Abstract
Feline mammary carcinoma is highly malignant and generally associated with a poor prognosis, although studies suggest the range of survival times in affected cats is broad. Histologic grading of these tumors is achieved using the Elston and Ellis system, originally developed for human breast cancer. In cats, however, classification using this method has variable prognostic value. Therefore, objectives of this study were (1) to evaluate the Elston and Ellis grading system for feline mammary carcinoma in a predominantly spayed population and (2) to determine whether modification of this system or development of a novel system improved the prognostic value of histologic grading. Survey data and histologic features for 108 carcinomas from 97 cats were analyzed with respect to overall survival. Elston and Ellis grading failed to correlate significantly with overall survival. Using multivariable analysis, lymphovascular invasion, nuclear form, and mitotic count each demonstrated independent prognostic significance (P = .008, <.001, and .004, respectively). Modifications of the Elston and Ellis system and a novel grading system were proposed based on these results; all showed significant correlation with overall survival (P < .001). Median survival times were 27, 29, or 31 months for grade I; 14, 12, or 14 months for grade II; and 13, 5, or 8 months for grade III carcinomas using the mitotic-modified Elston and Ellis, the revised Elston and Ellis, or the novel grading system, respectively. Based on this retrospective study, adoption of the species-specific systems as proposed here may improve the prognostic value of histologic grading for feline mammary carcinoma.
Keywords
Feline mammary chain tumors are encountered routinely in primary veterinary practice, accounting for 17% of neoplasms in female cats. 15 Reported malignancy rates generally range between 80% and 90%, 9,15 with fibroadenoma, fibroadenomatous hyperplasia, or duct ectasia being the most commonly encountered benign, dysplastic, or nonneoplastic masses, respectively. 9 The etiology is unknown but almost certainly multifactorial; reported risk factors include sex, 12 breed, 11,14 age, 38 hormone exposure, 21 and reproductive status. 26
Although feline mammary carcinomas tend to be biologically aggressive, survival times can vary significantly. 19,29,36 Prognosis in affected cats has been shown previously to be influenced by tumor diameter, 14,16,36,38,39 World Health Organization (WHO) stage and modified WHO stage, 14,34 extent of surgery, 16,25 and histologic grade. 4,34 The Elston and Ellis (EE) histologic grading system (also known as the Nottingham Grading System) has been widely adopted by veterinary investigators for the grading of feline mammary carcinoma. 2,4,19,28,32 –34,40 This system represents the “gold standard” in assessment of invasive human breast cancer, whereby distinct histopathologic features (ie, percentage tubule formation, nuclear pleomorphism, and mitotic count) are scored and added together to produce a grade, which then correlates with degree of malignancy and prognosis. 8,30,31 Use of this grading system by veterinary pathologists is based primarily on evidence suggesting that feline mammary carcinoma represents a suitable model for human breast cancer, particularly the more aggressive estrogen-negative carcinomas. 2,22,39,41
Castagnaro et al 4 found the EE grading system to have good predictive value for well-differentiated (grade I) and poorly differentiated (grade III) carcinomas. The same grading method was found to have high predictive value in queens with invasive carcinomas by Millanta et al. 19 More recently, Seixas et al 34 found histologic grade determined using the EE grading system to be an independent prognostic factor for middle-aged to elderly queens with mammary carcinoma. These studies present compelling preliminary evidence in support of histologic grading using the EE grading system as a predictor of overall survival (OS) time in unspayed female cats.
Matos et al 18 recently called attention to the need for a standardized grading system for feline mammary gland carcinomas, where early detection, reliable histologic characterization, and aggressive treatment may have a significant influence on survival times. The aim of the current study was to (1) determine whether histologic grade using the EE grading system is a reliable prognostic indicator for a predominantly spayed, North American population of female cats with mammary carcinoma and (2) determine whether modification of the EE grading system and/or development of a novel grading system would improve the prognostic value of routine histologic examination of feline mammary carcinoma.
Materials and Methods
Case Origin and Data Collection
Cases were drawn from 834 surgical biopsy specimens of mammary tumors originating from 789 domestic cats and submitted to either Veterinary Diagnostic Services (later Prairie Diagnostic Services, Inc) at the University of Saskatchewan between 1989 and 2011 or to the Veterinary Diagnostic Laboratories at Colorado State University between 2005 and 2010. Use of animal tissues in this study was approved by the University Committee on Animal Care and Supply at the University of Saskatchewan. A total of 329 surveys were distributed to referring clinics to obtain follow-up information based on the original medical case file. Partial or complete information on 225 cases comprising 255 excisional biopsy specimens (tumors) was obtained. The survey collected the patient signalment; size, location, appearance, and duration of mammary tumor(s); presence and location of lymph node and/or distant metastasis at initial diagnosis; presence and timing of local/regional recurrence or metastases; and the timing and nature of the eventual outcome. All cases were staged according to the modified WHO staging system. 15 Briefly, stage I and stage II included cats with primary tumors less than 2 cm and 2 to 3 cm in diameter, respectively, with no evidence of regional or distant metastases. Stage III included cats with primary tumors less than 2 cm or 2 to 3 cm in diameter with evidence of regional metastases or cats with tumors greater than 3 cm in diameter with or without evidence of regional metastases. Stage IV included any cat with evidence of distant metastases regardless of tumor size or regional metastases.
Inclusion/Exclusion Criteria
This study included only primary mammary tumors from female cats (spayed or intact) with complete survey data, a specimen of adequate quality, a confirmed histologic diagnosis of invasive mammary carcinoma, and a WHO clinical stage of I, II, or III. Except in cases where death or euthanasia occurred and was determined or suspected to be tumor related, at least 1 year of follow-up information was required for inclusion. Any cats receiving treatment beyond surgical excision of the tumor (ie, drug or radiation therapy) were excluded. Noninfiltrating (in situ) carcinomas were excluded. A tumor was considered primary when there was no prior history of mammary neoplasia indicated in the pathology report or survey data. Every effort was made to confirm mammary origin. In cases that initially presented with multiple mammary masses, all submitted tumors were evaluated independently. A total of 97 cats fulfilled the case definition, encompassing 108 tumors. Of those, 88 (81%) were single tumor submissions, while 20 (19%) were multiple tumor submissions from the same animal.
Processing of Tissues
Archived, paraffin-embedded tissues were retrieved, sectioned (4 μm), and stained with hematoxylin and eosin (HE). When it was necessary to confirm intravascular invasion, vascular endothelial cells were highlighted by immunohistochemical staining for Von Willebrand factor carried out on paraffin-embedded sections using a commercial staining platform (Benchmark staining platform; Ventana Medical Systems, Tucson, AZ) and a streptavidin-biotin detection system (BMK iVIEW DAB Paraffin detection kit; Ventana Medical Systems). The primary antibody (polyclonal rabbit anti–human Von Willebrand factor; Dako Canada, Inc, Mississauga, ON, Canada) was applied for 32 minutes at a dilution of 1:1000.
Histologic Assessment Criteria
Histologic slides were reviewed independently by 2 authors (S.W.M. and K.M.M.), with direct assistance from two ACVP board-certified veterinary pathologists (J.L.D. and E.S.). Assessors were blinded to the clinicopathologic (survey) data and clinical outcome; all discordant results were reviewed by S.W.M. and K.M.M. with a multiheaded microscope to reach consensus. When more than 1 section was available for examination, only the one containing the largest portion of the tumor was selected for evaluation. The only exception was assessment of lymphovascular invasion, where all available sections were screened. All tumors were initially classified according to the WHO subtypes described for malignant feline mammary carcinomas. 20 The current study included tubulopapillary, solid, cribriform, and squamous cell carcinomas, with most demonstrating more than 1 growth pattern. In these instances, the pattern comprising the largest relative portion of the tumor determined the subtype.
Tumor characteristics evaluated included percentage tubule formation, necrosis, squamous differentiation, inflammation, lymphovascular invasion, stromal response, chromatin vesiculation, nucleolar morphology, anisokaryosis, nuclear size, nuclear form, nuclear pleomorphism, and mitotic count. Percentage tubule formation was evaluated according to Elston and Ellis. 8 Briefly, the proportion of neoplastic cells within a given section exhibiting discrete tubule formation relative to the total tumor mass was expressed semi-quantitatively as a percentage. Necrosis and squamous differentiation were scored in similar fashion. Changes consistent with squamous differentiation included at least 2 of the following: decreased N:C ratio, increased cytoplasmic eosinophilia, increased cell size, and increased angularity of cell margin. Inflammation was classified subjectively as follows: (1) absent or very mild, (2) predominantly lymphoplasmacytic, or (3) neutrophilic or pleocellular. A tumor was positive for lymphovascular invasion if neoplastic emboli were clearly seen within a continuous, endothelium-lined vessel lumen. In cases where the assessment of lymphovascular invasion was equivocal or consensus could not be reached, additional sections were stained for Von Willebrand factor to highlight vascular endothelium and a definitive classification made. Stromal response encompassed desmoplasia, myofibroblastic/myoepithelial hyperplasia, or a combination of both; it was often not possible to definitively discriminate between these processes in HE-stained tissue sections. The stromal response was graded as normal (mild-moderate) or marked when limited primarily to the periphery of the tumor, or intratumoral when it dissected through the tumor in a trabecular fashion. Assessment of neoplastic cell nuclei was limited to the least differentiated and/or most invasive portions of the tumor in areas lacking appreciable squamous differentiation to reduce bias. Normal nuclear morphology was defined as that which may be found in normal mammary tubular epithelium. Subcategories for chromatin vesiculation, nucleolar morphology, anisokaryosis, nuclear size, and nuclear form were developed and assessed subjectively based on the range of morphology observed in all specimens. Chromatin vesiculation evaluated the predominance of euchromatin within the nuclear envelope as evidenced by areas of pallor or clear space. Categories included (1) none or mildly vesiculated, (2) moderately vesiculated, or (3) markedly vesiculated. Nucleoli were characterized as (1) indistinct, (2) small and prominent, or (3) large, multiple, or abnormally shaped. Anisokaryosis and nuclear size were expressed relative to adjacent tumor cells and nuclei in normal mammary epithelium, respectively. The nuclear form assessment derived from a high-power (40–60× objective), subjective evaluation of nuclear shape independent of other nuclear features or artifactual changes (Figs. 1–4, Suppl. Figs. 1–6). Deviations from a smooth nuclear contour and round or oval shape such as corrugation, angularity, clefting (indentation), or overtly ameboid shape were considered abnormal. Several fields were evaluated in the least differentiated and/or most invasive portion of the tumor, and the number of nuclei exhibiting abnormal form was estimated relative to the total number of nuclei within a given field and expressed as a percentage. Subcategories (≤5%, 6%–25%, or >25% abnormal) were then assigned. Mitotic figures were evaluated according to Elston and Ellis. 8 Briefly, mitotic figures were counted in 10 consecutive fields at the periphery of the tumor in the areas of highest proliferative activity. Microscope field diameter was maintained at 0.53 mm for all observations to standardize counts. 1 Care was taken to exclude apoptotic, pyknotic, or otherwise hyperchromatic nuclei, only including those that had definitively entered prophase or were in metaphase, anaphase, or telophase. In certain cases where tumor heterogeneity and high mitotic rate caused mitotic count to differ by more than 20% between 2 observers, they were repeated independently until acceptable concordance was attained. A correlation coefficient (Pearson’s R value) of 0.954 was obtained overall.
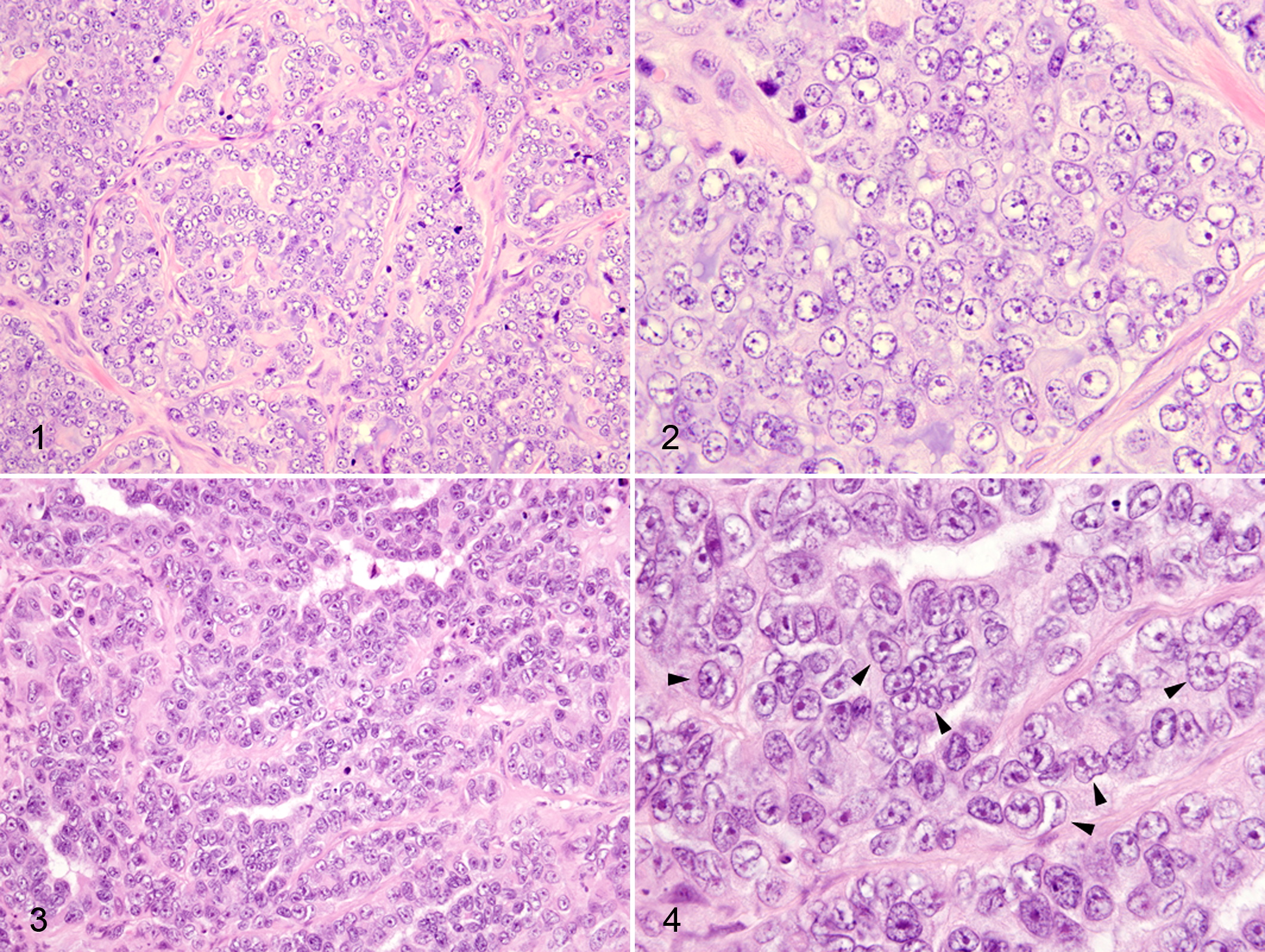
Histologic Grading
Initially, tumors were graded according to the EE grading system described for human breast cancer (Table 1). 8 In this system, carcinomas are scored according to 3 criteria: percentage tubule formation, degree of nuclear pleomorphism, and mitotic count. Briefly, the degree of tubule formation is assessed subjectively on low power and expressed as a percentage. When greater than 75% of the tumor parenchyma exhibits tubule formation, 1 point is assigned. Tubule formation between 10% and 75% is given a score of 2 points, and 3 points are assigned when <10% of the tumor parenchyma displays tubule formation. Nuclear pleomorphism scoring is conducted at high power (40×) in the least differentiated and/or most invasive portion of the tumor, typically along the periphery. Tumor cell nuclei that display uniform chromatin distribution with only mild variation in size, shape, and contour relative to normal nuclei are assigned 1 point. Enlarged nuclei with more vesicular chromatin and moderate variation in size and shape are given 2 points. Finally, 3 points are assigned when tumor cell nuclei exhibit marked vesiculation as well as marked variation in size and shape. Nucleoli are very often present in the latter 2 cases; multiple nucleoli or nucleolar atypia favor category 3.
Elston and Ellis (EE), Mitotic-Modified Elston and Ellis (MMEE), and Revised Elston and Ellis (REE) Grading Systems for Evaluation of Invasive Mammary Carcinoma in Female Cats.
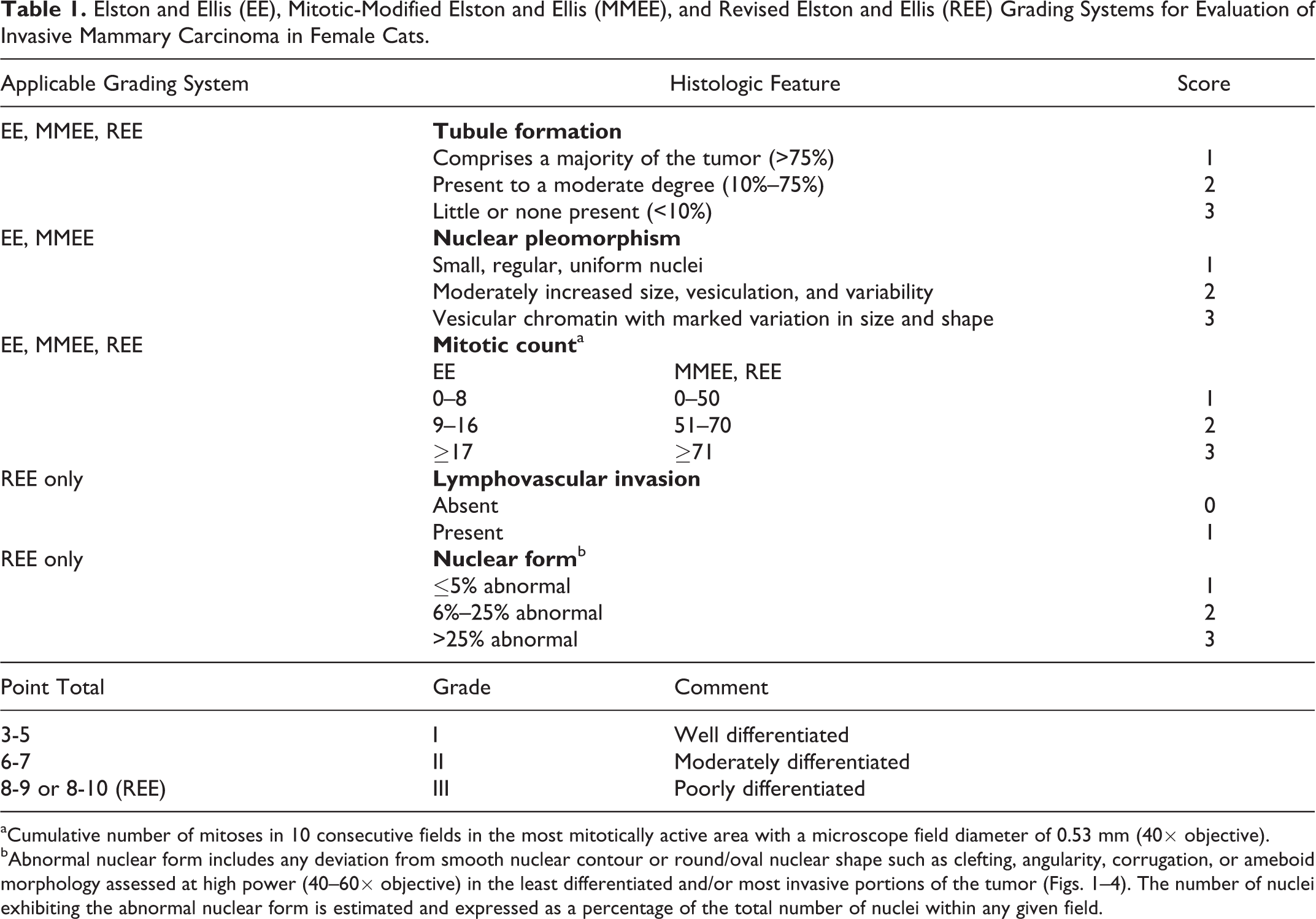
aCumulative number of mitoses in 10 consecutive fields in the most mitotically active area with a microscope field diameter of 0.53 mm (40× objective).
bAbnormal nuclear form includes any deviation from smooth nuclear contour or round/oval nuclear shape such as clefting, angularity, corrugation, or ameboid morphology assessed at high power (40–60× objective) in the least differentiated and/or most invasive portions of the tumor (Figs. 1–4). The number of nuclei exhibiting the abnormal nuclear form is estimated and expressed as a percentage of the total number of nuclei within any given field.
Mitotic counts were grouped as follows: a total of 0 to 8 mitotic figures in 10 high-power fields (40×) was given 1 point, 9 to 16 was given 2 points, and >17 was given 3 points. The score for each category was summed, and the total score for each tumor corresponded to a predetermined grade (I, II, or III), indicating well-differentiated, moderately differentiated, or poorly differentiated carcinomas, respectively.
Three new grading systems were designed, applied to our data, and analyzed with respect to OS. First, in the mitotic-modified Elston and Ellis (MMEE) grading system (Table 1), range subcategories within the mitotic count category of the EE grading system were modified to better accommodate the wide range and high magnitude of mitotic counts observed within the tumors we evaluated. Cutoff values between mitotic count subcategories were derived directly from tertile boundaries from our results. Next, with the revised Elston and Ellis (REE) grading system (Table 1), the EE grading system was modified further to include nuclear form scoring and lymphovascular invasion. Nuclear pleomorphism scoring was removed, and an additional point was added to the grading chart to accommodate the addition of lymphovascular invasion. Scoring and grading with the MMEE and REE grading systems was otherwise achieved in the same manner as described above for EE grading. Finally, a novel grading system was developed directly from the Cox proportional hazards analysis (Table 2). Hazard ratios were assigned to the presence of the independent prognostic factors (lymphovascular invasion, nuclear form, and mitotic count) using coefficients from the model and divided into grades. Mitotic count subcategories were derived directly from the median mitotic count observed in our results. Using the novel grading system, the absence of lymphovascular invasion together with less than or equal to 5% abnormal nuclear form and a mitotic count less than or equal to 62 in 10 high-power fields corresponded to grade I (low-grade carcinoma). The presence of any one of lymphovascular invasion, greater than 5% abnormal nuclear form, or a cumulative mitotic count greater than 62 yielded grade II (intermediate-grade carcinoma). Finally, if any 2 or all 3 of the aforementioned features were present, grade III (high-grade carcinoma) was assigned.
Novel Grading System for Evaluation of Invasive Mammary Carcinoma in Female Cats.
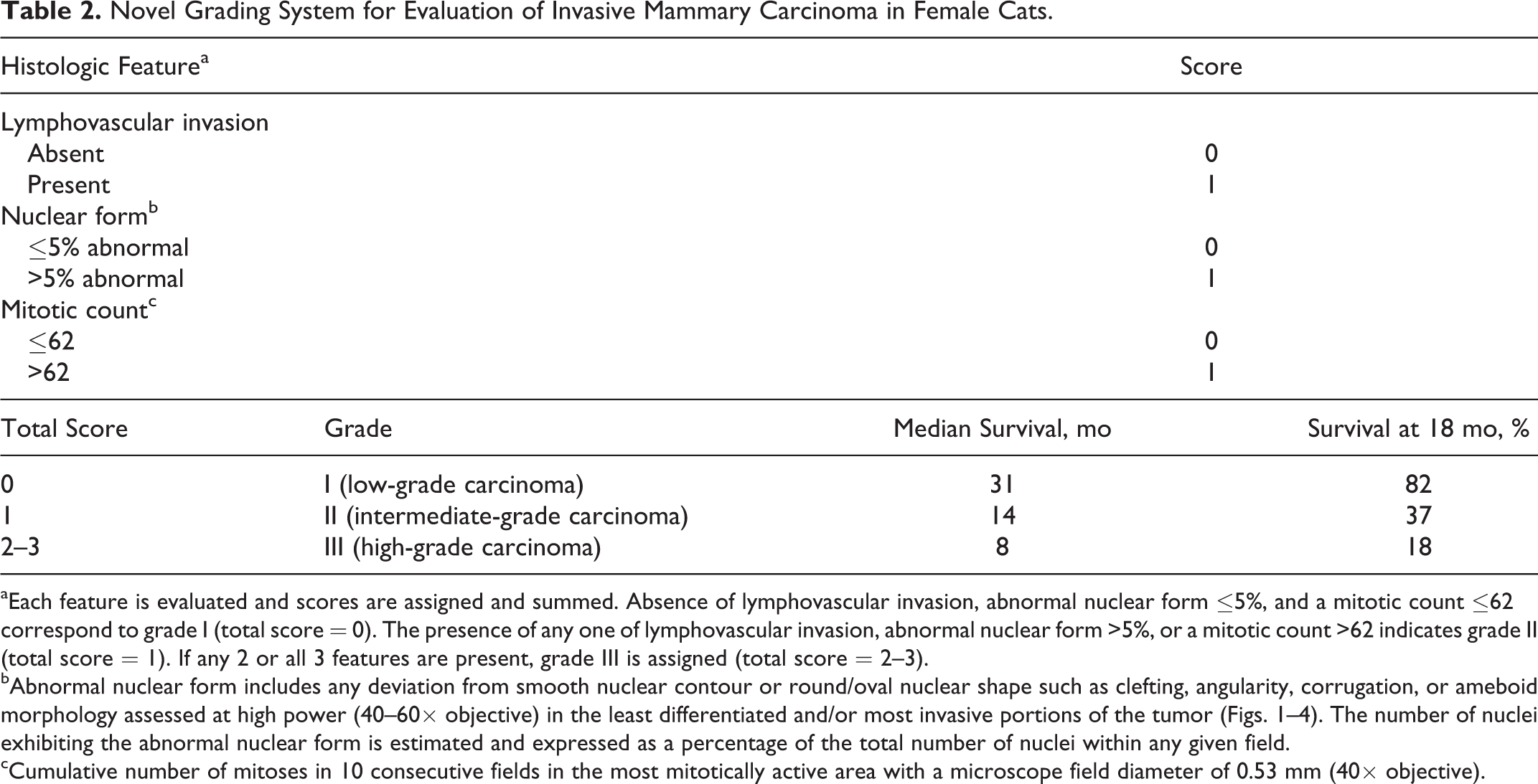
aEach feature is evaluated and scores are assigned and summed. Absence of lymphovascular invasion, abnormal nuclear form ≤5%, and a mitotic count ≤62 correspond to grade I (total score = 0). The presence of any one of lymphovascular invasion, abnormal nuclear form >5%, or a mitotic count >62 indicates grade II (total score = 1). If any 2 or all 3 features are present, grade III is assigned (total score = 2–3).
bAbnormal nuclear form includes any deviation from smooth nuclear contour or round/oval nuclear shape such as clefting, angularity, corrugation, or ameboid morphology assessed at high power (40–60× objective) in the least differentiated and/or most invasive portions of the tumor (Figs. 1–4). The number of nuclei exhibiting the abnormal nuclear form is estimated and expressed as a percentage of the total number of nuclei within any given field.
cCumulative number of mitoses in 10 consecutive fields in the most mitotically active area with a microscope field diameter of 0.53 mm (40× objective).
Statistical Analysis
Analyses were conducted using Prism (GraphPad Software, La Jolla, CA) and Stata (StataCorp LP, College Station TX) statistical software packages. Normality was assessed with the D’Agostino and Pearson normality test. To analyze the relationship between individual survey or histologic criteria/grading and OS, Kaplan-Meier curves were generated and compared using log rank or Gehan-Breslow-Wilcoxon tests. A Cox proportional hazards model was used to evaluate the effect of multiple criteria on OS, and hazard ratios were calculated using the Mantel-Haenszel method. For continuous variables, categorization was based on median values or tertiles unless otherwise stated. OS was defined as the period between the date of biopsy/excision and the date of death or last documented follow-up. Cases were grouped into 5 distinct outcomes: (1) alive, (2) lost to follow-up, (3) tumor-related death/euthanasia, (4) non–tumor-related death/euthanasia, and (5) death/euthanasia of unverified cause (but suspected to be tumor related). For statistical analysis of OS, outcomes 1, 2, and 4 were considered censored events. In all cases, a P value equal to or less than .05 was considered significant.
Results
Female cats in this study ranged in age from 3.5 to 20 years old (median, 11 years; mean [SD], 11.4 [3.5] years). Breeds included 65 domestic short hair (66.3%), 14 domestic long hair (14.4%), 5 domestic medium hair (5.1%), 6 Siamese (and crosses) (6.2%), 2 Maine Coon (and crosses) (2%), 1 Korat cross (1%), 1 Manx (1%), 1 Birman (1%), 1 Chinchilla (1%), and 1 Devon Rex (1%). Ovariectomy or ovariohysterectomy (spaying) had been performed previously in 82 cats (84.7%), while 15 (15.3%) were intact or spayed concurrently with mass removal. Tumor diameter was less than 2 cm in 51 cases (52.6%), between 2 and 3 cm in 18 cases (18.4%), greater than 3 cm in 19 cases (19.6%), and unknown in 9 cases (9.2%). On presentation to the referring clinic, 56 cats (57.7%) were reported to have a single mass, 40 (40.8%) had multiple masses, and the number of masses was unreported in 1. A total of 43 cases (43.9%) were classified as WHO stage 1, 15 (15.3%) as stage 2, and 32 (33%) as stage 3, with 7 (7.2%) unknown (survey information incomplete).
Results of Kaplan-Meier survival analysis of survey data and histologic features are listed in Tables 3, 4, and 5. Although tumor diameter was not a significant factor overall, those greater than 3 cm in diameter were associated with reduced OS (P = .046) compared with tumors less than 2 cm in diameter. More advanced WHO stage and the presence of lymph node metastases also showed statistically significant negative correlation with OS (both P = .01). Both WHO histologic subtype (P = .005) and the presence of lymphovascular invasion (P < .001) corresponded with OS. A weak association was found between tumors with >75% tubule formation and those with less than 10% tubule formation (P = .037) and OS, the latter conferring a poorer prognosis. The median survival of cats with solid or cribriform carcinomas (10 and 8 months, respectively) was less than half that of cats with the tubulopapillary subtype (21 months). Percentage of necrosis, the presence or nature of any inflammation, the stromal response, and the percentage of squamous differentiation were not statistically significant with respect to OS. In contrast, with the exception of chromatin vesiculation, all nuclear characteristics evaluated within the neoplastic population were significantly and inversely related to OS, including abnormal nucleolar morphology (P = .013), increased anisokaryosis (P = .013), increased size (P = .038), abnormal nuclear form (P < .001), increased nuclear pleomorphism score (P = .006), and increased mitotic count (P = .021). Visible nucleoli were absent in only 2 tumors (1.9%); nuclear size was increased relative to normal in 104 tumors (96.3%). The mean mitotic count was 67 mitotic figures per 10 high-power fields, with a median value of 62. When divided into tertiles, categories were 0 to 50, 51 to 70, and >71 mitotic figures per 10 high-power fields. In contrast, when mitotic counts were grouped according to the EE grading system, 1 tumor (1.0%) was given a score of 1, 4 (3.7%) a score of 2, and 103 (95.4%) a score of 3.
Kaplan-Meier Analysis of Selected Clinicopathologic Criteria With Respect to Overall Survival in Female Cats With Mammary Carcinoma.
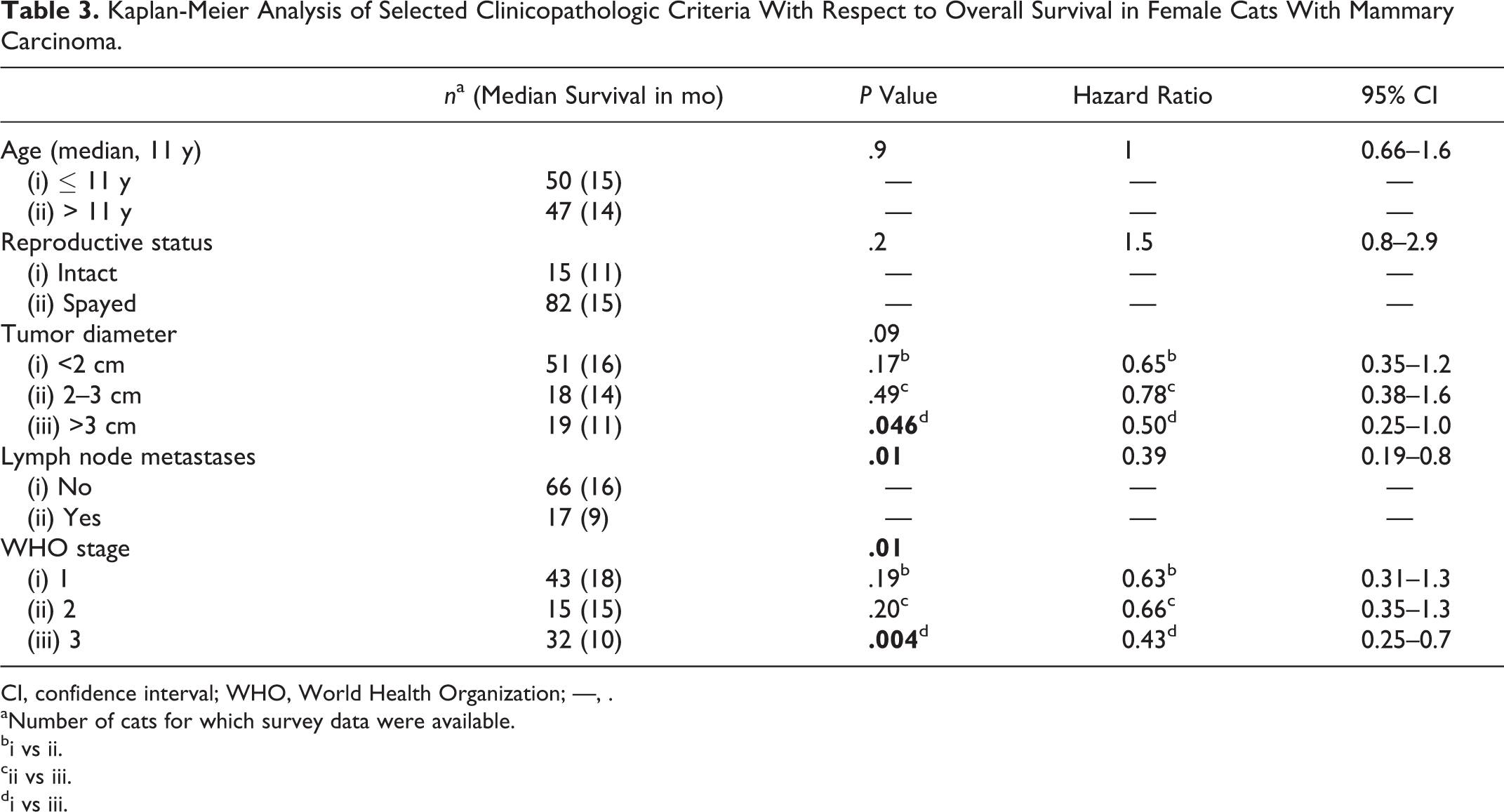
CI, confidence interval; WHO, World Health Organization; —, .
aNumber of cats for which survey data were available.
bi vs ii.
cii vs iii.
di vs iii.
Kaplan-Meier Analysis of Low-Power Histologic Criteria With Respect to Overall Survival in Female Cats With Mammary Carcinoma.
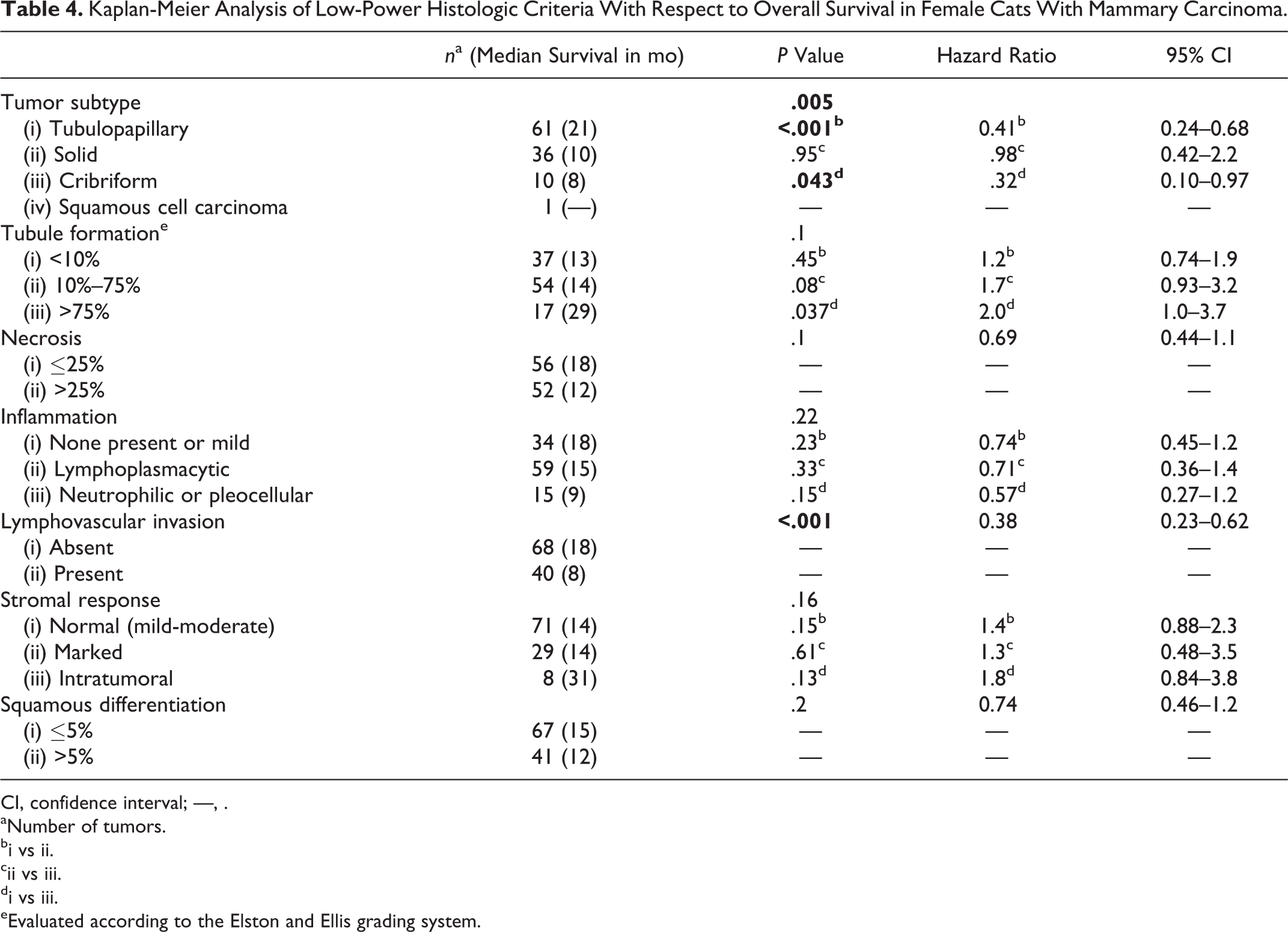
CI, confidence interval; —, .
aNumber of tumors.
bi vs ii.
cii vs iii.
di vs iii.
eEvaluated according to the Elston and Ellis grading system.
Kaplan-Meier Analysis of High-Power Histologic Criteria With Respect to Overall Survival in Female Cats With Mammary Carcinoma.
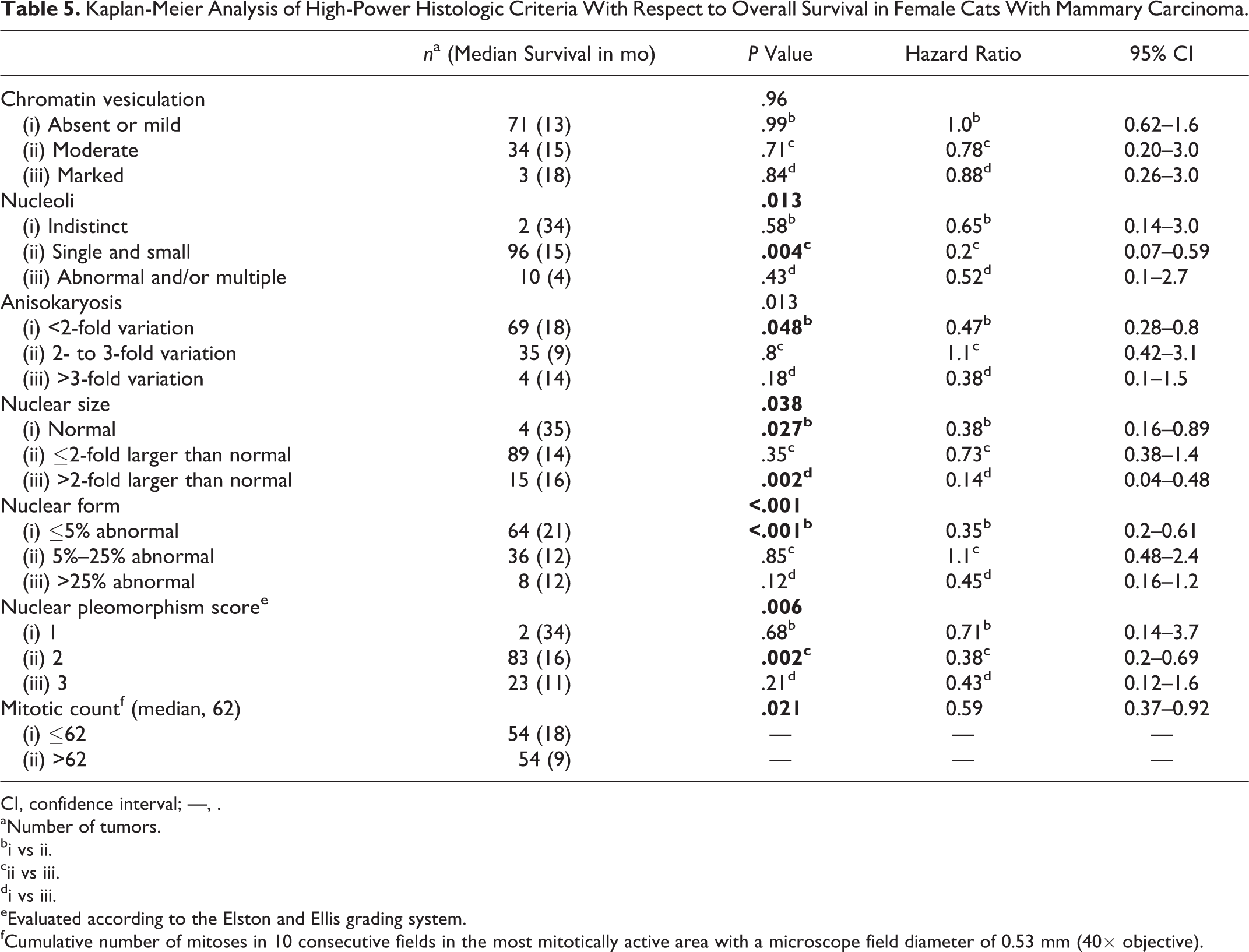
CI, confidence interval; —, .
aNumber of tumors.
bi vs ii.
cii vs iii.
di vs iii.
eEvaluated according to the Elston and Ellis grading system.
fCumulative number of mitoses in 10 consecutive fields in the most mitotically active area with a microscope field diameter of 0.53 mm (40× objective).
Only lymphovascular invasion, mitotic count, and nuclear form could be considered independent prognostic factors in the current study (P = .008, .004, and <.001, respectively) according to Cox proportional hazards analysis (Suppl. Table S1).
Overall, EE grading was not associated with OS (Fig. 5). A statistically significant difference was found between grades II and III (P = .026), with a median survival of 17 months (n = 63) and 13 months (n = 43), respectively. Only 2 tumors (1.9%) received a nuclear pleomorphism score of 1, and only 5 (4.6%) received a mitotic count score of either 1 or 2 (data not shown).
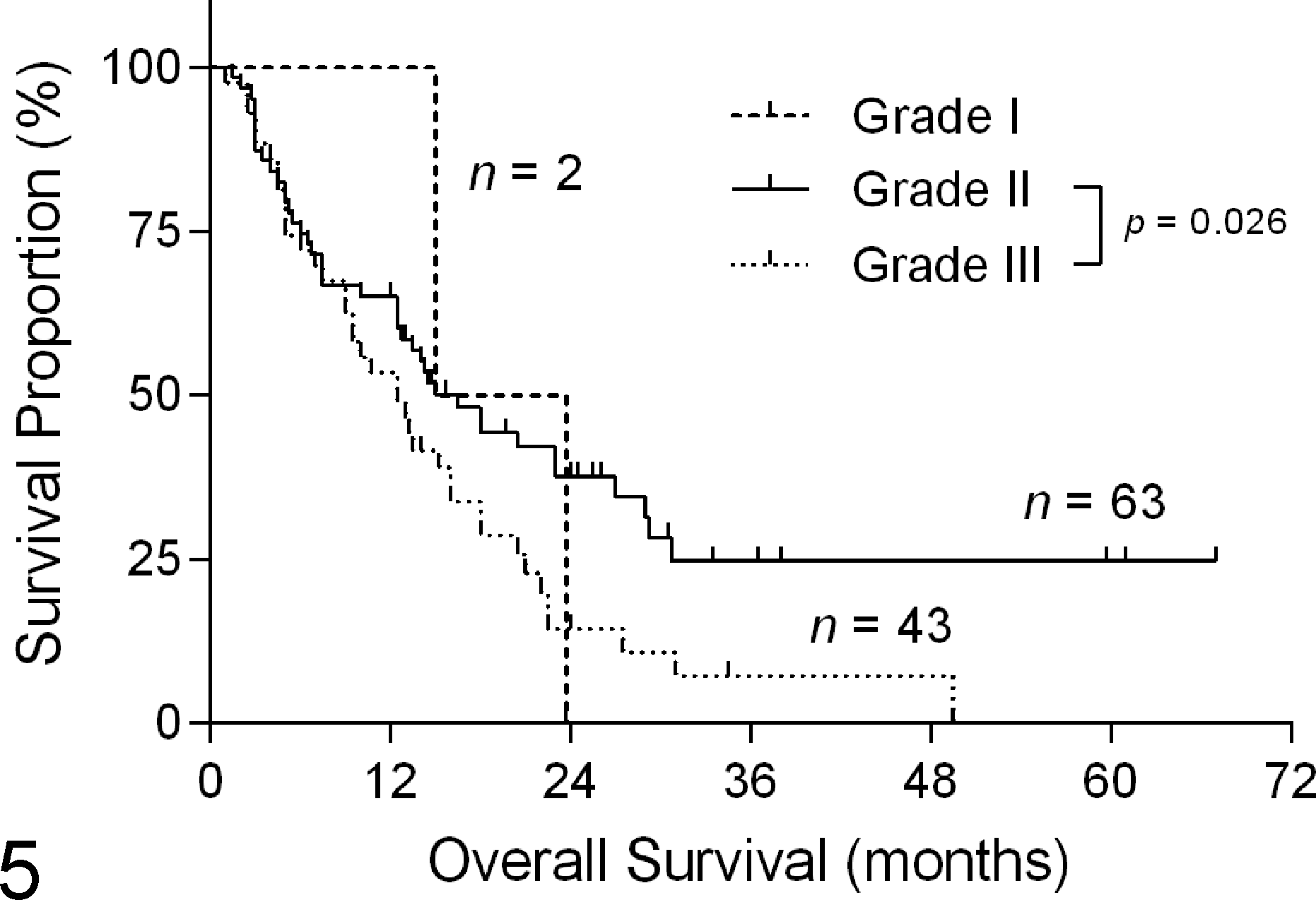
Kaplan-Meier curve depicting Elston and Ellis grade (Table 1) vs overall survival in female cats with invasive mammary carcinoma. Median survival was 19 months, 17 months, and 13 months for grade I, II, and III tumors, respectively. Hashmarks indicate censored events.
A strong association was found between the MMEE grading and OS (P < .001; Fig. 6), REE grading and OS (P < .001; Fig. 7), and novel grading and OS (P < .001; Fig. 8). The MMEE grading system classified 29 carcinomas (26.9%) as grade I (median survival of 27 months), compared with only 2 grade I carcinomas (1.9%) using the original EE grading system (median survival 19 months). Median survival was 14 months (n = 61) and 13 months (n = 18) for cats with grade II and III carcinomas, respectively. REE grading resulted in 32 carcinomas (29.6%) being classified as grade I (median survival 29 months). This was 2.5-fold longer OS than grade II carcinomas (median survival 12 months; n = 63) and 5.8-fold longer OS than grade III carcinomas (median survival 5 months; n = 13). When individual grades were analyzed separately, a statistically significant difference was not identified between grades II and III in the MMEE or the REE grading system. When tumors were graded histologically using the novel system, a statistically significant difference was present between grades I and II (P = .0012), grades II and III (P = .024), and grades I and III (P < .0001). Cats with grade III (high-grade) carcinomas had only one-fourth (25.8%) the survival time of cats with grade I (low-grade) carcinomas (median survival 8 months [n = 48] and 31 months [n = 22], respectively) and less than two-thirds (57%) the median survival time of cats with grade II carcinomas (median survival 14 months; n = 38).
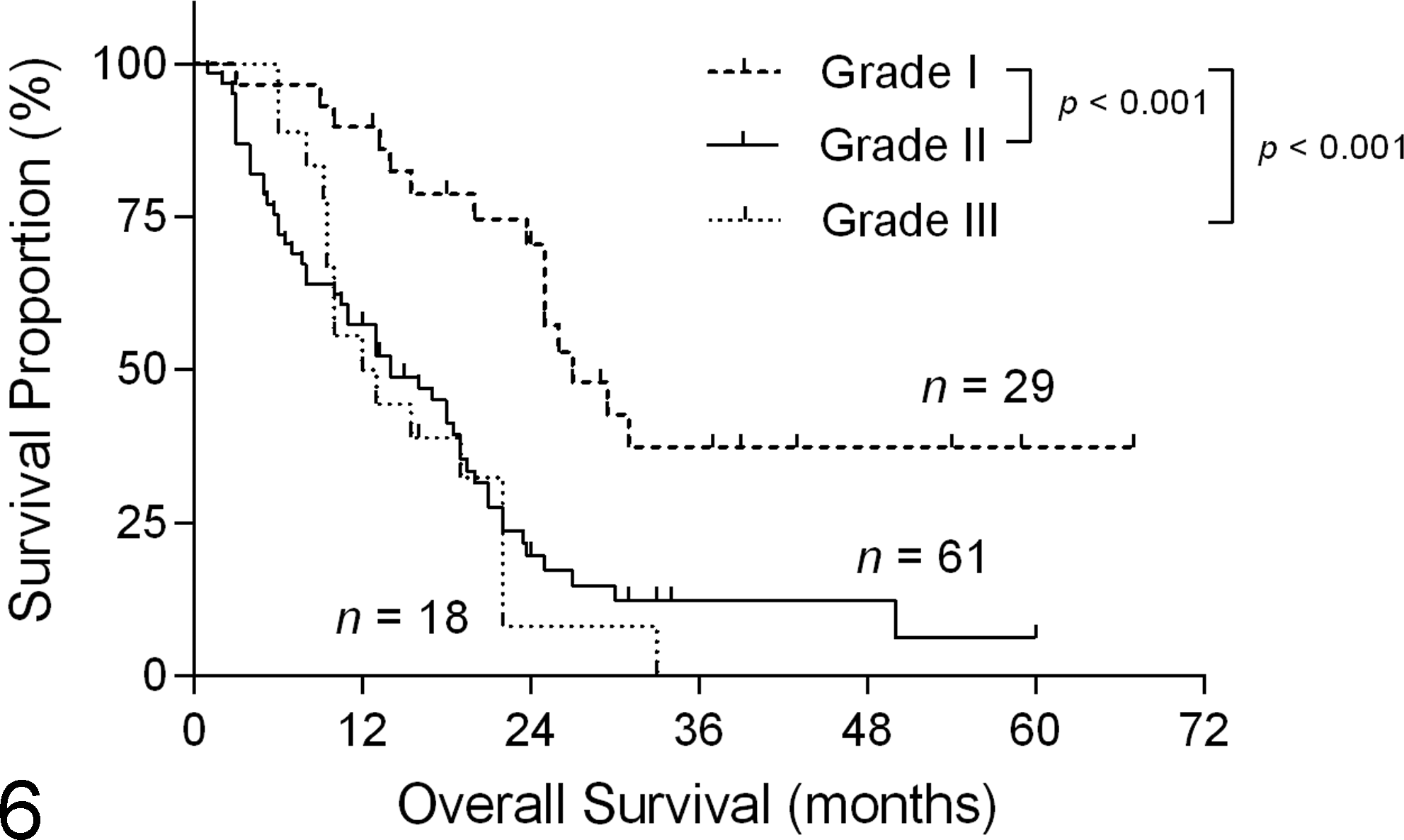
Kaplan-Meier curve depicting mitotic-modified Elston and Ellis grade (Table 1) vs overall survival in female cats with invasive mammary carcinoma. Median survival was 27 months, 14 months, and 13 months for grade I, II, and III tumors, respectively. Hashmarks indicate censored events.
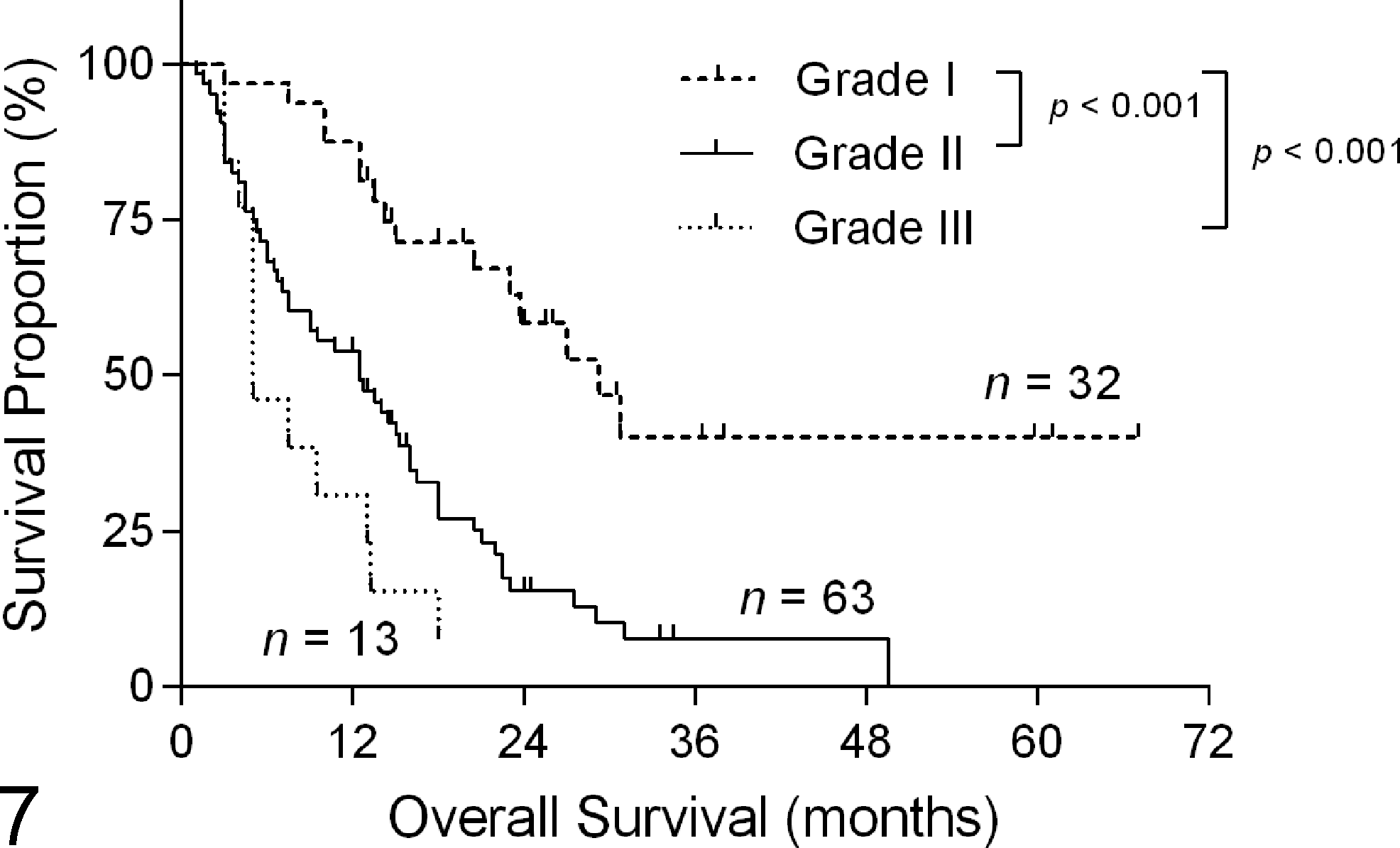
Kaplan-Meier curve depicting revised Elston and Ellis grade (Table 1) vs overall survival in female cats with invasive mammary carcinoma. Median survival was 29 months, 12 months, and 5 months for grade I, II, and III tumors, respectively. Hashmarks indicate censored events.
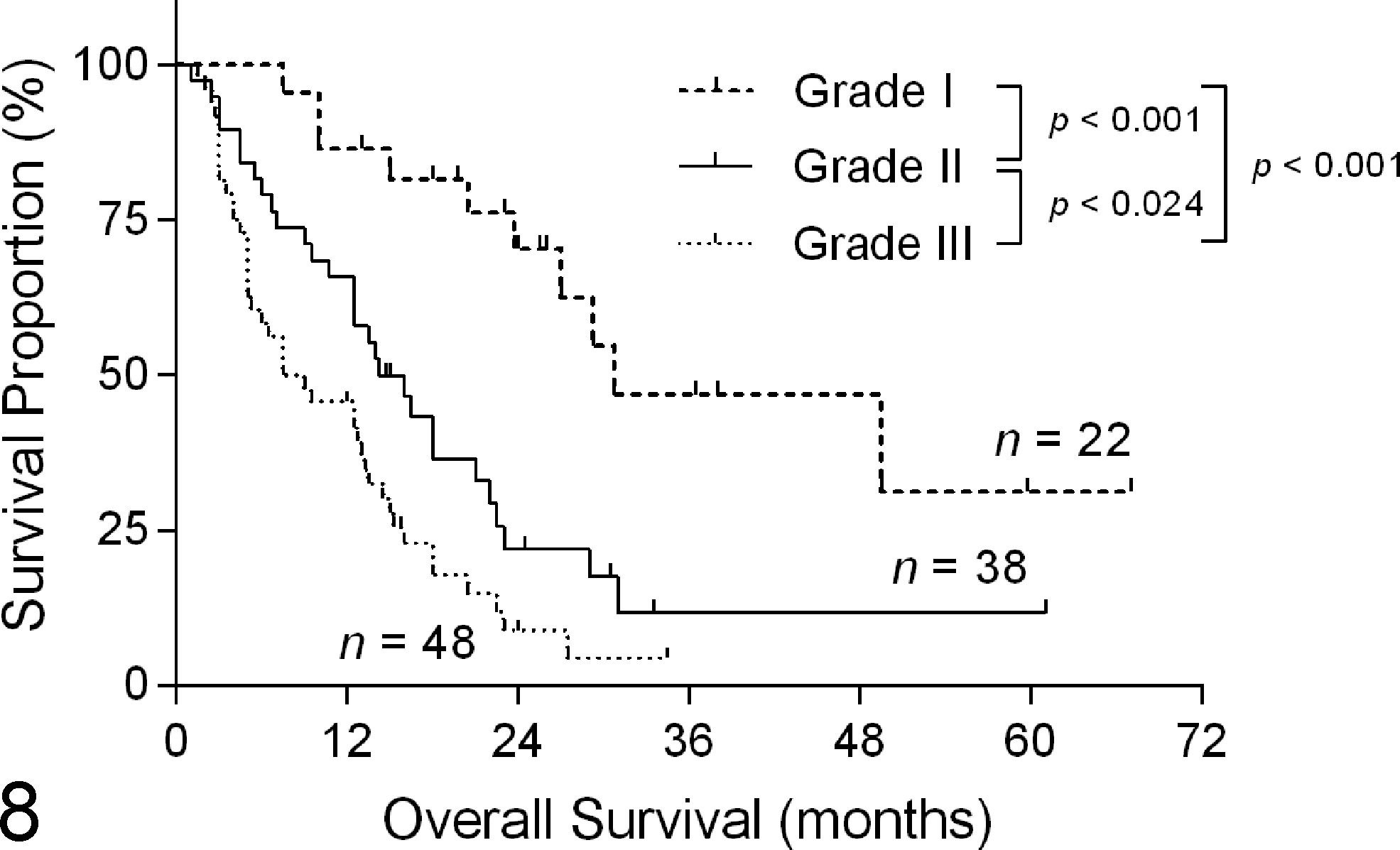
Kaplan-Meier curve depicting novel grade (Table 2) vs overall survival in female cats with invasive mammary carcinoma. Median survival was 31 months, 14 months, and 8 months for grade I, II, and III tumors, respectively. Hashmarks indicate censored events.
Discussion
To our knowledge, this is the largest histologic analysis of feline mammary carcinoma to date, aimed at evaluating and improving current grading methodologies. Due to the current lack of a globally accepted, reliable, species-specific grading system, the poor prognosis generally conferred upon cats with malignant mammary carcinomas in diagnostic practice is not typically revised as a result of histologic assessment. It was evident in this and previous studies, however, that survival times in affected cats are variable, with postsurgical survival extending for years rather than months in many cases. 19,29,36 Findings of this study support the use of modified EE grading systems or a novel grading system to improve the prognostic value of histologic grading for feline mammary carcinoma.
Tumor size (or diameter) is easily estimated at presentation to a primary practice and is a commonly used diagnostic indicator due to its reported correlation with prognosis in feline mammary carcinoma. 14,16,39 Our study demonstrated only a weak statistical association between tumor diameter and OS and only for tumors greater than 3 cm in diameter. This may be due to insufficient numbers as well as the inherent subjectivity involved in measurement of tumor diameter (calipers vs palpation, in vivo vs ex vivo, etc). Additional shortcomings and possible sources of error encountered when using tumor size (or volume) as a prognostic factor are described by Matos et al. 18 Recent reports support findings here, indicating that while larger tumors (>3 cm diameter) are associated with reduced survival times, tumor size alone appears to have little or no independent prognostic value when smaller than 3 cm in diameter. 25,36
The modified WHO clinical stage provides a useful prognostic indicator in cats with advanced clinical disease. 15 Stage 3 cats had significantly reduced OS compared with stage 1 cats in this study; a statistical association between WHO stage and OS has also been noted elsewhere. 34 Unfortunately, WHO staging relies heavily on tumor diameter and may therefore be inadequate for cats with more localized disease at the time of diagnosis or where regional metastases are not detected (stages I and II).
Application of the EE grading system to our study population resulted in inferior discrimination between tumors and lacked overall association with OS. Tumors were heavily skewed toward grade II or III (58.3% and 39.8% of carcinomas, respectively), resulting in underrepresentation of grade I (well-differentiated) carcinomas (1.9% of evaluated tumors). The asymmetry of this classification stemmed directly from nuclear pleomorphism and mitotic count results, both of which were heavily weighted toward higher scores (data not shown).
These results contrast with previous studies, where the EE grading system has shown significant prognostic utility in intact queens. 4,19,34 It was noted, however, in the prospective study by Castagnaro et al 4 that a grade II designation lacked prognostic value, a category that comprised a majority of the tumors they evaluated. Furthermore, survival analysis was not conducted in that study, limiting the conclusions that may be drawn. In the report by Millanta et al, 19 significant overlap is evident on the Kaplan-Meier curves depicting EE grade vs OS, and it is unclear to which group (grade) comparisons the statistical significance is attributable. The retrospective investigation by Seixas et al 34 is most comparable to the present study in both scope and objective. Similar to our findings, only 5.4% of tumors they evaluated were given a grade I designation using the EE grading system, perhaps further underscoring the need for a more discriminating grading method. Other possible explanations for the discrepant results found in this study include the inherent subjectivity associated with tumor grading, 23 differences in population-specific factors (ie, spayed vs intact, geographic location), differences attributable to study type and case definition, or an inherently deficient grading system.
To address the latter possibility, numerous morphological criteria were evaluated in the present study and analyzed with respect to OS. Tumor subtypes were included, despite their characterization as descriptive rather than prognostic. 20 Indeed, a significant association between tumor subtype and OS was found here and by Seixas et al 34 but not by Millanta et al 19 or Castagnaro et al. 4 This discrepancy may reflect the inherent subjectivity of histologic evaluation, the morphological heterogeneity of feline mammary carcinoma, or variable interpretation of the category descriptions. 20
Both the mitotic count and the presence of lymphovascular invasion demonstrated a strong correlation with OS, similar to findings in previous studies. 29,34 Mitotic index and histologic staging based on lymphovascular invasion were both found to be independent prognostic factors by Preziosi et al. 29 The prognostic significance of invasion was also recognized by Mandelli et al, 17 who developed a histologic grading system for feline mammary gland tumors based on this feature alone.
In this study, nucleolar morphology, anisokaryosis, nuclear size, nuclear form, and nuclear pleomorphism score all showed a significant correlation with OS in univariate analysis. Upon multivariable analysis, however, nuclear form was the only aspect of nuclear morphology shown here to be an independent prognostic factor in feline mammary carcinoma. Nuclear form was previously evaluated using nuclear morphometry software. DeVico and Maiolino 6 concluded that both the standard deviation and coefficient of variation of nuclear form were reliable prognostic factors in cats with mammary carcinoma. In a more recent study, however, Simeonov and Simeonova 35 found no statistical correlation between nuclear morphometric parameters and OS upon analysis of cytological samples from feline mammary carcinomas. Further investigation is warranted given the results of the present study.
In light of the multivariate analysis results together with the apparent shortcomings of EE grading when applied to the study population, 3 new grading systems were developed in an attempt to improve histologic grading of feline mammary carcinoma. First, as has been recently described for grading canine mammary carcinomas, 27 modifications of the EE grading system were proposed. The MMEE grading system features increased range subcategories within the mitotic count categoryto better accommodate the high median and broad range of mitotic counts encountered within our study population. Application of the MMEE grading system greatly improved detection of grade I (well-differentiated) carcinomas compared with the EE grading system, both in terms of the number of tumors recognized and median survival. The REE grading system represents a more extensive modification of the EE grading system to further improve tumor discrimination. Nuclear pleomorphism, a significant source of skewness within the data and long recognized as the most subjective criterion in the EE system, 7 was removed and replaced by nuclear form. Although still admittedly subjective, this allows the diagnostic pathologist to concentrate on a single aspect of nuclear morphology irrespective of concurrent nuclear features. Lymphovascular invasion was also added, recognizing its prognostic significance. REE grading again resulted in superior recognition of grade I (well-differentiated) carcinomas compared with EE grading. In addition, grade III (poorly differentiated) carcinomas were more adequately represented in terms of median survival. It should be noted, however, that neither the MMEE nor the REE grading system was able to show significant differences between grades II and III. In the latter case, this may be attributable to the low number of grade III carcinomas.
In practical terms, grading tumors with the MMEE grading system is virtually identical to using the original EE grading system. Potential drawbacks of the REE grading system include the additional time required for evaluation of a fourth criterion and familiarization with nuclear form scoring.
A novel grading system was also developed, based solely on those criteria found to be independent prognostic factors: lymphovascular invasion, mitotic count, and nuclear form. Only 2 subcategories were established for mitotic count and nuclear form to simplify the grading system. When applied to the study population, a statistically significant difference was found between all grades in terms of OS. Although fewer were identified relative to MMEE and REE grading, grade I (low grade) carcinomas were well represented. The presence of only 2 categories associated with each criterion greatly simplifies histologic scoring using this system, increasing the potential utility for the diagnostic pathologist. In addition, cats with grade I (low-grade) carcinomas were again more adequately represented by the novel grading system compared with the EE grading system.
A potential limitation associated with use of lymphovascular invasion in the REE and novel grading systems is the inconsistent presence between histologic sections of the same tumor. While it is true that the extent to which identifying lymphovascular invasion depends on which portion(s) or serial section(s) of the tumor are evaluated, the impact this has on its value as a prognostic indicator is unknown. There is suboptimal sensitivity associated with many histologic criteria evaluated on a 2-dimensional section of a 3-dimensional, heterogeneous tumor. Despite these potential shortcomings, vascular invasion has been included in histologic grading systems for other tumors, including squamous cell carcinoma of the tongue in dogs, 3 as well as for prognostication in feline primary lung carcinomas. 10
In contrast to dogs, where the protective effect of early ovariohysterectomy on the development of mammary carcinoma is well established, less is known about the impact of reproductive hormones on feline mammary carcinoma. Overley et al 26 demonstrated a clear protective effect of ovariohysterectomy, whereby those cats having undergone the procedure prior to 1 year of age showed an 86% reduction in risk of mammary carcinoma relative to intact cats. Unfortunately, scant information is available regarding possible effect(s) of spay status on tumor characteristics, grading, and OS in cats with mammary carcinoma. No difference was found between spayed and intact cats with regard to OS in the present study, although median survival was 66% higher in the former group. This may simply be due to a lack of statistical power, as only 15% (n = 16) of carcinomas were from intact animals. In dogs with mammary carcinoma, spay status has been shown to be significantly associated with survival. 5,27 Recently, Hughes and Dobson 13 called attention to the need for larger studies to investigate the effect of spay status on prognostic marker expression in feline mammary carcinomas. It is currently unknown to what extent the effects of endogenous estrogen and/or progesterone exposure may manifest histologically and/or immunohistologically in feline mammary carcinoma. If present, this may influence diagnostic evaluation, including any attempts at histologic grading. Further investigation is required in this area.
One of the strengths of the present study is that most tumor biopsy specimens originated from primary practices in rural, semi-rural, and urban areas rather than large referral centers, reducing bias and improving the generalizability of results. The primary limitation of the present study is its retrospective and multi-institutional nature. Patient data acquired from surveys may have been incomplete or inaccurate, and there was a clear bias favoring available specimens. Furthermore, recordkeeping, diagnostic workup, sample collection/processing, and diagnosis lack standardization in such studies. 37 Except in rare cases, no postmortem examinations were carried out to confirm the extent of disease or cause of death. In some cases, multiple masses were reported by the primary practitioner, but not all were submitted. Every attempt was made to include all potential primary tumors as recommended by Matos et al, 18 although it remains unknown whether unsubmitted tumors were in fact primary, higher grade, or mammary in origin and to what extent they influenced OS.
In summary, the use of the EE grading system for feline mammary carcinoma has not been adopted widely by diagnostic pathologists because it was developed for human breast cancer, it is time-consuming, and its use is not perceived to improve upon the generally poor prognosis associated with feline carcinomas. Application of the EE grading system in our study population resulted in inferior discrimination between tumors with respect to OS. Species-specific, reliable histologic grading of feline mammary carcinomas has the potential to significantly improve routine diagnostic evaluation and prognostication. Currently, no such grading system exists, although the MMEE, REE, and the novel grading systems introduced here may represent a step in this direction. All 3 significantly improved the prognostic value of histologic grading for feline mammary carcinoma, especially for thesubset of affected cats exhibiting prolonged survival that may otherwise go unrecognized. Finally, while the results and proposed grading systems presented herein are applicable to the study population, it is unclear to what extent they are applicable to the greater feline population. Future studies are required to validate these findings, elucidate the role of spay status if any, and provide further guidance as to the most appropriate grading system to use in female cats with mammary carcinoma.
Footnotes
Acknowledgements
We thank Phil Dillman and the Histology Laboratory as well as Melissa Koehnlein and Dale Godson in the Immunology Laboratory at Prairie Diagnostic Services, Inc for their expertise.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the WCVM Companion Animal Health Fund.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
