Abstract
The liver can be an indicator of the health of an individual or of a group, which can be especially important to identify agents that can cause disease in multiple species. To better characterize hepatic lesions in stranded harbor porpoises (Phocoena phocoena), we analyzed the livers from 39 porpoises that stranded along the Dutch coast between December 2008 and December 2012. The animals were selected because they had either gross or histologic liver lesions with minimal autolysis and no evidence of trematode (Campula oblonga) infection. The most common finding was a chronic hepatitis (22/39, 56.4%) that was often associated with significant disease reported in another organ system (18/22, 81.8%), of which 14 had chronic systemic disease. One case of chronic hepatitis was so severe as to mimic lymphoma, which could only be differentiated with immunohistochemistry. The other common lesions were lipidosis (11/39, 28.2%) and acute hepatitis (6/39, 15.4%), often in combination with mild chronic changes. Overall, although there were no consistent trends in etiology for the hepatic lesions, lipidosis was associated with starvation (8/11, 72.7%) and acute disease, and acute hepatitis was associated with bacterial infections and sepsis (6/6, 100%).
Keywords
The liver has important roles in homeostasis, metabolism, catabolism and immune function. The liver also processes many toxins, some of which affect environmental health including that of wildlife and humans. 4 Disease of the hepatobiliary system could lead to reduced metabolism of toxins and therefore an increased risk of other illnesses such as immune suppression or reproductive failure. 20 Therefore, the liver is not only a vital organ to investigate in any individual animal, but also for determining the health of a population.
Reported causes of hepatic lesions in case studies of cetaceans include a variety of bacterial, 12,21 viral, 19,23,28 trematode (Campula sp.), 3,9,12,15,23 and protozoal 23 infections, lipidosis, 3,23,30 amyloidosis, 26 hemochromatosis, 30 nonspecific reactive hepatitis, 9 and neoplasia. 18,24 With the harbor porpoise’s position at the top of the food chain, toxin accumulation can occur and there have been many studies assessing toxin and pollutant levels in marine mammal livers. 4,6,20 However, to date there have been conflicting reports of direct links between toxins and hepatic damage. 13,22
Many of the above investigations concerned stranded harbor porpoises (Phocoena phocoena), 12,15,23,28 but none have specifically focused on liver pathology. Two investigations that specifically studied hepatic lesions in cetaceans, but not porpoises, include a case series in a captive dolphin population 30 and a case series in free-living, stranded cetaceans (dolphins and whales only) from the Canary Islands. 9 Recently, a study of the effects of Campula spp parasitism on the hepatobiliary system in cetaceans found severe architectural distortion caused by the trematodes with fibrosis, nodular regeneration, biliary hyperplasia, and inflammation. 10
The goals of this study were to describe the histologic liver lesions in very fresh to fresh (decomposition condition code [DCC] 1 and 2) 14 stranded harbor porpoises and to identify potential causes. Animals with trematode (Campula oblonga) infections were excluded for 3 reasons: the lesions have been described previously in great detail, 1,3,9,10,12,15,23 the parasites markedly distort the architecture with inflammation and cirrhosis that would mask other lesions, 10 and all livers with trematodes would be graded as very marked chronic hepatitis with very marked fibrosis, 7,8 which would potentially skew data analysis. Our secondary goal was to determine if a grading system, adapted from Ishak et al, 7,8 would be useful in categorizing hepatic lesions in future studies.
Materials and Methods
Animals
In total, 745 harbor porpoises were submitted for routine necropsy between December 2008 and December 2012, which was performed according to a standard protocol based on the DCC 14 that includes macroscopic and microscopic evaluation of a range of organs, including the liver. Liver samples were collected for histologic examination in 4% neutral buffered formaldehyde, and some were cultured directly for bacteriological analysis and stored at –20°C and –80°C for future analysis.
A total of 130 cases were selected because they had evidence of macroscopic or microscopic liver changes noted in the necropsy reports and were within the very fresh (DCC1) and fresh (DCC2) categories. Cases were eliminated that had moderate to severe histologic distortion due to autolysis or freeze artifact (n = 39), had gross or histologic evidence of a trematode (C. oblonga) infection (n = 51, 39%), or the animal was treated in a rehabilitation center (n = 1). After review, 39 cases (30% of fresh porpoises) fit the selection criteria and were included in this study.
Histology
Sections of liver were routinely processed for histologic examination with hematoxylin and eosin (HE) stains. Periodic acid–Schiff (PAS) stain for fungal elements and hepatocellular inclusions, Gram stain for bacteria, and Perls’ Prussian blue stain for iron were performed on selected cases per standard protocols.
Grading Scheme
The grading of the hepatic inflammatory lesions was performed following a modified version of the grading scheme proposed by the WSAVA (World Small Animal Veterinary Association) for needle biopsy specimens obtained from canine livers, which was adapted from a human grading scheme from Ishak et al. 7,8 Individual parameters for acute or chronic hepatitis were evaluated in 3 areas of at least 10× objective by 3 authors (L.H., L.C.M.W., and R.I.K.). Each observer gave numeric values for each parameter for acute or chronic hepatitis (Table 1). Fibrosis was given an overall severity based on the 3 parameters combined. The mean of the values for all parameters was calculated per animal, and this value was designated as the grade or stage.
Grading Scheme for Inflammatory Liver Lesions.
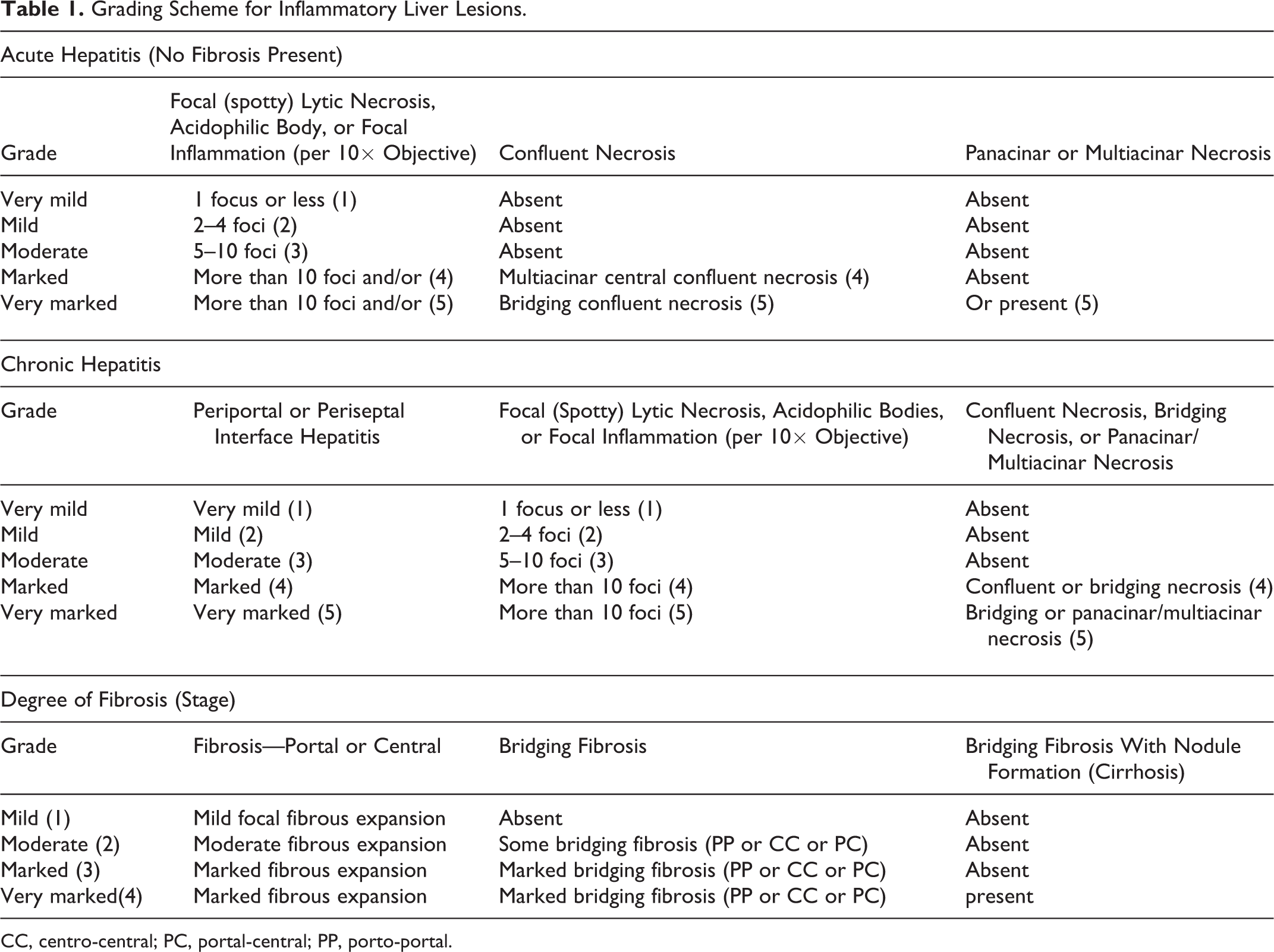
CC, centro-central; PC, portal-central; PP, porto-portal.
Immunohistochemistry
For all antibodies, formalin-fixed, paraffin-embedded liver sections were mounted on silan-coated slides and dried overnight at 55°C and then deparaffinized with xylene. For Toxoplasma gondii detection (n = 19), the slides were incubated with 1% H2O2 in methanol for 30 minutes, rinsed, incubated with 10% goat serum in phosphate-buffered saline (PBS) for 15 minutes, and incubated with rabbit polyclonal anti–T. gondii primary antibody (Biogenex, Fremont, CA) at 1:100 dilution at room temperature for 60 minutes. For CD3 and CD79A detection (n = 1), slides were pretreated with citrate buffer (pH 6.0) in a microwave for 15 minutes and then blocked with 1% H2O2 in methanol for 30 minutes. For CD3, the slides were incubated with 10% goat serum for 15 minutes and then a rabbit polyclonal anti-CD3 primary antibody (Cell Marque, Rocklin, CA) at a 1:200 dilution overnight at 4°C. For CD79A, the slides were incubated with 10% normal horse serum for 15 minutes and then a mouse monoclonal CD79A antibody (HM57; Santa Cruz Biotechnology, Dallas, TX) at a 1:80 dilution overnight at 4°C. Both the Toxoplasma antibody and CD3 antibody protocols used a secondary goat anti–rabbit biotin antibody (Vector Laboratories, Burlingame, CA) at 1:250 dilution for 30 minutes at room temperature. For the CD79A antibody, a horse anti–mouse biotin secondary antibody (Vector Laboratories) at 1:250 dilution for 30 minutes at room temperature was used. A secondary peroxidase-based method was used for all samples with detection of bound antibodies performed using the
Bacterial Cultures
Fifty-three samples from 22 animals were analyzed (liver, n = 20; lung, n = 20; spleen, n = 5; intestinal contents, n = 6; lymph node, n = 2). Samples were cultured on 5% sheep blood agar in anaerobic and aerobic conditions and on MacConkey’s agar. Lung tissue was also cultured on chocolate agar. After incubation at 37°C, cultures were examined for growth after 24 and 48 hours. Culturing for Brucella sp was performed on 15 animals as described previously 11 on lung, lung worms, liver, lymph node, and genital swabs.
Viral Testing
Livers were tested for herpes viruses (n = 9) and influenza A virus (n = 5) via polymerase chain reaction (PCR) or reverse transcription PCR (RT-PCR), respectively, on frozen (either –20°C or –80°C) liver tissue. Total nucleic acids were isolated on a MagnaPure LC system using the LC Total Nucleic Acid Isolation Kit (Roche Diagnostics, Almere, the Netherlands) according to the manufacturer’s protocol.
A nested herpes virus PCR was performed as described previously. 29 Briefly, 2 forward primers (HV-F1: 5′-GAYTTYGCNAGYYTNTAYCC-3′ and HV-F2: 5′-TCCTGGACAAGCAGARNYSGCNMTNAA-3′) and 1 reverse primer (HV-R1: 5′-GTCTTGCTCACCAGNTCNACNCCYTT-3′) were used in the first PCR. An aliquot of 2 μl from the first PCR reaction was used for a nested PCR with 1 forward primer (HV-F3: 5′-TGTAACTCGGTGTAYGGNTTYACNGGNGT-3′) and 1 reverse primer (HV-R2: 5′-CACAGAGTCCGTRTCNCCRTANAT-3′). Products of the PCR reactions were checked by electrophoresis on a 2% agarose gel for fragments of the correct size.
Influenza A virus was detected using a real-time PCR assay as described previously. 31 The following primers and probes were used: forward primer: 5′-CTTCTRACCGAGGTCGAAACGTA (bases 32–54), reverse primer: 5′-TCTTGTCTTTAGCCAYTCCATGAG (bases 149–172), probe 1: 5′-6-FAM-TCAGGCCCCCTCAAAGCCGAGA-BHQ-3′ (bases 74–95), and probe 2: 5′-6-FAM-TCAGGCCCCCTCAAAGCCGAAA-BHQ-3′ (bases 74–95). Amplification and detection were performed on an ABI7500 (Applied Biosystems, Foster City, CA) with the Taqman Fast Virus 1-Step Master Mix (Applied Biosystems).
Results
Animals
From the population of 39 harbor porpoises, there were 8 neonates (5 female, 3 male), 30 juveniles (12 female, 18 male), and 1 adult female animal. The animals stranded predominantly in the province of North Holland (n = 15) in the winter (n = 19), but all coastal provinces and seasons were represented (Suppl. Fig. S1).
Necropsy Results
Categories of causes of death included bycatch, infectious, emaciation, starvation, and trauma (Suppl. Fig. S2 and Suppl. Table S1). Starvation, considered an acute process, was used to designate animals with ample blubber and fat reserves and no gastrointestinal contents. Emaciation, considered a chronic process, was used to designate animals with minimal blubber and fat reserves whether or not there were gastrointestinal contents.
Signalment and stranding data for each category of hepatic lesion are presented in Supplemental Table S2. Within the chronic hepatitis group (n = 22), there were 3 very mild (Nos. 12, 18, and 29), 12 mild (Nos. 2, 11, 19, 24, 26, 28, 30, and 32–36), 6 moderate (Nos. 1, 5, 6, 9, 10, and 13), and 1 very marked hepatitis (No. 27). Most cases (15/22, 68.2%) had mild infiltrates in the portal triads, often consisting of few lymphocytes, plasma cells, and occasional eosinophils and mild fibrosis (Fig. 1). One remarkable case (No. 27) had marked lymphoid infiltration with mixed inflammation, including plasma cells, macrophages, and few eosinophils (Fig. 2a). The lymphocytes distorted the normal hepatic architecture, and there were multiple areas of bridging fibrosis and nodule formation (cirrhosis). Immunohistochemistry for CD3 and CD79A revealed a pattern of large nodules of B cells surrounded by coalescing areas of T cells (Fig. 2b,c). This change was also seen in the spleen and pulmonary lymph nodes (data not shown).
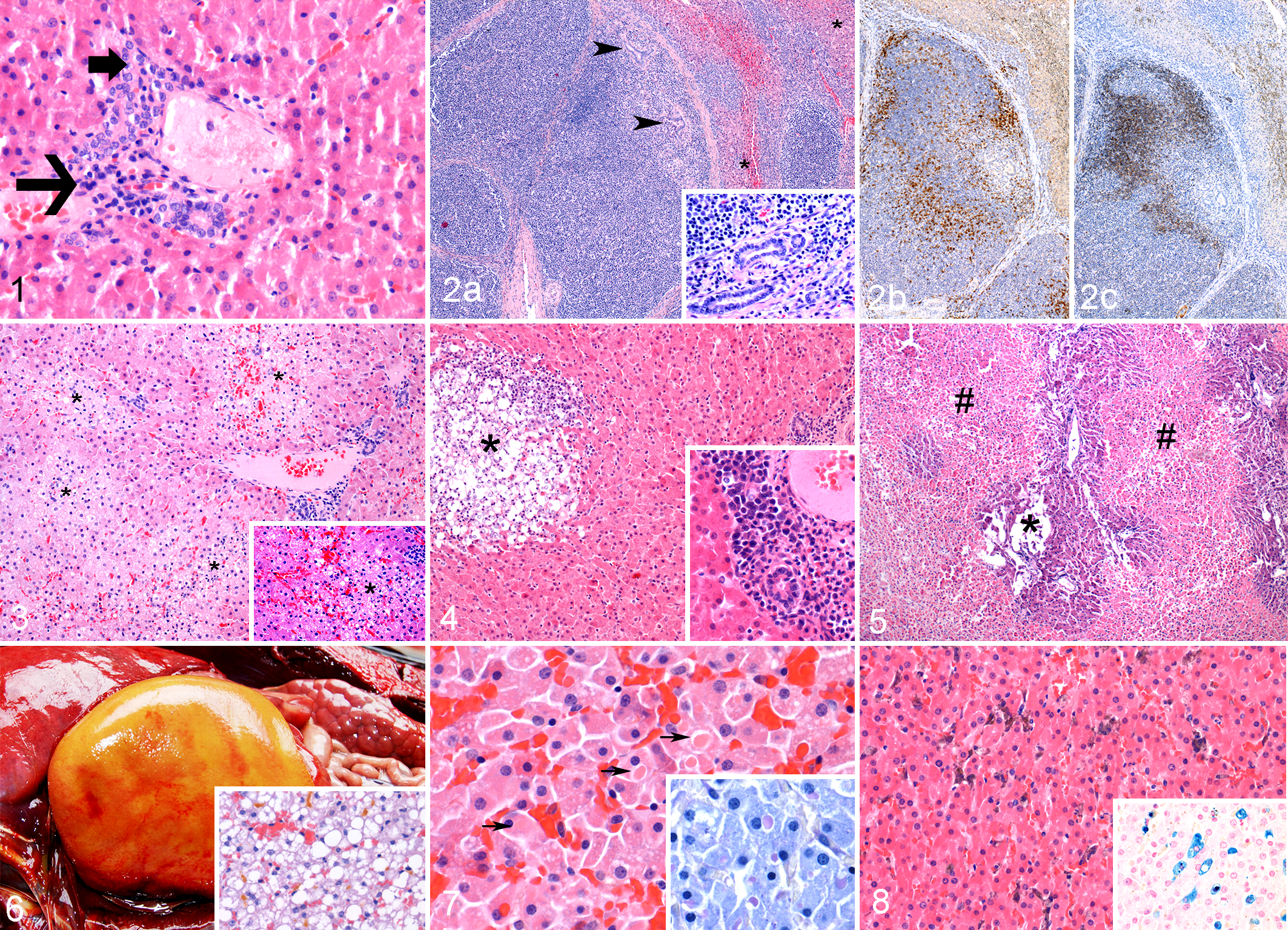
Liver, harbor porpoise.
Chronic hepatitis as the sole liver lesion was common (22/39, 56.4%), was distributed throughout the different age and sex groups of the study population (Suppl. Table S2), and was often associated (14/22, 65%) with chronic inflammatory lesions in other organ systems, most commonly pneumonia (n = 16), gastrointestinal disease (n = 11), or dermatitis (n = 8). In total, 35 of 39 cases had evidence of chronic hepatitis, and 13 were in combination with lipidosis (n = 9) or acute hepatitis (n = 4). No specific etiology or general class of agent was associated with chronic hepatitis (Table 2).
Results From Bacterial Cultures, Brucella sp Culture, and Testing for Influenza A Virus, Herpes Viruses, and Toxoplasma gondii.a
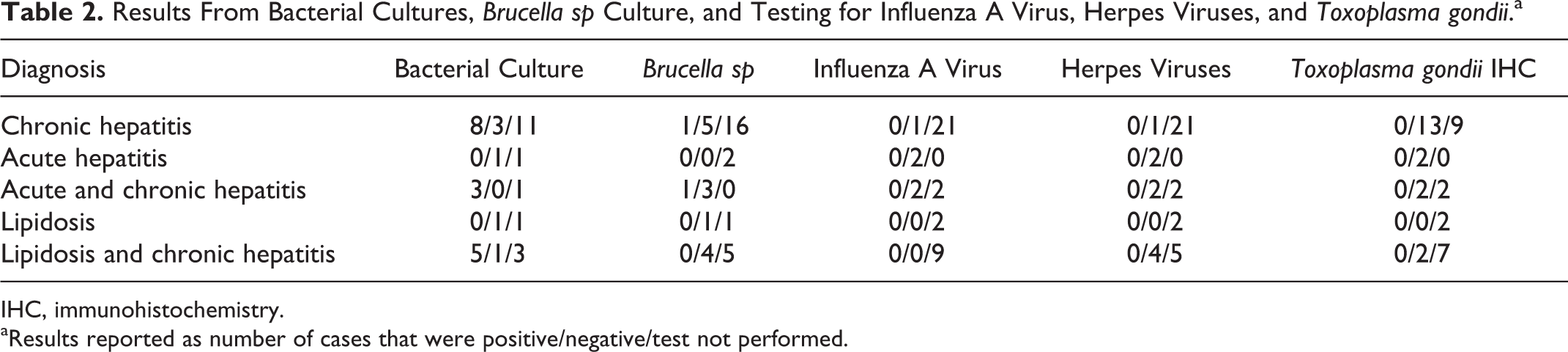
IHC, immunohistochemistry.
aResults reported as number of cases that were positive/negative/test not performed.
Within the acute hepatitis group (n = 2, Nos. 3 and 8), there was 1 moderate (No. 3) and 1 marked hepatitis (No. 8). All cases of acute hepatitis had spotty, multifocal, or confluent areas of necrosis or parenchymal inflammation with no evidence of fibrosis (Fig. 3). Four cases (Nos. 4, 15, 20, and 23) had both acute and chronic changes: (1) marked to very marked acute hepatocellular necrosis with no or suppurative inflammation and (2) moderate or very mild portal mononuclear infiltrates and mild portal fibrosis (Figs. 4, 5).
Categories of causes of death with acute hepatitis included bycatch (Nos. 3, 4, and 20), infectious (Nos. 15 and 23), and trauma (No. 8). Of the 4 animals with both acute and chronic hepatitis, all had chronic inflammatory lesions in another organ system, including parasitic pneumonia (Nos. 4, 15, and 20) or skin ulceration with bacteria (No. 23).
There were 2 cases of hepatic lipidosis with no inflammatory changes (Nos. 21 and 37) and 9 cases of mild to marked lipidosis in conjunction with very mild (n = 5) to mild (n = 4) chronic hepatitis (Nos. 7, 14, 16, 17, 22, 25, 31, 38, and 39). Grossly, the livers were markedly enlarged, yellow to orange, friable, and greasy on cross section (Fig. 6). Histologically, the cases with moderate or severe lipidosis (9/11; Nos. 7, 14, 16, 17, 21, 22, 25, 31, and 38) often had concurrent cholestasis with bile plugs in canaliculi (Fig. 6). One of the cases of mild lipidosis (No. 37) also had 10- to 15-μm eosinophilic cytoplasmic inclusions that were PAS-positive (Fig. 7).
In 3 cases of chronic hepatitis (Nos. 19, 24, and 26), there was significant pigment accumulation mainly within Kupffer cells and fewer hepatocytes that was often brown to green-black with HE and stained blue with Perls’ Prussian blue, which is consistent with iron accumulation (Fig. 8).
Ancillary Testing
A summary of the ancillary testing for infectious etiologies can be found in Table 2. All PAS reactions were negative for fungal organisms. Only 1 case (No. 19) had findings with Gram staining: few scattered Gram-positive and Gram-negative bacteria within multiple blood vessels but not within tissues or areas of inflammation, in association with mild chronic hepatitis, a tongue ulcer, and a positive culture for Bacillus bacteria.
All animals tested for T. gondii (n = 19), herpes viruses (n = 9), or influenza A virus (n = 5) were negative.
Of the 22 animals that had bacterial cultures performed, 6 had no growth (Nos. 3, 11, 26, 31, 32, and 37). The culture results were markedly variable and included Staphylococcus sp (No. 10), Streptococcus sp (No. 23), Klebsiella sp (No. 12), Bacillus sp (No. 18), Clostridium perfringens (Nos. 12, 19, 23, and 24), Vibrio alginolyticus (No. 38), Fusobacterium sp (No. 16), Enterobacter sp (No. 39), and many mixed cultures (Nos. 6, 13, 15–17, 19, 20, 22, 24, 27, and 38). Two animals were culture positive for Brucella sp in the lung, pulmonary lymph node, or within lung parasites (Nos. 15 and 26).
Discussion
The objective of this study was to provide detailed descriptions of hepatic lesions within harbor porpoises and to use an established, standard grading scheme to objectively evaluate these findings. Previous case reports tend to focus on grossly evident lesions and infections, such as parasites, and there are rare mentions of other changes, although these lesions could be important to determine cause of death or debilitation in these animals.
Common limiting factors in identifying more subtle hepatic lesions in stranded harbor porpoises include autolysis and parasitism. Indeed, 615 of 745 (82.6%) animals were eliminated from this study based on degree of decomposition alone. Although many of the carcasses were not useful, 39 animals fit the criteria of minimal postmortem artifacts and no identifiable trematodes and remarkably had easily identifiable hepatic lesions.
Stranding location was not correlated with any type of hepatitis or lesion in this data set. The animals within this study were predominantly neonatal and juvenile, which could be artificial due to elimination of those animals with trematode infections, which occurs more frequently in older animals. 12 There is also a predominance of strandings in the winter within this data set, but most trematode infections are noted in the summer and fall, perhaps lowering the number of cases within those seasons.
Most chronic hepatitis cases had very mild or mild portal inflammatory infiltrates with mild fibrosis, which was also a common finding in a case series of hepatic lesions in cetaceans that stranded on the Canary Islands 9 and was referred to in that study as a nonspecific reactive hepatitis. It has been suggested that these lesions could be associated with residual hepatic inflammation or systemic disease, 9 although 4 of 15 of the animals had no other significant inflammatory disease seen at necropsy. In fulminant trematode infections, there is portal inflammation and fibrosis, 10,12,15 and it is possible that these milder lesions may be associated with cleared or mild trematode infections that were not evident on postmortem examination.
All cases of moderate or marked chronic hepatitis were associated with significant chronic inflammatory disease in other organ systems (multiorgan inflammation [Nos. 5, 6, and 27] and pneumonia [Nos. 10 and 13]) or acute disease (skin ulcers with bacteria in Nos. 1 and 9). The hepatitis in these cases is more likely indicative of systemic disease rather than inflammation specifically targeted at the liver.
The animals with acute hepatitis did not have mild disease and were all classified as moderate to severe. Significantly, case No. 20, which had massive centrilobular to midzonal coagulative necrosis, also had evidence of severe trauma (loss of portion of tail) and acute, severe blood loss. The resulting hypoxia could explain the zonal pattern in the liver.
Four cases had evidence of acute hepatitis with mild to moderate chronic portal inflammation. There is evidence that chronic disease can lead to a generalized decrease in immune system function and an increased susceptibility to an acute insult, 20 which may be the case in these animals. The acute hepatitis may be due to sepsis or another infectious agent.
There were 11 cases (28.2%) of hepatic lipidosis, which has been reported in other studies. 9 Most cases (9/11) had moderate to severe lipidosis associated with moderate to severe cholestasis. The cholestasis appeared to be due to massive swelling of the hepatocytes, as it was not seen in cases of mild lipidosis. Intrahepatic cholestasis also occurs during anorexia due to decreased bile flow, although the mechanism is not clear. 25
Starvation was listed as the cause of death in most of the lipidosis cases, and the animals in our study with starvation listed as the cause of death (9 animals) often had hepatic lipidosis (8/9). The 2 pure lipidosis cases were both neonates that stranded in the spring both with no other significant lesions in the remaining tissues. Although lipidosis can be a normal finding within neonatal animals, 9 the diffuse distribution, severity, and cholestasis are not typical. With the absence of food within the digestive tract and the severe lipidosis, starvation was the most likely cause of death in these 2 animals and, at that age, is most likely due to separation from the mother. Starvation and hepatic lipidosis were also seen in animals with severe infectious disease (such as sepsis or pneumonia) or bycaught animals.
In one of the mild lipidosis cases without inflammation (No. 39), PAS-positive, eosinophilic inclusions were seen in the cytoplasm of many hepatocytes. Cytoplasmic inclusions are not expected with herpes virus infection, and this case tested negative for herpes viruses. Similar hyaline inclusions have been described previously in hepatic lesions in cetaceans that stranded in the Canary Islands. 9 The inclusions in that study were also PAS-positive and the ultrastructural findings were not consistent with viral inclusions.
One of the cases (No. 27) had spectacular lymphoid proliferation surrounding the portal triads and extending into the hepatic parenchyma. Only with immunohistochemistry could this proliferation be identified as nodular hyperplasia instead of lymphoma. Similar lesions have been described in older cats with chronic cholangiohepatitis with lymphoid proliferation and follicle formation. In these cats, the etiology is still debated in the current literature, with aerobic enteric bacteria being a leading cause, although not found in all cases. 5,27 In contrast to the research in cats, no cholangiohepatitis was seen and a mixed anaerobic bacterial population not consistent with postmortem overgrowth was cultured from the porpoise liver. This porpoise did have significant chronic inflammation in multiple other organ systems, which could explain the reactive lymphoid proliferation.
Three of the chronic hepatitis cases had pigment accumulation within Kupffer cells and hepatocytes that was confirmed as iron pigment with Perls’ Prussian blue stain. Iron accumulation can be characterized as hemosiderosis or hemochromatosis. Hemochromatosis has been reported in a captive dolphin population associated with lipidosis and hepatitis and was thought to be potentially associated with diabetes mellitus. 30 In human hemochromatosis, iron accumulation is primarily in hepatocytes and not within Kupffer cells. 30 In this study, the iron was in both cell types, and the livers had evidence of mild chronic hepatitis with no concurrent lipidosis. Therefore, the iron accumulation is most accurately characterized as hemosiderosis, which can be seen with anemia, dietary iron overload, viral infections, and fatty liver.
Although no toxicology testing has been performed on these livers, environmental pollutants such as polychlorinated biphenyls (PCBs) and toxins from cyanobacteria can cause hepatic lesions. PCBs most often bioaccumulate and cause hepatic neoplasms in laboratory rodents, fish, and humans. 2,16 No liver tumors were observed in any of the stranded harbor porpoises. PCBs have also been implicated in decreased immune function and reproductive function. 20 Reproductive function cannot be assessed in this population, since most cases are neonates or juvenile animals. Decreased immune function could lead to an increased susceptibility to infections, such as opportunistic bacterial infections or increased parasite loads, which are common in this population. Microcystin and nodularin toxins from cyanobacteria can cause acute, massive liver necrosis with hemorrhage in many animal species, including mammals. 17 In this case series, only 1 case (No. 20) had massive liver necrosis, but the lesion was more likely due to hypoxia after trauma and blood loss.
In conclusion, this study analyzed 39 cases of liver lesions in harbor porpoises that were not associated with trematode infection and found acute and chronic hepatitis, severe lymphoproliferative disease, hepatic lipidosis, and iron accumulation. Although chronic hepatitis has been reported, often in association with trematode infection, the other lesions have not been described previously. The ancillary testing performed did not identify etiologies for the acute or chronic hepatitis, although further studies, including toxicology and broader viral screening, could provide additional information. The grading system that we implemented provided more detailed information than was typically described in the case record and has the added value of uniform designations of lesion classification and severity. The records could therefore be extremely helpful in future studies of liver pathology and epidemiology of disease within the porpoise population if a widespread toxic or infectious event should occur.
Footnotes
Acknowledgements
We are highly appreciative of the hard work by our volunteers, especially Jaap van der Hiele, Kees Kooimans, and Arnold Gronert, who collected and transported the stranded animals, and for those that assisted with the necropsies. We are grateful to the other pathologists in the project for providing initial diagnoses for some of the gross and histologic lesions. Furthermore, we thank the histology and necropsy staff and technicians for specimen collection and preparation. Last, we greatly appreciate the manuscript review and critique provided by Guy Grinwis and Lineke Begeman and the PhotoShop assistance from Sarah Mills.
Author Contribution
Conception or design: SH, RIK. Data acquisition, analysis, or interpretation: SH, LH, LCMW, RIK. Drafting the manuscript: SH, RIK. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Dutch Ministry of Economic Affairs funded this ongoing study into the causes of death of stranded harbor porpoises along the Dutch coast.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
