Abstract
Two newly established canine histiocytic sarcoma (HS) cell lines, designated as PWC-HS01 and FCR-HS02, were obtained from brain and articular tumors, respectively. These 2 HS cell lines had phagocytic ability and modal chromosome aberrations. Although morphologic features of both HS cells were similar, immunocytochemical examinations revealed that the PWC-HS01 cell line expressed both dendritic cell (ie, S100, CD208, CD1, and CD4) and macrophage (ie, CD68, CD163, and CD204) markers. In contrast, the FCR-HS02 cell line was immunonegative for CD204 and CD68 but consistently positive for the dendritic cell markers. Moreover, reverse transcription polymerase chain reaction analyses confirmed histiocytic differentiation of both HS cell lines. These results suggest that HS from the central nervous system may have a tendency to be more undifferentiated compared with cases from other organs. In addition, the 2 newly established HS cell lines were also tumorigenic and metastatic in immunodeficient mice, supporting that these cell lines can be used as new tumor models for investigating canine histiocytic diseases.
Histiocytic sarcoma (HS), a form of histiocytic proliferative disorder (HPD), is a life-threatening, aggressive malignant tumor derived from histiocytes. Histiocytes can be classified into dendritic cells (DCs) and macrophages, and they play important roles in antigen presentation and phagocytosis, respectively, in the mononuclear phagocyte system. Unlike in humans, HSs in dogs have either a DC or macrophage origin, based on the findings of immunohistochemical studies of the neoplasms. 2,9 However, the majority of HS cases in dogs have DC differentiation, whereas macrophage differentiation is rarely illustrated. In the veterinary literature, primary HS in the central nervous system (CNS) is uncommon and can be classified into localized and disseminated forms similar to those in other organs. In the CNS, the localized form is more common in canine HS. 6,15,27,29,35
Tumor cell lines isolated from both human and animal sources have been widely used for many purposes including cancer and toxicologic research and novel antitumor drug development. To date, a few studies have demonstrated the establishment of canine HS cell lines isolated from the bone marrow, skin masses, and synovial masses. 3,25,31 To our knowledge, the morphologic characteristics as well as biological properties of canine HS cell line isolated from the CNS have not been described. Therefore, in the present study, an HS cell line (PWC-HS01) obtained from a brain tumor is compared with another new HS cell line (FCR-HS02) from an articular tumor. Cellular morphologic and immunocytochemical characteristics as well as molecular and biological behaviors of the cell lines are elucidated. The tumor xenotransplantation and metastasis assay, moreover, are performed using immunodeficient mice to evaluate biological properties of the 2 cell lines.
Materials and Methods
Animals
Tumor samples were obtained from 2 dogs. Dog No. 1 was an 11-year-5-month-old female Pembroke Welsh Corgi with a history of right hemiplegia and convulsion. Magnetic resonance imaging of the brain revealed a solitary multilobulated mass located in the left frontal lobe (Supplemental Fig. 1). No evidence of distant metastases was observed. Brain surgery requested by the owner was performed. Dog No. 2 was an 8-year-6-month-old male Flat-Coated Retriever suffering from right forelimb stiffness and non–weight bearing. A computed tomography scan revealed a mass located around the articular area of the right shoulder joint and proximal part of the humerus (Supplemental Fig. 2). Limb amputation as well as lymphadenectomy of the regional lymph nodes were performed. It is unfortunate that no follow-up data were available.
Cell Cultivation
Fresh tumor samples were finely minced and washed twice with sterile phosphate buffered saline (PBS; pH 7.4) containing 1% penicillin-streptomycin. The samples were then suspended in Dulbecco’s modified eagle medium (DMEM; Gibco™; Invitrogen, Tokyo, Japan) containing 0.25% crude trypsin and incubated at 4ºC for 16 to 18 hours. After centrifugation, the pellets were resuspended in new DMEM supplemented with 10% heat-inactivated fetal bovine serum (FBS; GE Healthcare Life Sciences, South Logan, UT, USA) and 1% penicillin-streptomycin (Wako Pure Chemical, Osaka, Japan), and filtered through a cell strainer (70 µm nylon; Falcon, Tokyo, Japan). The suspended cells were seeded into 100-mm petri dishes (Nunc™ delta surface; Thermo Scientific, Tokyo, Japan) and subsequently incubated at 37ºC in an atmosphere of 5% CO2 and 95% air.
Three months after the initial cultivation, tumor cells were cloned by a serial dilution method. After cells had grown to cover a cell culture plate, cloned cells were removed and seeded into 60-mm petri dishes with DMEM supplemented with 10% FBS. Media were replaced every 4 days and cell passages were performed weekly. Population-doubling time was determined using the procedure described previously. 8,25 The newly established canine HS cells from the 30th passage were used for all in vitro experiments and each study was repeated at least 5 times to verify the results.
Morphologic Characterization of the Established Canine HS Cell Lines
PWC-HS01 and FCR-HS02 cells (1 × 104 cells/ml) were seeded into 2-well chamber slides (Nunc™ Lab-Tek™ II CC2™ chamber slide system; Thermo Scientific, Rochester, NY, USA) and incubated at 37ºC in an atmosphere of 5% CO2 and 95% air. One week after cultivation, the cells were stained with Giemsa.
Immunophenotypic Characterization of the Established Canine HS Cell Lines
PWC-HS01 and FCR-HS02 cells were inoculated at 1 × 105 cells/ml in 100-mm petri dishes (Thermo Scientific, Tokyo, Japan) and incubated at 37ºC in an atmosphere of 5% CO2 and 95% air. One week after cultivation, these cells were detached by trypsinization and then centrifuged at 1000 rpm for 6 minutes using a cytocentrifuge (StatSpin® Cytofuge® 2 Cytocentrifuge; Beckman Coulter, Tokyo, Japan), and the slides with the cells were fixed with 4% paraformaldehyde. In cases of the detection of intracellular markers, cell membrane permeabilization was then performed by incubating the cells with 0.25% Triton X-100 prior to a blocking step. After rinsing with PBS, all slides were treated with 1% bovine serum albumin in PBS with Tween® 20 (PBST) to block nonspecific reactions. Primary antibodies listed in Supplemental Table 1 were applied at 4ºC overnight. The slides were subsequently incubated with fluorochrome-labeled secondary antibodies including fluorescein horse antimouse immunoglobulin G (IgG), fluorescein goat antirabbit IgG, or fluorescein streptavidin (Vector Laboratory, Tokyo, Japan) at room temperature for 1 hour and counterstained with DAPI (Vectashield®; Vector Laboratory). All slides were observed under a Zeiss LSM 700 laser scanning confocal microscope (Carl Zeiss Meditec, Tokyo, Japan).
Phagocytosis Assay
PWC-HS01 and FCR-HS02 cells (1 × 105 cells/ml) were seeded into 2-well chamber slides and incubated at 37ºC in an atmosphere of 5%CO2 and 95% air overnight. The medium was substituted by serum-free DMEM with zymosan A fluorescent BioParticle® (Invitrogen), and then the cells were incubated at 37ºC for 2 hours in an atmosphere of 5% CO2 and 95% air. Cultured cells were fixed in 4% paraformaldehyde and counterstained with DAPI. Phagocytosis (tumor cells engulfing the fluorescent particles) was observed under a Zeiss LSM 700 laser scanning confocal microscope.
Chromosome Analysis
One week after initial seeding (initial cell concentration; 1 × 105 cells/ml) within the 100-mm petri dishes, cultured HS cells were incubated with colcemid (KaryoMax®; Invitrogen), with the final concentration of 0.02 µg/ml at 37ºC for 2 hours in an atmosphere of 5% CO2 and 95% air, and subsequently treated with a prewarmed hypotonic 0.075 M KCl solution. After centrifugation, the pellets including cell lysates were resuspended in a fixative solution (methanol-acetic acid; 1:1), dropped down on a slide, and subsequently stained with the Giemsa solution. Two hundred metaphases were counted under a light microscope in an oil-immersion field (1000×) to estimate the modal chromosome numbers.
Western Blot Analysis
Extracted whole cell proteins by RIPA buffer (50 mM Tris-HCl, 150 mM NaCl, 1% Triton X-100, 0.5% sodium deoxycholate, 0.1% sodium dodecyl sulfate, 5 mM EDTA, 2 mM Na3VO4, 1 mM phenylmethylsulfonyl fluoride, 10 mM sodium fluoride, and complete tablets, mini EDTA-free, EASYpack; Roche Diagnostic, Mannheim, Germany) were used in this experiment. Twenty μg of protein samples diluted (1:1) with a loading buffer were electrophoresed on a 15% polyacrylamide gel (e-PAGEL®; ATTO Corporation, Tokyo, Japan) at 600 volts, 20 mA, for 90 minutes. The protein bands were transferred onto a polyvinylidene fluoride (PVDF) membrane (Immibilon®-P transfer membrane; Millipore Corporation, Billerica, MA, USA) at 20 volts, 200 mA, for 25 minutes. A chemiluminescence method (Amersham™ ECL™ prime Western blotting detection reagent; GE Healthcare, Little Chalfont, UK) was used for the detection of immunoblotting bands. Primary antibodies used were anti-CD204 (1:500; TransGenic, Kobe, Japan) and anti-β-actin (1:2000; Cell Signaling Technology, Tokyo, Japan).
Reverse Transcription Polymerase Chain Reaction
Total RNA was extracted from PWC-HS01 and FCR-HS02 cells using an RNA extraction kit (RNeasy Mini Kit; Qiagen, Tokyo, Japan) according to the manufacturer’s protocol. RNA concentration was then quantified using a NanoDrop 2000 spectrophotometer (Thermo Scientific, Wilmington, DE, USA) at a wavelength of 260/280 nm with an absorbance ratio between 1.8 and 2.0. In the reverse transcription process, cDNA was synthesized from RNA of each sample using a reverse transcription polymerase chain reaction (RT-PCR) kit (Prime Scipt™; Takara Bio, Tokyo, Japan) according to the manufacturer’s protocol.
RT-PCR amplification for canine CD11b, CD11c, and CD204 genes was performed. In addition, glyceraldehyde-3-phosphate dehydrogenase was also used as an internal control. The primers were constructed using the sequences appearing in the Primer 3Plus (www.bioinformatics.nl) or a previous report. 33 The sequences are detailed in Supplemental Table 2. For PCR analysis, DNA polymerase was activated at 95ºC for 3 minutes, and cDNA followed by 40 cycles of denaturation at 94ºC for 30 seconds, annealing at 52ºC or 60ºC for 1 minute, and extension at 72ºC for 30 seconds. The PCR products were mixed with a DNA loading buffer (Excellgen, Rockville, MD, USA) and then electrophoresed on a 2% agarose gel. To visualize the bands, a molecular imager, GelDoc™ XR+ System (BIO-RAD, Tokyo, Japan), was used.
Tumorigenicity and Metastasis Assays
Twelve 5-week-old female C.B-17/Icr-scid/scidJcl (SCID) mice (CLEA Japan, Tokyo, Japan) were randomly divided into 2 groups for PWC-HS01 and FCR-HS02 cell lines, respectively. The experiments regarding animal utilization were approved by the Animal Care and Use Committee of the Graduate School of Agricultural and Life Sciences at The University of Tokyo.
For determining tumorigenicity, 200 µl of cell suspension (3 × 106 cells/ml) in Hanks’ balanced salt solution (HBSS; Gibco; Invitrogen) were subcutaneously injected into the lower flank of the immunodeficient mice. Tumor formation at the injection site was observed for 2 months. The tumor volume was evaluated using the following equation: Tumor volume (mm3) = (Length × Width 2 ) / 2. 16 For determining metastatic properties, 200 µl of cell suspension (5 × 105 cells/ml) in HBSS were injected into the lateral tail vein of the SCID mice. Two months after injection, all animals were euthanized with isoflurane and grossly examined for tumor development including the presence of distant metastases. Visceral organs and tumor masses were immediately fixed in a 10% neutral-buffered formalin solution and subsequently embedded in paraffin. Immunohistochemistry (IHC) was performed as described previously. 27 Antigen retrieval methods and primary antibodies used for IHC are listed in Supplemental Table 3.
Results
Histologic Features of the Original Tumors From the Brain and Shoulder Joint
Both tumors contained numerous round- to polygonal-shaped cells with eosinophilic cytoplasm and a distinct cell border. Cytoplasmic vacuolation was occasionally observed. Their nuclei were round to polygonal with prominent nucleoli, mild to moderate atypia, and frequent atypical mitoses. Multinucleated giant tumor cells were sparsely present (Figs. 1, 2). Moderate infiltration of small lymphocytes was interspersed among the neoplastic cells and necrosis was common. In dog No. 2, tumor metastasis was present in adjacent lymph nodes.
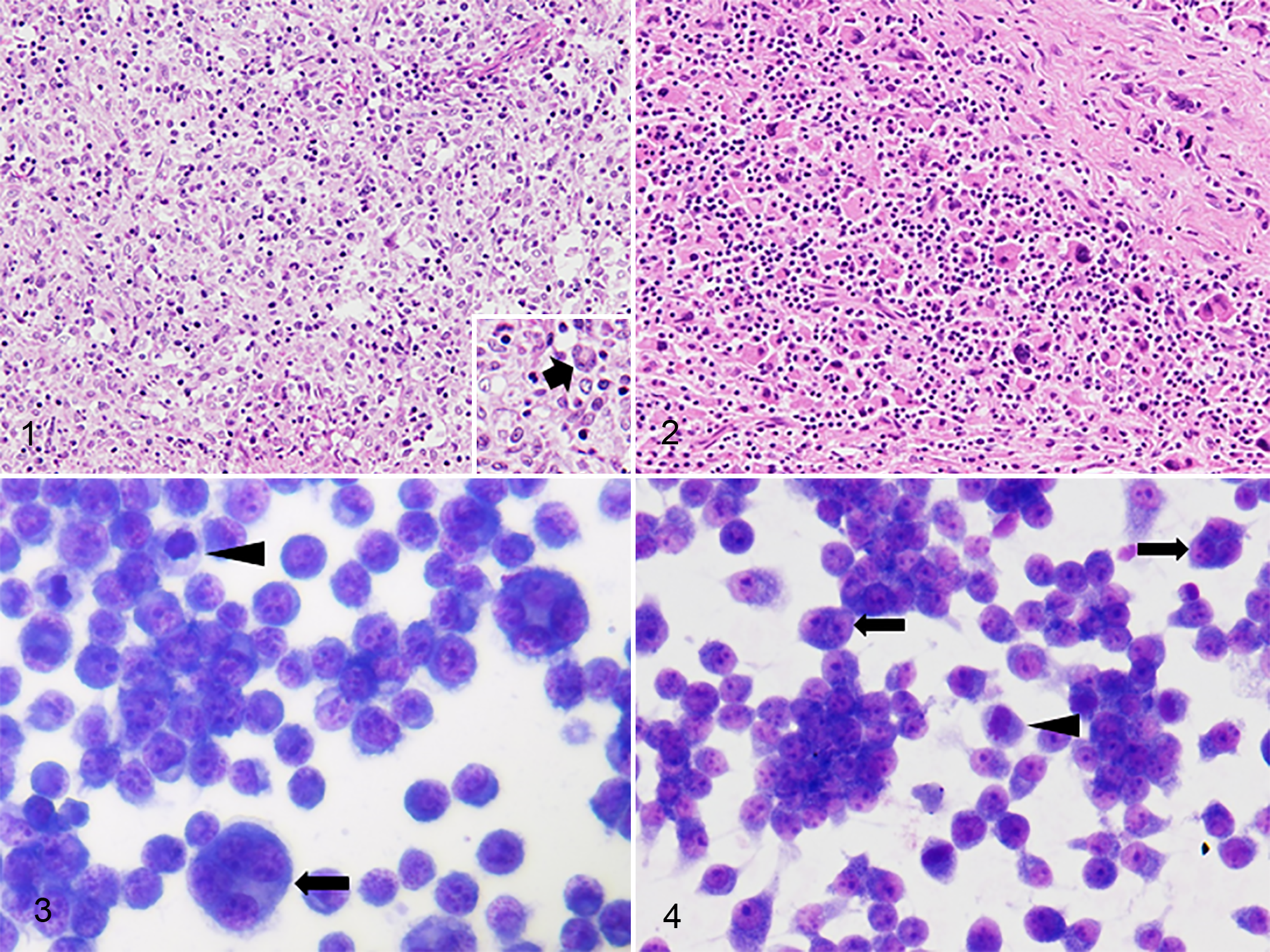
Histiocytic sarcoma. Dog.
Morphologic Characteristics of the Established Canine HS Cell Lines
In both HS cell lines, most cells were round to polygonal with abundant cytoplasm. Their nuclei were round with prominent single or multiple nucleoli, frequent atypical mitoses, and mild to moderate anisocytosis, anisokaryosis, and anisonucleoliosis. Bizarre binucleated and multinucleated giant cells were occasionally present. PWC-HS01 cells proliferated in monolayer, whereas FCR-HS02 cells usually formed proliferating clumps in culture (Figs. 3, 4). The population-doubling time of PWC-HS01 and FCR-HS02 was approximately 64.8 and 56.5 hours, respectively (Fig. 5, Table 1). Furthermore, the cells of both lines had adhesive properties to the cell culture vessel.
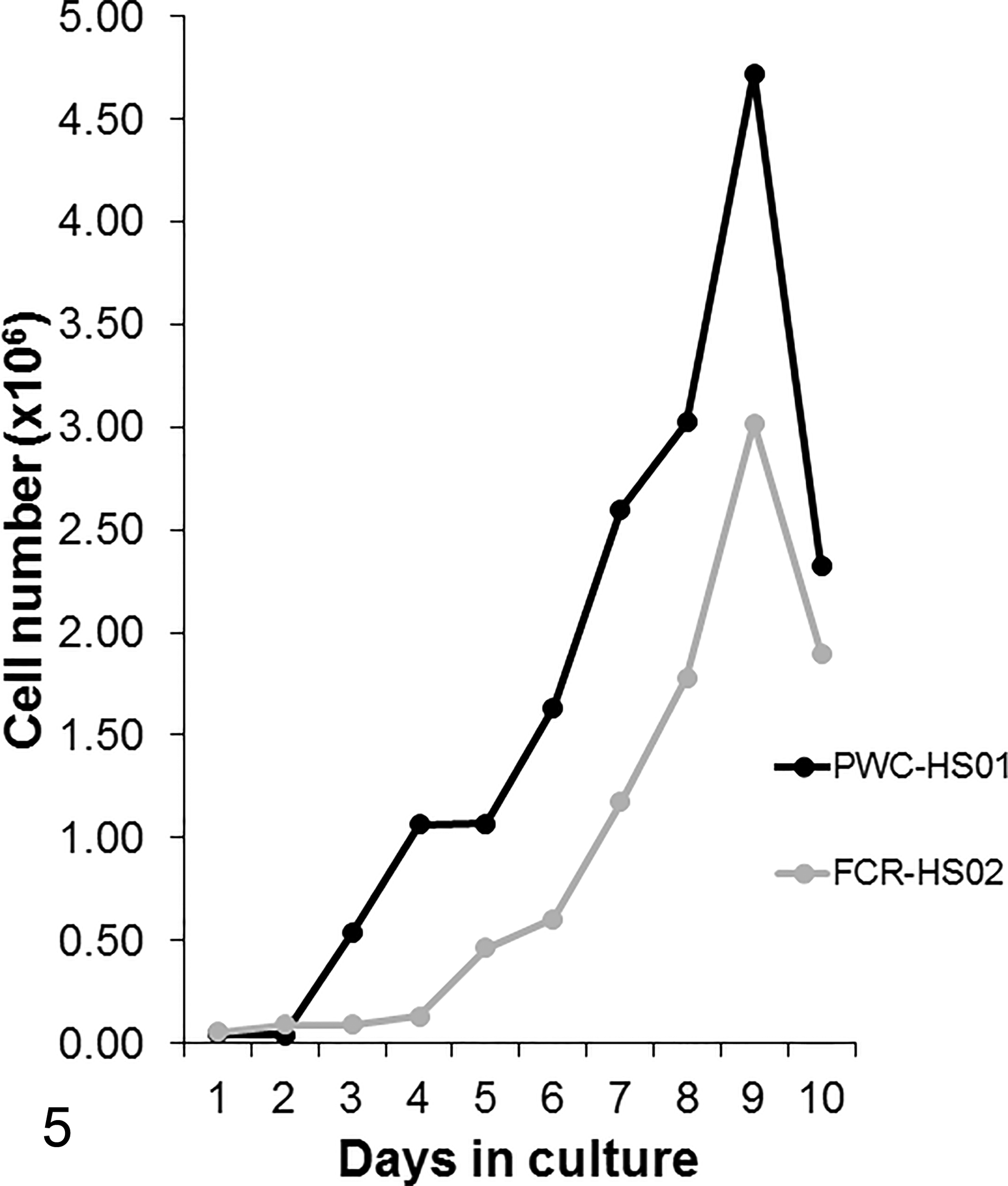
Histiocytic sarcoma cell lines (PWC-HS01 and FCR-HS02). Dog. Growth curves of newly established canine histiocytic sarcoma cell lines cultured in Dulbecco’s modified eagle medium supplemented with 10% fetal bovine serum. The population-doubling times of PWC-HS01 and FCR-HS02 cell lines are 64.8 and 56.5 hours, respectively.
Biological and Immunocytochemical Characteristics of HS Cell Lines.
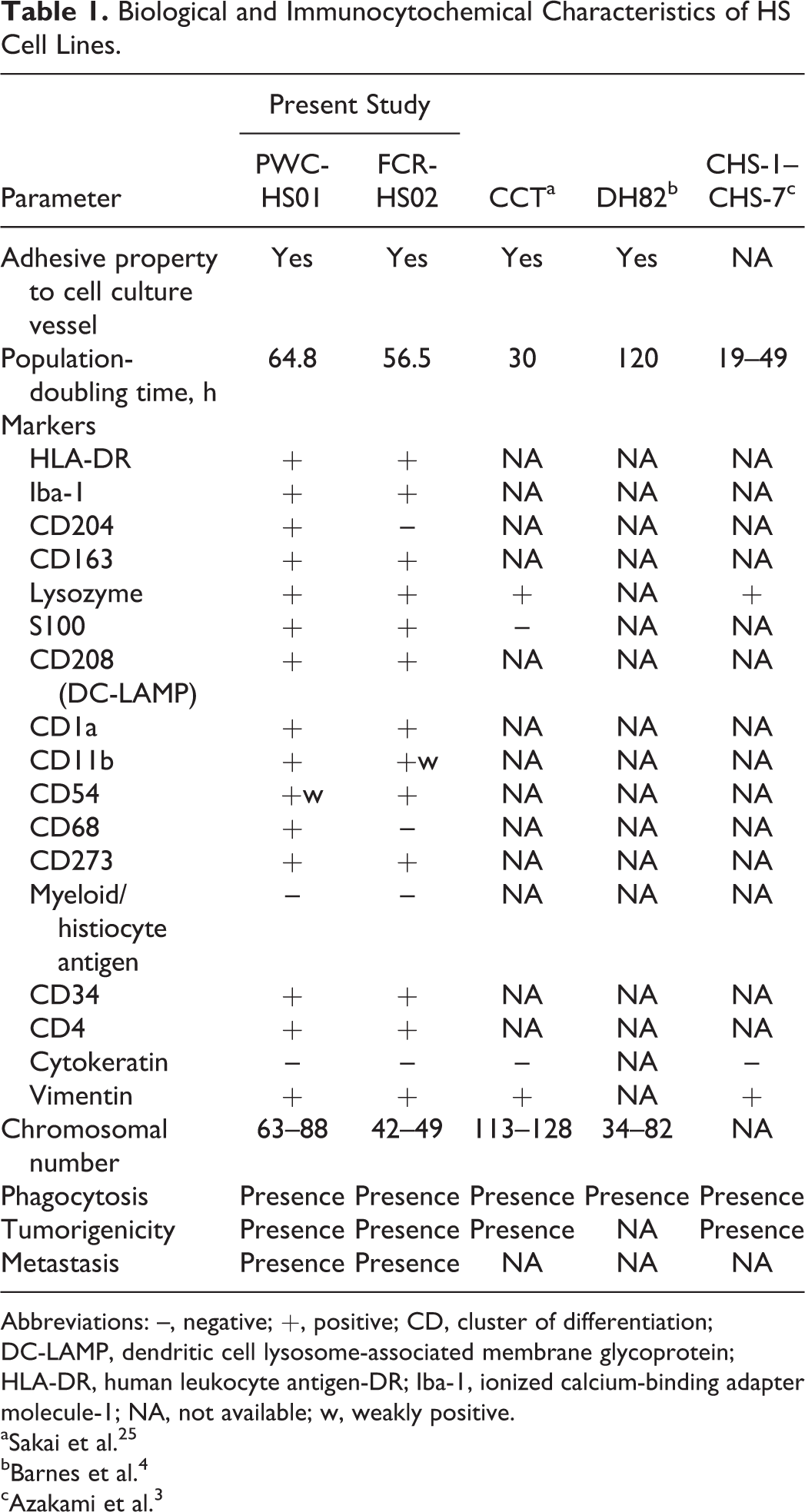
Abbreviations: –, negative; +, positive; CD, cluster of differentiation; DC-LAMP, dendritic cell lysosome-associated membrane glycoprotein; HLA-DR, human leukocyte antigen-DR; Iba-1, ionized calcium-binding adapter molecule-1; NA, not available; w, weakly positive.
aSakai et al. 25
bBarnes et al. 4
cAzakami et al. 3
Immunocytochemical Characterization of Canine HS Cell Lines
Immunocytochemical examination revealed that cells of both cell lines were consistently positive for HLA-DR, Iba-1, CD163, iNOS, lysozyme, S100, CD208, vimentin, CD34, CD4, CD1a, CD11b, and CD273. Variable or weak CD54 immunoreaction on the cell membrane was also observed. However, the cells of both lines were negative for cytokeratin AE1/AE3. PWC-HS01 cells were positive for CD68 and CD204, but FCR-HS02 cells were negative (Figs. 6, 7, Table 1).
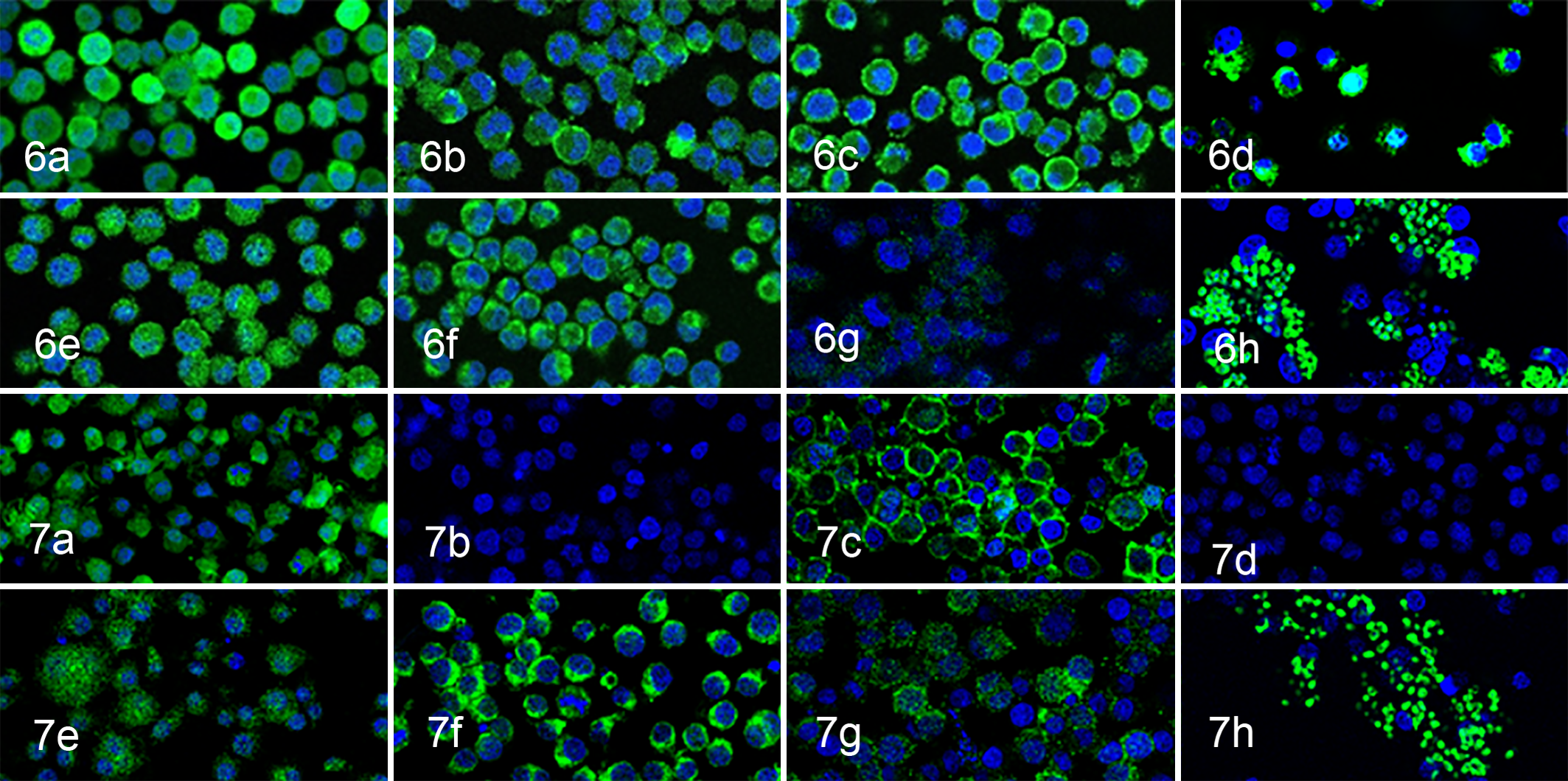
Phagocytosis and Chromosome Analyses
Both PWC-HS01 and FCR-HS02 cell lines had the capacity to engulf zymosan A, consistent with a histiocytic origin (Figs. 6, 7, Table 1). The modal chromosome number of the PWC-HS01 cell line was 63–88 (mode = 75, mean = 74.7), and that of FCR-HS02 was 42–49 (mode = 46, mean = 45.4) (Table 1).
Western Blot and RT-PCR Analyses
Extracted proteins from both HS cell lines were examined for the expression of CD204 by Western blotting. The result revealed that CD204 (72 kDa) was detected only in PWC-HS01 cells and not in FCR-HS02 cells (Fig. 8). The expression of CD11b mRNA was observed in both cell lines. Strong and moderate expression of CD11c mRNA was observed in PWC-HS01 and FCR-HS02 cells, respectively. FCR-HS02 cells showed a low expression of CD204 mRNA (Fig. 9).
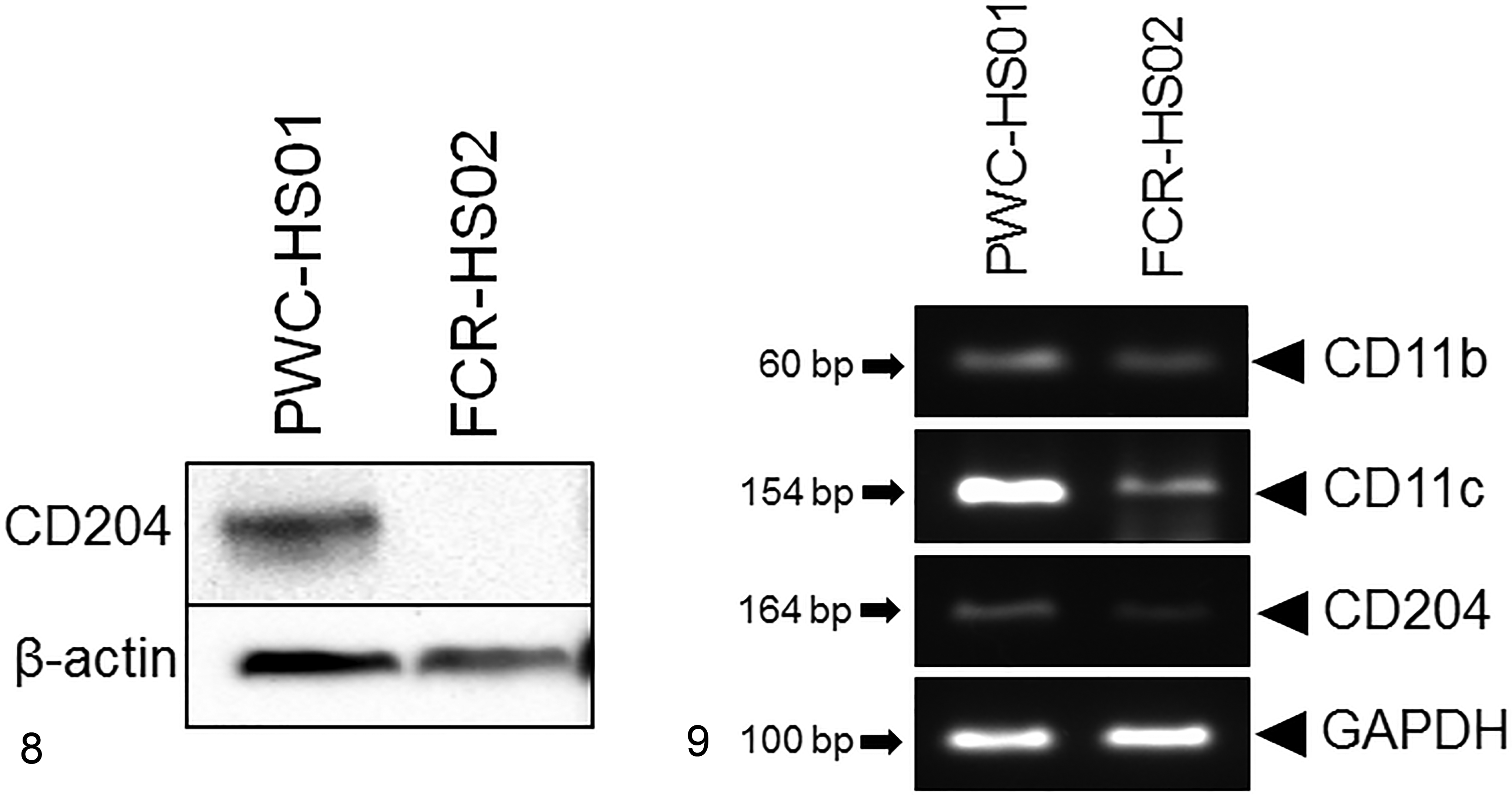
Histiocytic sarcoma cell lines (PWC-HS01 and FCR-HS02). Dog.
Xenotransplantation to Immunodeficient Mice
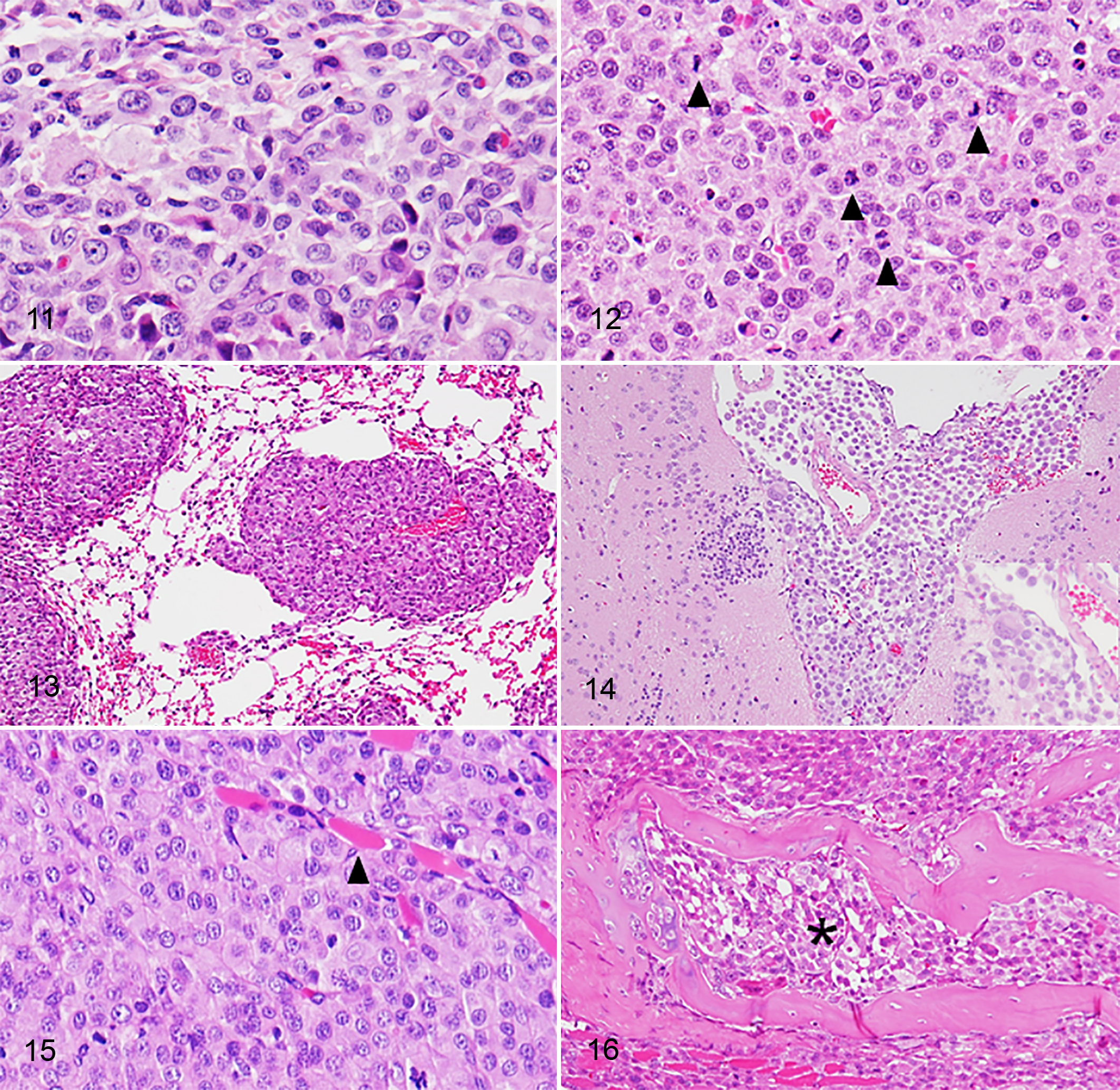
Subcutaneous masses were observed in all SCID mice injected with either cell line, 1 month after cell inoculation (Fig. 10, Supplemental Figs. 3, 4). The masses were dome-shaped and usually firm. Mild to moderate hemorrhage and necrosis were observed in the center of the masses. No distant metastases were detected in any of the mice. Tumors from the 2 cell lines showed the same histopathologic features. Numerous round- to polygonal-shaped anaplastic cells were arranged in a sheet with a solid pattern. Bizarre mononuclear cells were common. The cells had abundant eosinophilic cytoplasm. Their nuclei were mainly round to pleomorphic with 1 or multiple prominent nucleoli. Mitotic figures were frequent (> 100 mitoses/10 high power fields, 400×). Cytoplasmic vacuolation was notable in FCR-HS02 cells xenografted into SCID mice. Multinucleated giant cells in the original tumors were rarely detected in the xenotransplanted tumors. Moreover, necrosis was common in the center of the tumor tissues (Figs. 11, 12).
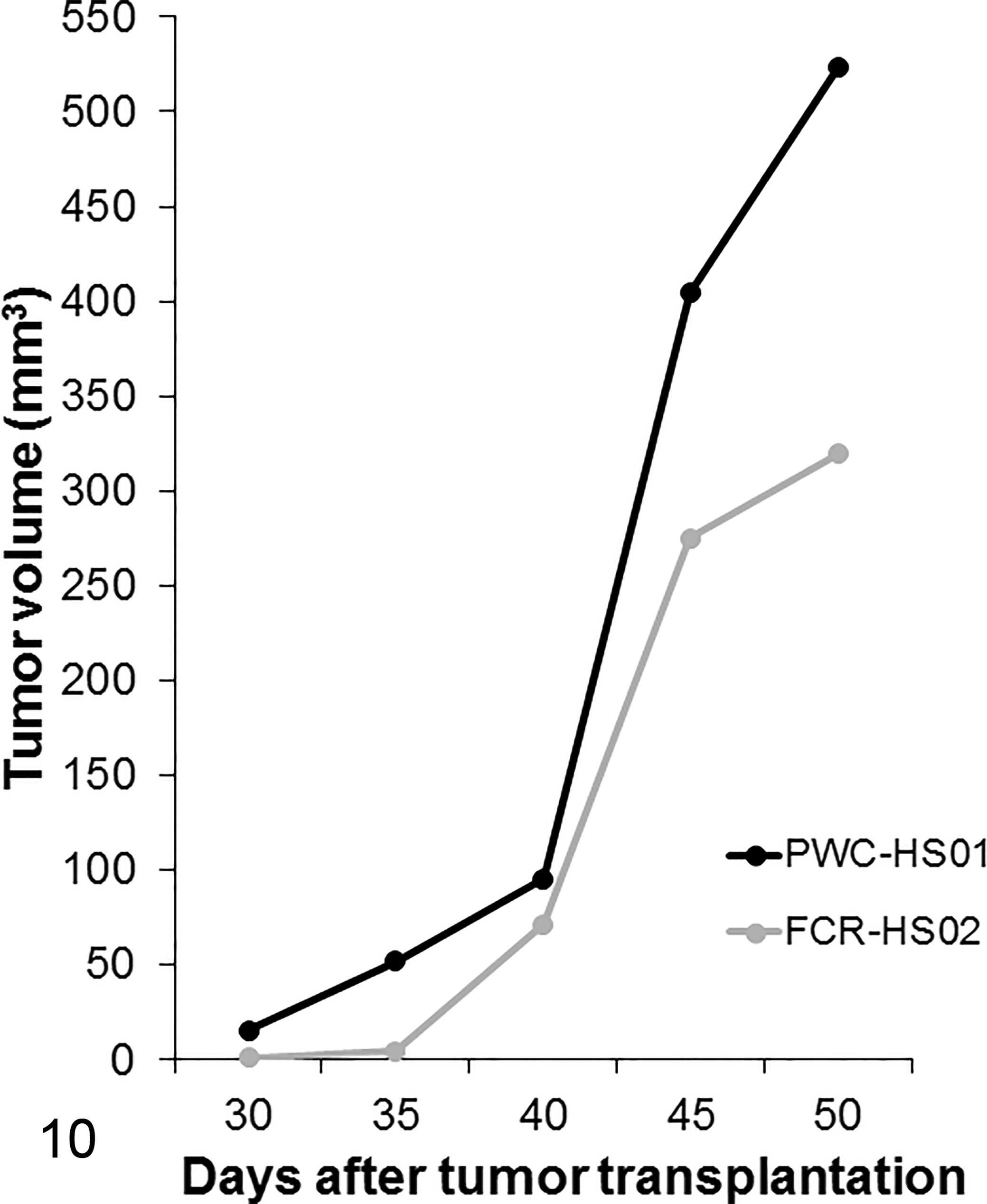
Histiocytic sarcoma. SCID mice. Thirty to 35 days after tumor inoculation in the mice, masses are grossly observed in all mice. Tumorigenicity assay.
Immunohistochemistry examination revealed that both tumor cell lines transplanted were consistently positive for HLA-DR, CD204, CD163, and vimentin. Immunoreactivities to Iba-1 and lysozyme were variable. None or very few cells were positive for S100. Furthermore, all the tumor cells were negative for CD208, unlike the original tumor. The IHC features are summarized in Table 2.
Comparison of Immunohistochemical Features of Primary, Xenotransplanted, and Metastatic Tumors.a
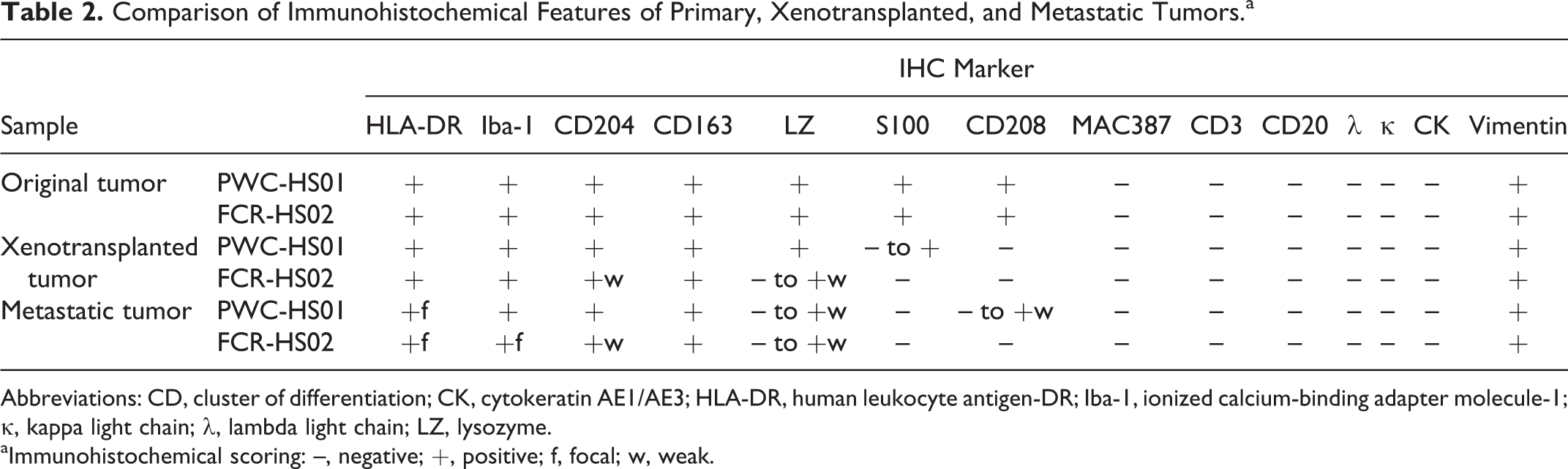
Abbreviations: CD, cluster of differentiation; CK, cytokeratin AE1/AE3; HLA-DR, human leukocyte antigen-DR; Iba-1, ionized calcium-binding adapter molecule-1; κ, kappa light chain; λ, lambda light chain; LZ, lysozyme.
aImmunohistochemical scoring: –, negative; +, positive; f, focal; w, weak.
Metastasis Assay
Metastatic lesions in the lung, hilar lymph nodes, and abdominal cavity were grossly observed in 2 of 3 mice intravenously injected with PWC-HS01 cells (Fig. 13). The lesions microscopically appeared also in the diaphragm, sternum, and cardiac muscle. The other mouse showed microscopic metastasis to the brain and meninges without any lesions in other organs (Fig. 14). Immunohistochemically, contrary to the original tumor, metastatic tumor cells lacked the expression of S100, whereas immunoreactivities to HLA-DR, lysozyme, and CD208 were variable. These metastatic tumor cells were consistently positive for Iba-1, CD204, CD163, lysozyme, and vimentin as were those in the original tumor (Table 2).
On the other hand, lesions in the thoracic cavity as well as abdominal cavity were commonly observed in all the mice injected with FCR-HS02 cells. In addition, metastatic masses in the skin were detected. Histopathologically, tumor cells invading the lung parenchyma and bone marrow were also observed in 1 mouse (Figs. 15, 16). Contrary to the original tumors, all the metastatic tumors lacked expression of S100 and CD208. The immunoreactivities to HLA-DR and lysozyme were variable. However, the metastatic tumor cells were positive for Iba-1, CD204, and CD163 (Table 2, Supplemental Figs. 5, 6).
Discussion
Herein, we report successful establishment of new HS cell lines, PWC-HS01 and FCR-HS02, which were derived from brain and articular HS tumors, respectively. Morphologic characteristics of these cells were similar to those of normal histiocytes and of other canine HS cell lines established previously (Table 1). 1,3,4,12,22,31
The results of immunocytochemical examination revealed that PWC-HS01 cells were positive for HLA-DR, Iba-1, and CD204, similar to those of the original tumor, indicating histiocytic differentiation. It is interesting that the cells expressed both macrophage (CD204, CD163, iNOS, lysozyme, CD68, and CD11b) and DC (S100, CD1a, CD4, CD54 [weak], and CD273) markers. The RT-PCR results also showed the expression of both CD11b (a macrophage marker) and CD11c (a DC marker) mRNA. 7,9,20,21,24,29 These findings indicate that canine primary CNS HS expresses both macrophage and DC phenotypes, which is an aberrant phenomenon in other organ systems. 20,27 Moreover, PWC-HS01 cells were intensely positive for iNOS and CD163, suggesting that primary CNS HS may have features of both M1 and M2 macrophages.
Contrary to PWC-HS01 cells, FCR-HS02 cells were immunonegative for macrophage markers such as CD204 and CD68 yet consistently positive for DC markers such as S100, CD208, CD1a, and CD54. These observations may support that FCR-HS02 cells have a DC phenotype, which is consistent with the characteristics of HS derived from the extraneural organs described previously. 9,14,17,19,20 However, FCR-HS02 cells also expressed the lysozyme antigen, a macrophage marker. This aberrant phenomenon suggests that cell dedifferentiation has occurred in the cell line, resulting in a variety of molecular expressions. 13,17 The results of Western blotting and RT-PCR support the immunocytochemical characters of FCR-HS02 cells. Moreover, the expression of CD11b (low intensity) and CD11c (high intensity) mRNA was also detected. This strongly supports the DC phenotype of FCR-HS02 cells. 11
In addition, both established cell lines were also strongly immunopositive for CD34, a marker for hematopoietic stem and progenitor cells. 18 This finding supports that these cultured HS cells can revert to their progenitor cells.
It is interesting that although the 2 cultured HS cells reverted to the undifferentiated progenitors, high phagocytic capability was detected. In accordance with the phagocytosis ability, Golovkina et al 10 have illustrated that the presence of high FBS in cell culture medium leads to low phagocytic activity of cells. Furthermore, a high cell culture density also affects the phagocytosis of macrophages. 30 A low cell density as well as serum-free medium used in the present study may stimulate the phagocytic activity of both PWC-HS1 and FCR-HS02 cells.
In the xenotransplanted mice, all the tumor masses were confined to the dermis and/or subcutis. Spontaneous metastases to distant organs were not detected grossly or microscopically. These findings suggest that a certain surrounding microenvironment is relevant to tumor escape from the primary injected sites. 23,26 To assess the metastatic activity, suspended cells were intravenously injected into immunodeficient mice. Microscopic as well as gross metastatic lesions were observed in the dermis, subcutis, lung, diaphragm, sternum, cardiac muscle, abdominal cavity, bone marrow, and brain. However, other visceral organs such as the spleen and liver were not affected. It is interesting that there was only 1 mouse that developed microscopic metastatic lesions in the brain. Detailed mechanisms of the brain metastasis of PWC-HS01 remain unclear, although the cell line might have a tissue-tropism. To clarify the mechanism, further studies are needed. In addition, Tominaga et al 28 and Wilhelm et al 32 demonstrated that cultured tumor cells from the primary tumor in non-CNS organs can bind to blood–brain barrier endothelial cells and subsequently induce endothelial cell destruction, resulting in brain metastasis. These observations support the invasive capacity of the newly established canine HS cell lines.
Variable immunoreaction (negative to strongly positive) to HLA-DR, Iba-1, lysozyme, S100, and CD208 was observed in both xenotransplanted and metastatic tumor cells, whereas cultured PWC-HS01 and FCR-HS02 cells (prior to injection into mice) consistently exhibited all the markers used. It is interesting that both established cell lines also expressed CD34, a hematopoietic stem and progenitor cell marker, whereas those of the original tumor were negative (data not shown). Despite that all the HS cells in the original tumors consistently expressed CD204, those of the FCR-HS02 cell line were negative for CD204. This observation strongly supports that the original HSs may revert to their progenitors (dedifferentiation) by cell cultivation. 5,34 Moreover, cultured tumor cells may redifferentiate into another cell phenotype through in vivo reproliferation, depending on the tumor microenvironment, resulting in phenotypic alteration. 5
Unlike HS cases in extraneural organs, primary cerebral HS shares both macrophage and DC phenotypes, indicating that these tumor cells appear to have progenitor-like or stem-like features. These observations reflect the biological behavior of primary HS in the CNS, having a tendency to be poorly differentiated compared with HS from the other organ systems. However, further prospective studies of canine HS in the CNS are required to confirm these findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
