Abstract
Canine perivascular wall tumors (PWTs) are a group of subcutaneous soft tissue sarcomas developing from vascular mural cells. Mural cells are involved in angiogenesis through a complex crosstalk with endothelial cells mediated by several growth factors and their receptors. The evaluation of their expression may have relevance since they may represent a therapeutic target in the control of canine PWTs. The expression of vascular endothelial growth factor (VEGF) and receptors VEGFR-I/II, basic fibroblast growth factor (bFGF) and receptor Flg, platelet-derived growth factor B (PDGFB) and receptor PDGFRβ, transforming growth factor β1 (TGFβ1) and receptors TGFβR-I/II, and cyclooxygenase 2 (COX2) was evaluated on frozen sections of 40 PWTs by immunohistochemistry and semiquantitatively scored to identify their potential role in PWT development. Statistical analysis was performed to analyze possible correlations between Ki67 labeling index and the expression of each molecule. Proteins of the VEGF-, PDGFB-, and bFGF-mediated pathways were highly expressed in 27 (67.5%), 30 (75%), and 19 (47.5%) of 40 PWTs, respectively. Proteins of the TGFβ1- and COX2-mediated pathways were highly expressed in 4 (10%) and 14 (35%) of 40 cases. Statistical analysis identified an association between VEGF and VEGFR-I/II (P = .015 and .003, respectively), bFGF and Flg (P = .038), bFGF and PDGFRβ (P = .003), and between TGFβ1 and COX2 (P = .006). These findings were consistent with the mechanisms that have been reported to play a role in angiogenesis and in tumor development. No association with Ki67 labeling index was found. VEGF-, PDGFB-, and bFGF-mediated pathways seem to have a key role in PWT development and growth. Blockade of tyrosine kinase receptors after surgery could represent a promising therapy with the aim to reduce the PWT relapse rate and prolong the time to relapse.
Keywords
Canine perivascular wall tumors (PWTs), previously grouped under the term hemangiopericytoma, are a group of soft tissue sarcomas (STSs) developing from vascular mural cells (MCs), and they are characterized by variable differentiation, ranging from pericytic to myoid. 5,36 PWTs arise more commonly in the subcutaneous tissue of the extremities and are characterized by a low metastatic potential and an intermediate rate of local recurrence. 4,5,47 The major prognostic factors in canine PWTs are tumor size, completeness of margins, and site and depth of the lesion. 4
MCs, from which PWTs arise, are involved in angiogenesis and blood vessel stabilization through a complex crosstalk with endothelial cells (ECs), a process mediated by several growth factors (GFs), including vascular endothelial GF (VEGF), basic fibroblast GF (bFGF), platelet-derived GF B (PDGFB), and transforming GF β (TGFβ). 2 VEGF, PDGFB, and bFGF have been demonstrated to have a role in the development of several human STSs, including gastrointestinal stromal tumors, uterine leiomyosarcomas, and dermatofibrosarcoma protuberans, 13,32,43 and in canine STSs and feline vaccine-associated sarcoma in cats. 1,16,19,28 Based on in vivo and in vitro studies, these GFs have been regarded as targets for the therapy of STSs in humans 9,12,32 and dogs. 27,31,37 To the best of our knowledge, the involvement of GFs and their receptors in the pathogenesis of PWTs and their potential therapeutic value have not been investigated.
COX2 is an inducible enzyme involved in inflammation that is also responsible for pulmonary MC proliferation in hypoxic conditions, through TGFβ-mediated modulation. 35,45 COX2 expression has been demonstrated in several canine and feline tumors, and its inhibition is considered a promising adjuvant therapy. 6,8,29,30,38,39 –41
The aims of this study are as follows: to assess the expression of several GFs pathways—including VEGF, bFGF, PDGFB, and TGFβ1 and their receptors VEGFR-I (Flt-1), VEGFR-II (Flk-1), PDGFRβ, Flg, TGFβR-I, TGFβR-II, and COX2—to identify a high expression of molecules and their potential role as therapeutic targets; to detect possible associations between GFs and GF receptor expression, similar to what occurs during the crosstalk between MCs and ECs throughout angiogenesis when expression of these proteins may be up- and downregulated (regulatory loops); and to assess the presence of associations between the high expression of GFs and Ki67 labeling index or mitotic index to identify a putative role of GFs in neoplastic cell proliferation.
Materials and Methods
Inclusion Criteria and Case Selection
All tumors included in the study had to fulfill the following criteria: available fresh tissue, origin from the skin and subcutis of dogs, and histopathologic features diagnostic for PWT (perivascular whorls, bundles from tunica media, staghorn vessels, and placentoid growth). 5 Cases expressing GFAP and/or S100 and negative for markers of myoid differentiation (smooth muscle actin, calponin, desmin, smoothelin, and myosin) were excluded.
Tissue Handling
One part of each excised tumor was fixed in 10% neutral buffered formalin and routinely processed for histology, and 1 portion (diameter range of the sample, 5–10 mm) was snap-frozen in isopentane cooled at the freezing temperature in liquid nitrogen.
Histologic Evaluation
Histologic parameters assessed in each tumor included mitotic index, percentage of necrosis, and histologic grade, which were assessed according to the literature. 17
Immunohistochemistry
Immunohistochemistry for Ki67 was performed on formalin-fixed, paraffin-embedded tissues using the clone MIB-1 (Dako, Goldstrup, Denmark; dilution, 1:600) after heat-induced antigen retrieval (pressure cooker, citrate buffer, pH 5.6). Ki67 expression was evaluated as the labeling index and was defined as the percentage of Ki67-positive cells. The count of Ki67-positive cells was performed in 10 high-power fields (400×), with at least 1000 cells counted for each case, using the manual count tool of the Image Pro Plus 6.3 analysis software (Media Cybernetics Inc).
Immunohistochemistry was performed on cryostatic sections to exclude a peripheral nerve sheath tumor, to confirm a diagnosis of PWT, and to subtype canine PWTs, applying antibodies that identify S100, GFAP, vimentin, smooth muscle actin, calponin, desmin, smoothelin, and myosin, as previously reported.
5
Subtyping of PWTs was performed according with the following criteria
5
: Cases expressing only vimentin and characterized by perivascular whorls were classified as angiofibroma. Cases expressing vimentin and smooth muscle actin with predominant staghorn vessels were classified as hemangiopericytoma. Cases with a mature muscular phenotype (smooth muscle myosin and/or smoothelin positive) and any of the perivascular histological pattern were classified as angioleiomyoma or angioleiomyosarcoma based on the degree of atypia of neoplastic cells. Cases with intermediate myoid differentiation (smooth muscle actin, calponin, and variably desmin) and perivascular (pericapillary) whorls were classified as myopericytoma. Cases with intermediate myoid differentiation and perivascular (adventitial) whorls were classified as adventitial tumors.
For the evaluation of GF pathway and COX2 expression, 5-μm cryostatic sections were air-dried, fixed in cold acetone (3 minutes at 5°), and stored at –70°C. Dilution and source of primary antibodies are listed in Supplemental Table 1.
Sections were incubated at room temperature for 1 hour with the primary antibodies and 30 minutes with the appropriate biotin-linked secondary antibody (Dako, Goldstrup, Denmark; dilution 1:200).
The immunoreaction was visualized for all reactions with amino-9-ethyl-carbazole chromogen (AEC, Kit, Vector, Burlingame, CA, USA). Sections were counterstained with Mayer’s hematoxylin and mounted with glycerine. As positive controls, sections of granulation tissue were used. The different cellular components of the wall of intratumoral vessel served as internal positive and negative controls. Negative controls consisted of the substitution of specific primary antibodies with an isotype-matched, irrelevant monoclonal antibody (for monoclonal primary antibodies), irrelevant polyclonal antibody (for polyclonal antibodies), or omission of the primary antibody. Scoring of the GFs pathways and COX2 was performed independently by 2 pathologists in a semiquantitative manner, examining the entire sample at intermediate magnification (200×), and evaluating the percentage of positive neoplastic cells, according with the following scoring system: 0, negative; 1, ≤20%; 2, 21%–50%; 3, 51%–70%; 4, ≥70%. Since the majority of the GFs and GF receptors examined in MCs can be expressed during normal angiogenesis, these molecules were considered highly expressed if staining was present in >70% of neoplastic cells (score 4). On the contrary, since COX2 is seldom expressed by MCs, it was considered highly expressed when expressed by >20% of neoplastic cells according with scoring systems reported in the literature (score, ≥2). 41 Intensity of the staining and extracellular staining were not included in the scoring system. Variation of the staining intensity within a sample was recorded. Differences between the scores of the 2 pathologists were discussed jointly to achieve an agreement.
Statistical Analysis
The association between the expression of GFs and GF receptors and between COX2 and GFs was evaluated for each couple of variables by the odds ratio (OR) statistics estimated by the logistic regression model. OR is a measure of the strength of the association between 2 variables. In the case of independence, OR is equal to 1; OR values near to 1 indicate a weak association; and OR values further from 1 indicate the presence of a strong association. The strength of association was classified as follows: weak, >1.2 and ≤1.5; moderate, >1.5 and ≤3; strong, >3. 33 Because of the low number of tumors included in some of the categories (eg, COX2 high expression), only univariate analysis was performed.
The relationship between Ki67 labeling index and high expression of GF, GF receptors, and COX2 was evaluated by linear regression models. Residuals analysis was performed to evaluate the assumption underlying the regression parameters inference. Taking into account the sample size, only regression models with a maximum of 4 variables were considered to avoid unreliable results. KI67 was the dependent variable and the following models were considered: (1) VEGF + VEGFR-I + VEGFR-II, (2) PDGFB + PDGFRβ + flg + bFGF, (3) COX2. In each model GF, GF receptors, and COX2 were included as dummy variables. A P value of .05 was considered significant and indicative of a putative association between variables. The association between GF high expression and PWT subtypes was not evaluated, because of the small number of cases included in some diagnostic categories.
Results
Diagnosis, Histologic Parameters, and Ki67 Labeling Index
We collected 40 cases of canine PWTs: 18 grade I, 18 grade II, and 4 grade III. Two cases were classified as hemangiopericytoma, 26 myopericytoma, 5 angioleiomyoma, 2 angioleiomyosarcoma, 2 adventitial tumors, and 3 angiofibroma. The major histologic pattern, the immunohistochemical phenotype, and specific diagnosis of each case are listed in Supplemental Table 2. Size (largest diameter) of the tumor was collected in 36 of 40 cases and ranged between 0.5 and 20 cm (median, 5 cm). Mitotic index evaluated in 10 high-power fields ranged between 0 and 48 (median, 4). Ki67 labeling index ranged between 0.8% and 36.2% (median, 7.20%). In 31 cases, necrosis was absent; in 7 cases, it was present in <50% of the tumor; and in 2 cases, it was present in >50% of the tumor.
Immunohistochemistry for GFs, Their Receptors, and COX2
The scores of the immunohistochemical expression of VEGF, VEGFR-I, VEGFR-II, PDGFB, PDGFRβ, bFGF, Flg, TGFβ1, TGFβR-I, TGFβR-II, and COX2 for each case are listed in Supplemental Table 3.
VEGF was highly expressed in 32 of 40 cases (80%; Fig. 1), VEGFR-I in 33 cases (82.5%; Fig. 2), and VEGFR-II in 32 cases (80%; Fig. 3). VEGF, VEGFR-I, and VEGFR-II were simultaneously highly expressed in 27 of 40 cases (67.5%). Cases negative for VEGF, VEGFR-I, and VEGFR-II were 1, 0, and 1, respectively. In intraneoplastic vessels, MCs were always VEGFR-II negative (Fig. 3). PDGFB was highly expressed in 37 of 40 cases (92.5%; Figs. 4, 5) and PDGFRβ in 32 cases (80%; Fig. 6). PDGFB and PDGFRβ were simultaneously highly expressed in 30 of 40 cases (75%). Cases negative for PDGFB and PDGFRβ were 0 and 3, respectively. In intraneoplastic vessels, MCs were PDGFB negative (Fig. 5) and PDGFRβ positive (Fig. 6). bFGF was highly expressed in 29 of 40 cases (72.5%; Fig. 7) and Flg in 21 cases (52.5%; Fig. 8). bFGF and Flg were simultaneously highly expressed in 19 of 40 cases (47.5%). No cases were negative for bFGF and Flg. TGFβ1 was highly expressed in 9 of 40 cases (22.5%) and had low expression in 31 cases (77.5%; Fig. 9). TGFβR-I was highly expressed in 16 of 40 cases (40%; Fig. 10) and TGFβR-II in 24 cases (60%; Fig. 11). TGFβ1, TGFβR-I, and TGFβR-II were simultaneously highly expressed in 4 of 40 cases (10%). Cases negative to TGFβ1, TGFβR-I, and TGFβR-II were 1, 1, and 0, respectively. COX2 was highly expressed in 14 of 40 cases (35%), while 21 cases were negative to COX2 (52.5%; Fig. 12). In a minority of cases, the intensity of the staining for VEGF, bFGF, PDGFB, TGFβ1 and their receptors VEGFR-I, VEGFR-II, PDGFRβ, Flg, TGFβR-I, TGFβR-II was more intense in a subgroup of neoplastic cells, ranging from 5% to 10% of positive cells. For each molecule evaluated, the expression was cytoplasmic. Graphs summarizing immunohistochemical results are depicted in Figure 13, 14, and 15.
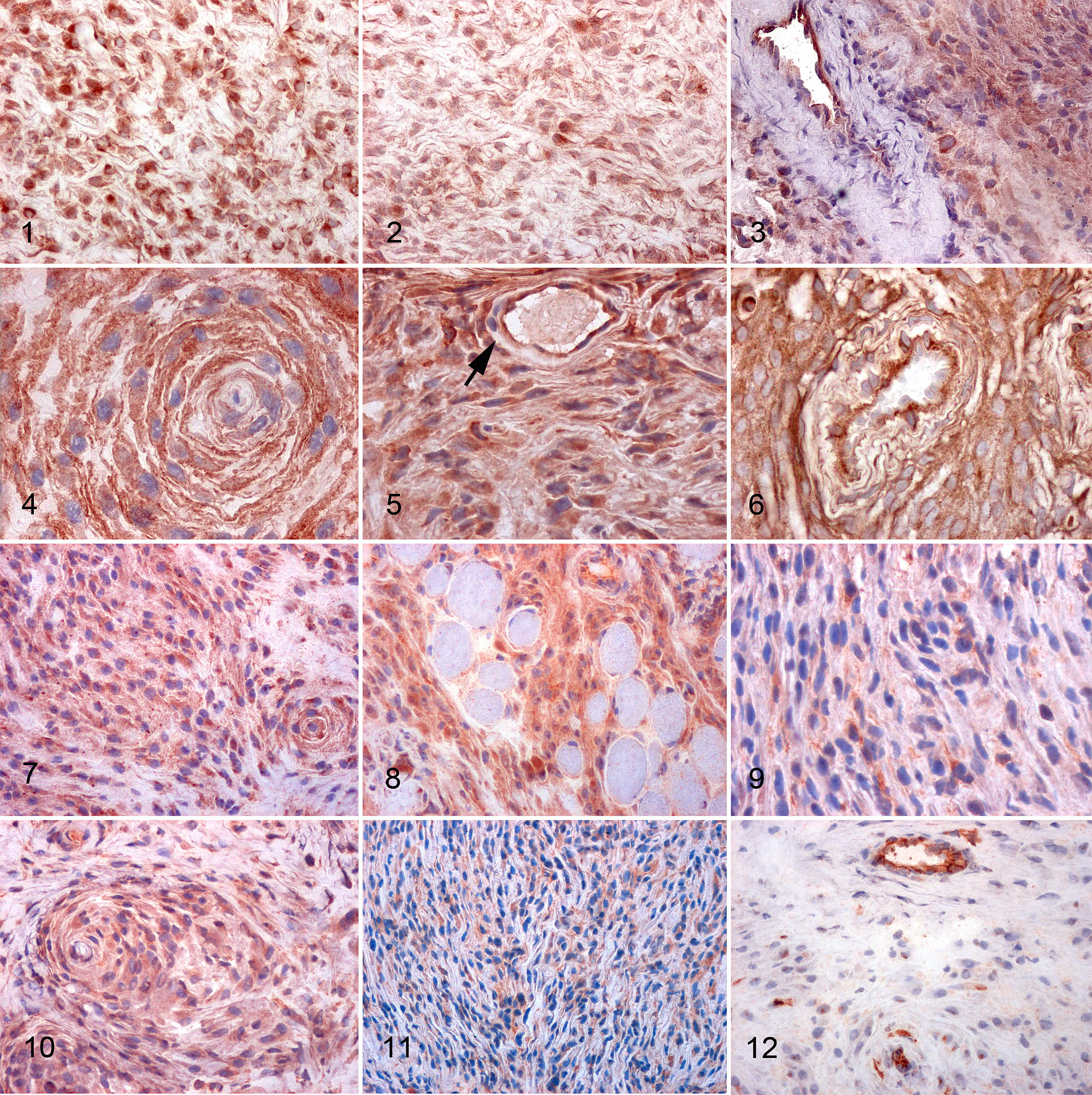
Perivascular wall tumors, dog, subcutis. Immunohistochemistry for vascular endothelial growth factor (VEGF) and receptors VEGFR-I and VEGFR-II, platelet-derived growth factor B (PDGFB) and receptor PDGFRβ, basic fibroblast GF (bFGF), Flg, transforming growth factor β1 (TGFβ1) and receptors TGFβR-I and TGFβR-II, and COX2. ABC method, AEC chromogen, hematoxylin counterstain.
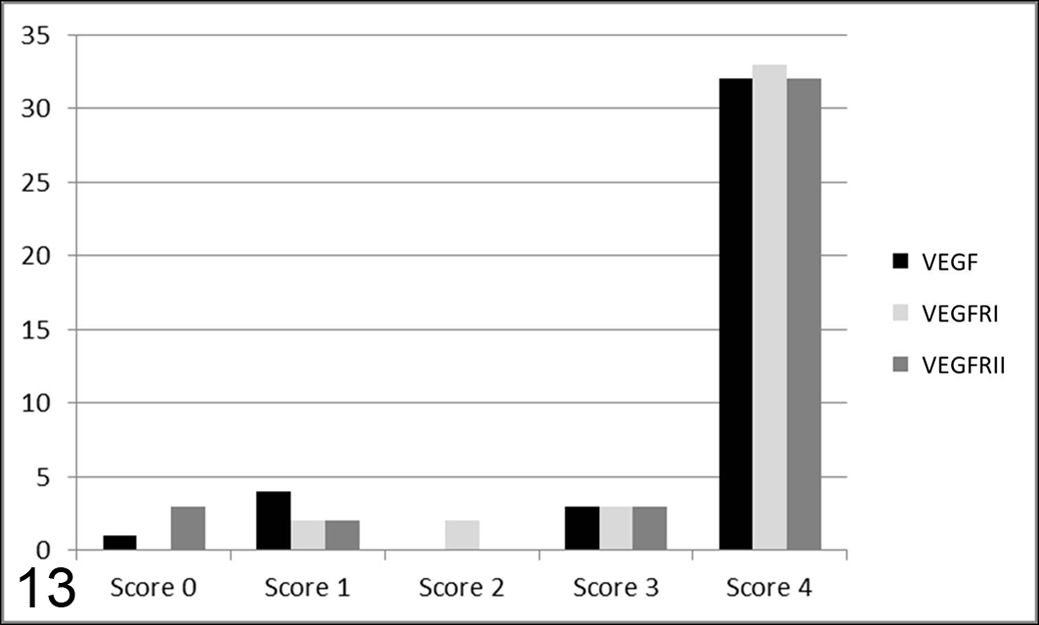
Graphs summarizing the number of cases (y-axis) in each score class (x-axis) for vascular endothelial growth factor (VEGF) and receptors VEGFR-I and VEGFR-II. More than 75% of cases received score 4 for these 3 markers.
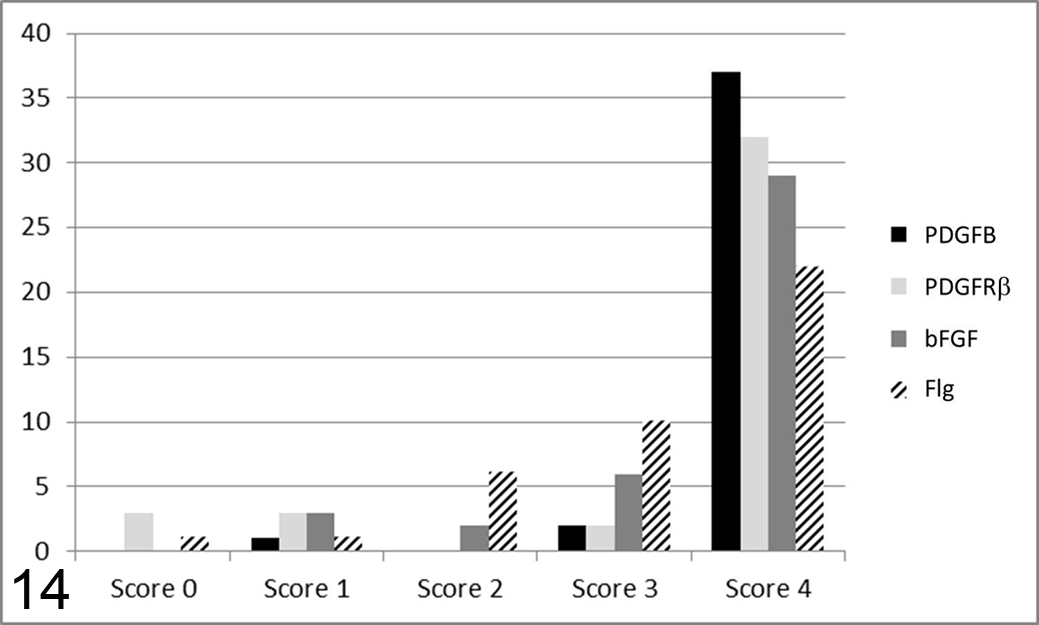
Graphs summarizing the number of cases (y-axis) in each score class (x-axis) for platelet-derived growth factor B (PDGFB) and receptor PDGFRβ, as well as basic fibroblast GF (bFGF) and Flg. More than 75% of cases received score 4 for PDGFB and PDGFRβ, and 50% of the cases received score 4 for bFGF and Flg. A statistically significant association was found between bFGF and PDGFRβ.
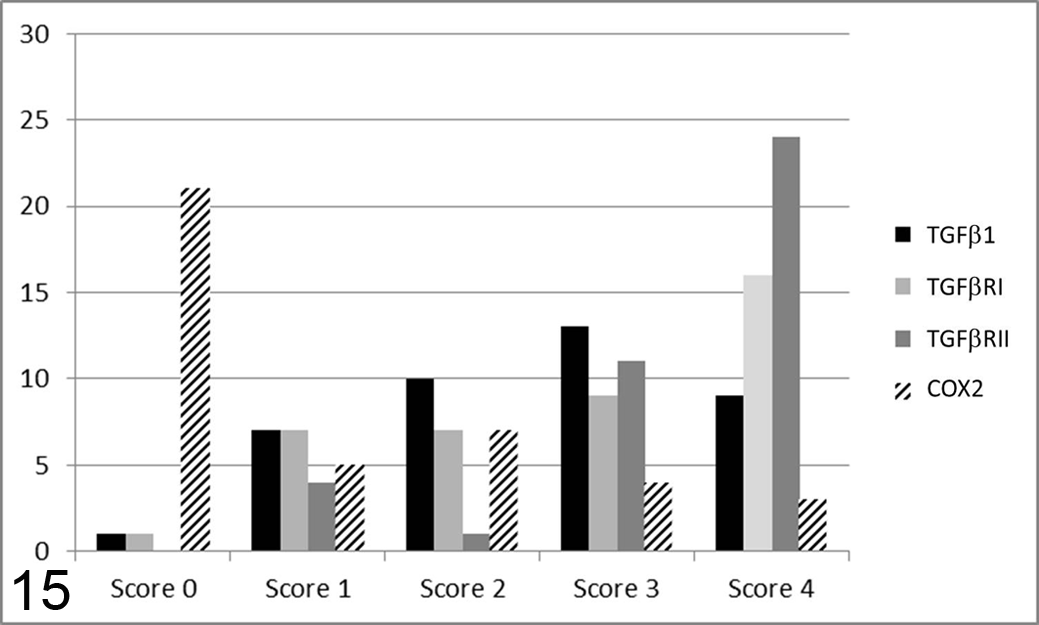
Graphs summarizing the number of cases (y-axis) in each score class (x-axis) for transforming growth factor β1 (TGFβ1) and receptors TGFβR-I and TGFβR-II, as well as COX2. More than 50% of the cases received score 4 for TGFβR-II. TGFβ1 and TGFβR-I were less expressed, and COX2 was negative in the majority of cases.
Statistical Analysis
The odds of high expression of GF receptor was more than 3 in tumors with high GF expression relative to those with low GF expression (P < .05); specifically, a statistically significant and strong association was found between high expression of VEGF and VEGFR-I (OR = 9.667; P = .015), VEGF and VEGFR-II (OR = 16.111; P = .003), bFGF and Flg (OR = 5.067; P = .038), and bFGF and PDGFRβ (OR = 16.2; P = .003). The odds of high expression of TGFβ1 were 12 times greater in cases with high expression of COX2 relative to those with low expression of COX2 (P = .006). Weak nonstatistically significant associations were found between TGFβ and TGFβR-I (OR = 1.27, P = .757) and between COX2 and bFGF (OR = 0.92, P = .911). Moderate nonstatistically significant associations were found between TGFβ and TGFβR-II (OR = 2.88, P = .229), between PDGFB and Flg (OR = 2.63, P = .447), and between COX2 and VEGF (OR = 1.80, P = .511).
Concerning Ki67 labeling index, residuals analysis suggested the use of logarithmic scale. No significant relationship was found between Ki67 labeling index and VEGF, VEGFR-I, VEGFR-II (F 3,36 = 0.6703, P = .5758), between Ki67 and PDGFB, PDGFRβ, flg, bFGF (F 4,35 = 0.2989, P = .8766), and between Ki67 and COX2 (F 1,38 = 0.4791, P = .4931).
Results of regression models suggested a weak association between Ki67 labeling index and the above-mentioned variables being near to 1.00 (from 0.8 to 1.3)—specifically, the estimated ratio between the average of Ki67 in high versus low expression of GFs and in high versus low expression of COX2 (see supplemental material).
Discussion
Most canine PWTs expressed molecules involved in VEGF-, PDGFB-, and bFGF-mediated signaling pathways. The statistically significant association between GFs and GF receptor expression observed in PWTs recapitulated the upregulatory loops involved in MCs and EC crosstalk described in vitro. 7,34
Immunohistochemistry revealed the expression of VEGF, VEGFR-I, and VEGFR-II in a high percentage of cells in most PWTs. VEGF, which is normally produced by MCs during angiogenesis, binds to VEGFR-I/II on ECs, stimulating EC proliferation. 42 Interestingly, PWT neoplastic MCs were characterized by the aberrant expression of VEGFR-II. VEGFR-II is normally absent in MCs, as reported in the literature 48 and as evidenced in the intratumoral vessels of tested cases. This finding can be related to the concurrent activation of an autocrine and paracrine pathway stimulating proliferation of neoplastic cells and intratumoral angiogenesis. Furthermore, the association between the high expression of VEGF and VEGF receptors was statistically significant. This finding suggests the presence of an upregulatory pathway between VEGF and VEGFR-I/II that has been already demonstrated in vitro 7 and that, in canine PWTs, may cause the amplification of the autocrine/paracrine loop involved in neoplastic cell proliferation. This hypothesis has been suggested also for canine STSs and osteosarcomas, where VEGF and VEGFR-I/II expression by neoplastic cells and their presence in the serum of affected dogs have been reported. 15,16,19 As a consequence of these promising observations, therapies targeting VEGF have been applied to canine STSs utilizing vaccine and virotherapy approaches. 27,37
The PDGFB-PDGFRβ pathway seemed also involved in canine PWT development. During angiogenesis, PDGFB is produced by ECs and binds to PDGFRβ that is expressed by MCs. 24,26 This process leads to MC recruitment and vascular wall stabilization. 24,26
PDGFB is usually produced by ECs and not MCs, 24,26 a finding confirmed by intratumoral normal vessel expression pattern in this report. Interestingly, PWT neoplastic MCs highly expressed PDGFRβ and PDGFB, suggesting the presence of an additional autocrine and paracrine pathway involved in PWT development and growth, characterized by aberrant expression of PDGFB. Upregulation of the PDGFB pathway has been demonstrated in canine hemangiosarcoma, feline injection site sarcoma, 3,28 and human sarcomas, 32 in which it has been also targeted for therapeutic purposes. 9,12
The bFGF-Flg pathway was also highly expressed by canine PWTs. bFGF is a potent angiogenic factor that stimulates EC and MC proliferation. 21,46 While bFGF can directly stimulate EC proliferation, the presence of PDGFB is necessary for bFGF-mediated MC proliferation in vivo since bFGF and PDGFBB upregulate PDGFRβ on ECs and Flg on MCs, respectively. 34 The significant association between bFGF and PDGFRβ high expression suggested a major involvement also of this upregulation loop in canine PWT development.
The fourth pathway investigated in this report involved TGFβ1, TGFβR-I, and TGFβR-II. The majority of PWTs were characterized by the expression of TGFβ1 by <70% of neoplastic cells. TGFβ1 and its receptors are normally expressed in ECs and MCs and are involved in the induction of MCs from undifferentiated mesenchymal cells. 44 A further role of the TGFβ superfamily is to regulate COX2 secretion in hypoxic tissues; hypoxia itself represents an additional trigger for MCs proliferation. 35,45 Simultaneous high expression of TGFβ1 and the 2 corresponding receptors was evident in 4 of 40 PWTs. Based on these findings, the TGFβ1 pathway seems to play a minor role in canine PWTs pathogenesis. Interestingly, in this study, TGFβ1 expression was significantly associated with COX2 expression, supporting the presence of a link between these 2 molecules, as demonstrated by previous studies. 35,45
COX2 was detected in a minority of tumors. This result was expected since COX2 expression has been reported mainly in epithelial tumors, 14,20 while its presence in mesenchymal neoplasm is inconstant and rarely has a prognostic relevance. 11,18,23,25
Mitotic index and MIB-1 labeling index of PWTs were similar to what has been reported, 4,47 and a statistical association with the expression of GFs was not identified. This result, although deceiving, is not surprising, since neoplastic cell proliferation rate is likely the result of the interaction among several GFs, GF receptors, and other cell cycle regulators, but the number of cases included in this study did not allow the evaluation of association patterns of multiple GFs. An alternative hypothesis is that an alteration of the downstream intracellular signaling pathways induces an inefficient expression of GF and GF receptors, hampering signal transduction. Evaluation of the expression levels of the molecules involved in signal transduction could better elucidate this hypothesis. As a last point, the expression of GFs involved in angiogenesis, such as bFGF, PDGFB, and VEGF, may derive from a hypoxic microenvironment (as often occurs for neoplastic lesions) inducing angiogenesis not directly correlated with neoplastic cell proliferation. Nevertheless, this latter hypothesis seems improbable given the distinctive lack of expression of specific GF receptors by MCs of intratumoral blood vessels. The association between TGFβ and its receptors and between PDGFB and Flg was accounted for but surprisingly not detected in PWTs. For these cases, an OR >2 may be still suggestive of an association that nevertheless is not significant, likely as the consequence of the evaluation of a statistical insufficient number of cases in the modalities of the examined parameters.
Interestingly, the pathways with the highest expression in canine PWTs are mediated by tyrosine kinase receptors. This finding suggests that tyrosine kinase receptor blockade may represent a promising therapy to control canine PWT recurrence. If this hypothesis is substantiated by future clinical studies, results could parallel observations reported for some human STS subtypes 9,10,32 and in a few studies in veterinary medicine that attempted target therapies toward specific GFs. 27,31,37 Noteworthy, the efficacy and safety of the use of a commercially available tyrosine kinase inhibitor for canine mast cell tumor therapy has been already reported. 22
However, based on the correlations between different GF and GF receptor expressions identified in this work, an approach aimed at multiple pathway inhibition (mainly VEGF, PDGFB, and bFGF) seems a possibly more promising approach for PWT control.
In summary, we have identified the high expression of 3 signaling pathways that may play a role in PWT pathogenesis and that represent good candidates for the control of local recurrence, which is the most common canine PWT relapse. 4,47
Footnotes
Acknowledgement
This work was supported by the International Society of Veterinary Dermatopathology research grant 2010.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the International Society of Veterinary Dermatopathology research grant 2010.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
