Jung AG, Heidelberg, Germany). All histologic slides were stained with hematoxylin and eosin and von Kossa stain.4
Histologic analysis
All histologic samples were evaluated via blind study by one pathologist (M. Kik). Tumors were defined as OS when osteoid was produced by the tumor cells. Histologic subtypes of OS were defined as osteoblastic, chondroblastic, fibroblastic, teleangectatic, or combinations thereof.26 Histologic variables included tumor cell pleomorphism, amount of tumor matrix, tumor cell density, tumor necrosis, estimated number of multinucleated giant cells, estimated whirl formation, number of mitoses, tumor growth into vessels, and tumor grade. All variables were scored subjectively using a predetermined classification system, adapted from previously reported human13 and veterinary19,26 systems.
Percentage difference in tumor cell pleomorphism (TCP) was evaluated on a scale of 0 to 4 (0 = all nuclei identical; 1 = less than 25% TCP; 2 = 25–50% TCP; 3 = 50–75% TCP; 4 = more than 75% TCP). The amount of tumor matrix was evaluated on a scale of 1 to 3 (1 = more than 50% tumor matrix; 2 = 25–50% tumor matrix; 3 = less than 25% tumor matrix). Tumor cell density was evaluated on a scale of 1 to 4 (1 = less than 25% tumor cells; 2 = 25–50% tumor cells; 3 = 50–75% tumor cells; 4 = more than 75% tumor cells). Tumor necrosis was evaluated on a scale of 0 to 3 (0 = no necrosis present; 1 = less than 25% necrosis; 2 = 25–50% necrosis; 3 = 50–75% necrosis; 4 = more than 75% necrosis). The number of multinucleated giant cells (MNGC) was estimated on a scale of 0 to 3 (0 = no MNGC; 1 = minimal number of MNGC; 2 = moderate number of MNGC; 3 = large number of MNGC). The whirl formation was estimated on a scale of 0 to 3 (0 = no whirl formation; 1 = minimal whirl formation; 2 = moderate whirl formation; 3 = maximal whirl formation). Number of mitoses was calculated by adding the number of cells in mitosis of three randomly selected high-power fields (400×). Signs of tumor growth into the vessels or tumor cells within the vessels were evaluated throughout the entire biopsy specimen. If present, lymph nodes were also evaluated for possible metastasis. OS was scored grade III if tumor growth into the vessels or tumor cells within the vessels or lymph nodes were present. Tumor grade was determined according to a classification schedule (Table 1) if tumor growth into the vessels or tumor cells within the vessels or lymph nodes were not present or could not be evaluated.
Classification for tumor grade determination using a predetermined histologic scores for canine osteosarcoma.

Survival data
Of the 166 dogs, 12 were lost to follow-up. Fifty-five dogs were euthanatized. The remaining 99 dogs were used to calculate the following survival data: DFI (days), ST (days), whether the patient was alive at the moment of evaluation, occurrence of metastasis, recurrence of the tumor, and the location of metastases. Follow-up evaluations were performed 4–6 weeks after initial diagnosis, every 3 months for 1 year, and every 6 months thereafter or when clinical signs of recurrence or metastatic disease occurred.
Statistical analysis
Frequency distributions were calculated and categorical data were compared using chi-square analysis. Normally distributed continuous and interval categoric data were analyzed using an analysis of variance. Multivariate analysis was performed using the general linear model procedure to evaluate interactions between different dependent variables and independent variables. Histologic scores of variables of primary tumors were compared with those of their metastases using a paired t-test. If variables were not normally distributed, a logarithmic transformation was performed. If logarithmic transformation did not result in normal distribution a nonparametric test, such as the Wilcoxon signed-rank test, was used for comparison. Histologic scores of core biopsy specimens were compared with those of the primary tumor using the Wilcoxon signed-rank test for paired variables.
The influence of variables on DFI and ST was calculated using Cox proportional hazards multivariate analysis. Factors evaluated included age, weight, sex, tumor location and side (left, right), AP, type of surgical excision, type of chemotherapy, histologic subtype, MNGC, whirl formation, and histologic tumor grade. The analysis was repeated without histologic tumor grade but with the following additional histologic variables: TCP, number of mitoses, necrosis, amount of tumor matrix, tumor cell density, and the presence of tumor cells in the tumor vessels. Dogs that were lost to follow-up, died from non-tumor-related disease, or were free from metastasis at the time of analysis were censored in the survival analysis. Differences with a P value of <0.05 were considered significant.
Results
Clinical data
In total, 175 primary tumors and 34 metastases from 166 dogs were available for analysis. Of the 175 primary samples, nine were obtained using a core biopsy instrument. Forty-five breeds were represented, with a mean age of 7.3 years (range, 6 weeks–13.4 years). A male:female ratio of 1.05 was observed. The weight was known in 141 dogs and averaged 36.6 kg (range, 7–70 kg). Seven dogs were classified as small/medium, 78 as large, and 56 as giant. The most common locations of the OS were distal radius (n = 31), rib (n = 16), proximal humerus (n = 15), distal femur (n = 15), extraskeletal (n = 14), distal tibia (n = 11), mandible (n = 11), and maxilla (n = 11). The tumor was located on the right side in 83 dogs, on the left side in 69 dogs, and in the middle in 8 dogs.
FNAB of the primary tumor was performed in 35 dogs. Mesenchymal tumor or OS was the clinicopathologic diagnosis for 22 (63%) aspirates. The clinicopathologic diagnosis of tumor was not significantly associated with either histologic tumor grade or subtype. Fifteen of 49 dogs (30.6%) had an increased AP level compared with reference values. There were no significant differences in mean AP levels for different tumor grades.
Histologic data
The frequencies of the scores for the histologic variables were calculated for 166 primary tumors and 34 metastases in 166 dogs (Table 2). There were seven grade I (4.2%), 34 grade II (20.5%), and 125 primary grade III (75.3%) OS. The 34 metastases were all grade III tumors. Nine histologic subtypes were observed in this study (Table 3). The type of matrix (osteoid, cartilage, or fibrous tissue) defined the histologic subtype. Of 50 lymph nodes that were evaluated for metastatic disease at presentation, 12 (24%) harbored metastases. One hundred seventeen of the 152 (70.5%) primary tumors for which tumor vessels were available for evaluation and all metastases showed invasion of neoplastic cells into the tumor vessels. Tumor vessel invasion could not be evaluated in 14 primary OS.
Frequencies of histologic scores for variables in 166 primary and 34 metastatic osteosarcomas.
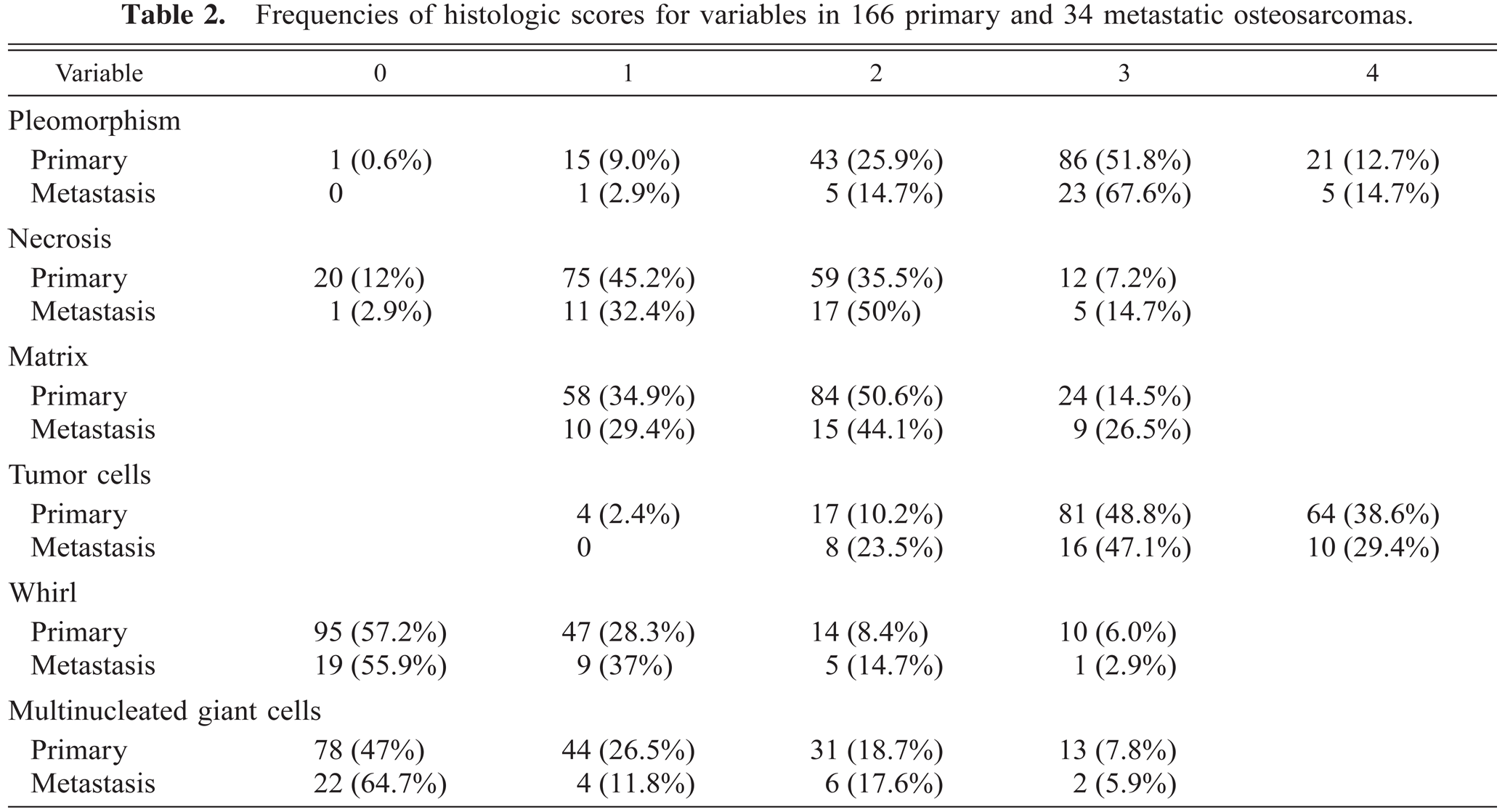
Frequencies of different histologic subtypes of 166 primary and 34 metastatic canine osteosarcomas.
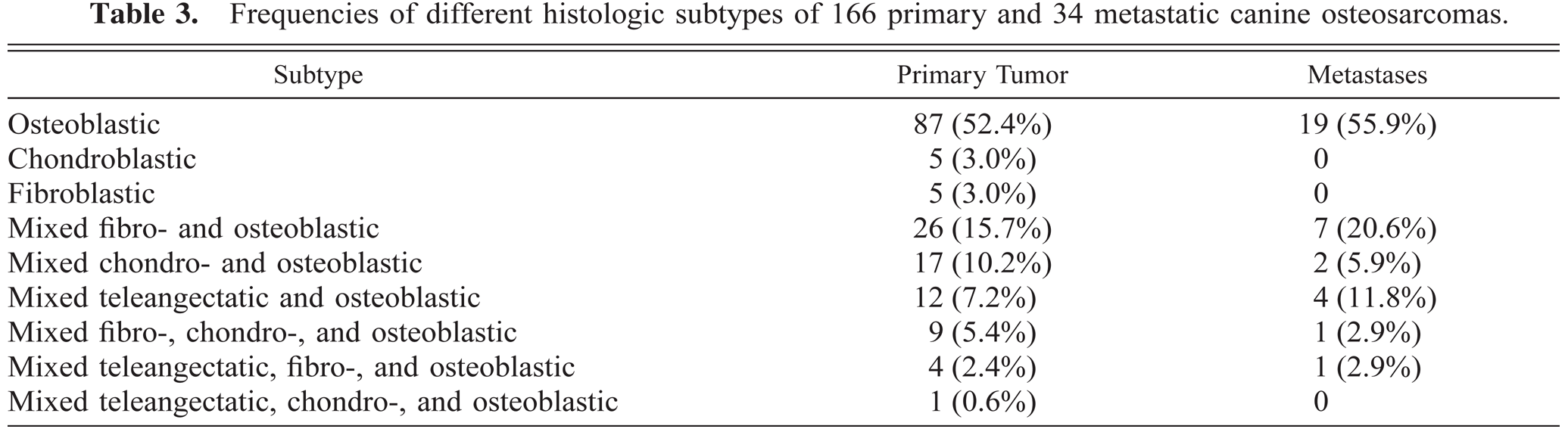
No significant differences were observed among any of the histologic variables evaluated (for which sufficient data for both the primary and the metastatic tumor were available) when the primary tumor was compared with the correspondent metastasis in 29 dogs. The metastasis and primary tumor had a similar histologic subtype and formed an identical matrix in 17 of 29 dogs (59%). All 29 metastases and 26 primary tumors were classified as grade III, and three primary tumors were classified as grade II. When the data for all primary tumors were combined, the mean score for amount of necrosis was significantly higher in the metastases (1.76 ± 0.74) than in the primary OS (1.38 ± 0.79; P = 0.01). The differences among all other histologic variables were not significant.
A core biopsy sample predicted the correct subtype in three of nine (33%) OS and the correct grade in seven of nine (78%) OS. Scores of all the other histologic variables did not differ significantly between the biopsy specimen and primary tumor.
Follow-up and survival data
Of the 99 dogs that were not euthanatized or lost to follow-up, 17 were treated conservatively. Local tumor control consisted of marginal resection of the tumor in eight dogs, limb amputation in 45 dogs, total resection other than limb amputation in 25 dogs, and regional perfusion with cisplatin in four dogs. Sixty-one dogs did not receive any other adjuvant chemotherapy, 27 received a maximum of four doses of lobaplatin (35 mg/m2), four received four doses of carboplatin (150 mg/m2) and doxorubicin (15 mg/m2), one received four doses of cisplatin (60 mg/m2), one received five doses of doxorubicin (30 mg/m2), and one received low-dose methotrexate. Most dogs (n = 138, 89.6%) had already died at the time of the study. Of these 138 dogs, 12 dogs died from causes other than the primary OS or metastatic disease.
Of the 110 dogs on which a postmortem examination was performed, metastases were present at the time of death in 72 dogs (65.5%) and occurred in the lungs in 48 dogs (66.7%), bone in 10 dogs (13.9%), nonpulmonary visceral organs in seven dogs (9.7%), subcutaneous tissues in five dogs (6.9%), and simultaneously in lungs and bone in two dogs (2.8%). There was local recurrence in all eight dogs with marginal resections, half of the 16 dogs with total resections, and one of 44 dogs with amputations for which postmortem examinations were performed.
When survival data for the 99 dogs were evaluated by multivariate analysis, increased hazard ratios (HRs) were observed for incomplete surgical resection (HR = 8.49, 95% confidence interval [CI]: 3.1–23.4; P < 0.001), histologic grade III tumors (HR = 5.9, 95% CI: 2.0–17.3; P = 0.001), and increased plasma AP levels (HR = 1.003, 95% CI: 1.001–1.005; P = 0.003). Increased HR for histologic grade and increased AP were also observed when the data were stratified for type of surgical resection. Dogs not receiving any chemotherapy had a significantly increased HR (HR = 17.3, 95% CI: 4.8–62.0; P < 0.001) compared with dogs that did receive chemotherapy in the group with plasma AP levels within the normal reference range. HRs were not significantly different between these groups in dogs with increased plasma AP levels. Increased HRs for a shorter DFI were observed for dogs with increased plasma AP levels (HR = 1.004, 95% CI: 1.001–1.007; P = 0.005), dogs that did not receive chemotherapy (HR = 14.6, 95% CI: 3.0–71.6; P < 0.001), and histologic high-grade tumors (HR = 3.8, 95% CI: 1.5–9.9; P = 0.006). If instead of tumor grade, the five histologic variables that predicted tumor grade were evaluated, significant HRs for shorter ST were found with the use of chemotherapy (HR = 6.1, 95% CI: 2.1–18.1; P = 0.001), presence of tumor cells in the blood vessels (HR = 8.3, 95% CI: 2.3–30.2; P = 0.001), and increased plasma AP level (HR = 1.003, 95% CI: 1.001–1.005; P = 0.02). For DFI, increased hazard was associated with two variables: tumors with a high number of mitoses (HR = 1.06, 95% CI: 1.02–1.10; P = 0.002) and dog that did not receive chemotherapy (HR = 12, 95% CI: 2.6–51.6; P = 0.002).
Discussion
Although the clinical data presented in this study are comparable to those of previous studies,8,19,20,22,23,26 the histologic data are more difficult to interpret because classification systems were rarely used in previous studies. In 1972, Misdorp and Hart26 presented data for 144 dogs with OS that were evaluated by multivariate analysis. In their study of 144 cases of OS, 47% were osteoblastic, 23% were osteoblastic-chondroblastic, 14% were osteoblastic-fibroblastic, and 15% were osteoblastic-chondroblastic-fibroblastic.26 The findings of the present study are comparable, when teleangectatic OS was not considered a separate group. The importance of the subdivision is artificial because the histologic multipotentiality of the tumor, similar to that in human OS, eliminates any significant differences in DFI and ST among the subgroups.18 Prognosticators that were used earlier, such as fibroblastic25,26 and teleangectatic18 OS, were not associated with a significant better or worse prognosis in this study. Division into subclasses does not appear to be warranted.
Histologic grade provides a better assessment of the biologic aggressiveness of the tumor.13 Existing grading systems were adapted to better suit the canine OS. High grade (grade III) OS in general has a high cell:matrix ratio, poor differentiation of immature matrices, and malignant cytologic characteristics, such as degree of pleomorphism, mitotic index, vascular invasion, amount of necrosis, and invasiveness.13,30 High-grade tumors were associated with significantly decreased ST and DFI in this study, similar to ST and DFI for other mesenchymal tumor types such as hemangiosarcoma28 and splenic,34 synovial cell,39 and feline soft-tissue sarcoma.9 In two studies, tumor grade in a group of dogs with OS was evaluated using multivariate analysis, but higher risk was not associated with higher tumor grade.19,36 Both studies, however, included relatively small populations and one specific tumor site. Tumor grading is a relatively simple procedure without additional costs and should be performed before further therapeutic measures are undertaken. Grade I and grade II OS, which represent approximately 25% of the OS reported in this study, have a significantly better prognosis than grade III OS. All researchers that use multivariate analysis to evaluate whether a certain therapy is successful in treating canine OS should, therefore, be aware that the grade of OS may significantly influence the outcome.
Several specific histologic variables, such as pleomorphism, proportion of tumor cells, amount of matrix, necrosis, and invasiveness, did not show any significant effect on survival data. The histologic variables associated with an increased risk were the number of mitoses and presence of ingrowth of tumor cells in blood vessels (blood vessel invasion). The number of mitoses (or mitotic index) has been used in other studies to calculate a histologic score and appears to be an important determinant of the relative risk calculation for DFI and ST.19,36 Blood vessel invasion was observed by Misdorp and Hart26 in 71.4% of the samples evaluated but was not always associated with macroscopic metastatic disease. This frequency is identical to the results presented here.
Of all the variables that were evaluated as prognostic indicators for survival and DFI using multivariate analysis, only a few appeared to be significant. Although complete surgical resection without adjunctive chemotherapy should be viewed as a purely palliative procedure, it did increase survival compared with that for dogs that had incomplete or no resection at all. From an ethical standpoint, it is debatable whether conservative treatment (i.e., analgesics) is preferable to palliative resection, especially because resection will improve ST and, in the case of appendicular OS, amputation results in excellent function in most dogs.21 The second independent prognosticator was plasma AP level. An increased level of AP was associated with a shorter DFI and ST. This finding is similar to that of studies that evaluated the prognostic importance of AP activity in dogs with appendicular OS.12,15 HR for increased AP in both studies were comparable; increased AP values were not associated with more undifferentiated tumors or tumors with a higher grade.12 The Musculoskeletal Tumor Committee of the Japanese Orthopedic Association has modified the Tumor Node Metastasis (TNM) system to include serum AP levels, because AP value was also a strong prognosticator in human OS.1 This modification would allow a better correlation between survival rate and stage.14 The data obtained by Ehrhart et al.12 and Garzotto et al.15 and in the present study show that this modification may also apply for canine OS.
The response of OS to chemotherapy is variable. Some tumors appear to respond very well to adjuvant chemotherapy, but others show little or no effect. In dogs with AP values within the normal range, chemotherapy was a prognostic factor in this study. Dogs with an increased AP value did not benefit from additional chemotherapy, but dogs with an AP value within the normal range did benefit, similar to the situation for human OS.1,2 The source of the elevated AP value in dogs is bone-specific AP, which is produced by the neoplastic osteoblasts.12 In the present study, the specific isoenzymes were not determined. The results, however, were corrected for steroid-induced AP,38 and none of the dogs had clinical signs of overt liver, gastrointestinal, or bone diseases other than OS. Other liver enzymes were not elevated (data not shown). The exact role of AP in chemoresistance has not been elucidated. Chemorestistant tumor cells overexpressing AP may be multidrug resistant (MDR).11 Human MDR OS cells overexpress P-glycoprotein, which is a negative predictor of clinical outcome.3,33 Canine OS that overexpress AP also may be MDR. The specific relation between AP and P-glycoprotein, however, has not been established.
OS is known as a highly metastatic tumor that supposedly metastasizes through mainly hematogenous routes.29,40 The distribution of metastases was similar to that in previous studies.24,26 In the present study, however, lymphogenous metastases were present in approximately 25% of dogs. The routine evaluation of regional lymph nodes in the staging procedure of the OS is therefore necessary to fully comprehend the metastatic behavior of this tumor type. The TNM system is also applicable for canine OS.
In this study, the histologic variables of the primary tumor and the corresponding metastases were compared. Most histologic variables were identical, and the histologic subtypes corresponded in most cases. The metastasis often is a true reflection of the primary tumor in canine OS.
The advantage of obtaining a histologic biopsy specimen for canine OS is suggested by the results of this study. Published techniques have diagnostic values of approximately 82–94%, depending on the type of biopsy and whether the technique was performed adequately.31,37,41 In this study, all core biopsy specimens were obtained using a Michele trephine and resulted in a correct grade prediction in seven of nine OS cases. Histologic variables did not differ between the biopsy and the primary specimen, but the number of biopsy specimens examined is relatively small. Because histologic grade was predicted correctly in most cases, the evaluation of biopsy specimens is recommended and may aid in the selection and prediction of outcome for certain therapy protocols. Classification of OS is made easier, however, by a larger biopsy specimen, including the edge of the tumor. An incisional biopsy of the softer outer parts of the OS combined with a core biopsy from the calcified inner part will result in the best chances for accurate diagnosis. Ultrasound-guided FNAB of bone lesions is another reported way of obtaining a preliminary diagnosis. Blind FNAB of 35 OS were obtained in this study, and cytologic evidence for mesenchymal tumor or OS was observed in 63% of these samples. The advantages of FNAB over core biopsy are the relative ease and low costs of obtaining the sample. However, limited information about tumor subtype and grade is obtained by FNAB when compared with a core biopsy. We disagree with Samii et al.32 that sedation or anaesthesia, which increases the cost of the procedure, is not necessary, because the procedure is rather painful for the animal.
Canine OS presents with heterogenous histologic and clinical features, of which tumor grade, best predicted by blood vessel invasion and mitotic index, and AP levels seem to be most important for evaluating the prognosis for survival. Dogs with high-grade tumors and elevated AP should be carefully evaluated for the presence of possible metastatic disease before starting adjunctive therapy protocols.
Footnotes
Acknowledgements
The assistance of H. M. W. Priem-de Groen, W. Steenbergen-Botweg, J. E. Rofina, and D. J. Compton is greatly appreciated.
