Abstract
Newcastle disease (ND), caused by virulent strains of Newcastle disease virus (NDV), is a devastating disease of poultry worldwide. The pathogenesis of ND in quail is poorly documented. To characterize the ability of virulent NDV strains to replicate and cause disease in quail, groups of 14 two-week-old Japanese quail (Coturnix japonica) were experimentally inoculated with 108 EID50 (embryo infectious dose 50%) units of 1 of 4 virulent NDV strains: 2 isolated from quail (N2, N23) and 2 from chickens (Israel, Pakistan). At day 2 postinfection, noninfected quail (contact group) were added to each infection group to assess the efficacy of virus transmission. Tested NDV strains showed moderate pathogenicity, with highest mortality being 28% for the N2 strain and below 10% for the others. Two N2-inoculated birds showed neurological signs, such as head tremor and ataxia. Microscopic lesions were present in N2-, Israel-, and Pakistan-inoculated birds and consisted of nonsuppurative encephalitis. Contact birds showed no clinical signs or lesions. In both inoculated and contact birds, virus replication was moderate to minimal, respectively, as observed by immunohistochemistry in tissues and virus isolation from oropharyngeal and cloacal swabs. Strains originally isolated from quail resulted in higher numbers of birds shedding in the inoculation group; however, transmission appeared slightly more efficient with chicken-derived isolates. This study shows that virulent NDV strains have limited replicative potential and mild to moderate disease-inducing ability in Japanese quail.
Keywords
Newcastle disease (ND) is a devastating disease of poultry worldwide, responsible for millions in assets lost from the poultry industry due to morbidity, mortality, outbreak containment measures, and trade restrictions. 1,32 ND is caused by virulent strains of Newcastle disease virus (NDV) and is notifiable to the OIE (World Organisation for Animal Health). 36 NDV is an enveloped virus within the Avulavirus genus, Paramyoxoviridae family, Mononegavirales order. According to recent amendments by the International Committee on Taxonomy of Viruses, NDV has been renamed avian avulavirus (AAvV) 1. 6 However, for consistency with previous publications, the taxa name Newcastle disease virus was maintained in the present work. 32 NDV genome is composed of a single-stranded RNA molecule of approximately 15.2 kb, which contains 6 genes encoding for 6 structural proteins, from the 3′ to 5′, nucleoprotein (NP), phosphoprotein (P), matrix (M), fusion (F), hemagglutinin-neuraminidase (HN), and RNA-dependent RNA polymerase (L). 8,32 Since only virulent NDV strains cause ND, assessment of strain virulence is paramount for classification and regulatory purposes: The intracerebral pathogenicity index (ICPI) is the internationally recognized method to classify NDV virulence in vivo (see the following). 36
Although encompassed within a single serotype, NDV strains show marked genomic variability. 11,32 Phylogenetically, NDV strains are classified into 2 major groups: class I and II. 9,11 Class I is composed of a single genotype that typically includes nonvirulent strains isolated from waterfowl and shorebirds 11 (with 1 recorded exception 3 ). Class II is subdivided into 18 genotypes (indicated by Roman numerals) and multiple subgenotypes (indicated by lower case letters). 40 Genotypes III-IX and XI-XVIII encompass only virulent NDV strains. 11,28,32,40 Genotypes V, VI, VII, and VIII constitute the predominant genotypes currently circulating worldwide. 28 NDV strains of genotype VII have the widest worldwide distribution, and recently, NDV strains of novel subgenotype VIIi have been detected spreading rapidly through the Middle East (Israel), Asia (Pakistan, Indonesia), and Eastern Europe. 29,30 Viruses of genotype XIII have been detected mainly in Eurasia and Africa. Strains of subgenotype XIIIb were isolated in India and Pakistan during 2003 and 2013 and have been recovered predominantly from chickens, with the exception of a single strain recovered from Japanese quail. 13,30 Strains of genotype XVIIa have been isolated primarily from domestic poultry in West and Central Africa during 2006 to 2011. 40 Experimental challenges have confirmed that representative strains of subgenotype VIIi, XIIIb, and XVIIa are highly virulent for chickens. 38,41 Genotype VI includes strains highly adapted to Columbiformes species (referred to as pigeon paramyxovirus), 32 and subgenotype VIh was first identified in a pigeon in Argentina in 1997 and subsequently isolated from pigeons and doves in Kenya and Nigeria during 2007 to 2013. 13 These isolates can be virulent for chickens, especially after multiple passages. 15,23
Based on the severity of clinical signs elicited in infected chickens, NDV strains are divided (from least to most virulent) into asymptomatic enteric (avirulent), lentogenic, mesogenic, and velogenic. 7,32 Velogenic strains cause up to 100% morbidity and mortality in naïve chickens and can be divided into viscerotropic (velogenic viscerotropic NDV, VVNDV) and neurotropic (velogenic neurotropic NDV, VNNDV) based on their ability to cause necro-hemorrhagic lesions in the intestine or encephalitis, respectively. 7,32 Mesogenic strains cause respiratory and nervous signs, with moderate morbidity and mortality (up to 50%). Lentogenic strains usually cause minimal disease; however, respiratory disease can be observed in young chicks, especially if secondary bacterial infection is superimposed. 7,17,32
The ICPI is the internationally accepted method to classify the virulence of NDV isolates. 36 This method is conducted by scoring groups of 10 one-day-old infected chicks as dead, sick, or normal over an 8-day observation period. ICPI scores range from 0.00 to 2.00, with strains scoring ≥0.7 considered virulent and notifiable to the international community. 36 Velogenic strains have scores ≥1.5 and are considered highly virulent. 4 Sequencing of the F gene to deduct the amino acid sequence of the cleavage site is another method to classify the virulence of NDV strains. 36 NDV strains with multiple basic amino acid residues between position 113 and 116 plus a phenylalanine at position 117 of the F protein are considered to be virulent. 32,36 This polybasic configuration allows cleavage of the F protein by ubiquitous intracellular proteases, leading to systemic virus activation and multi organ infection. 44
NDV strains can infect over 200 species of birds. 7,32 Chickens, turkeys, pheasant, and other gallinaceous species are most severely affected by ND, 2,7,43 whereas ducks, although susceptible to infection, appear to be highly resistant to disease development. 7,35 Other species, such as pigeons and geese, have an intermediate susceptibility. 19,45,46
The pathogenesis of ND in quail is poorly understood, and few studies document the clinico-pathological features of NDV infection in this species. Description of a natural ND outbreak in a multispecies farm in northern India reported morbidity and mortality in 22-week-old Japanese quail (Coturnix coturnix japonica) of 20% and 30%, respectively, with central nervous signs and nonsuppurative encephalomyelitis predominating. 21 A velogenic NDV strain was isolated; however, no additional information was provided. 21 In at least 4 studies, experimental infection of 3- to 6-week-old Japanese quail with velogenic NDV strains showed partial success in reproducing disease using the oculo-nasal (mortality, 13%), 33 intracelomic (mortality, 25%), 18 and intramuscular (mortality, 3% and 40%) 37,39 routes. Clinical signs varied and included generalized weakness, 18,33,37,39 nervous, 37,39 and respiratory signs. 18,33 Reported gross lesions (not confirmed histologically) consisted of diffuse congestion of muscles (mainly pectoral and thighs) and splanchnic organs (liver, spleen), hemorrhagic proventriculitis, and enteritis. 18,33,37,39 In the only study 18 providing histopathological assessment, reported microscopic lesions included pneumonia, lymphoid depletion, and degeneration/necrosis of multiple organs (heart, liver, kidney) in intraperitoneally infected quail. In partial disagreement with these reports, an additional study showed that inoculation of 17-week-old Japanese quail with a velogenic NDV strain via the oculo-nasal route resulted in no morbidity or mortality. 25 In 2 of the aforementioned 5 studies, NDV isolation was attempted from cloacal swabs. In 1 study, 33 isolation was successful in 9 of 15 infected birds, with the last 2 positive samples identified at 14 days postinfection (dpi). The other study 25 did not report the number of birds actively shedding the virus; however, it seems that at least 1 bird (of 12) was positive at 5 and 14 dpi.
Although the studies discussed previously documented some clinico-pathological features of Japanese quail infected with NDV strains, there are several limitations to their experimental approach, including overall lack of systematic histopathological analysis, immunohistochemistry, and virus titration to evaluate the magnitude of virus replication/shedding. In addition, these studies included no information regarding the genetic classification (ie, genotype) of the strains used for inoculation, which may affect the pathogenesis. Further, clinical signs and lesions were reproduced mostly through inoculation of birds via parenteral administration (intramuscular, intracelomic), which is not a natural route of infection. 32 When NDV was inoculated through the oculo-nasal or oral routes, mortality, as reported in 3 of those studies, ranged from 0% 25,37 to 13%. 33
To better understand the pathogenesis and disease-inducing ability of virulent NDV strains in Japanese quail and clarify some conflicting reports in the literature, we inoculated groups of 2-week-old Japanese quail through the oculo-nasal route with 4 virulent NDV strains of different genotypes. We adopted a systematic approach to determine clinical signs, gross and histopathological lesions, and the extent and magnitude of NDV replication as assessed by virus isolation in mucosal secretions and immunohistochemistry in tissues. Further, transmission potential was assessed by contact transmission trials with naïve quail.
Materials and Methods
Viruses
The following 4 NDV strains were used for this experiment:
NDV/quail (Coturnix Japonica)/Nigeria/VRD17/N2/861/2004, referred here as N2 strain. This strain was isolated from quail in Nigeria in 2004 and classified within genotype XVIIa.
14
NDV/quail (Coturnix Japonica)/Nigeria/VRD08/385/N23/870/2008, referred here as N23 strain. This strain was isolated from quail in Nigeria in 2008 and classified within genotype VIh (data not shown).
NDV/broiler/Israel/KY50/826/2013, referred here as Israel strain. This strain has been classified within genotype VIIi
30
and was previously shown to have ICPI = 1.85 and be virulent for chickens in a pathogenesis experiment.
38
NDV/chicken/Pakistan/SPVC/Karachi/33/2007, referred here as Pakistan strain. This strain has been classified within genotype XIIIb
30
and was previously shown to have ICPI = 1.85 and be virulent for chickens in a pathogenesis experiment.
38
All strains were obtained from the repository of the Southeast Poultry Research Laboratory (SEPRL, Agricultural Research Service, ARS-USDA). Each virus was initially purified by limiting dilutions and propagated in 9- to 10-day-old specific pathogen-free (SPF) embryonating chicken eggs through chorioallantoic inoculation, as per standard procedure. 26
Birds
A total of 130 unvaccinated Japanese quail (Coturnix coturnix japonica) were obtained from a commercial farm and delivered to SEPRL at 11 days of age. Quail were housed in negative pressure isolators, with 14, 8, or 6 birds per cage (see experimental design) and acclimated for 3 days before beginning the experiment (14 days of age). Upon arrival, blood samples from 4 extra quail were drawn, and serum was evaluated using the hemagglutination inhibition (HI) test against LaSota NDV strain to ensure that birds were immunologically naïve for NDV. 5,26
Chicken eggs for virus propagation and titration as well as 1-day-old chickens for ICPI tests were obtained from the SEPRL SPF White Leghorn flock. Quail and day-old chickens were housed at the facilities of the SEPRL under BSL-3 enhanced agriculture conditions. Food and water were provided with ad libitum access, and birds were checked daily. The experiment was approved by and performed under the regulations of the SEPRL Institutional Animal Care and Use Committee.
ICPI Test
An ICPI test was conducted for N2 and N23 NDV strains as prescribed by the OIE. 36 Briefly, for each test strain, 10 one-day-old SPF chickens were inoculated intracranially with 0.05 ml of a 1:10 dilution of infective allantoic fluid in phosphate buffered saline (PBS). Over an 8-day period, birds were observed daily to score clinical signs as follows: normal = 0, sick or paralyzed = 1, dead = 2. The ICPI for each group was calculated as the average of each bird’s score over the observation period. 36 The ICPI score for the Israel and Pakistan strains was previously reported. 38
Experimental Design
The experimental design is outlined in Supplemental Fig. S1. Birds (n = 126) were randomly assigned into 1 of 3 experimental groups as follows: (1) inoculation group: 4 cages with 8 birds each and 4 cages with 6 birds each (n = 56, 8 cages); (2) contact group: 4 cages with 14 birds each (n = 56, 4 cages); and (3) control group: 2 cages with 8 or 6 birds each (n = 14, 2 cages). For the inoculation group, birds from one 8-bird cage and one 6-bird cage (n = 14) were inoculated with either N2, N23, Pakistan, or Israel NDV strains. Each bird was infected with 108 embryo infectious dose 50% (EID50) units of virus diluted in 0.1 ml of Brain Heart Infusion broth (BHI), half (0.05 ml) delivered in the left conjunctival sac and half (0.05 ml) in the nasal cavity via the choanal slit. Birds that served as negative control were inoculated with BHI alone.
At 2 dpi, oropharyngeal and cloacal swabs were collected from inoculated birds. After swab collection, naïve birds from each cage in the contact group were transferred to cages housing the inoculated birds as follows: 8 contact birds were transferred to the cage with 8 inoculated birds, and 6 contact birds were transferred to the cage with 6 inoculated birds. This resulted in 2 sets of cages (containing 12 and 16 birds per infection group, or 6 and 8 birds for control) and a ratio of inoculated to contact bird of 1:1 (Fig. S1). Birds for necropsy were sampled from the cages containing 16 birds. In the cages with 12 birds, necropsies were not performed to assess morbidity and mortality. Swabs were collected from all birds regardless of cage assignment.
At 2 dpi (right after transfer), 2 inoculated birds per group were euthanized (sodium pentobarbital, 100 mg/kg intravenous) for pathology (postmortem and collection of tissues for histopathology). At 4, 7, and 10 dpi (corresponding to 2, 5, 8 days postcontact [dpc]), all birds (from both sets of cages) were swabbed (oropharyngeal and clocal), and 2 to 3 inoculated and 2 contact birds (from the cages with 16 birds) were euthanized for pathology. Birds in the control group were sampled for pathology from both cages. Birds unable to move, eat, or drink were euthanized regardless of the sampling schedule or cage, and counted as dead for the next day.
Histopathology
The following tissues were collected for histopathology: eyelid, turbinates, pharynx, trachea, lung, esophagus, proventriculus, duodenum and pancreas, Meckel’s diverticulum, large intestine, cecal tonsils, heart, kidney, liver, brain, spleen, bursa, thymus, and head of femur. Tissues were fixed in 10% neutral buffered formalin for 52 hours and then transferred in 70% ethanol solution before being routinely processed onto positively charged slides for hematoxylin and eosin (HE) staining and immunohistochemistry (IHC). The femur and turbinates were decalcified in Cal-Ex solution (Fisher Scientific, Ottawa, Canada) for 12 hours prior to processing. Microscopic lesions were graded semiquantitatively in 3 distinct encephalic areas, including forebrain (telencephalon and thalamus), brainstem (midbrain and hindbrain), and cerebellum, 16,34 as well as in the intestinal ganglia present in sections of the proventriculus, ventriculus, and intestine.
Immunohistochemistry
IHC was performed to assess NDV replication in tissues and was carried out using the Novolink detection system kit (Leica, Newcastle, UK), as previously described. 34 The primary antibody was a mouse monoclonal antibody directed against ribonucleoprotein of LaSota NDV strain (Novus Biologicals, Littleton, USA), diluted 1:7500 in PBS with added 0.1% Tween 20 (PBS-T) and applied to tissues overnight at 4°C. Antigen retrieval was carried out by microwaving tissues for 5 minutes in 10% sodium citrate solution, followed by a 10-minute peroxidase and protein blocking steps (solutions provided with the kit). Signal was detected using 3,3′-diaminobenzidine (DAB) substrate. A formalin-fixed, paraffin-embedded spleen from a chicken previously inoculated with the Israel strain 38 was used as positive control for the technique. To assess the specificity of the antibody and the extent of nonspecific labeling, mouse serum (Leica, Newcastle, UK) was used instead of the primary antibody (isotype control) to optimize the technique and assess presence of nonspecific staining.
Virus Isolation and Titration of Shed Viruses
Oropharyngeal and cloacal swabs were collected and stored separately in tubes containing 1.5 ml of BHI with antibiotics (2000 U penicillin G/ml, 200 µg gentamicin sulfate/ml, 4 µg amphotericin B/ml; Sigma Chemical Co., St. Louis, MO). For processing, tubes were vortexed, left to settle, and the supernatant used for virus isolation (VI) and titration. VI and titration of positive samples were conducted in 9- to 10-day-old SPF embryonating chicken eggs, as previously described. 5,26 Titers were expressed as EID50/0.1 ml. 22
Statistical Analysis
Viral titers of oropharyngeal and cloacal swabs for inoculated and contact birds were analyzed separately using a 2-way ANOVA (variables: virus strain, timepoint) with multiple comparisons between groups (Tukey’s test). When possible, analyses were performed both including and excluding negative samples in the calculation of the group means (to assess differences considering all birds or only those actively shedding).
For the inoculated cohort, oropharyngeal swabs were considered for statistical analysis only at 2 and 4 dpi due to the high numbers of negative samples at later timepoints. For cloacal swabs, all timepoints (2, 4, 7, 10 dpi) were included. For the contact cohort, all timepoints (2, 5, 8 dpc) and negative samples were included in calculation of the group means as a test without negative samples could not be performed (it would have left entire groups without data, preventing multiple comparisons).
Contingency tables for both oropharyngeal and cloacal swabs were created to compare fractions of positive swabs between groups at 2 and 4 dpi in both inoculation and contact cohorts. Contingency tables were analyzed using the chi-square test (2 × 4 tables) and the Fisher’s exact test (2 × 2 tables).
In all calculations, samples that were positive by virus isolation but had titers below the sensitivity threshold (<100.5 EID50/0.1 ml) were tallied as 100.5 EID50/0.1 ml. All tests were considered statistically significant for P values <.05. Statistical analysis was carried out using GraphPad Prism for iOS, version 7 (GraphPad Software, La Jolla, USA).
Results
ICPI Test
ICPI scores for N2 and N23 strain were 1.96 and 1.76, respectively. The ICPI values for both Israel and Pakistan strains had been previously reported to be 1.85. 38 Based on these ICPI values, all 4 strains are virulent for chickens and can be considered velogenic strains. 4
Clinical and Gross Findings
A summary of mortality and clinical and gross findings is reported in Table 1. Gross lesions were observed only in a few birds throughout the experiment, were nonspecific, and consisted of multifocal hemorrhages in the eyelid and trachea and enlarged spleen (Table 1). Morbidity and mortality are reported in detail in the following.
Summary of Clinical and Gross Findings in 2-Week-Old Japanese Quail Infected With 4 Virulent Newcastle Disease Virus (NDV) Strains.
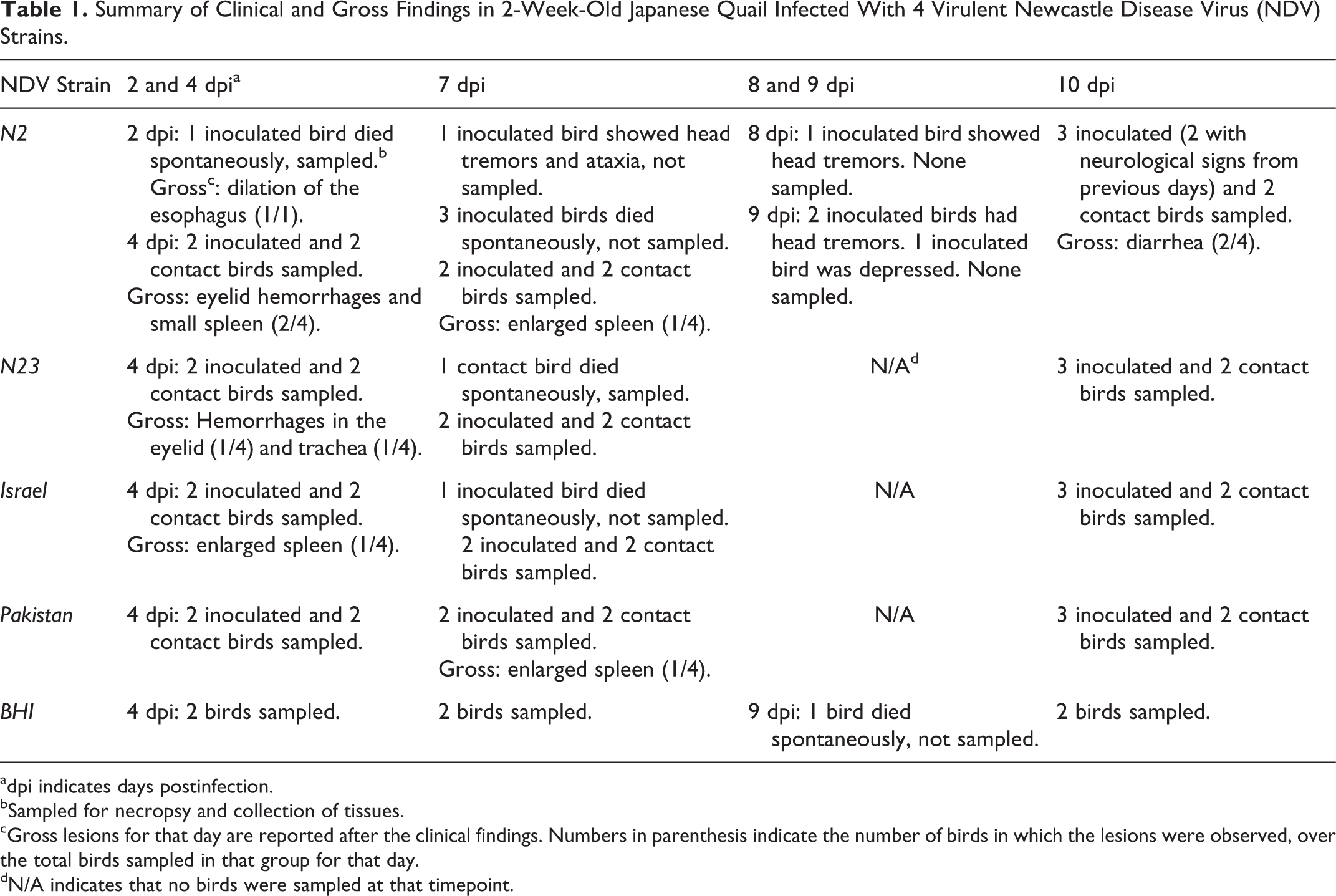
adpi indicates days postinfection.
bSampled for necropsy and collection of tissues.
cGross lesions for that day are reported after the clinical findings. Numbers in parenthesis indicate the number of birds in which the lesions were observed, over the total birds sampled in that group for that day.
dN/A indicates that no birds were sampled at that timepoint.
Inoculated birds
Considering both sets of cages, clinical signs were only observed in 3 of the N2-inoculated birds (3/14 initially inoculated birds; 21.4%) and consisted of head tremor and ataxia (n = 2; 1 at 7 and 1 at 9 dpi) and depression (n = 1; 9 dpi). Four (n = 4; 28.5%) and 1 (n = 1; 7.1%) dead birds (not euthanized) were recorded in the N2 and Israel inoculation groups, respectively. At 2 dpi, 1 N2-inoculated bird was found dead, showing dilated esophagus filled with ingesta. At 7 dpi, 3 N2- and 1 Israel-inoculated bird were found dead in the cage (not sampled for necropsy). No mortality was observed with the N23- and Pakistan-inoculated birds. Based on the cohort of birds that were not actively sampled for necropsy (cages with only 6 inoculated birds, Fig. S1), morbidity and mortality were 50% (n = 3, starting at 2 dpi) and 16.7% (n = 1, at 7 dpi) for the N2 inoculation group, respectively, and 0% for the other 3 groups.
Contact and control birds
One N23 contact and 1 control bird were found dead at 5 dpc and 9 dpi, respectively. Both birds had tested negative for virus isolation in oropharyngeal and cloacal swabs. No other deaths were recorded throughout the experiment in the contact and control groups.
Histopathology
In total, 60 birds were submitted for histopathology: 29 birds from the inoculation group, 25 from the contact group, and 6 control birds. Most significant lesions consisted of nonsuppurative encephalitis, which was observed in 2 N2- and 1 Israel-inoculated bird at 7 dpi and in 3 N2-, 2 Israel-, and 1 Pakistan-inoculated birds at 10 dpi (Table 2). Affected areas were randomly distributed to the forebrain, brainstem, and cerebellum, without obvious predilection for 1 specific site (Table 2). Lesions were characterized by multifocal, randomly distributed perivascular cuffs of inflammatory cells composed mainly of lymphocytes and fewer macrophages that spilled into the adjacent tissue and mild to moderate endothelial hypertrophy of affected vessels (Fig. 1). In most severely affected areas, neuroparenchyma showed neuronal necrosis, multifocal gliosis, and vacuolation of the neuropil (Fig. 2). In the cerebellum, there was segmental loss of Purkinje cells, perivascular accumulation of inflammatory cells that infiltrated the adjacent tissue, and multifocal gliosis of the molecular layer (Fig. 3). In 4 birds (1 N2- and 3 Israel-inoculated birds, Table 2), there was mild to moderate inflammation of the nervous ganglia throughout the proventriculus, ventriculus, and sections of intestines (Fig. 4) as well as inflammation of the splanchnic nerves (Fig. 5). Microscopic lesions were not present in the nervous tissue of N23-inoculated birds or any of the contact or control birds (Supplemental Figs. S2, S3). Overall, the most severe and extensive lesions were observed in N2-inoculated birds, both at 7 and 10 dpi. Presence of nervous lesions did not appear to be associated with the magnitude of shedding as several birds showing no lesions had positive oropharyngeal or cloacal swabs at 4, 7, and 10 dpi (Table 2). All birds showing nervous lesions tested positive by virus isolation at least at 1 timepoint, with the exception of 1 Israel- and 1 Pakistan-infected quail that showed no positive virus isolation results.
Semi-Quantitative Scoring of Microscopic Lesions in the Brain and Intestinal Ganglia of 2-Week-Old Japanese Quail Inoculated With 4 Virulent NDV Strains (Scoring Is Reported Individually for Each Inoculated Bird at 7 and 10 dpi).
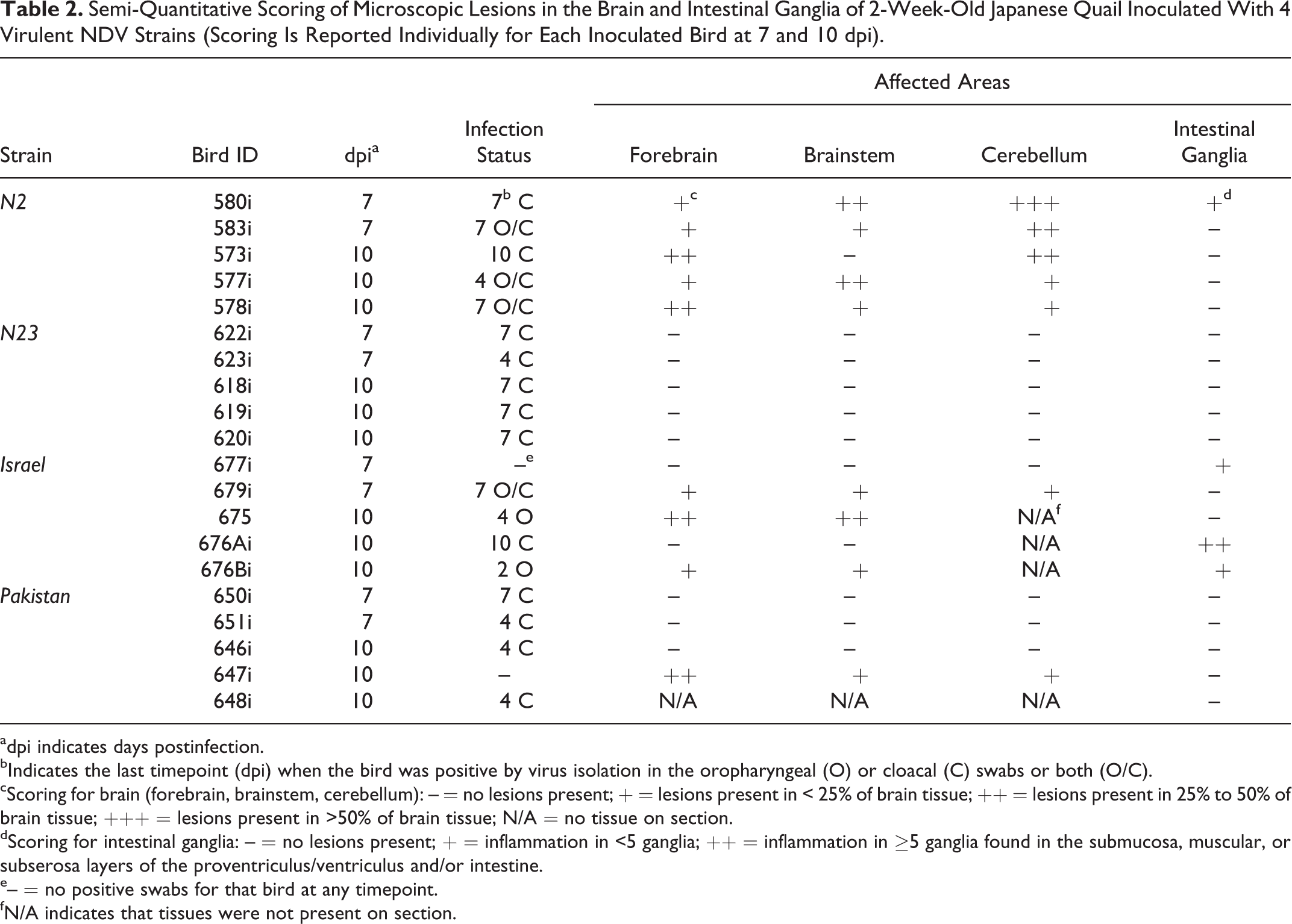
adpi indicates days postinfection.
bIndicates the last timepoint (dpi) when the bird was positive by virus isolation in the oropharyngeal (O) or cloacal (C) swabs or both (O/C).
cScoring for brain (forebrain, brainstem, cerebellum): – = no lesions present; + = lesions present in < 25% of brain tissue; ++ = lesions present in 25% to 50% of brain tissue; +++ = lesions present in >50% of brain tissue; N/A = no tissue on section.
dScoring for intestinal ganglia: – = no lesions present; + = inflammation in <5 ganglia; ++ = inflammation in ≥5 ganglia found in the submucosa, muscular, or subserosa layers of the proventriculus/ventriculus and/or intestine.
e– = no positive swabs for that bird at any timepoint.
fN/A indicates that tissues were not present on section.
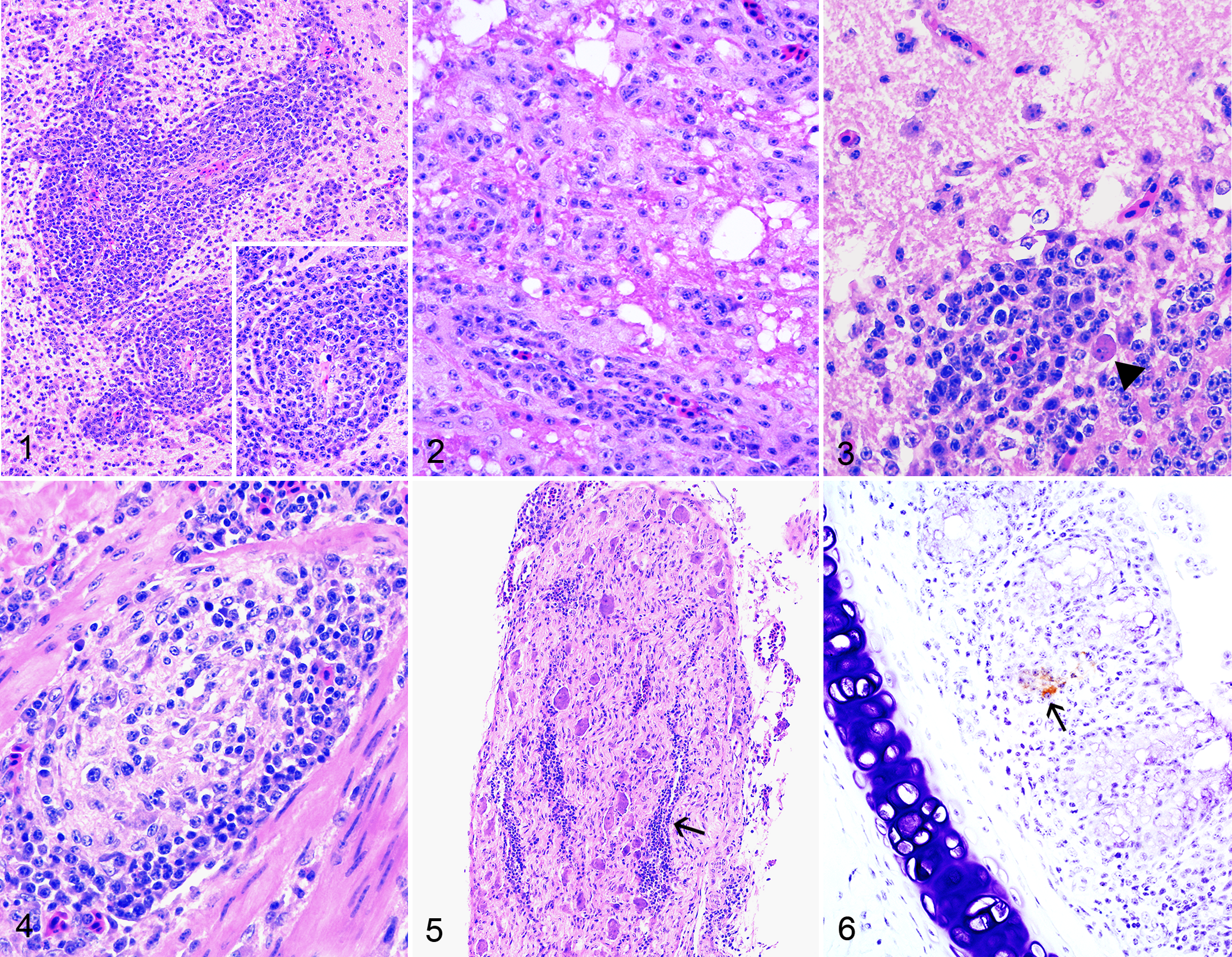
In 5 birds, there were severe microscopic lesions that were not considered to be NDV-induced. These consisted of: (1) granulomatous and heterophilic coelomitis and epicarditis with intralesional bacteria (1 control bird sampled at 7 dpi); (2) multifocal fibrinous and heterophilic bronchopneumonia and multifocal necrotizing rhinitis with intralesional bacteria (1 N2-inoculated bird that died at 2 dpi, Supplemental Fig. S4); (3) multifocal subacute fibrinoheterophilic epicarditis and celomitis with granulation tissue (Israel-inoculated bird sampled at 10 dpi); (4) chronic pericarditis, granulomatous pneumonia and airsacculitis with plant material, and heterophilic and granulomatous rhinitis (1 Pakistan-inoculated bird sampled at 7 dpi); and (5) severe blepharitis with intralesional bacteria and pulmonary edema (1 N23-contact bird that died at 5 dpc; this bird showed also marked autolysis).
Additional incidental lesions included accumulation of heterophils in rare bursal follicles (3 N2 contact birds; 2 N23-inoculated and 1 N23 contact bird; 1 Pakistan contact bird; 1 control bird) and moderate to severe inflammation of the Meckel’s diverticulum (3 N2-inoculated and 2 N2 contact birds; 4 N23-inoculated birds; 2 Israel-inoculated birds; and 2 Pakistan contact birds) that occasionally spread to the nearby tissue causing mild celomitis (Supplemental Fig. S5). Mild, focal epicardial fibrosis with hypertrophy of mesothelial cells was observed in 1 N23- and 1 Israel-inoculated bird (both sampled at 10 dpi).
Immunohistochemistry
In the control tissues from an Israel-inoculated chicken, 38 the primary antibody yielded a finely to coarsely granular staining within the macrophages in the splenic ellipsoids and in areas of necrosis (Supplemental Fig. S6) as well as in the bursa of Fabricius (Supplemental Fig. S7), while no reactivity was observed on tissues from a non-inoculated chicken. Using these same tissues, a mouse isotype control yielded no reactivity, confirming the specificity of the primary antibody (data not shown). In tested quail tissues, IHC reactivity was observed in a total of 7 birds: 1, 3, 2, and 1 birds inoculated with N2, N23, Israel, and Pakistan strains (Supplemental Table S1). Overall, immunolabeling intensity was weak and confined to scattered cells in the spleen (4 birds), turbinates (3 birds, Fig. 6), and thymus (2 birds). No immunoreactivity was observed in the areas where encephalitis was observed histologically.
Virus Isolation and Titration of Shed Virus
Results of virus isolation and titration from oropharyngeal and cloacal swabs are presented in Table 3, Figs. 7 and 8.
Summary of the Results for Virus Isolation and Titration From Oropharyngeal (O) and Cloacal (C) Swabs in Both Inoculation (In) and Contact (Co) Groups.
adpi indicates days postinfection.
bne, negative for virus isolation.
cIndicates the average titer calculated among all shedding birds in that group for that timepoint. All titers are expressed as EID50/0.1 ml.
dIndicates, in parentheses, the number of birds shedding (swabs positive by virus isolation) over the total sampled birds in that group for that timepoint.
eN/A indicates that sample was not collected.
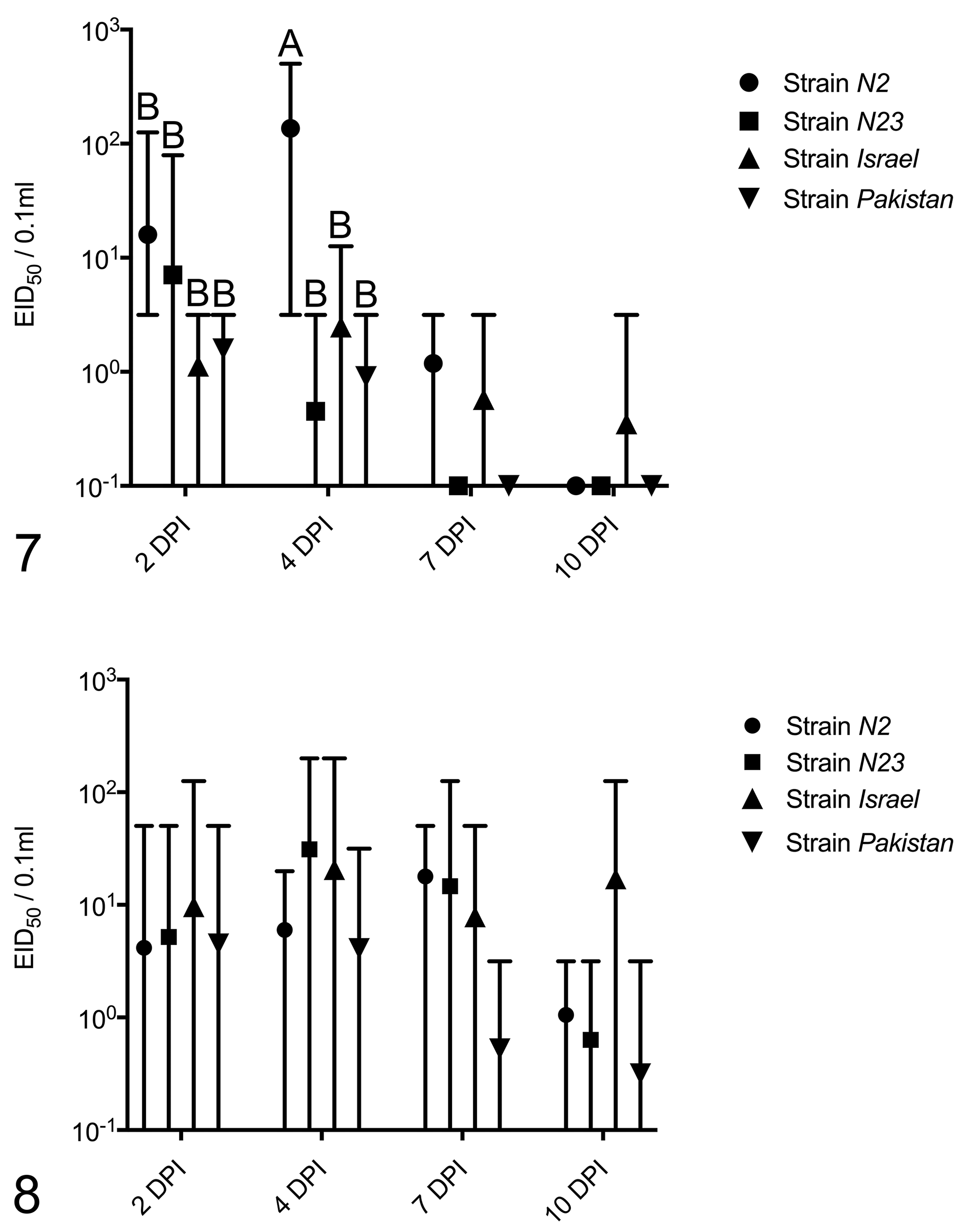
Inoculated birds
All NDV strains replicated in inoculated birds, with 100%, 79%, 93%, and 86% of quail inoculated with N2, N23, Israel, and Pakistan strains having at least 1 oropharyngeal or cloacal swab positive by virus isolation, respectively. Considering all sampling days (2, 4, 7, 10 dpi), N2 was positive in 73% and 51.2%, N23 in 16% and 50%, Israel in 33% and 39%, and Pakistan in 22% and 30% of all oropharyngeal and clocal swabs, respectively. Considering only shedding birds, strain N2 showed the highest average titer for oropharyngeal swabs at 4 dpi (102.13 EID50/0.1 ml; 13/13 birds shedding), and strain Israel had the highest average titer for cloacal swab at 4 dpi (101.76 EID50/0.1 ml; 5/14 birds shedding). The magnitude and the number of birds shedding through the oropharyngeal route drastically decreased from 4 to 7 dpi. Cloacal shedding was highest at 4 or 7 dpi for all inoculation groups (exception of Pakistan strain, highest at 2 dpi) and decreased at 10 dpi, with the exception of 4 birds inoculated with the Israel group (average, 101.59 EID50/0.1 ml, 4/9 birds shedding, 10 dpi).
Including negative swabs in the calculation of the group means, statistical analysis of oropharyngeal swabs showed that the average titer from N2-inoculated birds at 4 dpi was significantly higher than the other groups at 2 and 4 dpi (Fig. 7); no statistically significant differences were observed between the means of the cloacal titers at 2, 4, 7, and 10 dpi (Fig. 8) (2-way ANOVA with Tukey’s test for multiple comparisons). When negative samples were excluded from the calculations, no statistically significant differences were observed among the averages of oropharyngeal (2 and 4 dpi) and cloacal (2, 4, 7, and 10 dpi) titers (2-way ANOVA with Tukey’s test for multiple comparisons)
Considering oropharyngeal swabs, Fisher’s exact test showed that significantly more N2-inoculated quail shed virus at both 2 and 4 dpi (considered separately) than the other groups in the same day and that more N23-inoculated birds shed virus than Israel-inoculated birds at 4 dpi. Considering cloacal swabs, significantly more N2- and N23-inoculated birds shed than Pakistan-inoculated birds at 7 dpi.
Contact birds
At least 1 oropharyngeal or cloacal swab tested positive for virus isolation in 7.1%, 21.4%, 43%, and 26% of contact birds in the N2, N23, Israel, and Pakistan groups, respectively. Considering all sampling days (2, 5, 8 dpc), N2 was positive in 0% and 3%, N23 in 2.8% and 5.4%, Israel in 11.1% and 11.1%, and Pakistan in 2.7% and 11.1% of all oropharyngeal and clocal swabs, respectively. Including negative samples, comparison of titer averages (2-way ANOVA with Tukey’s test for multiple comparisons) showed that the Pakistan contact group at 5 dpc shed significantly higher amounts of virus from oropharyngeal swabs compared to the other groups at 2 and 8 dpc as well as more than the N2 and Israel groups at 5 dpc; no significant differences were observed in the magnitude of cloacal shedding (data not shown). Fisher’s exact test showed no differences between fractions of shedding birds in oropharyngeal or cloacal swabs among contact groups.
Virus isolation in relationship with incidental lesions
Among those 5 birds that showed severe microscopic incidental lesions (see histopathology section), the control bird sampled at 7 dpi and the N23 contact bird sampled at 5 dpc tested negative for virus isolation in both oropharyngeal and cloacal swabs. The N2-inoculated quail that died at 2 dpi tested positive in the oropharyngeal swab only (101.1 EID50/0.1 ml); the Pakistan-inoculated quail sampled at 7 dpi was negative at the time of sampling and had tested positive in a previous oropharyngeal (100.5 EID50/0.1 ml, 2 dpi) and clocal (100.5 EID50/0.1 ml, 4 dpi) swab; the Israel-inoculated quail sampled at 10 dpi was positive at the time of sampling for the cloacal swab (101.3 EID50/0.1 ml) and had tested positive previously at low titers (cloacal: 100.7 EID50/0.1 ml, 7 dpi; oropharyngeal and cloacal: 100.5 EID50/0.1 ml and 100.7 EID50/0.1 ml, respectively, 2 dpi).
Discussion
The 4 tested NDV strains showed mild to moderate disease-inducing ability in inoculated quail, with highest mortality being 28% among N2-inoculated birds and less than 10% for the others (considering all inoculated animals in both cages). This was mirrored by the low magnitude of virus replication, as observed by minimal IHC labeling, low virus titers in oropharyngeal and cloacal secretions, and low numbers of shedding birds. Similarly, no NDV-related morbidity and mortality were observed in contact birds, with only few shedding virus at low titers. In contrast, when naive chickens are infected with highly virulent NDV strains, mortality reaches 100%, immunohistochemical labeling is observed in most tissues, and the majority of birds shed virus at high titers, leading to high rates of infection in contact birds. 7,12,27,42 The findings of this work are in agreement with some studies, which reported no 25,37 to low mortality (13%) 33 when Japanese quail were infected by the oculo-nasal route with virulent NDV strains.
In the present study, the only microscopic lesions consistent with ND histopathology were observed in the brain and were characterized by nonsuppurative encephalitis and gliosis, which were initially observed at 7 dpi and progressed in intensity and prevalence (more birds involved) by 10 dpi. Nervous involvement at later stages of infection (after 5 dpi) is often observed with ND. 7,16 Although involvement of the central nervous system (brain) was prevalent, inflammation was also observed in the peripheral nervous tissue (splanchnic nerves and intestinal ganglia) of 4 birds. Tropism of virulent NDV strain for the peripheral nervous tissue has been previously documented 42 ; therefore, it is possible that the observed ganglioneuritis might have been the consequence of virus replication at those sites. No lesions in the lymphoid organs, a hallmark of infection with highly virulent NDV strains in chickens, 7,32,41,42 were observed, which may account for the low morbidity and mortality recorded during this experiment.
Low mortality associated with absence of lymphoid necrosis and sporadic involvement of the nervous system has been commonly reported in avian species that are partially resistant to ND development (compared to chickens), such as pigeons, 45 cormorants, 24 ducks, 10 and different species of pet birds, 20 upon infection with highly virulent NDV strains. On the other hand, chickens—which are highly susceptible—develop nervous lesions when infected with attenuated NDV strains that allow for a longer course of the disease (ie, mesogenic pathotypes) 7,32 or when birds have partially protective antibody titers against NDV before infection. 31 In the present study, HI assay on a representative subset of birds before commencing the experiment showed that quail were serologically naïve against NDV. Additionally, all 4 NDV strains used in the present study were highly virulent according to standard pathogenicity tests (ICPI > 1.7), and the Israel and Pakistan strains behaved as VVNDV strains in chickens, as previously shown by our group. 38 Lastly, quail were inoculated with a high virus dose (108 EID50 units/bird), which is significantly higher (approximately 3 logarithmic units) than what is normally used to infect White Leghorn chickens of similar age by our group (105 EID50 units/bird), 38,42 suggesting that failure to reproduce a severe course of disease characterized by high morbidity and mortality was not caused by too low an infectious dose.
Among the birds submitted for histopathology in this study, 5 showed severe microscopic changes not associated with NDV infection in the literature. 7 These consisted of necrotizing and granulomatous lesions within the epicardium, celomic cavity, air sacs, and lungs. Considering that 2 of these birds were negative for virus isolation and 3 were positive at low titers in sampled swabs, there is strong evidence that these lesions were not related to NDV inoculation and were considered likely to be caused by bacterial infections or—when observed in the lesions—inhalation of plant material. Meckel’s diverticulitis and accumulation of heterophils in rare follicles of the Bursa of Fabricius were also observed in a few birds, and these were considered incidental lesions. Presence of these background lesions is likely a reflection of the fact that quail were not SPF.
Data of VI and titration showed that all 4 strains could infect Japanese quail; however, the magnitude of virus replication and shedding was low, leading to minimal transmission to contact birds. Scant immunolabeling for NDV NP in tissues of inoculated birds further suggests a limited replicative ability of these strains in quail. No immunolabeling was noted in the areas of nonsuppurative encephalitis at 7 and 10 dpi, and no signal was observed during previous timepoints in the brain of sampled quail. It is possible that by day 7 dpi, the virus was cleared from the nervous tissue or that given the low magnitude of replication, NDV antigen in tissues might have been below the detection limit for the IHC assay. It should be noted that even in those instances when immunolabeling for NDV is observed in the brain of infected chickens, signal is commonly multifocal and involves only rare neurons. 16
Highest morbidity (nervous signs and depression) and mortality, as well as significantly highest numbers of shedding birds and maximal magnitude of oropharyngeal virus shedding, were observed in birds inoculated with the N2 strain, which is a quail-derived isolate. These data suggest that the N2 strain (ICPI = 1.96) may be more adapted to the host and possibly more pathogenic or quail-derived than chicken-derived isolates. However, the other quail-derived strain in this study (N23) caused no morbidity or mortality, perhaps due to the relatively lower ICPI score (1.76). Alternatively, it is possible that N23 strain might not have been fully adapted to quail: Since genotype VIh commonly circulates in pigeon and dove species, 13 it is conceivable that isolation of N23 strain from quail might have been incidental.
In summary, oro-nasal inoculation of highly virulent NDV strains in quail resulted in infection of the majority of birds (≥79%) but limited tissue replication as observed by virus titration in swabs and IHC, leading to low transmission rates (≤43%). Most likely as a reflection of limited virus replication, highest mortality was low (28%) and observed in N2-inoculated birds. Lesions in inoculated birds consisted of encephalitis from 7 dpi onward, which is commonly observed in partially resistant birds when inoculated with highly virulent NDV strains. These results suggest that Japanese quail have a certain degree of natural resistance to NDV infection and development of lesions. In addition, more severe clinical signs and lesions caused by a quail-derived isolate suggest that host adaptation may play a role in increasing NDV virulence for this species.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818767996 - Newcastle Disease Virus Infection in Quail
Supplemental Material, DS1_VET_10.1177_0300985818767996 for Newcastle Disease Virus Infection in Quail by Leonardo Susta, Diego Segovia, Timothy L. Olivier, Kiril M. Dimitrov, Ismaila Shittu, Valerie Marcano, and Patti J. Miller in Veterinary Pathology
Footnotes
Acknowledgements
We would like to acknowledge Dawn Williams-Coplin for technical assistance. The mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. The USDA is an equal opportunity provider and employer.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by USDA-ARS CRIS 6040-32000-072. Mr. Segovia’s salary was partly funded through the Undergraduate Research Assistantship (URA) program of the University of Guelph.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
