Abstract
H-type bovine spongiform encephalopathy (H-BSE) is an atypical form of BSE in cattle. During passaging of H-BSE in transgenic bovinized (TgBoPrP) mice, a novel phenotype of BSE, termed BSE-SW emerged and was characterized by a short incubation time and host weight loss. To investigate the biological and biochemical properties of the BSE-SW prion, a transmission study was conducted in cattle, which were inoculated intracerebrally with brain homogenate from BSE-SW–infected TgBoPrP mice. The disease incubation period was approximately 15 months. The animals showed characteristic neurological signs of dullness, and severe spongiform changes and a widespread, uniform distribution of disease-associated prion protein (PrPSc) were observed throughout the brain of infected cattle. Immunohistochemical PrPSc staining of the brain revealed the presence of intraglial accumulations and plaque-like deposits. No remarkable differences were identified in vacuolar lesion scores, topographical distribution patterns, and staining types of PrPSc in the brains of BSE-SW– vs H-BSE–infected cattle. PrPSc deposition was detected in the ganglia, vagus nerve, spinal nerve, cauda equina, adrenal medulla, and ocular muscle. Western blot analysis revealed that the specific biochemical properties of the BSE-SW prion, with an additional 10- to 12-kDa fragment, were well maintained after transmission. These findings indicated that the BSE-SW prion has biochemical properties distinct from those of H-BSE in cattle, although clinical and pathologic features of BSW-SW in cattle are indistinguishable from those of H-BSE. The results suggest that the 2 infectious agents, BSE-SW and H-BSE, are closely related strains.
Keywords
Bovine spongiform encephalopathy (BSE), which belongs to a group of prion or transmissible spongiform encephalopathy (TSE) diseases, is a fatal, progressive neurodegenerative disorder in cattle. The prion disease is characterized by the development of spongiform changes and accumulation of an abnormal isoform of a prion protein (PrPSc), which is thought to be a posttranslational conformational change of the normal, host-encoded cellular prion protein (PrPC) in the brain of infected hosts. 36 Three distinct strains of BSE have been recognized to date, based on the molecular features of the proteinase K–resistant PrPSc (PrPres) in Western blotting: a classical (C) type and atypical high (H) and low (L) molecular mass-type BSEs. 3,10,14,19 The origins of BSEs are still unclear; however, based on epidemiologic data, C-BSE is probably caused by the consumption of meat and bone meal and milk replacer from C-BSE–infected or scrapie-infected ruminants. 43,44 On the contrary, both atypical BSEs have been considered spontaneous or sporadic forms of the disease in cattle over 8 years of age, with similarities to the sporadic form of Creutzfeldt-Jacob disease (CJD). 3,11,19
Experimental transmission of H-BSE by intracerebral challenge has been confirmed in cattle, 1,16,22,23,27,30 in transgenic mice overexpressing bovine PrP, 7,12,32 and in wild-type mice. 5,6 However, transmission to ovinized transgenic mice 2 and Syrian hamsters 28 is inefficient because of the potential existence of a species barrier. Interestingly, the fact that H-BSE can be transmitted to hamster PrP-transgenic mice is an example of overcoming the species barrier. 33 The biological and biochemical properties of H-BSE prions clearly differ from those of C-BSE prions with respect to their transmission to cattle 1,22,23,30 and mice, 5,8 including incubation period, PrPres molecular profiles in Western blotting, neuropathology, and immunohistochemical analysis. However, H-BSE can acquire a C-BSE–like phenotype after serial passages in rodent models. 4,6,42 Recently, serial transmission of a Canadian H-BSE 15 in transgenic bovine PrP-overexpressing (TgBoPrP) mice led to the emergence of a different phenotype of prion strain. 25 The strain, named BSE-SW, was characterized by a shorter incubation period and induced characteristic clinical signs of weight loss and lack of constant chewing of the bedding in infected TgBoPrP mice. 32 The biochemical signature of BSE-SW is the presence of an N-terminally truncated PrPres fragment, PrPres 1, with a molecular weight close that of C-BSE in cattle. 25 This finding suggests the existence of an unidentified form of atypical BSE in cattle 39 or that C-BSE might have developed from an atypical BSE. 4,6,7,13,42 Therefore, back transmission of BSE-SW from TgBoPrP mice into the original host, cattle, was performed to evaluate any alteration or consistency in the biological and biochemical properties of the agent after transmission. In H-BSE transmission studies, PrPSc accumulation was detected throughout the peripheral nervous tissues, adrenal gland, and skeletal muscles of cattle, 22,30,31,34 while lymphoid tissues and enteric nervous system revealed no involvement. 22,30 Therefore, in this study, distribution of PrPSc in the extracerebral tissues of cattle challenged intracerebrally with BSE-SW was assessed by immunohistochemical and biochemical methods compared to H-BSE.
Materials and Methods
Ethics Statement
Animal experiments were performed in strict accordance with the regulations in the Guide for the Care and Use of Laboratory Animals of the National Institute of Animal Health (approval ID: 04-III-6) in accordance with Guidelines for Proper Conduct of Animal Experiments, 2006 by the Science Council of Japan. 37 Procedures involving animal subjects were approved by the Institutional Animal Care and Use Committee at the National Institute of Animal Health (approval ID: 13-055 [cattle transmission study], 15-030 [mouse transmission study], and 15-005 [mouse follicular dendritic cell (FDC) assay]) with all possible effort made to minimize pain and discomfort of the animals. All surgeries were performed under xylazine sedation and ketamine anesthesia in cattle and under sevoflurane anesthesia in mice.
Sources of Inocula and Transmission to Cattle
The BSE-SW strain had emerged after a third intracerebral subpassage of a Canadian H-BSE brain-derived field isolate 15 in TgBoPrP mice. 25 TgBoPrP mice express approximately 8-fold higher PrPC in the brain than cattle. 38 After 2 further passages of the strain in TgBoPrP mice, a 10% brain homogenate (weight/volume) was prepared from whole brains of infected mice at the terminal stage of the disease. Three 3- to 4-month-old female Holstein calves were inoculated intracerebrally with 1 ml of the homogenate. Brains of 3 sham-inoculated heifers culled at 20 months of age served as controls. At necropsy, the brains were removed and fixed for pathologic examination and immunohistochemistry (IHC) or frozen for Western blotting.
Histopathology
Collected brains and cerebella were cut sagittally at the midline. The left cerebral hemisphere, cerebellum, spinal cord at the level of cervical (C8) and lumber (L6) enlargement, and more than 80 peripheral tissue samples were fixed in 10% buffered formalin (pH 7.4) containing 10% methanol at 37°C for 3 days. Coronal slices of each brain were cut serially at 3- to 4-mm thickness. The formalin-fixed tissues were treated with 98% formic acid for 60 minutes at 20°C to reduce infectivity, dehydrated, embedded in paraffin wax, sectioned at 4 μm, stained with hematoxylin and eosin (HE), and used for IHC. Lesion profiles were determined in the HE-stained sections by scoring the vacuolar changes in 17 different brain areas as previously described. 30,41 Selected brain sections were stained with phenol Congo red and examined under a polarizing microscope to confirm amyloid deposition using its characteristic dichroism. 18
PrPSc IHC
Dewaxed sections were placed on silanized glass slides, treated with 3% hydrogen peroxide at 20°C for 10 minutes, incubated with 10 μg/ml of proteinase K (0.03 U/mg; Nakarai, Kyoto, Japan) in phosphate-buffered saline (PBS, pH 7.4) containing 0.1% Triton-X at 20°C for 10 minutes, and immersed in 150 mM sodium hydroxide at 60°C for 10 minutes. 35 After epitope retrieval with a combination of chemical and enzymatic pretreatments, 35 the sections were incubated with monoclonal antibody (mAb) T1 40 or F99/97.6.1 (VMRD, Pullman, WA) followed by an anti-mouse, universal horseradish peroxidase (HRP)–conjugated polymer (Nichirei Histofine Simple Stain MAX-PO [M]; Nichirei, Tokyo, Japan) as the secondary antibody and visualized with 3,3′-diaminobenzidine tetrachloride containing 10 mM imidazole as the chromogen. Peripheral tissue sections were also immunostained using the highly sensitive tyramide signal amplification biotin system (PerkinElmer, Boston, MA) as described previously. 29 Finally, the sections were slightly counterstained with Mayer’s hematoxylin. Negative controls were incubated with nonimmune mouse IgG (1:30 dilution; Dako, Glostrup, Denmark) in PBS (pH 7.4) instead of the primary antibody.
Western Blotting Analysis
Proteins were extracted from the frozen tissues and subjected to Western blotting analysis of PrPres according to previously described methods. 30 Briefly, tissue samples (200 ± 10 mg) were homogenized in PBS using a multibead shocker. The homogenate (125 µl) was mixed with an equal volume of buffer containing 4% (w/v) Zwittergent 3–14 (Merck, Darmstadt, Germany), 1% (w/v) Sarkosyl, 100 mM NaCl, and 50 mM Tris-HCl (pH 7.6) and incubated with 0.25 mg collagenase, followed by incubation with proteinase K (final concentration, 50 μg/ml) at 37°C for 30 minutes. Proteinase K digestion was terminated by the addition of 2 mM 4-(2-aminoethyl) benzenesulfonyl fluoride hydrochloride (Pefabloc; Roche Diagnostics, Basel, Switzerland). The brain samples were mixed with equal volumes of a 2-butanol/methanol mixture (5:1) and centrifuged at 20 000 × g for 10 minutes. PrPres from the peripheral nervous tissues, extracerebral central nervous tissues, and lymphoid tissues was extracted by phosphotungstic acid precipitation as described previously. 30 The extracts were electrophoresed on NuPAGE Novex 12% Bis-Tris gels using NuPAGE MOPS-SDS running buffer (Life Technologies, Carlsbad, CA). The proteins were transferred onto an Immobilon-P PVDF membrane (Millipore, Billerica, MA). The blotted membrane was probed with mAbs P4 (R-Biopharm, Darmstadt, Germany), 6H4 (Prionics, Schlieren, Switzerland), or SAF84 (SPI-bio, Montigny le Bretonneux, France) at 20°C for 60 minutes and then incubated with HRP-conjugated anti-mouse IgG (Jackson ImmunoResearch, West Grove, PA) for 60 minutes at 20°C. Signals were developed with a chemiluminescent substrate (SuperSignal; Thermo Fisher Scientific, Rockford, IL). After proteinase K treatment, some samples were deglycosylated with N-glycosidase F (PNGase F; New England Biolabs, Beverly, MA), according to the manufacturer’s instructions. The signal intensity and glycoform ratio of PrPres were calculated with Fluorochem software (Alpha Innotech, San Leandro, CA).
Infectivity Assay of BSE-SW Prions in TgBoPrP Mice
To estimate the infectivity of BSE-SW and H-BSE prions in the brains of TgBoPrP mice, an end-point titration assay was conducted. BSE-SW–infected and H-BSE–infected TgBoPrP mouse brain homogenate was serially diluted with PBS, and each dilution was intracerebrally inoculated into TgBoPrP mice (Table 1). To calculate the 50% lethal dose in 1 g of cattle brain tissue (LD50/g), the same volumes of 10-fold dilutions of homogenates were inoculated into the right hemisphere. Infectious units (IU; LD50/g of the brain) were calculated by Behrens-Karber’s formula. A standard curve was generated for the relationship between incubation period and LD50/g.
Comparison of Survival Times and Infectious Units in TgBoPrP Mice Infected With a Novel Phenotype of Bovine Spongiform Encephalopathy (BSE-SW) and With H-Type Bovine Spongiform Encepalopathy (H-BSE) as Determined by an End-Point Titration Assay.
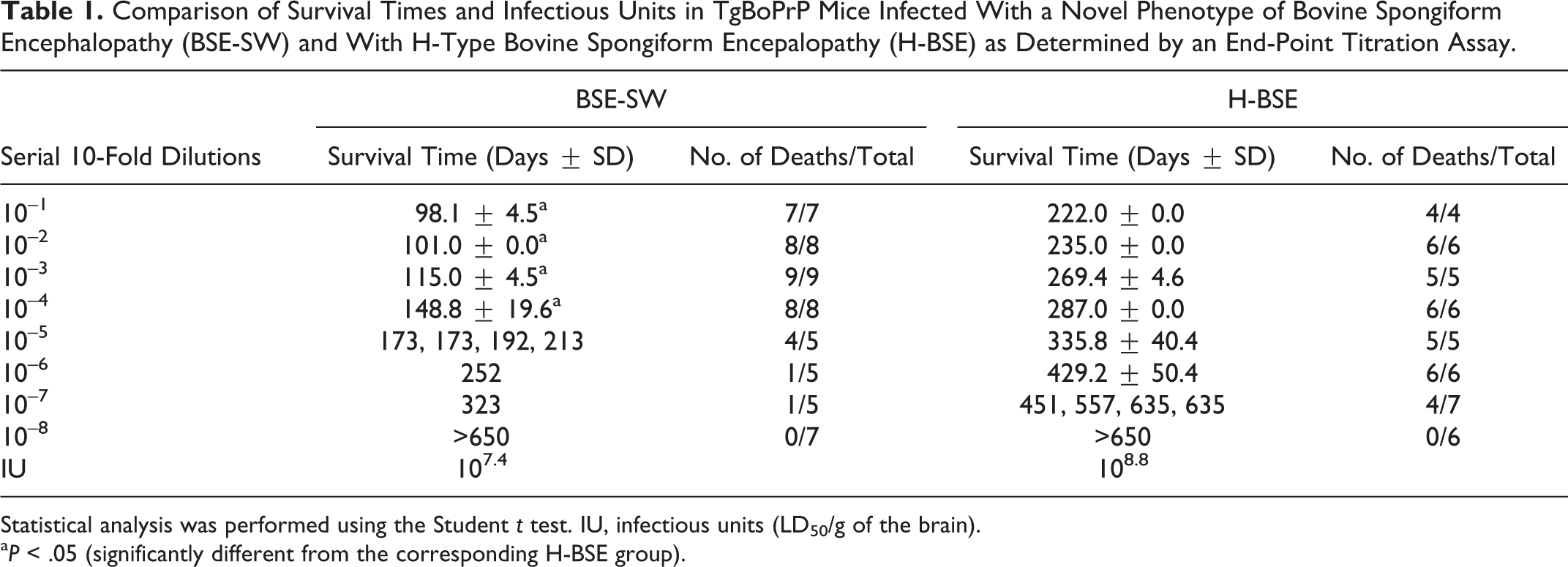
Statistical analysis was performed using the Student t test. IU, infectious units (LD50/g of the brain).
a P < .05 (significantly different from the corresponding H-BSE group).
Back Transmission of BSE-SW Isolates From Cattle Into TgBoPrP Mice
To evaluate disease phenotypes and molecular characteristics of 2 distinct H-BSE and BSE-SW prions before and after passage in cattle, 20 μl of 10% brain homogenates from diseased cattle was intracerebrally injected into TgBoPrP mice. The brains of diseased mice were removed and fixed for pathologic examination or stored at –80°C for Western blotting analysis, as described previously. 25,32
Splenic FDC Bioassay
Brain homogenates (10%) were prepared from C-BSE–, H-BSE–, and BSE-SW–infected cattle as described above. Female outbred ICR (CD-1) mice (n = 5 in each group, 3 weeks old; Japan SLC, Shizuoka, Japan) were intraperitoneally inoculated with 60 µl of 10% brainstem homogenate from cattle infected with C-BSE, H-BSE, or BSE-SW. Inoculated mice were sacrificed at 75 and 150 days postinoculation (dpi) for the splenic FDC assay as a rapid PrPSc detection method. 20 Half of the spleen was frozen for Western blotting, and the remaining half was fixed for histopathology and IHC. PrPSc accumulation in the FDC of the spleen of wild-type mice inoculated with C-BSE at 60 days after inoculation has been confirmed 26 but not with H-BSE at the end of their life span. 5
Results
BSE-SW and H-BSE Induce Similar Clinical Signs but Have Different Incubation Periods
Detailed clinical signs of BSE-SW–infected cattle have already been described elsewhere. 25 Briefly, the animals displayed behavioral changes between 11.5 and 12.5 months after inoculation. The clinical signs of the disease were similar in the 3 challenged animals (case No. 1, code 2263; case No. 2, code 9772; case No. 3, code 2744) and were comparable to those of intracerebrally H-BSE–infected cattle reported in Japan 30 and elsewhere. 1,22,23 Although the number of recipients was too small for statistical analysis, the mean incubation period of 14.8 ± 1.5 (average ± standard deviation) months of BSE-SW was shorter than that of the original H-BSE isolate (18.7 ± 1.6 months), 30 as a result of a difference in the mean clinical duration after disease onset.
BSE-SW and H-BSE Have Similar Pathologic Features in Bovine Brain
Histopathologic analysis revealed that vacuolar lesion scores in 17 different brain areas were similar among the 3 animals (Fig. 1). Neuropil but not neuronal vacuolation was consistent throughout the brain. The highest mean lesion scores were obtained for the thalamic nuclei and neuropil of the central gray matter of the midbrain and the lowest for the caudal cerebral cortices and cerebellar cortex. Spongiform changes were not conspicuous in other brainstem nuclei. Mild vacuolation was detected in the neuropil of the gray matter in the spinal cords of all animals. In addition, amyloid-plaque deposits visualized with Congo red stain under polarized light were scattered throughout the cerebral white matter (data not shown).
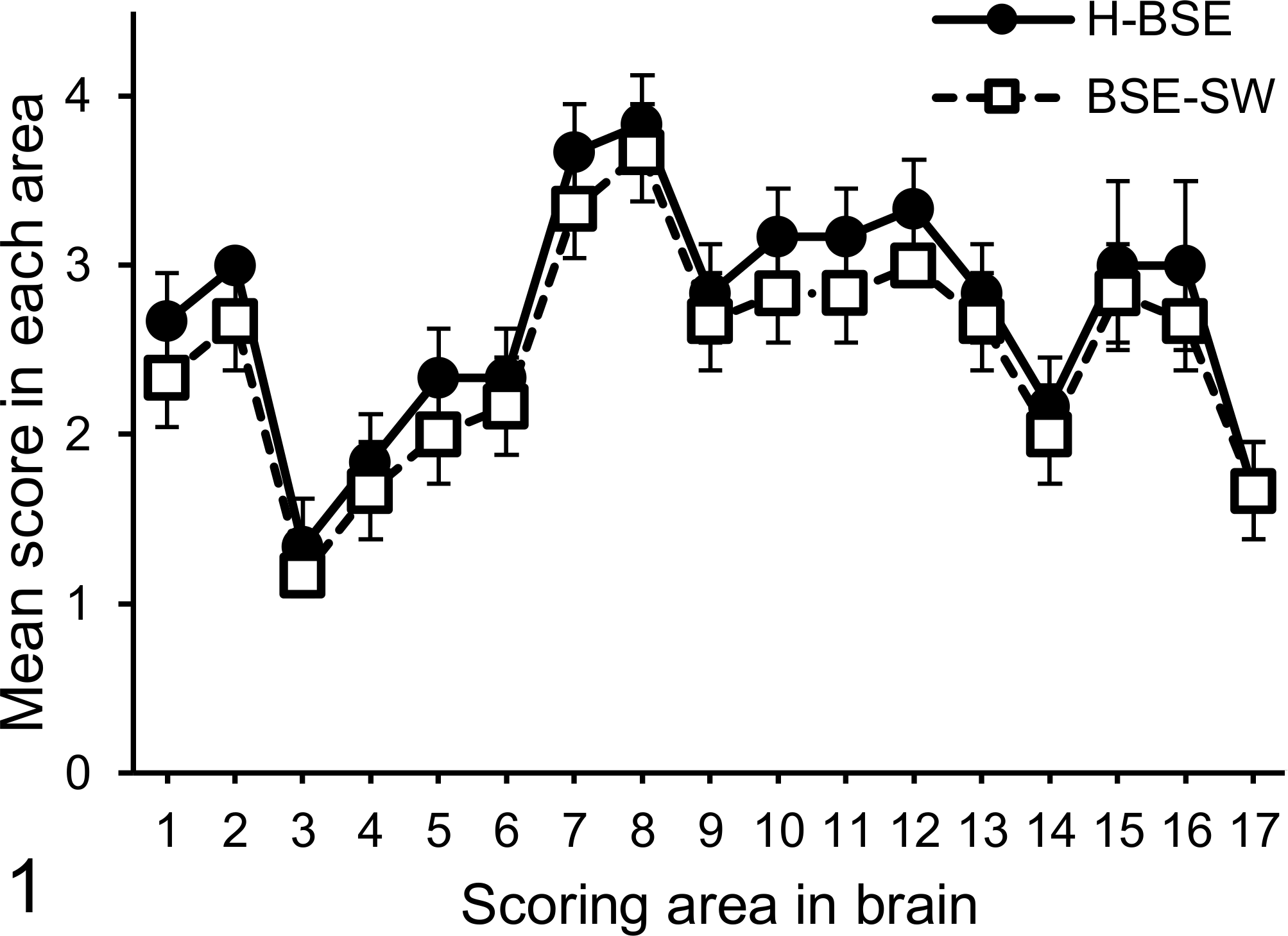
Lesion profiles of cattle inoculated with BSE-SW and H-BSE. Vacuolation was scored on a 0 to 5 scale in the following 17 brain areas: 1, nucleus of the solitary tract; 2, nucleus of the spinal tract of the trigeminal nerve; 3, hypoglossal nucleus; 4, vestibular nuclear complex; 5, cochlear nucleus; 6, cerebellar vermis; 7, central gray matter; 8, superior colliculus; 9, medial geniculate nucleus; 10, hypothalamus; 11, dorsomedial nucleus of the thalamus; 12, ventral intermediate nucleus of the thalamus; 13, frontal cortex; 14, septal nucleus; 15, caudate nucleus; 16, putamen; and 17, claustrum. Data represent the mean ± standard deviation (n = 3). The lesion scores for H-BSE are taken from a previous study. 30
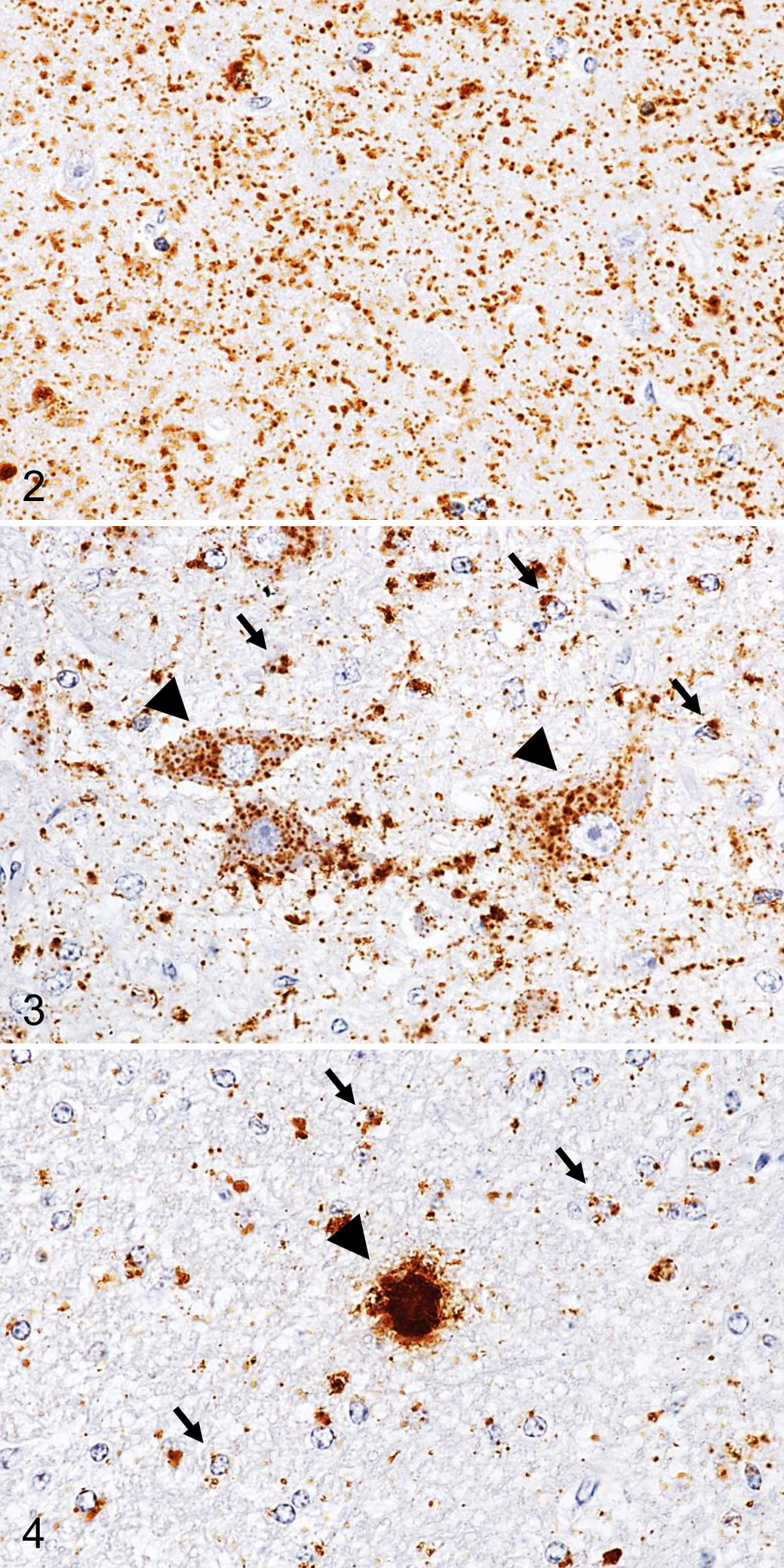
PrPSc IHC demonstrated variable staining patterns, including fine particulate, coarse granular, intraneuronal, perineuronal, intraglial, linear, stellate, and plaques, throughout the brain of each animal. The most conspicuous patterns were fine particulate and coarse granular deposits in the neuropil of the gray matter of the brain and spinal cord (Fig. 2). The most characteristic PrPSc deposit was intraglial type, which was diffusely distributed throughout the white matter of the brain and spinal cord (Fig. 3). Plaques were sparsely detectable in basal ganglia, thalamus, deeper layers of the cerebral cortex, and subcortical white matter (Fig. 4). Stellate-type PrPSc staining was also present in the cerebral cortex.
Biochemical Features of TgBoPrP Mouse-Derived BSE-SW in Cattle
PrPres was widespread throughout the brain and spinal cord (Fig. 5). The specific biochemical properties of PrPres in cattle infected with BSE-SW have been previously described. 25 Briefly, the unglycosylated fragment of PrPres derived from BSE-SW migrated slightly faster on Western blotting than that from H-BSE, which resembles C-BSE in cattle (Suppl. Fig. S1). With mAb SAF84, a multiple banding pattern with an additional 10- to 12-kDa fragment was detected for both BSE-SW and H-BSE samples (Fig. 5 and Suppl. Fig. S1). In contrast to H-BSE in cattle, the BSE-SW PrPres signal was undetectable when analyzed with mAb P4 (Suppl. Fig. S1). After PNGase treatment, deglycosylated PrPres with a molecular mass of approximately 17 kDa was detected, which was similar to PrPres from C-BSE detected with mAb SAF84 and different from that of H-BSE, which was approximately 19 kDa (Suppl. Fig. S2). An additional 10- to 12-kDa PrPres band detected in H-BSE with mAb SAF84 was conserved in BSE-SW. PrPres with mAb 6H4 revealed similar glycoform patterns in BSE-SW and H-BSE (Fig. 6).
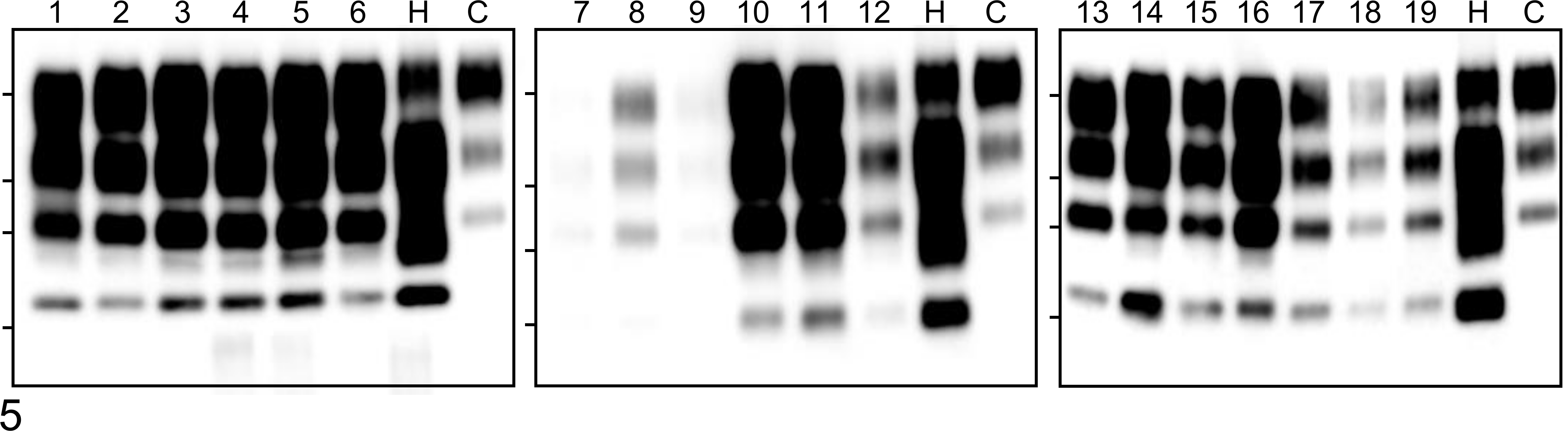
Western blot analysis of proteinase K–digested disease-associated prion protein (PrPres) in the brain and spinal cord from a BSE-SW–infected calf (case No. 2) probed with monoclonal antibody SAF84. Lane numbers are indicated as follows: 1, olfactory bulb; 2, frontal cortex; 3, putamen; 4, parietal cortex; 5, piriform cortex; 6, occipital cortex; 7, frontal white matter; 8, parietal white matter; 9, occipital white matter; 10, caudate nucleus; 11, thalamus; 12, hypothalamus; 13, hippocampus; 14, midbrain (superior colliculus); 15, brainstem at the level of the obex; 16, cerebellar vermis; 17, cerebellar white matter; 18, cervical enlargement (C8); 19, lumber enlargement (L6). As controls, brains of H-BSE– (H) and C-BSE–infected (C) cattle were used. The bars on the left in each panel represent molecular mass markers of 28, 19, 14, and 6 kDa.
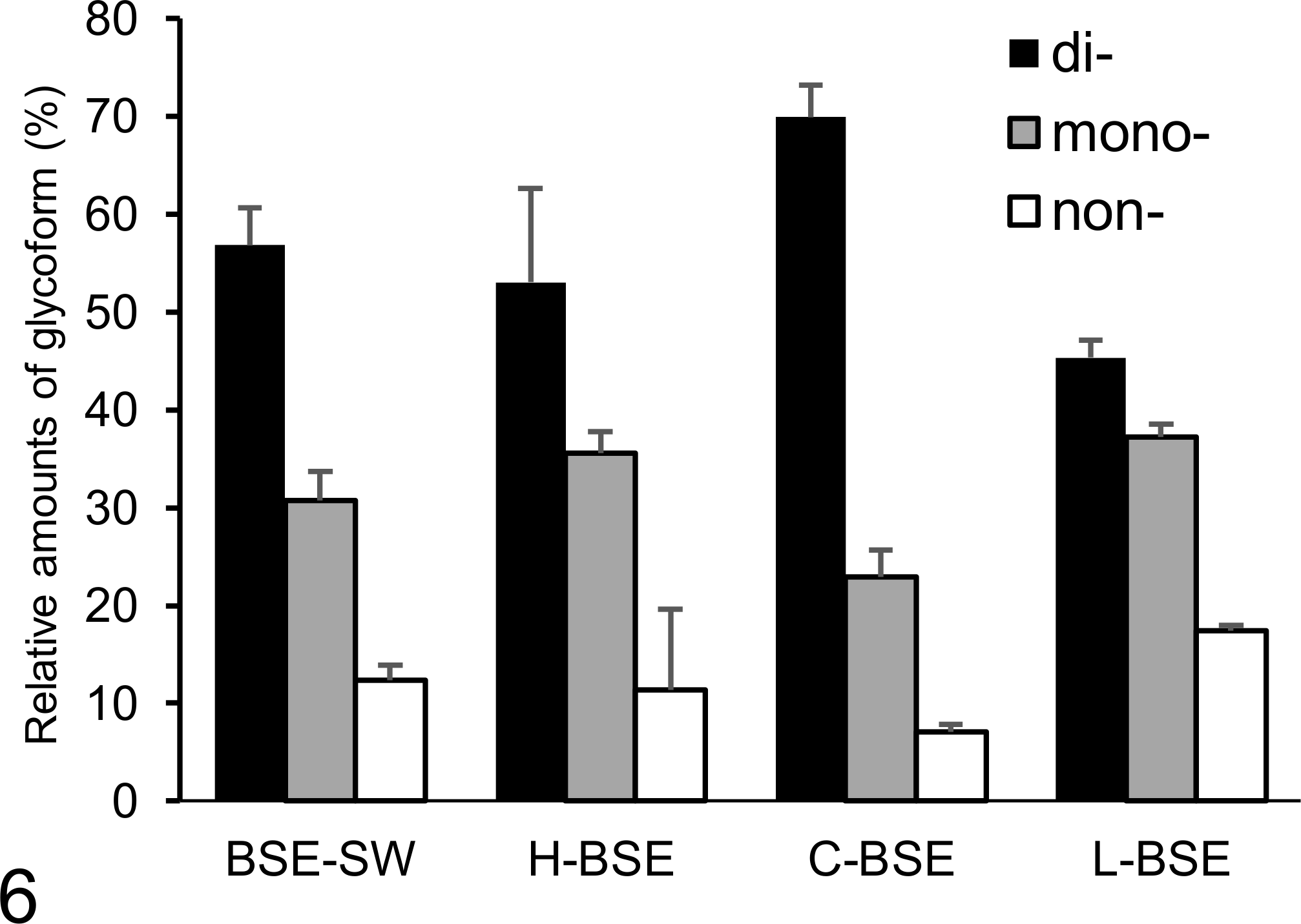
Relative amounts of di-, mono-, and non-glycosylated forms of proteinase K–digested disease-associated prion protein in BSE-SW, H-BSE, C-BSE, and L-BSE prion-infected animals analyzed with monoclonal antibody 6H4. The results are shown as the mean ± standard deviation in triplicate experiments. Bar diagram indicates diglycoslated form (black column), monoglycosylated form (gray column), and non-glycosylated form (white column).
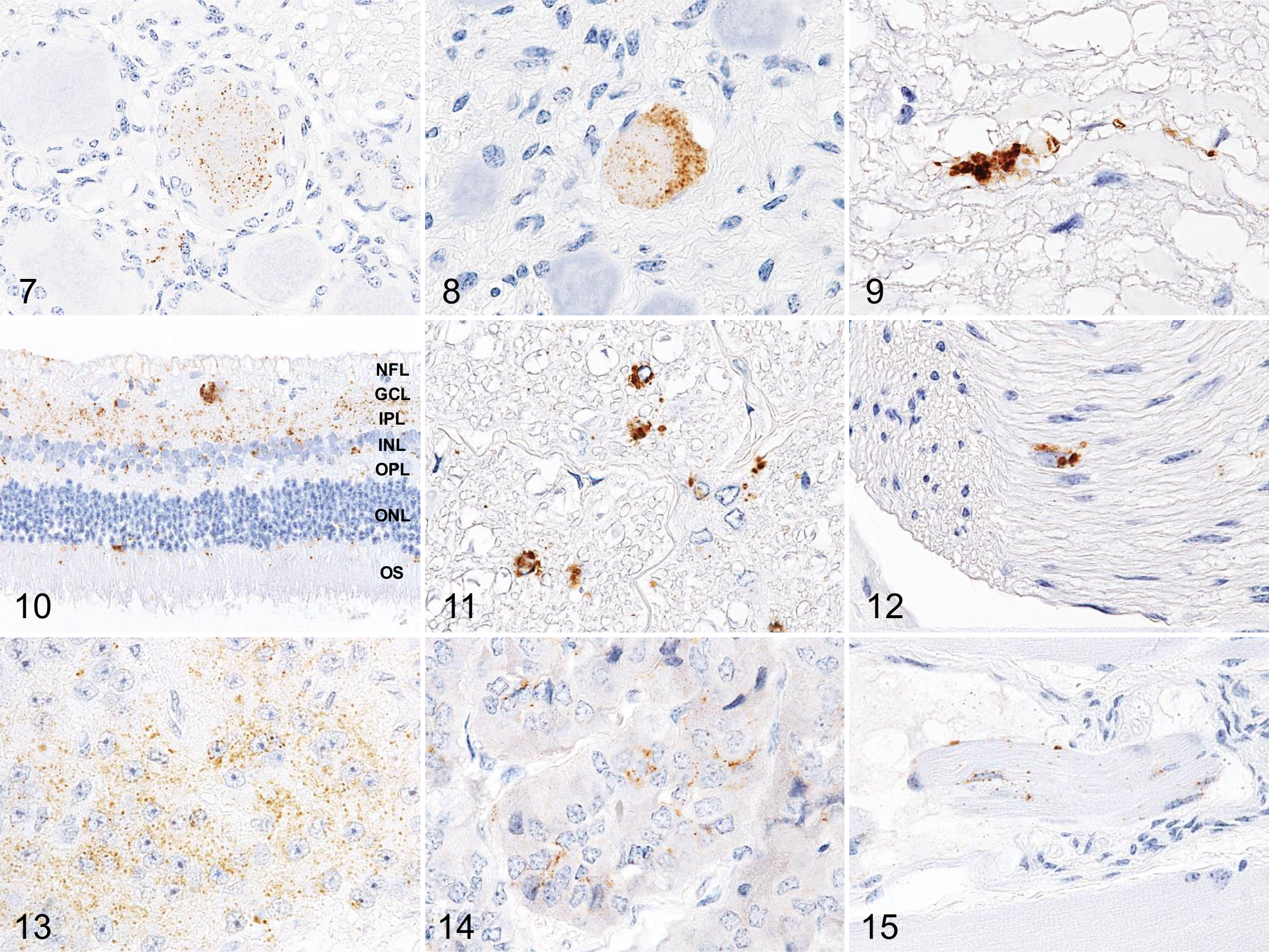
PrPSc Distribution in Extracerebral Tissues
IHC (Figs. 7–15) and Western blotting (Fig. 16) showed that PrPSc was widespread in the extracerebral central nervous tissues (retina, optic nerve, and neurohypophysis), nerve ganglia (trigeminal, dorsal root, stellate, cervical cranial, nodose, and coeliac and mesenteric ganglia), peripheral nerves such as cauda equina and vagus nerve, pineal gland, adrenal medulla, and the muscle spindles of the ocular muscle. In contrast, no PrPSc was detectable in any of the lymphoid tissues, including intestinal Peyer’s patches and in the enteric nervous system (Suppl. Table S1). Moreover, PrPSc deposition was not detected in skeletal muscle samples, including the masseter, pectoral, intercostal, triceps brachii, longissimus thoracis, psoas major, gluteus medius, quadriceps femoris, and semitendinosus muscles, indicating no involvement of the muscle spindles.

Western blot analysis of proteinase K–digested prion protein (PrPres) in the extracerebral tissues of a BSE-SW–infected calf (case No. 3) probed with monoclonal antibody SAF84. The lane numbers are indicated as follows: 1, optic nerve; 2, retina; 3, neurohypophysis; 4, cauda equina; 5, dorsal root ganglion; 6, trigeminal ganglion; 7, cranial cervical ganglion; 8, stellate ganglion; 9, sympathic trunk; 10, coeliac and mesenteric ganglion; 11, cranial vagus nerve; 12, facial nerve; 13, hypoglossal nerve; 14, brachial nerve plexus; 15, median nerve; 16, sciatic nerve; 17, tibial nerve; 18, adrenal medulla; 19, ileum; 20, colon. The PrPres signal in the extracerebral tissues is indicated below each lane in the lower panels as follows: +, positive; –, negative. The bars on the left in each panel represent molecular mass markers of 28, 19, 14, and 6 kDa.
Infectivity Assay of BSE-SW Prions in TgBoPrP Mice
The infectivity titer of the TgBoPrP mouse brain homogenates infected with BSE-SW was 107.4 ID50/g tissue, which was lower than that from H-BSE isolate from TgBoPrP mice (Table 1 and Suppl. Fig. S3). Significant differences were present in the survival days between TgBoPrP mice inoculated with BSE-SW and H-BSE (P < .05).
Retransmission of BSE-SW From Cattle to TgBoPrP Mice
TgBoPrP mice (n = 5) infected with BSE-SW from cattle developed disease at 141.0 ± 7.1 dpi, which was longer than the mean incubation period of 97.3 ± 3.7 days in BSE-SW–adapted TgBoPrP mice. 25 However, the clinical signs and pathologic and molecular features of retransmitted mice were indistinguishable from the BSE-SW–infected TgBoPrP mice previously reported 25 (data not shown).
FDC Assay in Wild-Type ICR Mice
All ICR mice (n = 5) intraperitoneally inoculated with the C-BSE prion isolated from cattle showed PrPSc deposition in the spleen at 75 and 150 dpi, with a 100% attack rate, as indicated by both Western blotting and IHC analyses. In contrast, PrPSc deposition was not detected in ICR mice inoculated with H-BSE or BSE-SW prions from cattle at 75 or 150 dpi (data not shown).
Discussion
The present study aimed to characterize the pathologic and biochemical features of BSE-SW in cattle, a prion variant newly emerged from H-BSE after serial passages in bovinized mice, 25 in comparison with H-BSE. The clinical signs in cattle infected with BSE-SW were consistent with those previously described for H-BSE–infected cattle 22,30 and included difficulty in rising and dullness, without display of nervousness or hyperesthesia to acoustic or tactile stimuli. However, behavioral changes can vary depending on the experimental conditions. 22,27 Despite the common clinical signs, the incubation period of BSE-SW in cattle (14.8 ± 1.5 months postinoculation [mpi]) was shorter than that of original H-BSE inoculum (18.7 ± 1.6 mpi), 30 while the time of clinical onset—between 11.5 and 12.5 months after inoculation—was similar for both prions. The incubation period or clinical duration of the disease in cattle challenged with H-BSE can vary among experiments. 1,17,22 Although the exact reasons for shorter incubation periods or clinical durations remain unknown, this may be due to differences in inoculum titers of the agents (Suppl. Fig. S3) or host/environmental factors rather than differences in inoculum strains. 27 Alternatively, the BSE-SW prion may be more virulent than the H-BSE prion in cattle. The shortened incubation period in cattle was in accordance with that observed in BSE-SW– as compared to H-BSE–infected TgBoPrP mice.
Vacuolar lesion scores in the cattle infected with BSE-SW resembled those described for H-BSE–infected cattle. 30 The PrPSc staining types were characterized by intraglial labeling in the white matter throughout the brain and spinal cord in addition to plaque-like deposits in the cerebral white matter, which was consistent with previous studies in cattle experimentally challenged with H-BSE. 22,30 In addition, regional distribution patterns of PrPSc caused by BSE-SW were identical to those of H-BSE. 30 Thus, on the basis of pathologic results, including vacuolar lesion scores, PrPSc neuroanatomical distribution patterns, and PrPSc staining types, BSE-SW shows more commonalities than differences with H-BSE in cattle.
The widespread PrPSc accumulation in extracerebral tissues detected by IHC as well as Western blotting may result from the centrifugal spreading of prions from the brain via nerve pathways during the clinical stage of the disease. 1 However, extracerebral involvement of PrPSc in cattle infected with BSE-SW was smaller than that in animals infected with H-BSE and seemed to be influenced by the shorter clinical duration. No striking difference was identified in the topographical tropism of PrPSc between H-BSE and BSE-SW prion-infected cattle. The absence of PrPSc lymphotropism in cattle and wild-type mice infected with BSE-SW was in accordance with the results reported for H-BSE.
With regard to strain-specific molecular features, similar to H-BSE, 9 BSE-SW was characterized by the presence of 2 cleaved forms of PrPres, PrPres 1 (N-terminally truncated) and PrPres 2 (C-terminally truncated), as shown by Western blotting. The characteristic multiple-banded patterns with an additional 10- to 12-kDa fragment analyzed with mAb SAF84 in TgBoPrP mice infected with BSE-SW were reproduced in cattle, indicating that both PrPres fragments of BSE-SW and uncleaved PrPres were consistently maintained after transmission into cattle. Usually, PrPres 1 is detectable using N-terminal–specific antibodies, such as P4 (bovine PrP epitope 101-WGQGGSH-107). However, the proteinase K cleavage site of PrPres 1 of BSE-SW in cattle, which was analyzed with mAbs P4, 6H4, and SAF84 (Suppl. Fig. S1), was distinct from that of H-BSE, indicating that PrPSc of BSE-SW has a different conformation than H-BSE. 25 Interestingly, with mAb 6H4, bands from BSE-SW closely resembled those from C-BSE. However, glycoform patterns from BSE-SW were similar to those from H-BSE. The unique strain-specific molecular features of BSW-SW previously described in TgBoPrP mice, 25 such as molecular weight, proteinase K cleavage sites, and glycoform profiles, were fully conserved after the transmission into cattle. The PrPres 1 of approximately 17 kDa from BSE-SW in TgBoPrP mice was more sensitive to guanidine hydrochloride treatment than that of H-BSE. 25 The biochemical evidence supports the concept that changes in the conformational stability of PrPres 1 may contribute to PrPSc diversity. Therefore, changes in replication environment may play an active role in generating new PrPSc variants to fit the new environment. 24 Artificial conditions, such as overexpression of PrP transgenes in TgBoPrP mice, most likely influence PrPSc diversity during serial transmission.
Traceback transmission is useful to maintain or alter strain-specific features of isolates before and after passage during cross-species transmission. 21 However, inoculation of BSE-SW isolated from cattle back into TgBoPrP mice failed to produce the traceback phenomenon 21 of the original H-BSE, even after further passages in TgBoPrP mice. In this study, back transmission of H-BSE from TgBoPrP mice into cattle was not carried out. However, the original H-BSE disease phenotype is expected to reemerge in cattle after transmission from TgBoPrP and/or wild-type mice infected with H-BSE owing to the traceback phenomenon.
In conclusion, no distinct differences in clinical and pathologic features were detected between BSE-SW and H-BSE in cattle, and biochemical properties of BSE-SW in cattle were well conserved from BSE-SW in TgBoPrP mice but distinct from those of H-BSE in cattle. The differences in molecular properties between BSW-SW and H-BSE did not influence the disease phenotype in cattle, suggesting that these 2 infectious agents are closely related strains. The fact that BSE-SW can transmit to cattle provides an important insight for devising control measures for atypical BSE. Further research is needed to prevent the spread of yet another form of atypical BSEs and for risk assessment and disease control measures of atypical BSEs.
Footnotes
Acknowledgements
We thank Naomi Furuya, Naoko Tabeta, Ritsuko Miwa, Junko Yamada, Noriko Shimozaki, and the animal caretakers for excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded, in part, by a subsidy of National Agriculture and Food Research Organization (NARO) and TSE research (H26-Shokuhin-Ippan-004) from the Ministry of Health, Labour and Welfare, Japan. The funders of the work did not influence study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
