Abstract
Tissue microarrays (TMAs) represent a useful technique for the simultaneous phenotyping of large sample numbers and are particularly suitable for histopathologic tumor research. In this study, TMAs were used to evaluate semiquantitatively the expression of multiple antigens in various canine central nervous system (CNS) neoplasms and to identify markers with potential discriminative diagnostic relevance. Ninety-seven canine CNS neoplasms, previously diagnosed on hematoxylin and eosin sections according to the World Health Organization classification, were investigated on TMAs, with each tumor consisting of 2 cylindrical samples from the center and the periphery of the neoplasm. Tumor cells were phenotyped using a panel of 28 monoclonal and polyclonal antibodies, and hierarchical clustering analysis was applied to group neoplasms according to similarities in their expression profiles. Hierarchical clustering generally grouped cases with similar histologic diagnoses; however, gliomas especially exhibited a considerable heterogeneity in their positivity scores. Multiple tumor groups, such as astrocytomas and oligodendrogliomas, significantly differed in the proportion of positive immunoreaction for certain markers such as p75NTR, AQP4, GFAP, and S100 protein. The study highlights AQP4 and p75NTR as novel markers, helping to discriminate between canine astrocytoma and oligodendroglioma. Furthermore, the results suggest that p75NTR and proteolipid protein may represent useful markers, whose expression inversely correlates with malignant transformation in canine astrocytomas and oligodendrogliomas, respectively. Tissue microarray was demonstrated to be a useful and time-saving tool for the simultaneous immunohistochemical characterization of multiple canine CNS neoplasms. The present study provides a detailed overview of the expression patterns of different types of canine CNS neoplasms.
Keywords
Tumors of the central nervous system (CNS) in domestic animals have been intensely investigated over the past years with respect to both clinical and pathologic aspects. 13,31 An estimated 80% to 90% of brain tumors diagnosed in domestic animals occur in dogs and cats. 13 Canine CNS neoplasms have been reported with a comparatively high incidence of 14.5 per 100 000 dogs, 9,13,31,33 whereas human CNS neoplasms are reported with a relatively lower incidence of 4 to 5 per 100 000 individuals. 13,33 The continuous adaptation and advancement of the World Health Organization (WHO) classification of CNS tumors in domestic animals over the past decades is intensely influenced by novel insights into oncogenesis and tumor cell development and mirrors the ongoing scientific progress in the field of veterinary neuro-oncology and neuropathology, respectively.
Tissue microarrays (TMAs) represent a high-throughput technique for the simultaneous investigation of large sample numbers. Numerous small cylindrical paraffin wax–embedded tissue samples from different donor blocks are symmetrically arranged in 1 single recipient block, which can subsequently be used for various examinations on the DNA, RNA, and protein level. Tissue microarrays hold the advantages of a rapid, time- and cost-effective analysis of large sample numbers and allow standardized laboratory conditions as well as internal controls. 39 However, common limitations may concern poor representation of heterogeneous tissue samples and loss of tissue discs during processing. 12,38,39 The general usefulness of TMAs for the investigation of canine and feline CNS neoplasms has been validated earlier by comparing 10 archived paraffin wax blocks from CNS neoplasms with 2 punch biopsies from each respective tumor. 39 Staining with hematoxylin and eosin (HE) and immunohistochemistry were conducted under equal laboratory conditions. This previous study reported an accuracy of 95% for punch biopsies compared with the original donor blocks and thus highlighted TMA as a sufficient and reliable method for the simultaneous immunohistochemical characterization of canine CNS neoplasms. Moreover, recent studies on canine and feline mammary tumors have similarly reported TMA to preserve the molecular profile of tumor markers, 22 thus emphasizing TMA as a cost-effective and rapid method to phenotype multiple tumors simultaneously with a consistent methodology.
In dogs, meningiomas represent the most common intracalvarial neoplasm, 3,13,20,32 while gliomas, including astrocytoma and oligodendroglioma, are the second most common 31,32 followed by choroid plexus tumors. 3,13 Primary and secondary CNS neoplasms are assumed to occur with an equal incidence of approximately 50%. 3
Despite versatile scientific efforts, the determination of correct tumor diagnosis using HE-stained sections is still challenging, especially for highly malignant tumors with marked anaplasia. Moreover, interpretation of margins from surgical biopsy material has increasingly become important. 13 Both the histomorphologic growth pattern and the distinct cellular and nuclear characteristics are considered for a final histologic diagnosis. Hence, an accurate tumor diagnosis is an essential prerequisite for the prediction of patient survival and for choosing appropriate therapeutic strategies. Especially in human oncology, sophisticated molecular pathology on both the gene and protein level has gained increasing relevance during the past decades aimed to design novel therapies, which target specific molecules. 29 Here, although not applied routinely, immunohistochemistry may help to better determine the full spectrum of cellular characteristics. 13
An extensive pool of monoclonal and polyclonal antibodies has been established on canine CNS tissue over the past years 13,18,39 and has a long history of use on canine CNS neoplasms. 2,19 The commercial availability of these antibodies supports the identification of unique expression profiles in certain canine CNS neoplasms and may help to detect novel tumor cell markers with potential discriminative significance.
The present study aimed to apply TMA for a comprehensive immunohistochemical profiling of a total number of 97 canine CNS neoplasms. The objectives of the present study were (1) to reveal both similarities and differences in the antigen expression profile of multiple canine CNS neoplastic entities and (2) to identify potential markers, which might provide diagnostic tools that help to differentiate between canine CNS tumors and differentiation stages, respectively.
Materials and Methods
Case Collection
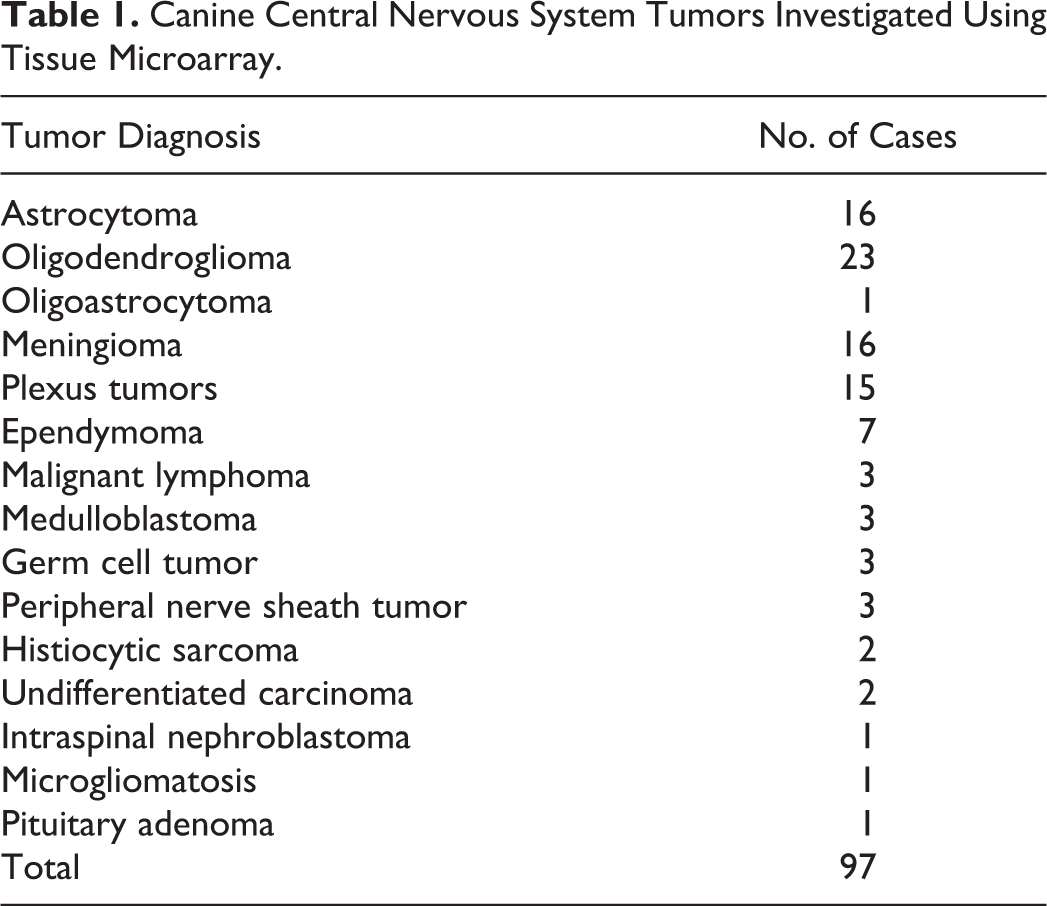
The archive of the routine necropsy material of the Department of Pathology, University of Veterinary Medicine, Hannover, Germany, was screened for all available cases of primary canine CNS neoplasms, which occurred between 1980 and 2003. Secondary CNS tumors and pituitary neoplasms were excluded from the screening of the archive; however, 1 case, which was initially diagnosed as a primary CNS neoplasm, was reclassified as a pituitary adenoma and is thus also included in the present study (Table 1). An additional number of 15 randomly chosen cases of primary CNS tumors were kindly provided from the Department of Veterinary Pathology, Justus-Liebig-University, Giessen, Germany, and 18 additional cases were allocated from the Department of Veterinary Bioscience, Ohio State University, Columbus, Ohio. The case collection finally resulted in a total number of 97 canine CNS neoplasms, which are included in the present study (Table 1).
Canine Central Nervous System Tumors Investigated Using Tissue Microarray.
Representative tumor areas were selected for histology and fixed in 10% formalin for at least 24 hours. Subsequently, tissue was routinely processed in paraffin wax, cut at 3-μm thickness, and stained with HE. Histologic diagnosis of each neoplasm was done by board-certified veterinary pathologists (W.B., P.W.) according to the recent international WHO classification for tumors of the nervous system of domestic animals.
Tissue Microarray
The validity of TMA has been demonstrated on some representative cases of the included tumors in an earlier study, which reported no significant differences between histologic and immunohistochemical findings in whole tissue sections as compared with TMA spots. 39
For the global investigation of 97 canine intracranial neoplasms, TMA assembly was performed as described in an earlier study. 39 Briefly, representative tumor areas were microscopically selected on HE-stained sections and highlighted with a permanent marker on the section and similarly on the respective donor block. Areas presenting hemorrhage, necrosis, and calcification were strictly excluded from further investigations. Subsequently, 2 cylindrical core samples of 1.2 mm in diameter were manually taken with a punch needle from the center and periphery of each paraffin wax–embedded neoplasm. 39 In total, 5 TMA blocks, each of them containing 60 punch biopsies, including appropriate negative control tissue such as normal canine cerebrum and cerebellum, were commercially constructed (Zytomed, Berlin, Germany). The 2 cores from the same tumor were located in direct proximity on the respective TMA block. One single TMA was prepared with tissue specimen as positive controls. On this TMA section, canine liver, large intestine, kidney, spleen, lymph node, skin, cerebrum, cerebellum, 1 squamous cell carcinoma, and a lymphoma were included. Finally, 3-μm serial sections were cut from the TMAs, collected onto coated slides (Superfrost Plus slides, Menzel Co., Braunschweig, Germany), stained with HE, or underwent further processing for immunohistochemistry.
TMAs included a total case number of 97 canine CNS neoplasms (Table 1). For the present study, cases were grouped into gliomas (n = 40), meningiomas (n = 16), plexus tumors (n = 15), ependymomas (n = 7), lymphoma (n = 3), medulloblastoma (n = 3), germ cell tumors (n = 3), peripheral nerve sheath tumors (PNSTs; n = 3), histiocytic sarcomas (n = 2), undifferentiated carcinomas (n = 2), an intraspinal nephroblastoma (n = 1), 1 case of microgliomatosis (n = 1), and a pituitary adenoma (n = 1). The glioma group was composed of astrocytomas (n = 16, including glioblastoma), oligodendroglioma (n = 23), and an oligoastrocytoma (n = 1). Moreover, based on the initial histologic observation of the degree of nuclear pleomorphism, cellularity, and overall cellular differentiation, astrocytomas, oligodendrogliomas, and ependymomas were subgrouped into anaplastic and nonanaplastic types.
Immunohistochemistry
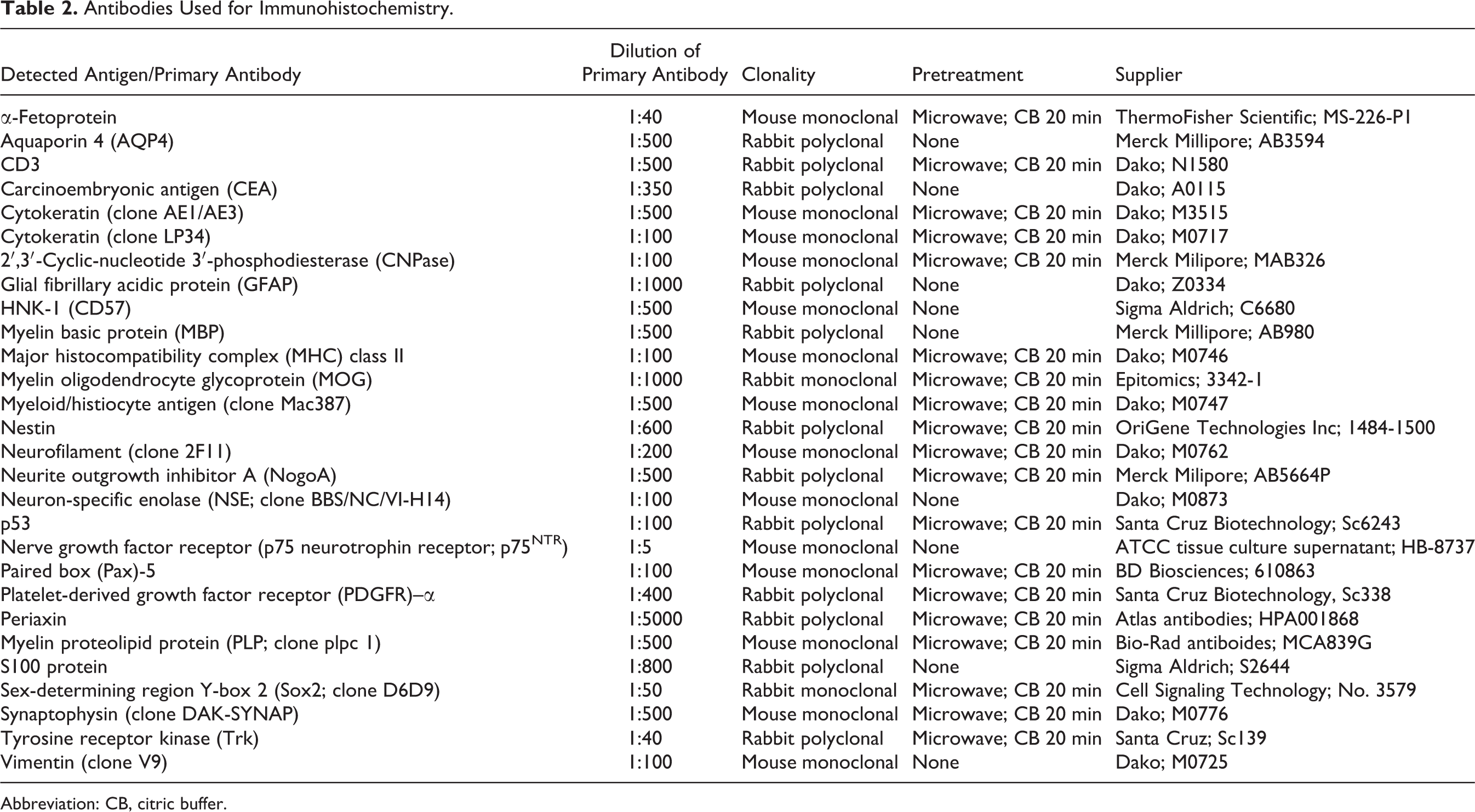
To evaluate the expression profile of the neoplasms, 28 monoclonal and polyclonal antibodies (Table 2) were applied for immunohistochemistry as previously described. 39 Briefly, unstained TMA sections were dewaxed in Roticlear (Roth, Karlsruhe, Germany) and isopropanol for 5 minutes, respectively, and rehydrated in a descending series of alcohol. Endogenous peroxidase activity was blocked by a 30-minute incubation in 0.5% H2O2 in methanol. Antibody retrieval was performed depending on the primary antibody, followed by blocking of unspecific protein binding with normal goat or horse serum diluted 1 in 5 in phosphate-buffered saline (PBS) for 20 minutes. The primary antibody was added in the respective dilution (Table 2) and incubated overnight at 4°C. Subsequently, a secondary biotin-labeled antibody (goat-anti-mouse and goat-anti-rabbit, respectively) was applied at a dilution of 1 in 200 in PBS for 30 minutes at room temperature, followed by incubation with avidin-biotin-peroxidase complex (ABC; Vector Laboratories, Burlingame, CA). Visualization of the reaction was done by incubation with fresh and filtered 3, 3′-diaminobenzidine tetrahydrochloride (DAB; Sigma Aldrich, Taufkirchen, Germany) and 0.05% H2O2 for 10 minutes at room temperature. Finally, slides were slightly counterstained with Mayer’s hematoxylin, dehydrated in an ascending series of alcohol, cleared in acetic acid-n-butylester (EBE, Roth, Karlsruhe, Germany), and mounted using Roti-Histokit (Roth). Between each reaction step, the slides were washed threefold with PBS. To prevent dehydration during processing, slides were kept in a humidified chamber (Coverplates Sequenza, Pittsburgh, PA). For appropriate negative controls, the primary antibody was substituted by the following sera: ascites fluid from Balb/cJ mice (dilution 1 in 1000 in PBS with 1% bovine serum albumin) for monoclonal antibodies and rabbit serum (dilution 1:3000 in PBS with 1% bovine serum albumin) for polyclonal antibodies.
Antibodies Used for Immunohistochemistry.
Abbreviation: CB, citric buffer.
Semiquantitative Evaluation, Hierarchical Clustering, and Statistical Analysis
The immunohistochemical analysis of TMA sections was performed using a semiquantitative scoring system ranging from 0 to 3 points, in which 0 points indicated no immunopositive tumor cells, 0.5 points 5–10% positive tumor cells, 1 point 10–50% positive tumor cells, 2 points 50%–90% positive tumor cells, and 3 points >90% positive tumor cells. For each neoplasm, 2 cylindrical tissue samples, from the center and the periphery of the tumor, were investigated separately. The positivity scores were given as the mean value of points of both TMA spots according to the above-mentioned scoring system. The obtained data were directly registered in tabular worksheets (Microsoft Excel 2010, Microsoft Corporation, Redmond, WA) and further processed for hierarchical clustering analysis with complete linkage and default distance settings (Genesis, Institute for Genomics and Bioinformatics, Graz University of Technology, Austria). Clustering arranged tumors and markers according to similarities and overlaps in their expression profiles. In addition, a statistical groupwise comparison of the proportion of immunopositive and immunonegative tumor types (including different types of glioma, meningioma, ependymoma, plexus tumors) and dignity (anaplastic versus nonanaplastic) was assessed for each marker using multiple Fisher exact tests applying GraphPad Prism 5.0 (GraphPad Software Inc., La Jolla, CA). For these analyses, tumors were judged positive if they at least had an immunopositivity score of 0.5, and all tumors with score 0 were defined as negative. Statistical significance was designated as P ≤ .05.
Results
Hierarchical Clustering Analysis
Five of the 97 cases had to be excluded from hierarchical clustering analysis because of insufficient immunohistochemical data (i.e., loss of too many tissue discs during preparation). Figure 1 visualizes a clustered heat map of 2576 (92 tumors times 28 markers) distinct spots corresponding to each single semiquantitative score value. Following hierarchical clustering analysis, 92 canine CNS neoplasms were visually grouped into 6 distinct clusters (A–F) according to the degree of similarities in their immunohistochemical positivity score profiles (Fig. 1).
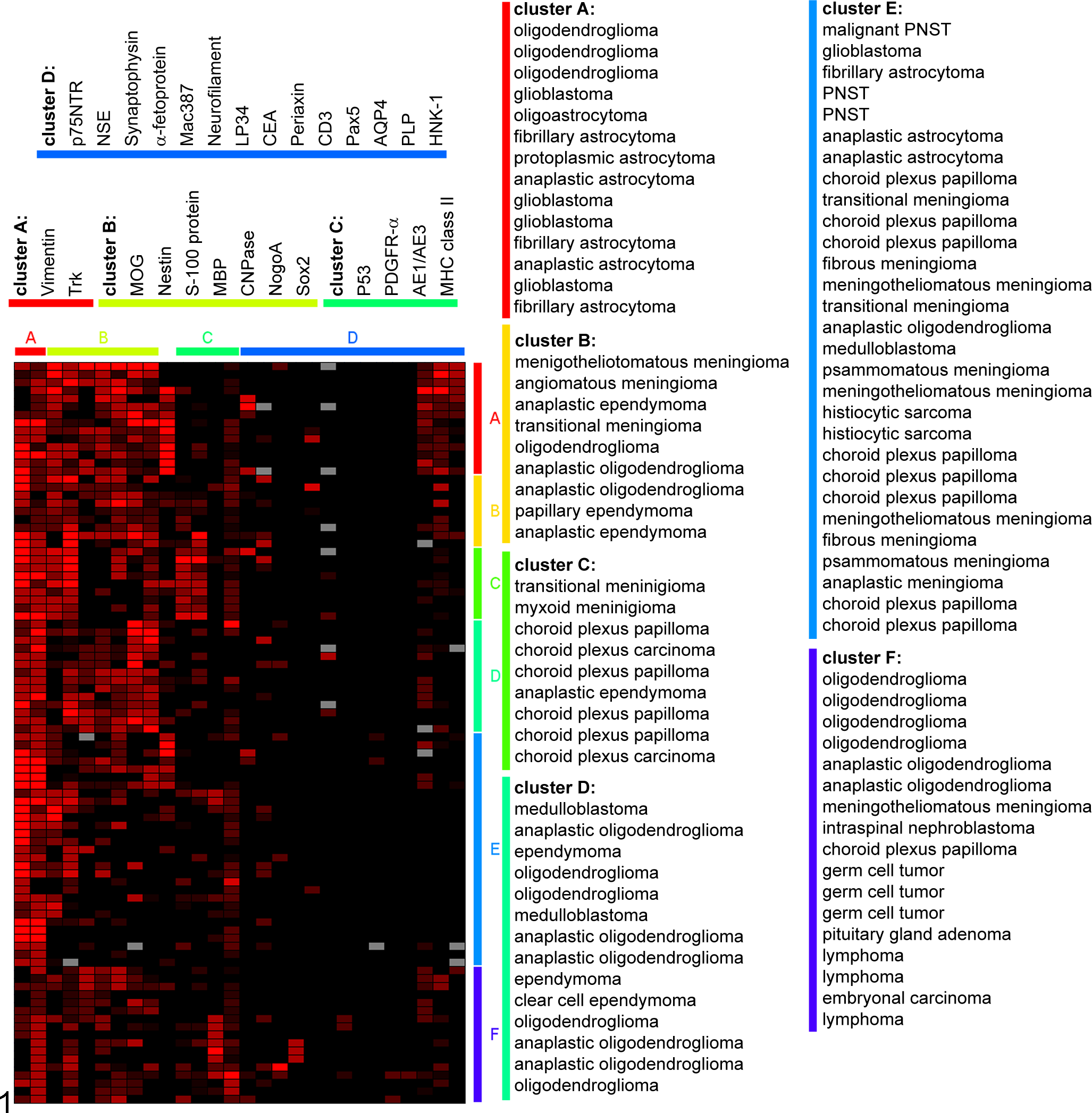
Hierarchical clustering analysis of 92 canine central nervous system tumors investigated on tissue microarray (TMA) sections in the present study. The different shades of red coloration in the spots indicate increasing semiquantitative expression scores (0.5 [pale red]–3 [bright red]), whereas the black staining represents absent expression of the respective antigen (semiquantitative score 0). The gray spots in the figure depict TMA spots where the evaluation was not possible because of loss of the respective tissue discs during immunohistochemical processing. Clustering was performed with the software Genesis (Institute for Genomics and Bioinformatics, Graz University of Technology, Austria) and resulted in 6 clusters, which grouped the tumors (A–F; right side), and 4 clusters, which grouped the markers (A–D; upper side), according to similarities in their immunopositivity profiles. Glial fibrillary acidic protein (GFAP) was not grouped into a cluster (spots between marker clusters B and C). AE1/AE3, clone for cytokeratin; AQP4, aquaporin 4; CEA, carcinoembryonic antigen; CNPase, 2′,3′-cyclic-nucleotide 3′-phosphodiesterase; HNK-1, CD57; LP34, clone for cytokeratin; Mac387, clone for myeloid/histiocyte antigen; MBP, myelin basic protein; MHC class II, major histocompatibility complex class II; MOG, myelin oligodendrocyte glycoprotein; NSE, neuron-specific enolase; p75NTR, p75 neurotrophin receptor; Pax5, paired box 5; PDGFR-α, platelet-derived growth factor receptor alpha; PLP, proteolipid protein; PNST, peripheral nerve sheath tumor; Sox2, sex-determining region Y-box 2; Trk, tyrosine kinase receptor.
Cluster A contained 14 of 37 gliomas (10 astrocytomas, 3 oligodendrogliomas, and 1 oligoastrocytoma), with most of the astrocytomas exhibiting anaplasia (n = 6), whereas the oligodendrogliomas exhibited no anaplasia. In cluster B, 9 tumors with an equal distribution of meningioma, ependymoma, and oligodendroglioma were present, and 6 of these tumors were nonanaplastic. Cluster C comprised 9 cases including 6 choroid plexus tumors, 2 meningiomas, and 1 ependymoma. Only 2 of these plexus tumors were carcinomas. In cluster D, 9 oligodendrogliomas (nonanaplastic, n = 4; anaplastic, n = 5), 3 ependymomas, and 2 medulloblastomas were clustered. The large cluster E contained 29 heterogenous cases including 10 meningiomas, 8 plexus tumors, 4 astrocytomas, 3 PNSTs, 2 histiocytic sarcomas, 1 medulloblastoma, and 1 oligoastrocytoma. In this group, only 1 meningioma showed anaplasia, whereas the other 9 meningiomas were nonanaplastic. All of the plexus tumors in cluster E were papillomas. However, 3 of the 4 astrocytomas in cluster E had an anaplastic phenotype. Cluster F included 17 neoplasms: 6 oligodendrogliomas, 3 germ cell tumors, 3 lymphomas, 1 meningioma, 1 nephroblastoma, 1 plexus tumor, 1 pituitary gland adenoma, and 1 embryonal carcinoma.
Summarized, gliomas were distributed in 5 of 6 clusters with oligodendrogliomas in group A (14%), B (14%), D (41%), E (5%), and F (27%), whereas astrocytomas were clustered exclusively in cluster A (71%) and E (29%). Meningiomas were assorted to clusters B (19%), C (13%), E (63%), and F (6%), with one anaplastic meningioma in cluster E. Plexus tumors clustered in groups C (40%), E (53%), and F (7%) with both choroid plexus carcinomas (13%) in cluster C. The ependymomas were distributed throughout clusters B (43%), C (14%), and D (43%), with 43% of anaplastic ependymomas arranged in clusters B and C. The miscellaneous tumors were distributed over clusters D (2%), E (35%), and F (53%).
Analogous to the individual tumor cases, the applied antibodies were clustered according to overlaps in their immunopositivity score profiles (Fig. 1). Hierarchical cluster analysis separated the markers into 4 clusters (A–D). Cluster A contained only vimentin and tyrosine receptor kinase (Trk), which were expressed by the vast majority of the investigated tumors (Fig. 1). Cluster B contained 7 investigated antigens including myelin oligodendrocyte glycoprotein (MOG), nestin, S100 protein, myelin basic protein (MBP), cyclic nucleotide phosphodiesterase (CNPase), NogoA, and the transcription factor Sox2. Cluster C contained 4 antigens: the tumor suppressor p53, platelet-derived growth factor receptor–α, the cytokeratin cocktail (clone AE1/AE3), and major histocompatibility complex class II. Cluster D contained the following antigens: p75 neurotrophin receptor (NTR), neuron-specific enolase, synaptophysin, α-fetoprotein, myeloid/histiocyte antigen (clone Mac387), neurofilament, another cytokeratin cocktail (clone LP34), carcinoembryonic antigen, periaxin, CD3, Pax5, aquaporin (AQP) 4, proteolipid protein (PLP), and human natural killer–1. Glial fibrillary acidic protein (GFAP) was not grouped into a cluster because of low overlaps in the immunopositivity scores compared with the other markers (Fig. 1).
Statistical Analysis
A groupwise comparison for each marker was done for the tumor groups and within each group for different dignities (anaplastic vs nonanaplastic) using multiple Fisher exact tests in order to identify markers with discriminative potential. Multiple markers were able to distinguish between the different tumor entities including gliomas, meningiomas, ependymomas, and plexus tumors, respectively (Table 3, Suppl. Table 1). GFAP, S100 protein, AQP4, and p75NTR significantly discriminated between astrocytomas and oligodendrogliomas (P < .05; Figs. 2–9, Table 3, Suppl. Table 1). Fifteen of 16 investigated astrocytomas (>90%) expressed GFAP (Fig. 2), while GFAP was expressed by only 5 of the 23 (22%) investigated oligodendrogliomas (Fig. 3). In contrast, a relatively lower proportion of astrocytomas was immunopositive for S100 protein (8 of 16 positive [50%]; Fig. 4) as compared with a higher proportion of oligodendrogliomas (21 of 23 [91%]) being positive for this protein (Fig. 5).
Suggested Markers for the Differentiation Between Combinations of Common Morphological Differential Diagnoses of Canine Central Nervous System Neoplasms Based on Significanta Differences in the Proportions of Immunopositive and Immunonegative Tumors in the Present Tissue Microarray Study.

Abbreviations: AE1/AE3, clone for cytokeratin; AQP4, aquaporin 4; CNPase, 2′,3′-cyclic-nucleotide 3′-phosphodiesterase; GFAP, glial fibrillary acidic protein; MBP, myelin basic protein; MHCII, major histocompatibility complex class II; MOG, myelin oligodendrocyte protein; NSE, neuron-specific enolase; PLP, proteolipid protein; Sox2, sex-determining region Y-box 2.
a As determined by multiple Fisher exact tests.
b Glioma includes both oligodendrogliomas and astrocytomas.
c Hematopoietic tumor includes malignant lymphoma and histiocytic sarcoma.
d Embryonic tumor includes medulloblastoma, germ cell tumor, and intraspinal nephroblastoma
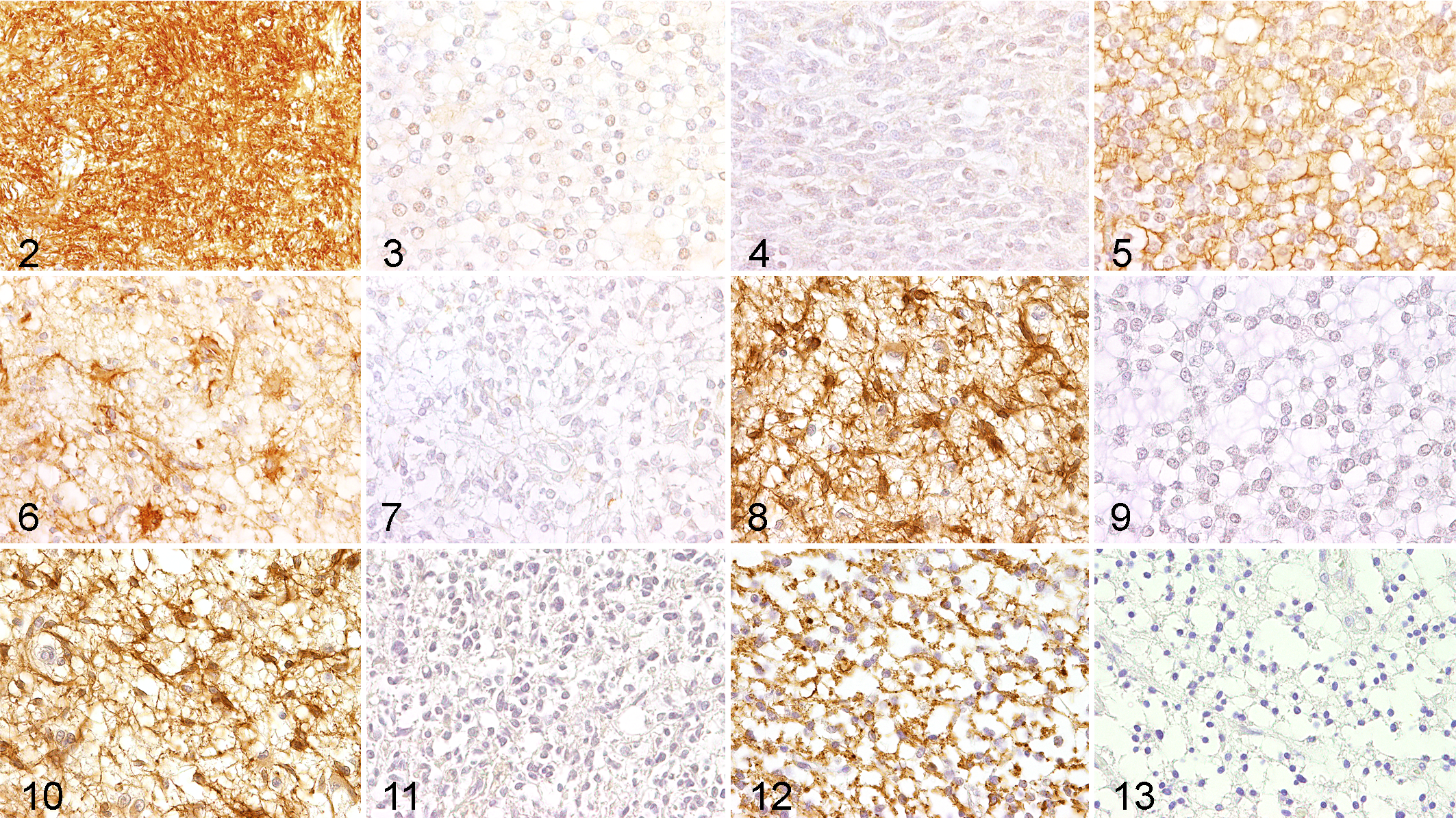
Similar to GFAP, the vast majority (15 of 16; 94%) of the investigated astrocytomas expressed AQP4 (Fig. 6), while AQP4 was expressed by 14 of 23 (61%) oligodendrogliomas (Fig. 7). Even though p75NTR was expressed by only 4 of 16 (25%) of the investigated astrocytomas (Fig. 8), not a single oligodendroglioma was immunopositive for this marker (Fig. 9). Interestingly, the neurotrophin receptor p75NTR was exclusively expressed in nonanaplastic astrocytomas compared with anaplastic counterparts (P = .02; Figs. 10, 11). Of the nonanaplastic oligodendrogliomas, 8 of 13 (62%) were PLP positive (Fig. 12). However, only 2 of 10 (20%) anaplastic oligodendrogliomas were positive for PLP (Fig. 13). A Fisher exact test revealed a significant difference in the proportion of PLP-expressing anaplastic oligodendrogliomas versus nonanaplastic counterparts (P = .04).
Figure 14 illustrates differences in the percentages of immunopositive astrocytomas versus oligodendrogliomas for the discriminative markers GFAP, S100 protein, AQP4, and p75NTR.
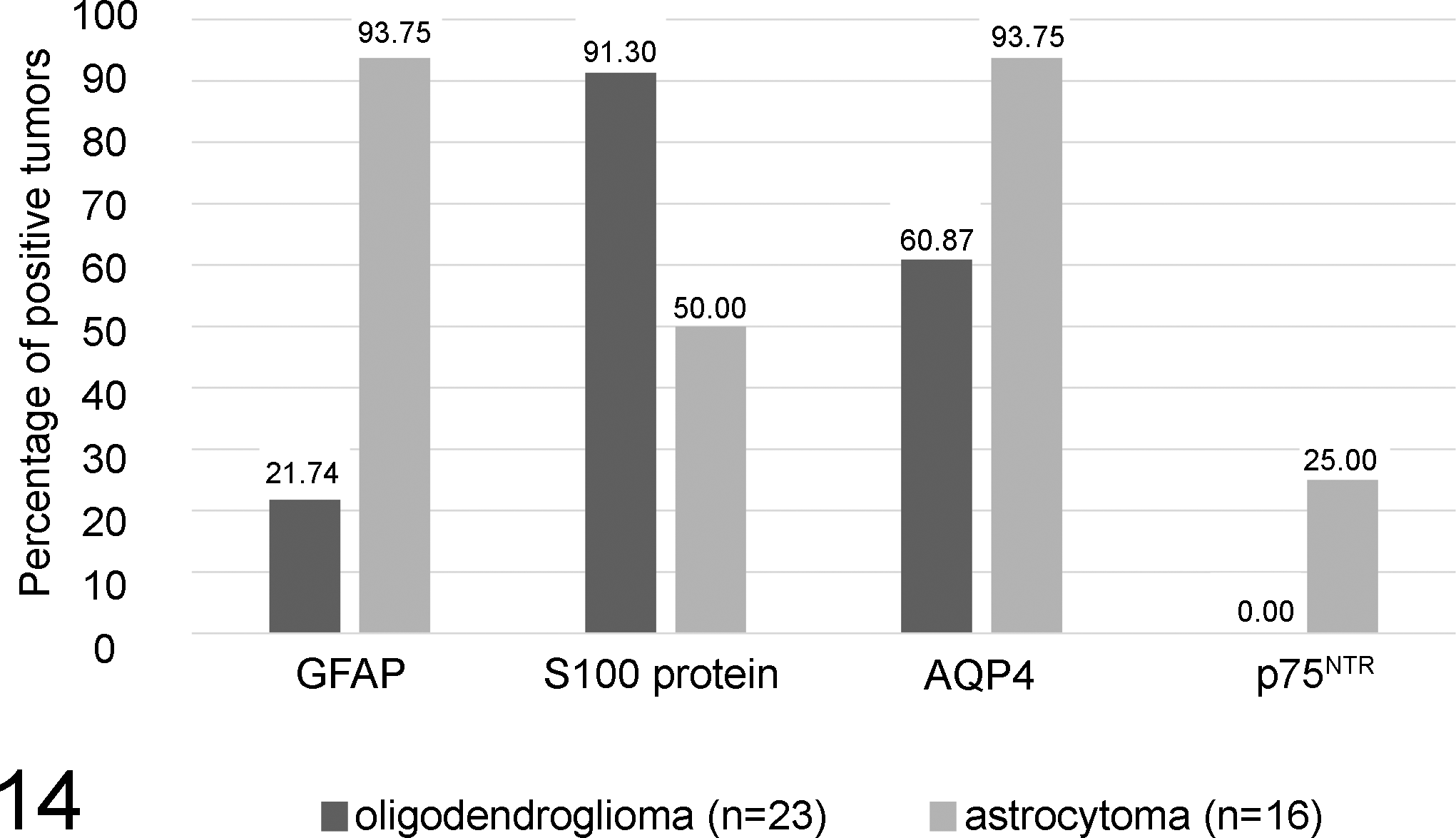
Scale bars illustrating the percentage of immunopositive astrocytomas compared with oligodendrogliomas for the markers glial fibrillary acidic protein (GFAP), S100 protein, aquaporin 4 (AQP4), and p75 neurotrophin receptor (p75NTR). Tumors were judged positive if they exhibited a minimum semiquantitative score of 0.5 (at least 5%–10% of tumor cells positive), while all tumors with score = 0 (no tumor cells positive) were defined as negative. Note the relatively higher proportions of astrocytomas being positive for GFAP, AQP4, and p75NTR as compared with oligodendrogliomas. P75NTR expression was absent in oligodendrogliomas, whereas the other markers were variably expressed in a relatively lower proportion of oligodendrogliomas. In contrast, S100 protein is expressed by a high proportion of oligodendrogliomas compared with astrocytomas.
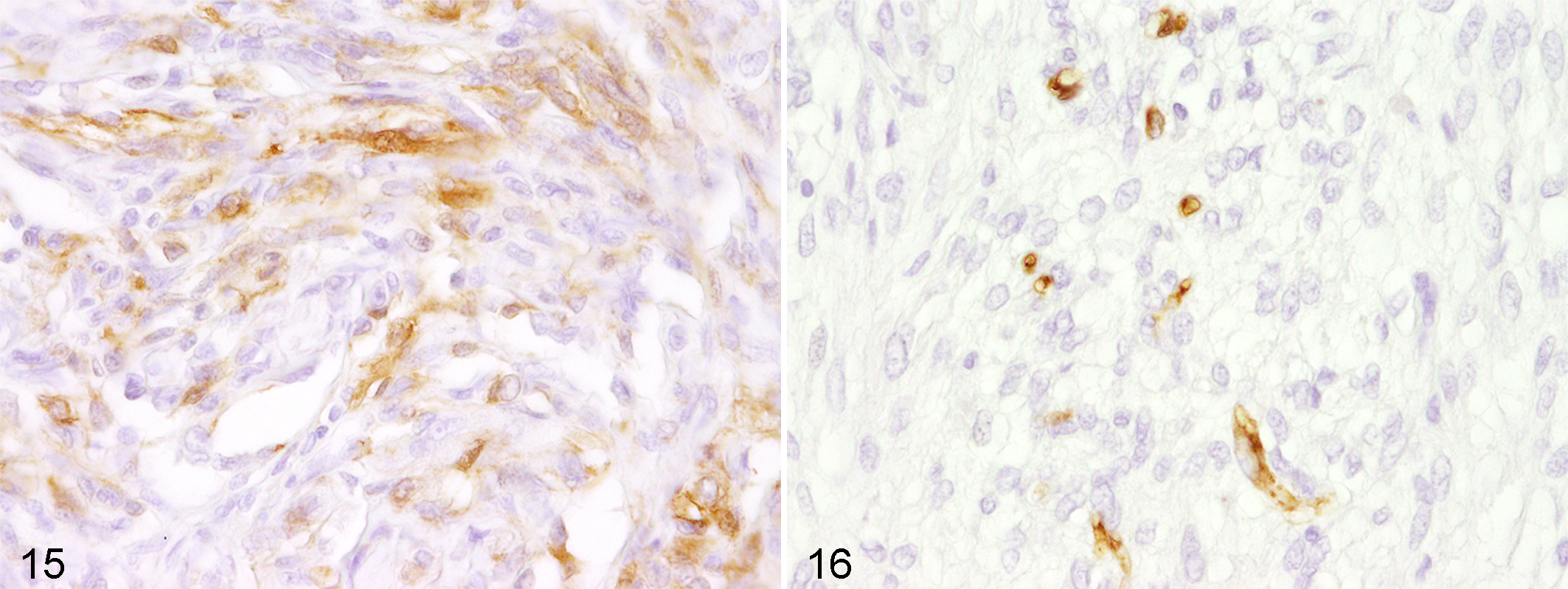
Interestingly, periaxin, a marker for myelinating Schwann cells, 30 was expressed in 2 of 3 PNSTs (Figs. 15, 16). None of the other 94 neoplasms expressed this antigen.
Discussion
In this study, TMA was used for the global analysis of antigen expression patterns in various canine central nervous system neoplasms. Even though HE staining represents the gold standard for initial tumor diagnosis in veterinary diagnostics, immunohistochemistry displays a helpful tool for certain diagnostic settings. Here, immunohistochemical analyses might, for instance, help to predict tumor dignity, to exclude differential diagnoses, and to investigate biopsy margins. 13 Using TMAs, whose general suitability for veterinary tumor research has been validated, 22,39 this study gives new insights into phenotypes of canine CNS neoplasms.
In the present study, hierarchical clustering analysis was used to group tumors with similar expression profiles and markers with similar distribution among the tumors, respectively. Hierarchical clustering analysis represents a tool that is, for instance, commonly used in large-scale transcriptomic studies. 36 However, clustering analyses have also been applied for immunohistochemical data on tissue arrays of human CNS neoplasms. 11 In fact, the amount of data generated by TMA is relatively huge, thus requiring analytical methods, originally known from large-scale proteomic and transcriptomic studies. In the present study, hierarchical clustering analysis was demonstrated to be a suitable tool to illustrate similarities and differences in the expression profile of the investigated tumors and markers, respectively. In an ideal setting, one would expect all tumors of a similar diagnosis to be within a single cluster and tumors of another entity within a different cluster. However, the relative heterogeneity and the high overlap of the immunohistochemical expression profiles in this analysis (i.e., tumors with different histopathological diagnosis exhibiting relatively similar immunohistochemical profiles and thus clustering together) clearly demonstrate that HE staining remains the gold standard for a definite diagnosis and that immunohistochemistry alone, even with a multitude of markers, is not sufficient for a definite diagnosis. In fact, for a correct histopathological diagnosis, the entire morphological and phenotypical characteristics have to be taken into account, including special features such as rosette formation and other patterns. In particular, meningiomas and gliomas exhibited a relatively high phenotypical heterogeneity compared with other tumor categories. This is especially true for oligodendrogliomas, which were distributed among 5 of 6 total clusters, thus suggesting that they represent a neoplastic entity with a relatively heterogeneous phenotypical pattern. In contrast, astrocytomas were clustered in only 2 of 6 clusters, suggesting comparatively higher immunophenotypical homogeneity as compared with oligodendrocytic tumors.
However, clustering of markers resulted in an expected grouping of myelin-related proteins such as MOG, MBP, CNPase, and NogoA, which showed similar expression patterns among the tumors. Likewise, CD3, myeloid/histiocyte antigen, and Pax5 were in a single cluster, all representing markers for lymphoid cells. The remaining applied antibodies were haphazardly distributed among the marker clusters. While the markers among the first 3 clusters revealed relatively high expression levels in multiple tumors, markers from the fourth cluster were only rarely expressed throughout the neoplasms, potentially suggesting a higher specificity of these markers for distinct neoplastic entities. Vimentin and tyrosine receptor kinase (Trk; cluster A) were expressed by the vast majority of the investigated tumors (Fig. 1), suggesting that these markers have a relatively low specificity. In a second approach, we conducted multiple Fisher exact tests to identify markers with discriminative potential among the tumors. The major drawback of this approach is that if multiple tests are done simultaneously, an inflation of the overall type I error may occur. However, a correction of multiple testing is usually not performed on TMA data, and thus few false-positive results among the tests are generally accepted. 11 The analysis pointed out that multiple markers were able to discriminate between tumor groups (Suppl. Table 1). For instance, GFAP, S100 protein, AQP4, and p75NTR significantly discriminated between canine astrocytomas and oligodendrogliomas.
GFAP is the principal intermediate filament of mature astrocytes and is expressed in normal as well as in neoplastic astrocytes. 10,23 Consistent with previous observations, a higher proportion of astrocytomas was positive for GFAP as compared with oligodendrogliomas in the present study. This is in overall concordance with numerous reports which describe a high GFAP expression in astrocytomas but infrequent expression in oligodendrogliomas. 10,13,19,25,37 These results emphasize GFAP as an appropriate and readily available marker for the diagnostic differentiation of astrocytomas from oligodendrogliomas. In fact, GFAP positivity is a long-accepted means of diagnosis of astrocytoma, 13 even though GFAP expression is not unique to astrocytomas.
S100 protein is a dimeric calcium-binding protein, commonly found within the nervous system but also expressed in a variety of extraneuronal tissues. 8,25 In the present study, a significantly higher proportion of oligodendrogliomas was positive for S100 protein as compared with astrocytomas. This is consistent with a previous report on S100 protein expression in an oligodendroglioma from a dog. 27 Likewise, veterinary case reports of oligodendrogliomas in different species such as white-tailed deer, cow, and African lion similarly report immunopositivity for S100 protein. 7,15,35 However, canine 13 as well as human astrocytomas may similarly exhibit S100 protein immunopositivity to a variable extent, 5 which is in concordance with the present study, in which S100 protein was expressed by 50% of the investigated astrocytomas.
AQP4 is a key molecule involved in the maintenance of cellular water and ion homeostasis in the central nervous system. 4 The balance of the blood-brain barrier depends on astroglial polarity, which is directly influenced by AQP4 and other molecules such as dystroglycan, agrin, and matrix metalloproteinases. 26,40 In human glioblastoma multiforme, a dislocalization and redistribution of AQP4 has been confirmed as a major reason for the development of vasogenic edema and an increase of intracerebral pressure. 26,40 In the present study, AQP4 was expressed in a higher proportion of canine astrocytomas (94% of astrocytomas were positive) compared with oligodendrogliomas. However, nearly 61% of the latter also expressed AQP4. Thus, the present results suggest that AQP4 might, within these limitations, represent a suitable supplemental marker for the differentiation of astrocytomas from oligodendrogliomas. Interestingly, this is in concordance with a TMA study on human brain tumors showing a similar discrimination regarding AQP4 expression in oligodendrogliomas and astrocytomas, respectively. 11
The transmembrane glycoprotein p75NTR is a member of the tumor necrosis factor receptor superfamily and was originally isolated as a nerve growth factor receptor that is presumably involved in glioma migration and invasion. 14 Despite the fact that oligodendrocytes have previously been reported to be capable of expressing p75NTR, 17 in the present study, positivity for p75NTR was not observed in any oligodendroglioma, thus suggesting that this marker allows a clear discrimination between canine oligodendroglioma and astrocytoma. Moreover, the present results highlight that p75NTR is exclusively expressed in nonanaplastic astrocytomas compared with anaplastic astrocytomas, thus indicating a correlation between increased expression levels of p75NTR and a relatively higher differentiation of canine astroglial tumor cells. Likewise, an inverse association of p75NTR expression during malignant transformation of human prostate cancer cells has been reported. 16,28 In fact, in human prostate cancer, p75NTR has been proposed as a metastasis suppressor. 16,28
PLP is one of the major proteins of CNS myelin that is mainly expressed in mature oligodendrocytes. 1,24 PLP is reported to correspond to 50% of myelin proteins in normal CNS tissue and is functionally involved in the maintenance of myelin sheath stability. 1 Gene expression levels of MBP and PLP in human oligodendrogliomas are significantly higher compared with astrocytomas. 6 The significantly higher expression of PLP in nonanaplastic oligodendrogliomas compared with anaplastic oligodendrogliomas, as demonstrated in the present study, suggests that a higher degree of cellular differentiation is associated with PLP immunopositivity in canine oligodendrogliomas.
Periaxin represents a protein that is expressed by myelinating Schwann cells. 30 This myelin sheath–stabilizing protein is reported to be expressed relatively early during both Schwann cell–mediated myelination and remyelination and occurs prior to other proteins of the peripheral myelin sheath. 30 In the present study, periaxin was expressed in 2 of 3 PNSTs, whereas none of the other investigated 94 neoplasms expressed this marker. Despite the low number of investigated cases, these results indicate a considerable specificity of periaxin for canine PNSTs. However, further investigations are needed to support these findings. Underlining this observation, in Tasmanian devils, periaxin represents a highly specific marker for the transmissible devil facial tumor, and transcriptome analyses have demonstrated Schwann cell origin of this neoplastic entity. 21,34
Conclusion
Even though HE staining remains the gold standard for definite diagnosis of CNS tumors in domestic animals, immunohistochemistry is doubtlessly of high value for certain aspects of diagnostic neuro-oncologic veterinary pathology. However, the results of the hierarchical clustering analysis also demonstrate that there is a considerably high overlap in the immunohistochemical expression profile of morphologically distinct canine CNS tumor entities, suggesting a certain degree of antigenic or even histogenic relatedness.
Besides well-known and established markers for CNS tumor diagnosis such as GFAP and S100 protein, this study highlights AQP4, p75NTR, and periaxin as novel and promising candidates, broadening the panel of markers for the diagnostic immunohistochemical differentiation of canine CNS neoplasms. In particular, in addition to the well-known marker GFAP, AQP4 and p75NTR might represent useful supplemental markers for the differentiation of canine astrocytomas from oligodendrogliomas. High expression levels of PLP might indicate a higher degree of differentiation in canine oligodendrogliomas. Likewise, positivity for p75NTR might indicate a higher degree of differentiation in canine astrocytomas. Moreover, the present results suggest periaxin to represent a useful marker for the detection of canine PNSTs, which, however, needs to be confirmed in future studies.
Lastly, TMAs demonstrated to be a highly suitable methodological approach for the simultaneous, cost-effective, and time-saving immunohistochemical phenotyping of a multitude of different canine CNS neoplasms, although loss of some tissue discs during staining procedures has to be anticipated. The present results emphasize TMAs as a promising and useful tool for future investigations in a veterinary pathology setting, such as immunophenotyping of other neoplasms and complementing the panel of novel markers, respectively.
Footnotes
Acknowledgements
We gratefully thank Professor Emeritus Steven E. Weisbrode (Department of Veterinary Biosciences, The Ohio State University, Columbus, OH) for providing tissue from cases included in this study. Franziska Heinrich received financial support from the German National Academic Foundation (Bonn, Germany). We are thankful to Petra Grünig, Bettina Buck, Kerstin Schöne, Caroline Schütz, Christiane Namneck, and Claudia Hermann for excellent technical support.
Author contribution
F. Heinrich and I. Spitzbarth contributed equally to this work and should be considered as co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
