Abstract
Canine spindle cell mammary tumor (CSCMT) is an infrequent canine mammary tumor (CMT) composed of spindle or fusiform cells, which represents a challenge for pathologists and clinicians. Mammary tumors submitted for histopathology from 1998 to 2013 and compatible with CSCMTs were retrospectively selected. The tumors were diagnosed based on the hematoxylin and eosin (HE)–stained section; malignant tumors were graded using a canine soft tissue sarcoma grading scheme and a canine mammary tumor grading scheme, and they were further assigned a diagnosis based on immunohistochemistry (IHC) for pancytokeratin, cytokeratin 14, p63, calponin, vimentin, Ki-67, CD31, desmin, myosin, smooth muscle actin, glial fibrillary acidic protein, and S-100. The origin of the tumors was assessed as mammary, skin, or unknown. The prevalence of CSCMT was 1% of all CMTs. CSCMTs included 3 benign tumors (1 angioma and 2 benign myoepitheliomas) and 67 malignant tumors that after IHC were diagnosed as malignant myoepithelioma (64%), carcinoma and malignant myoepithelioma (19%), hemangiosarcoma (8%), undifferentiated sarcoma (5%), peripheral nerve sheath tumor (3%), and fibrosarcoma (2%). The diagnosis based on the HE-stained section differed from the diagnosis after IHC in 75% of the malignant cases. The majority of malignant CSCMTs were solitary (57%) large tumors (6.42 ± 3.92 cm) with low metastatic potential and high survival rate (8% tumor-related mortality). Higher sarcoma grade was associated with older age (P = .034) and greater tumor size (P = .037). Malignant CSCMTs need to be evaluated by IHC to ensure the histotype and the relatively benign clinical behavior, despite their large size.
Canine mammary tumor (CMT) is the most prevalent neoplasm in female dogs, representing between 25% and 50% of all tumors, 15 and is malignant in 30% to 50% of cases. 7,12,15 The histological diagnosis of CMTs is difficult due to their histological complexity, since they can be of epithelial, myoepithelial, or mesenchymal origin and may develop from multiple combinations of the listed cell types. 7 The most frequent types of CMTs are complex adenoma, benign mixed tumor, complex carcinoma, and solid carcinoma. 7,12 There is an infrequent group of CMTs, composed of spindle cells, that have a similar appearance with routine techniques (hematoxylin and eosin [HE]) and therefore need further immunophenotyping to be properly diagnosed. 11 They represent a diagnostic challenge for veterinary pathologists and clinicians, since there is scarce information on how to differentiate these histological types, and the knowledge of their biological behavior is limited or absent. 7 The authors of this study have used the term canine spindle cell mammary tumors (CSCMTs) to encompass these neoplasms. Their common histological feature is that they are composed of spindle or fusiform cells. Following the current histologic classification of CMTs, 7 several histological subtypes are included under this term: carcinoma and malignant myoepithelioma (with minimal epithelial proliferation), benign and malignant myoepithelioma, squamous cell carcinoma–spindle cell variant, spindle cell carcinoma, fibrosarcoma, and hemangiosarcoma. That is, CSCMTs comprise carcinomas (epithelial and/or myoepithelial origin) and sarcomas of uncertain origin that are indistinguishable on a morphological basis.
According to the scant available literature, CSCMTs are infrequent tumors. 7 Fibrosarcomas are considered the most frequent type of mammary sarcoma in the dog. 5 In a classical study, 8 fibrosarcomas and osteosarcomas bear the lowest survival time among CMTs. There is no information regarding the behavior of other CSCMT types, except for spindle cell carcinoma and malignant myoepithelioma (with prominent epithelial component) that has been classified as an aggressive type of tumor. 12
Given the low prevalence and lack of studies concerning CSCMTs, specific data regarding their epidemiology, morphology, immunohistochemical characteristics, and biological behavior are missing. As an example, some CSCMTs usually diagnosed as myoepitheliomas could correspond to perivascular wall tumors or peripheral nerve sheath tumors of mammary origin that have never been demonstrated to exist in the female dog. The aims of the current study were to retrospectively collect a reliable number of CSCMTs from 3 different institutions; determine their epidemiology, histological subtypes, and grade of malignancy by morphological and immunohistochemical evaluation; and determine their clinical and prognostic features, when possible.
Materials and Methods
Selection of Cases and Patient Information
Cases of dogs with mammary tissues submitted for histopathology and compatible with CSCMTs from 1998 to 2013 (16 years) were retrospectively selected from the databases of 3 different institutions: (1) Hospital Clinico Veterinario, Veterinary Medicine School, Complutense University of Madrid (UCM), Spain; (2) Animal and Veterinary Research Centre (CECAV), University of Trás-os Montes and Alto Douro (UTAD), Vila Real, Portugal; and (3) DIMEVET, University of Milano, Italy. The criteria of inclusion/exclusion were as follows: tumor with preponderance of spindle cell morphology was the single criterium of inclusion; osteosarcomas, chondrosarcomas, carcinosarcomas, mixed tumors, and carcinoma and malignant myoepithelioma with evident epithelial proliferation were excluded (only carcinomas and malignant myoepitheliomas with minimal, practically imperceptible, scored as less than 5% epithelial proliferation were accepted for the study).
Data of the animals were annotated (age, breed, reproductive status). The following tumor-associated information was also recovered: tumor size, presence and type of other mammary tumors (if any), and lymph node and distant metastases of CSCMT at presentation. Information related to the follow-up period, determined for a minimum of 2 years, was retrieved from the clinical files and/or obtained by phone calls to the owners or referral veterinarians.
Histopathology
The selected tumors were morphologically reviewed on HE-stained sections. Diagnoses retrieved from electronical archives were, when necessary, reviewed and updated according to the current histological classification of CMTs. 7 The origin of the neoplasms as mammary, skin, or unknown was determined by examining the HE-stained section, based on the presence of the corresponding histological structures (ie, for mammary gland tumors, the existence of mammary acini and the corresponding depth of the main tumor). Doubtful cases were classified as unknown.
In addition, 2 different histological grading systems were applied: a canine soft tissue sarcoma grading system that takes into account tissue differentiation, mitotic count, and percentage of necrosis (grades I, II, III), 4 which might be applicable and useful even in nonmesenchymal mammary neoplasms, and, when applicable, the grading system for canine mammary carcinomas, based on the evaluation of the percentage of tubule formation within the carcinoma areas, nuclear pleomorphism of the malignant component, and mitotic count (grades I, II, III). 10 Mitotic counts were performed in 10 high-power fields (400×, diameter of the field of view = 0.55 mm) using an Olympus (Germany) BX50 microscope. Masson’s trichrome stain (n = 76) and periodic acid–Schiff reaction (PAS) (n = 81) were performed in a subset of cases. Collagenization of the stroma (Masson’s trichrome) was evaluated as negative/scant and moderate/strong. The presence of PAS-positive membranes was categorized as positive or negative.
Immunohistochemistry
Immunohistochemistry (IHC) for the identification of different markers was performed for malignant tumors when paraffin blocks contained adequate residual tissue (n = 77), following previously established protocols, including positive and negative controls (Table 1). IHC was performed on malignant tumors regardless of the origin (mammary gland, skin, or unknown), and a second diagnosis was assigned based on the HE and IHC findings. Pancytokeratin (AE1/AE3), cytokeratin 14 (CK-14), p63, calponin (calp), and vimentin (vim) immunoexpression was used to distinguish cells of epithelial, myoepithelial, and mesenchymal origin 11 (Table 2). Positive IHC labeling was defined as the presence of more than 10% of immunoreactive cells and used in conjunction with the cellular morphology and localization of the evaluated antigen. 11 Myoepithelial cells were identified by their spindle morphology and at least 1 of the corresponding phenotypic markers (Table 2). In a context of monomorphic spindle cells (based on HE stain) with phenotypically identified myoepithelial cells, AE1/AE3-positive spindle cells were considered myoepithelial cells (immunohistochemical diagnosis: malignant myoepithelioma), whereas AE1/AE3-positive cuboidal cells arranged in groups or tubules were identified as epithelial cells (immunohistochemical diagnosis: carcinoma and malignant myoepithelioma). Some cases compatible by morphology (based on HE stain) with hemangiosarcoma, peripheral nerve sheath tumors (PNSTs), or perivascular wall tumors (PWTs) required additional immunohistochemistry (n = 13). For these cases, expression of CD31, desmin, myosin, smooth muscle actin (SMA), glial fibrillary acidic protein (GFAP), and S-100 antigens was investigated (Table 2). Ki-67 proliferation index was determined by counting positive and negative nuclei in 10 high-power-fields (40×), using a computer-assisted image analyzer (ImageJ; National Institutes of Health, Bethesda, MD). Using the IHC results, a final diagnosis was established, and the origin of the tumors (mammary, skin, unknown) was reassessed, including as mammary those cases with myoepithelial proliferations identified by morphology and IHC. Finally, only cases of mammary origin with IHC diagnosis were selected (n = 67).
Primary Antibodies Used for Immunophenotyping Canine Spindle Cell Malignant Mammary Tumors.
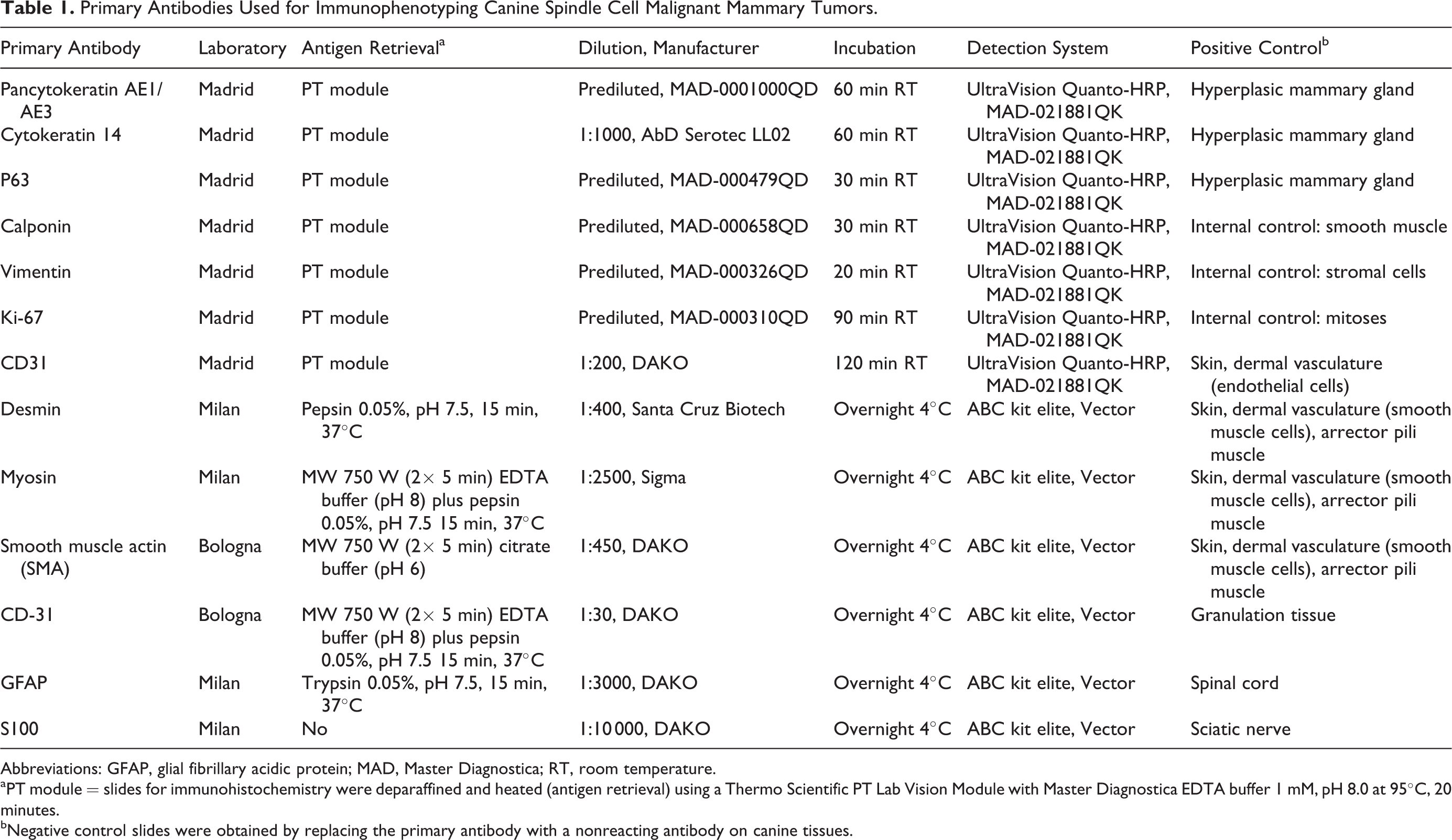
Abbreviations: GFAP, glial fibrillary acidic protein; MAD, Master Diagnostica; RT, room temperature.
aPT module = slides for immunohistochemistry were deparaffined and heated (antigen retrieval) using a Thermo Scientific PT Lab Vision Module with Master Diagnostica EDTA buffer 1 mM, pH 8.0 at 95°C, 20 minutes.
bNegative control slides were obtained by replacing the primary antibody with a nonreacting antibody on canine tissues.
Immunohistochemistry of Malignant Canine Spindle Cell Mammary Tumors.a
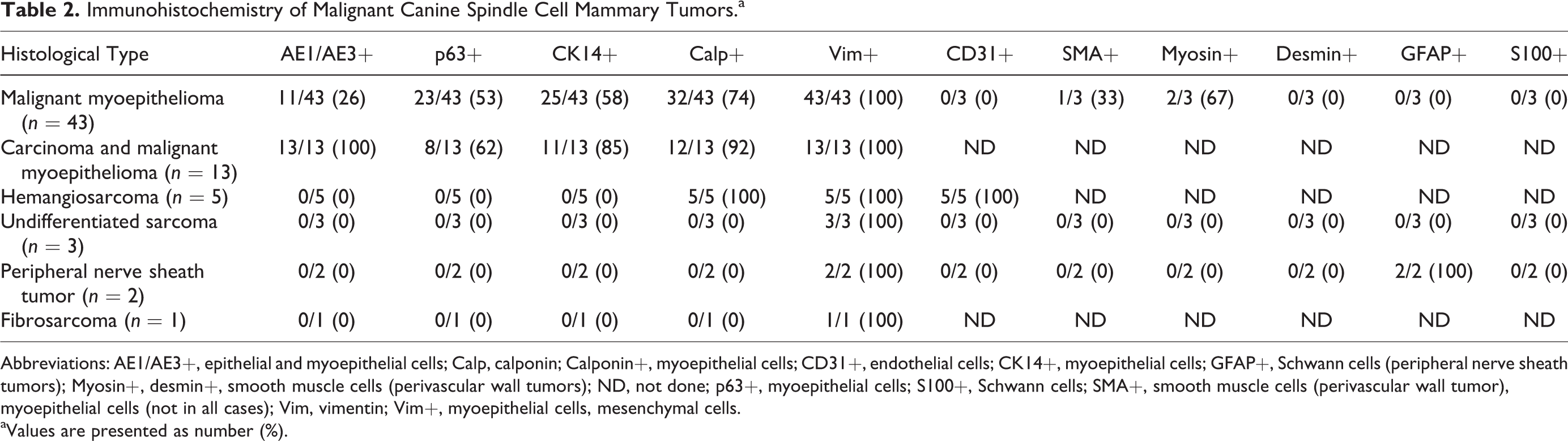
Abbreviations: AE1/AE3+, epithelial and myoepithelial cells; Calp, calponin; Calponin+, myoepithelial cells; CD31+, endothelial cells; CK14+, myoepithelial cells; GFAP+, Schwann cells (peripheral nerve sheath tumors); Myosin+, desmin+, smooth muscle cells (perivascular wall tumors); ND, not done; p63+, myoepithelial cells; S100+, Schwann cells; SMA+, smooth muscle cells (perivascular wall tumor), myoepithelial cells (not in all cases); Vim, vimentin; Vim+, myoepithelial cells, mesenchymal cells.
aValues are presented as number (%).
Statistical Analysis
Statistical analysis was performed with the statistical software IBM SPSS 25 (SPSS, Inc, an IBM Company, Chicago, IL), and differences were considered statistically significant at P < .05. The categorical variables analyzed and the categories established were as follows: institution of origin (Madrid, Vila Real, Milan), breed, spay status (intact/spayed), tumor size (T0, T1, T2), 16 lymph node metastasis and distant metastasis at presentation (yes/no), other CMTs (yes/no), other malignant CMTs (yes/no), other benign CMTs (yes/no), diagnosis based on examination of HE-stained sections, sarcoma grade (I, II, III), mammary tumor grade (I, II, III), diagnosis based on IHC, origin (mammary, skin, unknown), development of recurrence and/or metastasis (yes/no), and death by CSCMT (yes/no). The numerical variables analyzed were age, tumor size (cm), number of other CMTs, number of other benign CMTs, number of other malignant CMTs, mitotic count, and Ki-67 index.
Results
Pathologic Diagnosis of CSCMT
Of 9936 mammary tumors (mammary dysplasia was not included), 103 canine tumors with spindle cell morphology were selected. These had been submitted for histopathology as mammary tumors from 99 female dogs. The prevalence of spindle cell tumors of the mammary region was 1.03% (95% confidence interval [CI], 0.91–1.15) of all CMTs: 1.09% in Madrid, 0.83% in Milan, and 1.13% in Vila Real.
All the CSCMT cases, regardless of their origin (mammary, skin, or unknown), were histopathologically reviewed. After microscopic review on the HE slides, the neoplasms were reclassified as perivascular wall tumor/peripheral nerve sheath tumor (PWT/PNST) (21/103, 20.4%), fibrosarcoma (17/103, 16.5%), undifferentiated sarcoma (15/103, 14.6%), malignant myoepithelioma (13/103, 12.6%), carcinoma and malignant myoepithelioma (11/103, 10.7%), angioma (10/103, 9.7%), hemangiosarcoma (8/103, 7.8%), spindle cell carcinoma (6/103, 5.8%), and benign myoepithelioma (2/103, 1.9%).
Masson’s trichrome stain (n = 76) and PAS reaction (n = 81) were performed in cases of any origin (mammary, skin, unknown). The Masson’s trichrome stain was able to discriminate fibrosarcoma (n = 4) from malignant myoepithelioma (n = 43) by the presence of collagen deposition (moderate/strong vs none/scant, respectively) (P = .007).
Taking into account the HE findings and the IHC results (Figs. 1–12), the 103 CSCMT cases were finally classified as mammary (n = 80, IHC performed in 67 cases), skin (n = 11, IHC n = 4), and unknown origin (n = 12, IHC n = 6). One angioma and 2 benign myoepitheliomas were the only benign CSCMTs.
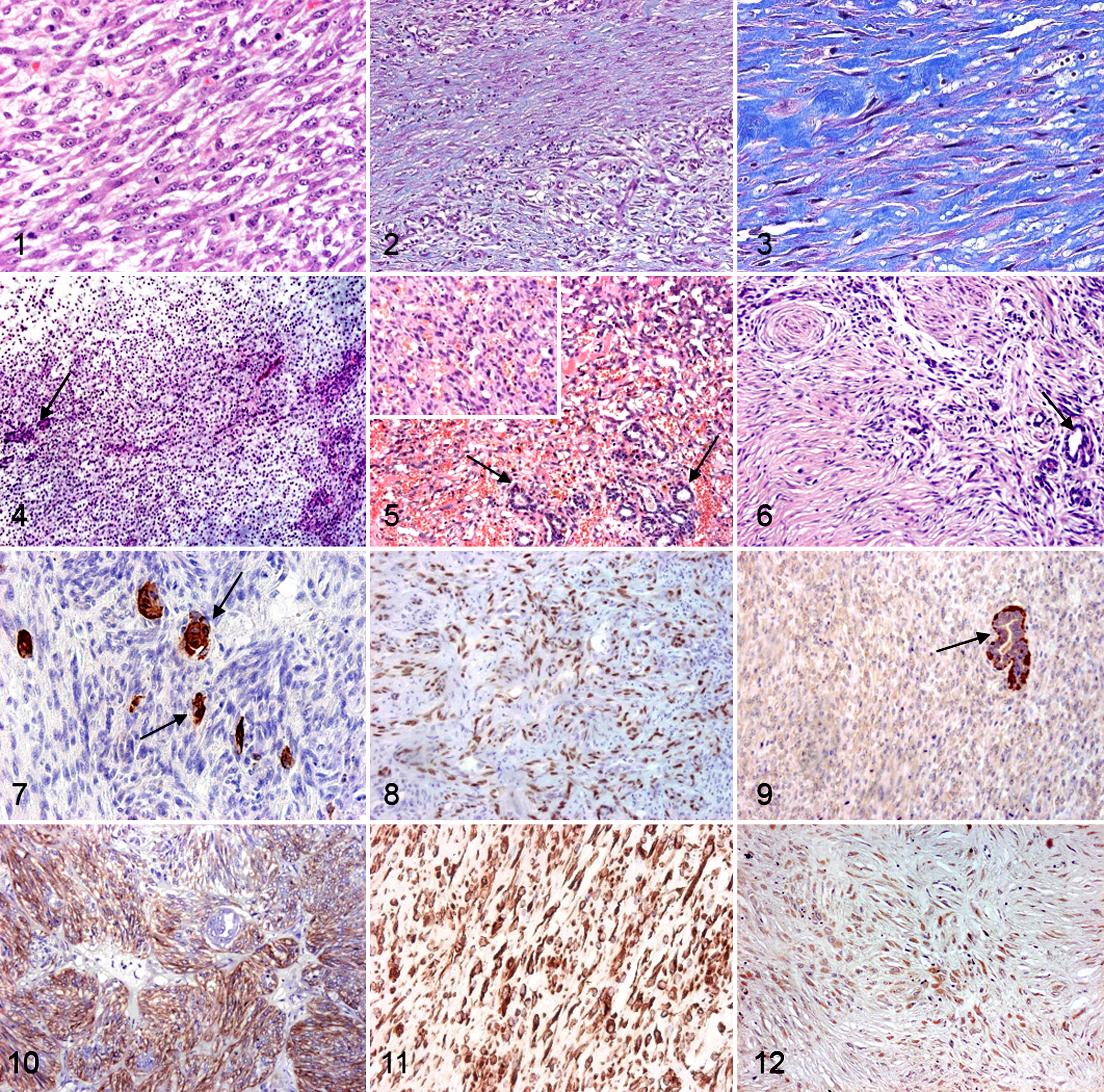
Malignant canine spindle cell mammary tumors, mammary gland, dog.
Immunolabeling of the malignant CSCMTs of mammary origin (n = 67) is summarized in Table 2. After IHC, the diagnoses were malignant myoepithelioma (43/67, 64%), carcinoma and malignant myoepithelioma (13/67, 19%), hemangiosarcoma (5/67, 7%), undifferentiated sarcoma (3/67, 4%), PNST (2/67, 3%), and fibrosarcoma (1/67, 1%). The diagnoses after IHC differed from HE-based diagnoses in 50 of 67 (75%) of the malignant cases (Table 3).
Canine Spindle Cell Mammary Tumors: Diagnoses Based on Histopathology and With Addition of Immunohistochemistry.
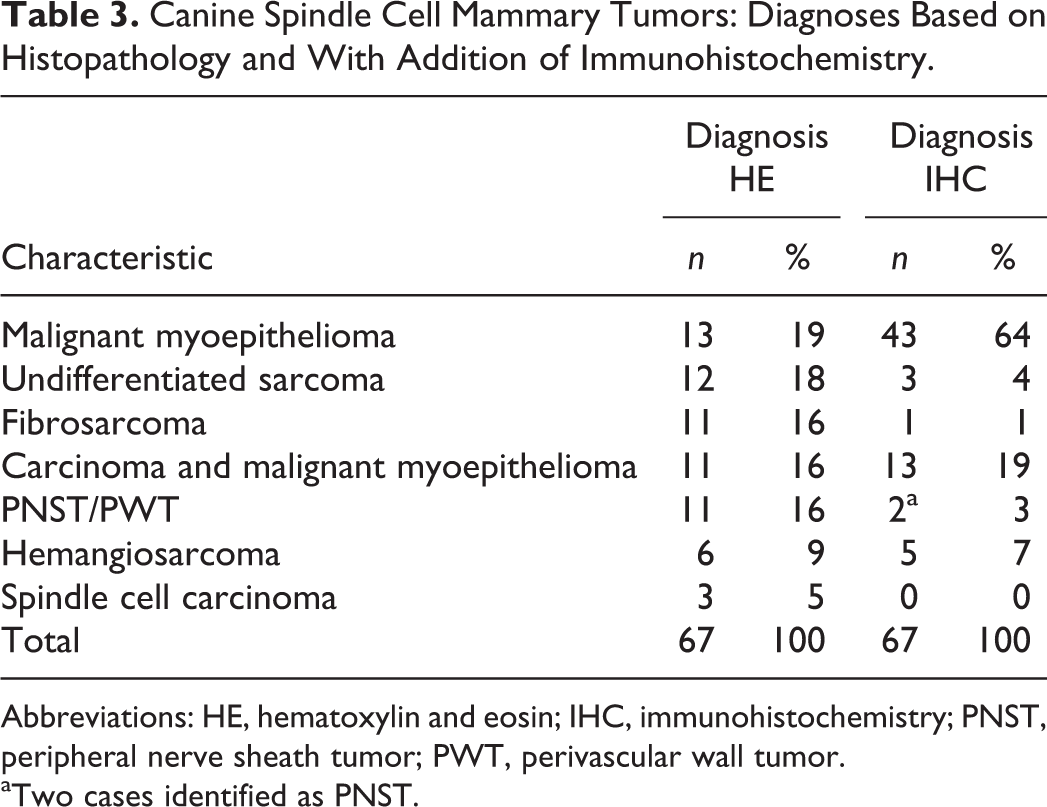
Abbreviations: HE, hematoxylin and eosin; IHC, immunohistochemistry; PNST, peripheral nerve sheath tumor; PWT, perivascular wall tumor.
aTwo cases identified as PNST.
Regarding immunohistochemistry for myoepithelial markers (p63, CK14, and calponin; Table 4) in the histotypes with myoepithelial proliferation (malignant myoepithelioma and carcinoma and malignant myopithelioma), expression of only 1 of the 3 myoepithelial markers was most frequent in malignant myoepithelioma (21/43, 49%) compared to carcinoma and malignant myoepithelioma (1/13, 8%) (P = .008). Immunolabeling for both CK14 and calponin was most frequent in carcinoma and malignant myoepithelioma (5/13, 38%) (P = .027). Immunolabelling for both p63 and CK14 was found in 1 case of carcinoma and malignant myoepithelioma and in none of the malignant myoepitheliomas.
Immunolabeling for Myoepithelial Markers in Malignant Canine Spindle Cell Mammary Tumors With Myoepithelial Proliferation.a
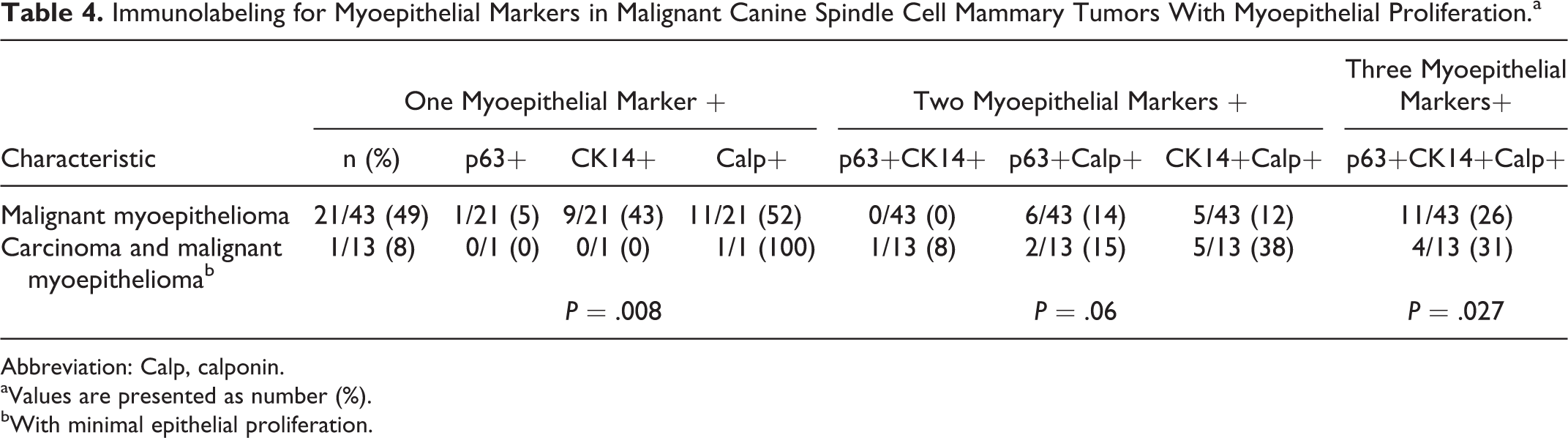
Abbreviation: Calp, calponin.
aValues are presented as number (%).
bWith minimal epithelial proliferation.
Epidemiology, Grading, and Clinical Data of Malignant CSCMTs
After considering the histomorphology based on HE and IHC, 70 CSCMTs were included: 3 benign (2 benign myoepitheliomas and 1 angioma) and 67 malignant. The following data refer only to the malignant neoplasms (malignant CSCMTs). The mean age of female dogs with malignant CSCMTs was 10.26 ± 2.23 years (minimum = 5, maximum = 15). Fourteen breeds were represented, and, among these, mongrels and German Shepherds were the most common ones (37% and 20%, respectively). Some breeds were more prevalent depending on the institutions but with no significant differences (P = .051). At presentation, most of the female dogs were intact (41/67, 61%), and the majority of malignant CSCMTS were solitary (38/67, 57%) large tumors (6.42 ± 3.92 cm, minimum = 1, maximum = 20; T3 >5 cm = 53%). In dogs with other mammary tumors, the malignant CSCMT was the only malignant tumor in 67% of the cases (45/67). Information on regional lymph node involvement and distant metastases of malignant CSCMTs at presentation was available in 32 cases: 2 dogs had lymph node metastatic disease (2/32, 6%; 1 malignant myoepithelioma and 1 undifferentiated sarcoma), and 1 animal had distant metastasis (1/32, 3%, malignant myoepithelioma).
Information on tumor grades according to the different histotypes is provided in Table 5. According to the 2 different grading systems applied on HE sections (prior to IHC), soft tissue sarcoma grading (n = 64) and mammary tumor grading (n = 33), most neoplasms were soft tissue sarcoma grade III and mammary grade II: sarcoma grade I, 14.1%; sarcoma grade II, 34.4%; sarcoma grade III, 51.6%; mammary grade I, 21.22%; mammary grade II, 63.6%; and mammary grade III, 15.2%. Mammary tumor grade could only be applied to malignant myoepithelioma and carcinoma and malignant myoepithelioma. The 2 cases with lymph node involvement and the case with distant metastases were grade III tumors (sarcoma and mammary grades). Mitotic count in 10 high-power fields (HPF) (n = 67) and Ki-67 (n = 27) proliferation index of malignant CSCMTs were high (mitotic count median = 14.00, interquartile range = 17.25; Ki-67 median = 44.93%, interquartile range = 35.2). There was a nonsignificant (P = .13) positive correlation between mitotic count and Ki-67 index (Pearson’s coefficient = 0.30). Ki-67 medians varied significantly among the malignant CSCMT histotypes (P = .041): carcinoma and malignant myoepithelioma, 22.59% (n = 4, interquartile range = 21.45); hemangiosarcoma, 36.27% (n = 5, interquartile range = 28.63); malignant myoepithelioma, 50.81% (n = 17, interquartile range = 34.49); and undifferentiated sarcoma, 61.92% (n = 1).
Grading of Malignant Canine Spindle Cell Mammary Tumors.a
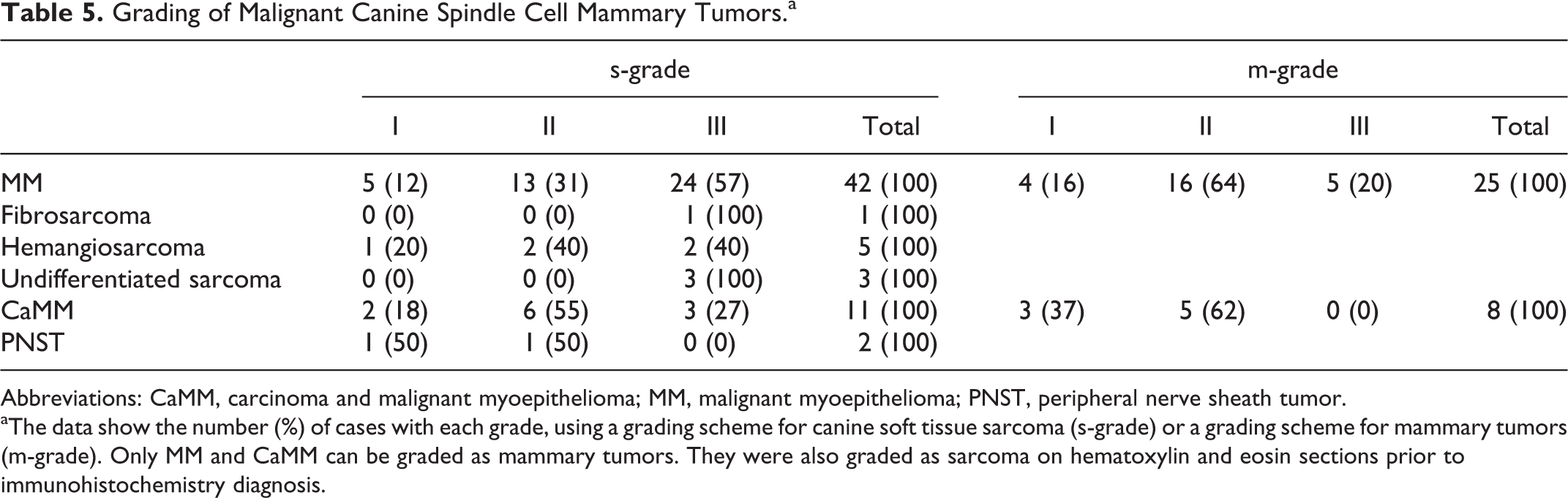
Abbreviations: CaMM, carcinoma and malignant myoepithelioma; MM, malignant myoepithelioma; PNST, peripheral nerve sheath tumor.
aThe data show the number (%) of cases with each grade, using a grading scheme for canine soft tissue sarcoma (s-grade) or a grading scheme for mammary tumors (m-grade). Only MM and CaMM can be graded as mammary tumors. They were also graded as sarcoma on hematoxylin and eosin sections prior to immunohistochemistry diagnosis.
Two-year postsurgery follow-up information regarding local recurrences and distant metastases was available in 28 cases. Five animals had local recurrence (5 malignant myoepitheliomas of sarcoma/mammary grades II/III), and 3 developed distant metastases (2 malignant myoepitheliomas of sarcoma/mammary grades II/ III and 1 hemangiosarcoma sarcoma grade III). In total, the percentage of recurrences/metastases was low (8/28, 29%). Information regarding 2-year postsurgery survival was available in 25 cases. Twelve animals were alive after this period (48%). Only 2 of 13 animals died of causes related to the malignant CSCMT (malignant myoepitheliomas of sarcoma/mammary grades II and III; mortality 8%) while the rest of dogs (n = 11) died due to other cancers (n = 6, 24%; multiple mast cell tumor, splenic primary hemangiosarcoma, limb osteosarcoma, apocrine adenocarcinoma, large oral neoplasia with lymph node invasion, and metastatic mammary carcinoma) or other causes (n = 5, 20%).
Relationships Among Variables
Tumor size was inversely associated with the presence of other mammary tumors: large malignant CSCMTs were usually solitary (mean 7.56 cm, P = .042). Moreover, when large malignant CSCMTs coexisted with other mammary tumors, it was more frequent that mammary tumors were benign (mean 7.43 cm, P = .025). Tumor size (cm) was associated with the presence of lymph node metastasis at presentation, although without statistical significance (P = .053).
High soft tissue sarcoma grade was associated with age as follows: older dogs had more frequently grade III tumors. Bonferroni test statistically separated the age of dogs of grades I and III (mean age 8.67 ± 1.02 and 10.79 ± 1.97 years, respectively; P = .034). Sarcoma grade was also associated with the size of tumors (P = .037), having T3 tumors (>5 cm) higher grade (61% of T3 tumors were of grade III). There were no associations regarding follow-up variables.
Discussion
To our knowledge, this is the first study of CSCMTs. The retrospective design is justified by the low prevalence of these neoplasms, 20 and due to its retrospective nature, some of the information regarding clinical and follow-up data could not be retrieved. To collect a representative number of cases, it was necessary to gather cases from 3 different institutions. We also decided to include only tumors with a predominance of spindle cells (excluding tumors with recognizable patterns as mixed tumors or osteosarcomas and excluding carcinomas and malignant myoepitheliomas with evident epithelial proliferation), since they are the ones that represent a diagnostic and clinical challenge.
The human histological classification includes many types of mesenchymal tumors that are located in the breast. 18 Any neoplasm appearing in the mammary region, regardless of the tissue of origin, is considered a breast tumor. In the present study, only neoplasms of proven mammary tissue origin were considered. After excluding cases with no mammary tissue origin and those that lacked sufficient tissue for additional investigations including immunohistochemistry, 67 tumors from 67 female dogs were included, obtaining a large, reliable, and representative series of malignant spindle cells tumors of the mammary gland. In addition, 3 spindle cell mammary neoplasms were benign (2 benign myoepitheliomas and 1 mammary angioma) and were not further considered in this study.
Once the corresponding IHC was performed on malignant CSCMT cases, malignant myoepithelioma and carcinoma and malignant myoepithelioma (with minimal epithelial proliferation) were the most frequent histotypes encountered. Surprisingly, spindle cell carcinoma and squamous carcinoma–spindle type were not represented in this series of cases. In our opinion, since these histotypes were not identified after recruiting cases of 3 institutions in a period of 16 years, their existence is anecdotical and questionable. It is possible that some of these cases could be diagnosed as carcinomas (spindle cell, squamous spindle cell) only on a morphological basis. In fact, the initial diagnosis of spindle cell carcinomas was changed to malignant myoepithelioma and carcinoma and malignant myoepithelioma based on the IHC findings.
Another surprising finding was the low prevalence of mammary fibrosarcomas (n = 1) in this case series. It is interesting to note that 11 tumors were originally diagnosed as fibrosarcomas of mammary origin based on morphology. Following IHC, the number decreased to only 1 case. This finding contrasts with current literature 5 that defines fibrosarcomas as the most prevalent type of primary mammary sarcoma, accounting for up to 16.7% of malignant CMTs and having also the worst prognosis. 8 The only fibrosarcoma of this study (prevalence 1%) was graded as sarcoma grade III and had neither lymph node involvement nor distant metastasis at presentation. Unfortunately, no follow-up information could be retrieved at 2 years after surgery to verify these findings. On the contrary, a surprisingly high frequency of mammary hemangiosarcomas (5/67, 7%) was observed.
According to morphology, 11 myoepithelial neoplasms were first identified but, as they contained extensive whorls characteristic of PNST/PWT, they were further classified as compatible with PNST/PWT based on histomorphology (HE). After IHC, these cases were rediagnosed as malignant myoepithelioma (n = 5), carcinoma and malignant myoepithelioma (n = 2), PNST (n = 2), and undifferentiated sarcoma (n = 2). PNSTs can occur not only in the peripheral nervous system but also in soft tissue and various organs. 17 To our knowledge, these 2 cases are the first description of canine mammary PNSTs. PNST cases were malignant, although grading systems varied in their categorization: they were graded as I, III (sarcoma grade), and II (mammary grade). The histogenesis of PNSTs is still not completely clarified, and the nomenclature remains confusing. 19 Cutaneous PNSTs belong to a very heterogeneous group of neoplasms that arise from neural crest–derived cells like Schwann cells, perineural cells, or fibroblasts. They can exhibit a benign or malignant biological behavior. 19 The diagnosis of PNST is primarily based on the combination of histological and immunohistochemical features, since there is not an unequivocally accepted immunohistochemical panel to support the PNST diagnosis. 19 The histological features of PNSTs may resemble PWTs; therefore, distinguishing PNSTs arising in non-PNS tissue from PWTs is considered difficult. Furthermore, in companion animal practice, PNSTs and PWTs have been treated as soft tissue sarcomas (STSs) together with other spindle cell tumors that arise in the soft tissue such as fibrosarcoma, rhabdomyosarcoma, leiomyosarcoma, myxosarcoma, and malignant mesenchymoma. 17 In our cases, the positive reaction to GFAP was considered diagnostic for PNST.
Although a panel of PNST/PWT markers (myosin, actin, desmin, S-100, GFAP) was applied in cases suspicious for PNST/PWT on HE-stained sections, 1,2 no PWTs could be identified. Nevertheless, in the authors’ opinion, mammary PWT remains possible considering the compatibility of the histologic features with PWT.
In total, the diagnosis based on the HE-stained section differed from the diagnosis after IHC in 50 of 67 (75%) of the malignant cases. Therefore, similar to previous suggestions, 11 IHC is essential for the proper diagnosis of CSCMTs. Nevertheless, according to the present study, Masson’s trichrome stain could help to discriminate between fibrosarcomas (of any origin) and malignant myoepithelioma (P = .007), representing an inexpensive alternative to IHC in routine diagnosis of these cases.
The most frequent tumors diagnosed in the present study were myoepithelial cell tumors (malignant myoepithelioma and carcinoma and malignant myoepithelioma). Moreover, many of the other tumor types not identified by IHC as myoepithelial proliferations contained spindle cells with myoepithelial morphology, even if these myoepithelial cells did not express the expected IHC markers of myoepithelial cells. As it has been extensively published, canine mammary tumors may have a more prominent myoepithelial proliferation compared to other species such as humans and cats. 7,14,18 Myoepithelial cell proliferation, in general, is associated with a better prognosis in mammary tumors. 10,14,22 Some authors postulate that myoepithelial cells may have a dual function as both tumor suppressors and promoters. 14 According to this theory, myoepithelial cells behave differently depending on their degree of differentiation. Differentiated myoepithelial cells would be associated with a better prognosis, while in malignant myoepithelioma, undifferentiated myoepithelial cells would be associated with a worse prognosis, promoting tumor progression. In our study, only 2 malignant myoepitheliomas had a malignant behavior.
Several markers have been proposed for canine myoepithelial cells, among them, basal cell markers such as CK14, smooth muscle–specific proteins such as α–smooth muscle actin (SMA) and calponin, and protein p63. 3,6,11,14,22 These markers are gradually lost in the nonbasal types of myoepithelial cells. 3,11 For this reason, the use of 2 or more markers is recommended to ensure precise identification of myoepithelial cells. 11 In our series of cases, it was rare for myoepithelial tumors to be positive for both p63 and CK14. Malignant myoepitheliomas more frequently expressed only 1 of these markers (P = .008), indicating less differentiated myoepithelial cells. Moreover, the differences found in the expression of myoepithelial markers in malignant myoepithelioma compared to carcinoma and malignant myoepithelioma seem to indicate that they are truly different histological types, even when epithelial cell proliferation is minimal. Consequently, and considering also that they might differ in their malignant behavior, the identification of carcinoma and malignant myoepithelioma with minimal epithelial proliferation by IHC is strongly recommended.
In the mammary gland, a basal/myoepithelial cell has been considered to have stem cell properties, 14 and the plasticity of canine spindle mammary cells in vitro and in vivo has been demonstrated: spindle cell tumors gave rise to spindle cell tumors in nude mice, and several contained bone tissue, a sign of plasticity. 21 In diagnostic procedures, the naming of tumors is generally based on their pattern of differentiation (based on the evidence provided by morphology and antigen expression), and the cell of origin usually is speculative and inferred in routine caseloads. As it has been stated previously, most of the tumors of this study had myoepithelial proliferations observed in HE-stained sections and not identified by expressed myoepithelial markers on IHC. Some of the tumors of the study (hemangiosarcoma, PNST, and undifferentiated sarcoma) are considered sarcomas, and the cell of origin could be located in the mammary stroma. Nevertheless, considering the plasticity of myoepithelial cells and their potential as stem cells, myoepithelial cell marker expression can be lost as myoepithelial cells undergo dedifferentiation, and the mentioned histological features, including the hypothesis of a myoepithelial origin of these mammary sarcomas, could still be valid. Mechanistic studies using myoepithelial cells in culture are needed to demonstrate or refute this hypothesis, although it is worthy to mention that all hemangiosarcomas of the study were positive for calponin, a myoepithelial and smooth muscle marker. Another marker, α–smooth muscle actin, has been found to be positive in cutaneous canine hemangiosarcoma. 13
Clinically, the malignant CSCMTs were in general large, single, minimally or nonaggressive tumors that rarely affected regional lymph nodes or developed distant metastases. Histological grading systems are used to predict the biological behavior of tumors. 10 Here, 2 different grading systems were used to further characterize the tumors: a system for canine soft tissue sarcoma 4 and a system for canine mammary carcinoma. 10 In this study, the application of the grading system was based on evaluation of HE-stained sections prior to IHC. The pathologists used the soft tissue sarcoma grading system in all cases, while the mammary tumor grading system was applied only in tumors compatible with malignant myoepithelioma and carcinoma and malignant myoepithelioma. Neither system could be fully validated in malignant CSCMTs in terms of prognosis, since tumors of grades II and III of both systems developed recurrence and/or metastasis in the follow-up period. It would have been desirable that only grade III tumors had this aggressive behavior. In contrast, the soft tissue sarcoma grade could be applied to all tumors. However, the mammary tumor grading system better represented the general low malignancy of the cases as most of them were grade II. Notably, tumors that had lymph node involvement or distant metastases at presentation were of grade III by both systems. We recommend that if IHC is performed and a definitive diagnosis is obtained, mammary sarcomas should be graded using the soft tissue sarcoma grading, and only the spindle cell mammary carcinomas (malignant myoepithelioma and carcinoma and malignant myoepithelioma) should be graded using the mammary tumor grading scheme. The application of grading criteria to tumor types for which the grading system has not been validated might be inappropriate. The fact that most of the female dogs were intact, some malignant CSCMTs were synchronous with other mammary tumors (29/67, 43%), and the statistical associations found with the other mammary tumors in terms of growth (size) and benign/malignancy could indicate that some hormonal influence might be present. In a previous report, the presence of progesterone receptors but not estrogen receptors has been described in cell lines derived from canine mammary spindle cell tumors. 9 Further studies on the hormone dependency of malignant CSCMTs would be desirable.
In conclusion, malignant CSCMTs are rare mammary tumors with a generally benign behavior. These tumors must be evaluated by IHC to determine the histotype and to indicate the relatively benign clinical behavior. It is recommended to histologically ensure the mammary origin of the neoplasms and to consider tumor size and the presence or absence of other mammary tumors as important clinical features in relation with the histological malignancy. Finally, Masson’s trichrome stain should be used routinely, prior to IHC, to help the definitive diagnosis. Further studies are necessary on the possible stem cell properties of myoepithelial cells and their diagnostic and prognostic significance.
Footnotes
Acknowledgements
We thank Pedro Aranda for his technical assistance and LIVET (Laboratory of Veterinary Immunohistochemistry, UCM) for the financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
