Abstract
Canine mammary tumors (CMTs) are morphologically and biologically heterogeneous, prompting several attempts to classify such tumors on the basis of their histopathological characteristics. Recently, molecular-based analysis methods borrowed from human breast cancer research have also been applied to the classification of CMTs. In this study, canine mammary neoplasms (n = 648) occurring in Korea from 2008 to 2011 were analyzed according to the histological classification and grading system proposed by Goldschmidt et al. Furthermore, randomly selected mammary carcinomas (n = 159) were classified according to the molecular subtype using immunohistochemical characteristics. Canine mammary neoplasia accounted for 52.6% (648/1250) of the tumors in female dogs, and 51.7% (340/648) of these were malignant. All of the carcinoma-anaplastic subtypes were grade III tumors (5/5, 100%), while most of the carcinoma-tubular subtypes (15/18, 83.3%) and carcinoma arising in a complex adenoma/mixed-tumor subtype (115/135, 85.2%) were grade I tumors. Tumor cell invasion into lymphatic vessels was most common in the comedocarcinoma, carcinoma-anaplastic, and inflammatory carcinoma subtypes. The most frequently occurring molecular subtype (70/159, 44%) was luminal A. However, the basal-like subtype was the most malignant and was frequently associated with grade III tumors and lymphatic invasion. The carcinoma-solid subtypes were also often of the basal-like subtype. Reclassification of CMTs using the newly proposed histopathological classification system and molecular subtyping could aid in determining the prognosis and the most suitable anticancer treatment for each case.
Mammary glands originate from apocrine sweat glands, and canine mammary gland tumors (CMTs) are the most common neoplasm in female dogs, with approximately 50% being described as malignant.18,25
CMTs were previously classified by histological type according to the published criteria of the World Health Organization (WHO).9,17 However, numerous factors, such as hormones, receptors, diet, obesity, irradiation, and genetic components, are involved in CMT progression. 16 Variations in these factors render CMTs both morphologically and biologically heterogeneous. Therefore, Goldschmidt et al 7 have proposed a new histological classification and grading system based on modifications to the WHO criteria.
Recently, gene expression profiling has revealed that the expression levels of certain hormonal receptors (estrogen receptor [ER] and progesterone receptor [PR]), human epidermal growth factor receptor 2 (HER-2), and basal markers correlate with tumor prognosis and clinical outcome. These differences have allowed the subdivision of tumors into various molecular subtypes in human breast cancer (HBC) studies.21,26 The HBCs have been categorized into the following 5 molecular subtypes: luminal A (ER positive and HER-2 negative), luminal B (ER and HER-2 positive), HER-2 overexpressing (ER negative and HER-2 positive), basal-like (ER and HER-2 negative and positive for any basal marker), and normal-like (all negative). 27 Although guidelines for the treatment of HBCs are based on the molecular subtype, application of gene expression profiling is not easy for routine diagnosis. Therefore, immunohistochemistry (IHC) panels are used as a substitute for gene expression panels. 19 Several veterinary studies have also attempted to classify malignant mammary tumors by molecular subtype and have confirmed that their expression patterns are similar to those of HBCs.5,24
Although CMTs have been well studied in Europe and the United States, the incidence and histopathological and molecular characteristics of CMTs in Asia remain unclear. In this study, we analyzed cases of CMT recorded in Korea between 2008 and 2011 and evaluated the histological type and grade according to the new histological classification and grading criteria. We also assessed the molecular subtype using IHC surrogate panels.
Materials and Methods
Samples
Canine mammary tumor samples taken between 2008 and 2011 were obtained from the histopathological database of the Department of Veterinary Pathology, Konkuk University Animal Teaching Hospital, Seoul, Korea. All tissues were fixed in 10% neutral-buffered formalin, processed routinely, and embedded in paraffin wax. Sections (4 μm thick) were stained with hematoxylin and eosin (HE).
Clinical Features
The mean age of the dogs was calculated, and the locations of the masses with respect to the lateral (left or right) and longitudinal (thoracic, abdominal, or inguinal mammary gland) axes were recorded. Furthermore, the frequencies of mammary tumors and hyperplasia/dysplasia were analyzed according to breed. In addition, the patient’s reproductive status (intact or spayed) at the time of mastectomy was assessed.
Histopathology
The samples were divided into hyperplasia/dysplasia, benign tumors, and malignant tumors according to the criteria of Goldschmidt et al. 7 Histological classification was also performed on the basis of the criteria proposed by Goldschmidt et al 7 by means of HE staining and IHC analysis. If more than 1 mass was present in a particular case with a female dog, only the mass with the greatest tumor progression and highest malignancy was selected for inclusion in this study. 15 Tumors were classified as grade I, II, or III based on evaluation of the formation of tubular structures, nuclear pleomorphism, and mitotic indices. 4 Microscopic evidence of lymphatic invasion and necrosis was also examined.
Immunohistochemistry
Tissue specimens randomly selected among the canine mammary carcinomas (n = 159) were immunostained to determine their molecular subtypes.11,12 To confirm the histological types of the tumors, immunolabeling to p63, smooth muscle actin (SMA), and vimentin was performed only in those tumors originating from myoepithelial cells.6,11
Immunohistochemical analysis was performed on formalin-fixed tissue sections (Table 1). In brief, the sections were dewaxed in xylene, hydrated through a graded series of ethanol solutions, and washed 3 times in 0.1 M phosphate-buffered saline (PBS; pH 7.4). Endogenous peroxidase activity was blocked by incubating the sections in 3% hydrogen peroxide in PBS at room temperature (RT) for 20 minutes and then washing them 3 times in PBS. Heat-induced epitope retrieval (Tris-EDTA, pH 9.0; citric acid, pH 6.0) was performed using a microwave oven at high power (650 W) for all primary antibodies except the anti–HER-2 antibody. After pretreatment, the serial sections were washed 3 times in PBS, overlaid with the primary antibody, and incubated. Isotype-matched immunoglobulins of irrelevant specificity (Table 1) were used for negative control staining. Normal mammary glands and normal to hyperplastic lesions adjacent to tumors were used as positive controls for all antigens except HER-2. Canine mammary carcinomas with HER-2 overexpression were used as positive controls for HER-2 by referring to our previous study. 11 The EnVision rabbit/mouse reagent conjugated to peroxidase (DakoCytomation, Glostrup, Denmark) was applied for 20 minutes at RT to visualize the immunolabeling. The slides were subsequently washed 4 times in PBS and developed with 3,3′-diaminobenzidine (DAB) as chromogen. The sections were then counterstained with Gill’s hematoxylin, dehydrated, and mounted.
Primary Antibodies and Immunostaining Protocols Used in This Study.
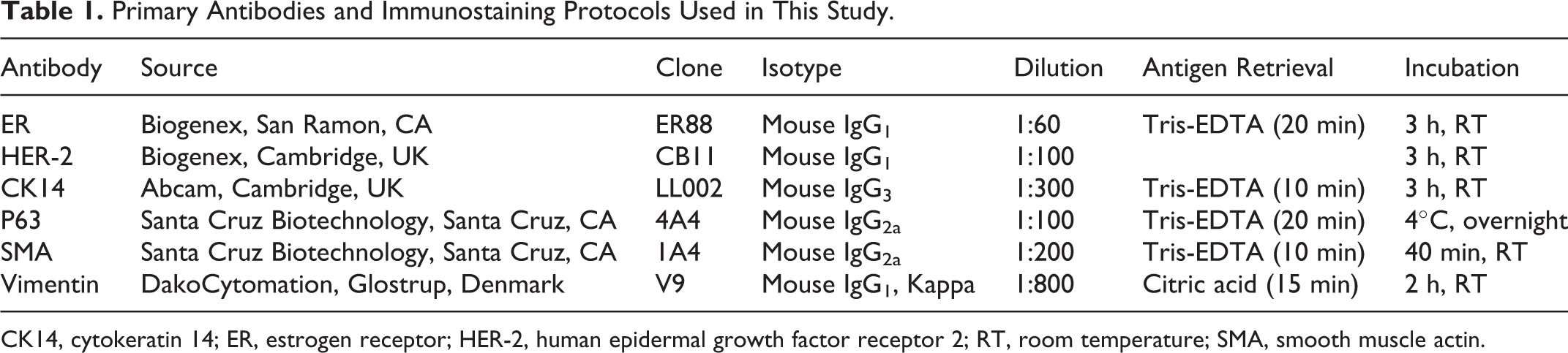
CK14, cytokeratin 14; ER, estrogen receptor; HER-2, human epidermal growth factor receptor 2; RT, room temperature; SMA, smooth muscle actin.
Immunohistochenistry Scoring
The IHC results were scored as follows. Nuclear ER immunoreactivity in more than 10% of the total number of tumor cells was considered a positive signal. HER-2 immunolabeling was measured according to the HercepTest scoring system (DakoCytomation), and weak to strong complete membrane staining of more than 10% of tumor cells was defined as overexpression of HER-2. Staining of more than 5% of tumor cells for cytoplasmic cytokeratin 14 (CK14) and nuclear p63 expression was considered a positive signal. Expression of SMA and vimentin in the cytoplasm of tumor cells originating from myoepithelial cells was confirmed.
The molecular subtype was classified as luminal A (ER positive/HER-2 negative), luminal B (ER positive/HER-2 positive), HER-2 overexpressing (ER negative/HER-2 positive), basal-like (ER/HER-2 negative and CK14 or p63 positive), or normal-like (all negative). 5
Statistical Analyses
All statistical analyses were performed using the Statistical Package for Social Sciences (SPSS, Inc, an IBM Company, Chicago, IL) software program for Windows, version 17. Continuous variables were expressed as the mean ± SD and categorical data as frequencies and percentages. Pearson’s χ2 test, Fisher’s exact test, and analysis of variance (ANOVA) were used to compare the groups. P < .05 was considered to indicate statistical significance.
Results
Samples
The total number of canine cases submitted to the Department of Veterinary Pathology, Konkuk University Animal Teaching Hospital, Seoul, Korea, from 2008 through 2011 was 2072 (822 male dogs and 1250 female dogs).
Tissues from the mammary glands of female dogs were obtained in 658 cases, representing 31.8% (658/2072) of all canine cases and 52.6% (658/1250) of cases in female dogs. Of these, 29 (4.4%) showed hyperplasia/dysplasia, 289 (43.9%) were benign tumors, and 340 (51.7%) were malignant tumors.
Clinical Features
Table 2 summarizes the clinical characteristics of the cases used in this study. The mean ± SD age of all affected dogs was 9.1 ± 2.7 years (range, 1.6–19 years; the age was unknown in 65 cases). The mean ± SD ages of dogs with hyperplasia/dysplasia, benign tumors, and malignant tumors were 9.0 ± 2.5 years (range, 3–16 years), 8.6 ± 2.6 years (range, 1.6–16 years), and 9.6 ± 2.8 years (range, 3–19 years), respectively. Dogs with malignant tumors were significantly older than dogs with benign tumors (ANOVA test, P = .00). A total of 24 breeds were represented (n = 583; the breed was unknown in 75 cases; Fig. 1), but most cases (n = 525) occurred in 6 breeds: Shih Tzu, Yorkshire Terrier, Maltese, Poodle, mixed breed, and Cocker Spaniel. Maltese dogs had more benign (83/160, 51.9%) than malignant (72/160, 45%) tumors. In contrast, Cocker Spaniels and mixed-breed dogs had more malignant (39/61, 63.9% and 35/57, 61.4%, respectively) than benign (19/61, 31.1% and 21/57, 36.8%, respectively; Fisher’s exact test, P = .017) tumors. The mean ± SD age of mixed-breed dogs (10 ± 3.1 years) was higher than that of Maltese dogs (8.8 ± 2.7 years) or Cocker Spaniels (8.1 ± 1.8 years; ANOVA test, P = .001). Although Cocker Spaniels had more malignant tumors than benign tumors, the mean age of Cocker Spaniels was similar to that of Maltese dogs, which had more benign tumors than malignant tumors.
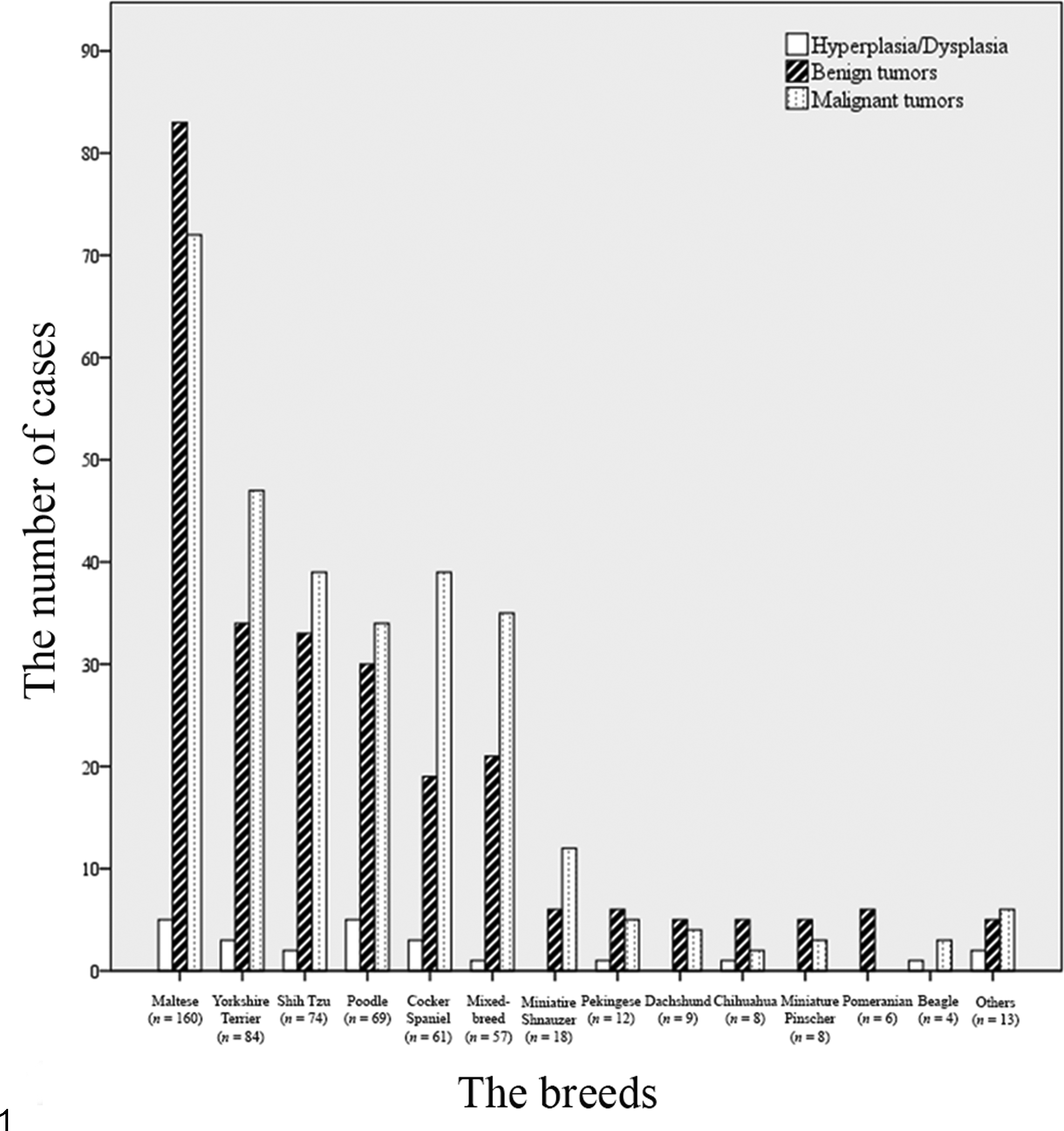
Analysis of canine mammary hyperplasia/dysplasia and mammary neoplasms according to the patient’s breed. Others: Great Pyrenees (n = 1), Spitz (n = 1), French Bulldog (n = 1), Lhasa Apso (n = 2), Pug (n = 1), German Shepherd (n = 2), Rottweiler (n = 1), Doberman Pinscher (n = 1), Jindo (n = 1), Old English Sheepdog (n = 1), and Golden Retriever (n = 1).
Clinical Characteristics of and Necrosis in Canine Mammary Gland Tissues.
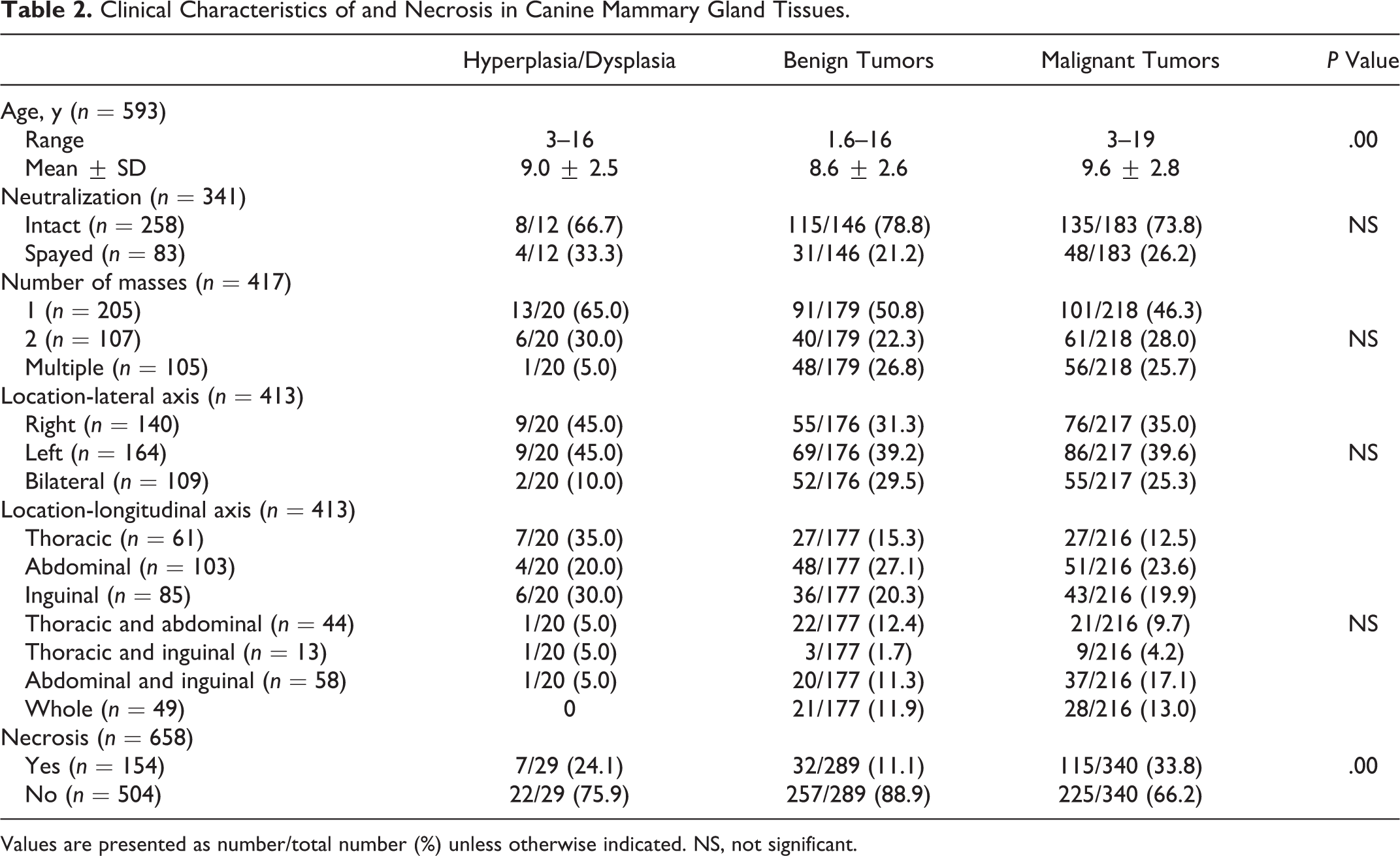
Values are presented as number/total number (%) unless otherwise indicated. NS, not significant.
The reproductive status (spayed or intact) at the time of surgery was established in only 341 of the 658 dogs. Most dogs were intact (n = 258 intact and n = 83 spayed), but no significant difference in the proportions of mammary tumors and hyperplasia/dysplasia was observed between these 2 subgroups. In addition, more than 1 mass was present in approximately 50% of cases, and the proportions of cases showing single (205/417, 49.2%) or plural (212/417, 50.8%) masses were not significantly different.
Unilateral masses (304/413, 73.6%) were more common than bilateral masses (109/413, 26.4%), but the numbers of masses on the right (140/413, 33.9%) and left (164/413, 39.7%) sides were not significantly different. The frequency of masses was highest in the abdominal (third and fourth) mammary glands (103/413, 24.9%), but no significant difference was observed with respect to the longitudinal axis.
Histopathological Findings: Necrosis, Histological Classification, Grade, and Lymphatic Invasion
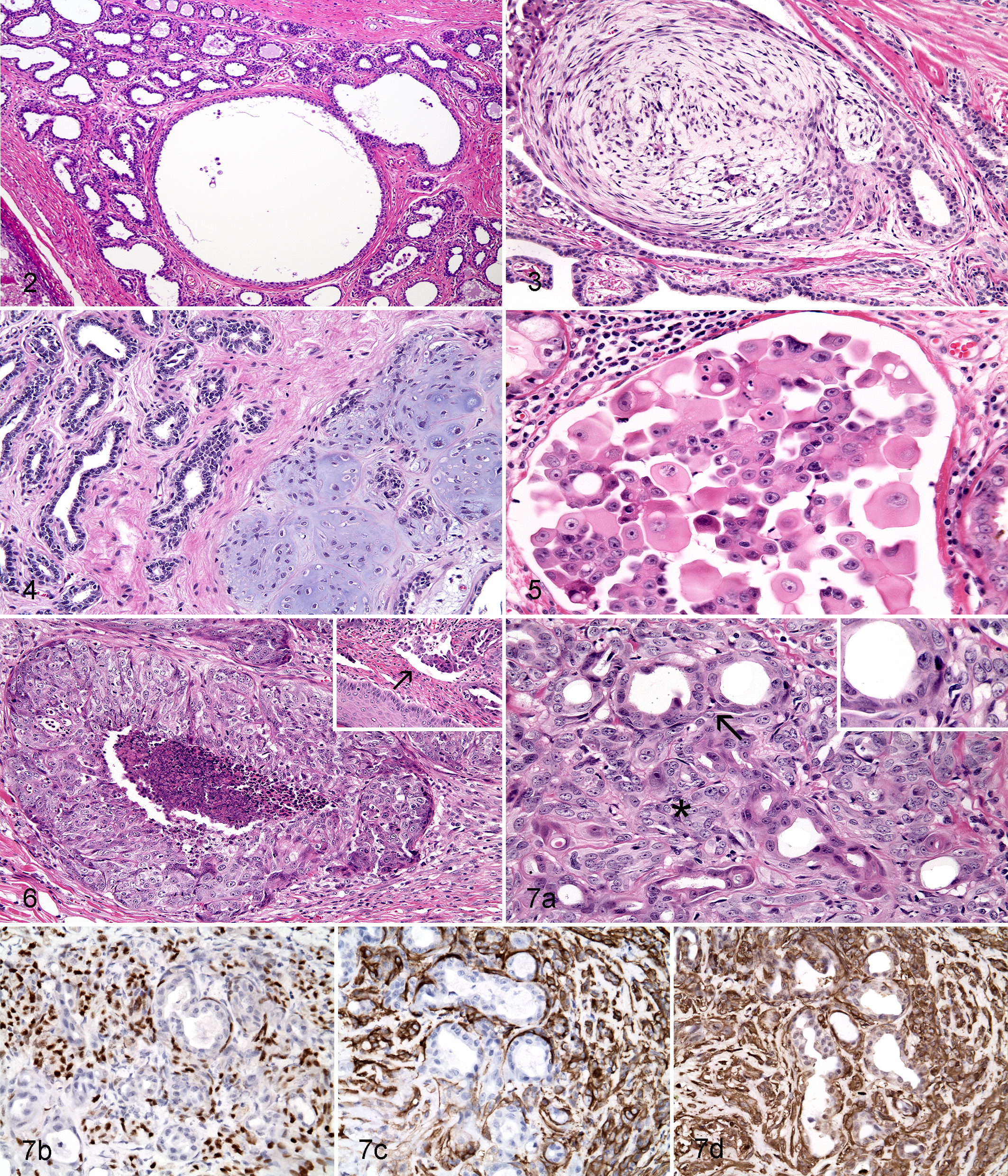
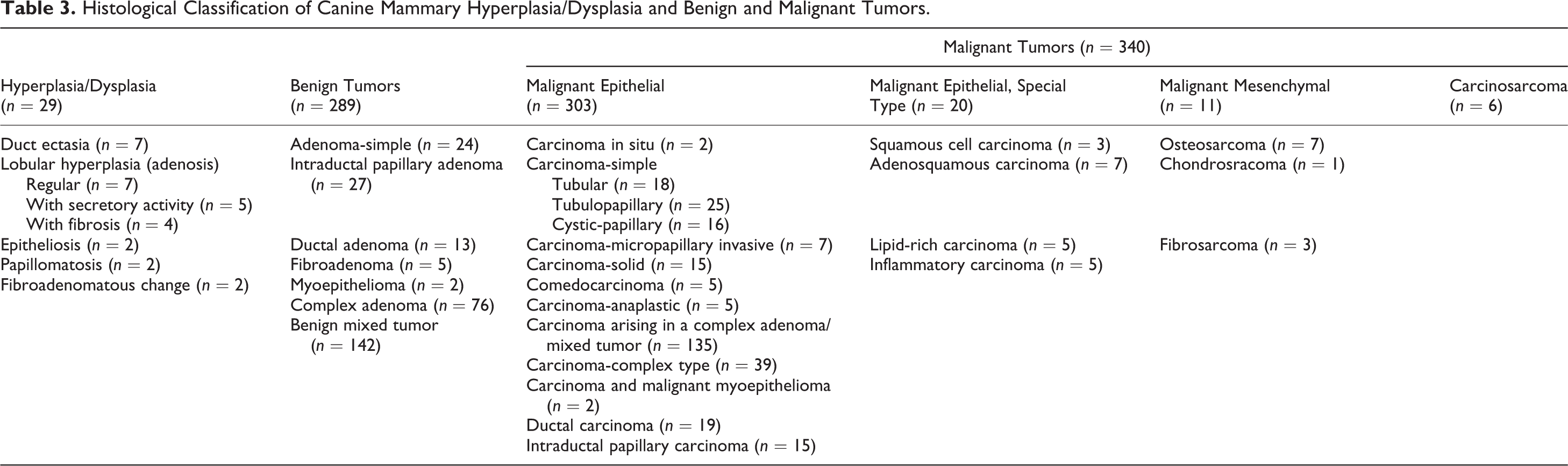
Necrosis was present in 154 of the 658 cases (hyperplasia/dysplasia, 7/29, 24.1%; benign tumors, 32/289, 11.1%; malignant tumors, 115/340, 33.8%; Table 2). Necrosis was detected in more malignant tumors than benign tumors (Pearson’s χ2 test, P = .00). Most of the infiltrating inflammatory cells were lymphocytes, but mast cells, plasma cells, and macrophages were also identified. Table 3 summarizes the histological classification of the mammary gland cases. Representative histological types are shown in Fig. 2 (hyperplasia/dysplasia), Figs. 3 and 4 (benign tumors), and Figs. 5, 6, and 7 (malignant tumors). From the IHC results for p63, SMA, and vimentin, myoepithelioma was identified in 2 cases of a total of 289 benign tumors. In addition, carcinoma and malignant myoepithelioma were identified in 2 cases of a total of 340 malignant tumors (Fig. 7). Table 4 summarizes the histological grade and presence of lymphatic invasion of the malignant mammary tumors (n = 340). Tumors were classified as grade I (n = 222, 65.3%), grade II (n = 74, 21.8%), or grade III (n = 44, 12.9%). Lymphatic invasion was identified microscopically in 40 cases (11.8%).
Histological Classification of Canine Mammary Hyperplasia/Dysplasia and Benign and Malignant Tumors.
Histological Grade, Lymphatic Invasion, and Molecular Subtype of Canine Malignant Mammary Tumors.

Basal-like, ER negative/HER-2 negative and CK14 or p63 positive; HER-2 overexpressing, ER negative/HER-2 positive; luminal A, ER positive/HER-2 negative; luminal B, ER positive/HER-2 positive; normal-like, all negative.
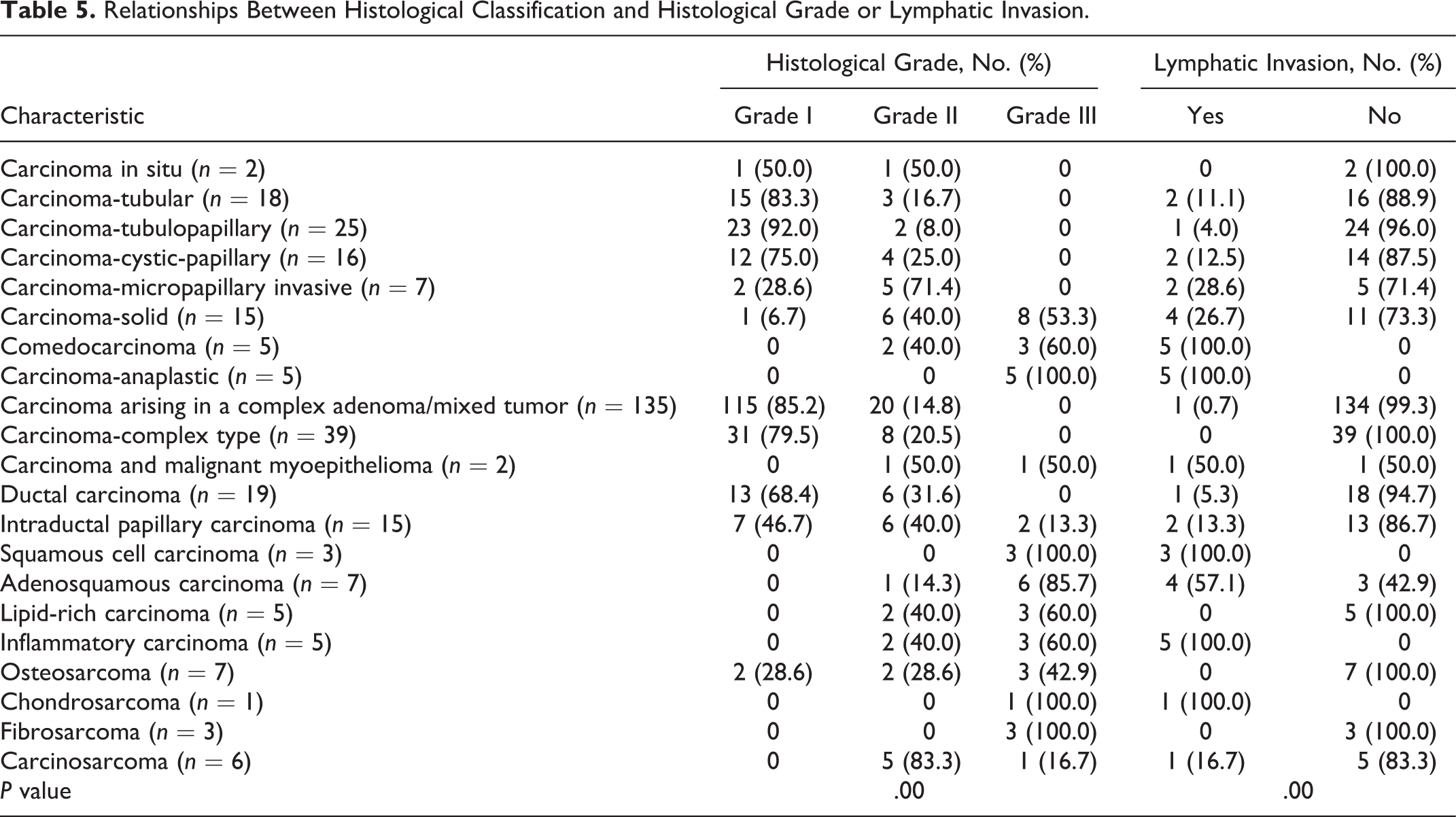
Carcinoma-anaplastic (5/5, 100%), adenosquamous carcinoma (6/7, 85.7%), and some sarcomas (7/11, 63.6%) were classified as grade III tumors, while carcinoma-tubular (15/18, 83.3%), carcinoma-tubulopapillary (23/25, 92%), and carcinoma arising in a complex adenoma/mixed tumor (115/135, 85.2%) were mostly classified as grade I tumors (Fisher’s exact test, P = .00; Table 5). Lymphatic invasion was highly associated with comedocarcinoma, carcinoma-anaplastic, and inflammatory carcinoma (Pearson’s χ2 test, P = .00; Table 5).
Relationships Between Histological Classification and Histological Grade or Lymphatic Invasion.
Molecular Subtypes
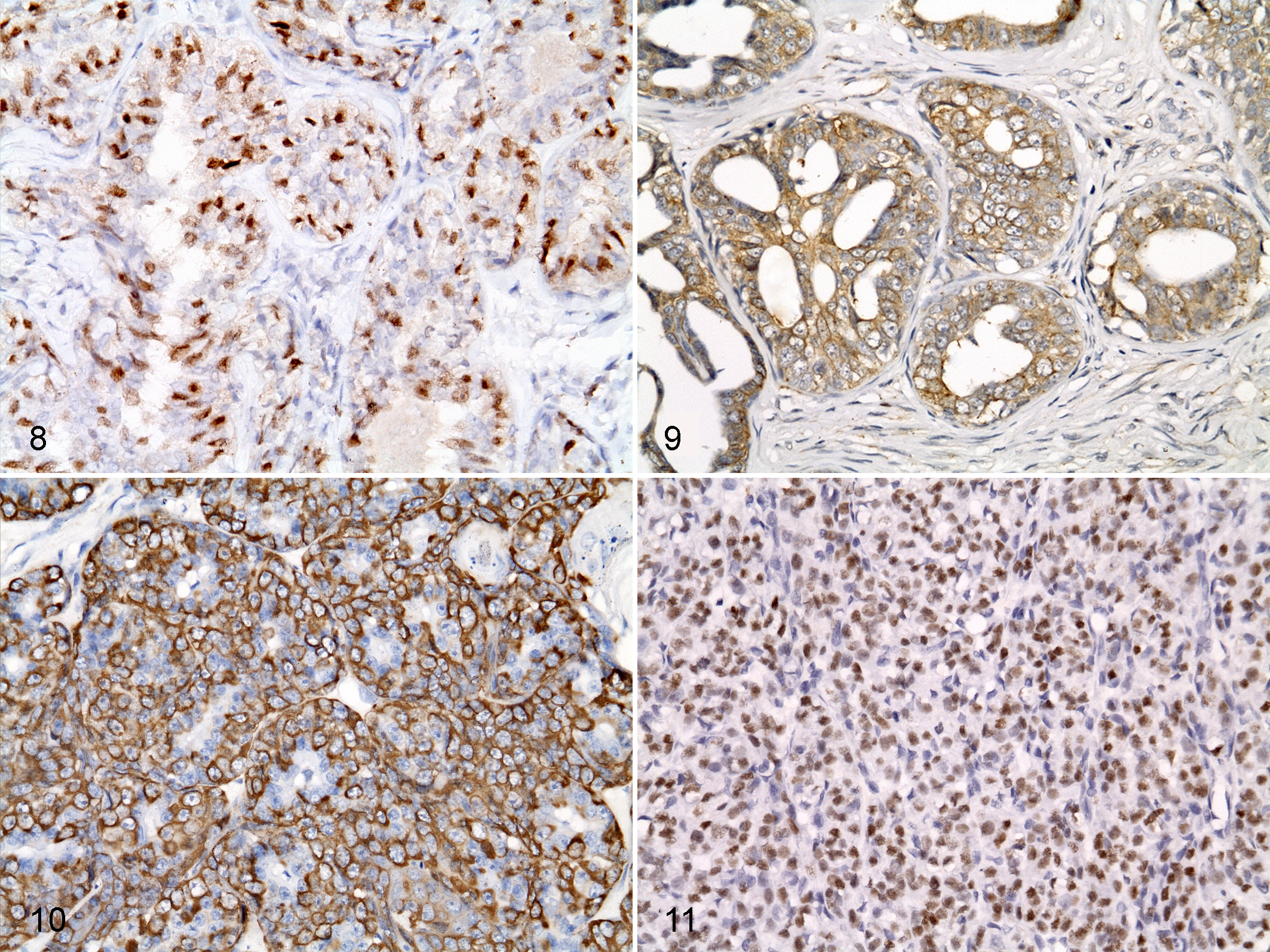
Of the 340 mammary carcinomas, 159 were randomly selected for molecular subtyping (Table 4). Of these, 106 cases (66.7%) of canine mammary carcinomas were ER positive, whereas 45 cases (28.3%) overexpressed HER-2. Mammary carcinomas were categorized as luminal A (n = 70), luminal B (n = 36), HER-2 overexpressing (n = 9), basal-like (n = 39), or normal-like (n = 5) according to the IHC results (Figs. 8–11). Luminal A (ER positive and HER-2 negative) was the most frequent subtype (70/159, 44%), and normal-like was the least frequent subtype (5/159, 3.1%).
Associations Between Molecular Subtype and Histological Classification, Histological Grade, and Lymphatic Invasion
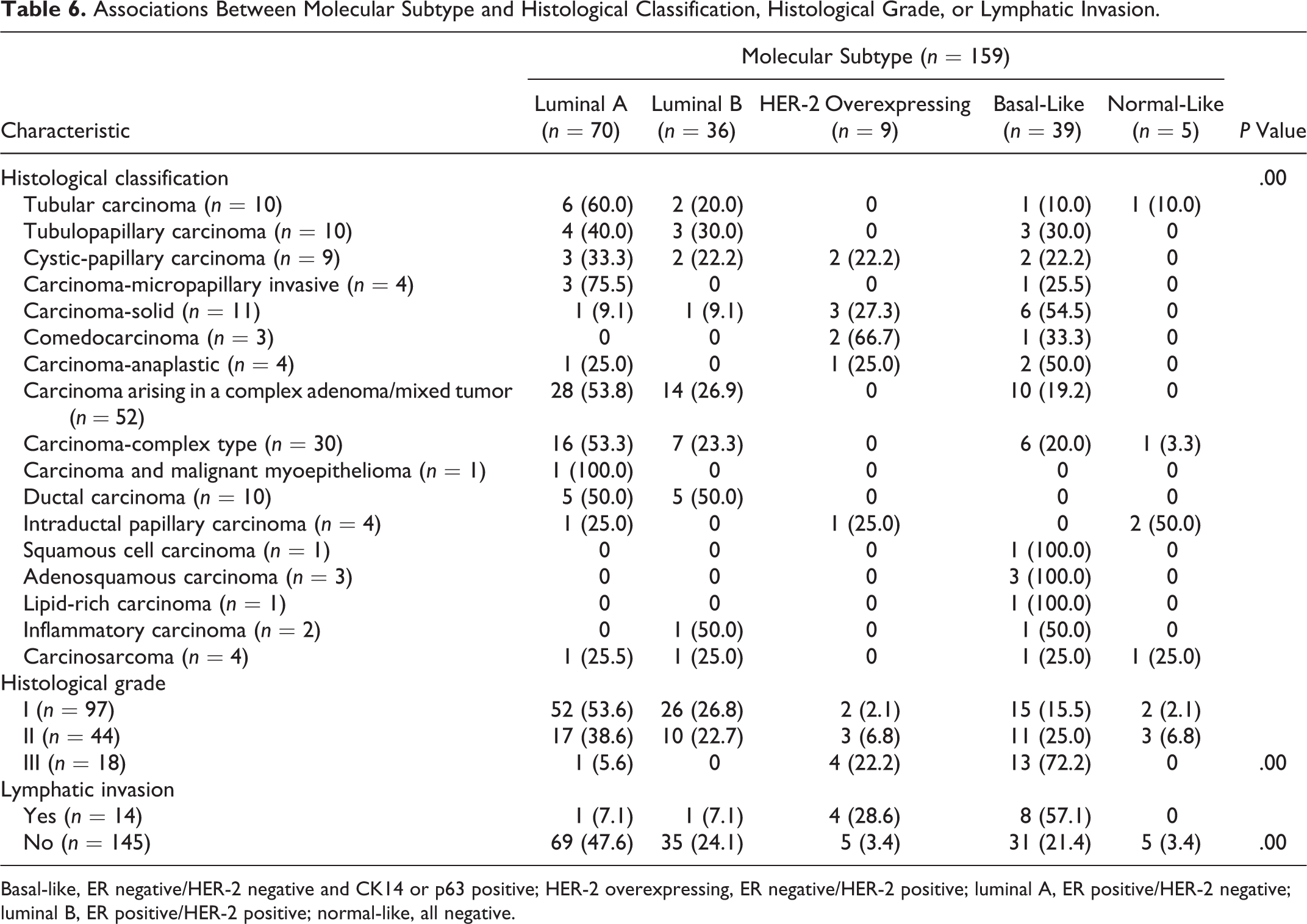
Table 6 summarizes the associations between molecular subtype and other variables in cases of CMT. Histological classification was significantly associated with molecular subtype (Fisher’s exact test, P = .00). Carcinoma-tubular (6/10, 60.0%) and carcinoma arising in a complex adenoma/mixed tumor (28/52, 53.8%) were frequently categorized as luminal A, whereas carcinoma-solid (6/11, 54.5%) was frequently categorized as basal-like.
Associations Between Molecular Subtype and Histological Classification, Histological Grade, or Lymphatic Invasion.
Basal-like, ER negative/HER-2 negative and CK14 or p63 positive; HER-2 overexpressing, ER negative/HER-2 positive; luminal A, ER positive/HER-2 negative; luminal B, ER positive/HER-2 positive; normal-like, all negative.
Histological grade was also significantly associated with molecular subtype (Fisher’s exact test, P = .00). Luminal A was the most common subtype of grade I (52/97, 53.6%) and II (17/44, 38.6%) tumors, while basal-like was the most common subtype of grade III tumors (13/18, 72.2%). Eight (57.1%) of all cases of lymphatic invasion (n = 14) occurred in basal-like tumors, while 69 (47.6%) of all cases of nonlymphatic invasion (n = 145) occurred in luminal A tumors (Fisher’s exact test, P = .00).
Discussion
This study analyzed canine mammary gland tissue from 658 samples that were taken between 2008 and 2011 from submissions to the Department of Veterinary Pathology, Konkuk University Animal Teaching Hospital, Seoul, Korea. Canine mammary hyperplasia/dysplasia was present in 27 cases, while CMTs were seen in 629 cases; of these, 54% were diagnosed as malignant tumors. The mean ages of dogs with benign and malignant tumors were similar to those reported in a previous study. 25 Malignant tumors were associated with an older mean age (9.6 ± 2.8 years), which supports previous findings that older dogs are more likely to have malignant tumors than younger dogs.25,28 Interestingly, the Cocker Spaniels with mammary masses were a similar age to the Maltese dogs in this study, although Cocker Spaniels had a higher proportion of malignant tumors and Maltese dogs had a higher proportion of benign tumors. On the other hand, in mixed-breed dogs, older age was associated with a high proportion of malignant tumors. It seems that the relationship between age and the benign/malignant ratio differs according to breed; this is likely to be a result of hereditary differences in genes such as BRCA1/2.11,23 It is known that early spaying can decrease the risk of mammary tumor development when compared with late spaying and no spaying. 8 In this study, there were no significant differences in the proportions of mammary tumors and hyperplasia/dysplasia between dogs that had been spayed or were intact at the time of mastectomy. This may reflect the fact that age at the time of spaying was not investigated in this study.
Recently, Goldschmidt et al 7 proposed modifications to the WHO classification criteria to create a more morphologically diverse and detailed classification system for CMTs. This system describes and proposes a nomenclature for tumors and hyperplasia/dysplasia. Some neoplasms (eg, carcinoma-solid and carcinoma-anaplastic) retain the same names, while others (eg, carcinoma-micropapillary invasive and ductal carcinoma) are separated from their previously assigned types. In addition, this classification adds new types such as inflammatory carcinoma, which reflects clinical signs associated with inflammatory reactions such as erythema, heat, and pain (Fig. 6). Results from IHC analysis can also be incorporated into this system to differentiate cells of myoepithelial origin (Fig. 7). As p63, SMA, and vimentin are simultaneously expressed in myoepithelial cells, but not in epithelial cells, primary antibodies against these 3 proteins are used to distinguish cells of these types. Among the mammary tumors investigated in this study, benign/malignant myoepithelioma occurred infrequently. However, tumors that are derived from myoepithelial cells should be identified by IHC for p63, SMA, and vimentin to allow detailed classification of mammary tumors.
We used this new histological classification and grading system for analysis of the CMTs in this study. The dilation of ducts and growth of lobules were most often observed in mammary hyperplasia/dysplasia. Benign mammary tumors predominantly comprised complex adenomas (epithelial and myoepithelial cell proliferation) and benign mixed tumors (epithelial and myoepithelial cell proliferation with cartilage and/or bone metaplasia), whereas malignant mammary tumors were most often seen occurring as part of complex adenomas/mixed tumors. Other studies have also reported that complex adenomas and benign mixed tumors were the most common benign mammary tumors, along with adenoma-simple. Similarly, carcinoma-simple was the most prevalent, and carcinoma arising in a complex adenoma/mixed tumor was reported to be common in malignant tumors.1,9,16 In the current study, histological classification was significantly associated with both the grade and the presence of lymphatic invasion. The highest grade and frequency of infiltration of tumor cells into lymphatic vessels were observed for carcinoma-anaplastic, which is consistent with findings from a previous study. 22
In HBCs, it is well known that luminal subtypes (ER positive) have a better prognosis, while the basal-like subtype (ER and HER-2 negative and basal marker positive) is generally associated with an aggressive clinical history, poor prognosis, resistance to hormonal and targeted chemotherapies, and limited treatment choices. 3 In contrast, some studies have described no association between the basal-like subtype and prognostic factors. 13 In the present study, classification of CMTs into molecular subtypes revealed luminal A (ER positive and HER-2 negative) to be the most common subtype (44%) of canine mammary carcinoma, while very few tumors (3.1%) were normal-like (ER, HER-2, and basal marker negative). The histological classification was related to molecular subtype. Furthermore, histological grade and lymphatic invasion were also associated with molecular subtype. Basal-like tumors were usually high grade with a high rate of lymphatic invasion, whereas luminal A tumors were usually low grade with a low rate of lymphatic invasion. These results are very similar to those reported in HBCs. 3
Despite differences in the methodologies of previously reported studies, a consistent finding has been that low ER expression and HER-2 overexpression are associated with a poor prognosis in veterinary medicine.2,10,14,20 However, the application of molecular subtyping is still uncertain.5,24 In this study, we have provided evidence of a relationship between molecular subtype and histological grade or lymphatic invasion, although additional in-depth studies are needed. Reclassification of CMTs occurring in predominantly small dog breeds in Korea from 2008 to 2011 could have great clinical value and may lead to improved assessment of tumor prognosis and more accurate selection of the most appropriate anticancer therapies for each case.
Footnotes
Acknowledgements
This article represents part of Keum Soon Im’s PhD thesis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, andor publication of this article: This paper was supported by the Konkuk University in 2013.
