Abstract
We investigated, in a cross-sectional study, the prevalence of antibodies against canine influenza A virus (CIV) H3N2 in serum samples collected from dogs and cats using a commercial ELISA and a hemagglutination inhibition (HI) test. Samples were obtained from 519 cats and dogs from 13 states within the United States. Data were analyzed for potential risk factors with positive sera (vs. negative sera) by logistic regression. Odds ratios and their 95% confidence intervals (CIs) were calculated by exponentiation of the regression coefficients. Ten dogs (2.21%; 95% CI: 1.05–3.98%) and 6 cats (8.96%; 95% CI: 3.36–18.48%) tested seropositive for CIV H3N2 by HI. One feline sample (1.49%; 95% CI: 0.04–8.04%) and 16 canine samples (3.53%; 95% CI: 2.01–5.61%) tested seropositive by ELISA for influenza A virus. There were no apparent associations between seropositivity and putative risk factors. All positive animals were from Indiana or Illinois; however, CIV H3N2 seroprevalence was not common in Illinois and Indiana.
Keywords
Influenza A virus (IAV; order Articulavirales, family Orthomyxoviridae, genus Alphainfluenzavirus) is a rapidly evolving virus commonly observed in mammals and birds. 1 IAV is a negative-sense RNA virus with segmented genome that has the ability to re-assort frequently, which gives rise to multiple reassortant strains of the virus. 12 Newly emerging, novel subtypes are common results of interspecies jumps, usually formed as the result of 2 differing strains being propagated in a single cell that gives the opportunity for viral genome swapping between different strains of IAV, or are the result of the innate ability of IAVs to undergo sequence mutation. 12
In 2007, canine influenza A virus (CIV) H3N2 was discovered in dogs in 3 veterinary clinics in South Korea during an outbreak of respiratory disease affecting dogs, and spreading among kennels and other facilities. 2 The virus, isolated from nasal swabs from infected dogs, was identified as an IAV that exhibited close homology to common IAVs in birds. 2 Originating with an avian lineage in South Korea and China, this H3N2 subtype began to infect dogs on multiple continents, causing clinical signs that included lethargy, coughing, sneezing, fever, and nasal discharge. 12 CIV H3N2 was initially thought to be contained within China, South Korea, and Thailand; however, this H3N2 strain (homology confirmed by sequencing analysis of hemagglutinin and neuraminidase genes) sparked a canine influenza pandemic in the United States in April 2015.2,6,11 Although CIV H3N2 may have been introduced to the United States via migrating wild birds or infected dogs traveling from Asia, the actual mode of entrance is unknown. Since its arrival in the United States, multiple CIV H3N2 outbreaks have occurred, initially in locations in and around Chicago, IL. From the Chicago area and Midwest region, the virus migrated to other states within the United States and into Canada (Office of the Chief Veterinarian for Ontario, Veterinary Advisory, Confirmed clusters of canine influenza in southern Ontario, https://oahn.ca/wp-content/uploads/2018/03/CIV-Disease-Advisory-2018-03-02-FINAL-1.pdf). The virus spreads via respiratory droplets (released when the dog sneezes, barks, or coughs) and direct contact with other infected hosts or fomites (https://www.avma.org/KB/Resources/Reference/Pages/Canine-Influenza-Backgrounder.aspx). 2 Cats can also be infected by the virus when exposed via direct dog-to-cat or cat-to-cat transfer, and can develop clinical disease as well as serum antibodies against H3N2 CIV. 3
Given the ability of CIV to spread quickly among groups of animals, efficient detection of CIV is very important. 8 Delaying detection could result in infected animals coming into contact with numerous naive individuals, thus leading to a widespread outbreak or the emergence of a new strain of CIV capable of infecting other species in close contact with dogs, such as cats and humans. To date, the most accurate method of detecting CIV genomic copies in acute infections is PCR. To detect infection by the virus, serologic tests such as hemagglutination inhibition (HI) assays or ELISA are utilized. 2 Virus isolation can be used for detection of viral particles, but with lower sensitivity and specificity.
We hypothesized that CIV seroprevalence would be higher among animals from the greater Chicago area than animals from other locations. The goals of our cross-sectional retrospective study were 1) to determine the prevalence of CIV seroreactivity in canine and feline serum samples using ELISA and HI methods designed to detect antibodies against IAV (e.g., CIV H3N2), with the understanding that ELISA will detect antibodies to all CIV, whereas HI will detect antibodies more specifically to CIV H3N2, and 2) to evaluate potential risk factors such as age, breed, location of residence, or recent travel to the Chicago area.
Sera from 452 dogs and 67 cats collected between April and August 2015 were obtained retrospectively from the Clinical Pathology Laboratory, Purdue University Veterinary Teaching Hospital (West Lafayette, IN). Sera were maintained frozen at −20°C until tested. The Purdue Animal Care and Use Committee (PACUC) approved the study.
A commercial ELISA kit (IDEXX, Westbrook, ME) targeting antibodies against IAV was used according to the manufacturer’s directions. HI testing was completed in accordance with methods described previously, using in-house reagents. 1 The cutoff titer for determining positivity of samples was determined through receiver-operator characteristic analysis. The cutoff titer for the HI test was determined at 100.0% sensitivity and 98.1% specificity to be 1:32 compared to the ELISA as reference, given that this commercial ELISA has been validated by IDEXX for detection of IgG antibodies to IAV H3N2 in canine and feline species.
Seroprevalence was calculated as the number of positive sera divided by the total number of sera tested × 100%. A 95% confidence interval (CI) using an exact binomial distribution was calculated for the seroprevalence of H3N2 virus. The associations of potential risk factors with positive sera (vs. negative sera) were assessed by logistic regression. Odds ratios and their 95% CIs were calculated by exponentiation of the regression coefficients.
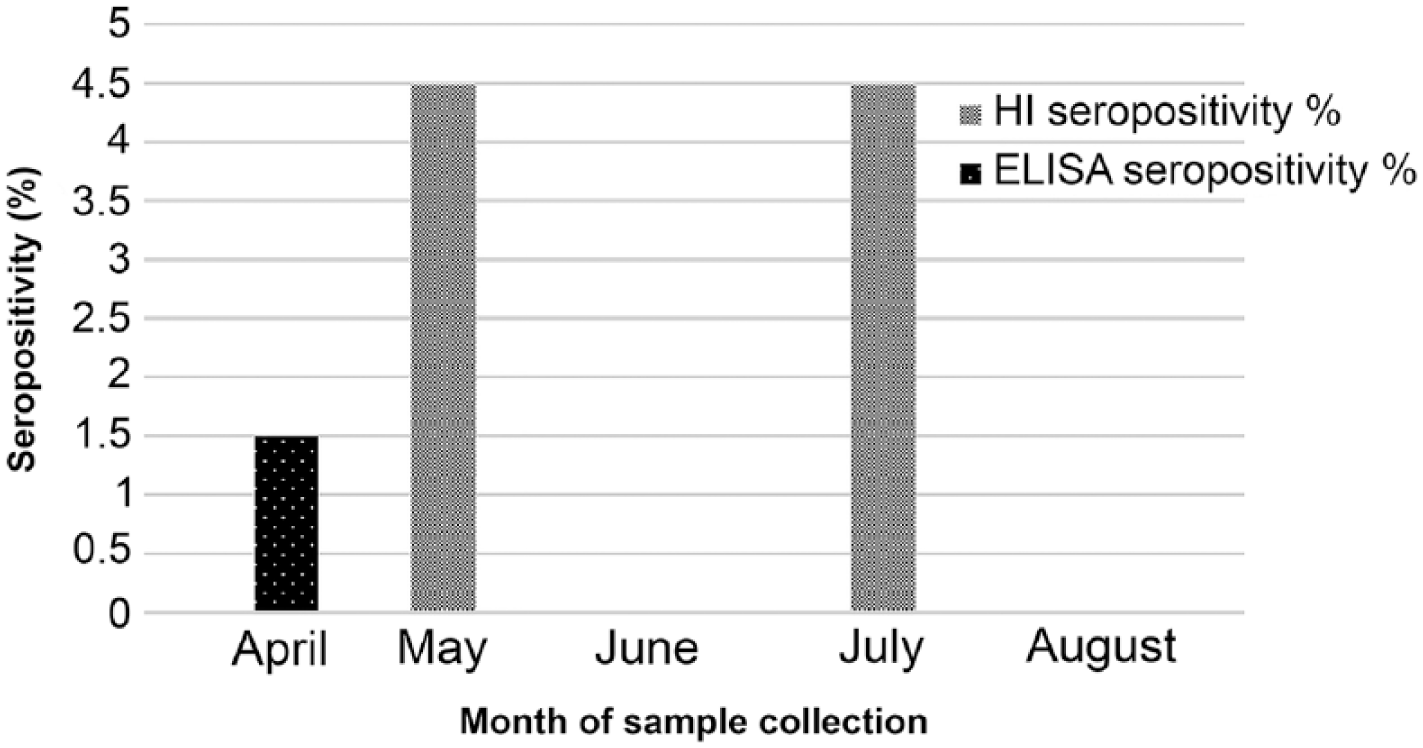
Animals tested were from Alabama, California, Iowa, Illinois, Indiana, Maryland, Maine, Michigan, Missouri, New York, Ohio, Tennessee, Virginia, and Wisconsin (Table 1). The majority of animals were from Indiana and Illinois.
Total number of canine and feline sera tested, and hemagglutination inhibition (HI)-positive and ELISA-positive canine and feline sera per state.
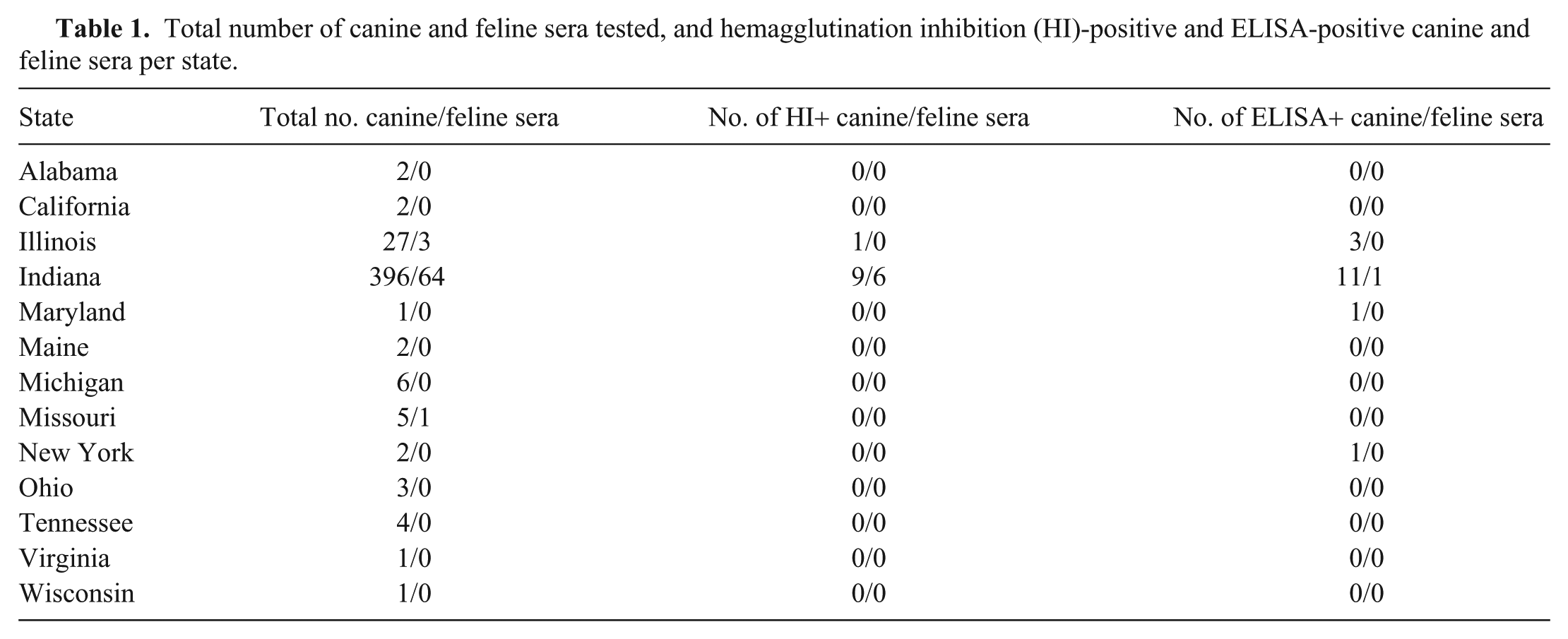
One feline sample from Indiana collected in April 2015 was ELISA-positive for IAV (1.49% seropositive; 95% CI: 0.04–8.04%; Fig. 1, Table 1). This sample was not positive in the HI test. The 66 ELISA-negative feline samples were obtained during the spring and summer of 2015 from cats residing in Illinois, Indiana, and Missouri. Six feline sera were considered positive for CIV H3N2 via HI testing (8.96% seropositive; 95% CI: 3.36–18.48%). These samples were collected in May and July 2015 from cats from Indiana, making the CIV H3N2 seropositivity rates for May and July 4.48% (95% CI: 0.93–12.53%; Fig. 1). The HI-negative samples included feline sera from Illinois, Indiana, and Missouri (Fig. 2). None of the HI-positive samples were ELISA-positive.
The distribution of ELISA and HI seropositivity rates (%) for feline samples by month (Apr–Aug 2015). Three samples collected in each of the months of May and July 2015 were HI-positive (4.5%).
Samples received from different states and tested by ELISA and HI for canine influenza A virus (CIV) H3N2. Sixteen canine samples (3.53%) were seropositive by ELISA for influenza A virus, and 10 dogs (2.21%) and 6 cats (8.96%) were seropositive for CIV H3N2 by HI.
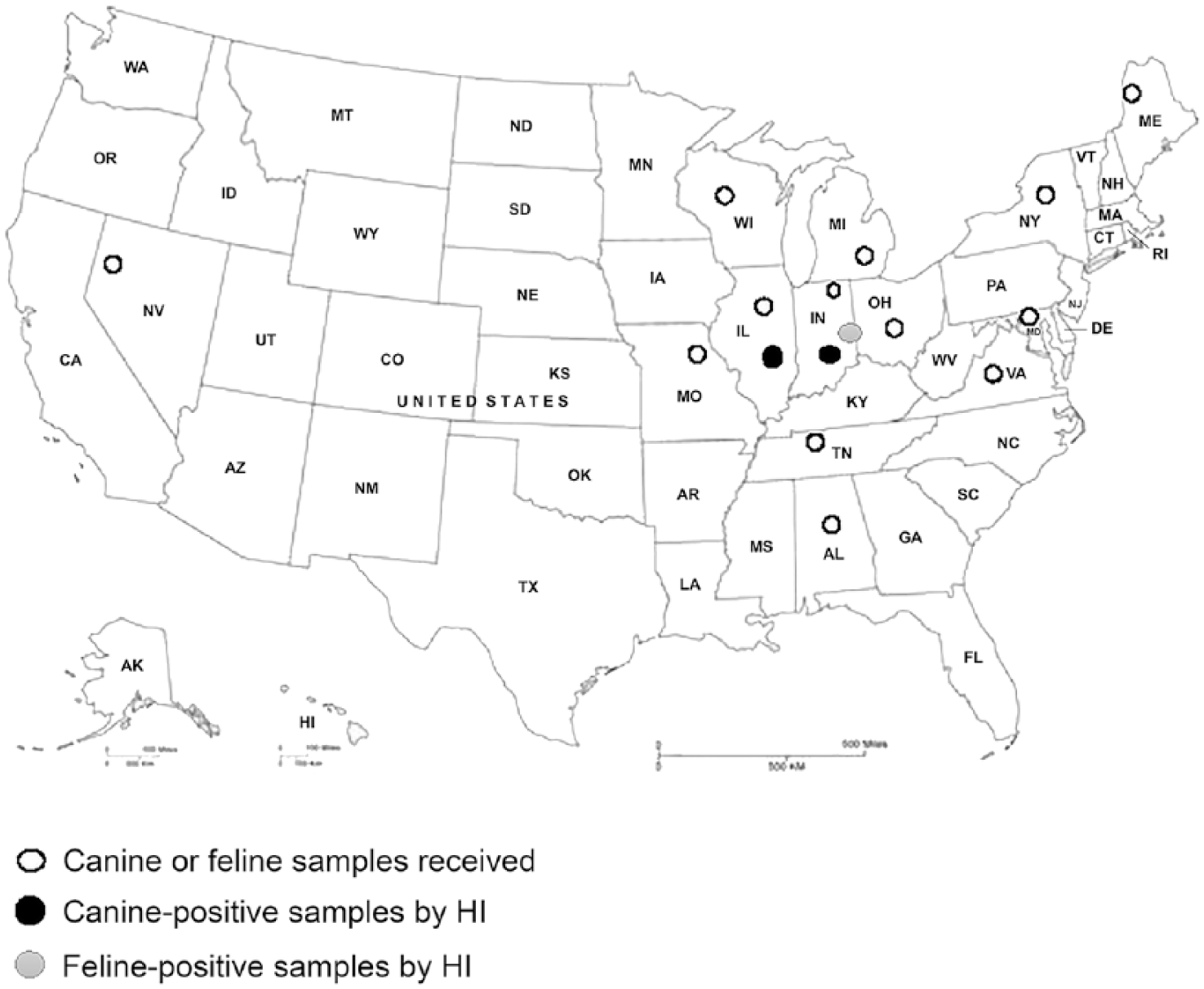
Four canine sera were HI-positive and ELISA-negative, 6 canine sera were ELISA-positive and HI-positive, and 10 canine sera were ELISA-positive and HI-negative. HI-positive samples were obtained from dogs residing in Illinois and Indiana during May, June, and August 2015; seropositivity rates for those months were 1.1% (95% CI: 0.36–2.56%), 0.88% (95% CI: 0.24–2.25%), and 0.22% (95% CI: 0.01–1.22%), respectively (Fig. 3). HI-negative dogs were from many states across the United States (Fig. 2). Overall, 10 canine serum samples (2.21%; 95% CI: 1.05–3.98%) of the 453 tested were positive for H3N2 on HI testing. Sixteen canine serum samples (3.53%; 95% CI: 2.01–5.61%) were positive for IAV by ELISA. ELISA-positive dogs were from Illinois, Indiana, Maryland, and New York. ELISA-negative dogs came from Alabama, California, Iowa, Illinois, Indiana, Maine, Michigan, Missouri, New York, Ohio, Tennessee, Virginia, and Wisconsin (Table 1). There were no significant associations between seroprevalence and age, breed, location of residence, or recent travel to the Chicago area.
The distribution of seropositivity rates (%) by ELISA and HI for canine samples by month (Apr–Aug 2015). These ELISA-positive samples were collected in May–August 2015, giving seropositivity values of 1.77%, 0.88%, 0.44%, and 0.44%, respectively. The HI-positive samples were collected in May, June, and August 2015, giving seropositivity rates of 1.10%, 0.88%, and 0.22%, respectively.
We found that 2.21% of canine sera were positive for CIV H3N2 and 3.53% for IAV. For feline sera, 8.96% were positive for CIV H3N2 and 1.49% for IAV. The results of HI testing and ELISA were not in total agreement, given that 11 (2%) animals were positive on ELISA but not on HI testing. All animals positive on HI testing were also positive on ELISA. A sample can be ELISA-positive but HI-negative because the ELISA detects IgG antibodies against any IAV strain, 4 not only H3N2. 6 A sample could be ELISA-negative but HI-positive because HI detects early-responding IgM as well as IgG antibodies, whereas ELISA primarily captures the later-responding IgG antibody; thus, IgM normally developed during earlier stages of infection is not detectable by ELISA, creating the potential for false-negatives. 10 This result could also be the consequence of nonspecific inhibition, which commonly yields false-positives in HI testing.5,7
In 2007, the seroprevalence of CIV H3N2 was found to be ~0.48% in Korean dogs. 6 In 2010, 34.8% of tested dogs in South Korea were seropositive for CIV H3N2; none of the tested cats were seropositive.8,9 Overall, most CIV testing in Korea showed seropositivity rates of ~5% in dogs. 2 Feline samples from northern China collected during 2010–2014 were analyzed and determined to have a CIV H3N2 strain seroprevalence rate of 0.7%. 9
Our results show that a low proportion of animals tested were infected by CIV H3N2, suggesting that the virus was spreading at a low rate in the population. No specific correlation to any factors such as age, breed, location of residence, or recent travel to the Chicago area that contribute to infection in these species or spreading of the virus was noted. The virus continues to circulate in the United States and Canada. (https://www.avma.org/KB/Resources/Reference/Pages/Canine-Influenza-Backgrounder.aspx; https://www.wormsandgermsblog.com/2018/10/articles/animals/dogs/canine-influenza-update-ontario-oct-25-2018/).6,9 Practitioners should continue to consider CIV in a differential diagnosis for animals with clinical signs and histories that are consistent with CIV exposure and illness, as well as take appropriate precautions to prevent transmission of this contagious pathogen among animals in their care. ELISA and HI tests, in addition to PCR testing, can be useful in establishing a diagnosis and documenting evidence of exposure.
Footnotes
Acknowledgements
We thank Brenda Turner, Donna Schrader, and Ariel Werner from the Indiana ADDL for assisting with ELISA testing, procurement of turkey red blood cells, and virus propagation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received internal financial support for the research, authorship, and/or publication of this article.
