Abstract
In 2015, H3N2 canine influenza emerged in dogs in the greater Chicago area. During this time, a 10-year-old German Shepherd dog presented to the referring veterinarian with lethargy and coughing that quickly progressed to death. This report describes the macroscopic and microscopic lesions and the molecular testing performed to identify the novel North American H3N2 strain of canine influenza. The larynx, pharynx, and trachea were covered by a fibrinonecrotic membrane. Bilaterally, the lungs had multifocal subpleural necrosis of the caudal lung lobes with hemorrhage, congestion, and pulmonary edema. Staphylococcus pseudointermedius was isolated from the lung. Mycoplasma cynos was identified by real-time polymerase chain reaction from nasal passages, oropharynx, larynx, trachea, and cranial lung lobes. The neuraminidase gene sequence from the influenza virus isolated obtained from this dog had ≥98% homology to the strain circulating in the Chicago area.
Canine influenza virus (CIV) was first discovered in Florida racetrack Greyhounds in 2004 and identified as subtype H3N8. 1,2 Over the next 2 years, serologically positive Greyhounds were found in 9 states and infected dogs with acute respiratory disease in 26 states. 1 In 2007, an H3N2 avian-origin influenza A virus caused a disease outbreak in pet and farmed dogs in South Korea. A subsequent outbreak in Jiangsu Province, China, was caused by an H3N2 strain that had greater than 99% homology to the South Korean strain. 4,5,8
A CIV H3N2 virus was isolated from a 10-year-old German Shepherd dog that resided in Cook County, Illinois (in the greater Chicago area). In February 2015, the dog was boarded in a kennel for 2 days, received an unspecified “flu vaccine” on day 1 of boarding, and 5 days later was presented to the referring veterinarian for anorexia and coughing. The physical exam showed a body temperature of 38.8°C, pulse of 110 beats per minute (bpm), respiratory rate of 24 breaths per minute, and pink mucous membranes with a capillary refill time of less than 2 seconds. Radiographs of the thoracic cavity revealed an interstitial lung pattern, suggestive of canine influenza pneumonia. The dog was hospitalized and given supportive therapy. On the third day of hospitalization, the dog was vomiting but quiet with a temperature of 39.4°C, pulse of 112 bpm, and tachypnea with an arterial oxygen saturation ranging from 61% to 68%. Abnormalities on complete blood count included leukopenia (1680 per μl; reference range, 6060–18 760 per μl) characterized by neutropenia (290 per μl; reference range, 2960–11 640 per μl), lymphopenia (320 per μl; reference range, 1060–6100 per μl) with band neutrophils, and thrombocytopenia (48 per μl: reference range, 188 000–484 000 per μl). Serum biochemistry panel revealed hypoalbuminemia (1.9 g/dl; reference range, 2.2–3.9 g/dl) and elevated urea (39 mg/dl; reference range 7–27 mg/dl) and alkaline phosphatase (269 U/l; reference range, 26–262 U/l). The dog became progressively dyspneic with a temperature of 39.4°C that progressed to death. Specimens from nasal mucosa were collected using polyester-tipped swabs in viral transport media. The necropsy was performed by the referring veterinarian, and unfixed tissues (lung, trachea, tracheobronchial lymph node, esophagus, tongue, heart, duodenum, pancreas, ileum, liver, kidney, and spleen) were shipped on ice overnight to the Wisconsin Veterinary Diagnostic Laboratory (WVDL).
Tissues were evaluated by macroscopic and microscopic examination, electron microscopy, polymerase chain reaction (PCR), bacterial and viral cultures, and sequence analysis of the virus isolate. For histopathology, tissues were fixed in formalin, and hematoxylin and eosin–stained sections were routinely prepared.
Tissues collected at the WVDL (cranial lung, tracheobronchial lymph node, liver, pancreas, ileum, and spleen) were homogenized, and DNA and RNA were extracted with a magnetic particle bead-based system. A broadly reactive quantitative real-time reverse transcriptase polymerase chain reaction (RT-qPCR) directed to the matrix gene of influenza A was used to identify positive specimens in a real-time PCR instrument (Applied Biosystems 7500 fast; Thermo Fisher Scientific, Singapore). 6 A respiratory multiplexed RT-qPCR panel (canine distemper, canine respiratory coronavirus, canine herpes virus, canine parainfluenza virus, canine adenovirus 2, Streptococcus zooepidemicus, Bordetella bronchiseptica, and Mycoplasma cynos) was performed on the nucleic acid extracted from the nasal mucosal swabs using a method developed and validated in the WVDL.
Virus isolation was performed in Madin-Darby canine kidney (MDCK) cells (ATCC CCL 34) and 9- to 11-day-old embryonated chicken eggs. Viral growth was monitored by RT-qPCR for a drop in threshold concentration (CT) values. Cultures negative for viral growth were subjected to 1 blind passage for 4 to 5 days for both the egg and MDCK cultures.
Grossly, the mucosa of the larynx and the proximal trachea was congested, markedly ulcerated, and multifocally covered by a fibrinonecrotic membrane (Fig. 2). All lung lobes were diffusely dark red, soft, with marked edema on cut section and random areas of subpleural hemorrhage (Fig. 1).
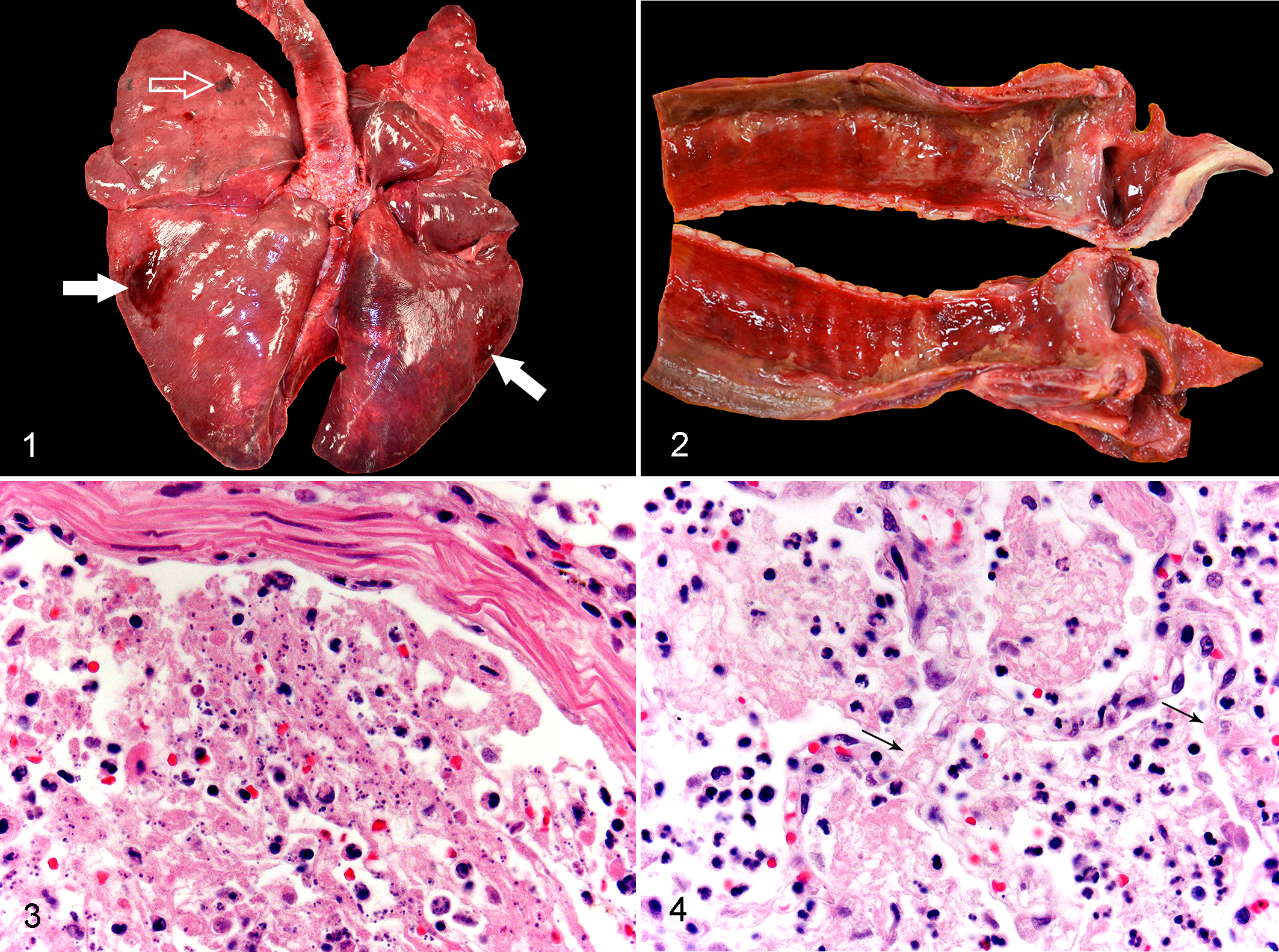
Influenza, lung dog.
Microscopically the bronchi, bronchioles, and alveoli contained necrotic debris, fibrin, hemorrhage, flocculent protein-rich edema degenerate neutrophils, and resident foamy macrophages (Figs. 3, 4; Suppl. Fig. S1). In some areas, inflammation and necrotic debris surrounded bronchial glands with mild to moderate replacement of small numbers of glands and cartilage. Frequent bronchioles lacked an epithelial lining, and the lumen contained neutrophils with necrotic cellular debris (Fig. 3). Alveolar septa were segmentally replaced by fibrin and necrotic debris while remaining septa were thickened by fibrinous exudate, viable and degenerate neutrophils, rare fibroblasts, and karyorrhectic debris (Fig. 4). Occasional foci of subpleural necrosis had severe hemorrhage, fibrin, and edema. A small number of small-caliber blood vessels contained fibrin thrombi (Suppl. Fig. S1). The pharynx and trachea revealed diffuse loss of the mucosal epithelium and lamina propria with replacement by fibrin, necrotic debris, hemorrhage, and edema admixed with large numbers of degenerate neutrophils, lymphocytes, plasma cells, and rare macrophages (Suppl. Figs. S1–S4). Necrosis extended into and surrounded cartilaginous rings multifocally with complete obliteration of the trachealis muscle (Suppl. Fig. S4).
The RT-qPCR assay for the matrix gene of influenza A was positive on nasal, oral, tracheal, and bronchial swabs and from samples of cranial lung and tracheobronchial lymph nodes (CT values of 20, 34.3, 36.4, 28.1, 31.8, and 39.6, respectively). The virus was not detected by RT-qPCR in liver, spleen, pancreas, ileum, or kidney. A multiplexed RT-qPCR panel for other canine viral pathogens was negative. M. cynos was identified by RT-qPCR from nasal, oropharynx, larynx, trachea, and cranial lung swabs (CT values 24–31). Microbial cultures of lung revealed numerous colonies of Staphylococcus pseudointermedius.
The neuraminidase gene of the H3N2 virus isolated in this case had ≥98% homology with that from the Chicago outbreak (A/canine/Illinois/12691/2018 [H3N2]; GenBank number KT002638.1) and the H3N2 CIV isolates from China and South Korea.
The present case shows lesions associated with the novel North American canine influenza virus H3N2. The lesions were consistent with those seen in dogs affected by the H3N2 virus in South Korea and China while less similar to the H3N8 canine influenza outbreak in Florida Greyhounds. Lesions common to both H3N2 and H3N8 infections were necrotizing tracheitis and bronchiolitis: a mononuclear cellular infiltrate surrounding and replacing tracheal and bronchial submucosal glands; sloughed luminal epithelial cells admixed with necrotic debris, degenerate neutrophils, edema, macrophages, and fibrin; and thickening of alveolar septa by a similar cellular inflammatory infiltrates admixed with hemorrhage, fibrin, and edema or complete loss (necrosis) of septa. The dogs in South Korea and the present H3N2-infected dog had alveolar necrosis with hyaline membrane formation, pulmonary vasculitis, and multifocal perivascular hemorrhage, whereas a limited number of cases of H3N8 infections had pulmonary vasculitis and thrombosis. 4
Clinically, 2 syndromes have been noted in natural and experimental H3N8 infection of dogs: a protracted clinical course causing moderate to severe morbidity followed by recovery and a peracute fatal form. 4 In the current H3N2 infection, the clinical course was approximately a 1-week duration, including coughing, depression, retching, fever of 39.4°C, sneezing, progressive lethargy, hypoxemia, and elevated respiratory rate. Previously reported cases had indications of subacute lesions, including squamous metaplasia (repair) in the trachea (South Korean H3N2) and erosion of the tracheal epithelium (Florida H3N8). The absence of these lesions in the current case most likely reflects the acute nature of the disease.
In this case, coccobacilli were noted in the inflammatory debris of the bronchioles as well as within the cytoplasm of neutrophils and macrophages. PCR revealed M. cynos and bacterial culture isolated S. pseudointermedius. The role of M. cynos in this case is not known.
The influenza A virus, specifically H3N8 and H3N2, has demonstrated rare interspecies transmission without viral reassortment. In the Florida outbreak, the entire H3N8 genome was transmitted from the equine lineage to racing Greyhounds 3 and subsequently to felines. 7 Similarly, full genomic transmission of H3N2 occurred between avian and canine species in South Korea, then identified with greater than 99% homology in dogs in Jiangsu, China. 5 In the current case, the neuraminidase gene of the H3N2 influenza virus had ≥98% homology with that from the Chicago outbreak (A/canine/Illinois/12691/2018; H3N2). Comparatively, the gross and microscopic lesions noted in this case are much more severe than the previously demonstrated H3N2 infections in China, South Korea, and Thailand. There are several possibilities for this occurrence, including the following: (1) the H3N2 virus is a novel virus in the immunologically naive dog population in the United States; (2) the virus obtained deadly virulence factors as it passaged from dog to dog; (3) this particular dog’s genetic and immunological status was permissive for severe infection; and (4) secondary bacterial infections exacerbated the effects of infections by the influenza virus. Answers to the first 3 questions will depend on future investigations into the virulence factors of the novel Illinois H3N2 virus, the location and type of canine influenza receptors used by the H3N2 virus, and the immunological reactions of dogs to influenza viruses.
Footnotes
Acknowledgements
We thank Dan Christensen and Craig Radi for their assistance with the photomicrographs and electron microscopy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
