Abstract
In many health-related fields, there is great interest in the identification of biomarkers that distinguish diseased from healthy individuals. In addition to identifying the diseased state, biomarkers have potential use in predicting disease risk, monitoring disease progression, evaluating treatment efficacy, and informing pathogenesis. This review details the genetic and biochemical markers associated with canine primary glaucoma. While there are numerous molecular markers (biochemical and genetic) associated with glaucoma in dogs, there is no ideal biomarker that allows early diagnosis and/or identification of disease progression. Genetic mutations associated with canine glaucoma include those affecting ADAMTS10, ADAMTS17, Myocilin, Nebulin, COL1A2, RAB22A, and SRBD1. With the exception of Myocilin, there is very limited crossover in genetic biomarkers identified between human and canine glaucomas. Mutations associated with canine glaucoma vary between and within canine breeds, and gene discoveries therefore have limited overall effects as a screening tool in the general canine population. Biochemical markers of glaucoma include indicators of inflammation, oxidative stress, serum autoantibodies, matrix metalloproteinases, tumor necrosis factor–α, and transforming growth factor–β. These markers include those that indicate an adaptive or protective response, as well as those that reflect the damage arising from oxidative stress.
Currently identified biomarkers of canine glaucoma typically lack disease specificity. Combined with our limited understanding of glaucoma etiopathogenesis, caution must be used to ensure each biomarker is used for the purpose for which it has been developed, investigated, or validated.
In this article, we review molecular and biochemical markers that have been identified in canine primary glaucoma and how these might be used and further developed to enhance our understanding and management of glaucoma (Table 1). A biomarker is a measurable anatomic, physiologic, biochemical, or molecular parameter indicative of a normal or pathologic process, disease state, or a response to an intervention. 16 In practice, biomarkers include tools and technologies that can aid in understanding the prediction, cause, diagnosis, progression, regression, or outcome of treatment of disease. 60 A good biomarker should be precise and reliable, distinguish between normal and diseased states, and differentiate between different diseases. 78 There are 2 major types of biomarkers: 60 biomarkers of exposure, which are used in risk prediction, and biomarkers of disease, which are used in screening, diagnosis, and monitoring of disease progression. The most important criterion for a relevant biomarker is its disease specificity. 36
Use of Molecular and Biochemical Biomarkers in Canine Primary Glaucoma.
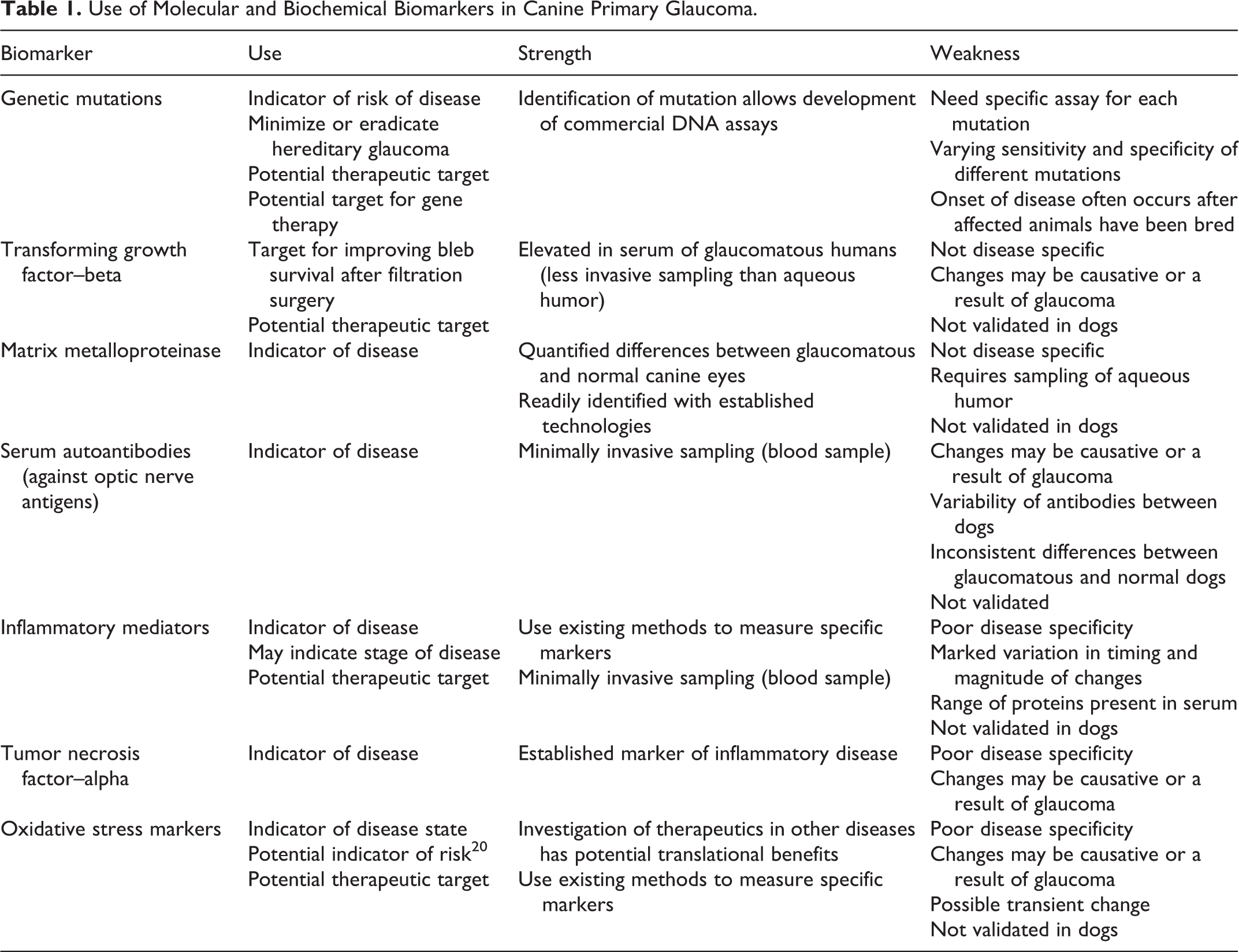
There is no ideal biomarker for glaucoma. Currently available clinical diagnostic tests (eg, tonometry, gonioscopy) provide a very limited measure of risk of glaucoma and an imprecise measure of disease progression in dogs. While an elevated intraocular pressure (IOP) is considered a constant risk factor in canine glaucoma 74 and a major risk factor in human glaucoma, the need for therapeutic interventions that address factors other than IOP is supported by the presence of normal-tension glaucoma and intraocular hypertension without glaucoma in humans, as well as the progression of glaucomatous optic nerve damage and vision loss despite an IOP within the normal range. Other proposed mechanisms involved in glaucoma include neuroinflammation, 59 hypoxia/ischemia, 8,84 increased extracellular glutamate and excitotoxicity, 21 oxidative stress, 23 obstruction of axoplasmic flow, 9 and deprivation of trophic factors. 77 By identifying accurate biomarkers of these processes in the canine glaucoma patient, the stage of disease or effect of an intervention might be more precisely identified.
Similar to the use of biomarkers in other medical fields (eg, oncology), the use of identifying biomarkers to provide a binary “yes/no” in the diagnosis of glaucoma is likely to be ineffective due to the complex, multifactorial, and incompletely understood nature of the disease and the fact that most biomarkers are not unique to glaucoma. Multiple biomarkers that create a molecular “phenotype” or biomarker set that can be used to recognize unique patterns in canine glaucoma patients might allow the earlier detection of disease, lead to additional therapeutic targets and therapeutics, and influence management of human glaucoma through validation of the dog as an animal model of glaucoma.
While genetic markers have been used and developed over time, the methods use just three classes of markers: protein variants (allozymes), DNA sequence polymorphisms, and DNA repeat variation. 85 The principle of allozyme markers is that protein variants in enzymes can be distinguished according to differences in size and isoelectric point caused by amino acid substitutions. 85 The subsequent development of DNA manipulation techniques led to a shift from enzyme-based to DNA-based markers, providing a more direct and sensitive method of detecting variations. Molecular markers in medicine involve molecules, reactions, and/or pathways altered by pathologic conditions. 36
Genetic Markers
Genetic markers provide information about allelic variation at a given locus 85 and are typically a biomarker of exposure (indicator of risk). 60 They exist before the disease/outcome occurs and are generally independent of other exposures. The most common types of genetic markers include restricted fragment length polymorphisms (RFLPs), amplified fragment length polymorphisms (AFLPs), random amplified polymorphic DNA (RAPD), simple sequence repeats (SSRs or microsatellites), and single-nucleotide polymorphisms (SNPs).
The identification of specific genetic mutations in several breeds of dog has led to the development of commercial DNA assays that help identify dogs at risk of the disease and remove them from the breeding population. The value of genetic screening as a tool to minimize or eradicate hereditary glaucoma is reliant on the presence of an assay for the specific mutation, the specificity and sensitivity of that mutation for identifying the risk of glaucoma, and use of the test in breeding programs.
Canine primary angle closure glaucoma (PACG) is multifactorial, with numerous studies indicating it is not a mere genetic trait. 18,27,32,49 The fact that the pathophysiology of glaucoma is incompletely understood and indeed likely varies between types of glaucoma, between species, and possibly between breeds of dog means that screening dogs and determining their risk of disease will continue to develop with improved understanding of the glaucomas. For example, progressive pectinate ligament abnormalities have been identified in some breeds predisposed to primary glaucoma (Flat Coated Retrievers, Eurasier dogs). The fact that pectinate ligament abnormalities and clinical glaucoma may develop despite prior test results of “normal” or “unaffected” has significant implications on the screening of breeding stock. 19,82,102
Although genetic markers are detected only in certain populations (Table 2), finding these markers is critical in identifying the cause and pathogenesis of disease, 78 and research provides potential screening and therapeutic targets in both canine and human glaucoma patients. However, the mutated genes play a very limited role in the actual pathogenesis and typically do not explain the usual clinical picture. 36,100 While alteration in expression status of only one gene is almost never disease specific, a biomarker set is particularly valuable for the creation of highly precise diagnostic approaches. 36
Genetic Mutations Associated With Canine Primary Glaucoma.
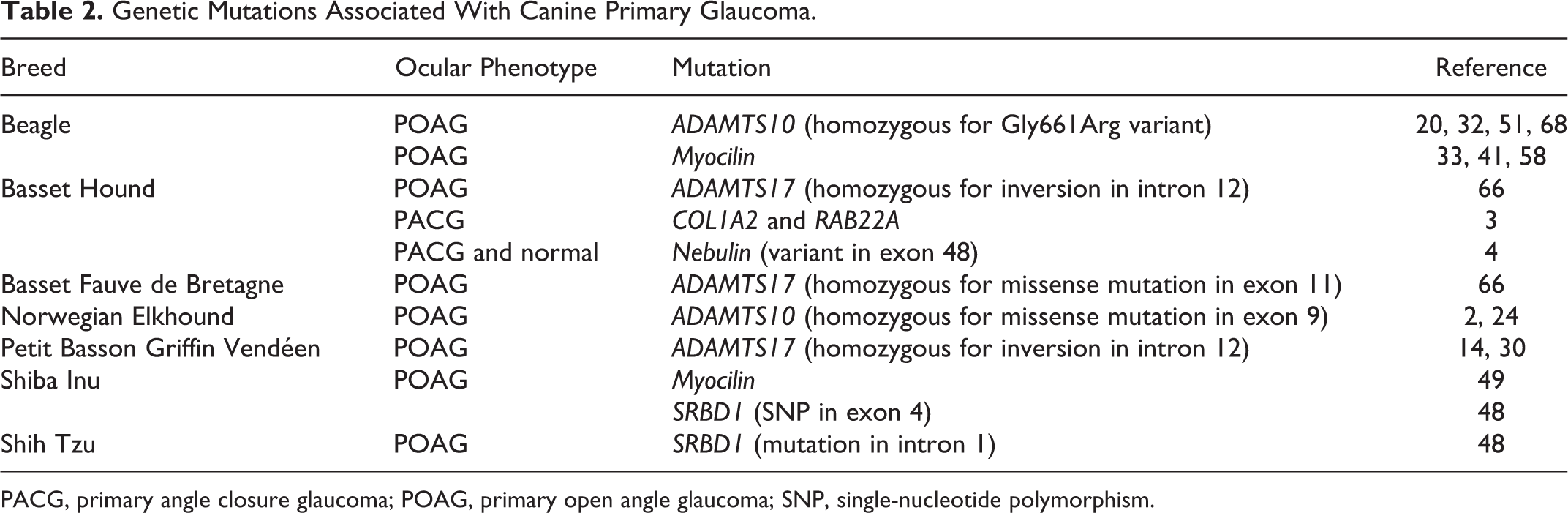
PACG, primary angle closure glaucoma; POAG, primary open angle glaucoma; SNP, single-nucleotide polymorphism.
ADAM Metallopeptidase With Thrombospondin Type 1 Motif 10 (ADAMTS10)
A missense mutation (Gly661Arg) in the ADAMTS10 gene was associated with primary open angle glaucoma (POAG) in a research colony of Beagles. 32,51 This mutation is positioned in the cysteine-rich domain and is hypothesized to disrupt protein folding, leading to instability. 51 This gene is also associated with fibrillin mutations and is important in microfibril formation and function. 51 The mutation has been excluded as a cause of primary glaucoma in the American Cocker Spaniel, Australian Cattle Dog, Chihuahua, Jack Russell Terrier, Siberian Husky, Shiba Inu, Shih Tzu, and Yorkshire Terrier. 52
Glaucoma in Norwegian Elkhounds was mapped to the known canine POAG locus, including the ADAMTS10 candidate gene, and subsequently identified as a missense mutation in exon 9. 2 The recessively segregating mutation results in an alanine to threonine change (p.A387T) in a highly conserved functional metalloprotease domain of the protein, which likely impairs ADAMTS10 function, leading to POAG in the homozygous dogs.
The Norwegian Elkhound mutation is different from that in Beagles, suggesting the pathogenesis may be different in these breeds. Mutations in the Norwegian Elkhound affect a highly conserved residue in the metalloprotease domain, which plays a role in remodeling of connective tissue. 24 Human metalloprotease domain mutations have revealed abnormalities in the cellular cytoskeleton, suggesting abnormal interactions with the extracellular matrix. These abnormalities may eventually result in defective microfibrils and glaucoma through alterations in biomechanical properties of tissue and/or through effects on signaling through transforming growth factor–β (TGF-β), which is known to be elevated in the aqueous humor of glaucoma patients. 53
Recent investigations have demonstrated significant alterations in the posterior scleral collagen microstructure before the onset of clinical glaucoma in ADAMTS10-mutant dogs 20 and significant stiffening of the posterior sclera associated with age in ADAMTS10-mutant dogs that did or did not have glaucoma. 68 The rate of stiffening with age was similar between dogs with and without glaucoma despite exposure to progressively elevated IOPs in dogs with glaucoma, which might have affected the scleral extracellular matrix. 35 Changes to the rate and degree of scleral stiffening with age suggest a weaker capacity for handling an occasional rapid increase in IOP as the eye ages. 68
ADAM Metallopeptidase With Thrombospondin Type 1 Motif 17 (ADAMTS17)
Glaucoma was found in 38 of 366 Petit Basson Griffon Vendéen (PBGV) dogs and was similar to that described in the Beagle and Norwegian Elkhound. 14 All 38 were negative by commercially available DNA testing for the ADAMTS10 mutation (responsible for POAG in the Beagle) and the ADAMTS17 mutation (responsible for primary lens luxation in several breeds). However, the locus for POAG in the PBGV was successfully mapped in a genome-wide association (GWA) study using exome sequencing. 30 Follow-up by genome sequencing of an individual case identified an inversion mutation with a breakpoint disrupting the ADAMTS17 gene.
While screening of these PBGVs did not identify mutations known to be associated with POAG in Beagles or primary lens luxation in terrier breeds, 14 mutations in these or related genes are possible. As lens zonules consist of microfibrils, it seems possible that relevant genetic mutations (such as those encoding microfibril proteins) would directly affect them. Further investigations are required to determine whether there is a direct association between POAG and lens luxation and/or whether zonular weakness and breakdown occur as part of the primary disease process or are secondary to buphthalmia associated with chronic elevations in IOP.
In the Basset Hound, a homozygous 19-bp deletion in exon 2 was present in all POAG-affected dogs. 66 The deletion is predicted to alter the reading frame of the transcript and introduce a premature stop codon at the 5′ end of exon 3, which would result in a truncated and aberrant protein if the RNA were stable and not subjected to nonsense-mediated decay. If the transcript is translated, the protein is expected to be truncated by 86%, including the entire catalytic domain, which would be expected to lead to complete loss of protein function.
All affected Basset Fauve de Bretagne dogs tested were homozygous for a missense mutation in exon 11, causing a glycine to serine amino acid substitution (G519S) in the disintegrin-like domain of ADAMTS17, which is predicted to alter protein function. 66 Unaffected Basset Fauve de Bretagne dogs were either heterozygous for the mutation (5/24) or homozygous for the wild-type allele (19/24).
While identifying the ADAMTS17 gene as the site of an identified mutation in these 3 breeds, these findings demonstrate the breed specificity of specific mutations associated with glaucoma and highlight the difficulties present in undertaking any widespread genetic screening program.
Myocilin
Myocilin protein was identified in the anterior chamber of Beagles with POAG, 58 but its role in POAG was not clarified. Aqueous myocilin protein levels increased significantly in primary and secondary glaucomatous eyes in many canine breeds, including Beagles, while they were low in Beagles with primary cataract, diabetic cataract, and normotensive eyes. 33 Changes in myocilin levels in the aqueous humor outflow pathway may be associated with increased IOP and subsequent glaucoma in Beagles. 41 There have been no reports on the relationship between myocilin gene mutations and the incidence or pathogenesis of canine PACG. 49
Myocilin sequence variants were identified in glaucomatous and normotensive Shiba Inu dogs with both open and closed iridocorneal angles (ICAs). However, these myocilin polymorphisms appear to be independent of the occurrence of glaucoma and the ICA grade. The observation of myocilin mutations/polymorphisms in Shiba Inu dogs indicated that there were more alterations in exon 1 than in exon 3 and no mutations or polymorphisms detected in exon 2. 49 This is similar to glaucoma in humans, in whom many amino acid mutations in exon 3 are known to cause glaucoma, 29 but exon 2 mutations are not reported in human POAG or PACG. 13,37,81
While only 4% of human POAG patients have mutated myocilin, this is the only known genetic mutation associated with both human and canine glaucoma. Given its presence in glaucoma in both species, as well as having been identified in 2 canine breeds (Shiba Inu and Beagle), further research might be useful to identify this mutation in other breeds predisposed to glaucoma.
Collagen Type I Alpha 2 (COL1A2) and RAB22A
The gene COL1A2 on chromosome 14 and RAB22A on chromosome 24 were identified as susceptibility loci on a GWA analysis of Basset Hounds with clinically confirmed PACG. 3 These results suggested that variation in multiple loci determined the risk for canine PACG in Basset Hounds, in fitting with the complex nature of PACG inheritance. 3
Nebulin (NEB)
Ahram et al 4 identified an NEB-based variant g.5588214 A→G in exon 48 (p.2051 Lys→Arg) and confirmed the association of this variant with PACG in Basset Hounds. This gene was predicted to affect protein function due to a possible pathogenic amino acid substitution within a highly phylogenetically conserved region. However, the disease-associated allele appears to be quite common in the US Basset Hound population. Furthermore, homozygosity for the risk allele was observed in 33% of the unaffected animals, and heterozygosity was observed in a small fraction of the affected dogs, suggesting additional factors are required for development of glaucoma.
S1 RNA Binding Domain 1 (SRBD1)
A significant association of SRBD1 polymorphisms and glaucoma was identified in Shiba Inus and Shih Tzus. 48 In Shiba Inus, the strongest association with glaucoma in SRBD1 was observed at rs22018513, which is a synonymous SNP in exon 4. Two other SNPs, rs8655283 and rs22018514, were also significantly associated with glaucoma; however, these significant associations were calculated only secondarily from a strong linkage disequilibrium with rs22018513. In Shih Tzus, however, rs22018513 was not associated with glaucoma. Only rs9172407 in intron 1 of SRBD1 showed a statistically significant association in Shih Tzus, while this association was not found in Shiba Inus. These results suggest that rs22018513 and rs9172407, or a respective neighboring polymorphism, may be a causative factor for glaucoma in Shiba Inus and Shih Tzus, respectively. SRBD1 polymorphisms were associated with canine glaucoma and human glaucoma independent of IOP, 48 suggesting that SRBD1 polymorphisms may affect a common disease condition in canine and human glaucoma.
Biochemical Markers in Canine Glaucoma
A biochemical marker is a biochemical variable associated with a disease (directly or indirectly) and might be any biochemical compound (antigen, antibody, enzyme, hormone, etc) that is sufficiently altered to provide diagnostic or predictive value. 16,36 Biochemical markers may differentiate specific diseases and guide therapy. 78 Unlike genetic markers, which primarily serve as a marker of disease risk and are independent of other factors, many biochemical markers of glaucoma are nonspecific, and their presence must be interpreted in context. Biomarkers obtained from biological samples (eg, blood) may indicate subclinical disease, stage, or severity of disease. They may be present due to their causative role in glaucoma or as a result of the disease process. Potential uses of these biomarkers include 60 identification of “at-risk” individuals or those in “preclinical” stages; reduction in disease heterogeneity in clinical trials or epidemiologic studies; monitoring the progression of disease, including induction, latency, and clinically detected phases; and as a direct target for therapeutic interventions.
TGF-β
TGF-β signaling has been implicated in the pathogenesis of ocular, neurodegenerative and vascular diseases, as well as remodeling of extracellular matrix. 25,62 The association of microfibril genes Latent Transforming Growth Factor Beta Binding Protein 2 (LTBP2) with primary congenital glaucoma in humans 5,46 and ADAMTS10 with POAG in Beagles 51,52 led to a hypothesis that microfibril defects cause glaucoma. 51,53 Elevated levels of TGF-β1 have been identified in the plasma of POAG humans 54 and elevated aqueous humor TGF-β levels found in glaucomatous compared with normal eyes. 31,65,75 Microfibrils are located in the extracellular matrix of a variety of tissues, where they are a major reservoir of latent TGF-βs 54 and play a central role in TGF-β signaling. 43,79,80 In the eye, microfibrils are found in the trabecular meshwork, inner wall of Schlemm’s canal, iris, ciliary body, and optic nerve head. 40,86,99
The overlap in the cellular and tissue changes in glaucoma and those induced by TGF-β implies that dysfunctional TGF-β signaling could be involved in the pathogenesis of glaucoma in humans, monkeys, pigs, and mice. 15,17,75 TGF-β plays a role in scarring and fibrosis following glaucoma surgery and is a therapeutic target for improving survival of the conjunctival filtration bleb (minimizing fibrosis, which would otherwise encapsulate the draining aqueous humor) in glaucoma surgery. 55 Preclinical and clinical research supports the role of TGF-β in glaucoma of humans and other animals, 15,17,75 although whether these proteins are always causative or are induced as a consequence of glaucoma remains undetermined. A main target of TGF-β activity is the trabecular meshwork. Because TGF-β1 and TGF-β2 are potential modulators of aqueous outflow, extracellular matrix remodeling, and inflammation in glaucomatous eyes, these proteins are currently being investigated as therapeutic targets. 75 Thus, knowledge of TGF-β in canine glaucoma patients combined with advances made in other species may be translated to managing the progression of glaucoma in dogs.
Matrix Metalloproteinases
Intraocular matrix metalloproteinase (MMP) activity and its relationship to the aqueous humor outflow pathway have been quantified in normal and glaucomatous canine eyes, 97,98 and elevated levels of intraocular MMP-2 and MMP-9 have been documented in dogs with glaucoma. 97
Immunohistochemistry has been used to identify MMP-2 and MMP-9 in the ICA of normal and glaucomatous eyes and specifically localized within trabecular meshwork cells, the extracellular matrix, and inflammatory cells. 98 Quantitative assessment of immunolabeling revealed greater immunolabeling of MMP-2 and MMP-9 within the ICA tissue, iris, and cornea in secondary glaucomas compared with primary glaucomas. 98 Nonetheless, there was no difference in MMP activity between primary and secondary glaucomas, 97 and it is the activity (rather than the quantity of protein) that is most biologically meaningful. Future prospective investigations are required to determine if these changes directly mediate the glaucomatous changes or arise as a result of the disease process. 97,98
Autoantibodies Against Optic Nerve Antigens
Significant differences in the levels of autoreactivity at certain bands (increased autoreactivity at 40 and 53 kDa and decreased autoreactivity at 48 kDa) were identified in serum autoantibodies against optic nerve antigens between dogs with and without goniodysgenesis-related glaucoma. 76 However, the study did not address whether immune-mediated mechanisms are involved in the initial pathogenesis or are a consequence of the disease that may in turn accelerate or intensify the disease process. 76 Further studies are required given the population studied (advanced glaucoma vs healthy dogs), the high variability in autoantibodies among individual dogs, and the considerable overlap between groups.
Inflammatory Markers
Inflammation can result from a variety of tissue insults, including infections, trauma, autoimmunity, and malignancy. These stimuli lead to the production of cytokines that up- or downregulate expression of acute-phase proteins. 70 There is marked variation in the timing and magnitude of this response among different acute-phase proteins.
More marked inflammatory changes have been identified in the earlier stages of glaucoma with substantially decreased inflammatory changes in the more chronically damaged retinas. 59 The decreased expression of crystallins in the retina may reflect an early, transient event in glaucoma pathogenesis. 1,47,60,73 In a cohort of dogs considered at risk of developing glaucoma (healthy Eurasier dogs), there was no difference from control dogs in plasma levels of inflammatory markers, including C-reactive protein (CRP; a biomarker of the early phase of inflammation), haptoglobin (which increases over a few days and remains high for longer), and albumin (decreased synthesis during inflammation). 19
It is not definitively known whether photoreceptor cell loss or functional decline is a feature of advanced glaucoma, 42,64,94,103 and several photoreceptor cell-specific genes are expressed at decreased levels in glaucomatous retinas, 47 while others mediating the neuroinflammatory response are expressed at higher levels in advanced glaucomatous retinopathy. Prominent functional categories of genes with elevated expression in glaucomatous retina compared with normal retina include antigen presentation, complement activation, lysosomal and proteasome activity, acute-phase proteins, inflammation signaling, and apoptosis, as well as decreased expression of genes involved in neuronal development and maintenance, cell adhesion, calcium transport and binding, transcriptional regulation, and synaptic transmission. 47
It has been suggested that glaucoma is not initiated by an immune response to specific retinal antigens, but rather the variety of immunoreactive molecules identified was due to the exposure of numerous epitopes during the rapid neuronal cell death. It is therefore conceivable that an immune response could accelerate retinal degeneration that was initiated by elevation in IOP. 47
Tumor Necrosis Factor–α
Modulation of tumor necrosis factor–α (TNF-α) and its receptors is correlated to the development of glaucoma, with increased immunoreactivity in glaucomatous canine retinas. 47 Significantly higher TNF-α levels were detected in the aqueous humor of all dogs with intraocular disease compared with dogs with normal eyes, 26 and TNF-α levels were significantly higher in those with acute anterior uveitis compared with those with PACG. 26
Oxidative Stress Markers
Oxidative stress is an increase over physiologic values in the intracellular concentrations of reactive oxygen species (ROS). 39 This is reflected by changes in the levels of antioxidant defenses that can be increased (protective response) or depleted due to ROS action. 87
Retinal ganglion cell (RGC) death in glaucoma is associated with the generation and effects of ROS and reactive nitrogen species. 63 Oxidative stress (reduced glutathione, decline in superoxide dismutase) occurs in the glaucomatous optic nerve head, trabecular meshwork cells, and connective tissue. 7,45,83 It is likely important in the retinal damage that occurs in glaucoma, 12,28,50,91 with an apparent transient nature of the oxidative stress suggested, and may affect endothelial cell function. 22,23,63 Neurons, especially RGCs, are particularly sensitive to oxidative stress in glaucoma, which results in reduced levels of antioxidants within the neurons. 11,22
Immunolabeling for malondialdehyde (MDA) and nitrotyrosine (NT), markers of free radical formation and oxidative stress, was identified in acutely but not chronically glaucomatous retinas. 23 While the location of the highest level of immunostaining for NT was not described, the highest levels for MDA were seen in the nuclei of neurons, including RGCs, photoreceptors of the outer nuclear layer (ONL), and putative neurons of the inner nuclear layer (INL), suggesting an increase in oxidative stress in these cells.
Glutathione peroxidase (GP) activity was significantly lower in a group of “at-risk” dogs (Eurasier dogs) compared with control dogs. 19 Higher plasma taurine concentration and lower GP activity were observed in adult male Eurasier dogs, suggesting an increased vulnerability to oxidative stress with age in this group. Plasma concentrations of methionine and cysteine in this cohort of dogs were similar between groups. 19
Glutathione immunostaining was decreased in RGCs, INL neurons, and Müller cell processes in minimally damaged regions of glaucomatous retinas but was increased in severely damaged regions, suggesting oxidative stress may occur before the redistribution of glutamate. 23 The fact that Müller cells and astrocytes were less heavily labeled than neurons and the finding of glutathione depletion in Müller cell processes suggest that these neighboring glial cells may also undergo oxidative stress, but to a lesser degree than neurons, or that glutathione is upregulated more readily in these cells compared with neurons. In canine glaucoma, the majority of cells remaining in severely damaged regions of retinas appeared to be glial cells. These Müller cells and astrocytes contained high levels of both glutathione and taurine, suggesting they were resistant to further oxidative stress. 23
Circulating autoantibodies against heat shock proteins have been identified in humans with glaucoma, and exposure to these antibodies can induce apoptosis in cultured retinal ganglion cells. 89,90,95,96 Oxidative stress and TGF-β2 expression are both believed to be greater in glaucomatous versus healthy eyes and are both able to promote expression of heat shock protein 27. 6,89
It is important to note that these markers are indicators of the presence of oxidative stress. Some of these markers (eg, glutathione, GP) represent an adaptive or protective response to oxidative stress, while others (MDA, NT) are indicative of the damage arising from the oxidative stress.
Future Work
In clinical trials, “hard” or “true” end points are clinical events directly relevant to the patient. 61 For glaucoma, the true end points would be significant loss of vision with decrease in quality of vision or quality of life, or development of functional disability. 56 The development and use of validated surrogate end points will allow clinical trials and evaluation of interventions targeted at a much earlier stage of disease, with potential for greater effects, compared with what is achieved with current treatments. Surrogate end points have the potential to offer shorter, less expensive trials with reduced sample size requirements while allowing observation of a greater number of end points during follow-up than could be achieved with observation of a true end point. 56
While many studies have investigated the genetics of glaucoma by linkage studies, candidate gene reports, and GWA investigations, gene discovery has limited population effects, 38 and specific causal mutations generally differ between humans and dogs. In contrast, biochemical marker candidates have more similarities between human and canine glaucoma patients, suggesting common pathophysiological mechanisms. Therefore, progress in developing biochemical markers of glaucoma in humans will likely have translational benefits in veterinary medicine.
Where candidate biomarkers have been identified in the aqueous humor of glaucomatous compared with normal canine eyes, use of the sampling technique to obtain data from large populations has been precluded by the invasive nature of this sampling technique and the requirement for specialized training, which constrains the validation and verification of the findings. Body fluid biomarkers have the advantages of less invasive collection methods and thus a better acceptance by patients or animal owners. 104 Despite ease of blood sampling, identifying potential biomarkers in plasma or serum poses difficulties because of the high sample complexity, low concentrations of individual markers, and degradation of biomarkers. 10,16,57 Analysis of more proximal samples (eg, tears or aqueous humor) may present higher sensitivity and specificity for screening and monitoring glaucoma-related biomarkers. 92 The tear proteome is a rich source of diagnostic markers and easily sampled. 104 Studies in human glaucoma patients have detected alterations in tear samples related to antiglaucoma medications, 71,101 largely based on analysis of single targeted markers. 34,67,69,88 With the development of very sensitive proteomic techniques, repeated analyses are possible despite the small volume of tears that can be collected from a patient. 104 Development and progress in sample fractionation and mass spectrometry as a major analytical tool for large-scale protein studies have been a focus to help uncover the key molecular processes in human glaucoma. 92,93,104
The use of the dog as a model of glaucoma could provide another population for trials to validate and verify techniques and candidate biochemical markers in human glaucoma. Longitudinal studies involving dogs of at-risk breeds or colonies of purpose-bred dogs with an identified predisposition to glaucoma could potentially allow correlation of changes in biomarkers with stage of disease. 44
While many potential biomarkers have emerged from laboratory and pilot studies, validation in independent experiments or large-scale clinical studies are lacking. 38,72,104 Consideration must be given to the limitations of available techniques. 104 While gel-based techniques provide information (eg, molecular weight, presence of posttranslational protein modifications), they are unable to detect low-abundance proteins, while mass spectrometry techniques cover a larger proteome range compared with gel-based techniques. 104 Regardless of the technique used, standardized collection procedures, power analyses to determine appropriate samples sizes, and subsequent verification of findings are required. These limitations in biomarker development are likely associated with the multifactorial nature of the glaucomas themselves, as well as the variation in techniques and procedures available to investigators.
Conclusion
Challenges in glaucoma research include limitations in our understanding of disease mechanisms, limited knowledge of potential therapeutic targets, a paucity of preclinical models, and limitations in the ability to detect early development and progression of the disease. Novel tools to improve screening and diagnosis, as well as assess disease progression, are therefore required. The identification, validation, and appropriate use of biomarkers could help address this gap and could inform pathogenesis and identify therapeutic targets. This will require studies that identify candidate biomarkers, evaluate their association with disease, validate the biomarker (or more likely a biomarker set) for the targeted use (eg, early detection, disease progression), and show effects of interventions on both the biomarker and clinical outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
