Abstract
To characterize the biological and biochemical properties of H-type bovine spongiform encephalopathy (BSE), a transmission study with a Canadian H-type isolate was performed with bovinized transgenic mice (TgBoPrP), which were inoculated intracerebrally with brain homogenate from cattle with H-type BSE. All mice exhibited characteristic neurologic signs, and the subsequent passage showed a shortened incubation period. The distribution of disease-associated prion protein (PrPSc) was determined by immunohistochemistry, Western blot, and paraffin-embedded tissue (PET) blot. Biochemical properties and higher molecular weight of the glycoform pattern were well conserved within mice. Immunolabeled granular PrPSc, aggregates, and/or plaque-like deposits were mainly detected in the following brain locations: septal nuclei, subcallosal regions, hypothalamus, paraventricular nucleus of the thalamus, interstitial nucleus of the stria terminalis, and the reticular formation of the midbrain. Weak reactivity was detected by immunohistochemistry and PET blot in the cerebral cortex, most thalamic nuclei, the hippocampus, medulla oblongata, and cerebellum. These findings indicate that the H-type BSE prion has biological and biochemical properties distinct from those of C-type and L-type BSE in TgBoPrP mice, which suggests that TgBoPrP mice constitute a useful animal model to distinguish isolates from BSE-infected cattle.
Transmissible spongiform encephalopathies (TSEs), such as bovine spongiform encephalopathy (BSE) in cattle, scrapie in sheep, and Creutzfeldt–Jacob disease (CJD) in humans, are fatal neurodegenerative disorders. Although initial studies indicated that the causal agent of BSE represented a uniform strain of prion, now described as classical (C-type) BSE, 2 different phenotypes of BSE, called atypical BSE, have been recently identified in Italy 8 and France. 5 Currently, atypical BSE cases are classified into at least 2 groups—H-type and L-type, which, when compared to C-type, involve higher or lower molecular masses of the misfolded isoform of prion protein (PrPSc) as demonstrated by Western blot analysis. 6 The biological and biochemical properties of atypical BSE prions related to transmission to mice clearly differ from those of C-type BSE prions, including incubation period, proteinase K–digested (PK-digested) PrPSc (PrPres) profiles by Western blot analysis, neuropathology, and immunohistochemical analysis.1,3,6
Although C-type BSE is the result of consumption of BSE prion–contaminated feed, the origins of H-type and L-type BSE are unknown. Possible hypotheses on the origin of atypical BSE include: (1) the existence of different BSE strains, (2) a different source or unrecognized form of TSE agents, (3) genetic mutations in the prion protein (PrP) gene, and (4) spontaneous or sporadic forms of the diseases in cattle, as limited to old age with similarities to the sporadic form of CJD. 1
As a result of rapid BSE screening tests and surveillance programs, H-type BSE was originally identified in 2003 in France 5 and subsequently in other European countries, including Sweden, Poland, Germany, the Netherlands, the United Kingdom, the United States, and Canada. Interestingly, most of the atypical BSE cases were found in various breeds of animals older than 8 years of age. The only exceptions are 2 L-type cases: one occurred in Japan in a 23-month-old steer and one in Belgium in a 64-month-old cow. The transmissibility of H-type BSE in France has been confirmed in tg540 mice 3 and in an inbred mouse model (C57B/6), 2 as well as in Germany in Tgbov XV mice. 6 The neuropathology and topographic distribution of disease-associated PrPSc on transmission is described in these mouse models. The purpose of the present study was to determine the transmissibility of the Canadian H-type BSE isolate to bovinized transgenic mice and to characterize the PrPSc by Western blot and immunohistochemical analyses in comparison to C-type BSE.
The Canadian H-type BSE was found in a 16-year-old Charolais crossbred cow, which showed recumbency before euthanasia. By confirmatory immunohistochemistry, PrPSc immunolabeling was detected in the neurons and neuropil of the obex region of medulla oblongata, but plaques were not. The brain sample infected with C-type BSE from a naturally occurring case in Japan was also used for this study. In sum, 10% brain homogenate (w/v) of H-type and C-type BSE was prepared in phosphate buffered saline (pH, 7.4) using a multibead shocker (Yasui Kikai, Osaka, Japan), centrifuged at 1,000× g for 5 minutes and the resulting supernatants were used as inocula.
Tg(BoPrP)-4092HOZ/Prnp0/0 (TgBoPrP) mice overexpressing the bovine PrP gene on a null mouse background highly susceptible to C-type BSE were kindly supplied by Dr S. B. Prusiner. 10 The inoculum was inoculated into the left cerebral cortex at the level of the thalamus of female 3-week-old TgBoPrP mice with 20 μl of each of the 10% brain homogenates through a 27-gauge disposal needle. Following inoculation, the clinical status of the mice was monitored daily to assess the onset of neurologic signs. All TgBoPrP mice inoculated with H-type BSE developed initial clinical signs, such as constant chewing of the bedding, without loss of weight or appetite before approximately 30 days before disease-related death. Later, moribund mice showed loss of appetite, wasting, inactivity, hunching, and inability to groom. The survival period of the first passage was 315.8 ± 11.6 days (n = 10). The subsequent passage of the brain homogenate of H-type BSE-infected TgBoPrP mice resulted in a shortened incubation period (230.4 ± 7.3 days, n = 10) with the same clinical signs. In contrast, first- and second-passage TgBoPrP mice infected with C-type BSE developed kyphosis, head twitching, and ataxia as the characteristic clinical signs; each mouse reached the terminal stage after an incubation period of 243.2 ± 7.2 days (n = 10) for the first passage and 215.8 ± 4.1 days (n = 10) for the second.
Symptomatic animals were sacrificed and examined for neuropathologic changes, including the histopathologic lesion profile. In addition, immunohistochemical and paraffin-embedded tissue (PET) blot analyses were done on formalin-fixed and formic acid–treated sections as previously described. 9 The biochemical properties of PrPres in the brain of infected mice were analyzed by Western blot with monoclonal antibodies (mAbs) 6H4 (Prionics, Schlieren, Switzerland) and SAF84 (SPI-bio, Montigny le Bretonneux, France) as previously described. 9 All samples were digested with 50 μg/ml PK at 37°C for 1 hour. Western blot analysis with mAb 6H4 (which recognizes bovine PrP amino acid residues 156–164) showed bands representing the higher molecular mass of approximately 19 kDa in the brain of H-type BSE-infected TgBoPrP mice, compared to a band of approximately 18 kDa in the brain of mice infected with C-type BSE prion (Fig. 1A). Moreover, Western blot analysis with mAb SAF84 that recognized the epitope located at amino acid residues 175–180 of bovine PrP yielded a different banding pattern of PrPres, including an additional ~14-kDa fragment (Fig. 1B). After deglycosylation by peptide-N-glycosidase F (PNGase F, New England Biolabs, Beverly, MA) treatment described previously, 9 a higher molecular mass of the unglycosylated PrPres form of H-type BSE was detected with 6H4 and SAF84 mAbs and was different from that of C-type BSE (Fig. 2). An additional ~14-kDa fragment was detectable with mAb SAF84 in H-type BSE (Fig. 2B). In addition, the relative amounts of the diglycosylated, monoglycosylated, and unglycosylated form of PrPres in H-type BSE were distinct from that of C-type BSE (Fig. 3), and these complex molecular features were conserved in the case of Canadian H-type BSE-infected TgBoPrP mice.
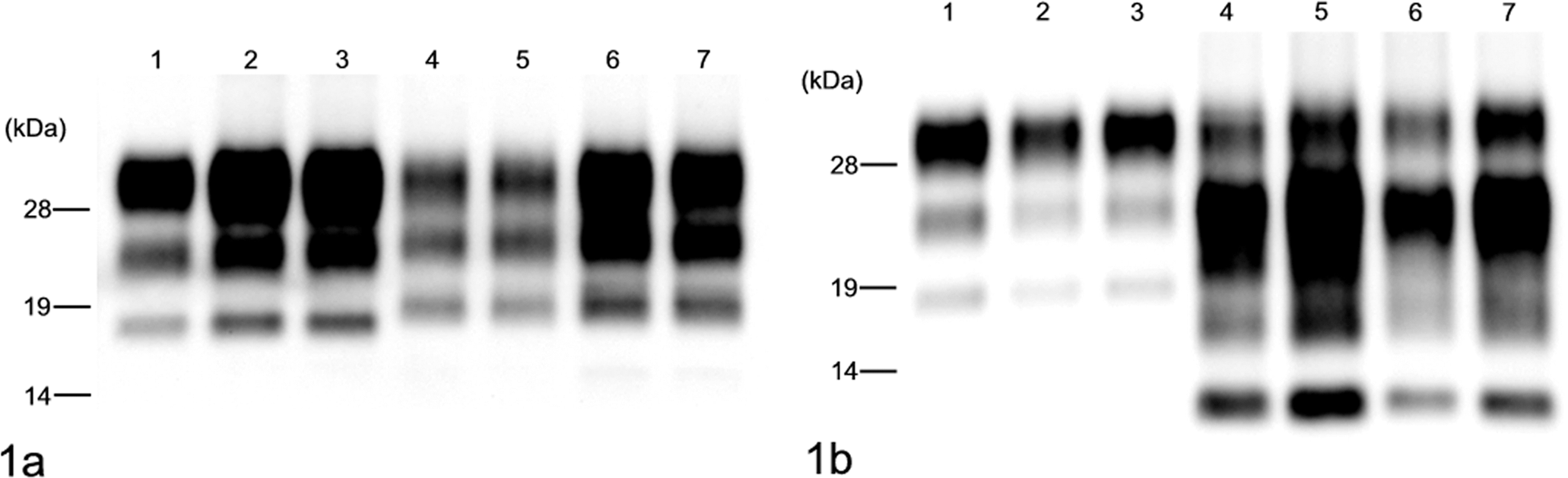
Western blot analysis of proteinase K–digested prion protein (PrPres) from C-type and H-type bovine spongiform encephalopathy (BSE) prion with monoclonal antibodies 6H4 (A) and SAF84 (B). Lane 1: Bovine brain with C-type BSE. Lane 2: TgBoPrP mouse inoculated with C-type BSE prion, first passage. Lane 3: TgBoPrP mouse inoculated with C-type BSE prion, second passage. Lane 4: Bovine brain with H-type BSE (naturally occurring case). Lane 5: Brain of a bovid inoculated intracerebrally with H-type BSE prion. Lane 6: TgBoPrP mouse inoculated with H-type BSE prion, first passage. Lane 7: TgBoPrP mouse inoculated with H-type BSE prion, second passage. Molecular masses are shown on the left (kDa).
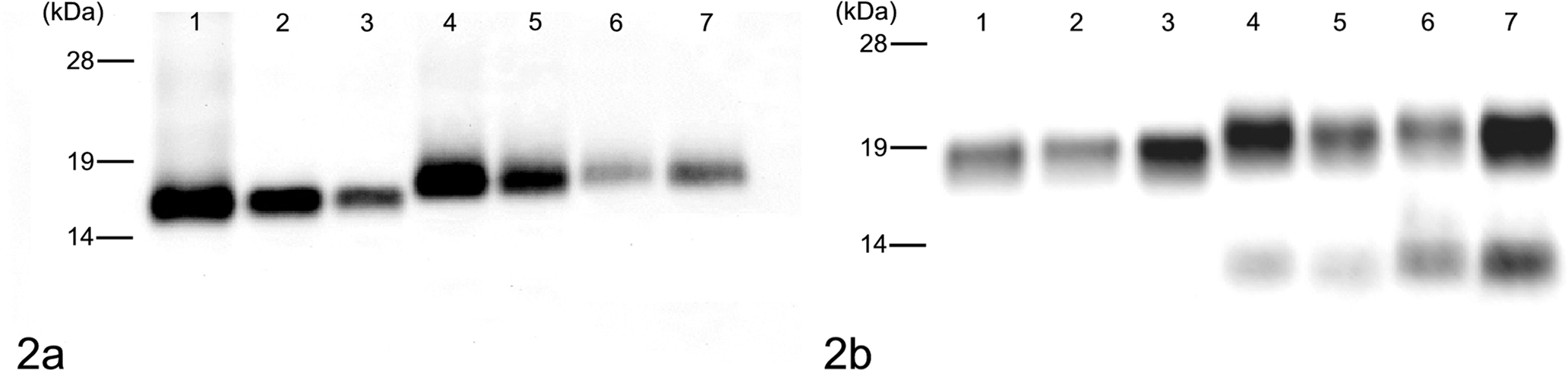
Western blot analysis of deglycosylated PrPres from C-type and H-type bovine spongiform encephalopathy prion, detected with monoclonal antibodies 6H4 (A) and SAF84 (B). The lane numbers are as listed in Figure 1. Molecular masses are shown on the left (kDa).
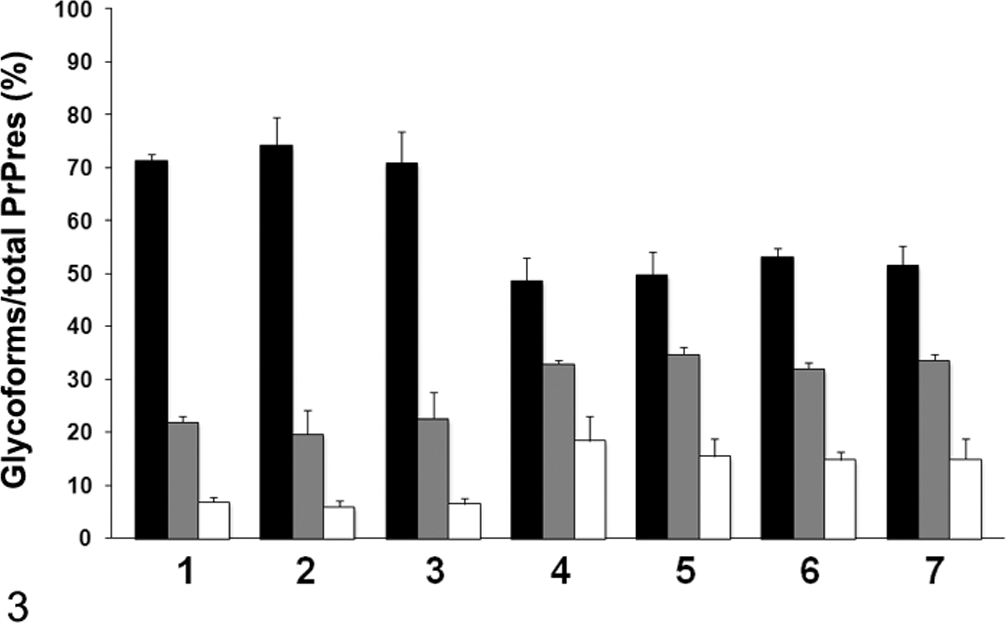
The relative amount of the diglycosylated, monoglycosylated, and unglycosylated form of PrPres in the C-type and H-type bovine spongiform encephalopathy prion–affected individual with monoclonal antibody 6H4. The lane numbers are as listed in Figure 1. The results are shown as mean ± standard deviation in triplicate experiments. Bar diagram indicates diglycoslated form (black column), monoglycosylated form (gray column), and unglycosylated form (white column).
Histopathologic examination showed mild to moderate spongiform changes characterized as neuropil vacuolation in the reticular formation of the midbrain, hypothalamus, thalamus, and septal nuclei in the first-passaged TgBoPrP mice (Figs. 4, 5A). Plaque-like deposits were frequently present—distributed bisymmetrically in subcallosal and adjacent periventricular area of the frontal lobe, basal ganglia, hippocampus, and corpus callosum. They corresponded to immunolabeled plaque-like deposits and/or large aggregates (Fig. 5B). Plaque-like deposits with a lucent core, but not large aggregates, displayed polarized and colored birefringency with the Congo red stain (data not shown). Such plaque-like deposits were occasionally present in the molecular layer of the cerebral cortex but were not detected in the thalamus or brainstem. Higher lesion scores in most brain areas, with the exception of cerebellar cortex (Fig. 4), and more conspicuous plaque-like deposits were featured in the second-passage mice than in the first-passage mice (data not shown).
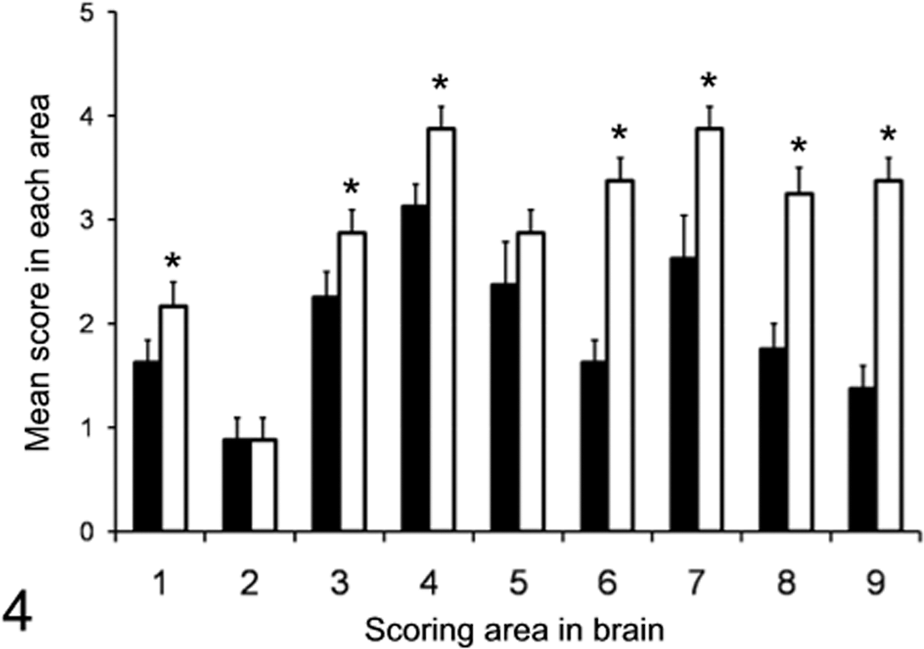
Histopathologic lesion profile of TgBoPrP mice inoculated with H-type bovine spongiform encephalopathy isolate. Vacuolation was scored on a 0–5 scale in the following brain areas: 1, dorsal medulla; 2, cerebellum; 3, midbrain; 4, hypothalamus; 5, thalamus; 6, hippocampus; 7, septal nuclei of the paraterminal body; 8 caudal cerebral cortex; 9, rostral cerebral cortex. Bar diagram indicates first (black column) and second passage of H-type bovine spongiform encephalopathy (white column). Data represent mean ± standard deviation (n = 10), and significant difference was determined by unpaired t test.
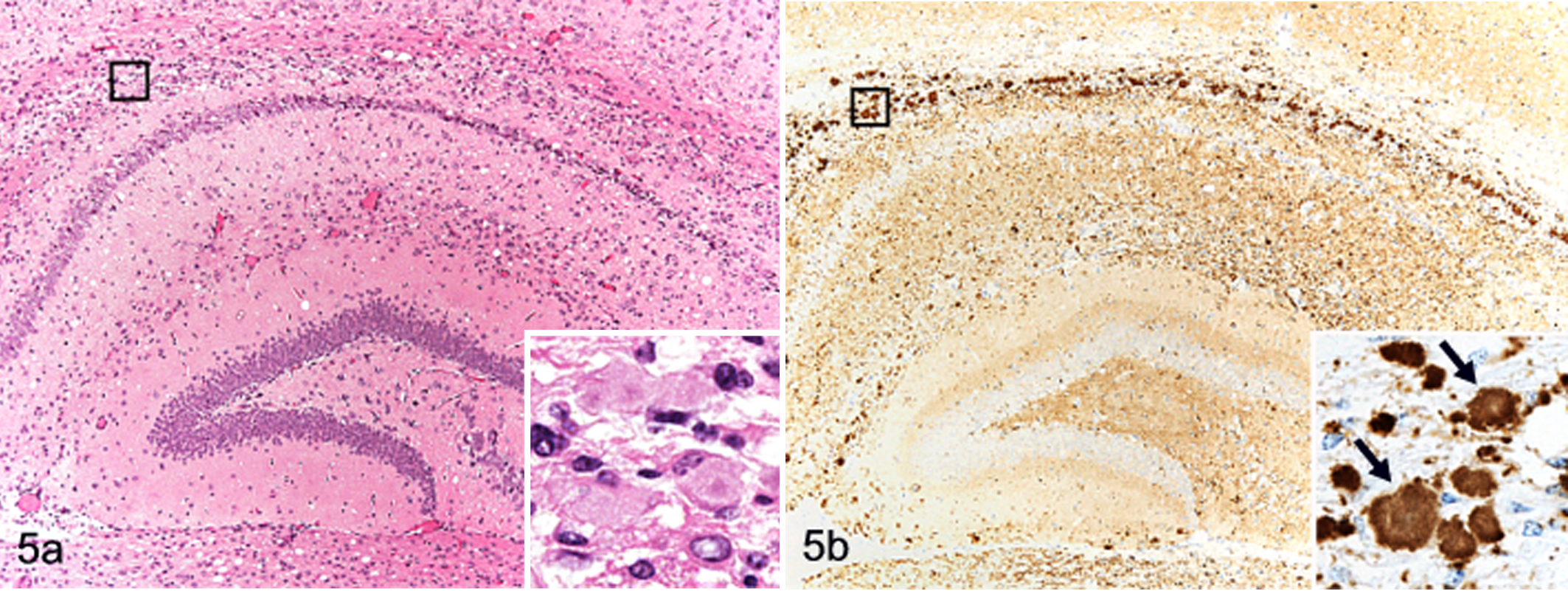
Brain; TgBoPrP mice inoculated with H-type bovine spongiform encephalopathy prion. A, mild to moderate vacuolation accompanied with plaque-like deposits is detectable in the hippocampus. Inset: higher magnification of the box. HE. B, granular PrPSc deposits and plaque-like deposits are conspicuous in the hippocampal region. Immunohistochemical labeling with monoclonal antibody F99/97.6.1 and hematoxylin counterstain. Inset: higher magnification of the box showing aggregates of various sizes and a plaque-like deposit with a lucent core (arrows).
There was a significant correlation between PrPSc deposition and neuropathologic changes in TgBoPrP mice inoculated with H-type BSE. As shown by immunohistochemistry with mAb F99/97.6.1 (VMRD, Pullman, WA) (Fig. 5B) and PET blot with mAb SAF84 (Fig. 6), immunolabeled PrPSc accumulation was most intense in the following brain locations: septal nuclei, interstitial nucleus of the stria terminalis, hypothalamus, accumbens, subcallosal regions, dorsal nucleus of the raphe, reticular formation of the midbrain, and raphe nuclei of the medulla oblongata. Signals mostly consisted of prominent deposition of fine and coarse granular PrPSc accompanied by PrP aggregates and spongiform changes in the neuropil. Lower levels of immunolabeled granular PrPSc and PrP aggregates were detected in the cerebellar cortex, thalamus, and dorsal and ventral medulla of the pons or medulla oblongata and moderately in the caudate nucleus, vestibular nuclei, and cerebellar nuclei. In contrast, PrPSc accumulation in C-type BSE-infected TgBoPrP mice was mainly detected in the interstitial nucleus and red nucleus of the midbrain, habenular nucleus of the thalamus, and reticular formation of the medulla oblongata (Fig. 7). The distribution patterns of PrPSc deposition were similar between the first and second passages of H-type and C-type BSE-infected TgBoPrP mice.
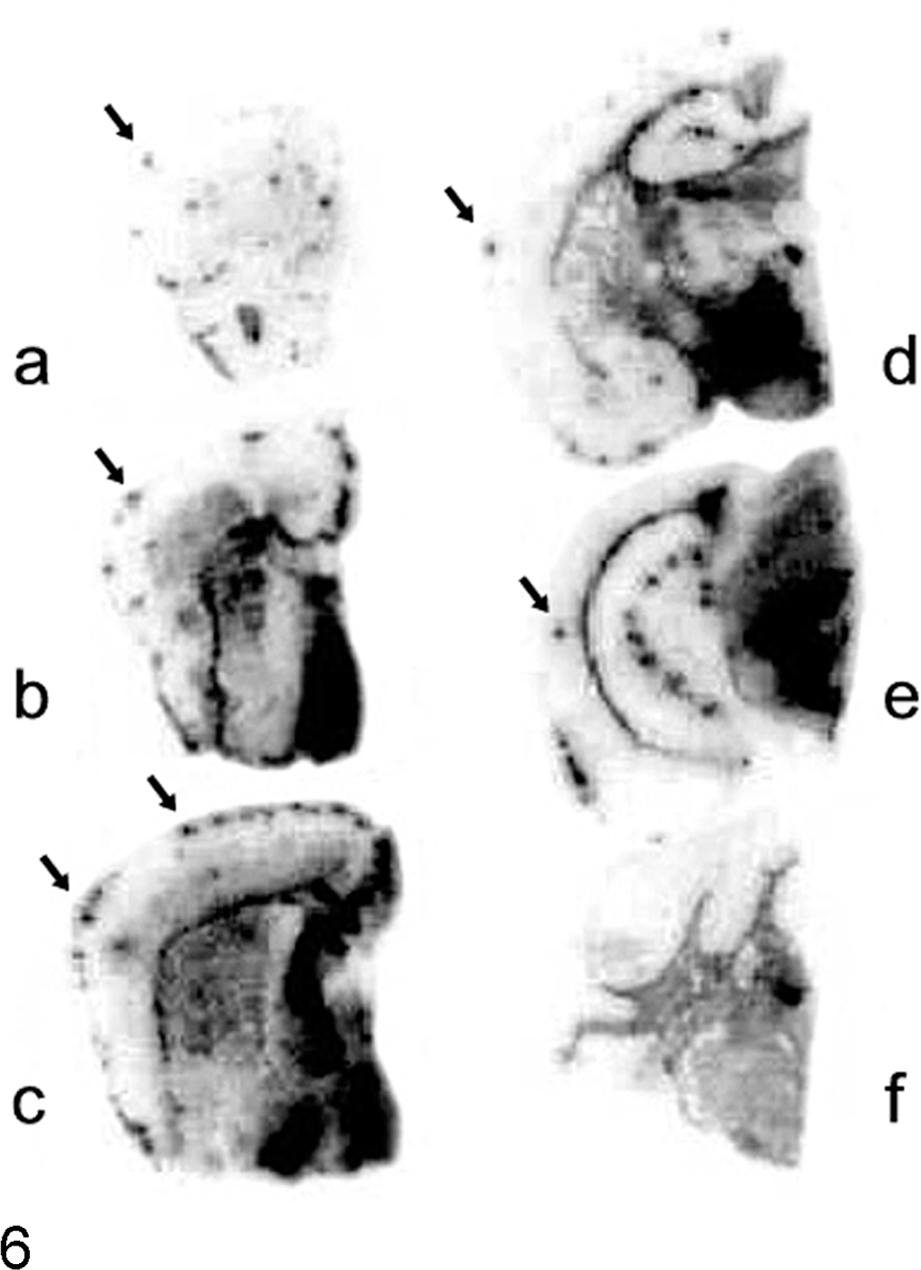
Brain; TgBoPrP mice. Paraffin-embedded tissue blot analysis showing the distribution of PrPSc in the brain of H-type bovine spongiform encephalopathy–infected TgBoPrP mice. Immunolabeled PrPSc is obvious in septal nuclei (B, C), subcallosal regions (D, E), hypothalamus (D), and reticular formation of the midbrain (E). Prion protein plaques are present as dots in the molecular layer of cerebral cortices (arrows). Blots corresponding to the brain areas at the level of frontal lobe (A), septal nuclei (B, C), hippocampus (D), midbrain (E), and medulla and cerebellum (F) were prepared with monoclonal antibody SAF84.
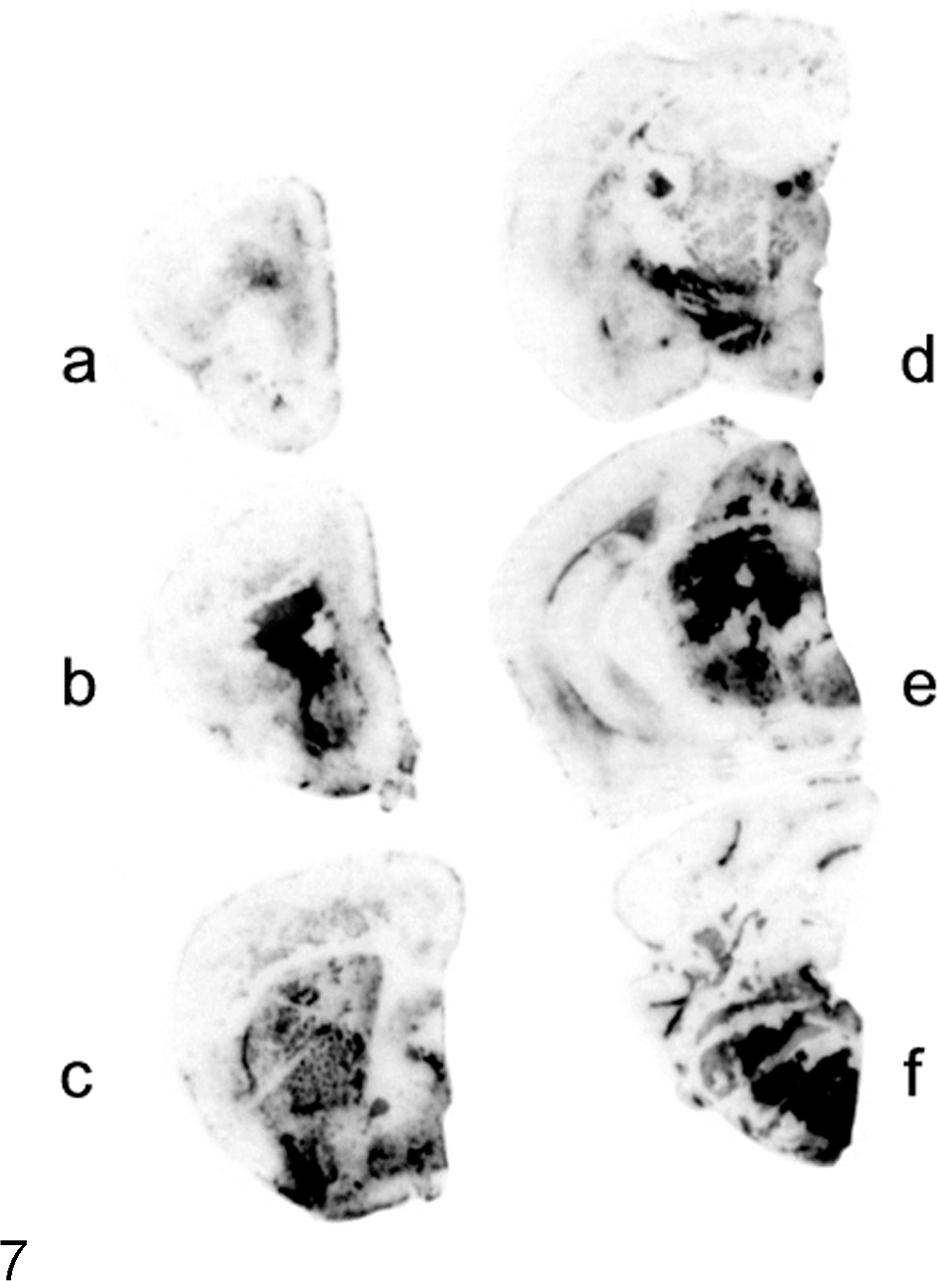
Brain; TgBoPrP mice. Paraffin-embedded tissue blot analysis showing the distribution of PrPSc in the brain of C-type bovine spongiform encephalopathy–infected mice. Immunolabeled PrPSc is marked in the reticular formation of the brainstem (E, F). Blots were prepared as indicated in Figure 6.
As in previous transmissions of atypical H-type and L-type BSE to mice, the present study showed that after transmission to TgBoPrP mice by the intracerebral route, the Canadian H-type BSE isolate clearly differs from C-type BSE with respect to incubation period, PrPres profile, and histopathologic and immunohistochemical analyses.1,3,6 Our findings provide support for the existence of different BSE prions using an animal model.
Reduced incubation periods are associated with multiple PrP copies in bovinized transgenic mice compared to wild-type mice. 10 Incubation periods in TgBoPrP mice (mean of 315 days) were similar to those reported in TgbovXV mice. 6 However, no further comparisons with the present findings are possible, because the clinical signs and neuropathology were not described in reports using H-type BSE in Tgbov XV mice. The survival periods in the first and second passages of tg540 mice were much longer than those in TgBoPrP mice. 3 Differences observed in spongiform degeneration and PrPSc accumulation in the brain and differences in the incubation periods at first- and second-passage mice may well be pathogenetically associated and may suggest that the primary passaged H-type BSE was not fully adapted to TgBoPrP mice. A third serial passage has been initiated and is ongoing to determine possible stabilization of properties. The discrepancy in incubation periods among TgBoPrP, Tgbov XV, and tg540 mice has several potential explanations, including differences in agents tested, differences in titers of inocula, and differences in expression levels of cellular PrP (PrPC) in the brain. Additionally, it is possible that there are differences in populations of PrPC-expressing cells, despite similar total expression levels of PrPC in the brains of the different models. The amount of PrPC in the brain of TgBoPrP mice was estimated to be at least 8-fold higher than that of the cattle brain, and similarly, tg540 mice were shown to have at least 8 times as much PrPC as wild-type FVB mice.3,10 An expression level of PrPC in Tgbov XV has not been published. However, both TgBoPrP and Tgbov XV mice were 10-fold more sensitive than cattle to infection with C-type BSE.7,10 It is likely that the expression levels and/or amino acid differences of PrPC in the brain in different transgenic mouse models play an important role in the incubation period differences. Previous studies have shown low levels of PrPSc in the medulla,3,5 and if this were representative of levels throughout the brain, it might suggest an equivalent reduced level of infectivity in H-type, compared to C-type, cases.
The present and previous studies prove that H-type BSE isolates have distinct biological and biochemical properties compared to those of C-type and L-type BSE.2,4 The PrPres of H-type BSE, as detected with the mAb SAF84 that recognized the C-terminal region of PrP, was characterized by a mixture of 2 fragments consisting of 19-kDa and ~14-kDa molecular weights, which possessed a different cleavage site in the N-terminal region with PK digestion. 4 The higher molecular mass of the unglycosylated PrPres molecules, which included an additional ~14-kDa fragment in the case of Canadian H-type BSE, was conserved in TgBoPrP mice. The unique molecular features of H-type BSE were also well preserved in wild-type mice.2,4
The H-type BSE isolate transmitted to TgBoPrP mice was characterized by the presence of PrP-immunolabeled amyloid plaques. Amyloid-positive PrP plaques have been detected in wild-type mice 2 but not in tg540 mice 3 challenged with H-type BSE. The presence of PrP plaques might be associated with a distinctive 14-kDa fragment detected with C-terminal antibodies in H-type BSE. 4 Therefore, the C-terminus-specific antibodies might be more useful than N-terminus- and middle region–specific antibodies for detailed analyses of the biological and biochemical properties in H-type BSE-infected animals.
In conclusion, the Canadian H-type BSE isolate was transmitted to TgBoPrP mice with biological and biochemical characteristics distinct from those of C-type BSE, suggesting that TgBoPrP mice are a useful animal model for distinguishing different isolates from BSE-affected cattle. Characterizing atypical BSE isolates and potentially unknown prion diseases in cattle using similar well-defined transmission studies may help to reveal origins and pathogenetic mechanisms of these diseases.
Footnotes
Acknowledgements
We thank Dr S. B. Prusiner, Institute for Neurodegenerative Diseases, University of California, for providing Tg(BoPrP)-4092HOZ/Prnp0/0 mice and Sandor Dudas for critically reading the manuscript. Expert technical assistance was provided by Ms M. Sakurai, N. Amagai, T. Murata, N. Tabeta, and the animal caretaker. This study was carried out under the guidelines of the Animal Ethical Committee and the Animal Care and Use Committee of National Institute of Animal Health.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
This work was supported by a Grant-in-Aid from the BSE and other Prion Disease Control Project of the Ministry of Agriculture, Forestry and Fisheries of Japan.
