Abstract
Hepatic lipodystrophy in Galloway calves is a fatal liver disease affecting a small proportion of the Galloway breed described in different parts of Europe and North America during the past decades. The clinical findings include a diversity of neurological signs. Clinical pathology findings frequently indicate hepatobiliary disease. Postmortem examination reveals an enlarged, pale yellow, and firm liver. Histologic lesions include hepatic fibrosis, hepatic lipidosis, and bile duct hyperplasia. To date, the etiopathogenesis remains obscure. Infectious causes, intoxications, and a hereditary origin have been considered. We describe hepatic lipodystrophy in Galloway calves from an extensively farmed cow-calf operation in southern Germany. Main clinical findings in 6 calves were consistent with hepatic encephalopathy. Clinical pathology findings in 5 of 6 tested animals revealed increased concentration of total bilirubin (maximum value [MV], 54 μmol/l; reference range [RR], <8.5 μmol/l), direct bilirubin (MV, 20 μmol/l; RR, <3.4 μmol/l), increased activity of gamma glutamyl transferase (MV, 162 U/l; RR, <36 U/l) and glutamate dehydrogenase (MV, 420 U/l; RR, <16 U/l). In addition, activity of glutathione peroxidase was decreased in all tested (n = 5) animals (MV, 61 U/g hemoglobin [Hb]; RR, >250 U/g Hb). Postmortem examination in 6 calves revealed a firm, diffusely enlarged yellow liver with a finely nodular surface. Histologic lesions included hepatic fibrosis, hepatic lipidosis, and bile duct hyperplasia. Our findings add to the existing data on hepatic lipodystrophy in the Galloway breed and outline a protocol to aid in the diagnosis of this disorder.
Keywords
Hepatic lipodystrophy in Galloway calves was first recognized in 1965 in Scotland with the first published report in 1982 by Stewart and others. 45 It has been reported in England, 7 Canada, 15 the United States, 31 and possibly Iceland. 26 Based on the small number of reports that were identified in the literature, animal-level prevalence (within the Galloway population) and herd-level prevalence (of all Galloway operations) of this disease appear to be relatively low. However, in affected herds, within-herd prevalence has been reported to be remarkably high, affecting up to 10% of the offspring in some cases. 7
Hepatic lipodystrophy has been reported to affect exclusively calves of the Galloway breed, with only 2 recently documented crossbred calves. 46 The objective of the present report was to describe the clinical and pathologic findings in cases of hepatic lipodystrophy in a Galloway herd in southern Germany.
Materials and Methods
Background
During June 2011 and February 2013, a total of 7 purebred Galloway calves were referred to the Clinic for Ruminants, University of Munich. The calves originated from an extensively farmed cow-calf operation herd with a total of 10 cows, 5 replacement heifers, and 1 breeding bull. The animals were on the same pasture year-round (approximate size 2 hectares). A shed was provided for weather protection and was bedded with straw purchased from a neighboring farm. All animals had free access to hay and grass silage from 2 different pastures of the farm as well as to water from the municipal water supply. No mineral supplementation was offered to the animals. Prior to the referral of the first calf, 3 calves at the age of 2 to 3 months died in April 2011, showing similar clinical signs according to the owner’s report. Postmortem examination was performed in 1 of the 3 calves at the Department of Veterinary Pathology of the Bavarian Health and Food Safety Authority, Oberschleissheim, and revealed a firm, diffusely enlarged yellow liver with a finely nodular surface. Histologic lesions included hepatic fibrosis, hepatic lipidosis, and bile duct hyperplasia. No lesions indicating white muscle disease were observed. Investigation of the trace element status via photometric analyses (Fe, iron; Cu, copper; Zn, zinc) and inductively coupled plasma optical emission spectrometry (ICP-OES; Se, selenium) of serum samples taken from 5 animals in June 2011 by the Bavarian Animal Health Service revealed a severe selenium deficiency. Examination of all forages did not lead to any evidence of the underlying cause of the reported disease. The owner observed serious behavioral changes in all affected calves several days prior to admission to the clinic. The animals at 1 to 10 weeks of age were depressed and were lying isolated from the herd, became progressively debilitated, showed only weak attempts to suckle, and became increasingly reluctant to follow their dams. They could easily be approached without eliciting a flight reflex. When forced to rise and trot, the calves showed a typical posture with the head carried very low and nodding listlessly. According to the owner’s opinion, the 7 calves that were referred to the clinic over a period of 20 months presented in various stages of the same disease. One calf died during transport and was sent to the Department of Veterinary Pathology of the Bavarian Health and Food Safety Authority, Oberschleissheim, for postmortem examination. A total of 6 calves were available for clinical examination and bloodwork. Immediately after arrival at the clinic, blood was taken from each animal by venipuncture of the jugular vein and placed directly into S-Monovette (Sarstedt, Nümbrecht-Rommelsfeld, Germany), anticoagulant (K3 EDTA, 1.6 mg/ml; Sarstedt), and blood gas Monovette (50 IU/ml of calcium-balanced lithium heparin; Sarstedt) tubes. Blood samples were processed immediately, and serum was harvested by centrifugation at 3000 rpm for 10 minutes at 25°C. All serological parameters, as well as the activity of glutathione peroxidase in whole blood, were determined using an automatic analyzing system (Automatic Analyzer Hitachi 911; Roche Diagnostics, Indianapolis, IN). Hematological analyses were performed with an automatic hematology analyzer (Sysmex F820; Sysmex, Norderstedt, Germany).
Necropsy of 6 calves was carried out at the Department of Veterinary Pathology of the Bavarian Health and Food Safety Authority, Oberschleissheim. Samples of liver, brain, myocardium, and kidney of all calves; major muscles of the thigh and shoulder of 4 calves (Nos. 1, 4, 5, and 6); spleen of 3 calves (Nos. 5, 6, and 7); lung of 3 calves (Nos. 1, 4, and 7); and hepatic lymph node of 2 calves (Nos. 4 and 6) were collected for light microscopy. Formalin-fixed, paraffin-embedded tissues were routinely processed for histological examination and stained with hematoxylin and eosin (HE). Liver specimens were stained with Goldner’s trichrome staining of connective tissue and Oil Red O for lipids.
Results
Clinical Signs and Clinical Pathology
At the time of hospitalization, 5 of the 6 calves predominantly demonstrated impairment of the sensorimotor system ranging from slight depression to coma with intermittent seizures and tenesmus. Table 1 shows baseline characteristics and results of clinical examination of the 6 calves at the time of hospitalization. Hematological and clinical chemistry findings are demonstrated in Table 2. Abnormal findings included an increase of liver-specific parameters in 5 calves and hypoglycemia in 4 calves. L-lactatemia could be detected in 2 calves and hypalbuminemia in 1 calf. In 5 of 5 tested calves, the activity of glutathione peroxidase was below the reference range (RR).
Baseline Characteristics and Clinical Findings at the Time of Hospitalization of 7 Galloway Calves Referred to the Clinic Over a Period of 20 Months.
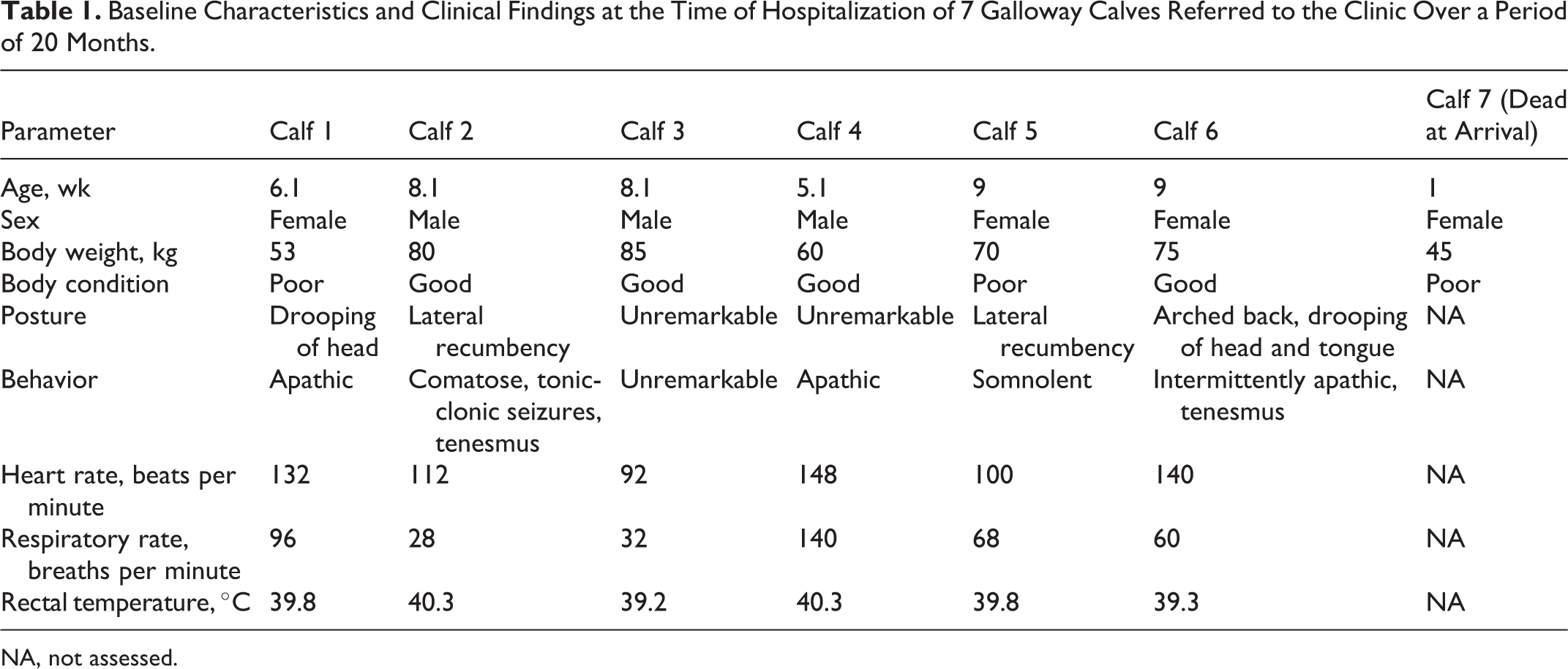
NA, not assessed.
Results of Hematological Analysis and Clinical Chemistry of 6 Galloway calves referred to the Clinic Over a Period of 20 Months.a
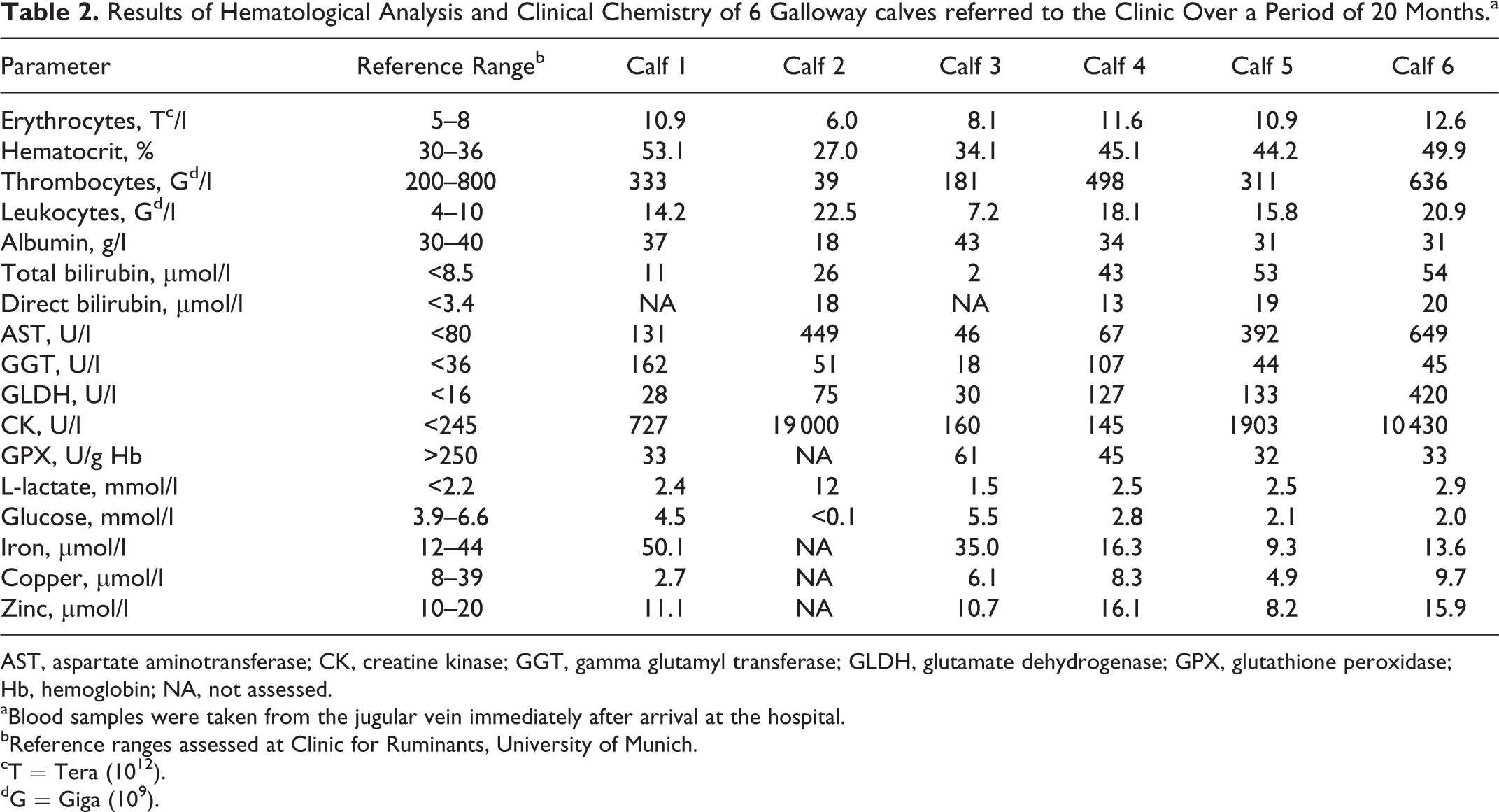
AST, aspartate aminotransferase; CK, creatine kinase; GGT, gamma glutamyl transferase; GLDH, glutamate dehydrogenase; GPX, glutathione peroxidase; Hb, hemoglobin; NA, not assessed.
aBlood samples were taken from the jugular vein immediately after arrival at the hospital.
bReference ranges assessed at Clinic for Ruminants, University of Munich.
cT = Tera (1012).
dG = Giga (109).
Treatment and Clinical Course
Based on the severity of clinical signs, calf No. 2 was euthanized immediately after completion of the clinical examination. Treatment for the remaining 5 calves consisted of vitamin E (tocopherol acetate, 10 mg/kg bodyweight [BW], subcutaneously [s.c.]) and sodium selenite (0.3 mg/kg BW, s.c.) in a combined preparation (Vitamin E-Selen-Lösung; CP-Pharma, Burgdorf, Germany), a nonsteroidal anti-inflammatory drug (meloxicam, 0.5 mg/kg BW, s.c.; Metacam, Boehringer Ingelheim, Ingelheim am Rhein, Germany), and an antimicrobial drug (gamithromycin, 6 mg/kg BW, s.c.; Zactran, Biokema SA, Merial, Hallbergmoos, Germany). Three calves (Nos. 4, 5, and 6) with hypoglycemia received a continuous intravenous infusion containing 5 liters of 0.9% sodium chloride solution (Isotone Natriumchlorid-Lösung ad usum veterinarium; B. Braun, Melsungen, Germany) and 1 liter of 40% dextrose solution (Glucose 40% ad usum veterinarium; B. Braun). One of these 3 calves (calf No. 5) died unexpectedly within several hours after arrival at the clinic. Calf No. 6 was euthanized 24 hours after arrival at the clinic because of rapid progression of the disease. Deterioration of the general condition and liver-specific parameters (aspartate aminotransferase [AST], 216 U/l [RR, <80 U/l]; gamma glutamyl transferase [GGT], 173 U/l [RR, <36 U/l]; glutamate dehydrogenase [GLDH], 63 U/l [RR, <16 U/l]) within 4 days was documented in calf No. 1. Five days after hospitalization, the calf was found in lateral recumbency showing tonic-clonic seizures and opisthotonus and was euthanized. A temporary improvement concerning the general condition and liver-specific parameters (AST, 58 U/l; GGT, 82 U/l; GLDH, 111 U/l) could be documented in calf No. 4. However, the general condition deteriorated peracutely 7 days after hospitalization. The calf showed central nervous signs (bawling, tonic-clonic seizures) and was euthanized. Calf No. 3 never showed any signs of disturbance of the general condition. Parameters of clinical chemistry on days of admission and discharge were unremarkable.
Pathological Findings
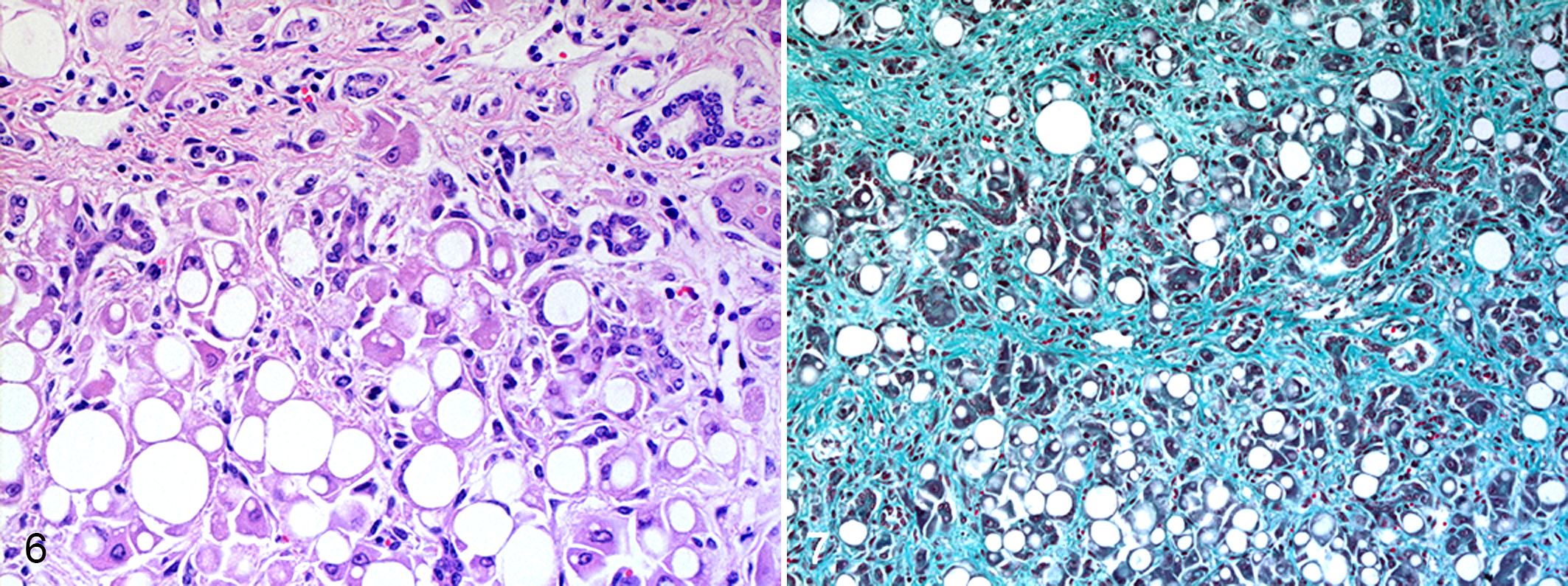
Postmortem examination revealed similar findings in all calves with some variation in the youngest calf (calf No. 7). The most obvious lesion was a firm, diffusely enlarged yellow liver with a finely nodular surface (Figs. 1–3). The gallbladder was severely distended with unremarkable bile (Fig. 3). Liver samples did not float in formalin. Histologically, livers were characterized by marked loss of hepatocytes, prominent hepatic lipidosis, severe diffuse hepatic fibrosis, and bile duct hyperplasia. Hepatocytes showed macrovesicular vacuoles staining with Oil Red O for lipids and displacing the nucleus to the periphery of the cell (Figs. 6, 7). Single necrotic hepatocytes were present as well as a small number of megalocytes. In the fibrotic areas, small foci of mononuclear inflammatory cell infiltrates were present. There was no accumulation of lipofuscin or any other pigments in hepatocytes or in Kupffer cells. Hepatic lymph nodes were moderately enlarged and edematous as confirmed histologically.
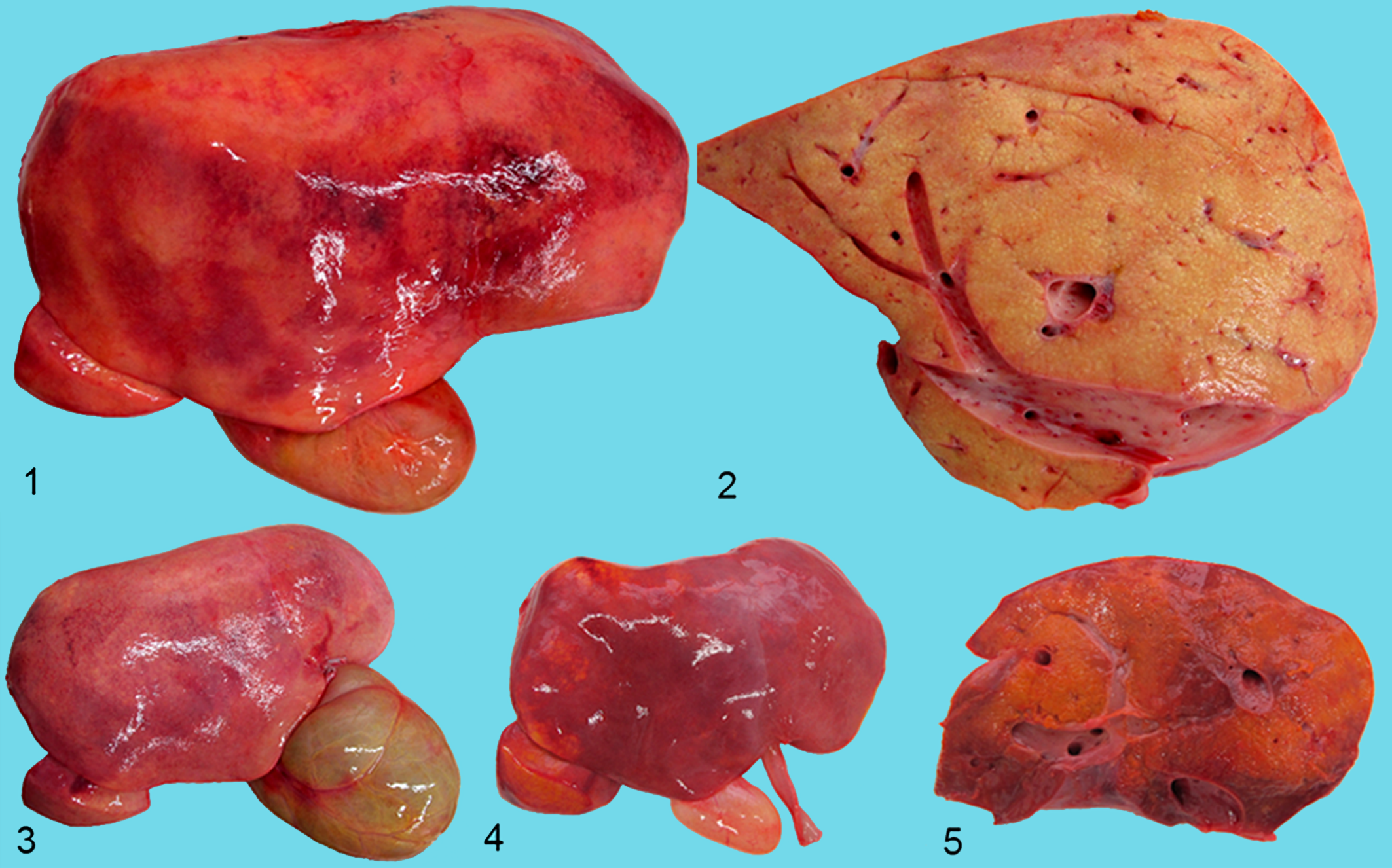
Hepatic lipodystrophy, liver and gallbladder; bovine.
Hepatic lipodystrophy, liver; bovine.
The liver of the youngest calf (calf No. 7) was enlarged and orange-brown in color, of nodular texture, and softer than normal (Figs. 4, 5). Hepatic lipidosis with macrovesicular vacuolization was the most obvious histological finding in this calf. There was only mild hepatic fibrosis that exhibited a portal distribution. Necrosis of small groups of hepatocytes and hepatocellular cholestasis were present. The gallbladder of this animal was filled with watery bile.
Furthermore, necropsy revealed obvious icterus of 2 calves (Nos. 6 and 7). Five of the 6 calves were emaciated, and 2 were cachectic (Nos. 1 and 5) as indicated by serous atrophy of epicardial fat.
Histologically, the brain of 2 calves (Nos. 4 and 6) showed acute spongiform changes, especially in the cerebral cortex. Due to mild autolysis, it was not possible to critically examine the brains of the other 4 calves for lesions of hepatic encephalopathy.
Light microscopic examination of major muscles of the thigh and shoulder and myocardium revealed no evidence of nutritional myodegeneration. Histologic examination of kidneys und lungs yielded moderate hyperemia. Further secondary findings were bronchopneumonia of the cranioventral regions of the lung (3 calves), enteritis (3 calves), and abomasal ulceration (2 calves). Spleen specimens showed no histological alterations. Bacteriological testing revealed no relevant infectious agents in thoracic and abdominal organs.
Liver samples of 2 calves (Nos. 2 and 7) were submitted to the Pharmaceutical Institute, University of Bonn, Germany, and analyzed via gas chromatography and gas chromatography–mass spectrometry. 40,55 A metabolite of pyrrolizidine alkaloids (PAs) was found in 1 calf (No. 2). Investigation of a liver sample of the same calf for concentration of selected mycotoxins via enzyme-linked immunoassay (aflatoxin, 14 deoxynivalenol [DON], 13 ochratoxin A 29 ) at the BioCheck laboratory services GmbH (Leipzig, Germany) revealed a concentration of DON of 99.6 μg/kg (RR, <20 μg/kg, assessed at BioCheck laboratory services GmbH), whereas concentration of aflatoxin and ochratoxin A was below the detection limit of 0.05 μg/kg. Moreover, concentration of hepatic copper and cobalt was analyzed from 1 calf (No. 7) at the Bavarian Health and Food Safety Authority (Oberschleissheim, Germany) using ICP-OES and inductively coupled plasma mass spectrometry (ICP-MS) as described in Section 64 of the German Food and Feed Code. Copper was determined to be below the reference range (23.8 mg/kg wet weight; RR, 30–150 mg/kg 39 ), whereas cobalt concentration was marginal but not deficient (0.015 mg/kg wet weight; RRs: deficient, <0.005; marginal, 0.005–0.017; adequate, 0.020–0.085 mg/kg wet weight 38 ).
Further Investigations
Investigation of serum samples from the respective dams yielded no elevation of liver-specific parameters. However, a severe reduction of activity of glutathione peroxidase could also be detected in adults (range, 9–15 U/g hemoglobin [Hb]; RR, >250 U/g Hb). In addition, selenium deficiency could be confirmed by measuring selenium by ICP-MS in the plasma of 5 cows (range, 5.3–11.4 μg/l; RR, >50 μg/l, assessed at the University of Veterinary Medicine Hannover, Foundation, Germany). A botanical investigation of a hay sample at the Pharmaceutical Institute, University of Bonn, Germany, did not give any evidence of PA-containing plants. Moreover, an inspection of the pasture was carried out by a botanist of the Bavarian Institute for Agriculture, in which a small amount of comfrey (Symphytum officinale) was found.
Discussion
To our knowledge, this is the first report on hepatic lipodystrophy in Galloway calves in Germany with a particularly high within-herd prevalence affecting 40% of the offspring in the years 2011 to 2013. While apparent herd-level and animal-level prevalence of the general Galloway population appear to be low, the true prevalence is not established. This might be further complicated by the fact that herd investigations, including necropsies, are only carried out in cases where a sudden increase of mortality within a herd is considered alarming. It is therefore possible that single cases of hepatic lipodystrophy might go unnoticed, and cases are attributed to concurrent calfhood diseases, such as diarrhea and pneumonia, which have been recorded in affected calves. 7,26
In most cases, the disease leads to clinical signs at 2 to 4 months of age. 26 The oldest calf was 5 months old. 26 In addition, abortions and stillborn calves have been reported. 7,26,46 While in the present case, most affected calves (5/6) have been within the characteristic age, 26 1 calf was only 1 week old and represents the youngest clinical case of documented hepatic lipodystrophy.
The predominant clinical signs as reported by the owner and documented after hospitalization show an involvement of the central nervous system consistent with hepatic encephalopathy as reported by others. 7,15,26,46
Laboratory parameters only have been documented in a total of 4 calves from 2 different studies, 7,26 with no general pattern of alterations. Although alterations of laboratory parameters in samples from calves in this study reflect different stages of the disease, a characteristic pattern of changes of liver-specific parameters was documented. Increased activity of AST in 4 cases might be at least partially due to muscle cell damage and can be attributed to the massive selenium deficiency leading to myodystrophy. This evidence is also underlined by the large increase in activity of creatine kinase (CK) in 2 calves (Nos. 2 and 6). 52 In this regard, the remarkable selenium deficiency as determined by activity of glutathione peroxidase, ICP-OES, and ICP-MS is noteworthy. Remarkable differences in the proportion of selenium-dependent and non-selenium-dependent glutathione peroxidase activity exist among different species. 25 It is therefore possible that Galloway cattle have a high proportion of selenium-dependent glutathione peroxidase in liver tissue compared to other species and breeds, resulting in a high susceptibility to oxidative stress and consequent hepatopathy when selenium intake is deficient. Considering the massive selenium deficiency, one should expect calves to be affected by coagulative necrosis of the skeletal muscles. The lack of such histological findings in skeletal muscles and myocardium might be due to the sufficient vitamin E intake through grazing, which might prevent some manifestations of selenium deficiency to a certain extent. However, because vitamin E status was not determined in any of the calves, this possible explanation remains to be tested. Hypoglycemia, as described in 4 cases, can be caused by massive liver cell damage, 43 which leads to L-lactatemia as previously described. 35,43
The case definition of hepatic lipodystrophy in Galloway calves is the presence of the aforementioned characteristic clinical signs in association with hepatic lipidosis, 7,15,26,46 which was documented at different degrees in all 6 animals necropsied. Hepatic lipidosis refers to a visible accumulation of triglycerides as round vacuoles in the cytoplasm of hepatocytes and can result from a variety of different disorders, including intoxications, nutritional deficiencies, metabolic disease, and inherited defects in lipid metabolism. 44 Several stages of hepatic lipid metabolism can be impaired by numerous toxins, causing hepatic lipid accumulation. 44 Among the toxins most relevant for cattle in Germany are aflatoxins, 5 DON, 48 and PAs. 20 DON and PA concentrations above the RR were detected in the liver of 1 affected calf.
Considering the age of affected calves (1 to 9 weeks in the present report plus aborted and stillborn calves in other reports 7,26,45 ), the possibility of transplacental transmission and transmission into the milk following ingestion of toxins by the dam has to be taken into account. Transplacental transmission of aflatoxins in calves causing liver cirrhosis has been reported, 1 as has been the case for transmission into the milk from animals fed with feeds containing aflatoxin B1. 11,34 Transfer of DON to milk following ingestion of the preformed toxin by the dam has been reported to be very low, with an estimated 0.0001% of the ingested dose excreted in milk. 36 Transplacental transmission of DON has been observed in rats 4 , pigs 49 , and humans 30 ; however, no detrimental effects on the liver of the respective fetuses could be detected. 4,49 No reliable data about fetotoxic effects of PAs have been reported. In contrast to reports of studies showing a transplacental transfer of PAs, 9,10,42 1 experimental study failed to detect abnormalities in fetuses after feeding a lethal dose to the respective dams. 20 While excretion of PAs into the milk could be detected, 17,28 no consensus of its effects after ingestion by the respective newborn and suckling animals exists. Dickinson and colleagues 6 could not detect any histological alterations of the liver or increased concentration of liver-specific parameters in newborn calves after feeding a nonlethal dose of PAs to the respective dams. In another experimental study, a lethal dose of Senecio jacobaea was fed to dams. 19 The respective calves showed increased concentration of liver-specific parameters. However, no characteristic histological alterations could be detected. 19 While in the present case, comfrey was the only PA-containing plant that was found in the pasture, it seems to be unlikely—according to the present knowledge—that single ingestion of comfrey can cause such severe clinical and pathological alterations, especially given the high prevalence of the described condition.
Notwithstanding, we cannot exclude with certainty that a possible intoxication of the calves has been caused by ingestion of more than 1 PA-containing plant other than comfrey. Due to the year-round grazing, animals in this herd have access to the sprouting young plants containing PAs in the spring, which have less of a bitter taste and, therefore, can be ingested, especially by inexperienced young animals. 33 Although the investigation of all forages did not yield any evidence for the presence of the toxins, we cannot exclude an interaction of more than one of the aforementioned toxins. Another aspect might be the fact that the young animals do not have a fully developed rumen and, therefore, are more sensitive to PAs than adult animals who have the capacity to inactivate certain toxins during ruminal passage. 54 It is therefore quite possible that the massive selenium deficiency would have had a negative impact on the individual resilience of affected calves not only to PA intoxication but also to other toxins such as aflatoxins and DON.
As a component of glutathione peroxidase, selenium has a direct influence on the glutathione homeostasis. The importance of intracellular glutathione on toxin elimination of PAs has been shown in numerous studies. 3,18,56 In addition, qualitative and quantitative species differences in the liver capacity of glucuronidation are said to be responsible for insensitivity to senecio-alkaloids of some animal species. 16
A further type of hepatic lipidosis associated with encephalopathy, named “white liver disease,” has been described concurrently with cobalt deficiency in sheep 22,24 and goats, 21 causing a decreased activity of the vitamin B12–dependent enzymes methyl-malonyl-CoA mutase 23 and methionine synthase 22 and a consequent accumulation of branched-chain fatty acids (BCFAs) in hepatocytes. 24 Histologically, livers were characterized by fatty degeneration of hepatocytes, cholangiolar hyperplasia, and hepatic megalocytosis similar to the findings observed in this study but also by a prominent lipofuscin accumulation in degenerated hepatocytes and in Kupffer cells, 24,50,51 which was absent in the cases described here. Hemosiderosis of the spleen is also common in ovine white liver disease. 51 In contrast, lipofuscin and hemosiderin accumulation is lacking in the organs of the presented calves. However, this characteristic manifestation of cobalt deficiency might be restricted to sheep and goats 2 due to a species-specific high rate of methyl-malonyl-CoA activity leading to higher rates of BCFA synthesis compared to cattle. 8 Finally, because hepatic cobalt concentration was determined to be within normal limits in calf No. 7, we consider a disorder similar to “white liver disease” in sheep and goats unlikely to be the cause of the findings in the calves described herein.
Another nutritional deficiency causing myopathy and hepatic lipidosis in weaned lambs is vitamin E deficiency. 27 However, vitamin E deficiency seems to be unlikely in animals on pasture. There was no evidence of myopathy from histological examination in the calves described herein.
A hereditary abnormality within the Galloway breed has to be considered as a possible cause or as a contributing factor in a multifactorial disease complex, as suggested by the breed predisposition of this disease. 7,26,45,46 Familial hyperlipoproteinemia in cats, 47 idiopathic hyperlipoproteinemia in dogs, 41 and congenital generalized lipodystrophies in humans 32 have been identified to be inherited defects in lipid metabolism. In the present case, all affected calves were the progeny of 1 bull that had been introduced into the herd in 2010 and was slaughtered in 2013. The most recent consultation with the owner revealed no new cases in 2014 and 2015. However, no genetic analysis has been performed to date.
The term hepatic lipodystrophy in association with Galloway cattle derives from the first report on “a fatal condition of pedigree Galloway calves” by Stewart and others. 45 The authors documented a characteristic clinical pattern and unique pathological findings that are similar to the ones described herein. To this date, several authors 7,15,26 have referred to this concept. In human medicine, the term lipodystrophy describes a group of syndromes in which the primary defect is a lack of intraabdominal or subcutaneous adipose tissue and an accumulation of fat depots in muscle tissue and central organs. 12 The cardinal features include insulin resistance, dyslipidemia reflected by hypertriglyceridemia, and liver steatosis. 53 According to the cause and the anatomical distribution of adipose tissue, lipodystrophies are classified as genetic or acquired and generalized or partial, respectively. 37 Although the increased concentration of serum lipids in 2 calves and triglycerides in 1 calf as reported by Macleod and Allison 26 offers some evidence that this disease of Galloway calves is a bovine form of lipodystrophy, the proof is still lacking. Further investigations, including testing of the concentration of insulin in blood possibly enhanced by a glucose tolerance test, as well as blood concentration of lipids, namely triglycerides, have to be carried out indicating the described phenomenon being a lipodystrophy as defined in human medicine. 37
Footnotes
Acknowledgements
We thank Dr Helmut Wiedenfeld (Pharmaceutical Institute, University of Bonn, Germany), Dr Martin Höltershinken (University of Veterinary Medicine Hannover, Foundation, Germany), and A. Lindner and associates (BioCheck Laboratory Services GmbH, Leipzig, Germany) for their invaluable technical assistance. We also acknowledge the collaboration of the farm owners during the investigation of these cases. This manuscript has been greatly improved by the thoughtful comments of an anonymous reviewer.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Bavarian State Ministry of Food, Agriculture and Forestry and the Bavarian Tierseuchenkasse (animal disease fund).
