Abstract
H-type bovine spongiform encephalopathy (BSE) has been identified in aged cattle in Europe and North America. To determine the localization of disease-associated prion protein (PrPSc) in the peripheral nerve tissues of cattle affected with H-type BSE, we employed highly sensitive immunohistochemical and immunofluorescence techniques with the tyramide signal amplification (TSA) system. PrPSc deposition was detected in the inferior ganglia, sympathetic nerve trunk, vagus nerve, spinal nerves, cauda equina, and adrenal medulla, using this system. Notably, granular PrPSc deposits were present mainly in the Schwann cells and fibroblast-like cells and occasionally along certain nerve fibers at the surface of the axons. In the adrenal gland, PrPSc immunolabeling was observed within the sympathetic nerve fibers and nerve endings in the adrenal medulla. Although our results were limited to only 3 experimental cases, these results suggest that the TSA system, a highly sensitive immunohistochemical procedure, may help in elucidating the peripheral pathogenesis of H-type BSE.
Keywords
Bovine spongiform encephalopathy (BSE), a transmissible spongiform encephalopathy of cattle, was first identified in the United Kingdom in 1986 and has subsequently spread to other European countries, Japan, and North America. The BSE agent was considered a single strain on the basis of the biological, neuropathological, and biochemical characteristics observed in field BSE cases and in BSE transmission studies using mice. Recently, 2 atypical forms of BSE, which were originally identified in France and Italy, have been detected in more than 70 cases in aged cattle worldwide. Atypical BSE agents are classified as H- and L-type BSE (also known as bovine amyloidotic spongiform encephalopathy) according to their abnormal prion protein (PrPSc) profiles and PrPSc deposition patterns in the brain, which differ from those of classical BSE. Furthermore, the transmissibility of H-type BSE compared to classical BSE has been confirmed in cattle by the intracerebral route. 9 Although a highly sensitive Western blotting technique using phosphotungstic acid precipitation showed accumulation of PrPSc in various peripheral nervous tissues in H-type BSE, the conventional immunohistochemical technique is unable to detect PrPSc, probably because of the relatively low levels of PrPSc in the peripheral nervous tissues or differences in detection sensitivity. 9 The aim of this study was to determine the localization of PrPSc in the peripheral nervous tissues of cattle affected experimentally with H-type BSE by using highly sensitive biotinylated tyramide-based procedures.
All experiments were performed in accordance with the guidelines of the Animal Ethical Committee and the Animal Care and Use Committee of the National Institute of Animal Health. Intracerebral transmission of H-type BSE has previously been reported in cattle (case No. 1, code 7749; case No. 2, code 9458; case No. 3, code 0728). 9 Two uninoculated calves served as controls and were sacrificed at the age of 27 months.
More than 150 tissues from the 3 cows affected with H-type BSE were fixed in 10% neutral buffered formalin containing 10% methanol. The peripheral tissues examined for PrPSc immunohistochemical reactivity were as follows: spinal nerves (C8, L6), dorsal root ganglia (C8, L6), cauda equina, trigeminal ganglion, inferior ganglion of the vagus nerve, superior cervical ganglion, stellate ganglion, sympathetic trunk, celiac and mesenteric ganglion complex, vagus nerve, facial nerve, hypoglossal nerve, phrenic nerve, accessory nerve, suprascapular nerve, brachial nerve plexus, median nerve, radial nerve, sciatic nerve, and tibial nerve. Formalin-fixed samples were treated in 98% formic acid for 60 minutes to reduce infectivity and then embedded in paraffin wax. Four-micrometer–thick serial sections were sliced onto silanized glass slides, stained with hematoxylin and eosin, and used for immunohistochemical analysis.
For PrPSc immunolabeling, dewaxed sections were immersed in 98% formic acid for 5 minutes, treated with 3% hydrogen peroxide at room temperature (RT) for 10 minutes, incubated with 10 μg/mL proteinase K (PK, 0.03 U/mg; Nacalai Tesque, Kyoto, Japan) in phosphate-buffered saline containing 0.1% Triton-X (PBST) at RT for 10 minutes, and then incubated in 150 mM sodium hydroxide at 60°C for 10 minutes. Immunohistochemical staining was performed using a tyramide signal amplification (TSA)-biotin kit (PerkinElmer, Boston, MA). Sections were incubated with 0.5% blocking reagent (PerkinElmer) for 30 minutes, monoclonal antibody (mAb) F99/97.6.1 (0.2 μg/mL; VMRD, Pullman, WA) for 60 minutes, then biotinylated anti-mouse IgG (1:300, PerkinElmer) for 30 minutes. Slides were then incubated with streptavidin-horseradish peroxidase (SA-HRP) (1:100, PerkinElmer) for 30 minutes, treated with biotinyl tyramide (1:50) as the amplification reagent for 5 minutes, and incubated again with SA-HRP (1:100) for 30 minutes before visualization with 3,3′-diaminobenzedine tetrachloride as the chromogen. Finally, sections were counterstained with Mayer’s hematoxylin.
For dual or triple immunofluorescence, selected sections were incubated in 98% formic acid for 5 minutes and then treated with 10 μg/mL PK in PBST for 10 minutes at RT followed by autoclaving at 121°C for 3 minutes in 10 mM citrate buffer (pH 6.0). After pretreatments, sections were incubated for 60 minutes with a primary antibody mix as follows: rabbit anti-protein gene product 9.5 (1:300, PGP9.5; Dako, Carpinteria, CA) for the axons of peripheral nerves and ganglionic cells of ganglia, rabbit anti-S100 (1:600, Dako) for the Schwann cells of peripheral nerves and satellite cells of ganglia, and rat anti-myelin basic protein (1:300; MBP, clone 12; Millipore, Billerica, MA) for the myelin sheath. After washing, sections were incubated with Alexa Fluor 546- or 647-conjugated goat anti-rabbit IgG (1:450; Molecular Probes, Portland, OR) for PGP9.5 and S100 or Alexa Fluor 546 goat anti-rat IgG (1:450, Molecular Probes) for MBP, as the secondary antibody. Sections were blocked and then incubated with mAb F99/97.6.1 (0.2 μg/mL) for 60 minutes. PrPSc immunofluorescence was visualized by the TSA fluorescence system (PerkinElmer) according to the manufacturer’s instructions. Immunofluorescence was evaluated using a Zeiss LSM 510 laser scanning confocal microscope (Carl Zeiss, Oberkochen, Germany). Sections incubated with non-immune mouse or rabbit IgG (1:30, Dako) instead of the primary antibody were used as negative controls for both TSA-biotin and TSA fluorescence amplification systems.
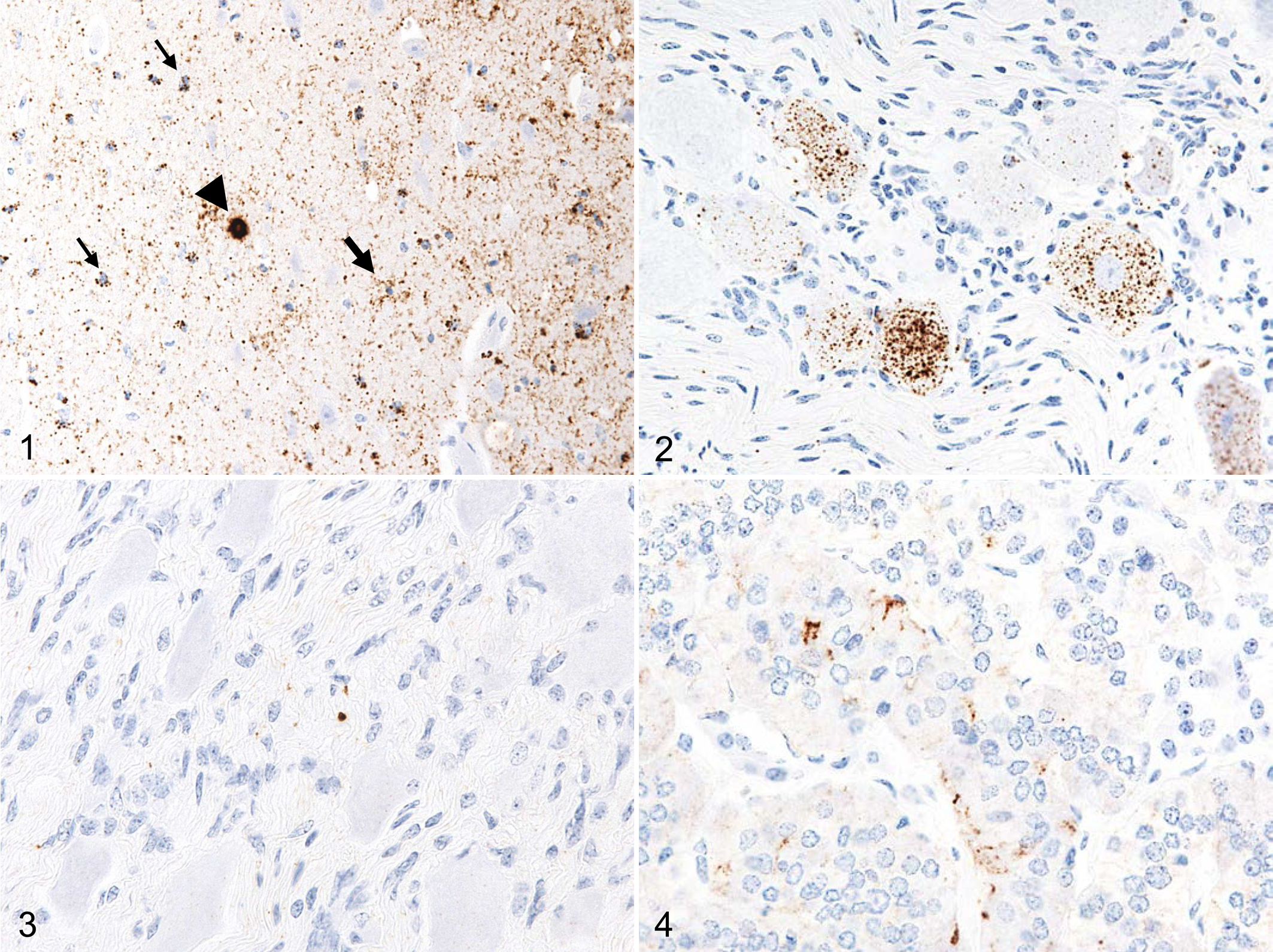
Neuropil, but not neuronal, vacuolation was obvious in all brain areas, especially in the nuclei of the thalamus and brainstem. Immunolabeled PrPSc was widely distributed throughout the brain and spinal cord (Fig. 1). Eight patterns of PrPSc immunolabeling including intraneuronal, perineuronal, intraglial, linear, fine particulate, coarse granular, stellate, and plaques were identified in the brain. The most conspicuous pattern of PrPSc was that of fine particulate and coarse granular deposits in the cerebral and cerebellar cortices and the nuclei of the thalamus, brainstem, and cerebellum. Stellate-type PrPSc deposition was detected in the cerebral and cerebellar cortices, basal ganglia, thalamus, hypothalamus, and hippocampus. Intraglial-type PrPSc deposition was conspicuous throughout the white matter of the brain and spinal cord. Spherical PrPSc plaques were sparsely located in the thalamus, basal ganglia, midbrain, pons, deeper layers of the cerebral cortices, and subcortical white matter. Immunolabeled PrPSc was observed in the retina, neurohypophysis, and optic nerve. In the trigeminal and dorsal root ganglia, PrPSc was mainly found accumulated in both ganglionic and satellite cells, using the conventional polymer immunodetection method as described previously. 9 In addition to the extracerebral tissues described above, positive PrPSc immunolabeling was detected in the adrenal gland, cauda equina, cervical spinal nerves, facial nerve, hypoglossal nerve, vagus nerve, sciatic nerve, and ganglia, such as the inferior ganglion of the vagus nerve, superior cervical ganglion, stellate ganglion, ganglia of the sympathetic trunk, and celiac and mesenteric ganglion complex, with the use of the TSA-biotin system. In these ganglia, not all neurons of ganglia exhibited intracytoplasmic labeling, but labeling was evident in the Schwann cells or fibroblast-like cells (Figs. 2, 3). In the cauda equina, dorsal roots of the cervical spinal nerves, facial nerve, hypoglossal nerve, vagus nerve, and sciatic nerve, granular PrPSc aggregate was mainly located in Schwann cells. In the adrenal gland, the PrPSc signal was evident at the intercellular fine processes of the nerve endings between the chromaffin cells of the adrenal medulla (Fig. 4). Immunolabeled PrPSc was not detected in the enteric nervous system such as the myenteric and submucosal plexi of the digestive tract. No specific immunolabeling was detectable in the lymphoid tissues with the TSA-biotin system.
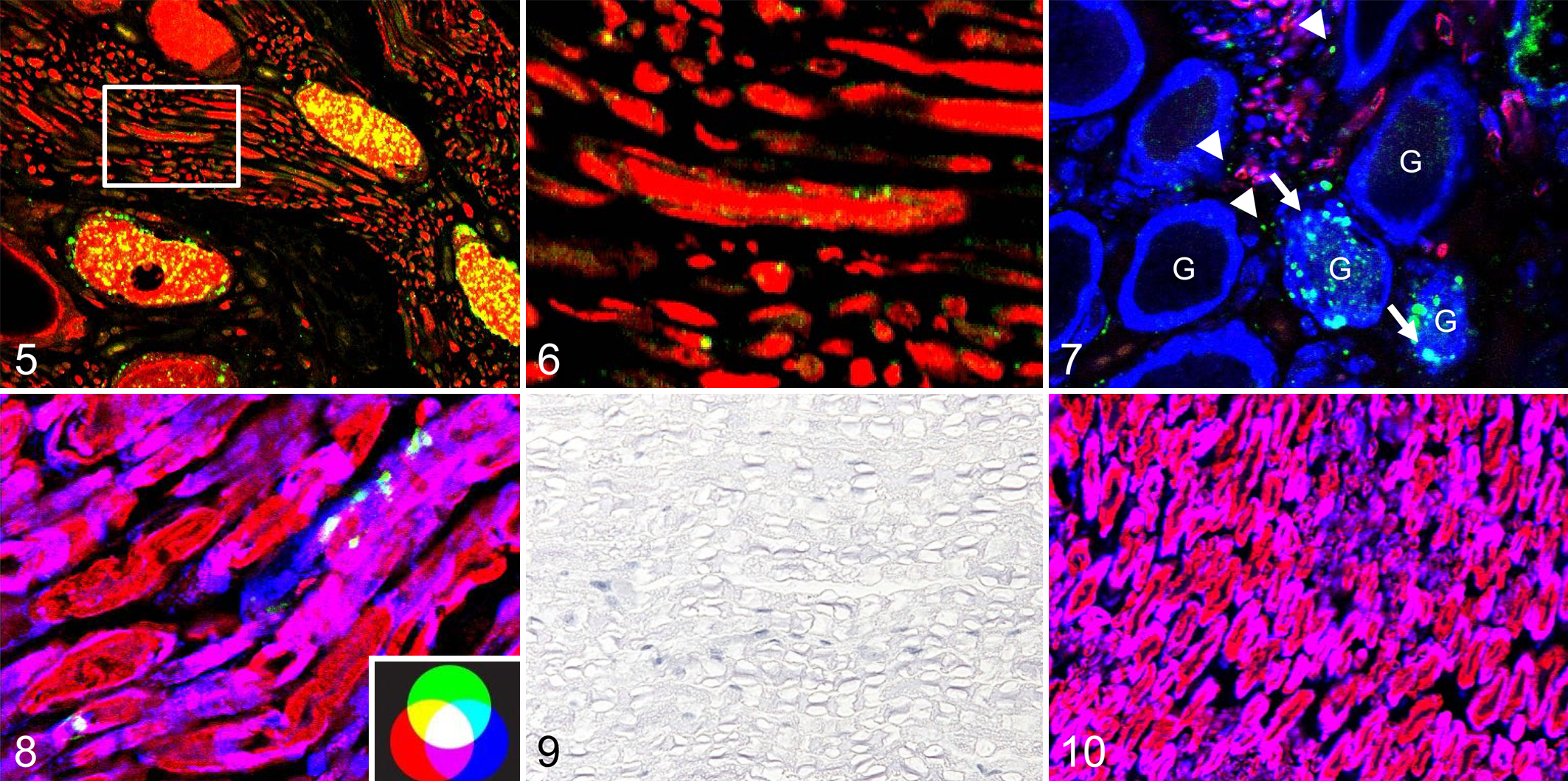
Using the dual or triple immunofluorescence technique, localization of PrPSc was visible in the ganglionic and satellite cells of the ganglia in the merged images (Fig. 5). In addition, the merged image showed that PrPSc granules were rarely located at the periphery of axons or within the axons adjacent to the ganglia (Fig. 6). Moreover, granular PrPSc was observed outside S100-positive cells (Fig. 7). In the cauda equina and spinal nerves, PrPSc coexisted mostly in the periphery of Schwann cells labeled with MBP and S100 (Fig. 8). No PrPSc immunolabeling was detected in the compact layers of the myelin sheath. In addition, no positive signal was detected in the sections from uninfected controls incubated with mAb F99/97.6.1 by both TSA-biotin and TSA-fluorescence methods, and no background immunostaining was observed in any sections of H-type infected animals by both methods when non-immune mouse and rabbit IgG, or PBS, were applied to the sections instead of the primary PrP-antibody (Figs. 9, 10).
The present study demonstrated that PrPSc granules in the peripheral nerves were localized mainly in the Schwann cells and fibroblast-like cells, and occasionally at the periphery of the axons. Deposition of PrPSc was observed at the cell surface of Schwann cells of the peripheral nerves. 1,4 –6,8 Normal cellular prion protein, PrPC, was mainly expressed at the cell surface of Schwann cells. 2 These results, therefore, suggest that Schwann cells may have a crucial role in prion propagation in the peripheral nerves. Thus, PrPSc accumulation in the central nervous system is commonly thought to be associated with a centrifugal spread of the infectious agent to the peripheral nervous tissues via neuronal rather than centripetal hematogenous progression of the agent by the non-axonal pathway. 2,3 Notably, small amounts of PrPSc were aggregated mainly in the Schwann cells, rather than in the axons, of the peripheral nervous tissues. 6
In addition, using the TSA-biotin system, immunolabeled PrPSc detected in the fibroblast-like cells present in the endoneurium of nerves seemed to be associated with the fluorescent signal located outside S100-positive cells that corresponded to Schwann cells. The fibroblast cell lines NIH/3T3 and L929 are susceptible to infection with mouse-adapted scrapie, suggesting that fibroblasts can be persistently infected with the agent and accumulate high levels of PrPSc after infection. 10
A PrPSc immunolabeling pattern in the adrenal medulla showing fine processes related to sympathetic nerve endings has been reported in sheep with scrapie. 7,8 PrPSc accumulation in the adrenal medulla, which consists of highly innervated tissue, may result from the spread of PrPSc from the brain via the sympathetic pathway.
In summary, our results demonstrated that this highly sensitive immunohistochemical procedure may be a valuable and practical tool for detecting minimal accumulation of PrPSc and for investigating the peripheral pathogenesis of H-type BSE in cattle.
Footnotes
Acknowledgements
We thank Naomi Furuya, Tomoko Murata, Mutsumi Sakurai, Junko Endo, Miyo Kakizaki, and Noriko Amagai for their expert technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. This study was supported by a Grant-in-Aid from the BSE and other Prion Disease Control Project of the Ministry of Agriculture, Forestry and Fisheries of Japan.
