Abstract
Lassa virus (LASV) infection causes an acute, multisystemic viral hemorrhagic fever that annually infects an estimated 100 000 to 300 000 persons in West Africa. This pathogenesis study evaluated the temporal progression of disease in guinea pigs following aerosol and subcutaneous inoculation of the Josiah strain of LASV as well as the usefulness of Strain 13 guinea pigs as an animal model for Lassa fever. After experimental infection, guinea pigs (Cavia porcellus; n = 67) were serially sampled to evaluate the temporal progression of infection, gross and histologic lesions, and serum chemistry and hematologic changes. Guinea pigs developed viremia on day 5 to 6 postexposure (PE), with clinical signs appearing by day 7 to 8 PE. Complete blood counts revealed lymphopenia and thrombocytopenia. Gross pathologic findings included skin lesions and congested lungs. Histologic lesions consisted of cortical lymphoid depletion by day 6 to 7 PE with lymphohistiocytic interstitial pneumonia at 7 to 8 days PE. Scattered hepatocellular degeneration and cell death were also noted in the liver and, to a lesser extent, in other tissues including the haired skin, lung, heart, adrenal gland, lymph nodes, thymus, and spleen. The first cell types to demonstrate staining for viral antigen were fibroblastic reticular cells and macrophages/dendritic cells in the lymph nodes on day 5 to 6 PE. This study demonstrates similarities between Lassa viral disease in human infections and experimental guinea pig infection. These shared pathologic characteristics support the utility of guinea pigs as an additional animal model for vaccine and therapeutic development under the Food and Drug Administration’s Animal Rule.
Lassa virus (LASV) infection causes an acute, multisystemic viral hemorrhagic fever that annually infects an estimated 100 000 to 300 000 persons in West Africa. 10,13,23,24,26,29,45 Approximately 5000 deaths from Lassa fever are recorded each year, and many who survive infection have partial or complete hearing loss. 13,24,26,31,42,44 Lassa fever is of particular concern and importance because of the large number of cases reported each year, the limited understanding of its pathogenesis, and the lack of a Food and Drug Administration (FDA)–approved vaccine. Medical reporting in Lassa fever–endemic regions is intermittent, so actual case numbers are hard to determine. 3,45 In parts of West Africa, 10% to 16% of hospital admissions are due to Lassa fever, and such a prevalent infectious disease causes disruptions at all levels of society. 3,10,32,38,45
Lassa fever was first identified in 1969 after 2 missionary nurses serving the community of Lassa, Nigeria, died from a previously unrecognized viral hemorrhagic fever. 3,10,45 LASV is a member of the Arenaviridae family, which are small, enveloped viruses with single-stranded RNA that are bisegmented with an L (large) segment and S (small) segment. 3,45 The reservoir is the multimammate rat, Mastomys natalensis, a ubiquitous rodent in many rural communities in West Africa. Infection occurs following ingestion, inhalation, or direct inoculation of the virus into mucous membranes or open wounds. 3,38,45 Transmission via close human contact and nosocomial transmission have also been reported. 19,34,35
The incubation period for LASV infection in humans is generally about 7 days but can vary from 3 to 21 days. 3,32,45 Clinical signs and symptoms vary widely between individuals, and most infections result in a nonclinical, self-limiting disease. In those that progress to clinical illness, initial signs include fever, lethargy, anorexia, retrosternal pain, myalgia, a sore throat, lumbosacral pain, and dizziness. 2,3,32 A maculopapular rash is frequently noted, and nonpruritic, nonexudative conjunctivitis is also common. 3,38 Patients often have pronounced pharyngitis that can be mistaken for an upper respiratory streptococcal infection. 3,45 Within the first few days of illness, coughing is also common. Gastrointestinal symptoms occur early during the clinical course and may include 1 or more of the following: nausea, vomiting, diarrhea, epigastric discomfort, and intense abdominal pain, which is often misdiagnosed as acute appendicitis. 3,45 With disease progression, increased vascular permeability results in decreased blood pressure and development of edema, especially in the facial region. In terminal cases, proteinuria, shock, and bleeding from the gums or phlebotomy sites are common sequelae. In those who do not die acutely, neurologic symptoms may arise weeks to months later, manifesting as confusion, tremors, ataxia, and seizures. 3,45 Severe cases progress to a comatose state or death. 3,11 One of the most striking sequelae of this disease is hearing loss. Up to one-third of clinically ill individuals develop partial or complete deafness, with no discernable correlation between severity of other disease symptoms and the occurrence of hearing loss. 12,45 Another unfortunate aspect of Lassa fever is its lethality among women in their third trimester of pregnancy; up to 95% of women at this stage of pregnancy abort and/or succumb to the disease. 10
Shortly after the onset of symptoms, clinical laboratory examination often reveals increases in serum levels of aspartate aminotransferase, alanine aminotransferase, and amylase as well as disturbances in electrolyte concentrations with varying levels of leukopenia with lymphocytopenia, thrombocytopenia, hemoconcentration, and proteinuria. 3,40 Overt hemorrhage is seen in less than 20% of cases, and disseminated intravascular coagulation occurs rarely if ever; loss of platelets and marked hemoconcentration are often more prominent features. 3
Guinea pigs are a commonly used rodent model for LASV. Outbred Hartley guinea pigs, when challenged with LASV Josiah strain, do not show uniform lethality, making them a less attractive animal model when compared with the inbred Strain 13 guinea pig. 27 It should be noted that a guinea pig–adapted strain of LASV-Josiah (GPA-LASV) has been used in Hartley guinea pigs for testing the antiviral drug favipiravir, and inoculation of the GPA-LASV strain resulted in 100% lethality in control guinea pigs in this study. 39
We used Strain 13 guinea pigs in an attempt to further characterize LASV-Josiah in a model that does not require species-specific viral adaptation. Strain 13 guinea pigs have also been used as a small-animal model for studying both New World (Junin and Machupo) and Old World (Lassa) arenaviruses, and we wanted to further evaluate this animal model. 27 Strain 13 guinea pigs have been used at this institute to test the antiviral utility of ST-193, a small-molecule inhibitor, and ribavirin, a guanosine analog, which have shown efficacy when given early during the course of disease. 9 Vaccine testing, including a DNA vaccine, has also been evaluated at this institute using Strain 13 guinea pigs. 8 Because this model has been so often used for both vaccine and therapeutic testing, our goal was to further characterize this animal model by describing the pathologic changes in Strain 13 guinea pigs during the course of disease following aerosol and subcutaneous exposure to LASV.
Materials and Methods
LASV Exposure and Sampling Scheme
Forty Strain 13 guinea pigs weighing between 350 and 600 g were aerosol exposed to a target dose of 1000 pfu LASV, Josiah strain. Guyton’s formula was used to determine minute volumes for guinea pigs. Plaque assays were performed on the virus-containing dilutions collected from the all-glass impingers after each aerosol exposure run. Actual doses received for guinea pigs in each aerosol run were then calculated as previously described. 21 (See Supplemental Table S1 for actual inhaled doses.) Strain 13 guinea pigs were sourced from an in-house colony maintained at the institute. Animals were randomized by a statistician into sampling groups by weight and sex. Groups of 4 guinea pigs were placed in wire mesh baskets inside the aerosol chamber and exposed to aerosolized virus for approximately 15 minutes. Plaque assays were performed on the starting concentrations and samples retrieved from the all-glass impinger for each exposure group.
After exposure, blood samples and tissues were collected from 5 guinea pigs per sampling time point. Sampling time points were scheduled at days 0, 3, 6, 8, 10, 12, 14, and 16 postexposure (PE). Complete blood counts (CBCs) and blood chemistry analyses were performed on samples collected from each guinea pig at each sampling time point. Day 0, unchallenged guinea pigs served as negative controls/baseline data for CBC and chemistry data. Guinea pigs were observed, weighed, and scanned for body temperature daily to record disease progression. Guinea pigs were implanted with IPTT-300 transponder chips (Bio Medic Data Systems Inc, Seaford, DE), which provided unique identification numbers for each animal and also provided body temperature data. Guinea pigs were scanned using BMDS data acquisition systems chip reader, and temperatures were recorded each day. A fever was defined by a temperature at or above 39.5°C.
A second study was done to determine if the course of disease and lesions were the same if Strain 13 guinea pigs were challenged subcutaneously. To address this question, 27 guinea pigs were challenged with 1000 pfu of LASV subcutaneously on day 0 and then were euthanized and necropsied on days 0, 2, 5, 7, 8, 10, 12, 14, and 15. Two additional guinea pigs that had no exposure to LASV were sacrificed at each time point to serve as controls. As with the aerosol group, complete blood counts and blood chemistry analyses were performed on samples collected from each guinea pig at each sampling time point and day 0, unchallenged guinea pigs served as negative controls/baseline data for CBC and chemistry data. Guinea pigs were observed, weighed, and scanned for body temperature daily to record disease progression.
Research was conducted under a USAMRIID Institutional Animal Care and Use Committee approved protocol in compliance with the Animal Welfare Act, Public Health Service Policy, and other federal statutes and regulations relating to animals and experiments involving animals. The facility where this research was conducted is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, International and adheres to principles stated in the Guide for the Care and Use of Laboratory Animals, National Research Council, 2011.
Virus Isolation, Preparation, Plaque Assay
Plaque assays of serum collected at each time point were performed for the aerosol study to generate viremia data. For a detailed description on plaque assay generation, see supplemental materials under the heading “Virus Isolation, Preparation, and Plaque Assay.”
Clinical Pathology
CBCs and serum chemistry were performed at the time of sacrifice for all guinea pigs. CBCs were performed using blood on a HemaVet 950 FS Hematology Analyzer (Drew Scientific, Oxford, CT) in accordance with manufacturer’s guidelines. Serum chemistry analysis was obtained from blood samples collected in sterile red-topped tubes. Serum chemistry analysis was performed on serum obtained from centrifugation at 1800g. An aliquot of 100 μL was analyzed for glucose, creatinine, blood urea nitrogen, calcium, albumin, total protein, alanine aminotransferase (ALT), alkaline phosphatase (ALP), total bilirubin, gamma-glutamyl transferase, and amylase. This particular panel did not include aspartate aminotransferase (AST). Results were compared with day 0, uninfected guinea pig control samples used as a baseline. Five randomly assigned samples were collected from each time point immediately prior to euthanasia.
Histopathology and Immunohistochemistry
Necropsies were performed on each guinea pig immediately following euthanasia in the USAMRIID biosafety level 4 laboratory. Tissues from all major organ systems were collected for both groups, immersion fixed in 10% neutral buffered formalin, and held in biocontainment for a minimum of 21 days. Histopathology samples were routinely processed, embedded in paraffin, sectioned, and stained with hematoxylin and eosin.
Lassa virus (IHC) was performed on replicate tissue sections for all guinea pigs. A detailed description of slide preparation and processing is given in the supplemental materials under the heading “Immunohistochemistry.”
Statistical Analysis
Student t test was used to compare day 0 (prechallenge) versus postchallenge samples. Statistical significance was assumed if P < .05. The complete statistical analysis and the individual animal data are provided as Supplemental Tables S6 and S7.
Results
Clinical Presentation of LASV Infection
In both the subcutaneous and aerosol-exposed groups, weight loss was apparent around day 8 to 10, with a general decrease in weight until the termination of the study (Fig. 1a). Both aerosol and subcutaneous exposure groups became febrile at approximately 8 days PE and remained so until the conclusion of the study (Fig. 1b). Serum viremia as measured by plaque assay was first detected in the range of 1 × 102 pfu/mL in guinea pigs exposed by the subcutaneous route on day 5 PE and the aerosol route on day 6 PE (Fig. 1c). Serum viremia increased after day 6 PE, reaching a peak in the range of 1 × 104 pfu/mL approximately day 10 PE, then maintaining this level until the end of the study (Fig. 1c). Petechial rashes were evident in most of the guinea pigs beginning at 12 days PE. A feature seen with aerosol exposure but not subcutaneous exposure was the development of conjunctival injection in a large percentage of guinea pigs; this finding was most likely related to the route of exposure.
Clinical Pathology
CBC analysis (Supplemental Tables S2 and S3) revealed guinea pigs in the subcutaneous group had a significant (P < .05) decrease in platelets on days 2 to 12 with a return to control levels on day 14. Those in the aerosol group did not have a significant decline, but there was a trend of decreasing platelet counts on days 10, 12, and 14. The mean platelet volume was significantly (P < .05) increased in both groups on days 12 and 14 as well as day 15 in the subcutaneous group and day 16 in the aerosol group; this is consistent with a regenerative platelet response. For hematology and chemistry values collected during this study, please see the attached supplemental materials (Supplemental Tables S2, S3, S4, S5).
A significant (P < .05) decrease in blood lymphocytes was noted in the subcutaneous group starting on day 2 and continuing through day 15; there was not a significant change in the aerosol group, although all time points trended lower in comparison with day 0 baseline values. Heterophils, the guinea pig analogue of neutrophils, were significantly (P < .05) increased in both groups on day 8 PE.
Clinical chemistry analysis (Supplemental Tables S4 and S5) revealed changes in multiple parameters involving serum enzymes, proteins, and electrolytes. On day 16 PE, ALT was increased in both groups, but the increase was not statistically significant. ALP levels showed no clear trends in either group. In both groups, there was a significant (P < .05) increase in globulins on days 12, 14, and 16 in the aerosol group and days 12, 14, and 15 in the subcutaneous group. A significant decrease in albumin was also noted in both groups on each of these days. Total protein levels were not significantly changed. There was a significant (P < .05) increase in serum urea on day 15 PE in the subcutaneous group. In several samples, there was evidence of both hemolysis and lipemia, which can make interpreting chemistry analytes difficult.
Gross Pathology
In general, gross lesions directly attributable to LASV challenge were mild and limited to the skin, lung, and lymph nodes. Gross lesions involving the skin were first noted in the day 12 PE guinea pigs and consisted of red, erythematous foci in the inguinal region. One of the day 14 PE guinea pigs also presented with multiple (i.e., 4–5) crusty, slightly scaly, red, raised lesions in the inguinal area that were approximately 2 to 4 mm in diameter. One of the day 15 PE guinea pigs had multiple foci of crusty and scaling skin on the ventral abdominal wall and inguinal region with slight hyperemia of the affected skin. Consolidation and firmness of the lung lobes became grossly apparent only on day 14 PE in several guinea pigs of both groups and remained until the end of the study. Grossly, the mesenteric lymph nodes were enlarged in 4 of 12 of the day 10 to 15 PE guinea pigs. No significant gross lesions were identified in the spleen, brain, thymus, liver, heart, kidney, adrenal glands, pancreas, gastrointestinal tract, or reproductive organs.
Histology and IHC
Histologic changes were generally mild and similar in nature and severity in guinea pigs challenged either subcutaneously or via aerosol. The location of antigen and temporal progression of lesions was similar in both groups of guinea pigs in this study, suggesting route of infection does not alter pathogenesis of LASV infection. Noticeable lesions first appeared on day 6 PE of the aerosol challenge group and day 7 PE of the subcutaneous challenge group. Lesions consisted of minimal to mild mononuclear inflammation and cell death in various tissues, as well as lymphocytolysis and lymphoid depletion in lymphoid tissues. Heterophilic tracheitis and laryngitis (in the aerosol-challenged group only), adrenal cortical necrosis, hepatitis, hepatic cell death, and individual cell death in hair follicle epithelium were noted as the disease progressed and were often associated with the presence of antigen via IHC. The organ systems with the most prominent histologic lesions will be discussed below. Differences between aerosol and subcutaneous challenge groups in the temporal development of particular lesions and presence of LASV antigen were, with rare exception, attributed to the difference in scheduled time points for these two serial sacrifices (see the Materials and Methods section); they do not likely represent real differences in pathogenesis of these 2 challenge routes. As such, unless otherwise noted, the findings below apply to both challenge groups, and time points listed represent a range that best fits lesion development in both groups. Lesions were attributed to LASV infection if IHC positivity was noted within or around histologic lesions.
Lungs
Interstitial pneumonia characterized by lymphohistiocytic inflammation resulting in mild thickening of the alveolar septa was first recognized in the day 8 PE guinea pigs and continued to increase in severity throughout the study (Figs. 2 and 3a,b). Additional histologic features of the interstitial pneumonia that also increased with severity over time included increased numbers of mononuclear cells, individually necrotic and apoptotic cells, congestion, hemorrhage, and interstitial edema. The mononuclear cells were interpreted as either alveolar and/or interstitial macrophages, which often formed clusters as the disease progressed and were also found around airways and blood vessels.
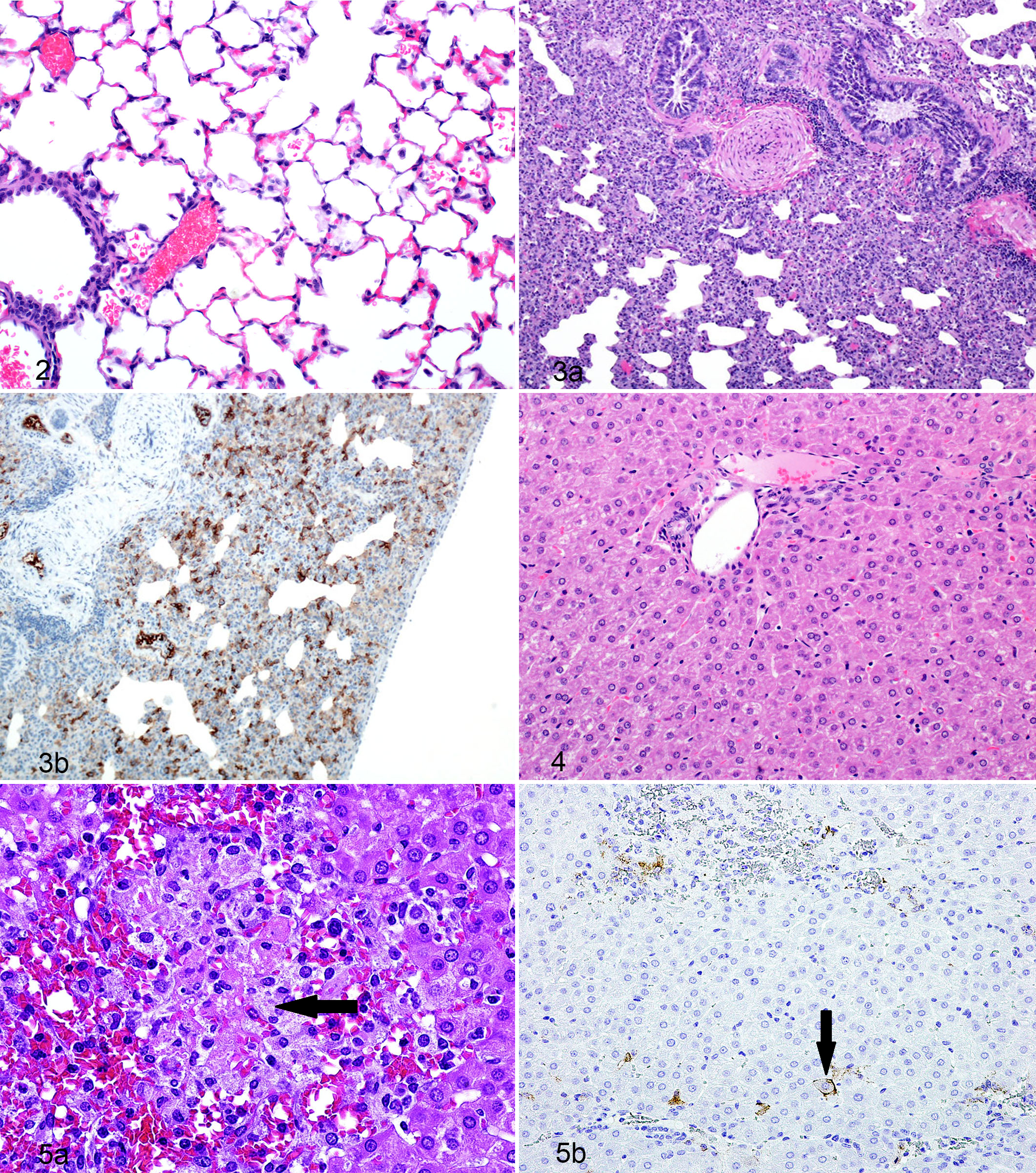
IHC of the lung sections revealed mononuclear cell cytoplasmic immunoreactivity first identifiable on day 6 PE in the aerosol challenge group and on day 7 PE in the subcutaneous challenge group, with a steady increase in both the numbers and intensity of immunohistochemical staining throughout the remainder of the study. Starting as early as day 8 PE, pneumocytes lining alveoli were often positive for viral antigen; by day 12 to 14 PE, depending on the individual guinea pig, positive viral antigen staining was also noted in the respiratory epithelium of larger airways.
Liver
Histologic lesions in the liver of both aerosol and subcutaneous challenge groups were minor and limited to scattered individual cell death (Figs. 4 and 5a,b) with or without mononuclear inflammation with portal and sinusoidal inflammation and crisp-edged vacuolar degeneration. Portal and sinusoidal inflammation was lymphoplasmacytic (nonsuppurative) and ranged from minimal to moderate. The individual cell death and inflammation were graded as minimal on day 7 PE and progressively increased to moderate by days 10 and 12 PE and remained fairly consistent thereafter. Vacuolar degeneration was first noted in the day 12 PE guinea pigs and peaked on days 14 to 16 PE, with a severity ranging from mild to moderate.
By day 7 PE, there was scant, multifocal, membrane-associated hepatocellular immunoreactivity throughout the liver with abundant membrane-associated immunoreactivity in mononuclear cells located at the periphery of necrotic foci. Day 8 PE immunoreactivity was similar with rare and faint intrasinusoidal mononuclear cell cytoplasmic- and membrane-associated or sinusoidal wall immunoreactivity. Throughout the remainder of the study, the LASV immunoreactivity (Fig. 5b) remained multifocal and associated with sinusoidal membranes, sinusoidal mononuclear cells, and scattered hepatocytes.
Heart
Beginning on day 8 PE in the subcutaneous challenge group, there were minute, multifocal foci of histiocytic and lymphoplasmacytic inflammation in the endocardium. By day 10 PE, guinea pigs of both aerosol and subcutaneous challenge groups had similar but slightly more distinct lesions. The inflammation began as minimal and became moderate by the end of the study (Figs. 6 and 7a,b). The inflammation tended to be most intense along the subendocardial connective tissue, with only a few cells and clusters of cells extending deeper into the myocardium.
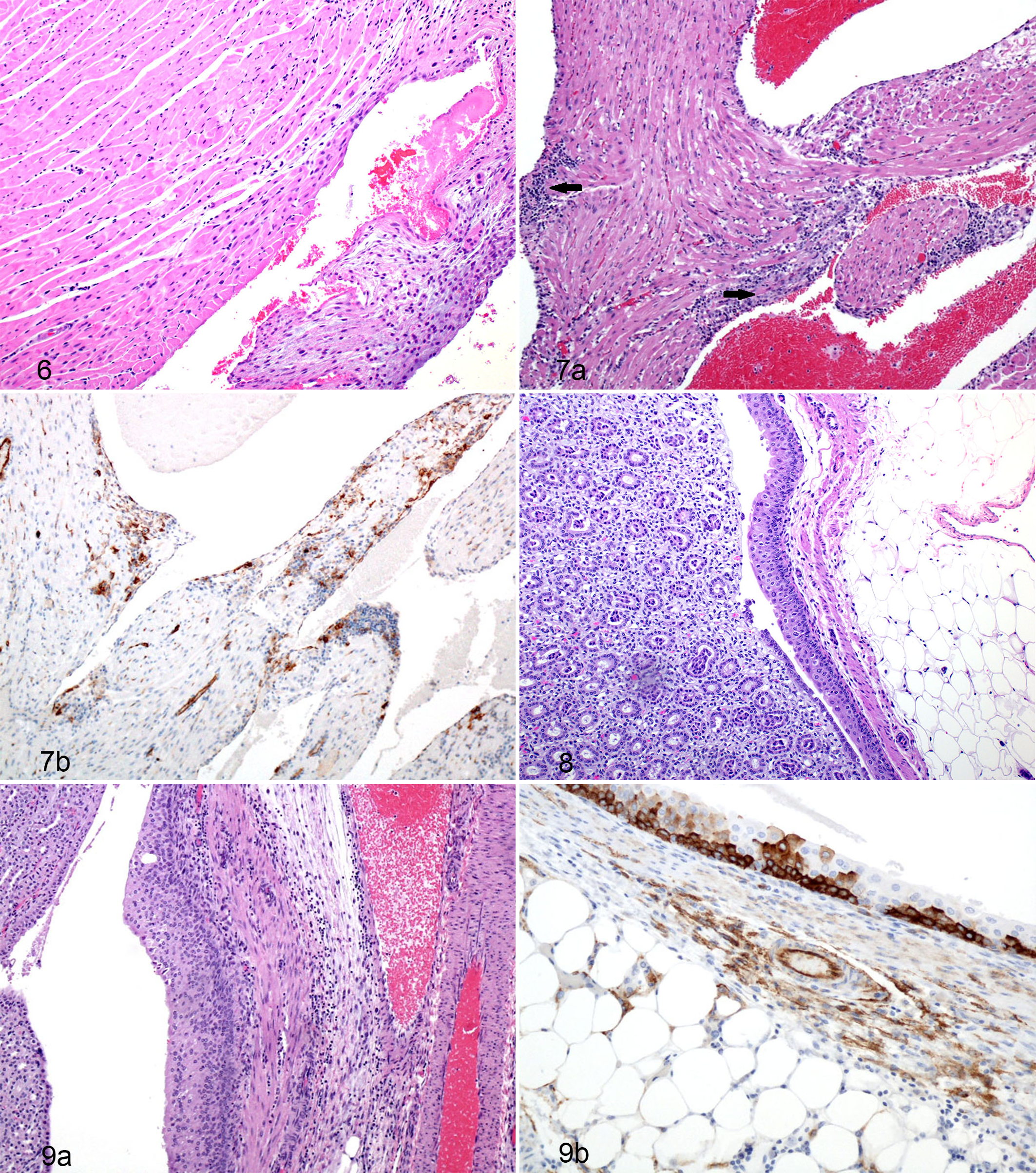
Immunohistochemically, there was cytoplasmic immunoreactivity in rare, scattered mononuclear cells in the endocardium on day 10 PE, which progressively increased in number and frequency through day 16 PE.
Kidneys
Varying degrees of nephrosclerosis and pelvic inflammation were present in the kidneys of each group of guinea pigs including the noninfected controls. There was an increase in the amount of perivascular, interstitial, and pelvic inflammation noted on day 7 PE, which peaked at day 14 PE and continued through day 16 PE (Figs. 8 and 9a,b). Also, in the day 15 PE guinea pigs, there was mild urothelial hyperplasia within the lining of the pelvis overlaying areas of mild inflammation. Immunohistochemistry revealed that day 10 and 12 PE, kidneys also demonstrated scant spindle cell cytoplasmic- and membrane-associated immunoreactivity in small foci of interstitial fibrosis. Finally, on day 15 PE, 2 of the 3 guinea pigs demonstrated strong cytoplasmic immunoreactivity in the basal layer of the uroepithelium (Fig. 9b) lining the renal pelvis as well as extending from the basal layer through the mucosa.
Adrenal Glands
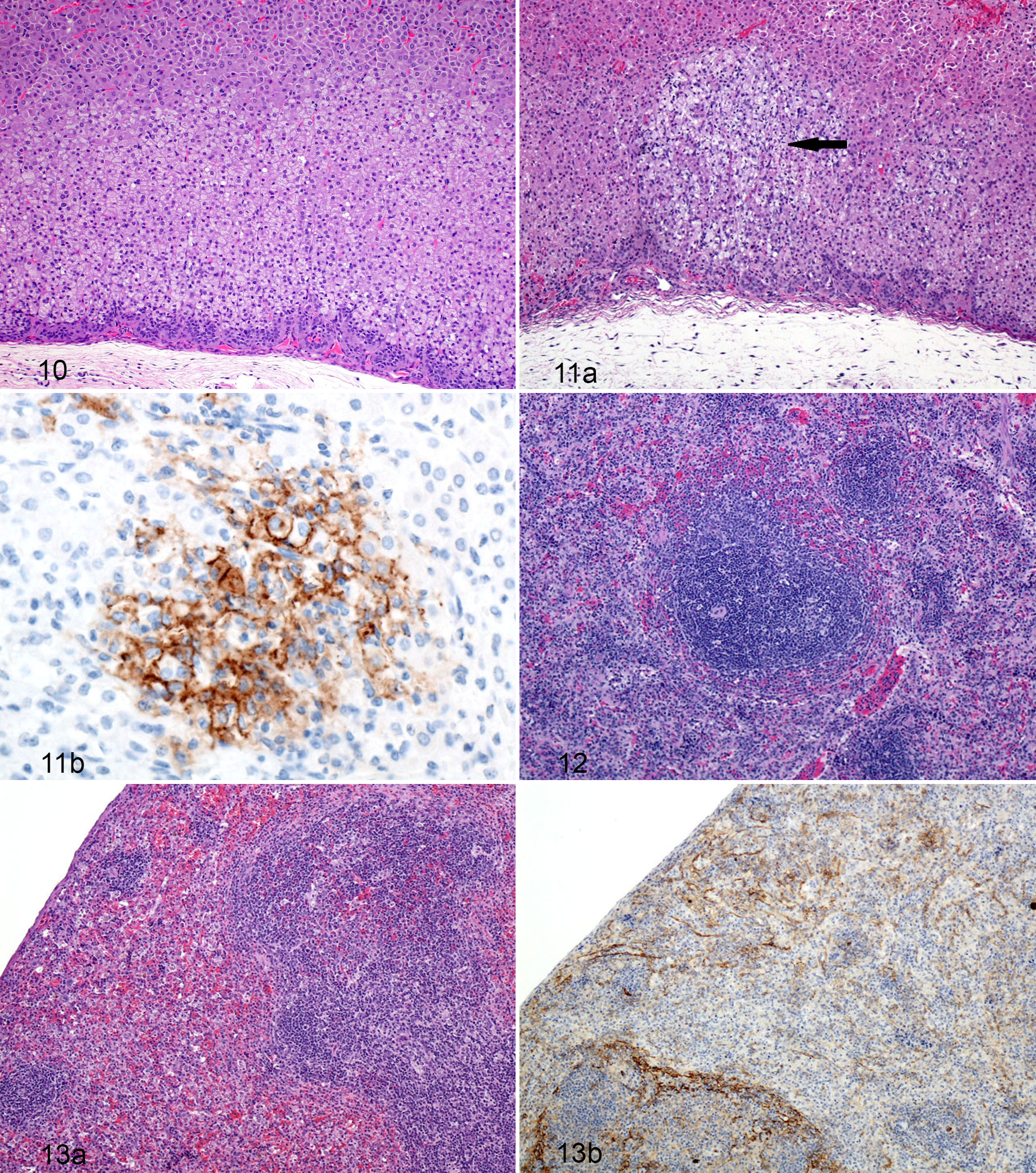
Most guinea pigs of both challenge groups exhibited rare, scattered single cell death in the adrenal cortex. Multifocally, individual cortical cells exhibited weak cytoplasmic immunoreactivity, and rare small clusters of cells exhibited both cytoplasmic- and membrane-associated immunoreactivity. Within the cortex of a day 8 PE guinea pig, there were multiple, well-circumscribed foci of vacuolar degeneration with scant apoptotic/necrotic cellular debris scattered in the interstitium. Interestingly, these foci were immunoreactive for LASV antigen. Only a few guinea pigs had distinct foci of numerous cells undergoing cell death (Figs. 10 and 11a,b). Throughout the remainder of the study, there was either minimal or a complete lack of histologic lesions within the adrenal glands; however, there was slightly increasing immunoreactivity within cortical cells and interstitial mononuclear cells.
Spleen
Histologic findings in the spleen were characterized by a mild heterophilic splenitis occurring on days 7 and 8 PE followed by minimal to mild lymphocytolysis and lymphoid depletion of the white pulp with perifollicular to diffuse proliferation of mononuclear cells occurring throughout the red pulp in challenged guinea pigs on days 10 through 15 PE (Figs. 12 and 13a,b). The diagnosis of heterophilic splenitis was based on the presence throughout the red pulp of increased numbers of heterophils; however, this diagnosis can be subjective because increased numbers of circulating heterophils (ie, blood heterophilia) can make the red pulp of the spleen appear inflamed, which was supported by the increases in heterophils on the CBCs.
The cellular proliferation in the red pulp was characterized by a nearly monomorphic population of blastlike mononuclear cells with a brisk mitotic rate; large, vesiculate nuclei; and moderate amounts of eosinophilic cytoplasm. These cells first appear at the periphery of the lymphoid follicles in the day 10 PE guinea pigs and obscure the line of distinction between the follicle and the red pulp. Throughout the remainder of the study, the proliferation of cells becomes more evident, as it was present throughout a larger percentage of the red pulp. This proliferation of cells likely represents extramedullary hematopoiesis.
Lymph Nodes
The first cells to demonstrate unequivocal viral antigen via IHC were the lymphoid fibroblastic reticular cells of the lymph nodes as well as macrophages/dendritic cells within lymph nodes and lung on day 5 or 6 PE in the subcutaneous and aerosol groups, respectively (Fig. 14a,b) To further confirm infection of macrophages and dendritic cells early in infection, we used a macrophage/dendritic cell marker, dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN) and stained with an immunofluorescent stain or used an in situ hybridization to corroborate our IHC staining and found LASV-infected macrophages/dendritic cells at the same time points as the first IHC staining of cells within lymph nodes (Figs. 15a,b and 16).
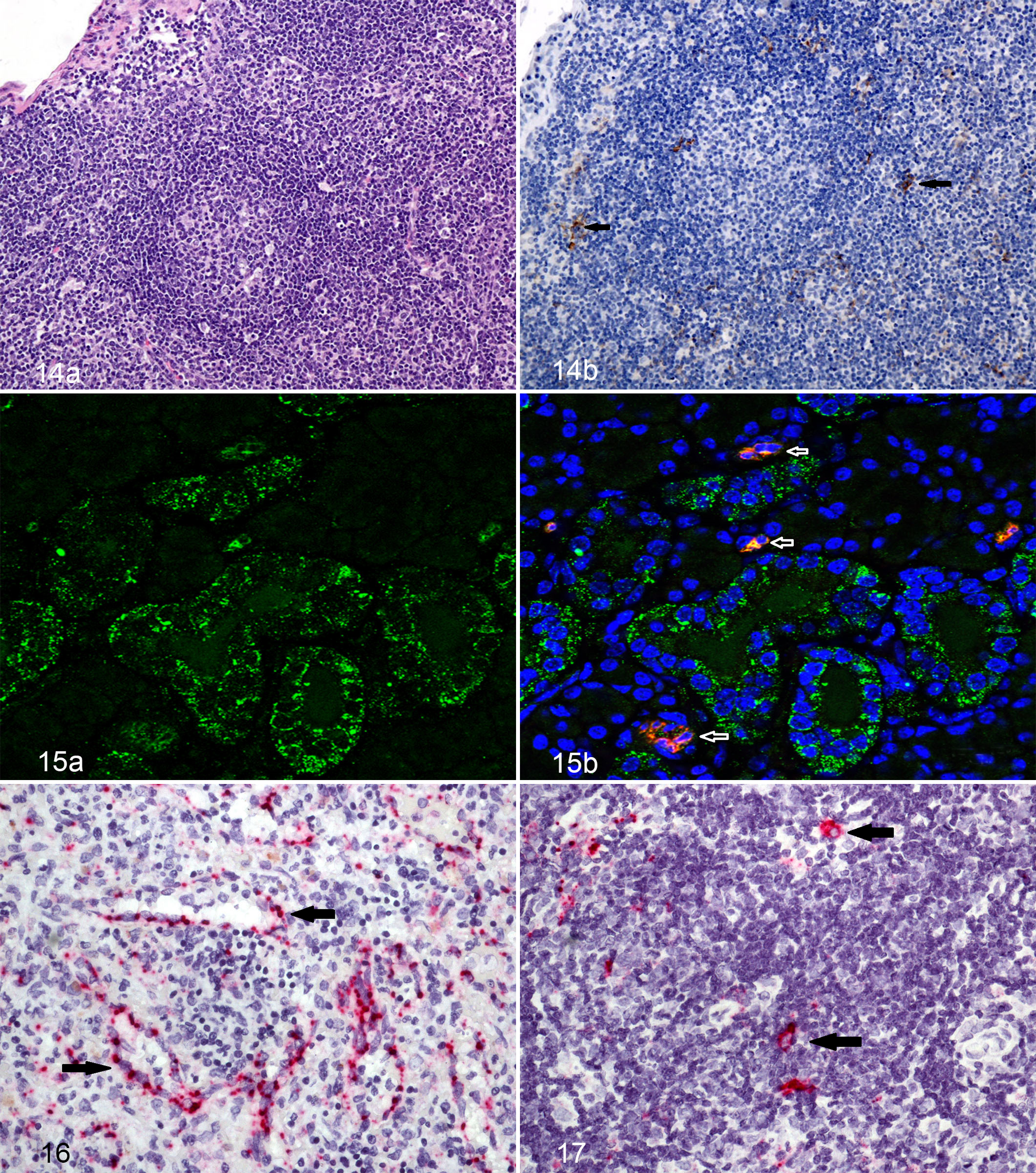
Lassa virus caused death of lymphocytes, which die via a bystander event, as noted by the complete lack of viral antigen staining noted in these cells. This finding has also been noted in other viral hemorrhagic fevers. 4 –6,17,18,22
Histologic findings of the submandibular lymph node and mesenteric lymph node were limited to mild lymphoid depletion characterized by lymphocytolysis and loss of clearly distinct primary and secondary lymphoid follicles. Depletion was first noted in the day 5 to 8 PE guinea pigs and persisted in the challenged guinea pigs through day 16 PE. Mononuclear cell proliferation was seen during later time points, especially in the mesenteric lymph node. Lymphocytolysis was less a feature in later time points than in earlier sampling groups. Immunoreactivity in the follicles of both lymph nodes was first noted in the day 5 and day 6 PE guinea pigs, where it increased in intensity and quantity through day 10 PE and then tapered on days 14 and 15 PE. Multifocally, there was light to moderate mononuclear cell membranous and cytoplasmic immunoreactivity as well as non–cell-associated immunoreactivity in both the medullary cords and sinuses. In addition, immunoreactive mononuclear cells were scattered throughout the cortex. There was no viral antigen observed within lymphocytes.
Because of nonspecific background staining of endothelial cells in multiple tissues noted in control guinea pigs, it was hard to determine via IHC if endothelial cells were infected or were merely staining because of indiscriminate antigen-antibody binding. In an attempt to investigate, in situ hybridization was performed on select samples to determine if viral nucleic acid was present in endothelial cells of infected guinea pigs. Endothelial staining was noted in both the heart and within lymph nodes (Fig. 17). The dearth of inflammation around any vessels noted to be positive for viral antigen or noticeable histopathologic changes in vessels suggests LASV does not cause overt cell death or vasculitis, at least in guinea pigs.
Thymus
Histologic findings in the thymus centered on depletion of lymphocytes within the cortex, which was first evident on day 8 PE and increased through the end of the study. Beginning on day 7 PE and continuing to the end of the study, there were increased numbers of tingible body macrophages (TBM) within the cortex. Lassa virus immunoreactivity within mononuclear cells was rare and weak on day 5 PE but progressively increased in quantity and intensity throughout the study, becoming more abundant in medullary mononuclear cells and within the cytoplasm of TBMs throughout the cortex. Multifocally, there were scattered immunoreactive mononuclear cells and non–cell-associated immunoreactivity located within both the cortex and medulla, epithelial (Hassall’s corpuscles) and lymphoid components. There was also immunopositivity of either dendritic cells or fibroblastic reticular cells, characterized by long, linear cytoplasmic processes that stained for antigen.
Bone Marrow
There were no appreciable histologic changes in the bone marrow in any of the guinea pigs from either challenge group. There was, however, scattered immunopositivity within mononuclear cells (macrophages/dendritic cells) in the bone marrow starting on day 8 PE and continuing to the end of the study. This likely represents circulating inflammatory cells that were infected with or had phagocytized viral antigen, with no apparent pathologic effect on the remaining components of the bone marrow.
Pancreas
Histologic lesions of the pancreas included acinar cell atrophy with loss of zymogen granules, vacuolar change affecting the cytoplasm, and replacement of parenchyma by fibrous connective tissue. The changes ranged from minimal to moderate and were first noted in the day 12 or day 14 PE group and persisted and increased through the day 15 and 16 PE groups.
Skin
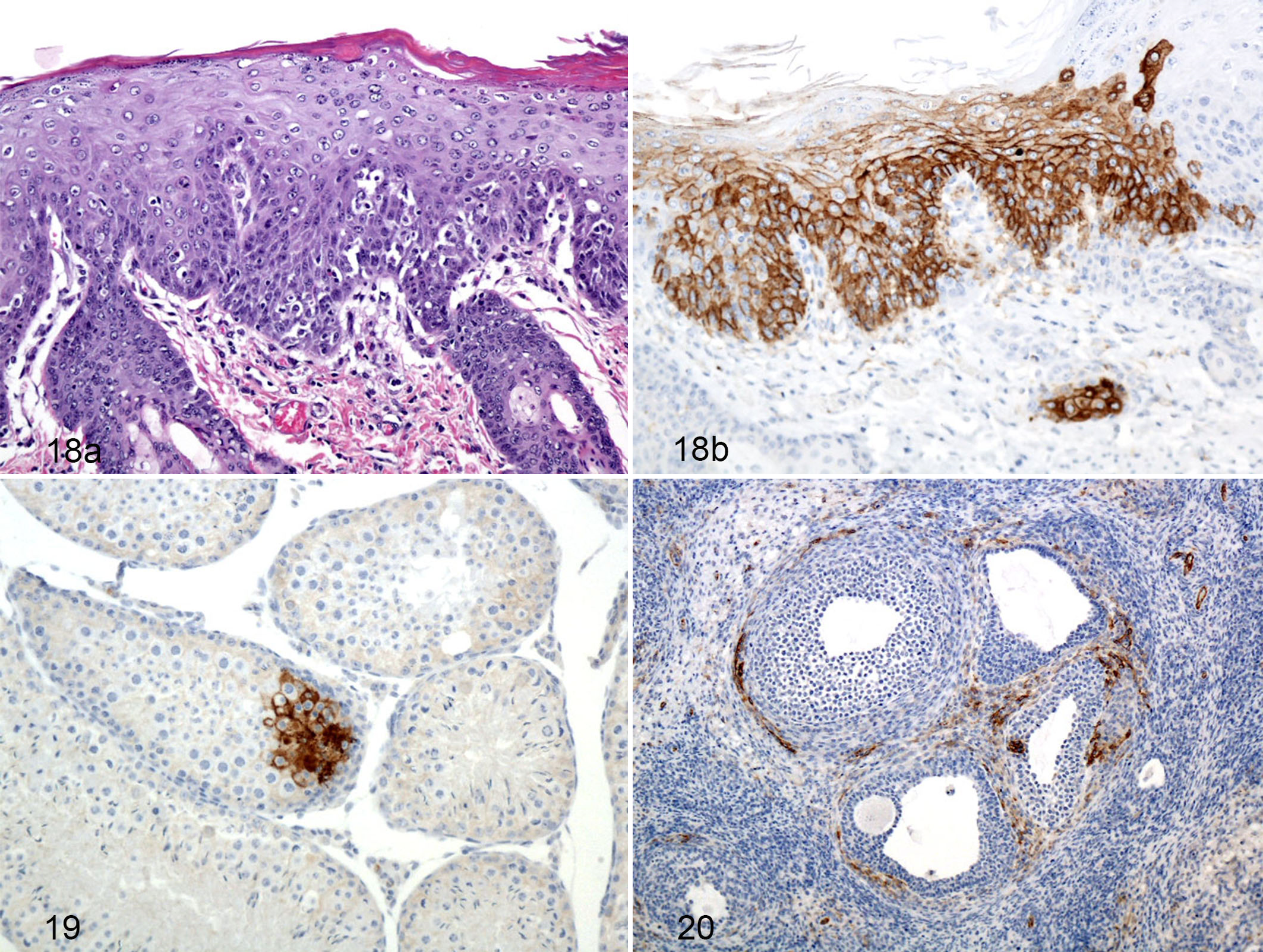
Histologic lesions were first identified in the day 10 PE group with 1 of 3 guinea pigs in the subcutaneous group and 1 of 5 in the aerosol group demonstrating a mild lymphocytic and histiocytic dermatitis with edema. These changes are indicative of hydropic change with single cell death. One of 3 guinea pigs in the day 12 PE subcutaneous group and 2 of 5 guinea pigs in the day 12 PE aerosol group exhibited dermal inflammation and epithelial hyperplasia (Fig. 18a,b). Five guinea pigs in the day 14 PE groups and all five guinea pigs in the day 16 PE group also had this lesion. Most interestingly, the multiple foci of inflammation and epidermal hyperplasia correlated directly to foci of epidermal immunoreactivity first recognized in the day 10 PE group and persisting through the days 12, 14, 15, and 16 of the PE groups. Lassa virus immunoreactivity occurred in clusters of epidermal squamous epithelial cells and was generally more intense in the basal layer with progressively less intensity as the squamous cells become keratinized and more superficial.
Reproductive Tissues
Histologic lesions associated with LASV were generally limited to male guinea pigs with mild epididymitis from the day 10 PE group onward.
Lassa virus antigen-specific IHC revealed that female guinea pigs from the day 7 PE group had small clusters of immunoreactive cells scattered throughout the submucosa of the vagina and cervix. One day 10 PE male guinea pig had a mild lymphocytic epididymitis that also contained immunoreactive mononuclear cells, and 2 of 3 guinea pigs in the subcutaneous group demonstrated cytoplasmic immunoreactivity in either the epithelial lining of the seminal vesicles or the squamous epithelium of the penis. One male day 12 PE guinea pig in the aerosol group had similar lesions and immunoreactivity. One male day 15 PE guinea pig exhibited strong epithelial cell apical cytoplasmic immunoreactivity within the ducts of the epididymis, and another had rare, small clusters of cytoplasmic immunoreactivity within germ cells lining the seminiferous tubules (Fig. 19). The day 14 PE guinea pigs demonstrated cytoplasmic immunoreactivity noted in clusters of parafollicular (thecal cells) and interstitial cells of the ovary as well as the uterine stroma with no substantial histologic lesions (Fig. 20).
Brain
Significant lesions within the brain were rare. Within the subcutaneous challenge group, 2 of 3 guinea pigs on day 10 PE exhibited minimal lymphoplasmacytic perivascular inflammation in the meninges and choroid plexus, and 1 day 16 PE guinea pig had similar inflammation. Immunohistochemistry for LASV antigen was positive in the choroid plexus of this same day 16 aerosol-exposed guinea pig.
Discussion
In this study, LASV was tested in guinea pigs to further elucidate disease-causing mechanisms and the temporal sequence of infection. The goal was to determine if the guinea pig is a suitable animal model for downstream studies under the FDA “Animal Rule” to develop countermeasures against Lassa fever. 41 In short, this study demonstrated that the Josiah strain of Lassa fever in Strain 13 guinea pigs had a similar incubation period, clinical course of disease, clinical pathologic findings, gross lesions, and histologic lesions compared with human and nonhuman primate cases of Lassa fever. * In addition, our findings suggest the route of infection, either through subcutaneous or aerosol inoculation, does not have an appreciable effect on the course of disease.
In general, gross lesions are sparse to absent in humans, 42,44 and guinea pigs in this study showed a similar paucity of gross lesions with the exception of the lung, lymph node, and skin lesions mentioned above. The gross lesions in the haired skin of the guinea pigs in this study directly correlated with dermal inflammation and epithelial hyperplasia. Regardless of challenge route, infected guinea pigs had firm, noncollapsing lungs indicative of interstitial pneumonia, and histologic lesions consisted of lymphohistiocytic interstitial pneumonia with increased number of alveolar macrophages. In humans, gross pathologic changes often include pulmonary edema and pleural effusion, with histologic lesions consisting of interstitial pneumonitis; thus, the guinea pig model corresponds to human disease.
Compared with reports in humans, guinea pigs have similar perturbations in their clinical pathology data following LASV infection. Thrombocytopenia, a common finding in human cases, was present in guinea pigs in our study. 42,45 In addition, human cases of Lassa fever often have elevated levels of liver enzymes in serum consistent with hepatic damage, such as ALT elevations. 3,42,45 Similar elevations were noted in guinea pigs in this study. In guinea pigs, ALT does not have the organ specificity it has in other species, and in this instance, some of this elevation may be due to the cardiac damage seen in several of the guinea pigs in this study. 20 AST was not evaluated in this particular study, and this enzyme should be included in future studies to determine correlations with human AST elevations. Lymphopenia was found in the guinea pigs of this study, 42,45 and lymphoid cell death and loss in lymphoid organs matched the lymphopenia identified on the hematology panels. Lymphopenia is commonly described in human patients infected with LASV as well, corroborating this animal model. The significant decrease in albumin levels of both challenge groups was most likely from vascular leakage. The overall lack of changes in total protein was most likely from the overall increase in globulins, which prevented an overall total protein decrease. This significant increase in globulin levels in both the challenge groups denotes a robust adaptive immune response with ongoing infection.
The most consistent histologic lesions reported in the sparse human literature on LASV pathology are hepatocellular necrosis, adrenal necrosis, splenic necrosis, myocarditis, interstitial pneumonia, and renal tubular injury. 42,44 With the exception of renal tubular injury, LASV-challenged guinea pigs in our study and previous studies by Jahrling et al developed all of these lesions to some extent. 27,43 The primary histologic lesions in our study consisted of degeneration or single cell death in multiple tissues, either alone or in combination with minimal to mild inflammation. The severity of lesions in all guinea pigs in this study was consistently minimal to mild, and this tendency for unspectacular histologic lesions despite this model being uniformly lethal mimics what is seen in fatal LASV cases in both human and nonhuman primates.
Endocardial lesions are noted in infected human patients. 43 Similarly, mild, multifocal, subacute endocarditis appears to be a fairly consistent, relatively specific LASV-associated lesion in guinea pigs with advanced disease (10 or more days PE).
Various epithelial tissues were affected beginning with the haired skin on day 10 PE and progressing to other epithelial cells at later time points to include epithelium of the reproductive organs, gall bladder, thyroid gland, and pancreas. These lesions, with the exception of the lesions in the haired skin, were generally minimal in severity. The only major difference in lesions between the two challenge routes was a mild increase in heterophilic inflammation noted in the upper respiratory tract of aerosol-challenged guinea pigs, most likely a function of the route of exposure.
As in the nonhuman primate model, 23 LASV antigen in guinea pigs followed a similar route of dissemination. Lassa virus appeared first in lymphoid tissues and subsequently spread to permissive parenchymal cells, most notably in the liver and to a lesser extent in the heart, lung, and adrenal gland, followed within a few days by antigen dispersal to the reproductive organs and skin.
Antigen was first associated with areas of hepatitis on day 6 to 8 PE, pneumonia on day 6 to 8 PE, and nephritis on day 10 PE. Lassa virus–specific immunoreactivity in the latter stages of infection in this and other studies is widely distributed. The ubiquitous Lassa viral receptor, alpha-dystroglycan, 3,37 at least partially explains this viral promiscuity. During later time points, antigen was concentrated within perivascular and interstitial spindle cells, mononuclear cells, and epithelial cells in multiple tissues. This suggests that Lassa viral infection of spindle cells occurs later in the disease course. If supporting tissues in and around vessels are affected by LASV, this could lead to systemic inflammation and vascular leakage. Histologic changes in the renal tubules as reported in the human literature were notably absent in the guinea pigs in our study. The reason for this discrepancy is unclear.
The infection of dendritic cells and macrophages as seen in this study is of particular importance because of their sentinel function within the immune system. Infection of these first-line defenders is a common finding in LASV infection in other species 23 and in other viral hemorrhagic fevers, and this often portends immune dysregulation in the host. 1,18,30 Previous in vivo and in vitro studies have shown that infection of dendritic cells by LASV impedes their ability to initiate an appropriate adaptive immune response, 1,23 and dendritic cells of guinea pigs also appear to respond in a similar fashion.
Antigen was also noted within reproductive tissues of both male and female gonads. In humans, transmission via semen has been reported up to 3 months after infection. 38 In humans, presence of viral antigen in semen appears to be effective at prolonging the transmission window for LASV beyond the initial acute stage of infection. Recent evidence has shown this also occurs with Ebola virus. 14 An animal model with corresponding gonadal viral infection may be helpful in future research that aims to study this aspect of the disease.
Significant neurologic lesions were not noted in this study; however, as neurologic pathology is typically identified in more chronic cases in the human literature, 3,11 it is possible that serial sacrifices beyond the day 16 endpoint of this study would be required to allow lesions in the nervous system to develop. This is evidenced by the observation of strong, specific immunoreactivity in the ependymal cells in the cerebrum of 1 guinea pig sacrificed on day 16 PE. Lassa virus in nonhuman primates has shown a neurologic predilection, 23 so further studies with additional time points are warranted to investigate this aspect of disease in guinea pigs.
The findings of this study indicate that LASV in Strain 13 guinea pigs, whether via inhalation or subcutaneous exposure, follows the same disease progression with largely indistinguishable lesions and viral kinetics as noted via IHC. This model also closely approximates Lassa fever in humans, 3,42,45 making it a viable model of infection. The incubation period of Lassa fever in Strain 13 guinea pigs is about 1 week, with hematology and chemistry results showing thrombocytopenia, lymphopenia, and increases in liver enzymes, which are all similar to the human course of disease. 3,42,45 Gross and histologic lesions are also very similar to those seen in Lassa fever patients to include lymphoid, hepatic, cardiac, and pulmonary changes, 42,44 and this supports the utility of the guinea pig as a viable animal model of disease. Future studies using this animal model may well lead to development of therapeutics and vaccines, which will ameliorate the harmful effects this terrible disease has on humankind. 1,4,21
Footnotes
Notes
Acknowledgements
We wish to gratefully acknowledge the numerous contributions of USAMRIID’s pathology personnel including William Aguilar, Neil Davis, Phil Fogle, Angela Grove, Gale Krietz, Christine Mech, Nancy Twenhafel, Taylor Chance, Paul Facemire, Don Nichols, and Brandon Wilke. We would also like to thank members of USAMRIID’s Virology Division, with a special thanks to Heather Esham, for help with animal observations. We would also like to thank the members of the Center for Aerobiological Sciences at USAMRIID for assistance with the aerosol exposures. The Veterinary Medicine Division performed phlebotomy on these studies and was instrumental in planning and performance of the study. A special thanks goes out to Dr Stephen Lockett and Kimberly Peifley at Optical Microscopy and Analysis lab at NCI-Frederick for assistance with confocal imaging. Finally, we would like to acknowledge the Special Pathogens Branch of the Centers for Disease Control and Prevention for providing USAMRIID with the original sample containing the LASV strain.
Author Contributions
T. M. Bell and C. I. Shaia contributed equally to the article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The views, opinions, and/or findings contained herein are those of the authors and should not be construed as an official Department of Army position, policy, or decision unless so designated by other documentation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received no financial support for the research, authorship, and/or publication of this article. The research was performed under USAMRIID project 195743 and was funded by a Defense Threat Reduction Agency, Joint Science and Technology Office (DTRA/JSTO) Translational Medical Technologies (TMT) grant TMTI_0045_09_RD_T.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
