Abstract
Histopathology remains the cornerstone for diagnosing canine mammary tumors (CMTs). Recently, 2 classification systems (the World Health Organization [WHO] classification of 1999 and the proposal of 2011) and 2 grading methods based on the human Nottingham grade have been used by pathologists. Despite some evidence that the histological subtype and grade are prognostic factors, there is no comprehensive comparative study of these classification and grading systems in the same series of CMTs. In this study, the 2 classifications and the 2 grading methods were simultaneously applied to a cohort of 134 female dogs with CMTs. In 85 animals with malignant tumors, univariable and multivariable survival analyses were performed. Using the 2 systems, the proportion of benign (161/305, 53%) and malignant (144/305, 47%) tumors was similar and no significant differences existed in categorization of benign tumors. However, the 2011 classification subdivided malignant tumors in more categories—namely, those classified as complex, solid, and tubulopapillary carcinomas by the WHO system. Histological subtype according to both systems was significantly associated with survival. Carcinomas arising in benign tumors, complex carcinomas, and mixed carcinomas were associated with a better prognosis. In contrast, carcinosarcomas and comedocarcinomas had a high risk of tumor-related death. Slight differences existed between the 2 grading methods, and grade was related to survival only in univariable analysis. In this cohort, age, completeness of surgical margins, and 2 index formulas adapted from human breast cancer studies (including tumor size, grade, and vascular/lymph node invasion) were independent prognostic factors.
Canine mammary tumors (CMTs) are the most frequent neoplasm in intact female dogs. Despite the efforts made on CMT research, which have contributed to a remarkable increase in knowledge of their biology, histological evaluation remains the cornerstone for classifying and grading CMTs. The intrinsic morphological heterogeneity of these tumors, with a frequent involvement of different cell populations, challenges the pathologist’s task of providing an accurate classification. For nearly 2 decades, most veterinary pathologists used the official histological classification for CMTs endorsed by the World Health Organization (WHO), 18 which included 6 benign tumor categories and 14 malignant subtypes. Several studies have underlined the prognostic value of this classification system, 3,14,18,21 while other authors have not found significant associations between histological type and survival variables. 5,22 More recently, Goldschmidt and coworkers 9 presented an alternative histological classification scheme for CMTs, recognized as the 2011 classification, which emphasizes architectural arrangements of neoplastic cells and involvement of myoepithelial cells in the neoplastic process. The 2011 classification is a complete and exhaustive classification scheme, 13 with 7 benign and 23 malignant subtypes, 9 and its prognostic value was recently demonstrated, 19,20 emerging also as an independent prognostic factor by multivariate analysis of a prospective cohort of CMTs. 20 Currently, veterinary pathologists face uncertainty regarding the histological classification of CMTs as it is not possible to compare the prognostic relevance of these histological classification systems based on published data, since they do not refer to the same cohorts of animals. Moreover, to our knowledge, there are no published reports comparing the prognostic value of the WHO classification and of the 2011 classification in the same series of female dogs with CMTs.
Traditionally, the grading of malignant CMTs has followed that of the human counterparts. In a reference book, Misdorp 17 adopted the numerical human grading method of Bloom and Richardson (1957), 2 which included 3 parameters: tubule formation, nuclear pleomorphism, and number of mitoses/hyperchromatic nuclei per high-power field. Each parameter is scored from 1 to 3 and the overall grade determined by the final combined score. However, during the past decade, the most used grading system for CMTs has been the so-called Nottingham histological grade (NHG) described by Elston and Ellis 8 in 1991. This method is a refinement of the Bloom and Richardson method, also including the assessment of tubule formation, nuclear pleomorphism, and mitotic counts. In NHG, the evaluation of tubule formation is semiquantitative (with percentages) and mitotic counts are scored considering the field area of the microscope used (Suppl. Table S1). 8 In CMTs, the NHG has been associated in univariate analysis with disease progression and cancer-related death, especially when grade I plus grade II were compared to grade III. 14,21,22 Recently, our group demonstrated that the computation of an index formula, adapted from the well-known human Nottingham Prognostic Index 11,15 and that included NHG, tumor size, and a variable related to the spread of the disease (vascular/lymph node invasion), had prognostic value in CMTs. 22 The diversity of cellular populations involved in CMTs, which often includes luminal epithelium, myoepithelium and mesenchymal components, and the great heterogeneity of their histological patterns, also challenged veterinary pathologists when grading the tumors. In a recent proposed grading scheme, Peña and colleagues 19 attempted to circumvent this by including all cell types in the assessment of the histological grade of CMTs. These authors described a canine adaptation of the human Nottingham histological grade (ca-NHG) system, which included modifications in the assignment of the parameters nuclear pleomorphism and mitotic count 19 (Suppl. Table S1). Recent studies demonstrated that ca-NHG provided independent prognostic information, 3,16,19 while others did not confirm that observation. 20 At this point, the number of CMTs series addressing the prognostic value of grading systems is relatively small, and to our knowledge, a comparative prognostic study of the NGH and ca-NHG is still missing. The aims of the present study were (1) to compare the WHO and 2011 histological classification systems in a prospective series of CMTs to identify advantages and limitations of each system when applied to diagnostic practice, (2) to evaluate the association of the histological diagnosis (performed according to each classification system) and histological grade (based on NHG and ca-NHG methods) with the overall survival, and (3) to evaluate the association between other relevant clinicopathological variables and survival.
Materials and Methods
Study Population and Histological Analysis
A cohort of female dogs with spontaneous CMTs diagnosed between January 2015 and July 2016 in the Veterinary Pathology Laboratory—ICBAS-UP at the University of Porto was prospectively included in this study. Dogs included in this study underwent surgery as the only treatment. All animals were treated with radical unilateral mastectomy (removal of an entire unilateral mammary chain) and/or partial mastectomy (removal of 1 to 3 glands, including the affected ones). Owners provided consent for surgery with curative intent, as well as the use of the material for research purposes. This protocol was approved by the institutional Ethics Committee (P151/2016).
Samples were immediately fixed in 10% buffered formalin after the surgery and routinely processed for histopathology. All the slides resulting from at least the largest transverse section of each tumor were screened (mean of 2 slides [range, 1–7] per benign tumor and mean of 4 slides [range, 1–18] per malignant tumor). All the regional and/or intramammary lymph nodes identified in the mastectomy specimens were sectioned (2 to 3 longitudinal sections), embedded, and submitted to histopathological examination. The presence of a malignant myoepithelial component was confirmed by immunohistochemistry using a panel of mouse monoclonal antibodies, including pancytokeratin (clone AE1/AE3 [Invitrogen, Camarillo, CA], 1:300 dilution, antigen retrieval in water bath with Target Retrieval Solution [Dako, Glostrup, Denmark]), vimentin (clone V9, Dako, 1:500 dilution, water bath with Target Retrieval Solution [Dako]), smooth muscle actin (clone HHF35, Dako, 1:300 dilution, water bath with Target Retrieval Solution [Dako]), and p63 (clone 4A4, Ventana, Tucson, AZ, ready-to-use, antigen retrieval in pressure cooker with sodium citrate buffer). The histological classifications and grades were assigned at the same time, blinded to survival outcomes. The histological diagnosis was established by consensus in a joint review by 3 pathologists (A.C., M.S., and P.D.-P.) using a multihead microscope, according to the WHO 18 and to the 2011 classification 9 systems. Histological grading was performed by consensus of the same 3 pathologists who classified the tumors, based on the criteria defined by the NHG 8 and by the ca-NHG method. 19 In the former, grading parameters were evaluated only in epithelial cells and sarcomas were excluded. Following the original publication, 19 inflammatory carcinomas, carcinosarcomas, and sarcomas were not graded by the ca-NHG method. The cutoffs for establishing the score of the mitotic counts of the NHG were adjusted to the high-power field (HPF) of the microscope (Nikon E600 Eclipse with a field diameter of 0.55 mm, HPF area of 0.237 mm2; Nikon, Tokyo, Japan), and the counting was performed in 10 HPFs that the observers judged to be the most mitotically active parts of the tumor (avoiding areas with necrosis and large amounts of stroma). This approach followed the current guidelines for human breast carcinomas. 7 Vascular invasion was defined as the presence of tumor emboli within endothelial-lined spaces without distinguishing between lymphatic and blood vessels. The lymph node was considered positive for metastases when 1 or more groups of 2 or more atypical, nonlymphoid and nonhematopoietic cells were seen in the subcapsular sinus or elsewhere within the node. In addition, the Nottingham Prognostic Index was adapted for use in CMTs from a human breast cancer study 11 and computed as veterinary-adapted Nottingham prognostic index, vet-NPI = (tumor size [cm] × 0.2) + NHG (1, 2, or 3, respectively, for grades I, II, and III) + evidence of vascular invasion and/or regional lymph node metastases (1 or 2, respectively, if absent or present), as previously described. 22 A similar index formula was computed but excluding the information about lymph node metastasis (veterinary-adapted, vascular-based Nottingham prognostic index vet-NPI-V).
Follow-up Study and Survival Data
The follow-up data were obtained by contacting the referring veterinarian and from the medical records. Disease-specific overall survival (OS) was calculated from the date of diagnosis to the date of the animal’s death/euthanasia due to the neoplastic disease. Animals that died or were euthanized for causes unrelated to the mammary tumor and those that were lost to follow-up were censored, respectively, at the time of death or at the time of their last clinical examination. In the subgroup of cases with multiple malignant tumors, a reference lesion was assigned for the survival study. Based on previous studies and evidence, the reference lesion was considered the tumor presenting peritumoral vascular invasion (primary criterion) or the one with the highest nuclear pleomorphism (secondary criterion) or, if necessary, the one with the largest diameter (tertiary criterion). 22 Necropsy was performed upon the owner’s consent.
Statistical Analysis
The concordance of the histological diagnosis in benign tumors according to the 2 classification systems was tested using the McNemar-Bowker test. Fisher exact method and χ2 test were used to assess the association between clinicopathological variables and number of disease-related deaths. Receiver operating characteristic (ROC) curves were used to assess the sensitivity and specificity of the vet-NPI and vet-NPI-V for predicting OS. Disease-specific survival curves were computed using the Kaplan-Meier product-limit estimates method, with log-rank (Mantel-Cox) tests being used to estimate the differences in survival fractions according to several clinicopathological parameters. Cox proportional hazard regression models were used to evaluate the prognostic role of the clinicopathological variables in univariable and multivariable analyses. Supplemental Table S2 describes the clinicopathological variables included in the regression analyses. Only the variables that proved to be significant in the univariable analysis were included in the multivariable regression models. Backward stepwise (Wald) selection was performed to arrive at the most parsimonious model. Cox regression coefficients were used to estimate the hazard of each independent variable of the model. The 95% confidence interval (CI) is given for all variables that figured in the final Cox regression model. The survival analyses were performed in the sample of cases with at least 1 malignant tumor for which follow-up data were available. All the analyses were performed using SPSS Statistics, version 24 (SPSS, Inc, an IBM Company, Chicago, IL). Statistical significance was set at 5%.
Results
Histopathologic Descriptive Findings
This study included 134 female dogs aged 5 to 18 years (mean, 9.9 years). In this cohort, 23 of 134 (17%) were spayed, 84 of 134 (63%) were nulliparous, and only 4 of 134 (3%) female dogs had been under contraceptive medication.
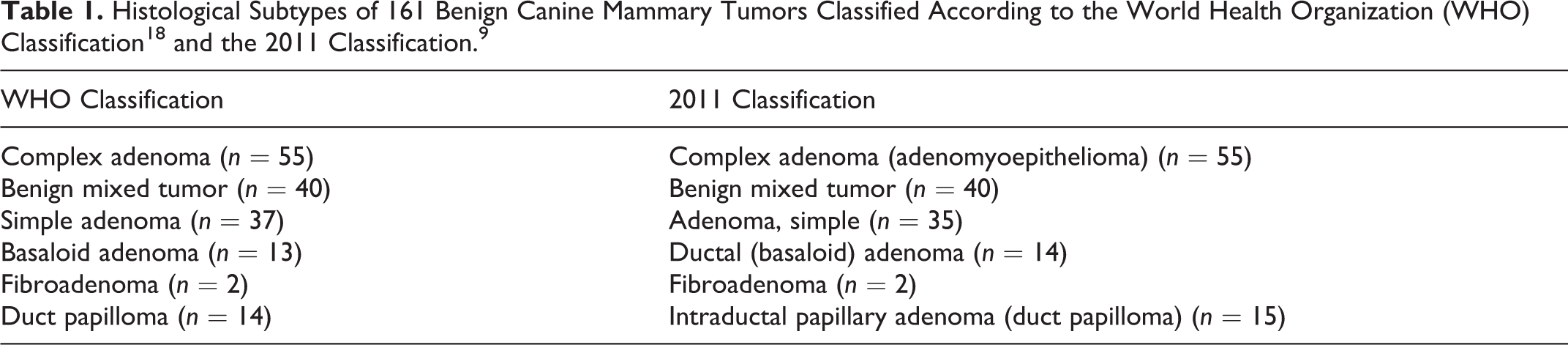
The number of cases assigned to each histological subtype, according to each classification system, is presented in Tables 1 and 2. In this cohort, multiple tumors were diagnosed in 80 of 134 (60%) of the cases. The proportion of benign and malignant tumors (161/305 [53%] vs 144/305 [47%], respectively) in this series was similar, irrespective of the classification system employed. According to both classification methods, the most frequent benign and malignant tumors were complex adenoma (55/161 [34%] in both classification systems) and complex carcinoma (49/144 [34%] and 23/144 [16%] according to the WHO and to the 2011 classification, respectively). Within the group of benign tumors, there were no significant differences between the 2 systems (Table 1). Only 2 WHO simple adenomas were reclassified by the 2011 classification, one as ductal (basaloid) adenoma and the other as intraductal papillary adenoma. One simple adenoma (WHO classification) was reclassified as ductal (basaloid) adenoma by the 2011 classification, as it presented slit-like lumina and bilayered cords with columnar luminal cells. Another simple adenoma was reclassified as intraductal papillary adenoma according to the 2011 classification, as multifocal intraductal growth with peripheral sclerosis and hyalinization was observed (features not described for duct papilloma in the WHO classification system).
Histological Subtypes and Grades of 144 Malignant Canine Mammary Tumors.a
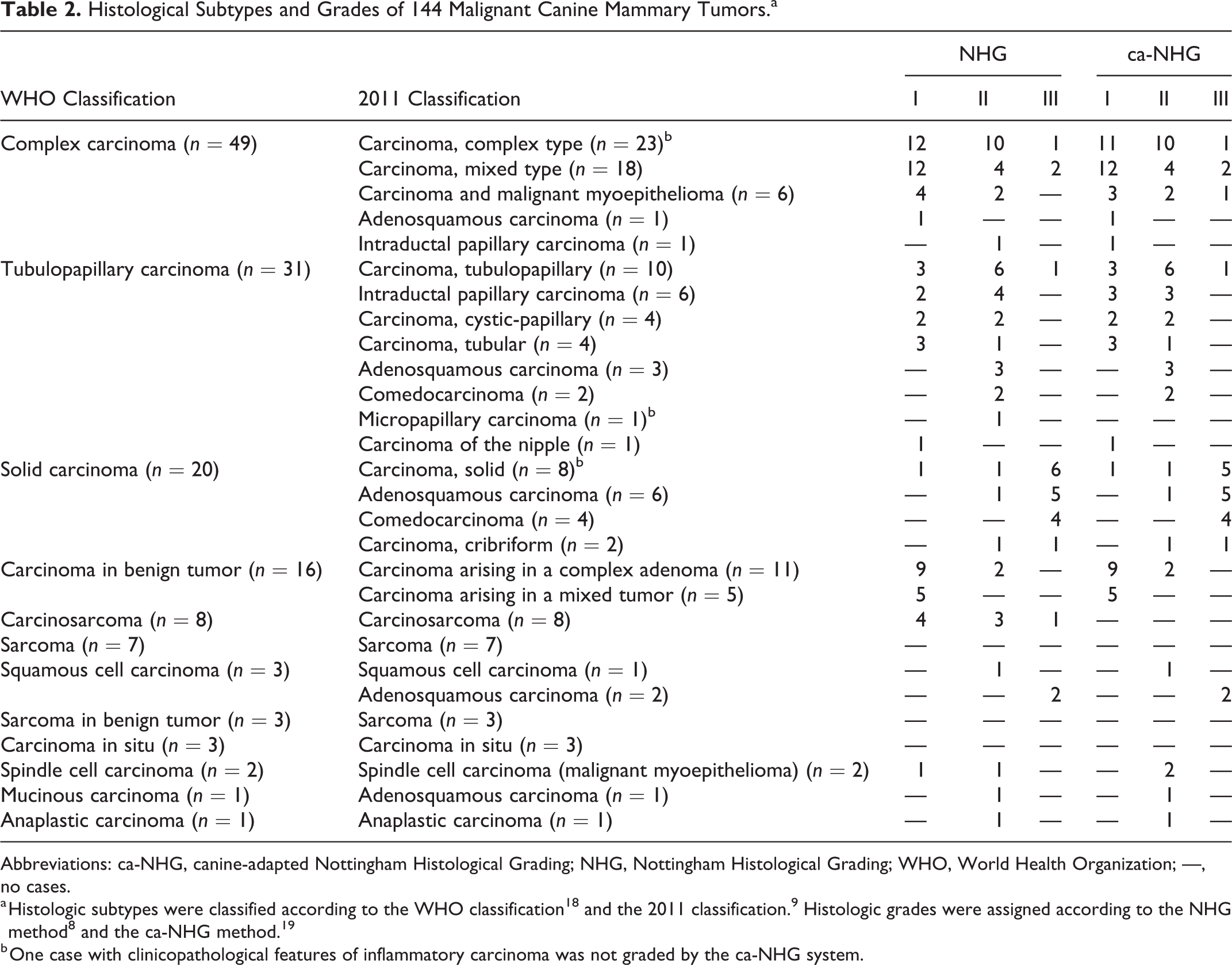
Abbreviations: ca-NHG, canine-adapted Nottingham Histological Grading; NHG, Nottingham Histological Grading; WHO, World Health Organization; —, no cases.
a Histologic subtypes were classified according to the WHO classification 18 and the 2011 classification. 9 Histologic grades were assigned according to the NHG method 8 and the ca-NHG method. 19
b One case with clinicopathological features of inflammatory carcinoma was not graded by the ca-NHG system.
In the group of malignant neoplasms, several differences in the histological diagnoses were found between the classification schemes (Table 2). Tumors diagnosed as complex carcinomas by the WHO system were divided by the criteria of the 2011 classification mainly into complex carcinoma, mixed carcinoma, and carcinoma and malignant myoepithelioma (Fig. 1a,b). Moreover, the 2011 classification subdivided the simple carcinomas into several subtypes: WHO tubulopapillary carcinomas were subdivided in 8 categories by the 2011 classification (Suppl. Figs. S1 and S2), and the majority of WHO solid carcinomas were divided into other categories (mainly adenosquamous carcinoma and comedocarcinoma) (Table 2, Fig. 2). The number of carcinomas in situ, carcinosarcomas, carcinomas arising in benign tumors, and anaplastic carcinomas was the same for both classifications. As the 2011 classification did not consider the category sarcoma arising in benign tumors (as the WHO classification did), these tumors were included in the sarcoma group. Another novel category of the 2011 classification was carcinoma of the nipple, and 1 case in this series was consistent with this entity. In addition, in 3 carcinomas (1 micropapillary, 1 solid, and 1 complex carcinoma), for which the clinician reported inflammatory signs, the histological hallmark of inflammatory carcinomas defined in the 2011 classification (ie, neoplastic emboli obliterating the dermal lymphatic vessels) was present.
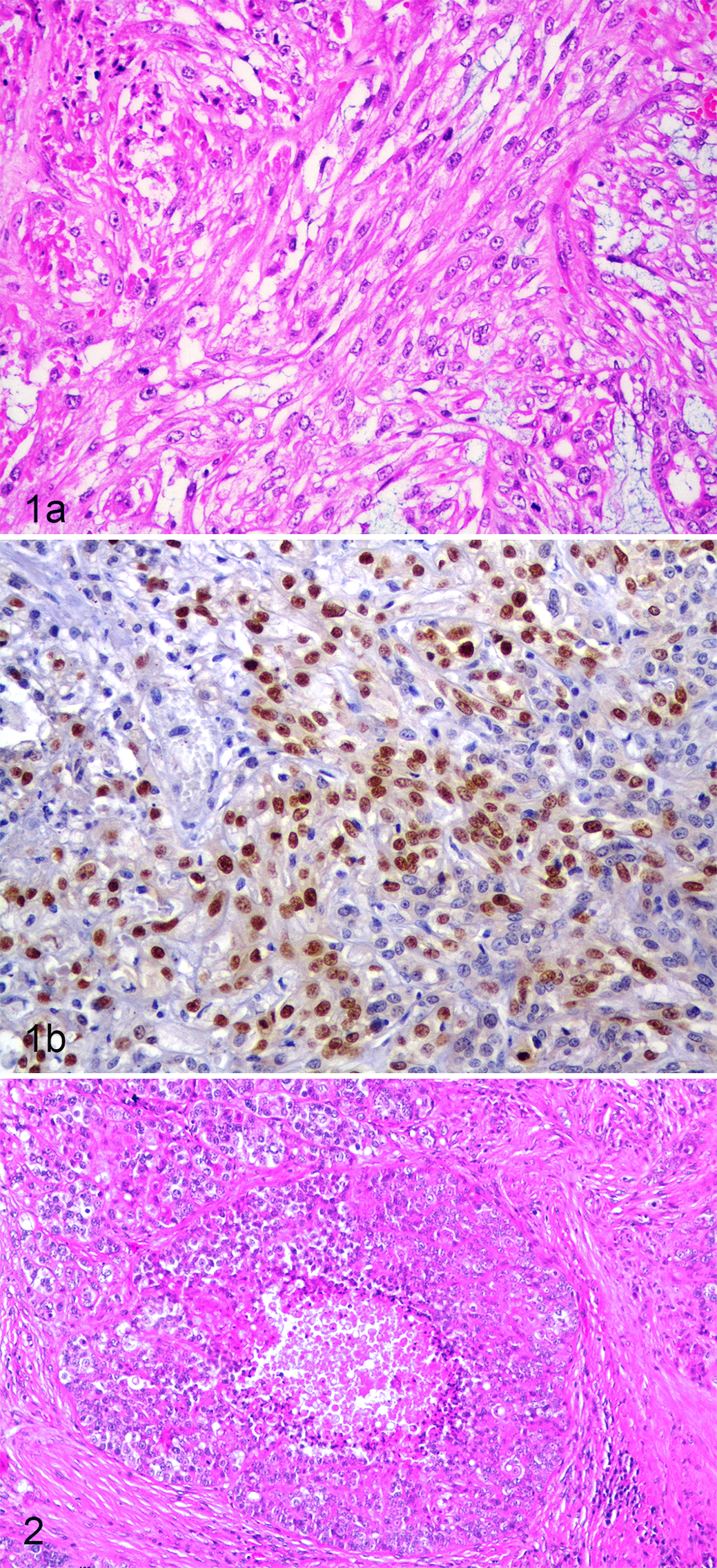
Carcinoma and malignant myoepithelioma, mammary gland, dog. (a) This malignant tumor, composed of epithelial cells arranged in tubules and fusiform cells, was classified as complex carcinoma by the World Health Organization classification. Hematoxylin and eosin (HE). (b) The fusiform cells display anisokaryosis, mitoses, and immunoreactivity for p63. Thus, the tumor was reclassified as carcinoma and malignant myoepithelioma by the 2011 classification criteria.
At the time of diagnosis, 14.6% (21/144) of malignant tumors showed peritumoral vascular invasion. One or more lymph nodes (inguinal and/or intramammary) were identified during the macroscopic pathological examination in 75 of 98 cases with at least 1 malignant tumor; of these, lymph node metastases were detected in 20 of 75 cases (26.7%).
Histologic Classification—Relationship to Survival
Two-year follow-up data were available for 85 bitches with at least 1 malignant tumor (53 of 85 had a single malignant tumor, with or without additional benign tumors). Cases fulfilling the criteria of inflammatory carcinoma of the 2011 classification were excluded from survival analysis regarding the histotype defined by the 2011 classification. Of the 85 cases, 26 of 85 (30.6%) died due to disease progression during the follow-up period (necropsy was performed in 4 cases and disseminated metastatic disease was confirmed histologically), 31 of 85 (36.4%) were alive 2 years after the surgery, 10 of 85 (11.8%) died of causes not related to the mammary disease, and 18 of 85 (21.2%) were lost to follow-up.
Both the WHO and the 2011 classification systems were significantly associated with tumor-specific OS (P < .001; Figs. 3, 4). According to the WHO classification, cases with complex carcinoma or with carcinoma or sarcoma arising in benign tumor had significantly better prognosis compared to all other subtypes (P = .002 and P = .05, respectively) (Fig. 5). None of the animals with a carcinoma or sarcoma arising in benign tumor died of neoplastic disease. On the contrary, all 5 animals with carcinosarcoma died of causes related to the disease (Fig. 6), and this subtype had a shorter OS (mean, 12.6 months; 95% CI, 5.6–19.6 months) compared to all other subtypes (mean, 20.0 months; 95% CI, 18.4–21.6 months; P < .001). When simple carcinomas (tubulopapillary, solid, and anaplastic) as a single group were compared to all other subtypes, no statistical association with OS was observed. Moreover, the subdivision of simple carcinomas into solid and tubulopapillary subtypes did not provide prognostic information. No difference was observed between the survival curves of cases with special types of carcinomas (squamous cell, mucinous, and spindle cell carcinomas) and cases with other types of malignant tumors.
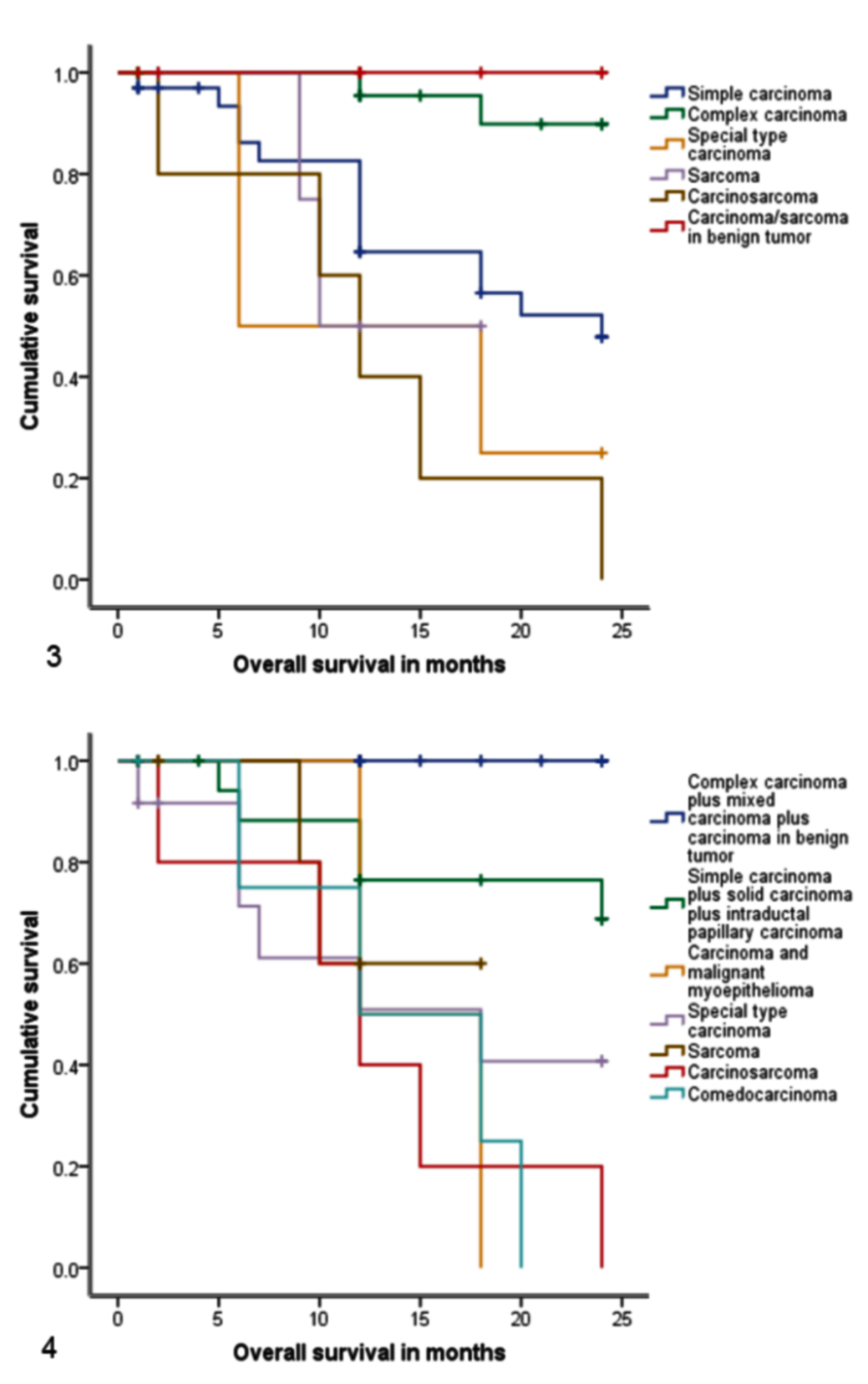
Kaplan-Meier overall survival curves according to histological classifications in 85 female dogs with malignant mammary tumors. Diagnoses of tumor type made in accordance with the World Health Organization classification were significantly associated with differences in survival. Compared to other categories, cases of carcinoma or sarcoma arising in benign tumors had significantly longer survival, and cases of carcinosarcoma had significantly shorter survival (P < .001). Vertical marks represent censored cases.
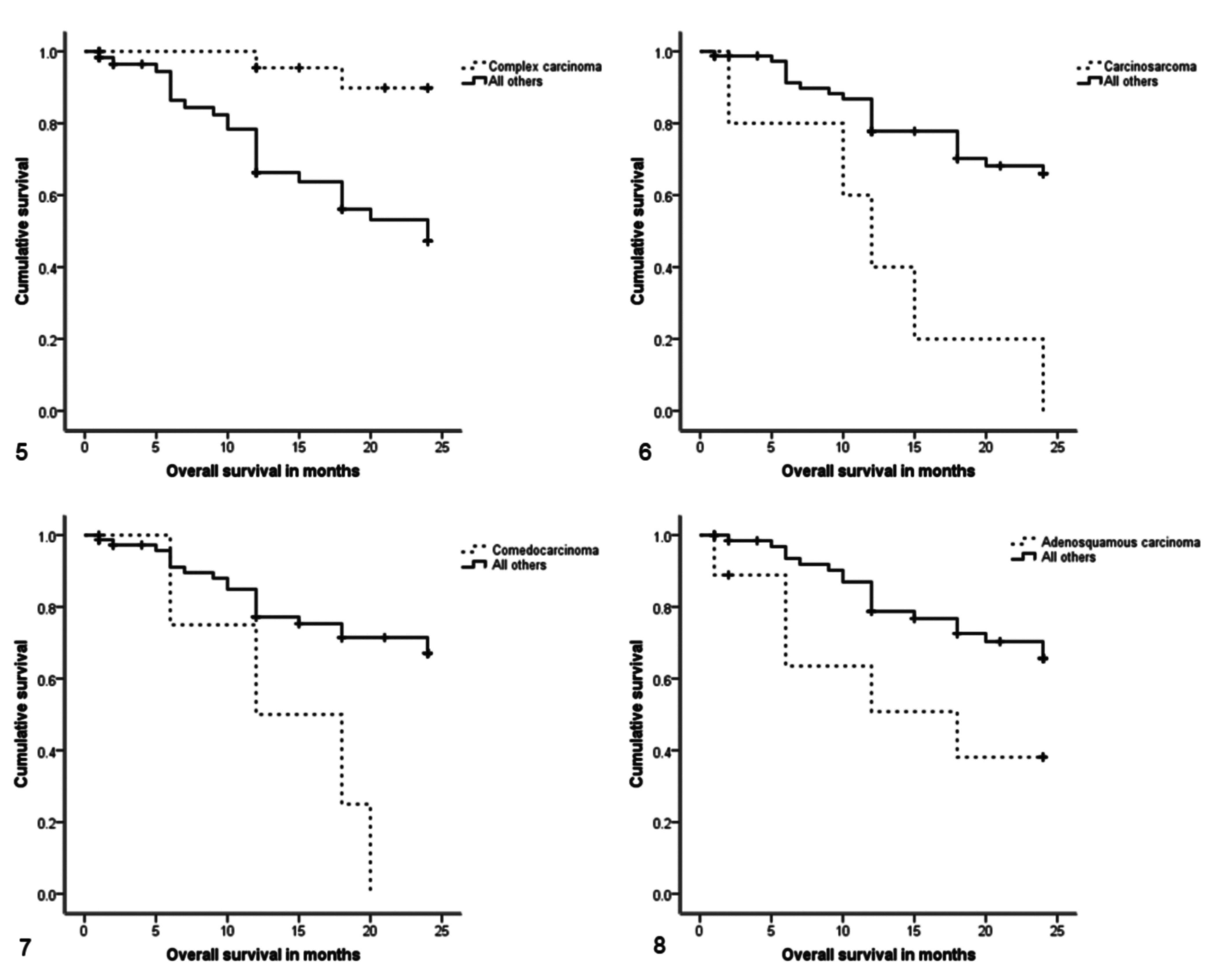
Regarding the 2011 classification system, the histological subtypes associated with better outcome (P < .001) were complex carcinoma, mixed carcinoma, and carcinoma arising in benign tumor; none of the animals with these tumors died of causes related to the neoplastic disease. On the other hand, carcinosarcoma, comedocarcinoma, and adenosquamous carcinoma had shorter survival (P < .001, P = .005, and P = .049, respectively) (Figs. 7, 8). As with the WHO classification, the OS of cases with simple carcinomas (including tubular, tubulopapillary, cystic-papillary, cribriform, and solid) and intraductal papillary carcinomas or cases with sarcomas (including osteosarcomas) was not significantly different from the survival of cases with other subtypes of malignant tumors.
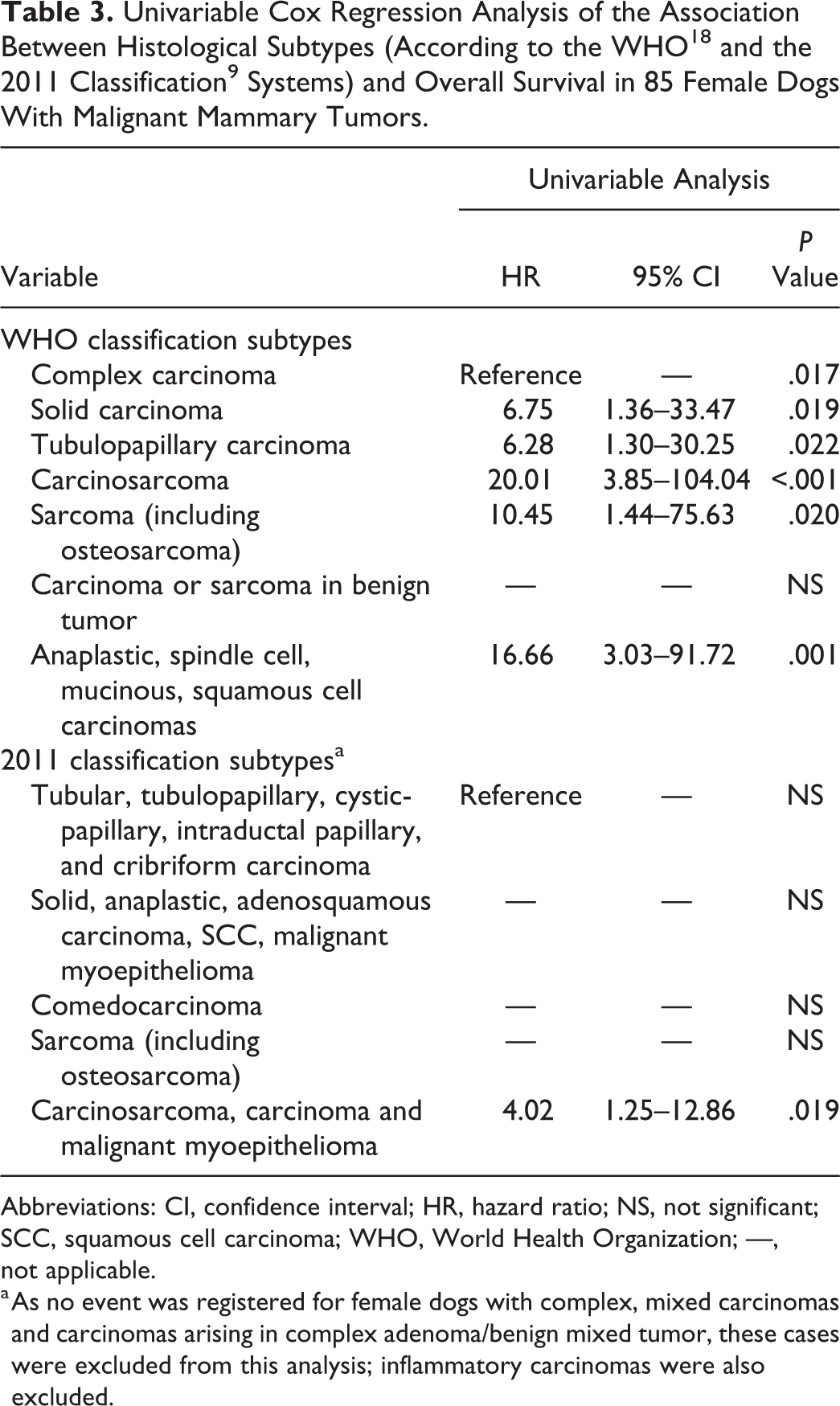
The association between the histological diagnosis and OS was also evaluated by univariable regression analysis. For that purpose, tumors were regrouped according to the established criteria of the original classification system and/or considering the results of the previously described survival analysis. The defined groups for each histological classification system and the results of the univariable regression analysis are presented in Table 3. In the WHO method, the complex carcinomas were assigned as reference for the computation of the hazard ratio, while in the 2011 classification, the reference lesion was the group of simple carcinomas plus intraductal papillary carcinomas (no event was registered for cases with complex carcinomas, mixed carcinomas, and carcinomas arising in complex adenoma/benign mixed tumor, so these cases were excluded from the regression analysis). The regression analysis confirmed the significant relationship between the WHO classification and OS, and carcinosarcoma was the subtype with the highest risk of disease-specific death. All other subtypes (except carcinomas or sarcomas arising in benign tumors) had a significantly higher risk of cancer-related death compared to complex carcinomas. For the 2011 classification, cases diagnosed with a carcinosarcoma or a carcinoma and malignant myoepithelioma had 4 times higher risk of disease-related death than cases with simple or intraductal papillary carcinomas. No difference was detected in the risk for dogs with other subtypes compared to simple or intraductal papillary carcinomas (Table 3).
Abbreviations: CI, confidence interval; HR, hazard ratio; NS, not significant; SCC, squamous cell carcinoma; WHO, World Health Organization; —, not applicable.
a As no event was registered for female dogs with complex, mixed carcinomas and carcinomas arising in complex adenoma/benign mixed tumor, these cases were excluded from this analysis; inflammatory carcinomas were also excluded.
Because of the low number of cases included in the present series, it was not possible to perform a meaningful analysis of the subtype-specific outcome of the following histotypes: anaplastic carcinomas, each type within the group of special-type carcinomas (either by the WHO or by the 2011 classification), as well as micropapillary invasive, ductal, and intraductal papillary carcinomas; carcinoma and malignant myoepithelioma; and each of the types of simple carcinomas defined by the 2011 classification.
Histologic Grade and Relationship to Survival
When the NHG and ca-NHG grading systems were applied to this series of 144 malignant tumors, there were no differences in the final grade or in the scores of each grading parameter in most cases. The few differences observed between the 2 grading systems were related mainly to mitotic count scores, less frequently to the nuclear pleomorphism scores, and also to the fact that, according to ca-NHG, malignant myoepitheliomas should always be assigned with score 2 in tubule formation. The nuclear pleomorphism scores by NHG and by ca-NHG were different in 2 complex carcinomas (reclassified as carcinoma and malignant myoepithelioma by the 2011 classification). The mitotic counts assigned by ca-NHG were lower in 6 cases compared to the score assigned by NHG, and in 2 cases, the scores assigned by ca-NHG were higher. After the sum of the scores, grade discrepancies were observed in 7 of 144 malignant tumors (4 complex carcinomas according to the WHO system, which were reclassified as carcinoma and malignant myoepithelioma by the 2011 classification, 1 complex carcinoma reclassified as intraductal papillary carcinoma, 1 tubulopapillary carcinoma reclassified as intraductal papillary carcinoma, and 1 spindle cell carcinoma reclassified as malignant myoepithelioma). Considering only the cases with follow-up data, the distribution of the grades according to the criteria of the NHG and ca-NHG methods is shown in Tables 4 and 5.
Relationship of Histologic Subtype According to the WHO Classification 18 and Histologic Grade According to the NHG and ca-NHG Methods, in Female Dogs With Malignant Mammary Tumors Included in the Survival Study.a
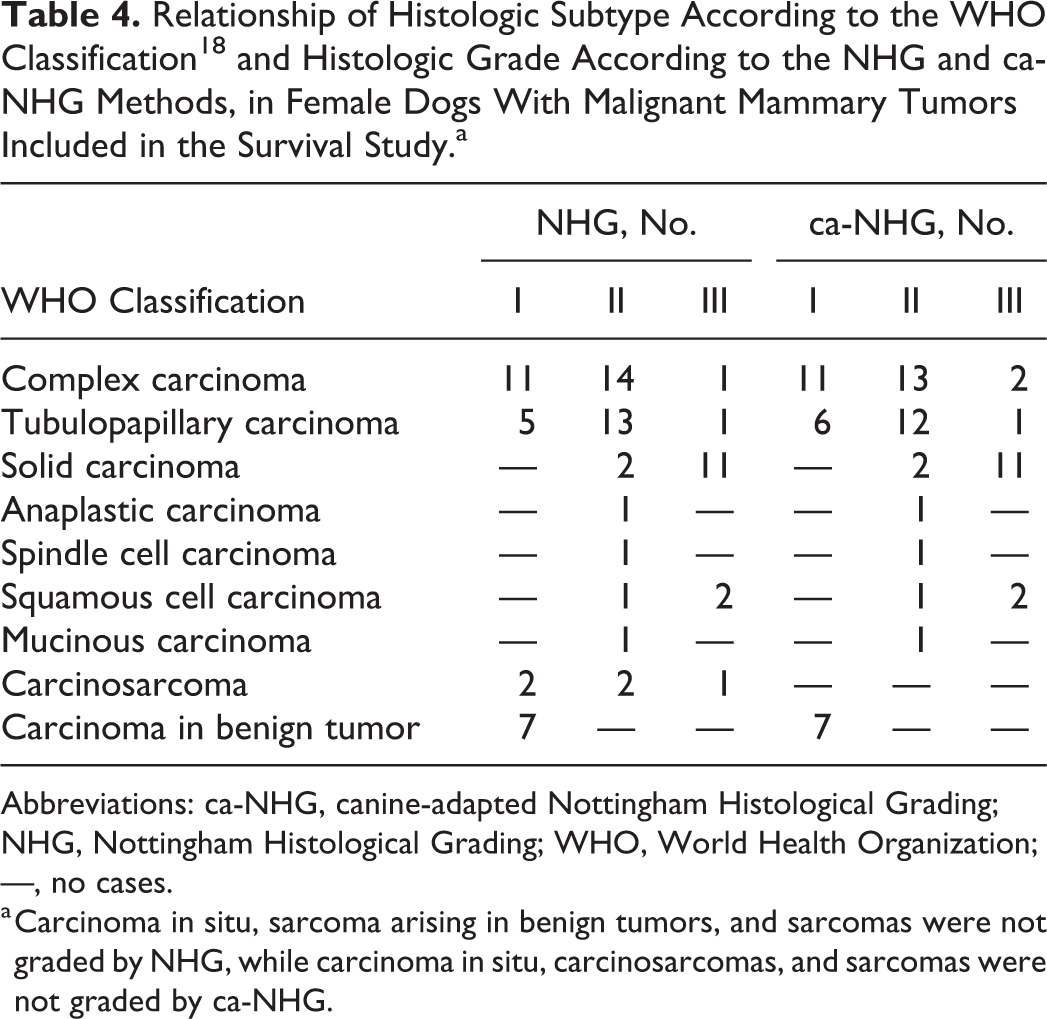
Abbreviations: ca-NHG, canine-adapted Nottingham Histological Grading; NHG, Nottingham Histological Grading; WHO, World Health Organization; —, no cases.
a Carcinoma in situ, sarcoma arising in benign tumors, and sarcomas were not graded by NHG, while carcinoma in situ, carcinosarcomas, and sarcomas were not graded by ca-NHG.
Relationship of Histologic Subtype According to the 2011 Classification Criteria 9 and Histologic Grade According to the NHG and ca-NHG Methods, in Female Dogs With Malignant Mammary Tumors Included in the Survival Study.a
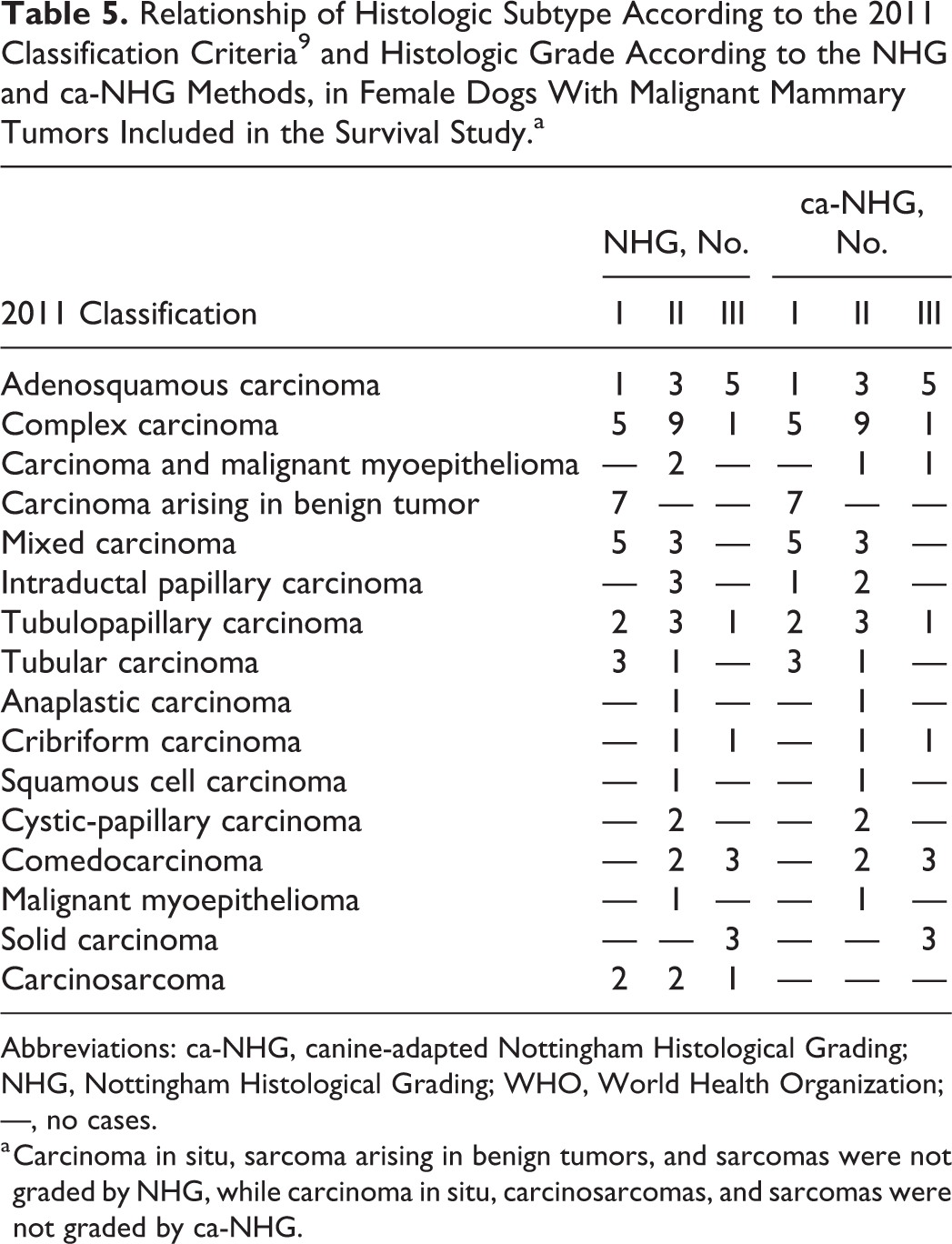
Abbreviations: ca-NHG, canine-adapted Nottingham Histological Grading; NHG, Nottingham Histological Grading; WHO, World Health Organization; —, no cases.
a Carcinoma in situ, sarcoma arising in benign tumors, and sarcomas were not graded by NHG, while carcinoma in situ, carcinosarcomas, and sarcomas were not graded by ca-NHG.
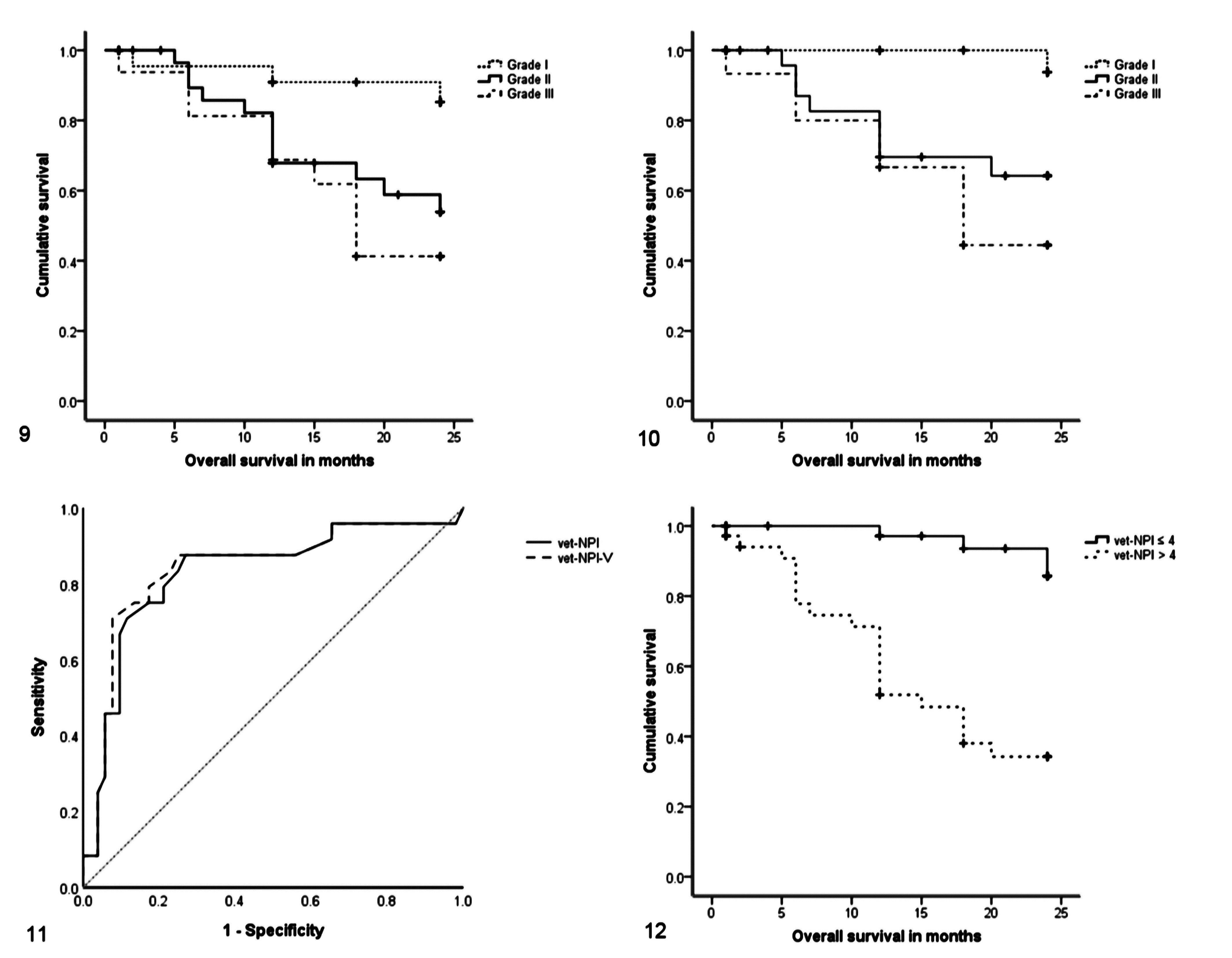
The histological grade was significantly associated with OS for both grading methods; OS was longer for cases with grade I tumors compared to cases with grade II or grade III tumors (P = .019 for NHG and P = .005 for ca-NHG) (Figs. 9, 10). The grading parameters of tubule formation and nuclear pleomorphism were related to the survival outcome, irrespective of the grading method employed (P = .036 for tubule formation by NHG and ca-NHG, respectively; P = .006 and P = .02 for nuclear pleomorphism by NHG and ca-NHG, respectively). Carcinomas with tubule formation scores of 1 or 2 had longer OS compared to those of score 3 (P = .026 for NHG and P = .012 for ca-NHG). Similar results were obtained for nuclear pleomorphism (P = .001 for NHG and P = .005 for ca-NHG). No association between mitotic count scores and OS was found for either grading method. The distribution of mitotic count scores was also analyzed within each subtype of tumors: the subgroup of simple, solid, and intraductal papillary carcinomas by the 2011 classification with mitotic activity scores of ≥2 was associated with a short OS. For all other histological subtypes, mitotic counts had no significant relation with OS.
Peritumoral vascular invasion was observed in 19 cases included in the survival study. Lymph nodes were available in 66 of 85 cases, and metastases were detected in 19 cases (16 of these also had vascular invasion, and 3 had metastases with no evidence of vascular invasion). Vascular invasion and lymph node metastases were both negatively associated with the OS in univariable regression analysis (Suppl. Table S2).
In the descriptive ROC curve analysis, the discriminative power of vet-NPI and vet-NPI-V for identifying tumor-associated death was high (area under the curve [AUC] = 0.83; 95% CI, 0.72–0.94, P < .001 for vet-NPI and AUC = 0.84; 95% CI, 0.73–0.95, P < .001 for vet-NPI-V) (Fig. 11). A cutoff of 4.0 for both indexes was selected for statistical purposes. This threshold allowed stratification of cases with significant differences in survival: cases with vet-NPI ≤4.0 had a mean OS of 23.4 months (95% CI, 22.5–24.3 months), whereas vet-NPI >4.0 had a mean and a median OS of 15.3 months (95% CI, 12.6–18.0 months) and 15.0 months (95% CI, 10.8–18.8) (P < .001), respectively; cases with vet-NPI-V ≤4.0 had a mean OS of 22.8 months (95% CI, 22.8–24.5 months), whereas vet-NPI-V >4.0 had a mean and a median OS of 15.1 months (95% CI, 12.4–17.7 months) and 15.0 months (95% CI, 11.2–18.8), respectively (P < .001) (Fig. 12).
In the final multivariable Cox models (Suppl. Table S2) after exclusion of nonsignificant variables, only age (cut-point of 12 years), surgical margins, vet-NPI, vet-NPI-V, and vascular invasion emerged as independent prognostic factors.
Discussion
This work represents the first comprehensive comparison of the WHO 18 and the 2011 classification 9 systems, as well as the NHG 8 and the ca-NHG 19 grading methods, performed in the same series of CMTs. Using both classification systems, the proportion of benign and malignant tumors was 4:4.5, which is in accordance with the existing literature. 10,17,23 The most frequent benign and malignant tumors were complex adenoma and complex carcinoma, respectively, independently of the classification scheme used, thus corroborating findings in other CMTs series. 19,20,24 Within the benign tumors, no significant differences were found between the 2 classification systems. Only 2 simple adenomas categorized by the WHO classification were reclassified differently by 2011 classification. This difference resulted from the fact that in the 2011 classification, the description of some benign tumor subtypes (including basaloid and intraductal papillary adenomas) was more detailed than that found in WHO classification, allowing the observer to appraise other subtle histological features presented in the tumor. As expected, the 2011 classification system subdivided malignant tumors in more categories compared to the WHO classification. This was particularly evident for simple carcinomas; for instance, in our series, the WHO tubulopapillary carcinomas and solid carcinomas were subdivided in 8 and 5 subcategories, respectively, according to the 2011 classification scheme. In our opinion, some of the simple carcinoma categories appeared subjective (such as tubular, tubulopapillary, or ductal carcinomas), while the diagnosis for others seemed relatively straightforward (such as comedocarcinoma). Despite this, in the 2011 classification system, there was no quantitative description of the proportion of specific morphological features, such as squamous or comedo areas, required for the diagnosis of adenosquamous carcinoma or comedocarcinoma. This limitation was also present in the WHO classification regarding other histological subtypes, such as mucinous carcinoma.
Herein, we confirmed the prognostic value of the WHO classification—namely, the better survival time in dogs with complex carcinomas and the worst outcome for those with carcinosarcoma, which is in accordance with findings from other authors. 3,18,21 However, in this series, it was not possible to support the association of mammary sarcomas with a worse prognosis 4,12 or the increased clinical aggressiveness of solid carcinomas described in other studies. 18 It is noteworthy that the carcinomas arising in benign tumors, diagnosed either according to the WHO or to the 2011 classification criteria, were not associated with disease-related deaths in this 2-year follow-up study. This is in line with a previous study 20 and reinforces the evidence that they resemble benign tumors in their biological behavior. However, it should be stressed that in both systems, the criteria for classification of a tumor as a carcinoma arising in benign tumor did not include a description of the number of foci and/or proportion of cells with malignant phenotype. Although further studies are needed to clarify the clinical value of this subtype of CMTs, we recommended that a comment about the expected benign clinical course of these tumors should be included in pathology reports.
Regarding the 2011 classification, the number of cases included in this series in some of the categories was too small to allow separate survival analysis. Considering the tumor categories with at least 5 cases, the 2011 classification clearly identified histological subtypes associated with a good prognosis (such as complex- and mixed-type carcinomas) and tumors associated with a poor survival (such as comedocarcinomas and carcinosarcomas), thus corroborating the findings of a recent study. 20 The 2011 classification scheme was more accurate than the WHO classification scheme, because it identified 2 subgroups (comedocarcinomas and adenosquamous carcinomas) within the WHO solid carcinoma category with a short survival time. Similarly, the 2011 classification scheme identified carcinoma and malignant myoepithelioma as distinctive from other WHO complex carcinomas, and this tumor type has been associated with a more guarded prognosis. 20 The present series included only 2 cases of carcinoma and malignant myoepithelioma with survival data, and both died of tumor-related causes during the follow-up period. This finding, suggests that the simultaneous proliferation of malignant epithelial and malignant myoepithelial cells increases the clinical aggressiveness of CMTs.
The histological grade (according to either the NHG or ca-NHG methods) was associated with OS, but the association was not independent, similar to the findings of some studies 20 –22 but contradicting others. 3,19 Variation in the interpretation of grading criteria (including interobserver variability) and different field diameters of the microscopes used in the different studies could have contributed to these discrepancies. To our knowledge, this is the first study where the 2 grading methods were applied to the same cohort of CMTs, and according to our findings, only slight differences existed between them. It is important to note that we excluded hyperchromatic nuclei for the assignment of the grading scores (although they were included in the evaluation of the nuclear pleomorphism of the ca-NHG method) because robust evidence has been built in human breast pathology that hyperchromatic nuclei represent apoptosis rather than mitosis. 7 In addition, the individual prognostic value of hyperchromatic/apoptotic nuclei in CMTs has never been identified, and a recent report failed to detect an association between the expression of apoptosis-related proteins and survival. 6 This exclusion might have contributed to underestimating the grade of some tumors, and this deserves clarification in further studies.
Interestingly, when histological grade, tumor size, and vascular/lymph node invasion were considered together in an index formula adapted from the validated human Nottingham Prognostic Index, 15 they showed an independent prognostic value. This finding confirmed a previous study of our group, where the index formula was adapted to CMTs for the first time. 22 Similar results were obtained when an alternative index formula including vascular invasion but not the lymph node status was computed. Based on these findings, we believe that the results of these prognostic index formulas could be routinely included in pathology reports, thus providing additional relevant prognostic data to clinicians. These index formulas should be validated in other prospective cohorts.
We also investigated the individual value of each of the grading parameters, a subject frequently neglected in prognostic studies of CMTs. In this series, tubule formation and nuclear pleomorphism were associated with OS for both grading methods—namely, when each parameter was classified as a 2-tiered system (score 1 or 2 vs score 3). As in the study by Carvalho and coworkers, 3 none of these parameters retained their association with survival in multivariable models. In addition, in this cohort, the mitotic counts had no prognostic value when all subtypes of tumors were considered. This observation confirmed previous findings from our group 21,22 but contradicted results of others. 3 Interestingly, mitotic activity seemed to be valuable for prognostication only for simple and for intraductal papillary carcinomas defined by the 2011 classification. Thus, the association of the grading parameters and prognosis indicates that they should be included in routine pathology reports of malignant CMTs, in addition to the overall histological grade that is more commonly reported.
In this study, age (<12 years) and the completeness of the surgical margins emerged as independent protective factors in CMTs, as previously reported. 1,12,23 This evidence reinforces the need for early diagnosis and a prompt and adequate surgical treatment. 1 As biologically expected, vascular invasion and the presence of lymph node metastases at the time of the diagnosis were relevant for prognostication in CMTs. However, only vascular invasion retained the prognostic value in the multivariable analyses, as previously reported. 22 The lower statistical significance of the association between lymph node metastasis and OS in Cox multivariable models may have resulted from missing data for this parameter in 19 cases. A similar trend was reported in other CMTs studies. 5,12,21
A limitation of this study was the relatively small number of cases, which precluded analyzing the association of some histological subtypes (especially those concerning the 2011 classification) with survival. Moreover, bias could have been introduced by obtaining the follow-up data from different clinicians. In addition, some cases enrolled in the survival study presented more than 1 malignant tumor, and only 1 tumor per animal was selected for the statistical analysis.
In conclusion, our findings demonstrated that the CMT classification systems were very similar in terms of the categorization of benign tumors, and both were prognostically relevant by identifying malignant tumors with a good prognosis and others with a high risk of cancer-related death. Large multi-institutional studies are warranted for evaluating the prognostic value of some categories in the 2011 histopathological classification system. Finally, it should be recommended that pathologists include the histotype defined by both classification systems in their routine reports of malignant CMTs. This will support clinicians and oncologists in their clinical and therapeutic decisions, as well as allow large, multi-institutional survival studies necessary for full standardization of the histological assessment of the CMTs.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818806968 - Canine Mammary Tumors: Comparison of Classification and Grading Methods in a Survival Study
Supplemental Material, DS1_VET_10.1177_0300985818806968 for Canine Mammary Tumors: Comparison of Classification and Grading Methods in a Survival Study by Ana Canadas, Miguel França, Cristina Pereira, Raquel Vilaça, Hugo Vilhena, Flora Tinoco, Maria João Silva, Jorge Ribeiro, Rui Medeiros, Pedro Oliveira, Patrícia Dias-Pereira, and Marta Santos in Veterinary Pathology
Footnotes
Acknowledgements
We thank the Veterinary Pathology Laboratory (ICBAS, University of Porto) for support, Prof. Paulo Cortez for support with the images, colleagues who had submitted samples for this work, and all the animals and owners involved in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.