Abstract
The expression of cell differentiation and proliferation markers of canine neuroepithelial tumors was examined immunohistochemically to identify the histogenesis of these tumors. Astrocytomas (n = 4) consisted of cells positive for glial fibrillary acidic protein (GFAP) and nestin and a few cells positive for doublecortin (DCX). Immunoreactive cells for receptor tyrosine kinases (epidermal growth factor receptor and c-erbB2) and their downstream molecules (phospho-extracellular signal-regulated kinase 1/2 and phospho-Akt) were often detected in astrocytomas, especially in medium- and high-grade tumors. Gliomatosis cerebri (n = 3) consisted of cells positive for ionized calcium–binding adaptor molecule 1 and GFAP, including a minor population of cells positive for nestin, DCX, and beta III tubulin, suggesting their glial differentiation. In choroid plexus tumors (n = 4), most tumor cells were positive for cytokeratins AE1/AE3 and 18, and few were positive for GFAP. The majority of cells of oligodendrogliomas (n = 5) were DCX positive, but the tumors also contained minor populations of cells positive for GFAP, nestin, or beta III tubulin. Primitive neuroectodermal tumors (PNETs; n = 2) consisted of heterogeneous cell populations, and the tumor cells were positive for nestin, beta III tubulin, and DCX, suggesting glial and neuronal differentiation. The major population of neuroblastoma cells (n = 3) were positive for beta III tubulin and DCX, suggesting single neuronal differentiation. As for antiapoptotic cell death molecules, most tumor cells in the choroid plexus tumors, PNETs, and neuroblastomas were intensely positive for Bcl-2 and Bcl-xL, whereas those in gliomatosis cerebri were almost negative. In astrocytomas, Bcl-xL-positive cells predominated over Bcl-2-positive cells, but the opposite was observed in oligodendrogliomas. The immunohistochemical results were analyzed by hierarchical clustering, and the constructed dendrogram clearly indicated a novel position of oligodendrogliomas: the primitive glial and neuronal differentiation.
According to the World Health Organization (WHO) classification of brain tumors of domestic animals, 18 neuroepithelial tumors are first classified into the following 8 groups: astrocytic tumors, oligodendroglial tumors, other gliomas, ependymal tumors, choroid plexus tumors, neuronal and mixed neuronal–glial tumors, embryonal tumors, and pineal parenchymal tumors. Of domestic animals, most neuroepithelial tumors are observed in dogs. 36 There is a report concerning the correlation between immunophenotype/genotype and the expression of p53 and EGFR genes in canine astrocytomas 35 ; however, the data concerning the expression of such markers that further characterize histogenesis of canine neuroepithelial tumors have not yet been reported. We therefore examined the expression of relevant markers that characterize the histogenesis of canine neuroepithelial tumors by immunohistochemistry to identify the histogenesis of these tumors.
To examine candidate molecules that determine putative glial or neuronal lineage specificity, selected cell differentiation markers were examined—including glial fibrillary acidic protein (GFAP), nestin, neurofilament (NF), beta III tubulin, doublecortin (DCX), cytokeratins, and ionized calcium–binding adaptor molecule 1 (Iba1). GFAP is a main intermediate filament protein of mature astrocytes, whereas nestin is a main intermediate filament protein of immature astrocytes. 10,37 Nestin has been extensively used as a marker for neural stem cells to astrocyte progenitors. 23,33 With NF, a distinct marker for mature neurons, the antibody for beta III tubulin has been used to identify the early phase of neuronal differentiation in human embryonic development. 15,16,20 DCX, which plays a crucial role in neuroblast migration, 25 is highly expressed in migrating neural precursor cells in the subventricular zone and rostral migratory stream during the development of the cerebral cortex. 25 Iba1is a macrophage/microglia lineage marker, and the antibody to Iba1has been used to recognize ramified and activated amoeboid microglia in the brain. 13,14 Cytokeratin AE1/AE3 is a broad spectrum of reactivity against high– and low–molecular weight cytokeratins but absent from reactivity to cytokeratin 18, one of the simple epithelial cytokeratins. 3
Expression of proliferation/apoptosis markers—including epidermal growth factor receptor (EGFR), c-erbB2, phospho-extracellular signal-regulated kinase 1/2 (pERK1/2), phospho-Akt (pAkt), p53, Bcl-2, Bcl-xL, cleaved caspase 3, and Ki-67—was also examined to determine the histogenesis of canine neuroepithelial tumors. Of receptors for tyrosine kinases (RTKs), EGFR and c-erbB2 play an important role during tumor transformation in human astrocytic tumors. 1,12,26 The RAS/MAPK pathway, located downstream of RTKs, provides an additional mitogenic stimulus that is often dysregulated in human astrocytic tumors. 28 The PI3K/Akt pathway, also located downstream of RTKs, exerts profound effects on tumor cell survival and antiapoptotic activities. 8,34 In sum, pERK1/2 and pAkt are included in the RAS/MAPK and PI3K/Akt pathways, respectively. Loss or mutation of the p53 tumor suppressor gene occurs in approximately one-third of human astrocytomas, and the loss of wild-type p53 activity increases genomic instability, which appears to accelerate neoplastic progression. 35 Bcl-2, identified in human follicular B-cell lymphoma, is known to function as an antiapoptotic factor by preventing all mitochondrial changes, including a cytochrome c release and a loss of membrane potential. 2,5 Rather than promote cell proliferation, Bcl-xL and Bcl-2 inhibit cell death in a way responsible for impaired apoptosis during tumorigenesis. 5,32 Caspase 3 is the ultimate executioner caspase; it is essential for the nuclear changes associated with apoptosis, and the cleaved activated form is a marker of apotosis. 19 Evaluation of the growth fraction by the Ki-67 proliferative index is highly predictive of the biological behavior in various canine tumors. 31
The aim of the present study is to reveal a characteristic combination profile of the markers in each type of canine neuroepithelial tumor, which will bring new information regarding its histogenesis and classification.
Materials and Methods
Tissue Samples and Histopathology
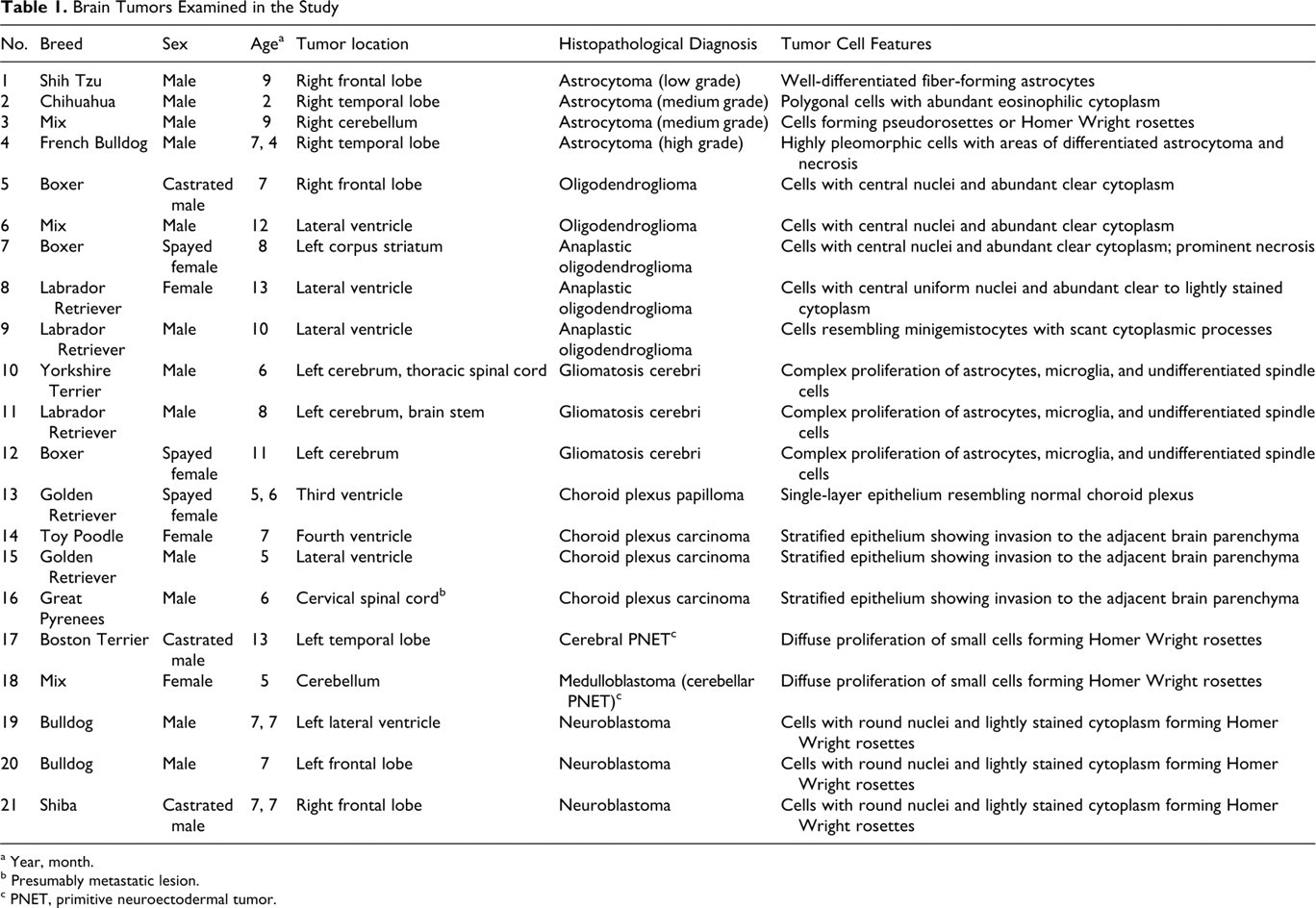
Twenty-one canine neuroepithelial brain tumors were collected from 17 necropsy and 4 surgical cases during the last 5 years (2005–2009). The tumor samples included 4 astrocytomas (1 low grade, 2 medium grade, and 1 high grade), 5 oligodendrogliomas (2 oligodendrogliomas and 3 anaplastic oligodendrogliomas), 3 gliomatosis cerebri, 4 choroid plexus tumors (1 choroid plexus papilloma and 3 choroid plexus carcinomas), 1 cerebral primitive neuroectodermal tumor (cerebral PNET), 1 medulloblastoma (cerebellar PNET), and 3 neuroblastomas. Table 1 shows the breed, sex, age, tumor location, pathological diagnosis, and major histological features of the cases.
Brain Tumors Examined in the Study
a Year, month.
b Presumably metastatic lesion.
c PNET, primitive neuroectodermal tumor.
Tissue samples were routinely fixed in 10% phosphate-buffered formalin solution. Paraffin sections were stained with HE. Histological classification of the brain tumors were determined according to the current WHO histological classification guidelines for domestic animal tumors. 18
Immunohistochemistry
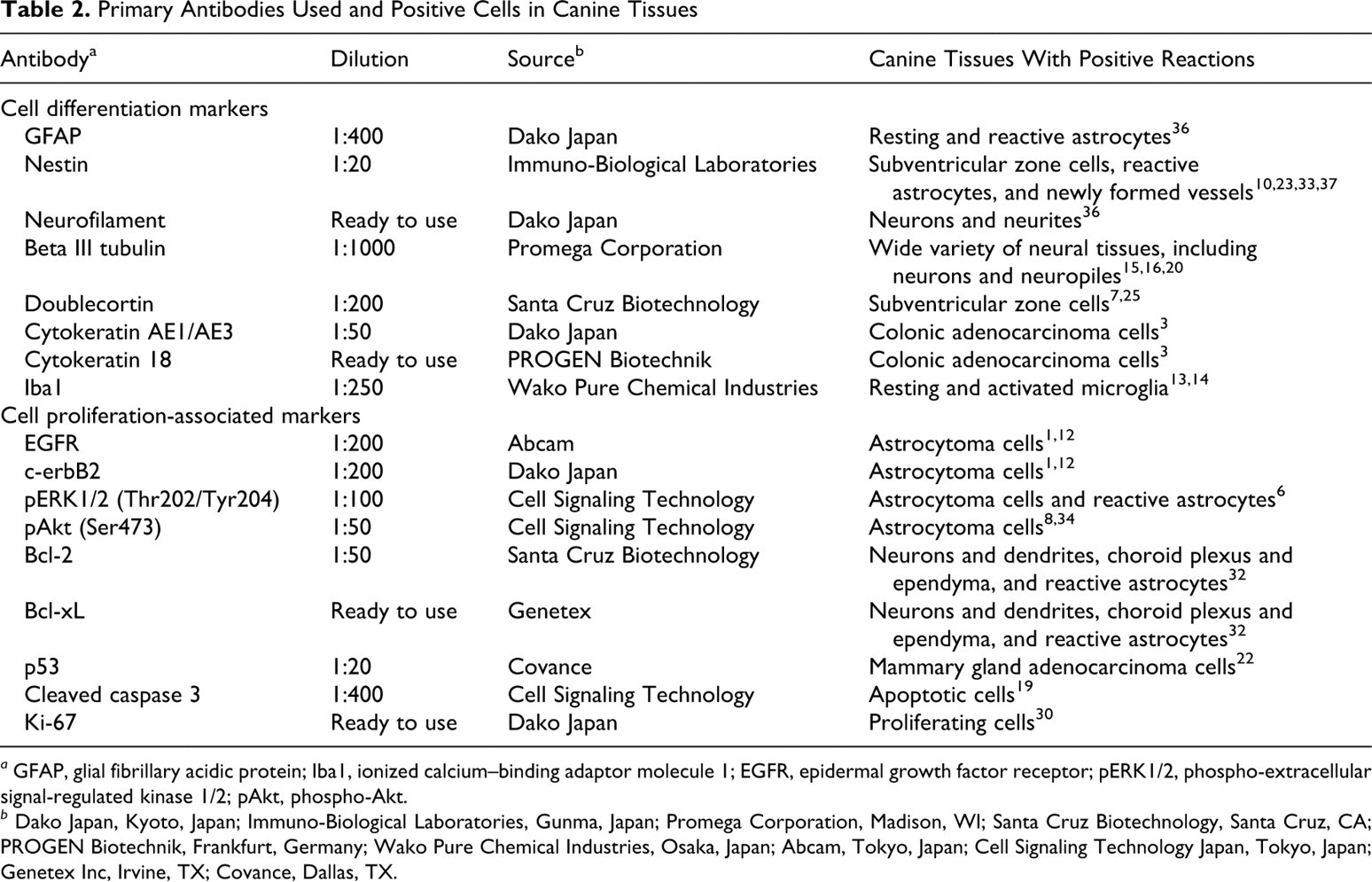
Immunohistochemistry was performed through the labeled streptavidin–biotinylated antibody method. Table 2 lists the primary antibodies and the positive reactions in canine tissues. Deparaffinized sections were first autoclaved at 120°C for 10 minutes in 10mM citrate buffer, pH 6.0, for antigen retrieval except for immunostains for GFAP, nestin, and NF. Tissue sections were then treated with 1% hydrogen peroxide–methanol for 30 minutes and incubated in 8% skim milk–tris-buffered saline at 37°C for 40 minutes to protect nonspecific reactions. The sections were incubated at 4°C overnight with the primary antibodies. Following 3 washings in tris-buffered saline, the sections were then incubated with either biotinylated anti-mouse, anti-rabbit, or anti-goat immunoglobulin G (1:400, KPL, Guildford, UK) at 37°C for 40 minutes and with horseradish peroxidase–conjugated streptavidin (1:300, Dako Japan, Kyoto, Japan) at room temperature for 40 minutes. Finally, the reaction products were visualized with 0.05% 3-3′-diaminobenzidine and 0.03% hydrogen peroxide in tris–hydrochloric acid buffer, followed by a counterstain with Mayer’s hematoxylin or methylgreen.
Primary Antibodies Used and Positive Cells in Canine Tissues
a GFAP, glial fibrillary acidic protein; Iba1, ionized calcium–binding adaptor molecule 1; EGFR, epidermal growth factor receptor; pERK1/2, phospho-extracellular signal-regulated kinase 1/2; pAkt, phospho-Akt.
b Dako Japan, Kyoto, Japan; Immuno-Biological Laboratories, Gunma, Japan; Promega Corporation, Madison, WI; Santa Cruz Biotechnology, Santa Cruz, CA; PROGEN Biotechnik, Frankfurt, Germany; Wako Pure Chemical Industries, Osaka, Japan; Abcam, Tokyo, Japan; Cell Signaling Technology Japan, Tokyo, Japan; Genetex Inc, Irvine, TX; Covance, Dallas, TX.
The percentage of immunoreactive cells was defined as the ratio of strongly labeled tumor cells to the total number of cells examined in 5 high-power fields (×400).
Cluster Analysis
A multivariate method of cluster analysis, the Ward method, was performed to obtain an objective classification for the results and a distance measure of each cluster. SPSS 15.0 was used for the calculation. Squared Euclidian distances between the clusters were used to construct a dendrogram.
Results
Histopathological Diagnoses
As mentioned, Table 1 summarizes the histopathological diagnoses and features of tumor cells in the cases.
Immunohistochemistry
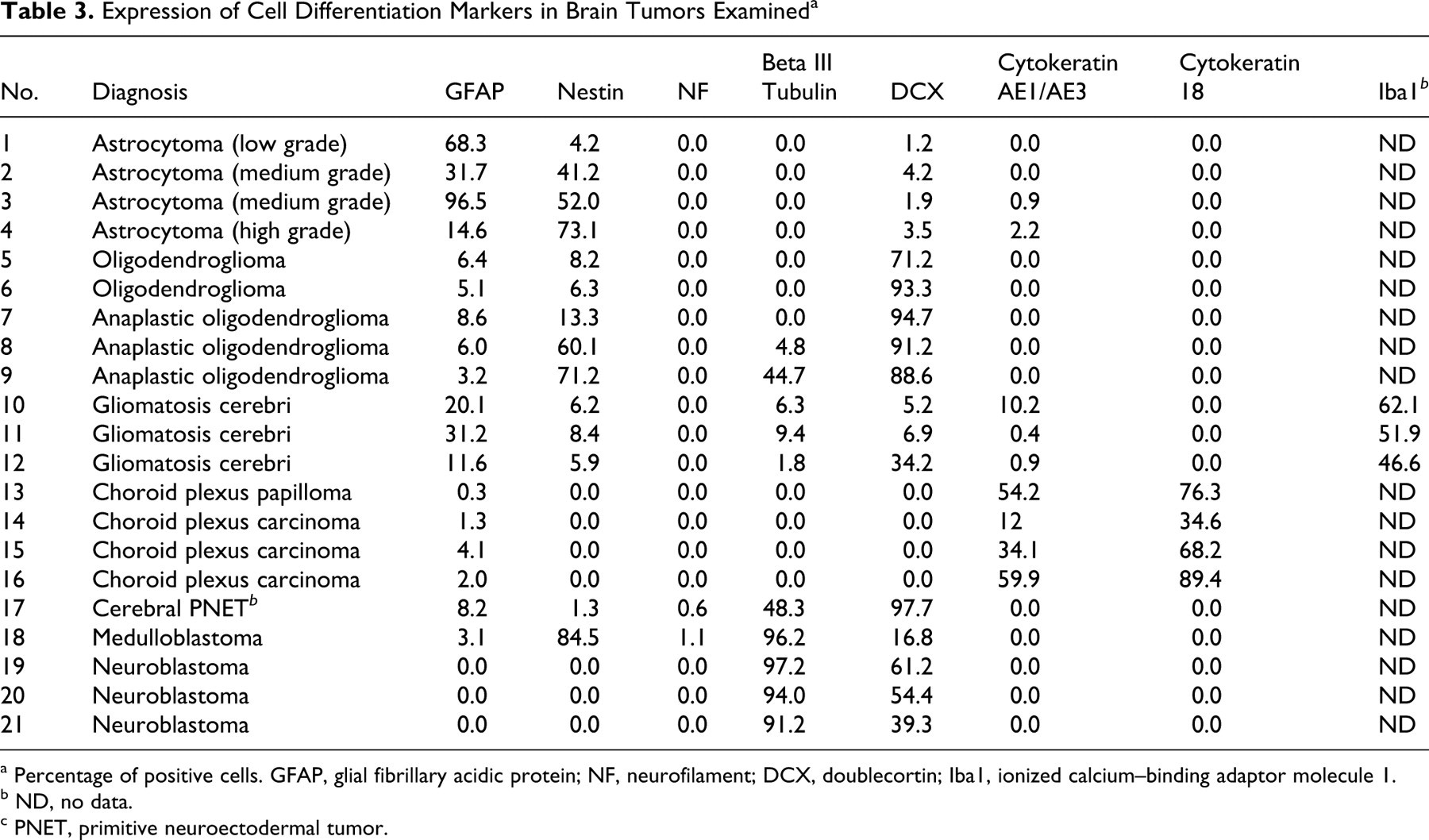
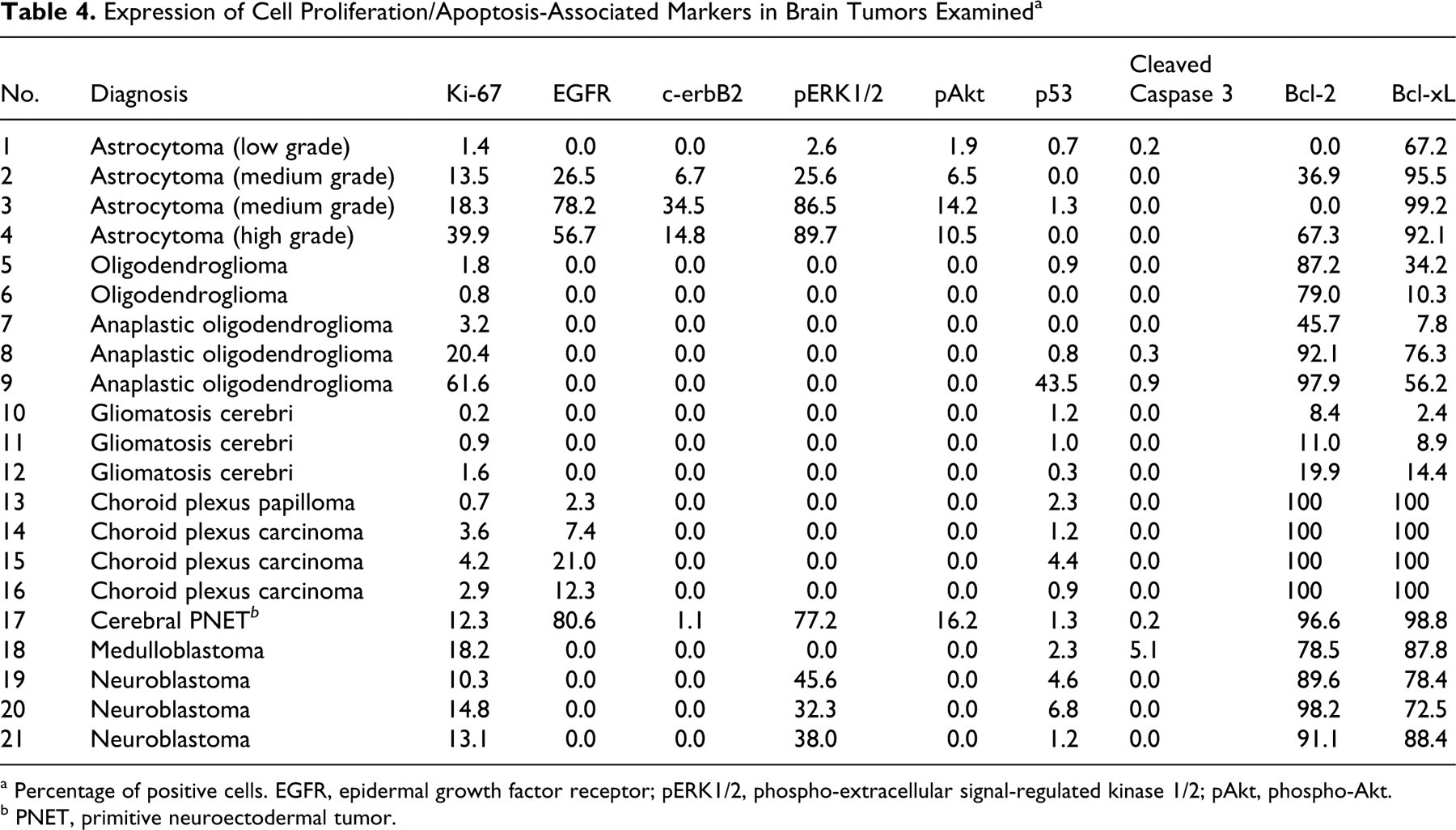
Tables 3 and 4 summarize the results of immunohistochemistry for cell differentiation markers and cell proliferation-associated markers, respectively.
Expression of Cell Differentiation Markers in Brain Tumors Examined a
a Percentage of positive cells. GFAP, glial fibrillary acidic protein; NF, neurofilament; DCX, doublecortin; Iba1, ionized calcium–binding adaptor molecule 1.
b ND, no data.
c PNET, primitive neuroectodermal tumor.
Expression of Cell Proliferation/Apoptosis-Associated Markers in Brain Tumors Examined a
a Percentage of positive cells. EGFR, epidermal growth factor receptor; pERK1/2, phospho-extracellular signal-regulated kinase 1/2; pAkt, phospho-Akt.
b PNET, primitive neuroectodermal tumor.
Astrocytomas
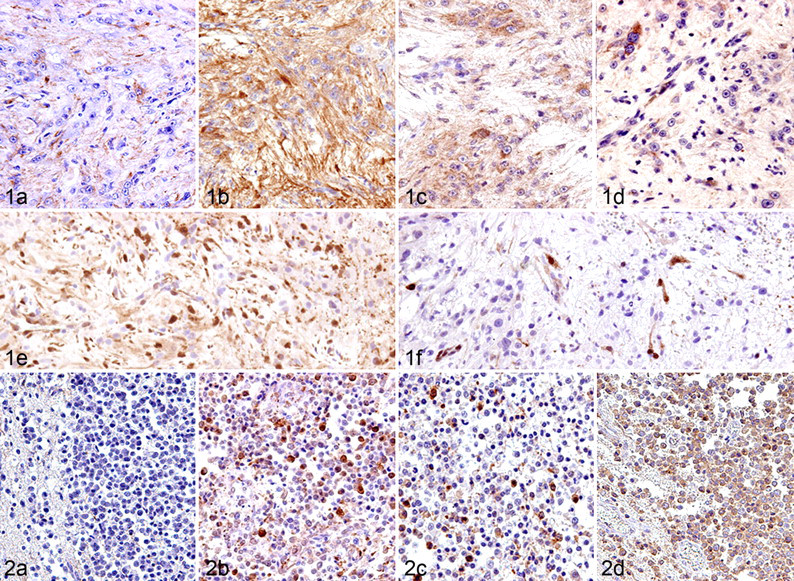
Immunoreactivity to GFAP (a mature astrocyte marker) varied among cases, although the number of GFAP-positive cells tended to be higher in the low-grade astrocytoma (Fig. 1). In contrast, the majority of tumor cells and their processes were strongly positive for nestin (an astrocytic precursor marker) in medium- and high-grade forms. A few tumor cells were weakly positive for DCX (a neuronal precursor marker), mainly in the margin of the lesion. A small number of cells positive for cytokeratin AE1/AE3 (an epithelial marker) were observed in 2 cases. Cells positive for NF (a mature neuronal marker), beta III tubulin (an early phase neuronal marker), and cytokeratin 18 (an epithelial marker) were not found in astrocytomas. The number of tumor cells with Ki-67-positive nuclei was high (13.5 to 39.9%) in medium- and high-grade tumors but low (1.4%) in low-grade ones. The number of tumor cells positive for EGFR, c-erbB2, pERK1/2, and pAkt was parallel to the tumor grade. For most cases, EGFR and c-erbB2 immunoreactivity was localized not only on the cell membrane but also in the cytoplasm, with pERK1/2 and pAkt immunoreactivity in the cytoplasm and the nuclei. The majority of tumor cells were positive for Bcl-xL in all astrocytomas, but the number of Bcl-2-positive cells varied among cases. Cells positive for p53 or cleaved caspase 3 were not found in astrocytomas.
Oligodendrogliomas
The majority of tumor cells were positive for DCX but negative for GFAP. Benign tumors consisted of few nestin-positive cells, whereas anaplastic tumors had many (Fig. 2). GFAP- and nestin-positive cells were present within a limited area and morphologically mimicked reactive astrocytes. Beta III tubulin–positive cells were observed in 2 anaplastic cases. Notably, minigemistocyte-like cells observed in case No. 9 were intensely positive for nestin, beta III tubulin, and DCX but negative for GFAP. In no cases were there any cells positive for NF, cytokeratin AE1/AE3, and cytokeratin 18. The percentage of Ki-67-positive tumor cells was generally low (0.8 to 3.2%), except for 2 anaplastic cases (20.4 and 61.6%). In 1 case of anaplastic oligodendroglioma (case No. 9), more than 10% of tumor cells exhibited p53 immunoreactivity, suggesting overexpression. 22,35,38 Contrary to the results of astrocytomas, the number of Bcl-2-positive tumor cells tended to be larger than that of Bcl-xL-positive cells. In oligodendrogliomas, no cells were detected positive for EGFR, c-erbB2, pERK1/2, pAkt, or cleaved caspase 3.
Gliomatosis cerebri
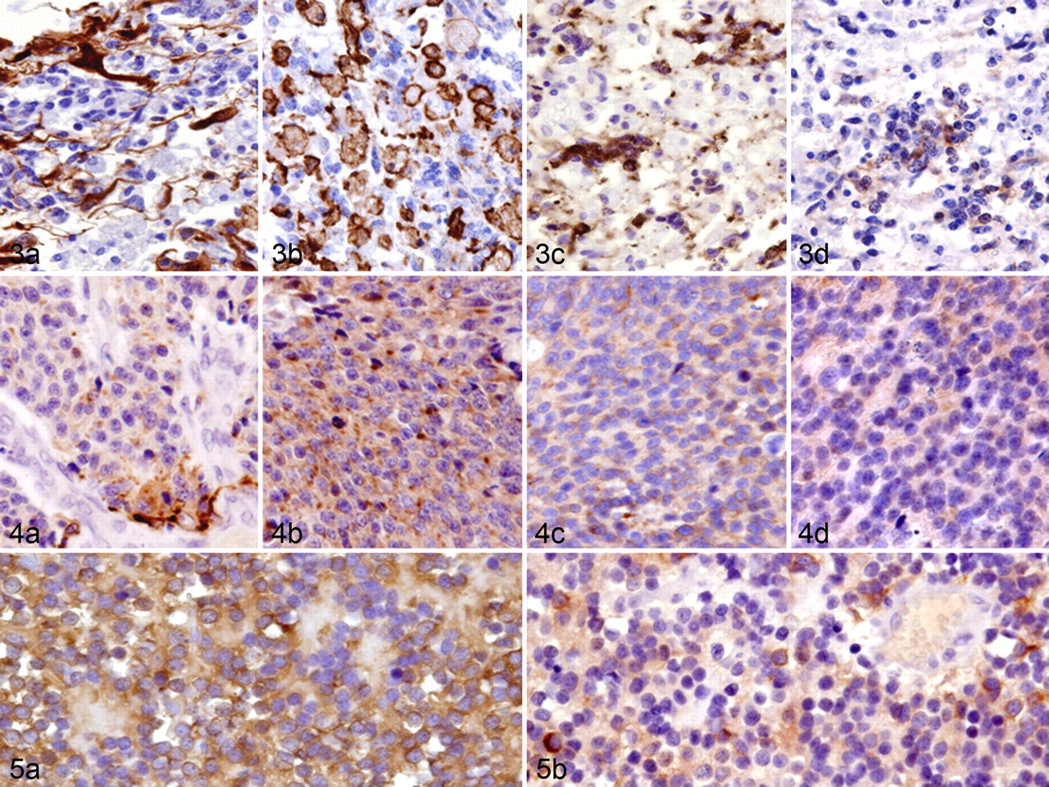
Gliomatosis cerebri is characterized by a diffuse infiltrative proliferation of small pleomorphic tumor cells, consisting of a heterogeneous cell population (Fig. 3). There was a small to considerable number of cells positive for Iba1 (a microglial marker), GFAP, nestin, DCX, beta III tubulin, or cytokeratin AE1/AE3. There were small foci of cells positive for GFAP, nestin, and cytokeratin AE1/AE3 in all 3 cases, and these cells were morphologically similar to astrocytes. Many Iba1-positive amoeboid microglia and rodlike cells were frequently found in the lesions. Small pleomorphic cells were negative for GFAP, cytokeratins, and Iba1 but positive for beta III tubulin, DCX, and nestin. Although NF-positive neurites were scattered in the tumor lesions, all tumor cells were negative for NF. The number of Ki-67-positive cells was extremely low (0.2 to 1.6%) in all cases. Bcl-2- or Bcl-xL-positive cells were rare to moderate. No cells positive for EGFR, c-erbB2, pERK1/2, pAkt, and cleaved caspase 3 were detected in gliomatosis cerebri.

A dendrogram demonstrating the similarity among the canine neuroepithelial tumors examined in the present study, as constructed by the Ward method for cluster analysis. PNET, primitive neuroectodermal tumor.
Choroid plexus tumors
Moderate to many tumor cells were positive for cytokeratin AE1/AE3 and cytokeratin 18. A small number of cells were focally positive for GFAP. No tumor cells were positive for nestin, DCX, beta III tubulin, and NF. EGFR-positive cells were rare to moderate. The number of immunoreactive cells for Ki-67 was low (0.7 to 4.2%). All tumor cells were positive for Bcl-2 and Bcl-xL in all cases. No cells positive for c-erbB2, pERK1/2, pAkt, and cleaved caspase 3 were detected in choroid plexus tumors.
Cerebral PNET
The cerebral PNET had a heterogeneous cell population and consisted of a small to considerable number of cells positive for GFAP, nestin, DCX, beta III tubulin, or NF. No tumor cells were positive for cytokeratin AE1/AE3 and cytokeratin 18. The number of Ki-67-positive cells was 12.3%. A small to considerable number of tumor cells were positive for EGFR, c-erbB2, pERK1/2, and pAkt. EGFR, c-erbB2, pERK1/2, and pAkt immunoreactivity showed the same localization with astrocytomas. The majority of tumor cells were positive for Bcl-2 and Bcl-xL. In this case, a small number of cells were found positive for p53 and cleaved caspase 3.
Medulloblastoma
Like cerebral PNET, medulloblastoma (cerebellar PNET) had a heterogeneous cell population, including a small to considerable number of cells positive for GFAP, nestin, DCX, beta III tubulin, or NF (Fig. 4). No tumor cells were positive for cytokeratin AE1/AE3 and cytokeratin 18. The number of Ki-67-positive cells was 18.2%. In this case, 5.1% of tumor cells exhibited cleaved caspase 3. The majority of tumor cells were positive for both Bcl-2 and Bcl-xL. No cells positive for EGFR, c-erbB2, pERK1/2, and pAkt were detected in this case.
Neuroblastomas
The majority of tumor cells were positive for beta III tubulin and DCX. No tumor cells were positive for GFAP, nestin, NF, cytokeratin AE1/AE3, and cytokeratin 18 (Fig. 5). The percentage of cells positive for Ki-67 varied from 10.3% to 14.8%; pERK1/2-positive cells were generally moderate, and the immunoreactivity was localized in the cytoplasm and nuclei. The majority of tumor cells were positive for both Bcl-2 and Bcl-xL. No cells positive for EGFR, c-erbB2, pAkt, and cleaved caspase 3 were detected in neuroblastomas.
Cluster Analysis
Figure 6 presents the results of the hierarchical clustering approach as a dendrogram. All cases were initially classified into 3 distinct clusters: cluster A, 3 gliomatosis cerebri and 4 astrocytomas; cluster B, only choroid plexus tumors; and cluster C, 5 oligodendrogliomas, 3 neuroblastomas, and 2 PNETs (including a medulloblastoma).
Discussion
The present study carried out a cluster analysis of 21 canine neuroepithelial neoplasms according to immunohistochemical expression profiles using 16 markers. As shown in Fig. 6, the neoplasms were classified into 3 clusters: cluster A, astrocytomas and gliomatosis cerebri; cluster B, choroid plexus neoplasms; and cluster C, oligodendrogliomas, neuroblastomas, and PNETs.
The present hierarchical clustering reveals that astrocytomas and gliomatosis cerebri are related neoplasms (cluster A). These neoplasms are classified as glial neoplasms in the WHO histological classification guidelines. 18 In 4 astrocytomas examined, the majority of neoplastic cells were positive for astrocytic markers (GFAP and nestin) and completely negative for neuronal markers (NF and beta III tubulin). In addition, there were a few DCX-positive cells. As reported, DCX is preferentially expressed in human invasive gliomas, and the immunoreactivity is more intense at the margin of the neoplastic lesion than at the center, suggesting high invasive capacity of the immunoreactive cells. 7 Similarly, canine gliomatosis cerebri, which are characterized by diffuse infiltrative proliferation of small pleomorphic neoplastic cells, consisted of GFAP-positive cells, Iba1-positive cells (most frequently found in the lesions), and small spindle cells positive for nestin, DCX, and beta III tubulin. Such a complex immunohistochemical profile of this type of neoplasms may support the idea that the neoplastic cells have variable differentiation potency with extreme invasiveness and motility. In addition, small spindle cells positive for nestin, DCX, and beta III tubulin may originate from subventricular zone cells. The results suggest that canine astrocytomas and gliomatosis cerebri are mainly directed to glial differentiation.
Choroid plexus neoplasms (cluster B) were far different from other neuroepithelial neoplasms. Choroid plexus neoplasms consistently expressed epithelial markers (cytokeratin AE1/AE3 and cytokeratin 18) and had specific morphological features, such as branching papillary structures formed by cuboidal to columnar epithelium, 4,29,36 thereby suggesting their direction to epithelial differentiation. These neoplasms contained small populations of GFAP-positive cells. This phenomena might be explained by the close relationship between choroid plexus and ependymal cells, the latter of which are known to express some glial markers, including GFAP. 4
Based on WHO histological classification guidelines, 18 oligodendrogliomas were classified as glial neoplasms, and neuroblastomas and PNETs, as embryonal neoplasms. However, the results of the cluster analysis indicate that these neoplasms are related and belong to the same cluster (Cluster C). All cases of this cluster predominantly or considerably consisted of DCX-positive and NF-negative cells, indicative of neuroblastic origin. The majority of neoplastic cells of 3 canine neuroblastomas, which were characterized by frequent Homer Wright rosette formation, were intensely positive for early-phase neuronal (beta III tubulin) and precursor (DCX) markers but not for astrocytic (GFAP and nestin) and mature neuronal (NF) markers. Therefore, neuroblastomas are composed of relatively homogeneous cell populations toward neuronal differentiation. In contrast, canine cerebral PNET and medulloblastoma consisted of heterogeneous cell populations, including cells positive for nestin, beta III tubulin, or DCX, as intermingled with a small number of GFAP- and NF-positive cells. This indicates that the neoplasms have glial and neuronal differentiation features. The majority of canine oligodendroglioma cells were DCX positive. The neoplasms also contained minor populations of GFAP- and/or nestin-positive cells. Although oligodendrogliomas are classified as glial neoplasms in the WHO guidelines, 18 the present results indicate that they are neoplasms not yet committed to specific differentiation and that the neoplasm’s cell of origin might be the progenitor cell, possessing the potential to differentiate into glial and neuronal cells. In fact, there is a report concerning the evidence for neuronal differentiation in human oligodendrogliomas with 1p19q codeletion. 9 The present results suggest that cluster C neoplasms (oligodendrogliomas, neuroblastomas, and PNETs) have immature or neuroblastic natures.
In this study, combined expressions of EGFR and c-erbB2 were mainly found in the malignant forms of astrocytomas and cerebral PNET. In the neoplasms, the expression of pERK1/2 and pAkt, located downstream of EGFR, was observed. These data are consistent with the observation made in mice gliomas that only astrocytomas had pAkt and pERK1/2 activities. 6,11,24 Combined expression of pERK1/2 and pAkt—that is, the cross talk between RAS/MAPK and the PI3K/Akt pathways—is possibly necessary for the pathogenesis of canine astrocytomas and cerebral PNET.
In a previous study dealing with 31 canine astrocytomas, 35 the expression rates of p53 and EGFR, as assessed by immunohistochemistry, were moderate—especially, that of EGFR, which was mainly higher in the malignant forms of astrocytomas. The present results show no or very little p53 expression in canine astrocytomas. However, EGFR immunoreactivity was found only in malignant forms of astrocytomas, consistent with the previous report, which suggests that some canine astrocytomas examined in this study have a deficit of p53 gene, as in human counterparts. 17,21,27,28
Additionally, the predominant expression of Bcl-2 and Bcl-xL observed in canine neuroepithelial neoplasms in the present study may indicate antiapoptotic mechanism in neoplastic cells. Such antiapoptotic mechanism concerning Bcl-2 and Bcl-xL expressions may be involved in the histogenesis of the neoplasms.
The present study clearly demonstrates that canine neuroepithelial neoplasms, except choroid plexus neoplasms, are of neural stem or progenitor origin with multipotent differentiation potency, although cell morphology and neoplasm location of each neoplasm are different. In conclusion, canine astrocytomas and gliomatosis cerebri are closely related neoplasms with glial differentiation. Although oligodendrogliomas are classified as glial neoplasms in the WHO guidelines, 18 the constructed dendrogram shows a novel criteria: primitive neuroepithelial neoplasms with glial and neuronal differentiations. Choroid plexus neoplasms had specific epithelial features and were far different from other neuroepithelial neoplasms.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
