Abstract
Primary hypoadrenocorticism, or Addison’s disease, is an autoimmune condition common in certain dog breeds that leads to the destruction of the adrenal cortex and a clinical syndrome involving anorexia, gastrointestinal upset, and electrolyte imbalances. Previous studies have demonstrated that this destruction is strongly associated with lymphocytic-plasmacytic inflammation and that the lymphocytes are primarily T cells. In this study, we used both immunohistochemistry and in situ hybridization to characterize the T-cell subtypes involved. We collected postmortem specimens of 5 dogs with primary hypoadrenocorticism and 2 control dogs and, using the aforementioned techniques, showed that the lymphocytes are primarily CD4+ rather than CD8+. These findings have important implications for improving our understanding of the pathogenesis and in searching for the underlying causative genetic polymorphisms.
Keywords
Primary hypoadrenocorticism, or Addison’s disease (AD), is believed to be an inherited disorder in dogs that involves the immune-mediated destruction of the adrenal cortex. 13 Evidence for the immune-mediated nature of the disease originates from histologic studies of autopsy specimens THAT demonstrated lymphocytic-plasmacytic inflammation in the adrenal cortex of acutely affected animals. 3,7,12 To date, the relative contribution of CD4+ versus CD8+ T cells to the lymphocytic inflammation observed in the adrenal cortex of dogs with AD has not been explored. A recent study of canine adrenal glands demonstrated a preponderance of T-cell (vs B-cell) infiltrates in dogs with adrenalitis and AD 5 ; however, specific T-cell subtypes were not evaluated.
Here, we used both immunohistochemistry (IHC) and in situ hybridization (ISH) on adrenal gland tissue collected from dogs with AD to examine the relative proportion of CD4+ versus CD8+ T lymphocytes. Based on prior studies in dogs showing an association between major histocompatibility complex (MHC) class II alleles and AD in certain breeds, we hypothesized that we would observe primarily CD4+ T cells in affected adrenal tissue. 8,11 However, we also reasoned that the concurrent presence of a strong CD8+ T-cell response might implicate the involvement of other genes or pathways in the pathogenesis of canine AD as well.
Animal Selection
We reviewed the autopsy database at the North Carolina State University, College of Veterinary Medicine, using the search terms
Our database search revealed 17 possible cases of dogs with AD. After reviewing the laboratory findings and clinical history, we selected the 10 dogs with a confirmed diagnosis of AD for histologic review. Five of these 10 cases were excluded based on the absence of lymphocytic adrenalitis. The remaining 5 dogs were included in this study and are described in Table 1. Affected dogs included 5 different breeds with an age range of 2.3 to 11 years (median, 7.9 years) at the time of autopsy. Two dogs had atypical and 3 dogs had typical AD.
Signalment, Clinical History, and Clinical Chemistry in Dogs With Primary Hypoadrenocorticisma
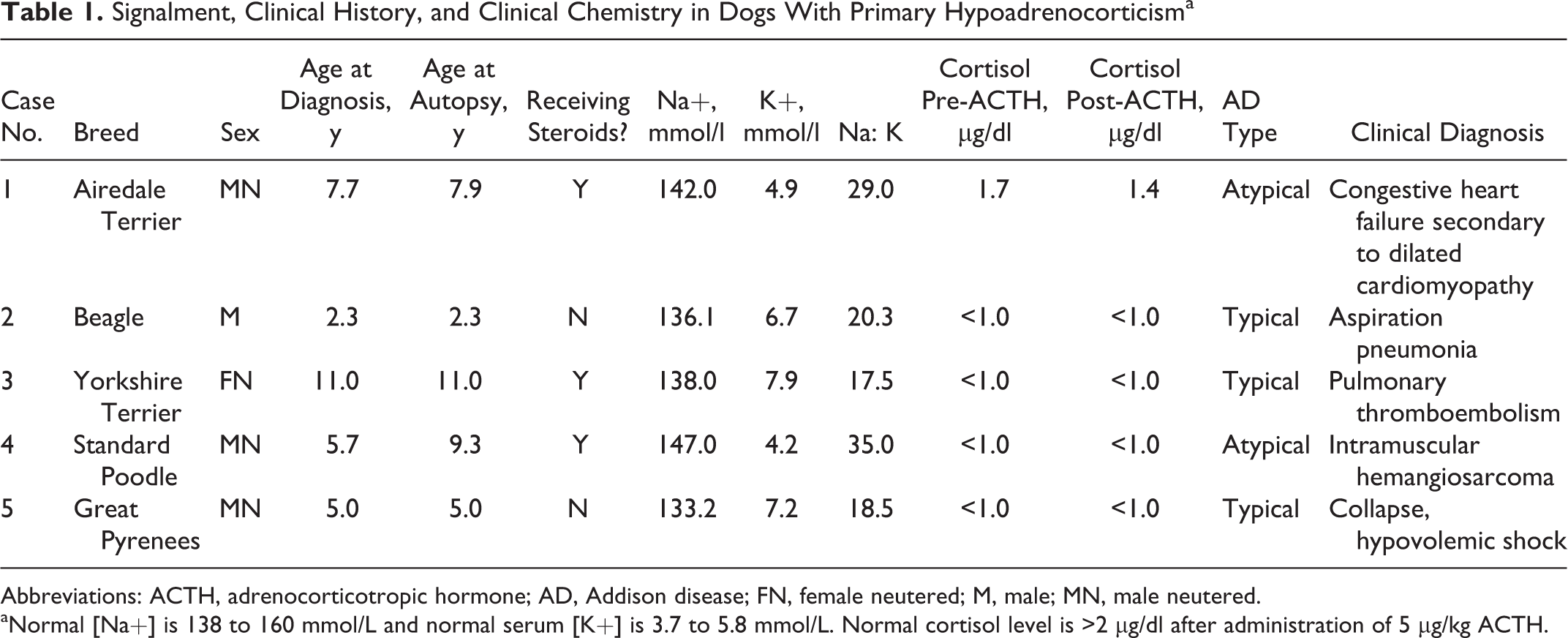
Abbreviations: ACTH, adrenocorticotropic hormone; AD, Addison disease; FN, female neutered; M, male; MN, male neutered.
aNormal [Na+] is 138 to 160 mmol/L and normal serum [K+] is 3.7 to 5.8 mmol/L. Normal cortisol level is >2 µg/dl after administration of 5 µg/kg ACTH.
We also searched the autopsy database over the same time period for cases in which adrenal gland tissue was collected but determined to be histologically normal for use as control samples. A subset of these cases was reviewed to ensure a normal cortical/medullary ratio and the absence of adrenocortical inflammation. We selected a 6.8-year-old spayed female mixed-breed dog with a premortem clinical diagnosis of acute kidney injury and an 8.8-year-old castrated male American Cocker Spaniel with a premortem clinical diagnosis of vomiting, diarrhea, and seizures.
Histological Evaluation
HE-stained sections from all samples were screened to ensure proper tissue fixation, freedom from remarkable microscopic artifacts, and adequate amount of adrenal gland tissue from each case for subsequent studies. Following review of the HE-stained sections, immunohistochemistry was performed on adrenal gland tissue sections using antibodies against CD3, CD4, and CD8, and in situ hybridization was performed on sections using probes for CD4 and CD8. These methods are described in the Supplemental Materials.
Each tissue section (1 section per animal per stain) was evaluated by a board-certified (American College of Veterinary Pathologists) veterinary pathologist (J.M.L.) who was blinded to the clinical diagnosis in each case. The sections were scored using a 4-tiered, semiquantitative method 6,9 based on the percentage of positively stained lymphocytes as follows: 0 = 0% to 2% positive cells, 1 = 2% to 10% positive cells, 2 = 10% to 50% positive cells, and 3 = greater than 50% positive cells. Given the small sample size as well as the semiquantitative nature of our scoring algorithm, we did not attempt to perform statistical analyses on our findings.
A summary of the overall histological findings, immunohistochemistry, and in situ hybridization is provided in Table 2. Cases 1, 2, and 3 generally showed multifocal, moderate cortical inflammation. In some areas, lymphocytes and plasma cells were admixed with small to moderate numbers of macrophages, some of which contained phagocytosed pigmented material. Areas with pigmented macrophages tended to make scoring of 3,3′-diaminobenzidine-stained lymphocytes more challenging. In case 4, the intensity of the inflammation was milder. In case 5, one adrenal gland had a band of severe lymphocytic-plasmacytic adrenalitis, along with scattered hemorrhages and several thrombosed small-caliber blood vessels.
Pathologic Findings in Adrenal Gland Sections of 5 Dogs With Primary Hypoadrenocorticisma
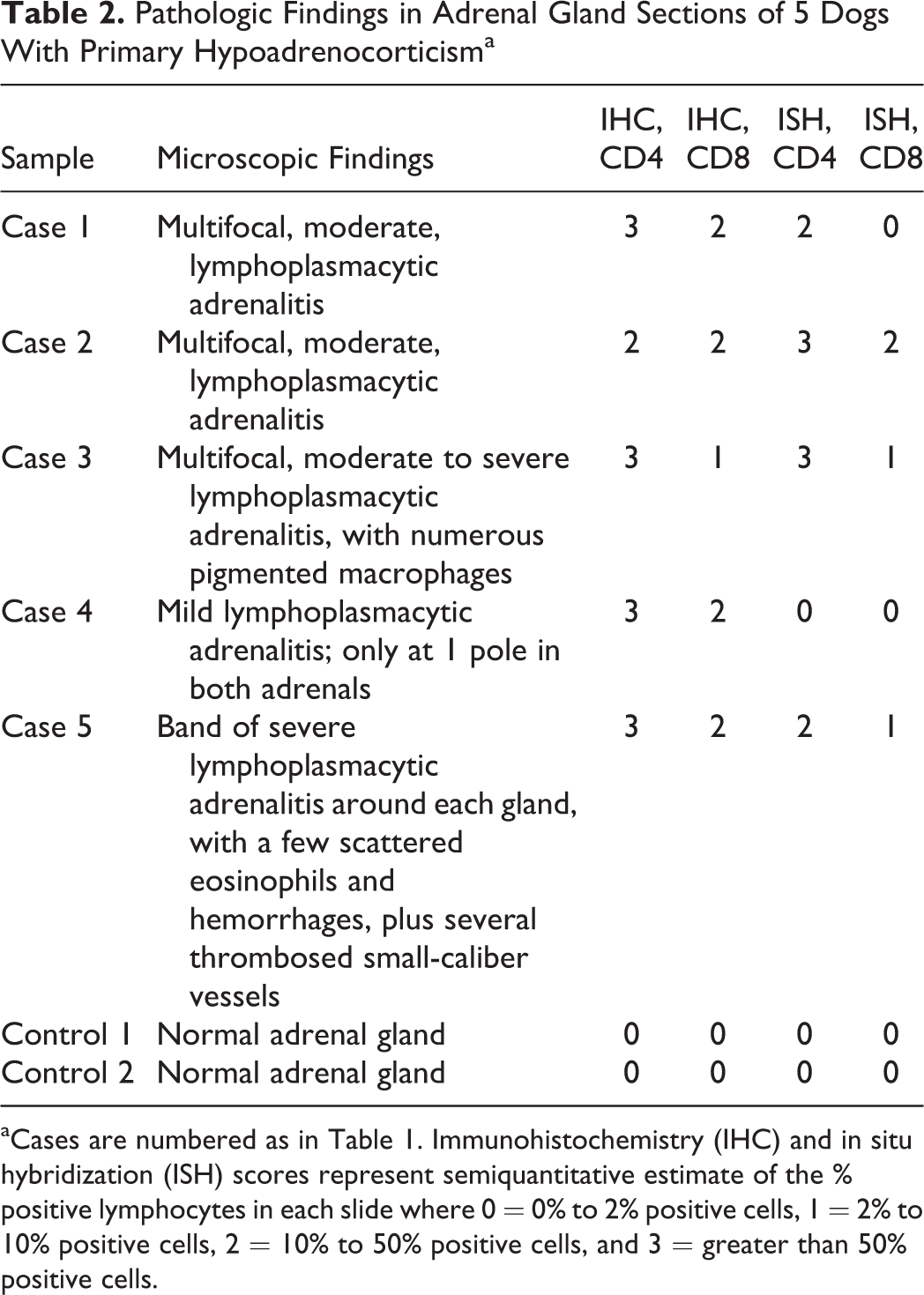
aCases are numbered as in Table 1. Immunohistochemistry (IHC) and in situ hybridization (ISH) scores represent semiquantitative estimate of the % positive lymphocytes in each slide where 0 = 0% to 2% positive cells, 1 = 2% to 10% positive cells, 2 = 10% to 50% positive cells, and 3 = greater than 50% positive cells.
In all sections, the staining quality and intensity for both the IHC and the ISH sections were judged to be very good, with an overall even staining distribution of the lymphocytic infiltrates. The CD3-stained sections were of good quality and served as positive controls for T-cell staining, to ensure the condition of these postmortem tissue specimens (adequate fixation and lack of significant postmortem autolysis). As expected, the 2 control adrenals were scored as 0 with no remarkable nonspecific staining of nonlymphocyte leukocytes.
Overall in the IHC results of the 5 cases evaluated, the lymphocyte population comprised mainly CD4+ cells, with fewer numbers of CD8+ cells present by comparison (Fig. 1). Staining extent for CD4 on 4 of 5 cases was scored as 3, while the fifth case was scored as 2. Regarding CD8 IHC, the staining results were somewhat more variable, with scores of 2 and 1, but with some sections having more problems with nonspecific staining.
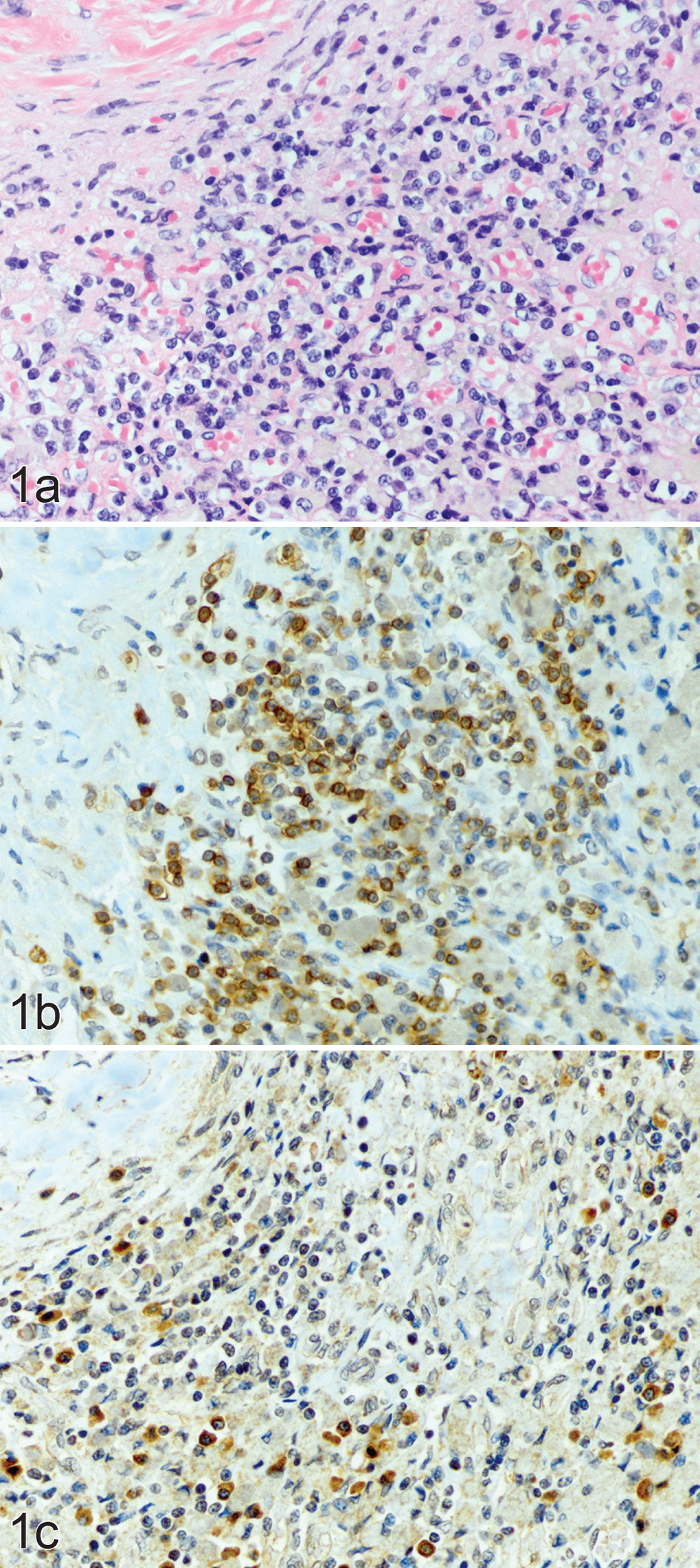
Primary hypoadrenocorticism, adrenal cortex; dog 3. (a) Lymphocytes, plasma cells, and foamy macrophages densely infiltrate the adrenal cortex. Hematoxylin and eosin. (b) Same area of the adrenal cortex, showing numerous CD4-positive lymphocytes with strong positive staining of the cytoplasmic membrane; scored as grade 3. Immunohistochemistry (IHC) for CD4. (c) Same area, showing fewer CD8-positive lymphocytes; scored as grade 1. IHC for CD8.
ISH staining followed a similar pattern to the IHC staining, with mainly CD4+ T cells and very few CD8+ cells by comparison (Fig. 2). With CD4, the AD cases were scored as either 2 or 3, whereas CD8 was again more variable. Two of the ISH sections had comparatively faint staining, likely due to the samples not being optimally fixed for ISH procedures. A summary of the semi-quantitiatve scoring for both IHC and ISH is shown in Fig. 3.
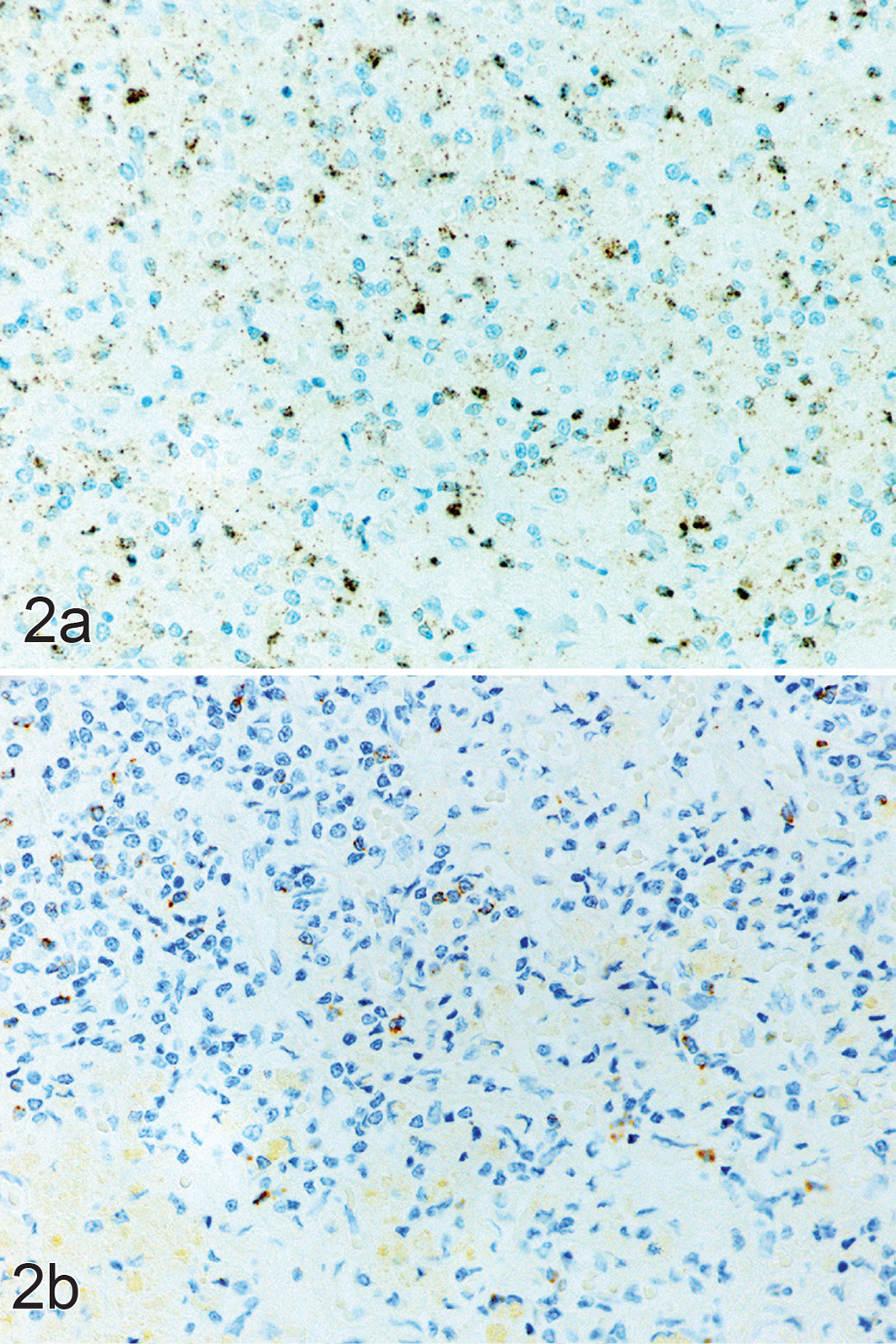
Primary hypoadrenocorticism, adrenal cortex; dog 3. (a) Same area of cortex as in Figure 1, showing numerous CD4-positive lymphocytes; scored as grade 3. In situ hybridization (ISH) for CD4. (b) Same area, with only a few scattered CD8-positive lymphocytes; scored as grade 1. ISH for CD8.
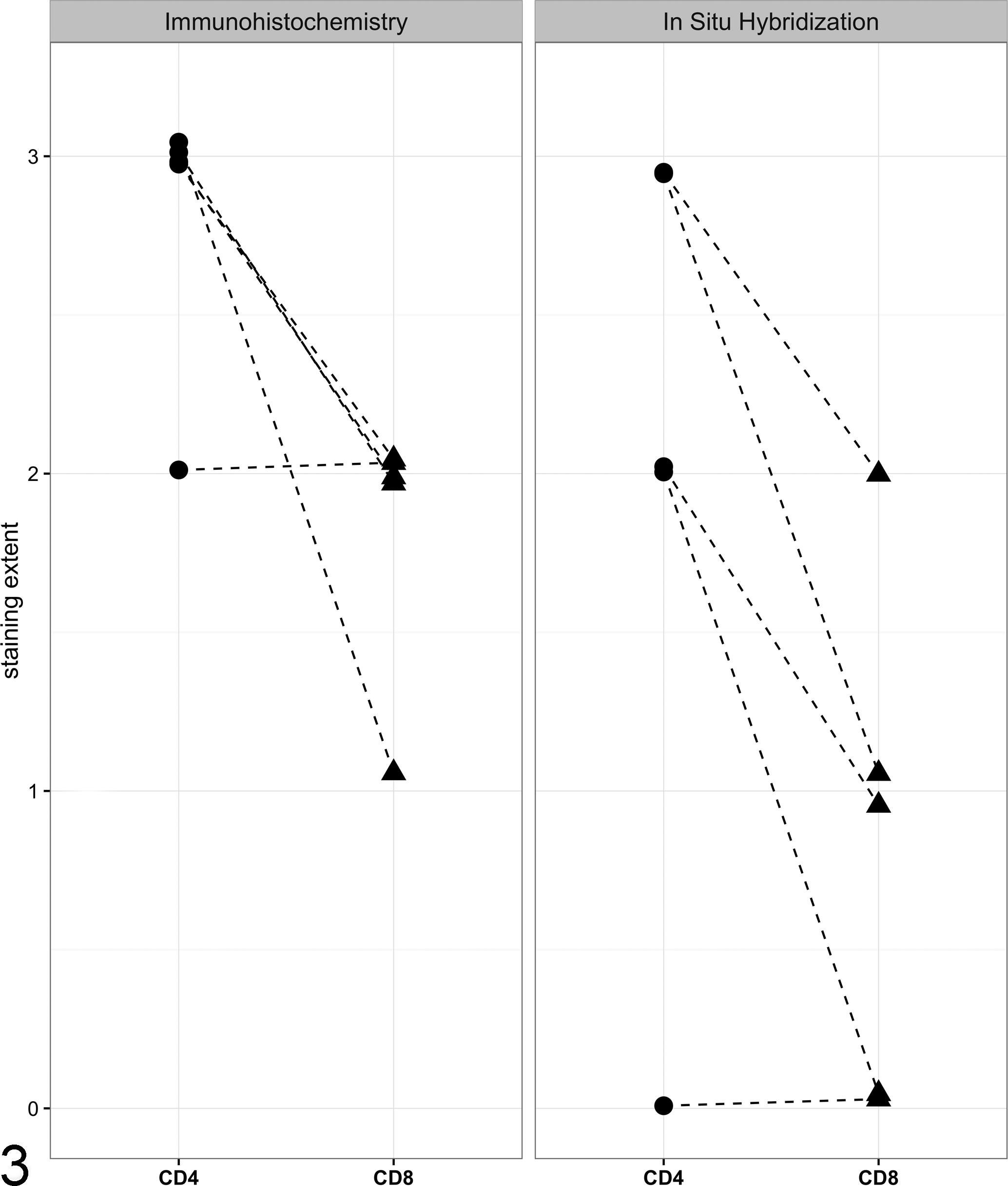
Semiquantitative scoring of lymphocytic inflammation in the adrenal glands of 5 dogs with primary hypoadrenocorticism demonstrating a predominance of CD4+ cells. Adrenal gland sections were evaluated using immunohistochemistry (IHC; left panel) and in situ hybridization (ISH; right panel) using antibodies (IHC) or probes (ISH) for CD4 and CD8, and the percentage of each cell type relative to the total number of lymphocytes was quantified on a scale of 0 to 3. CD4 staining is represented by circles and CD8 staining is represented by triangles; dashed lines connect the findings for each sample. A small amount of jitter was added to each point to permit visualization of overlapping values.
Using both IHC and ISH, our analysis showed that the preponderance of the lymphocytes in the adrenal cortices of the dogs we evaluated with AD are CD4+. This finding is consistent with the association of MHC class II genes and canine AD, 1,8,11 as certain CD4+ T-cell subsets (Tfh cells) directly interact with MHC class II molecules on the surface of dendritic cells. 10 It is also consistent with what is believed to occur in the adrenal glands of human patients with AD. 4 Furthermore, given that Tfh cells stimulate B cells to produce specific antibodies, further efforts to identify autoantibodies in dogs may be warranted. To date, autoantibodies have been detected against the steroidogenic enzyme CYP11A1 in approximately 25% of dogs 2 ; however, additional research in this area may yield insights regarding other autoantibodies in a larger percentage of the canine AD population.
In addition to noting a preponderance of CD4+ T cells in the adrenal glands of dogs with AD, we also found evidence of a small number of CD8+ T cells contributing to the inflammatory response. This finding suggests that the pathogenesis of AD is complex and that multiple pathways may be responsible for the immune-mediated destruction of the adrenal cortex in canine AD.
Beyond the implications of our study regarding the pathogenesis of canine AD, to our knowledge, this study represents the first report of both IHC and ISH staining of canine adrenal glands. Detection of specific molecular targets can be challenging even under the best of conditions, but the immunostaining of lymphocyte subsets was judged to be of good to excellent quality in all sections. Likewise, with the next-generation methodology of ISH (RNAscope technology), custom probes were sensitive enough to detect single messenger RNA transcripts in these fixed, paraffin-embedded canine tissues. Compared to flow cytometry, histology-based techniques allow visualization of the “tagged” cells within their morphological context and, in this case, their relationship to other inflammatory infiltrates and their relative distribution and concentration within the adrenal cortex.
One limitation of this study was the small sample size. The reason for the small sample size is largely a function of the retrospective nature of this work, as well as the fact that most of the adrenal glands we initially reviewed did not show evidence of active lymphocytic-plasmacytic adrenalitis. Another limitation of this study was that we were unable to control for the time after disease onset. As the nature of the inflammatory response is likely to change over time as the adrenal cortex is destroyed in AD, it is reasonable to expect that the composition of the lymphocyte infiltrates may change over time as well.
In summary, in this study of the adrenal glands of 5 dogs with AD, we showed that the primary lymphocyte population consists of CD4+ T cells rather than CD8+ T cells. Further studies are warranted to delineate specific CD4+ T-cell subpopulations that may yield further insights regarding the pathogenesis of primary hypoadrenocorticism in dogs.
Footnotes
Acknowledgements
We thank the staff at the Histology Core Research Facility at the University of North Carolina at Chapel Hill, the Histology Laboratory at North Carolina State University College of Veterinary Medicine, and WIL Research for their assistance with this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by a National Institutes of Health T32 training award provided to SGF (5T32OD11130-07). Additional funding was provided by the American Kennel Club Canine Health Foundation and the Poodle Club of America Foundation.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
