Abstract
Histopathology is considered the gold standard diagnostic method for canine mammary tumors. In 2011, a new histologic classification for canine mammary tumors was proposed. The present study was a 2-year prospective study that validated the 2011 classification as an independent prognostic indicator with multivariate analysis in a population of 229 female dogs, identifying subtype-specific median survival times (MST) and local recurrence/distant metastasis rates. Dogs with benign tumors and carcinoma arising in benign mixed tumors all had an excellent prognosis. Dogs with complex carcinoma and simple tubular carcinoma also experienced prolonged survival. Those with simple tubulopapillary carcinoma, intraductal papillary carcinoma, and carcinoma and malignant myoepithelioma had a more than 10-fold higher risk of tumor-related death. The prognosis was even worse for adenosquamous carcinoma (MST = 18 months), comedocarcinoma (MST = 14 months), and solid carcinoma (MST = 8 months). The most unfavorable outcome was for anaplastic carcinoma (MST = 3 months) and carcinosarcoma (MST = 3 months), which also had the highest metastatic rates (89% and 100%, respectively). Adenosquamous carcinoma exhibited the highest local recurrence rate (50%). In the same canine population, the tumor diameter was recognized as a strong predictor of local recurrence/distant metastasis and an independent prognosticator of survival in the multivariate analysis. Excision margins were predictive only of local recurrence, whereas lymphatic invasion and histologic grade were predictive of local recurrence/distant metastasis and survival, although only in univariate analyses. In conclusion, this study validated the 2011 classification scheme and provided information to be used in the clinical setting and as the basis for future prognostic studies.
Canine mammary tumors (CMTs) are the most common neoplasms in female dogs. 11 The mainstay of treatment is surgical removal, which is typically followed by histopathology. 19 Although there are new molecular techniques for subtyping CMTs, 3,5,7,18 their use is still limited to a research setting and implementation is needed before considering their application in routine diagnostic activity. Therefore, histopathology still represents the gold standard diagnostic method to classify and provide prognostic information about dogs with CMTs.
Despite no clear consensus on what information should be included in the histopathology reports for CMTs, various authors have suggested including the following information: tumor subtype, tumor grade, presence/absence of lymphatic invasion, margins of excision, and tumor diameter. 6,11,12,14
In 2011, a new detailed histologic classification of subtypes of CMTs was published by Goldschmidt and colleagues. 4 This new histologic classification (2011 classification) was based on the World Health Organization criteria previously published in 1974 and 1999 and included 30 CMT subtypes (23 malignant and 7 benign). Three subsequent studies investigated the prognostic significance of the 2011 classification. 5,12,14 Two studies retrospectively evaluated a population of 245 and 658 dogs, respectively, and found a correlation between the 2011 classification and histologic evidence of lymphatic invasion. 5,14 The third study was prospective, included 65 dogs, and found that the 2011 classification was able to predict disease-free survival, overall survival, local recurrence, and distant metastasis. However, the small number of animals limited the statistical evaluation to univariate analysis, and different subtypes of CMTs were grouped together in 1 category for analysis. 12 At the time of writing, there are no prognostic data available for each tumor subtype to be used in a clinical setting, and the 2011 classification has not yet been validated as an independent prognostic indicator in multivariate analysis.
The present study is a prospective 2-year follow-up study in which the prognostic value of the 2011 classification is investigated in a population of 229 female dogs. The study follows the recommended guidelines for the conduct and evaluation of prognostic studies in veterinary oncology.
20
Specific objectives of the study are as follows. To describe the clinical outcome (tumor-specific overall survival, rate of local recurrence, and rate of distant metastasis) of 14 CMT subtypes classified according to the 2011 classification. To investigate the prognostic significance of other histologic parameters (grade, lymphatic invasion, infiltration of the margins of excision, and tumor diameter). To validate the 2011 classification as an independent prognostic indicator in multivariate analysis.
Materials and Methods
Study Population and Inclusion Criteria
Female dogs were evaluated for inclusion in the study at the time of submission of a surgically removed mammary mass to the Diagnostic Service of Veterinary Anatomic Pathology of the University of Padua (Italy). Submission to the above-mentioned Diagnostic Service required a privacy and informed consent form that allowed research studies on the submitted material. The samples were not specifically collected for this study and they were submitted by veterinary clinical practitioners between January 2008 and December 2012 after surgery as therapeutic intervention. The study, therefore, did not require additional ethical approval. Dogs included in the study had a single mammary tumor (either benign or malignant) or multiple mammary tumors with only 1 of them being malignant, which was the tumor considered in the study. Dogs with different malignant mammary tumor subtypes or different benign mammary tumor subtypes were excluded from the study because of the difficulty in deciding which tumor would affect the follow-up. To be able to analyze each tumor subtype, a minimum of 8 cases for that subtype was the target during recruitment. Recruited cases were not included in prior studies. Only excisional biopsies were considered (incisional biopsies were excluded from the study). All recruited dogs had surgery as the only treatment modality and were free of metastasis on preoperative thoracic radiographs.
Histopathology
Samples of surgically resected mammary neoplasms and corresponding lymph nodes (when available) were fixed in 10% buffered formalin. The tumor diameter (largest tumor diameter) was measured postfixation at the time of trimming. Cross-sections of the tumors (3 to 6 sections, depending on the size of the tumor) were taken and routinely processed for histopathology. The histologic evaluation was performed jointly by 2 of the authors (R.R. and V.Z.) or in some cases by 3 of the authors (R.R., V.Z., and M.H.G.). At the time of the histologic examination, these authors were blind to the clinical follow-up. Neoplasms were histologically classified into subtypes following the 2011 classification. 4 According to this classification, some tumor subtypes are considered unambiguously malignant by their morphologic features (eg, solid carcinoma, comedocarcinoma, anaplastic carcinoma, adenosquamous carcinoma, and carcinosarcoma), whereas for other subtypes, benign and malignant forms are described (eg, complex adenoma vs complex carcinoma, simple adenoma vs simple tubular or tubulopapillary carcinoma, benign mixed tumor vs carcinoma arising in benign mixed tumor), and in these cases, the diagnosis of malignancy was based on the presence of at least 1 of the following features: pluristratification of the epithelial cells (more than 3 layers), necrosis (presence of at least 2 randomly distributed foci), marked nuclear pleomorphism (assessment in the fields with highest degree of anisokaryosis), or more than 3 mitoses in 10 high-power (400×) fields (assessment in the fields with the highest mitotic activity; diameter of the field of view = 0.55 mm). Identification of neoplastic malignant myoepithelium was performed according to previous studies. 13,14 Neoplasms were graded using the Peña system, 12 although focusing exclusively on the neoplastic epithelial component as previously done. 13,14 The status of the resected margins (free/not free of tumor) was also assessed, evaluating at least 3 cross-sections of the tumor. Lymphatic invasion, defined as infiltration of regional lymph nodes (when available) and/or lymphatic vessels at the periphery of the neoplasms, was also determined.
Follow-up
During the follow-up period, dogs received a complete physical examination and underwent imaging procedures (thoracic radiographs and abdominal ultrasound) every 3 to 4 months for a minimum of 2 years as part of the clinical monitoring performed by the veterinary clinical practitioners. Information recorded included the development of local recurrence, distant metastases, and tumor-related death. Local recurrence was defined as the development of a subsequent tumor in the same location and with the same histologic features as the original tumor. Metastatic disease was diagnosed using imaging procedures, supported by aspiration cytology when possible and ethically acceptable. Tumor-related death was clinically defined as spontaneous death or euthanasia due to tumor-related issues (local progression or systemic deterioration in animals with metastatic disease). It is unfortunate that owners did not give permission for necropsy examination.
Statistical Analyses
Data collected for each dog included age, breed, neuter status, tumor subtype, grade, lymphatic invasion, margins, and tumor diameter. Outcome measures were tumor-specific overall survival, development of local recurrence, and distant metastases. Animals that died for reasons unrelated to their tumors or that were still alive at the end of the follow-up period were censored because the event tumor-related death had not occurred.
Associations between tumor-specific overall survival, local recurrence, and distant metastases, measured as a binary outcome and age (quartiles), breed, neuter status, tumor diameter (< 1 cm, between 1 and 2 cm, between 2 and 5 cm, and > 5 cm), tumor subtype, grade, margins of excision, and lymphatic invasion, were examined using Pearson chi-square test. Kaplan–Meier survival plots and corresponding nonparametric log-rank tests and univariate Cox’s proportional hazard regression analysis (95% confidence interval) were examined for the parameters significant in the Pearson chi-square test (tumor subtype, grade, margins of excision, lymphatic invasion, tumor subtype). Multivariate Cox’s proportional hazard regression analysis was conducted including factors significant in the univariate analysis using a forward stepwise procedure. The statistical significance was set at 5%. Commercial software was used for the statistical analysis (IBM SPSS Statistics 21; IBM Corporation, Chicago, IL, USA).
Results
Characteristics of the Study Population
The individual-animal data are available in Supplemental Table 1.
Two hundred twenty-nine female dogs were included in the study. At the time of diagnosis, most of them (63%, 145/229) were intact. Twenty-two breeds were represented in the study, and among these, German Shepherd and Yorkshire Terrier were the most common (13% and 11%, respectively). The ages ranged from 3 to 18 years (median = 10.1 years). The median follow-up for censored dogs was 21 months, ranging from 2 to 39 months, whereas for cases that died of causes related to the mammary tumor, the median follow-up was 7 months with a range from 0 to 20 months. During the follow-up period, 33% (75/229) developed distant metastases, 13% (31/229) had local recurrence, and 26% (63/229) died from tumor-related causes. With Pearson chi-square tests, neuter status, age, and breed were found not to be associated with tumor-specific overall survival, local recurrence, or distant metastases.
Prognostic Significance of the Tumor Subtypes (2011 Classification)
Of the 229 tumors included in the study, 169 (74%) were malignant and 60 (26%) were benign. The 14 tumor subtypes included in the study and the number of cases for each category are described in Table 1. During the follow-up period, no events (distant metastases, local recurrence, or tumor-related death) were registered for dogs with benign tumors (complex adenoma, benign mixed tumor, simple adenoma) and dogs with carcinoma arising in benign mixed tumor. Therefore, these 4 tumor subtypes were excluded from the statistical analysis. The statistical analysis included the other 10 tumor subtypes.
Number of Cases for Each Canine Mammary Tumor Subtype Included in the Study.
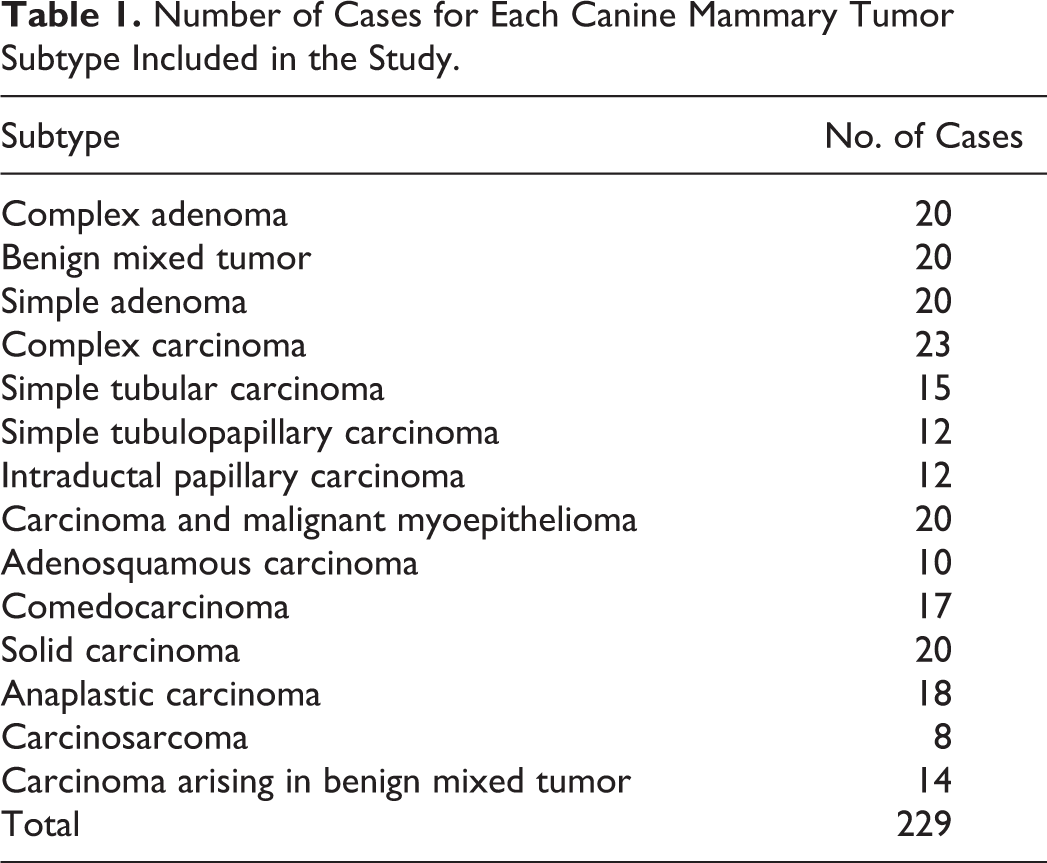
A significant association (log rank; P < .001) was found between the subtypes of the 2011 classification and the tumor-specific overall survival. Differences in overall survival are shown by the Kaplan–Meier curves (Fig. 1). Median survival time (MST), mean survival time (mST), 1-year and 2-year survival rates, and hazard ratio (HR) for tumor-related death (univariable Cox regression, reference category: complex carcinoma) for each subtype are provided in Table 2. Briefly, anaplastic carcinoma and carcinosarcoma were the most aggressive tumor subtypes, both having a MST of 3 months and a mST of 4.2 months. Animals diagnosed with these 2 tumors died within 1 year and had a risk of death more than 150 times higher than dogs with a complex carcinoma. A slower disease progression was recognized for adenosquamous carcinoma (MST = 18 months; mST = 12.6 months), comedocarcinoma (MST = 14 months; mST = 18 months), and solid carcinoma (MST = 8 months; mST = 15.6 months), although the risk of dying from tumor-related causes was still high (25 times more than complex carcinoma). Dogs with simple tubulopapillary carcinoma, intraductal papillary carcinoma, and carcinoma and malignant myoepithelioma were more likely to die than dogs with complex carcinoma (HR = 10.4–13). For these 3 subtypes, the MST was not reached during the follow-up period and the mST was more than 19 months. For the same 3 subtypes the 1-year survival rate varied from 70% to 83% and the 2-year survival rate varied from 50% to 67%. Dogs with simple tubular carcinoma and complex carcinoma experienced prolonged overall survival.
Median and Mean Survival Times (Months), Survival Rates, and Results of the Univariate Cox Regression for Each Subtype of Canine Mammary Tumor.a
Abbreviations: HR, hazard ratio; mST, mean survival time; MST, median survival time; nr, not reached; NS, nonsignificant.
a P < .05.
The 2011 classification showed an association with the development of local recurrence (chi-square; P = .012) and distant metastases (chi-square; P < .001). Recurrence and metastatic rates for each tumor subtype are included in Table 3. In parallel with the results of the survival analysis, the highest metastatic rates were registered for carcinosarcoma (100%) and anaplastic carcinoma (89%). Distant metastases were also common in dogs with comedocarcinoma (82%), solid carcinoma (65%), and adenosquamous carcinoma (60%). The adenosquamous carcinoma was also the tumor that most commonly recurred (50%), even more than anaplastic carcinoma (44%) and carcinosarcoma (37%).
Rates of Local Recurrence and Distant Metastasis for Each Tumor Subtype (169 Malignant Tumors).
Prognostic Significance of Histologic Grade, Lymphatic Invasion, Margins, and Tumor Diameter
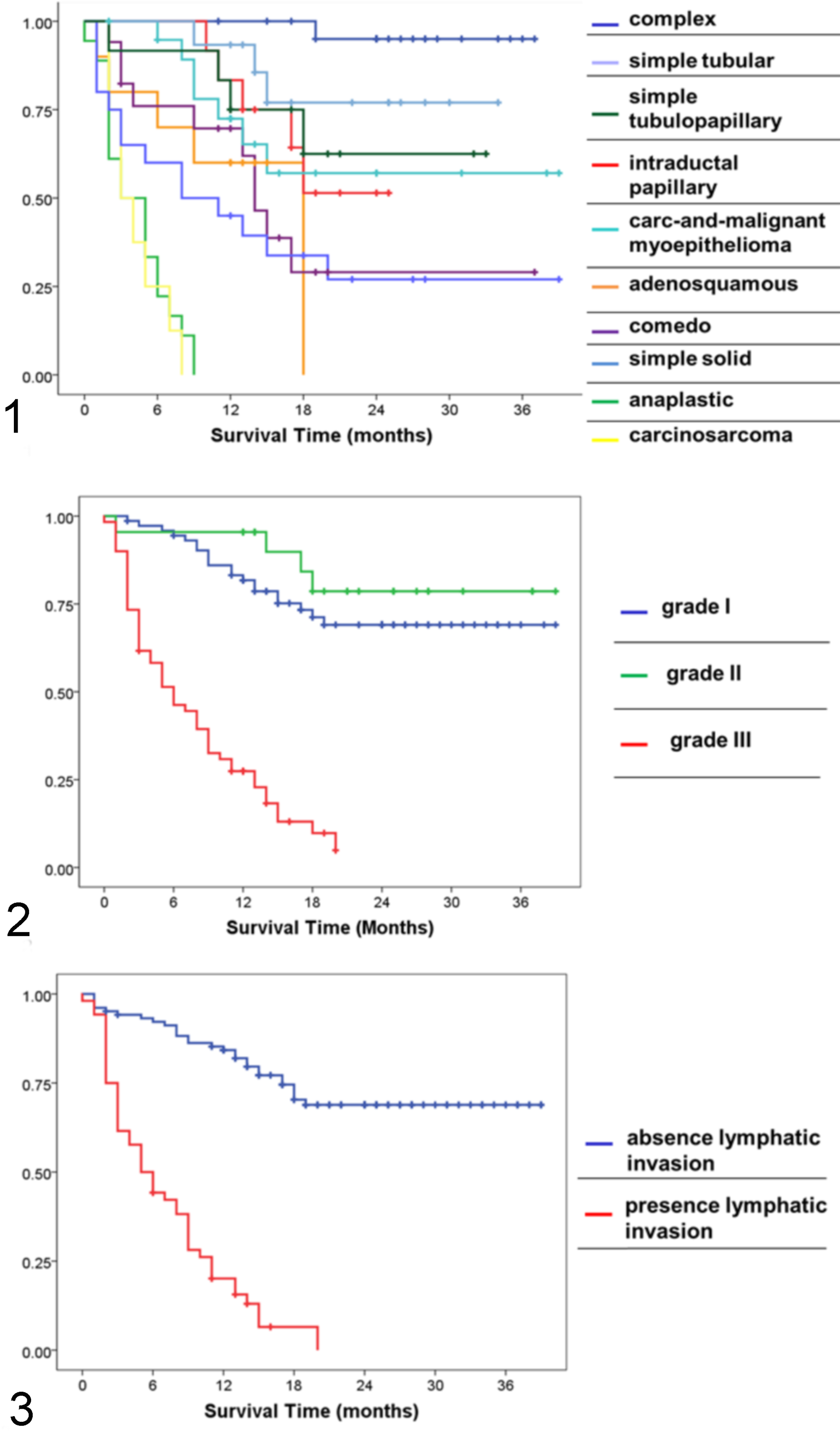
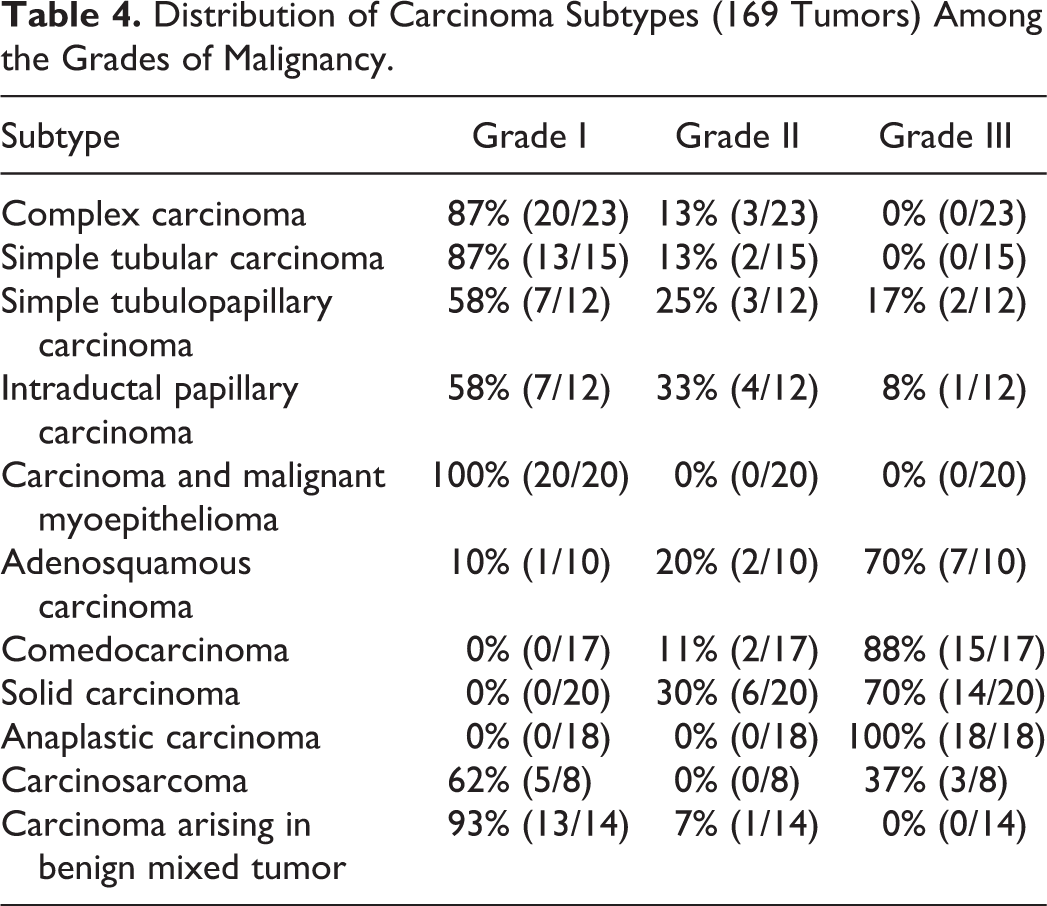
The 169 malignant tumors were graded as follows: 86 (51%) grade I, 23 (14%) grade II, and 60 (35%) grade III. The distribution of carcinoma subtypes among the grades of malignancy is described in Table 4. Carcinomas arising in benign mixed tumor, complex carcinomas, simple tubular carcinomas, simple tubulopapillary carcinomas, intraductal papillary carcinomas, carcinoma and malignant myoepitheliomas, and carcinosarcomas were mostly classified as grade I, whereas adenosquamous carcinomas, comedocarcinomas, solid carcinomas, and anaplastic carcinomas were usually grade III. The histologic grade was significantly associated with tumor-specific overall survival (log rank; P < .001), as shown by the Kaplan–Meier curves (Fig. 2). Grade III tumors had a MST of 6 months, a mST of 7.8 months, a 1-year survival rate of 27%, a 2-year survival rate of 0%, and a 7.1 times higher risk of death (univariate Cox regression; P < .001) compared with grade I. It is interesting that grade I and grade II tumors behaved similarly, both categories being associated with a prolonged tumor-specific overall survival (MST not reached for both grade I and grade II, mST of 30.3 months for grade I and 33.4 months for grade II, 1-year survival rate of 81% for grade I and 96% for grade II, 2-year survival rate of 69% for grade I and 78% for grade II) (Table 5). The histologic grade was also associated with local recurrence (chi-square; P = .015) and distant metastases (chi-square; P < .001), with grade III tumors being more aggressive (recurrence rate = 32%, metastatic rate = 87%) than grade I (recurrence rate = 12%, metastatic rate = 19%) and grade II (recurrence rate = 9%, metastatic rate = 30%) tumors (Table 5).
Distribution of Carcinoma Subtypes (169 Tumors) Among the Grades of Malignancy.
Survival Rates, Rates of Local Recurrence, and Rates of Distant Metastasis of the 169 Malignant Tumors According to Grade of Malignancy, Lymphatic Invasion, and Excision Margins.a
a Free or not free means that neoplastic cells were absent or present, respectively, at the excisional margin.
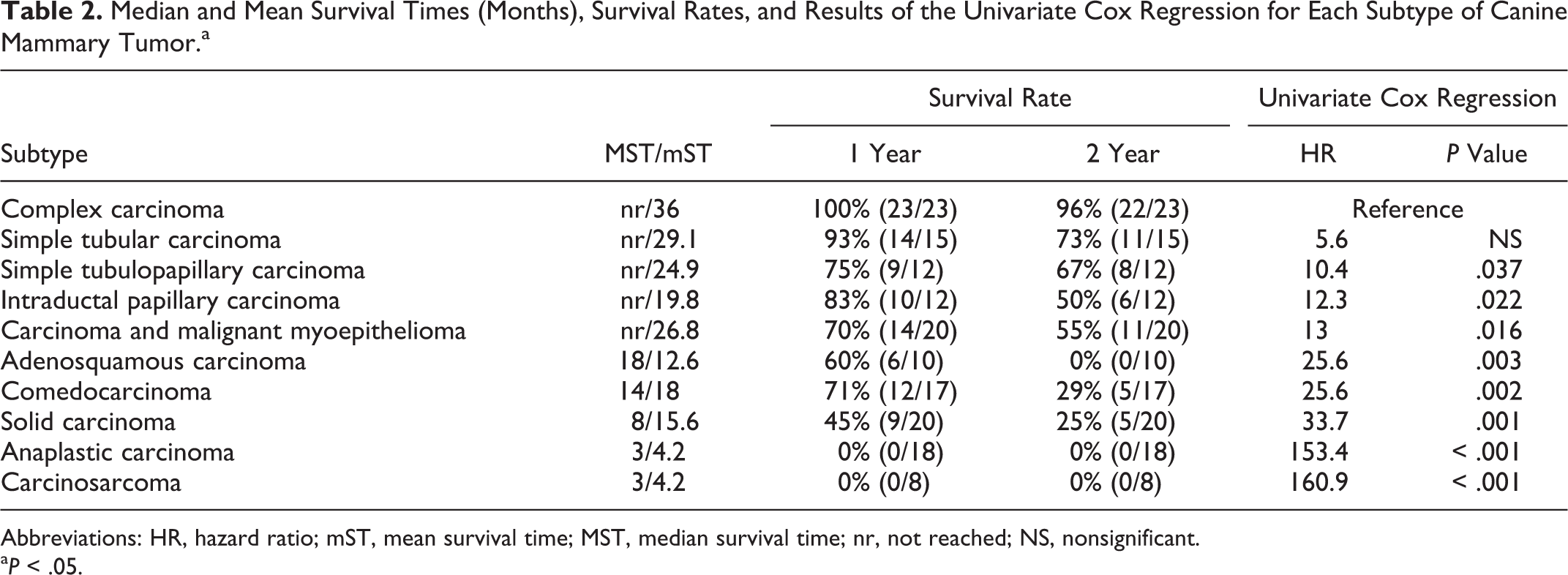
Lymphatic invasion, defined as infiltration of peritumoral lymphatic vessels (assessed in all dogs) and/or infiltration of regional lymph node (available for 92 dogs), was found in 31% (52/169) of the malignant tumor cases. Significant differences in tumor-specific survival (log rank; P < .001) (Fig. 3), risk of death (HR = 8.6, univariate Cox regression; P < .001), local recurrence (chi-square; P = .019), and distant metastases (chi-square; P < .001) were identified between dogs with and without lymphatic invasion. As expected, dogs with lymphatic invasion had a shorter tumor-specific overall survival (MST = 5 months, mST = 7.1 months, 1-year survival rate = 19%, 2-year survival rate = 0%) compared with dogs without lymphatic invasion (MST not reached, mST = 30.2 months, 1-year survival rate = 84%, 2-year survival rate = 69%) and more frequently developed distant metastases (88% vs 25%) and local recurrence (31% vs 13%) (Table 5).
Neoplastic infiltration at the excision margins was detected in 25% (42/169) of the dogs with malignant tumors. Neoplastic infiltration of margins of excision was significant for local recurrence (chi-square; P = .003) but did not affect the tumor-specific overall survival or distant metastases (Table 5).
The tumor diameter, which ranged from 0.2 to 9 cm (median = 3.5 cm), was significantly associated with tumor-specific overall survival (chi-square; P < .001), local recurrence (chi-square; P < .001), and distant metastasis (chi-square; P < .001).
Multivariate Cox Regression Survival Analysis
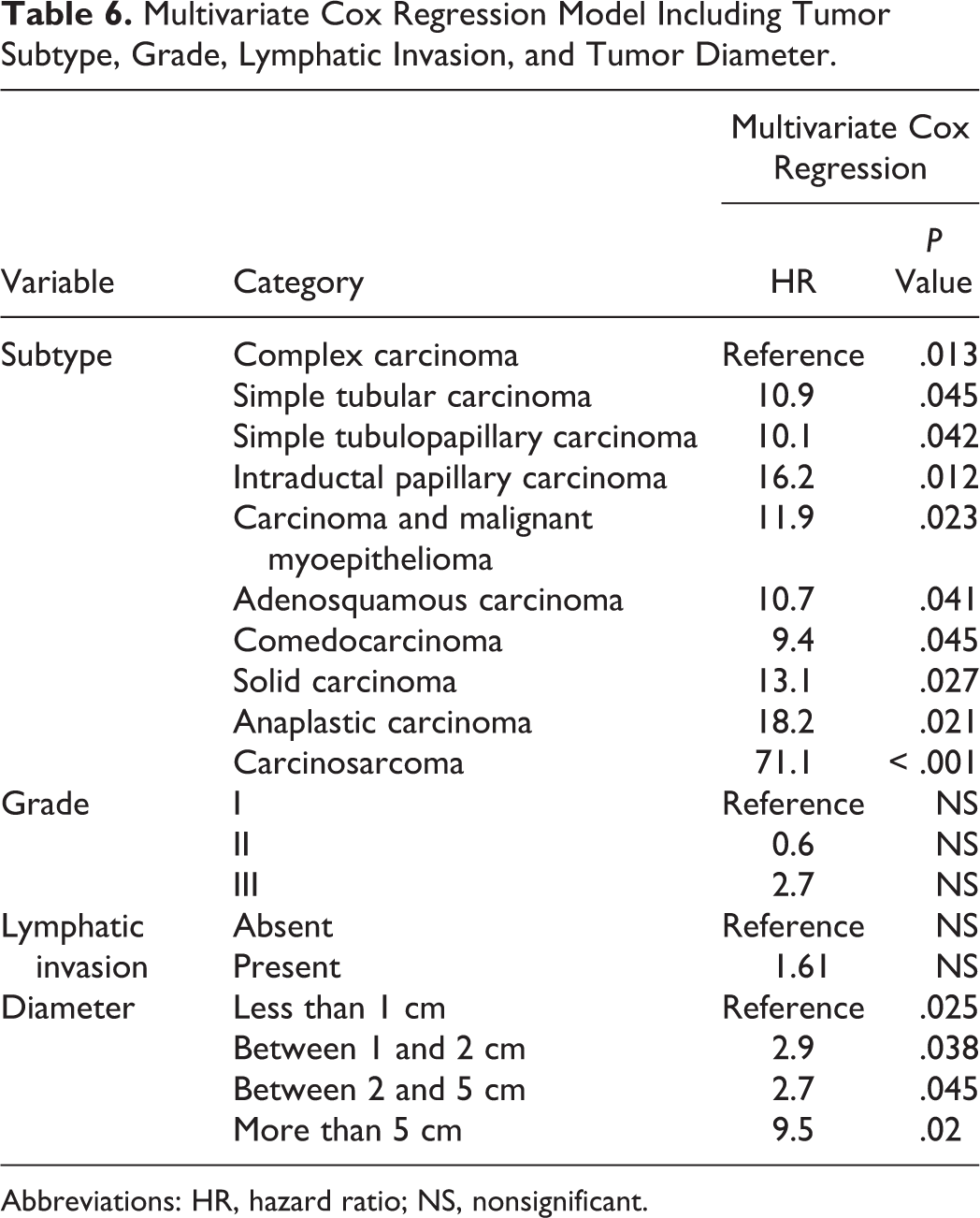
The multivariate Cox regression model included the tumor subtype, grade, lymphatic invasion, and tumor diameter (< 1 cm, between 1 and 2 cm, between 2 and 5 cm, and > 5 cm). The model was able to explain significantly the variability seen in the population (P < .001). The only 2 significant independent parameters were the tumor subtype (P = .013) and tumor diameter (P = .025), whereas grade (P = .098) and lymphatic invasion (P = .283) lost their prognostic value (Table 6).
Multivariate Cox Regression Model Including Tumor Subtype, Grade, Lymphatic Invasion, and Tumor Diameter.
Abbreviations: HR, hazard ratio; NS, nonsignificant.
Discussion
This 2-year prospective follow-up study demonstrated that the 2011 classification is a prognostic indicator for tumor-specific overall survival, local recurrence, and distant metastases in dogs affected by mammary tumors, supporting its application in routine diagnostic pathology. The main strength of the study is that it has been specifically designed to have an adequate number of cases for each tumor subtype to be able to analyze them separately and identify subtype-specific clinical outcomes. With this goal in mind, dogs with more than 1 malignant mammary tumor subtype and dogs that received adjuvant treatments (eg, chemotherapy) were not included in the study to prevent biases.
The independent prognostic significance retained by the 2011 classification in the multivariate analysis strengthens the biologic implications of the discovered association with the tumor-specific overall survival. The 14 subtypes analyzed in the study are the most common mammary tumor subtypes in dogs, 4,5,12 and the results obtained in terms of tumor-specific overall survival, recurrence, and distant metastases are considered potentially relevant information for clinicians and oncologists for prognostication and clinical decision making.
As expected, the 3 benign subtypes included in the study (complex adenoma, benign mixed tumor, and simple adenoma) did not recur or metastasize and were not associated with the animal’s death, suggesting that the histologic criteria of malignancy used (more than 3 layers of epithelial stratification, randomly distributed areas of necrosis, marked nuclear pleomorphism assessed in the areas of highest degree of anisokaryosis, and more than 3 mitoses in 10 high-power fields assessed in the fields with the highest mitotic activity) did not lead to underestimation of malignancy. It is interesting that carcinoma arising in benign mixed tumor, which was predominantly classified as grade I, had a biologic behavior similar to the benign subtypes. This result could support the hypothesis that this is a tumor in the early phases of malignant transformation, 4,14 or it could be argued that our histologic criteria of malignancy are too strict and might lead to an overestimation of the malignancy. Further research is warranted to refine the histologic criteria to differentiate this tumor from its benign counterpart (benign mixed tumor) and, in general, to understand the clinical implications of detecting foci of malignancy within an otherwise benign-looking mammary neoplasm.
Similar to previous studies, 1,2 we noted that anaplastic carcinomas had a worse prognosis than solid carcinomas, and solid carcinomas were more aggressive than simple tubulopapillary carcinomas and complex carcinomas. Besides, we discovered that carcinosarcomas had a mortality pattern similar to anaplastic carcinomas, with dramatically short tumor-specific overall survival times and high rates of local recurrence and distant metastases.
The biologic behavior of the adenosquamous carcinoma was also described for the first time with longer tumor-specific overall survival than anaplastic carcinoma and carcinosarcoma but a higher propensity for local recurrence (half of the cases). This tendency for local relapse might be attributable to the squamous component of the tumor, which when present in other anatomic locations is locally invasive (eg, cutaneous and oral squamous cell carcinoma). 19 On the contrary, comedocarcinoma was the most commonly metastatic tumor after anaplastic carcinoma and carcinosarcoma, which substantiates our previous findings on a different canine population. 14
The prognosis associated with canine intraductal papillary carcinoma had never been documented, and in this study, it appeared to be more favorable than that of other subtypes (eg, anaplastic carcinoma, carcinosarcoma, solid carcinoma, comedocarcinoma, adenosquamous carcinoma), although more cases are required to draw a more definitive conclusion on the behavior of this recently described subtype. It would also be interesting to compare the prognosis of the intraductal papillary carcinoma with that of the ductal carcinoma (not included in this study), which is believed to have a similar origin to the ductal system but displays a different type of growth and cell morphology (intraductal papillary carcinoma has papillae formed by columnar cells with moderate cytoplasm and open-faced nuclei, whereas ductal carcinoma has bilayered cords formed by cuboidal cells with scant cytoplasm and hyperchromatic nuclei). 4 These 2 duct-associated tumor subtypes have been recently described in cats, with preliminary follow-up data suggesting that they have a less aggressive behavior compared with other mammary carcinomas. 22
Survival analysis revealed a lower risk of tumor-related death for dogs with complex carcinoma compared with dogs with carcinoma and malignant myoepithelioma, confirming previous findings 13 and supporting the hypothesis of a protective role of well-differentiated myoepithelium as it occurs in complex carcinoma. 13 –15 In addition, the present study demonstrated that, despite being associated with a more guarded prognosis compared with complex carcinoma, the carcinoma and malignant myoepithelioma is not a very aggressive mammary malignancy in dogs, with most animals still alive 2 years after surgery. This is supported by the less aggressive pathologic features described by Yoshimura and colleagues 21 in tumors with a malignant myoepithelium compared with simple solid carcinomas.
It is rather surprising that there was no difference in the tumor-specific overall survival of dogs with simple tubular carcinoma compared with dogs with complex carcinoma. This contradicts previous results from our group 14 and other research groups 1,6,11 and is likely to be a reflection of the high proportion of grade I (87%) simple tubular carcinomas in this study compared with previous studies, suggesting that this finding should be interpreted with caution and that grade may be considered particularly relevant for the prognostication of this tumor subtype.
Less common mammary tumor subtypes such as lipid-rich carcinoma, micropapillary invasive carcinoma, and benign myoepithelioma were not included in this study due to the inadequate number of cases recruited. A multi-institutional approach in the future could help in overcoming this limitation, clarifying the biologic behavior also for these uncommon tumor entities.
Corroborating previous findings, 1,6,12 our study showed that grade, lymphatic invasion, and tumor diameter were associated with tumor-specific overall survival, recurrence, and distant metastases. However, lymphatic invasion and grade lost their prognostic power in the multivariate survival analysis since tumor diameter and subtype were able to significantly explain the variability in tumor-specific overall survival within this cohort of dogs.
As the size of the metastatic deposits in the lymph node has been recently demonstrated to have a significant effect on survival, 2 future studies should take into consideration this additional aspect, which could potentially be an independent prognostic factor superior to the simple assessment of presence or absence of lymphatic invasion.
With respect to the tumor grade, our survival rates were similar to those reported by other authors, 6,12 although they were slightly lower for grade I tumors. This could be due to intrinsic differences in the study populations and/or variations in how the grade has been calculated, as a certain degree of interobserver variability has been repeatedly documented in recent studies. 16,17 We adopted the grading system published by Peña and colleagues, 12 with the only difference being that we focused our evaluations on the neoplastic epithelial component only, as previously done. 6,9,13,14 Since we did not consider for grading purposes other tumor cell populations (myoepithelial and mesenchymal), an underestimation of malignancy in cases of carcinoma and malignant myoepithelioma and carcinosarcoma is possible, which could explain the lower survival rates of our grade I tumors compared with the study by Peña et al. 12 The reason for our choice is that specific criteria to estimate the malignancy of myoepithelial cells and mesenchymal cells in biphasic tumors, substantiated by the clinical outcome, have not yet been precisely established in the literature. We believe that further research on this topic is crucial in order to accurately and reliably prognosticate biphasic mammary tumors in dogs.
As in previous studies, we noted that it is the switch from grade I to grade III (not the switch from Grade I to Grade II) that is prognostic for tumor-specific overall survival. Grade II tumors behave similarly to grade I tumors, with prolonged overall survival times and low rates of local recurrence and distant metastasis. 9,12 This could open the debate about the utility of the grade II category in the clinical setting. Further research in this area may include the improvement of the 3-tier histologic grading system, as lately performed for feline mammary tumors modifying the range subcategories within the mitotic count and the assessment of the nuclear morphology. 10 An alternative could be the development of a 2-tier histologic grading system, as recently done for canine cutaneous mast cell tumors. 8
The ability of the neoplastic extension to margins to predict local recurrence, but not distant metastasis or tumor-specific overall survival, is intuitive and has been demonstrated for other canine malignancies, 19 but to the best of our knowledge, this is the first time that this is reported in CMTs.
In conclusion, this study validated in multivariate analysis the 2011 classification as an independent prognostic indicator in dogs with mammary tumors, suggesting that this could be used as the basis for future studies to look at other prognostic markers and therapeutic interventions. Subtype-specific clinical outcomes were described and these represent potentially relevant information for clinicians and oncologists for prognostication and clinical decision making. The tumor diameter was confirmed to be an independent prognostic factor in dogs with mammary tumors, and the excisional margins were found to be predictive of local recurrence only, similar to other canine neoplasms.
Lymphatic invasion and histologic grade were also proven to be prognostically relevant, although in this study population, they were not confirmed to be independent prognosticators.
Footnotes
Acknowledgments
We thank the staff of the Histology Laboratory of the Diagnostic Service of Veterinary Anatomic Pathology of the University of Padua (Italy) for the assistance with sample processing and the clinicians who have submitted the samples and kindly agreed to collect the follow-up data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Department of Comparative Biomedicine and Food Science of the University of Padua (Italy).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
