Abstract
The primary causes of mortality were identified in postmortem examination of 339 (90.9%) of 373 farmed mink (Neovison vison; syn. Mustela vison) from January 2009 through June 2014 at the Utah Veterinary Diagnostic Laboratory (Logan, Utah). Mink were raised under farm conditions in the Intermountain West in North America, except for 1 submission of mink from Wisconsin. In the 339 mink where cause(s) of death were established, 311 (91.7%) died from a single disease or condition, whereas 28 (8.3%) had 2 diseases or conditions contributing to death. Where cause(s) of death were evident, 11 diseases accounted for 321 (94.7%) of the diagnoses: bacterial pneumonia (67, 18.8%), Aleutian mink disease (61, 17.7%), mink viral enteritis (56, 16.2%), hepatic lipidosis (28, 8.1%), nutritional myopathy (24, 7%), bacterial enterocolitis (17, 4.9%), bacterial septicemia (16, 4.6%), starvation (15, 4.3%), epizootic catarrhal gastroenteritis of mink (14, 4.1%), pancreatitis (13, 3.8%), and bacterial metritis (10, 2.9%). In 34 (9.1%) animals, a cause of death was not evident. In an additional 16 (4.3%) of the mink, botulism was suspected from clinical history but could not be confirmed by laboratory testing. Control measures for the most common causes of death in farmed mink include testing and removal of positive animals (Aleutian mink disease), vaccination (Pseudomonas aeruginosa pneumonia, mink viral enteritis), avoidance of obesity in mink (hepatic lipidosis), and environmental management, including maintaining clean water cups, floors, feed troughs, cages, feed silos, feed truck tires, workers’ shoes, dining areas for farm personnel, leather mink handling gloves, street clothes, and coveralls.
Introduction
The Intermountain West is the second leading area of the United States for production of farmed mink (Neovison vison; syn. Mustela vison), after Wisconsin (Fur Commission USA. About Mink Farming. 2011. http://www.furcommission.com/farming/about-mink-farming/). Utah has ~70 mink farms that produce 1.2 million harvested animals annually, and an additional 30 mink farms are located in southern Idaho. Mink are produced principally for their pelts, although mink oil is often collected for cosmetics. Mink farming is a $292 million business in the United States (USDA, ISSN 1949-1549, National Agricultural Statistics Service, Agricultural Statistics Board, July 24, 2014. http://www.furcommission.com/documents/2012/10/2011-mink-production-in-the-us.pdf). Many veterinary diagnosticians are unfamiliar with diseases common in farmed mink. This descriptive case series reports the causes of mortality as determined from postmortem examination of 373 mink farmed in the Intermountain West over a 5.5-year period.
Materials and methods
Postmortem examination of all mink included in this descriptive case series occurred from January 2009 through June 2014 and all were in satisfactory postmortem condition; other mink with advanced postmortem degradation were disposed of rather than necropsied. Mink were presented in groups of 1–10 with a mean of 3.6 animals per submission. Juveniles were defined as mink with body weight ≤0.9 kg (corresponds to ~0–4 months of age), intermediate aged were defined as mink with body weight 0.91 to 1.19 kg (corresponds to ~4–10 months of age), and adults were defined as mink with body weight ≥1.2 kg (corresponds to ~10 or more months of age). 14 Mink with no indication of age or body weight recorded were defined as of unknown age. Sex of the mink was not routinely recorded. All mink were submitted from farming operations in Utah, Idaho, or Wisconsin. At postmortem examination, tissues and fluid samples were collected and stored at −20°C or in 10% neutral buffered formalin (NBF). Specific tissue and/or fluid samples collected depended on clinical signs reported before death and gross findings at postmortem examination. Where botulism was considered a likely cause of death, gastric and/or intestinal contents were sampled. Samples of lung, liver, kidney, and spleen were collected routinely and stored at −20°C. Similarly, samples of esophagus, stomach, intestine (multiple levels), pancreas, liver, trachea, lung, heart, skeletal muscle, kidney, urinary bladder, reproductive organs (ovaries, uterus, testes), spleen, lymph node, thymus (juveniles), adrenal glands, thyroid, and brain were collected and placed in NBF. Additional tissues and/or organs were collected on an individual basis. Once fixed, tissues were subsampled, processed, sectioned, and stained by routine methods. Tissues stained with hematoxylin and eosin were examined microscopically. Special stained replicate sections, including tissue Gram stains, periodic acid–Schiff, and Masson trichrome, were prepared and examined on a case-by-case basis.
Ancillary tests
Bacterial isolation was done by sampling tissues or body fluids and plating directly on trypticase soy agar with 5% sheep blood, chocolate blood agar, Columbia nutrient agar, and MacConkey agar. a Resulting colonies were identified to genus level using either of 2 commercial kits.b,c Botulism testing was conducted at the National Botulism Reference Laboratory, School of Veterinary Medicine at the University of Pennsylvania (Kennett Square, Pennsylvania) on samples of feed and gastrointestinal contents. A polymerase chain reaction (PCR)-based assay for Clostridium botulinum types A, B, and C toxin genes was used.
Aleutian mink disease was diagnosed most often by distinctive histologic findings in kidney, liver, and brain. For confirmation, virus-specific immunoglobulin G was detected using a lateral flow immunoassay d on heart blood obtained at postmortem examination. Alternatively, samples of kidney stored frozen were placed into viral transport medium e and sent to either the Diagnostic Center for Population and Animal Health (Lansing, Michigan) or the Wisconsin Veterinary Diagnostic Laboratory (Madison, Wisconsin) for PCR-based identification of viral DNA.
Mink viral enteritis was diagnosed by characteristic histologic changes in intestine, lymph nodes, and spleen. For confirmation, samples of intestine stored frozen were sent to Zoologix (Chatsworth, California) for PCR detection of viral DNA.
Nutritional myopathy was diagnosed by histologic examination of skeletal muscle and heart, combined with levels of hepatic selenium and vitamin E. Selenium was quantified using inductively coupled plasma–mass spectroscopy. Vitamin E testing was performed at the California Animal Health & Food Safety Laboratory System (Davis, California).
Determination of cause of death
Cause of death was established based on reported clinical signs, pathology, and results of ancillary tests. Where multiple diseases or conditions were diagnosed, death was attributed to the process(es) causing the most pathology in critical organs and/or tissues. As many as 2 diseases or conditions could be defined as causes of the death of 1 animal.
Electronic capture of data
Laboratory findings were entered into a laboratory information management system (LIMS) f that recorded owner and animal data, postmortem examination and other laboratory test results, and allowed for the composition of text to summarize case findings. The LIMS data was summarized by a records review of case reports by the authors and entered into data fields using a commercial spreadsheet. g The following data fields were populated: accession number, date, body weight, age (defined from body weight as described earlier), diagnosis(es) of cause(s) of death, and agent isolated (where applicable). Totals by category were calculated.
Results
During the 5.5-year study period, postmortem examinations were performed on 373 farmed mink. The mink originated from Utah (269 mink from 22 farms), Idaho (95 mink from 7 farms), and Wisconsin (9 mink from 1 farm). Causes of death (339 diagnosed, 34 with no diagnosis) are provided in Table 1. Approximately two-thirds of deaths with a diagnosis (236) were attributed to infectious disease, while the remaining one-third (103) resulted from noninfectious causes. In the 339 mink where cause(s) of death were established, 311 (91.7%) died from a single disease or condition, whereas 28 (8.3%) had 2 diseases or conditions contributing to death. Where cause(s) of death were evident, 11 diseases alone or in combination with other diseases accounted for 321 (94.7%) of the 339 diagnoses: bacterial pneumonia (67, 18.8%), Aleutian mink disease (61, 17.7%), mink viral enteritis (56, 16.2%), hepatic lipidosis (28, 8.1%), nutritional myopathy (24, 7%), bacterial enterocolitis (17, 4.9%), bacterial septicemia (16, 4.6%), starvation (15, 4.3%), epizootic catarrhal gastroenteritis of mink (14, 4.1%), pancreatitis (13, 3.8%), and bacterial metritis (10, 2.9%).
Causes of death in farmed mink.
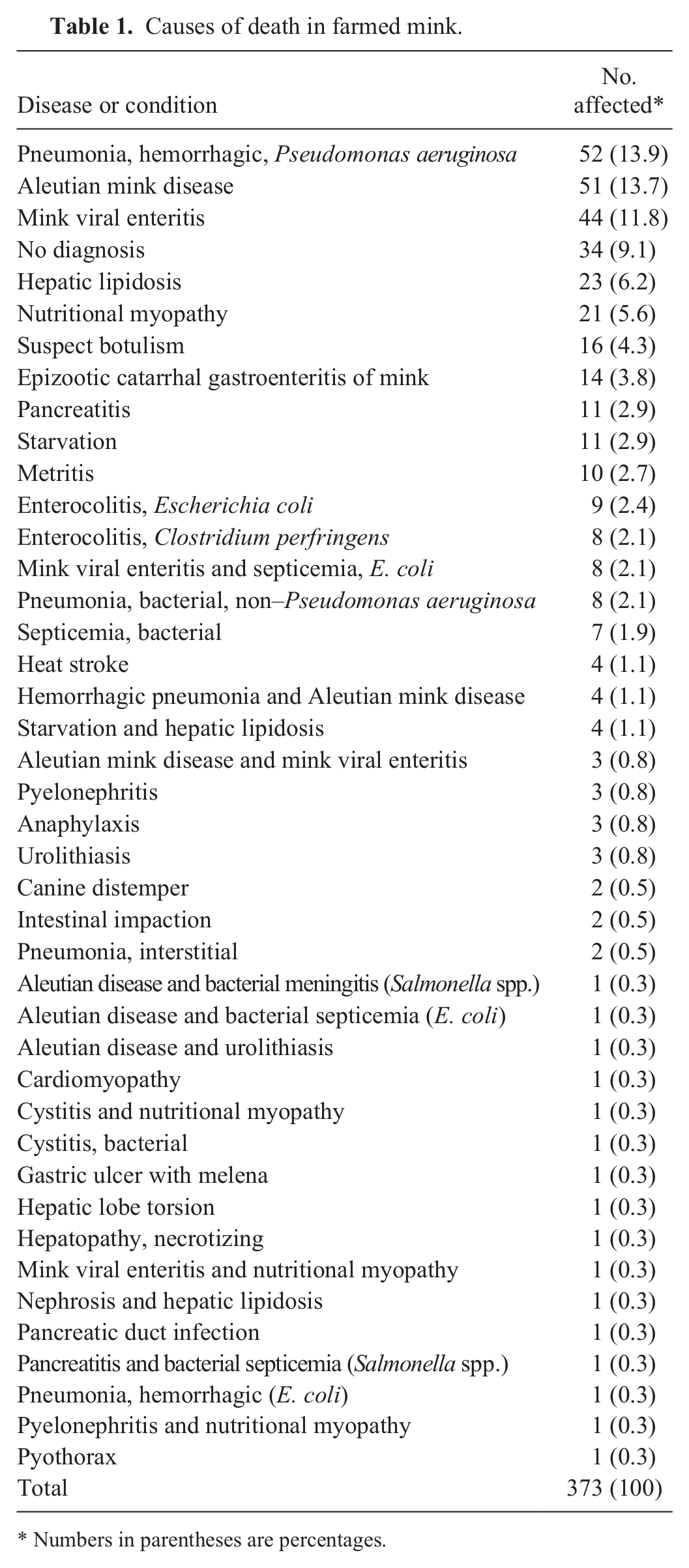
Numbers in parentheses are percentages.
Four infectious diseases alone or in combination with other diseases accounted for 53.9% of all mink deaths (n = 373): bacterial pneumonia (67 [52 Pseudomonas aeruginosa, 15 other], 17.8%), Aleutian mink disease (61, 16.4%), mink viral enteritis (56, 15.0%), and bacterial enterocolitis (17, 4.6%; Table 1). In an additional 16 (4.3%) mink, botulism was suspected from clinical history (flaccid paralysis) but could not be confirmed by laboratory testing. The following 2 categories of noninfectious causes of death alone or in combination with other diseases accounted for 19.0% of all mink deaths (n = 373): hepatic lipidosis, nutritional deficiencies, or starvation (60, 16.1%); and pancreatitis (noninfectious only; 11, 2.9%).
Principal causes of mortality
There were 104 juvenile mink necropsied (body weight: ≤0.9 kg). Seven diseases accounted for 71 (68.3%) of all juvenile mink deaths, shown in Table 2. There were 39 intermediate-aged mink necropsied (body weight: 0.91–1.19 kg). Four diseases accounted for 31 (79.5%) of all intermediate-aged mink deaths, shown in Table 3. There were 135 adult mink necropsied (body weight: ≥1.2 kg). Five diseases accounted for 92 (68.1%) of all adult mink deaths, shown in Table 4.
Principal causes of death in juvenile mink.

Numbers in parentheses are percentages.
Principal causes of death in intermediate-aged mink.

Numbers in parentheses are percentages.
Principal causes of death in adult mink.

Numbers in parentheses are percentages.
Discussion
The most common causes of mortality in farmed mink were similar among juvenile, intermediate, or adult aged animals. While 11 diseases accounted for nearly 95% of the deaths diagnosed in mink, the following 4 diseases that caused most of the death loss across all age groups: bacterial pneumonia (primarily caused by P. aeruginosa), Aleutian mink disease, mink viral enteritis, and hepatic lipidosis. Three of these diseases have consistently been reported as major causes of mink mortality in other studies from the United States and other regions of the world, despite widespread attempted control programs in some countries.7,10,12,17,20 The exception is mink viral enteritis; while it is described as a common mink disease, there are no refereed publications regarding its prevalence for more than 20 years.
Results are likely representative of the diseases on Utah and Idaho mink farms, representing the majority of mink production in the Intermountain West. With only 1 other submission of a group of mink from 1 farm in Wisconsin, a potential limitation of the study is that results may not represent mink diseases in other regions of the United States.
Pneumonia in mink caused by P. aeruginosa, often called fatal hemorrhagic pneumonia, has been described as an important cause of farmed mink death losses since 1953. The disease spreads rapidly, and mortality can be as high as 75%. The disease is more common during the fall, but can occur at any time of year.21,22 The pathogen is inhaled and spreads to all lung lobes via airways. Transmission between mink is rapid by aerosolization and inhalation. 15
Pseudomonas aeruginosa is more virulent in mink than in any other mammal as far as is known, but the basis for their unique susceptibility is unknown. The organism can be found in water cups, standing water, feed troughs, and unclean cages on mink farms. 10 Isolates from mink have been classified into more than 30 different P. aeruginosa types by various methods. Nearly always, all mink tested during an outbreak on a farm are infected with the same strain of P. aeruginosa, but the strains often change between outbreaks on a given farm. Sometimes, multiple farms in a single area, such as those served by the same mink food kitchen, are all infected with the same strain of P. aeruginosa within a short time. 10 (It has been the experience of the authors that mink producers often “blame” the feed first as the source of many outbreaks of various types of disease on their farms.)
Considerable interest in development of mink vaccines against hemorrhagic pneumonia caused by P. aeruginosa existed during the 1970s and 1980s. The vaccine is relatively expensive and the duration of protective immunity is short (~90 days), so vaccination is inconsistently used by mink producers worldwide. 10 However, mink producers in Utah and the Intermountain West are nearly all members of a large cooperative that routinely vaccinates 8-week-old kits against. P. aeruginosa with a single-dose bacterin toxoid vaccine. Sanitation is the other principal control measure, keeping feed troughs, water troughs, and cups clean, and cages as clean and dry as possible.
Aleutian mink disease is caused by Aleutian mink disease virus (AMDV; Carnivore amdoparvovirus 1), a parvovirus. This is a worldwide disease of financial importance in farmed mink, described for at least 60 years. The name comes from a mutation observed in the early 1940s in farmed mink that resulted in a dark blue coat color similar to the Aleutian fox; the disease was later first observed in these Aleutian mink.8,11,12,17 Disease signs with AMDV vary with the age and the coat color of mink, as well as the strain of the virus. 12 There are 4 recognized AMDV strains: Utah-1, Ontario (Canada), Montana, and Pullman (Washington). Aleutian, sapphire, or violet mink are usually more susceptible to clinical disease and death from AMDV than are pastel mink. Utah-1 and Ontario strains of AMDV usually infect more mink of any coat color than the Montana and Pullman strains.9,16 In this study, specific strains of AMDV were not determined. Clinical signs can include polydipsia, anorexia, gingivitis, hemorrhage, and depression, followed by death within weeks to months, or by a chronic condition with immunosuppression. Young kits may die with acute pneumonia caused by AMDV.2,8 In older mink, death is attributed to glomerulonephritis or secondary infection following immunosuppression. At postmortem examination, gingivitis, oral ulcers, multiple hemorrhages, arteritis, enlarged pale yellow kidneys with petechiae, pitting, or atrophy, splenic congestion and/or splenomegaly, or hepatic chronic passive congestion may be seen.8,12
Pathogenesis of AMDV is via hypergammaglobulinemia, lymphocytic–plasmacytic proliferation, and subsequent glomerulonephritis resulting in kidney failure, as well as immunosuppression. Histologically, AMDV infiltrates spleen, liver, kidney, lung, mesenteric lymph nodes, and intestine in conjunction with widespread arteritis.2,16
Eradication strategies have been attempted on some commercial mink farms worldwide, using a counterimmunoelectrophoresis (CIEP) test and removal of test-positive mink, and/or total depopulation and premises disinfection followed by restocking with a population of mink testing free of AMDV by CIEP testing. Nevertheless, AMDV is endemic on most commercial mink farms. 17 Since 2008, a PCR assay that amplifies a fragment of the NS1 gene of AMDV, with a reported sensitivity of 94.7% and specificity of 97.9%, has been used in eradication programs. However, the PCR has only been studied for use on spleen and lymph nodes postmortem, not as an antemortem test on serum or blood. 12
Persistence of AMDV in the environment on farms had long been postulated, but published studies regarding the virus in the environment on mink farms were lacking. A previous study compared 5 mink farms completely test negative for AMDV for 10 years with 3 farms on which 3%, 4%, and 25% of mink, respectively, tested positive for AMDV during the previous 2 years. Cotton-tipped sterile swabs were used to collect 79 samples from environmental sources for real-time PCR testing including feed silos and their stairways, feed truck tire ruts along the edge of farms, cage walls, workers’ shoes, dining areas for farm personnel including chairs and cabinets, leather mink handling gloves, inside surfaces of farm vehicles, street clothes, coveralls, mink feces, water on floors, and the truck drive-through disinfection baths. The virus was detected in every type of environmental sample on farms with AMDV, with 46 out of 49 (93.9%) samples testing positive for the virus. All 30 samples from the environment on the AMDV-free farms tested negative for the virus. 17 The results demonstrate that the environment is an important habitat for the virus, but how long AMDV may persist in various places on mink farms in the absence of any infected mink needs further investigation. The most widely recommended and applied control measures currently focus less on the environment than on test and removal strategies. However, some mink producers have told the authors that if the environment is not kept clean and dry, and rodents are not controlled, AMDV will always return to a completely closed AMDV-free mink farm within 2 years.
Mink enteritis was first reported in 1949, but the causative parvovirus, mink enteritis virus (MEV; Carnivore protoparvovirus 1 strain) was not identified until 1952, giving rise to the name mink viral enteritis. If clinical signs develop, the signs appear between 4 and 14 days postexposure. Diarrhea, anorexia, squinting, vomiting in some animals, and depression result. During the first 2 days of diarrhea, feces often contain mucus and intestinal casts, and thereafter consist of watery fluid. If death occurs, it is usually ~7 days postexposure, ~3 days following onset of clinical signs. Recovery is usually complete within 14 days postexposure.18,23
Pathology of MEV infection consists of fibrinous enteritis, hemorrhage and enlargement of the spleen, edema of gastrointestinal lymph nodes, intestinal epithelial necrosis, and lymphopenia. Histologic changes in the intestine include crypt epithelial cell necrosis with villus blunting and fusion, increased proportion of goblet cells in mucosal epithelium, and mildly increased numbers of lymphocytes in the lamina propria, especially in ileum. Seven to 8 days after infection, the duodenum, jejunum, and ileum often have nearly complete sloughing of intestinal mucosa. By day 10, surviving mink have near complete regeneration of intestinal mucosa, and, histologically, there are plasma cells and leukocytes throughout the lamina propria. Rarely, fibrin deposits and cellular debris may persist up to 14 days postexposure.19,23
Raccoons are highly susceptible to disease when inoculated experimentally with MEV and develop clinical signs and mortality similar to mink. Surviving raccoons as well as those that eventually died of the disease were found to shed MEV in their feces. Experimentally infected red foxes shed MEV intermittently in their feces. 3 The role of possibly naturally infected wild raccoons or red fox in spreading the disease to farmed mink, or the prevalence of MEV in those species in the wild, is not known.
There is great genetic and antigenic similarity between MEV and canine parvovirus, feline panleukopenia virus, and raccoon parvovirus. This has aided development of vaccines designed to immunize against MEV (as well as canine parvovirus and feline panleukopenia virus). In a challenge experiment, 3 different MEV vaccines each protected 5-month-old mink (6 per group, n = 18 total) inoculated on the same day as vaccination against signs of MEV; all 6 control mink administered a placebo contracted clinical signs on day 5 and were euthanized on day 8. 13 In another vaccine study, 10 mink of unspecified age were vaccinated with a modified live attenuated MEV vaccine and then challenged 7 weeks later with virulent MEV. None developed clinical signs, while 71% of control unvaccinated mink developed clinical signs. 5
The major control measure for MEV is vaccination. The mink producers in the cooperative in Utah and the Intermountain West routinely immunize 8-week-old kits for MEV with a single-dose modified live vaccine.
Hepatic lipidosis has been described in mink since 1948, but there have been few refereed publications concerning the disease. It was observed that farmed mink fed diets with relatively high fat in comparison to diets with relatively high protein had heavier livers with increased fatty infiltration at pelting. 6 Commercial mink food kitchens receive a varied supply of commodities that are blended into rations. Such commodities include bovine spleen, bovine liver, meat scraps, different species of fish, and poultry eggs. There has been speculation that species and amounts of fish or fish oil fed to mink affect hepatic lipidosis, but these claims have not been substantiated by controlled studies.1,7
Hepatic lipidosis in mink has been attributed partly to their body shape, with a high surface area-to-volume ratio being energetically inefficient. This likely contributes to the observed rapid mobilization of fat reserves when mink stop eating. Most mink that have stopped eating for 3–6 days, such as can result from other diseases or periparturient stress after birth of kits, rapidly increase the percentage of fat in their livers.4,7 In a previous study, the absolute amount of fat in mink livers increased nearly 6-fold after 5 days of not eating. 20 If mink are obese prior to anorexia, fatty infiltration of the liver is even more likely should they decrease or cease feed intake. 7 Recommended control measures for hepatic lipidosis are avoidance of obesity in mink, and efforts to minimize anorexia from other diseases or following the birth of kits and during the early nursing period.
Much of the mortality in farmed mink is ascribed to infectious diseases caused by viruses and bacteria. Many of the recommended and biologically sound control measures for control of mink disease involve environmental sanitation, immunization, control of obesity, and good overall management on mink farms.
Footnotes
Acknowledgements
We thank Dr. Jason Lott, River Valley Vet Hospital and consultant to Utah Fur Breeders, for his consultation and advice regarding the mink industry.
a.
All agars from Hardy Diagnostics, Santa Maria, CA.
b.
API test kits, bioMérieux Inc., Durham, NC.
c.
BBL Crystal, Becton Dickenson and Co., Franklin Lakes, NJ.
d.
Scintilla Development Co. LLC, Bath, PA.
e.
BD Universal viral transport system, Becton Dickenson and Co., Franklin Lakes, NJ.
f.
Vetstar animal disease diagnostic system, Advanced Technology Corp., Ramsey, NJ.
g.
Excel, Microsoft Corp., Redmond, WA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: General financial support for the activities of the diagnostic laboratory was received from Utah State University and the Utah Department of Agriculture and Foods.
