Abstract
Spontaneous age-related lesions of laboratory rabbits are not well documented in the contemporary scientific literature. A retrospective study of diagnostic necropsies of 36 rabbits >2 years of age found a number of common lung lesions. Fibromuscular intimal hyperplasia affected medium and to a lesser extent large pulmonary arteries and was present to a variable extent in all 36 rabbits >2 years of age. The lesions were characterized by fragmentation and/or reduplication of the internal elastic lamina (IEL), proliferation of smoothelin+/alpha-smooth muscle actin (α-SMA)+/vimentin− smooth muscle cells and fewer smoothelin−/α-SMA+/vimentin+ myofibroblasts, and intimal deposition of collagen without thrombosis, embolism, or evidence of pulmonary hypertension. Pulmonary emphysema, present in 30/36 rabbits, was characterized by the loss of alveolar septa; most affected rabbits did not have clinical signs of respiratory disease. In 8/13 rabbits of the inbred EIII/JC audiogenic strain, we identified a unique syndrome of granulomatous pneumonia containing hyaline brown to gray, globular to ring-like acellular material that was Alcian blue and periodic acid-Schiff positive. The material was immunoreactive for surfactant protein-A and had the ultrastructural appearance of multilamellar vesicles, suggesting a genetic defect in surfactant metabolism. Additionally, we found small benign primary lung tumors (fibropapillomas, 5 rabbits) not previously described. Other findings included heterotopic bone (5 rabbits), subacute to chronic suppurative bronchopneumonia, pyogranulomatous pneumonia with plant material, and pulmonary artifacts from barbiturate euthanasia solution.
Although spontaneous background lesions of aging laboratory mice and rats are well documented and described, 7,9,19,23,31 there is less information on spontaneous lesions of aging laboratory rabbits. 14,15,21 In part this is because of the types of acute or relatively short-term studies for which rabbits are commonly used. In some cases where age-related lesions are described, such as spontaneous pulmonary emphysema, 42 other more recent references are contradictory, based apparently on the lack of additional confirmatory reporting. 4,8 Other spontaneous age-related lesions, such as unique intimal proliferations in the pulmonary arteries, are poorly documented in the English language literature, 20 and can resemble iatrogenic lesions caused by experimental manipulation. 27
While most rabbits used in research are outbred New Zealand White (NZW) or Dutch-belted stock, inbred rabbits strains do exist, including the audiogenic (EIII/JC) strain. The inbred III/J parent rabbit strain was originally developed at the Jackson Laboratory in 1932 by Dr Paul B. Sawin using NZW rabbits from Dr William Ernest Castle. The audiogenic (EIII/J) substrain was selected for a predisposition to sound-induced seizures by Dr Richard R. Fox in 1961. 13 The strain came to Penn State Hershey in 1988 from the National Cancer Institute. 37
Rabbits have been used as an induced experimental model for a wide variety of pulmonary conditions, including hypertension, 29 tuberculosis, 36 and aspiration pneumonia. 51 Rabbits have also been proposed as a model for aging research. 14 Therefore, it is important to document and describe common age-related background lesions in this species in order to avoid misinterpretation or confusion with induced lesions, particularly in chronic studies. The genesis of this study was the finding of proliferative pulmonary artery lesions and emphysema in aged male NZW rabbits exposed to collimated gamma knife treatment of the neck, and the need to exclude experimental treatment as a cause of these lesions. 27,33 Herein we report common nonneoplastic and neoplastic background lung lesions in laboratory rabbits, including the identification and initial description of a novel surfactant metabolism defect in the inbred EIII/JC (audiogenic) rabbit strain.
Materials and Methods
All rabbits were on experimental or breeding protocols approved by the Penn State Hershey Medical Center College of Medicine Institutional Animal Care and Use Committee. All the transgenic rabbits and inbred audiogenic (EIII/JC) rabbits were produced in our facility, and the outbred rabbits were purchased from various vendors as indicated in the demographics section of the results. The rabbits were cared for and housed in an Association for the Assessment and Accreditation of Laboratory Animal Care-accredited animal facility at Penn State Hershey, in compliance with the standards set forth in the eighth edition of the Guide for the Care and Use of Laboratory Animals (Guide) and the Animal Welfare Act regulations. Except for pre-weaned litters, adult rabbits were singly housed in stainless-steel cages (Versa-Pak T.1411 cage liner; Blue Ridge Tissue Corp., Lenoir, NC) with an automatic water system and fed a commercial pelleted feed (2031 Teklad High Fiber Rabbit Diet for rabbits without litters and 8630 Teklad 15% Rabbit Diet for rabbits with litters; Harlan Laboratories, Madison, WI). Rabbits were given Timothy hay (Oxbow Animal Health, Oxbow Enterprises, Inc., Murdock, NE) for enrichment. The animal housing rooms were maintained with constant environmental conditions including temperature at 19°C
All necropsy records, gross photographs, and lung histology slides for rabbits >2 years of age, from 1989 to 2015, were reviewed. Following gross necropsy, tissues were fixed in 10% neutral-buffered formalin (NBF) and paraffin embedded, and sections were cut at 6 µm and stained with hematoxylin and eosin (H&E). Additional sections were stained with Verhoeff van Gieson elastin, Masson’s trichrome, Movat’s pentachrome, periodic acid-Schiff (PAS), Gomori’s methenamine silver (GMS), Sirius red for amyloid, Congo red, Wright-Giemsa, Alcian blue-PAS, von Kossa for calcium, Perl’s iron, and Mowry’s modification of the colloidal iron stain for acid mucopolysaccharides.
For semiquantitative scoring of arterial lesions, lungs were scored from 0 (normal) to 4 (severe) based on the number and size of affected vessels and extent of proliferation. Grade 1 lesions affected only a few medium pulmonary arteries (100–250 μm diameter) and consisted of rare fragmentation of the internal elastic lamina (IEL) with scant eccentric collagen accumulation between the endothelium and IEL. Increasing grades of lesions were progressively more florid and numerous, affecting more arteries (including small and large), with abundant circumferential extracellular matrix, reduction in luminal caliber, and extensive splitting, reduplication, and fragmentation of the IEL. Emphysema lesions were semiquantitatively graded from 0 (normal) to 3 (severe) based on the number, size, and geographic extent of alveolar septal loss and airspace dilatation. Emphysema was differentiated from overinflation by the frequent presence of free-floating segments of alveolar walls (representing dilatation of the pores of Kuhn) in the former. All slides were graded by a single pathologist (T.K.C.). Results were compared by Student’s t-test.
For immunohistochemistry, sections were mounted on charged slides, deparaffinized, and heat-induced antigen retrieval was performed in citrate buffer. Endogenous peroxide was blocked, and slides were incubated for 1 hour at room temperature with antibodies to alpha-smooth muscle actin (α-SMA, M0851, 1:50; Dako), smoothelin (ab8969, 1:200; Abcam), vimentin (ab80667, 1:200; Abcam,), or surfactant protein-A (SPA, 1:2000). The goat anti-rabbit SPA antibody was a gift from Dr Jeffrey A. Whitsett (Cincinnati Children’s Hospital, Cincinnati, OH). The antibody was generated by vaccination using purified SPA isolated from bronchoalveolar lavage fluid of rabbits. All microscopic images were obtained with an Olympus BX51 microscope and DP71 digital camera using cellSens Standard 1.12 imaging software (Olympus America, Center Valley, PA).
For electron microscopy, the lung was deparaffinized, postfixed in glutaraldehyde followed by osmium and potassium ferrocyanide, and embedded in Embed 812 plastic (Electron Microscopy Sciences, Hatfield, PA). Thin (1 µm) sections were cut and stained with toluidine blue to confirm orientation before imaging by transmission electron microscopy. Thin sections (70–90 nm) were mounted on copper grids and stained with 2% aqueous uranyl acetate and lead citrate. The sections were examined in a JEOL JEM 1400 transmission electron microscope (JEOL JEM, JEOL USA, Peabody, MA; Orius SC 1000, Gatan, Inc., Pleasanton, CA). An Orius SC 1000 bottom mounted CCD camera was used to capture the images.
Results
Demographics
From 1989 to 2015, a total of 431 diagnostic rabbit necropsies were performed; of these, there were 36 aged rabbits (>2 years old), including 10 that were between approximately 2 and 3 years old (5 males and 5 females) and 26 that were >3 years old (18 males and 8 females; details are given in Supplemental Table S1). The average age was 4.1 years (range 1.9-6.7). Twelve rabbits were from the inbred audiogenic (EIII/JC) strain. Remaining rabbits were outbred NZW stocks from Robinson Services (RSI:NZW, n = 12) (Robinson Services, Inc., Mocksville, NC), Millbrook Farms (Mfr:NZW, n = 2) (Millbrook Farms, Amherst, MA), Charles River Laboratories (Crl:KBL[NZW], n = 1) (Charles River Laboratories, Wilmington, MA), Myrtle’s Rabbitry (Myr:NZW, n = 1) (Myrtle’s Rabbitry, Inc., Thompsons Station, TN), and Hazleton Dutchland (Dla:NZW, n = 1) (Hazleton Dutchland, Denver, PA, later became Covance Research Products, Inc., Denver, PA). Five rabbits were transgenic for a human HLA (major histocompatibility complex, class I, A) allele: 4 on a Covance outbred background (Crp:NZW-Tg[HLA-A2.1]) and 1 on the inbred audiogenic background (EIII-Tg[HLA-A2.1]/JC). 25,26 Two rabbits were transgenic for a mutated ras gene under the control of a cottontail rabbit papillomavirus promoter, also on a Covance outbred background (Crp:NZW-Tg[URR-EJras]) (Covance Research Products, Inc., Denver, PA). 35 One rabbit was a Dutch-belted cross born in-house. Nineteen of these rabbits, including all the transgenic and most EIII/JC, were retired breeders not subjected to any experimental manipulations or anesthetic events. Ten of the 12 RSI:NZW rabbits had received experimental elastase-induced aneurysm of the right carotid artery followed by repeated magnetic resonance imaging (MRI) with or without gamma knife treatment. 46 One EIII/JC had been experimentally inoculated with kappapapillomavirus-2 (Shope papillomavirus) 18 months prior to euthanasia; the papillomas had regressed spontaneously. The Myr:NZW rabbit was used for antibody production. With the exception of a single 5.5-year-old RSI:NZW buck, none of the rabbits had clinical signs related to the respiratory system prior to death.
Fibromuscular Intimal Hyperplasia of the Medium and Large Pulmonary Arteries
Hyperplastic arterial lesions were striking and consisted of intimal deposition of collagen and proliferation of spindle cells, predominantly affecting medium muscular pulmonary arteries in a segmental manner, often with compression of the lumen (Fig. 1). Large elastic arteries were less commonly affected, with rare involvement of small arteries or arterioles. Affected arteries were frequently tortuous and reduplicated. Proliferations were adluminal to the IEL, which was itself multifocally fragmented or split (Fig. 2). In medium and small arteries, proliferations were nearly always circumferential, while large artery proliferations were usually eccentric and cushion-like, often occurring near branch points. Depending upon the severity, there was accumulation of scant to abundant mature collagen with a variable, often moderate to high cellularity (Fig. 3). Rare masses were coarsely sclerotic and paucicellular. In occasional rabbits, there was obvious medial smooth muscle cell hyperplasia. No pulmonary arterial thrombosis/thromboembolism was present in any of the rabbits.
By immunohistochemistry, intimal proliferations included smoothelin+/α-SMA+/vimentin− smooth muscle cells and fewer smoothelin−/α-SMA+/vimentin+ myofibroblasts (Figs. 4–6). Interestingly, some of the smoothelin-positive intimal cells were also vimentin positive, whereas the fully differentiated contractile smooth muscle cells of the media were smoothelin+/α-SMA+/vimentin−.
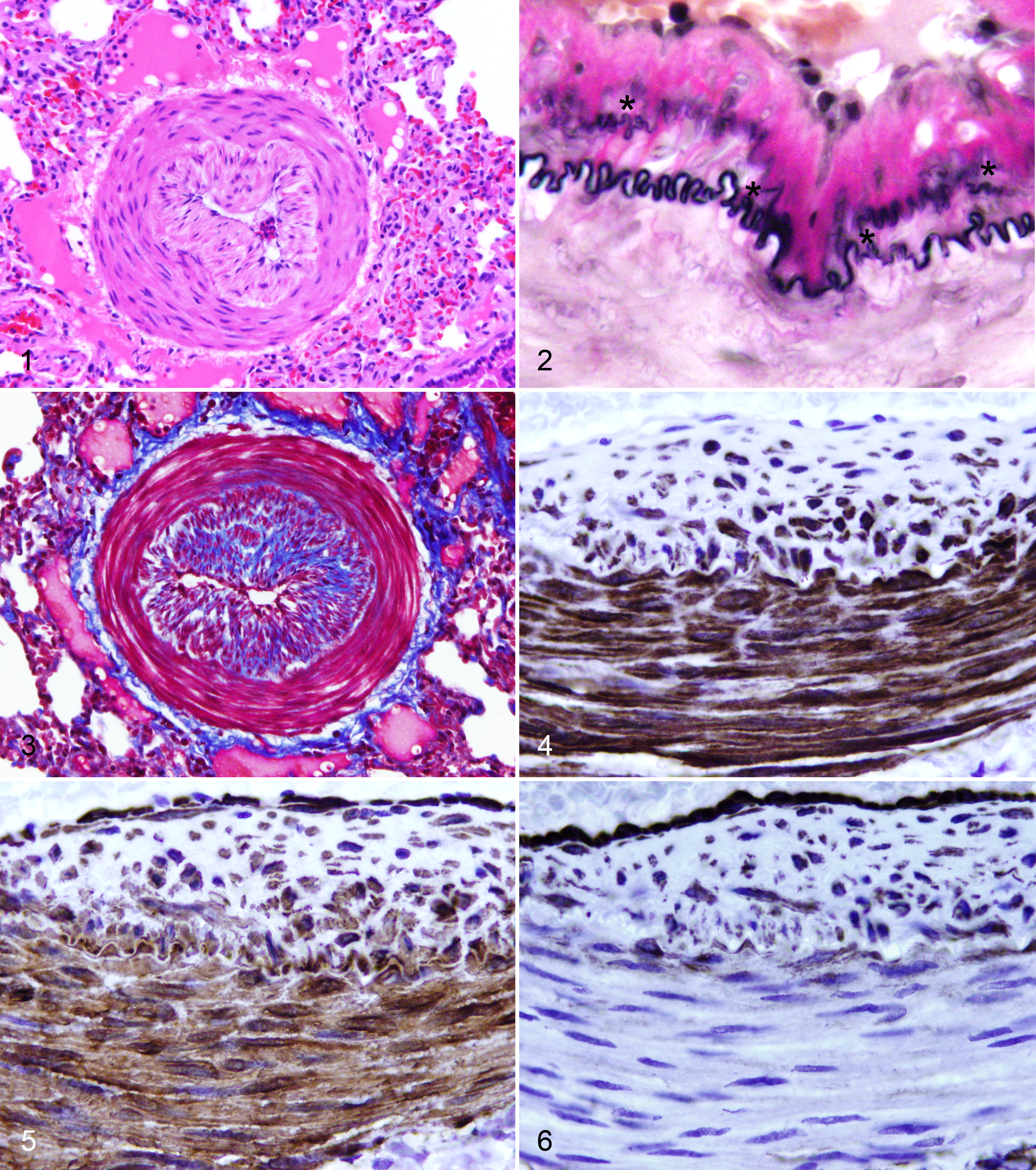
Lesions were scored on a semiquantitative (0–4) scale on the basis of the number of arteries affected and the severity of intimal proliferation. Lesions were present to some degree in all examined rabbits >2 years old, affecting both sexes and all strains and stocks. Although not present in rabbits of less than 1 year of age, there was only a weak correlation between age and intimal lesion score in rabbits older than 2 years (r2 = 0.27). There were no plexiform lesions, nor was there any significant smooth muscle hyperplasia in small arteries. By evaluating the ratio of ventricular weights, there was no evidence for compensatory right ventricular hypertrophy in affected rabbits as a consequence of these proliferations (Supplementary Fig S1). Similarly, comparison of heart weight and body weight was not significantly different between rabbits aged less than 2 years (n = 50) and rabbits aged greater than 3 years (n = 21, P = .38) or rabbits with severe pulmonary artery lesions only (n = 6, P = .61). One rabbit, a 3.9-year-old Crp: NZW-Tg(HLA-A2.1) breeder buck, was euthanatized due to right-sided congestive heart failure but had only mild pulmonary artery lesions.
Surfactant Pneumonia
Surfactant pneumonia was frequently grossly visible as irregular firm tan nodules in the cranioventral portions of lung lobes (Fig. 7). Histologically, lesions consisted of multifocal to coalescing intra-alveolar aggregates of large numbers of multinucleate giant cells, predominantly foreign body type, with epithelioid and foamy macrophages and few lymphocytes, macrophages, plasma cells, and nondegenerate heterophils (Figs. 8 and 9). Lesions occasionally extended into smaller bronchioles. There was frequent type II pneumocyte hyperplasia (adenomatosis) and minimal alveolar septal fibrosis. Free within alveoli and the cytoplasm of giant cells was characteristic hyaline brown to gray, globular to ring-like acellular material resembling so-called “Billups’ bodies.” The material was positive by Alcian blue and PAS stains (Fig. 10) as well as Mowry’s modification of colloidal iron and Movat’s pentachrome. The material was birefringent under polarized light; negative by Sirius red for amyloid, von Kossa, Perl’s iron, and GMS stains; and unstained by Wright-Giemsa and Masson’s trichrome stains. An antibody against SPA was reactive with the material (Fig. 11). Ultrastructurally, the material consisted of large concentric and whorling parallel lamellar arrays of electron dense material resembling the large extended form of multilamellar vesicles (Fig. 12) and were devoid of the complex quadratic lattice structure of tubular myelin. 1,24,49
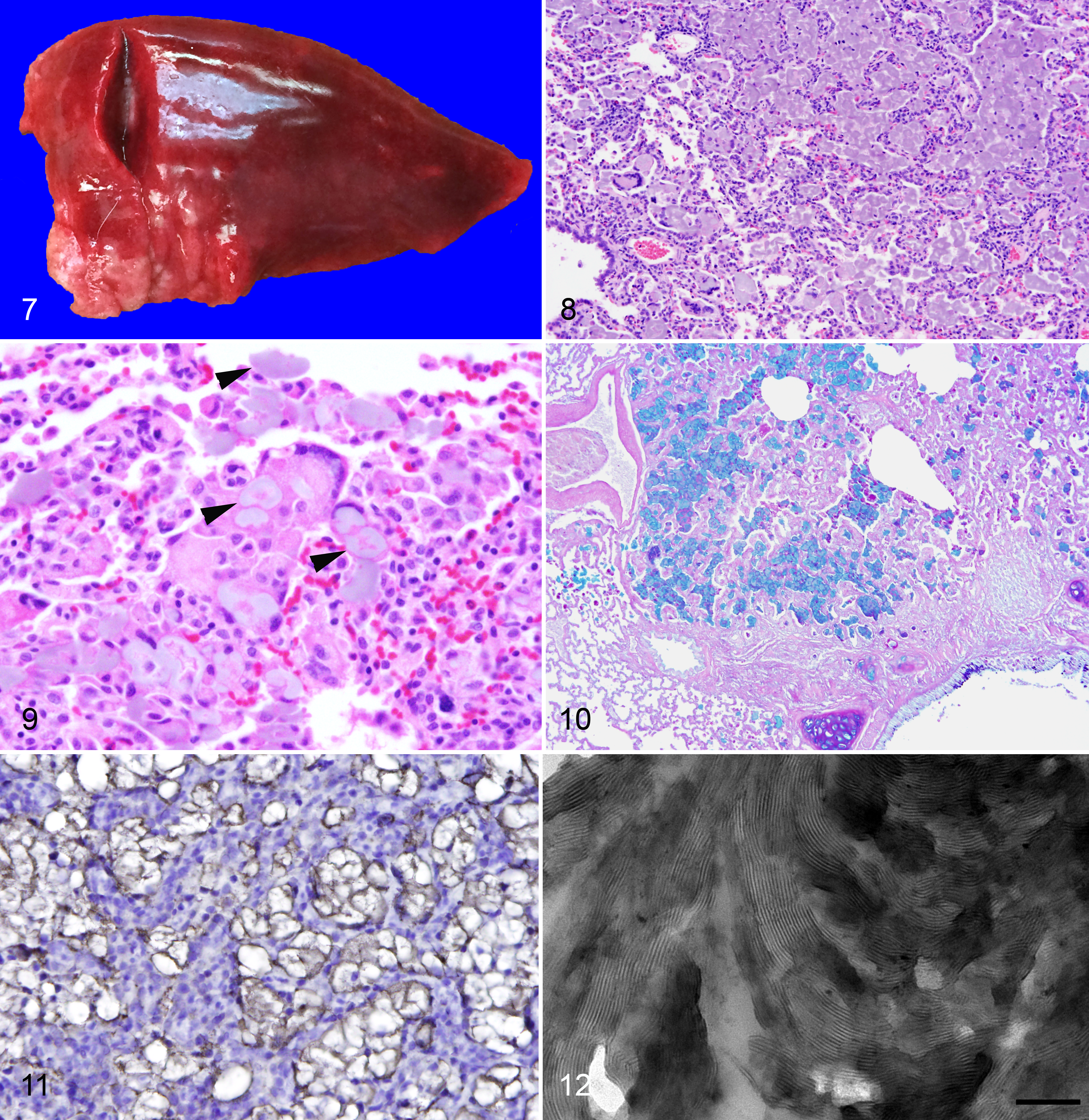
Surfactant pneumonia was present in 8/13 EIII/JC audiogenic rabbits >2 years of age (average 3.6 years, range 2–6; Supplemental Table S2) but was not identified in any outbred rabbits. Lesions were additionally present in 1 of 21 EIII/JC rabbits <2 years old (range 0.016–1.4 years): a single unmanipulated 1.3-year-old EIII-Tg(HLA-A2.1)/JC breeder doe. In all, 3 males and 6 females were affected. One rabbit, a 4.2-year-old doe, had concurrent but distinct chronic–active pyogranulomatous aspiration pneumonia with readily identifiable plant material (secondary to a large retropharyngeal abscess; not shown). None of the rabbits (including the doe with the abscess) were noted to be clinically dyspneic or in respiratory distress. While many of these rabbits had received oral medications at some point in their lives (usually fenbendazole and/or ponazuril), one rabbit had received no oral medications at any time. The same oral medications were given to many of the outbred rabbits as well.
Emphysema
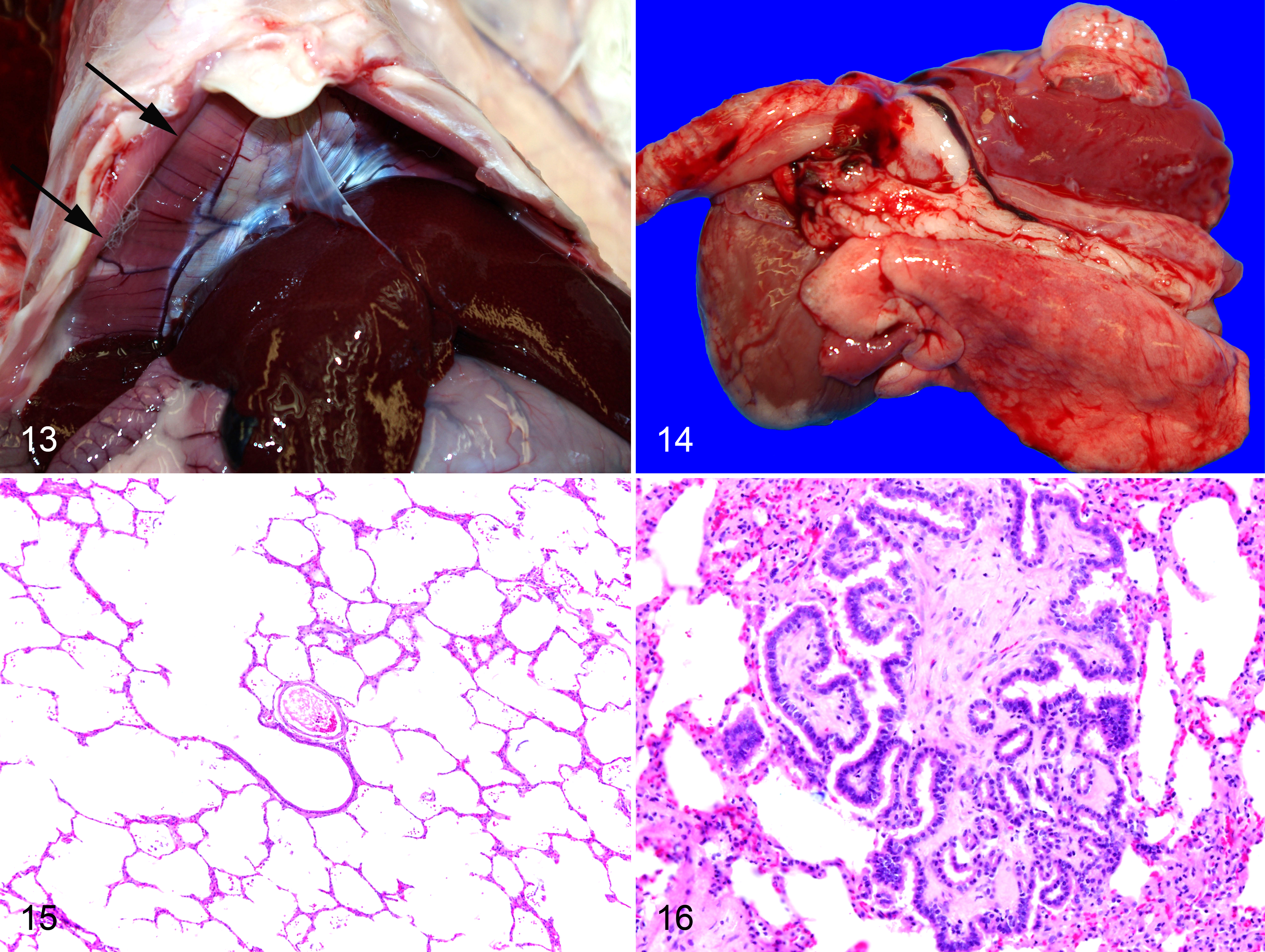
Emphysema was identified as the cause of morbidity in 1 rabbit, a 5.5-year-old RSI:NZW buck euthanatized due to sudden onset, clinically progressive tachypnea (64 rpm) with increased expiratory effort and abdominal breathing. At necropsy, there was a severe unilateral pneumothorax with emphysematous bullae and atelectasis of the ipsilateral lung (Figs. 13 and 14). There was no penetrating trauma, and the animal had not been anesthetized for experimental procedures since 13 days prior to euthanasia (MRI of carotid aneurysm under injectable anesthetic only; recovery on that date was uneventful). Emphysema was present to some degree in 30 of the 36 rabbits >2 years of age. Lesions were classified as mild (n = 9), moderate (n = 18), or severe (n = 3) based upon the degree and extent of loss of alveolar septa by histology (Fig. 15). No other rabbits had any clinical respiratory disease or dyspnea. None of the rabbits had airway obstruction that could result in overinflation. There was no correlation between age and emphysema score (r2 = 0.035). There was no significant difference in emphysema score between EIII/JC and outbred rabbits (P = .33).
Neoplasia
Metastatic neoplasia was present in the lungs of 2 EIII/JC rabbits. These included multiple large metastases of endometrial adenocarcinoma in a 2-year-old doe and rare intravascular tumor emboli from a large renal nephroblastoma in a 2.66-year-old breeder buck. Two additional does had endometrial adenocarcinomas with no observed pulmonary metastases.
Primary lung tumors were present in 5 (14%) of 36 rabbits. Tumors were incidental, not observed grossly, and appeared to develop within alveolar ducts. Tumor morphology was characteristic and consisted of a single small (<1 mm) expansile and noninvasive fibropapillomatous mass with a dense mature fibrous core covered by a single layer of crowded and hyperchromatic columnar epithelium (Fig. 16). Cells were rarely ciliated. Low numbers of plasma cells and macrophages were present in the stroma. Tumors did not occur in the context of an inflammatory or destructive process, such as organizing pneumonia. These tumors were found in the same 2.7-year-old EIII/JC buck with a concurrent nephroblastoma, a 2.1-year-old Mfr:NZW breeder doe, a 3.0-year-old EIII/JC breeder buck, a 3.2-year-old EIII-Tg(HLA-A2.1)/JC breeder doe, and a 5.5-year-old RSI:NZW buck with an induced carotid aneurysm. Only 1 of the EIII/JC rabbits had concurrent surfactant pneumonia, which was in an anatomically distinct location.
Other Lesions
Small nodules of mature and well-differentiated mineralized osteoid (heterotopic bone) were present in 5 rabbits, all >3 years old. Two EIII/JC breeder does (3.3 and 3.4 years old) and 1 EIII/J breeder buck (3.7 years old) had a subclinical mild subacute to chronic suppurative bronchopneumonia with gram-negative bacterial rods. Nasopharyngeal cultures were positive for Bordetella bronchiseptica in all 3 rabbits, and 1 was also Pasteurella multocida positive. A 2.1-year-old Crp:NZW-Tg(HLA-A2.1) breeder buck had a subacute pyogranulomatous pneumonia containing aspirated plant material, secondary to a large mandibular carcinoma. Artifact due to barbiturate euthanasia solution, causing perivascular edema, loss of detail and hypereosinophilia, 22 was rare and usually occurred when the dosage was given in excess.
Discussion
The genesis of this study was the finding of proliferative pulmonary artery lesions and emphysema in aged male NZW rabbits exposed to collimated gamma knife treatment of the neck, and the need to exclude experimental treatment as a cause of these lesions. 27,33 A comprehensive review of the institution’s archives demonstrated that these lesions were widespread in older rabbits of both sexes. Significant limitations of this study include the retrospective rather than prospective nature, variable rather than predetermined ages at death, multiple strains/stocks used, lack of emphysema as a primary end point (therefore, lungs were not insufflated with fixative to specified pressure), and variability in lung sampling (including location and number of samples) as a result of multiple personnel. While most rabbits were experimentally naive retired breeders, some did receive various experimental manipulations. These limitations may partially explain the inability to show strong correlations between age and lesion severity.
The cell types populating the arterial intimal lesions are of interest. In addition to smoothelin+/α-SMA+ and vimentin− contractile-type smooth muscle cells and vimentin+/α-SMA+/smoothelin− myofibroblast-like synthetic-type smooth muscle cells, there exists a population of triple-positive smooth muscle cells. These latter cells may represent a shift between the contractile and synthetic phenotypes. 44 Similar vimentin+/smoothelin+ cells have been observed in the neointima of saphenous veins used for coronary artery bypass grafts in humans. 28 The arterial lesions reported herein are distinct from those of pulmonary hypertension. The absence of plexiform lesions, the predominant medium and large artery distribution with only rare small artery involvement, and the absence of adventitial or interstitial fibrosis are inconsistent with pulmonary hypertension. 53 Further, there was no evidence of right ventricular hypertrophy in affected animals. However, invasive blood pressure measurements would be necessary to definitively exclude pulmonary hypertension as contributory.
There are no published descriptions of pulmonary artery proliferative lesions of rabbits in the English-language scientific literature, with only 2 limited and dated descriptions found in the non-English literature. 18,20 The paper by Grebenskaya (sometimes Grebenskaia) is the most comprehensive, evaluating a wide variety of ages (embryonic day 24 to 5 years old). The author noted splitting of the IEL and intimal collagen deposition as early as 3 months of age, progressing to eccentric cushion-like lesions in 3-year-old rabbits. However, there were no details on age-related incidence or the size of vessels affected, except to note that in some older animals large arteries are affected. In the English-language literature, there is only a single limited description of pulmonary arterial intimal “variations” in 20 male NZW rabbits, focusing on reduplication and splitting of the IEL of medium pulmonary arteries. 30 The authors did not report the ages of the rabbits used, only weights, and report no association with rabbit age.
Marini et al. noted inflammatory cell infiltrates in the walls of pulmonary arteries in association with the use of ketamine and xylazine anesthetics in rabbits, a change which could, with chronicity and repeated anesthetic episodes, potentially lead to intimal fibrosis (scarring). However, many of the rabbits examined in the current study were retired breeders that had never been anesthetized prior to the day of euthanasia. Interestingly, Marini et al. noted that, “[n]onocclusive, multifocal fibrous intimal plaques were observed in the large- and medium-caliber arteries of many rabbits,” but the age-group(s) in which the lesions were observed is not reported, and only the mean ages for the groups are provided. 32(p157)
The finding of accumulated surfactant concretions with a cranioventral (bronchogenic or aspiration) pattern inciting bland foreign-body-type inflammation in the audiogenic (EIII/JC) strain is quite unusual. Morphologically, histochemically, and ultrastructurally similar lung lesions with characteristic accumulated acellular material (Billups’ bodies) have been reported previously as pulmonary hyalinosis in dogs. In 1 study, lesions were predominant in brachycephalic breeds from urban settings, one-third of which had pulmonary or upper respiratory disease thought to predispose to aspiration. 5 The other study found lesions in 30 of 34 beagles exposed to airborne radon daughters (radioactive decay particles) with uranium ore dust and attributed the material to a degenerative response to injury with accumulation of endogenous secretions. 10 All but 1 of the affected EIII/JC described here were unmanipulated breeders, with no known airborne exposures (no history provided for the exception). Only 1 rabbit had upper airway disease potentially causing obstruction. We prefer the term “surfactant pneumonia” to hyalinosis because the former is more etiologically precise, while the latter shares a name with a histologically and biochemically distinct lesion in mice. 47
Alveolar type II epithelial cells synthesize and store surfactant phospholipids and proteins in secretory lamellar body vesicles containing surfactant in densely packed concentric bilayer membranes. The ABCA3 (ATP binding cassette subfamily A member 3) transporter on the lamellar body limiting membrane contributes to lamellar body transport of surfactant phospholipids. Lamellar bodies are exocytosed in a constitutive or regulated fashion under the control of catecholamines, purinoreceptor agonists, and cell stretching. 50 Fusion of the lamellar body limiting membrane with the cell surface membrane of alveolar type II epithelial cells releases lamellar body like dense surfactant particles that transform into contiguous lattices of tubular myelin and multilayered surface active film over the alveolar surface upon contact with air. The surfactant film at the air–liquid interface is critical for reducing surface tension and preventing alveolar collapse during breathing. 1,49 Secreted surfactant is continuously turned over through uptake and degradation by alveolar macrophages or reuptake via endocytosis by alveolar type II epithelial cells, leading to either degradation of spent surfactant components or recycling. Surfactant consists of approximately 80% phospholipid, 5%–10% cholesterol, and 5%–10% protein. Dipalmitoyl phosphatidyl choline is the most abundant and principal surface-active phospholipid of surfactant. The 4 surfactant proteins SPA, SPB, SPC, and SPD are differentially and critically involved in the biophysical phase transition from lamellar bodies to formation of the surface active film (SPB and SPC), tubular myelin ultrastructure (SPA and SPB), and turnover of spent surfactant (SPD). The occurrence of the surfactant lesion solely in our inbred rabbits strongly suggests an as yet unidentified genetic defect in surfactant production, ultrastructure, or degradation in the audiogenic strain. While no test breeding has been performed, a review of breeding records suggests an autosomal recessive inheritance with variable penetrance. A defect in the function of SPA or SPB would be a likely target based on the ultrastructural appearance of the material, although the histologic appearance of the lesions herein are clearly distinguishable from pulmonary alveolar proteinosis in people due to mutations in SFTPB (surfactant protein B), SFTPC (surfactant protein C), and ABCA3, which disrupt lamellar body biogenesis and formation of a patent surfactant film; or mutations in CSF2, which disrupts alveolar macrophage differentiation and degradation of surfactant. 12,24,50 The multilamellar vesicles devoid of tubular myelin, however, bear resemblance to the onion-like multilamellar vesicles (and emphysema) that characterize Adgrf5 (adhesion G protein-coupled receptor F5) (Gpr116)-deficient mice, which were associated with abnormal interaction of SPA with surfactant phospholipids and reduced reuptake of surfactant by alveolar type II epithelial cells. 2,52 Another intriguing possibility is BACH2 (BTB domain and CNC homologue 2), which is associated with defects in surfactant metabolism as well as epilepsy. 34,45 The actual incidence of the lesion may be significantly higher than reported here, due to sampling issues noted previously. The Penn State Hershey audiogenic rabbit colony was started with just 2 breeding pairs. Surfactant pneumonia lesions were present in 1 of the founder breeding females used to establish this colony (other founders not necropsied), as well as 1 of the offspring from the other founder pair. Because of the age-dependent nature of the lesions and a historically higher incidence of bacterial pneumonia, this entity may not have been noted previously in this strain. Alternatively, it may have been a more recent spontaneous mutation with a founder effect, considering the low number of animals used to start the PSUHMC colony. It is interesting to note that the only 3 rabbits with histologically identified subclinical bacterial bronchopneumonia in this study were audiogenic, suggesting that the abnormal surfactant metabolism may have functional consequences related to the clearance of opportunistic bacterial colonization. Further investigations into the exact biochemical defect(s) in surfactant metabolism within this strain are ongoing.
Strawbridge was the first to report spontaneous pulmonary emphysema in rabbits, and the aim of this study was not to recapitulate his detailed descriptions. 42 Rather, this study confirms that pulmonary emphysema is indeed a common finding in aged rabbits, with occasional clinical significance. The major reference textbooks on pathology of the laboratory rabbit either omit emphysema as a spontaneous disease 6,38 or acknowledge the findings of Strawbridge but dismiss them as being an historical anomaly or clinically irrelevant. 4,8 Although Strawbridge noted a marked increase in the incidence of emphysema in rabbits >2.5 years (62%) versus rabbits 6–18 months old (19%–39%), the present study only examined rabbits >2 years old and found no correlation between lesion severity and age in this group, likely due to study limitations. However, emphysema is rarely noted in younger rabbits. Although rabbits are reported to be highly sensitive to overdistension of alveoli during ventilation, 17 we excluded barotrauma as the cause of the lesions observed. Only the RSI:NZW rabbits with iatrogenic carotid aneurysm were anesthetized using positive pressure ventilation and only during the initial creation of aneurysm (occurring from 3.9 to 6.4 years prior to necropsy); all subsequent anesthetic events for imaging and gamma knife treatment used injectable agents only, with no intubation or ventilation. Two RSI:NZW rabbits necropsied within a week of ventilator use had only very mild lesions consistent with barotrauma (data not shown). Further, moderate to severe emphysema was present in multiple experimentally naive retired breeders that had never been anesthetized.
Primary pulmonary neoplasia is reported to be rare in rabbits. In the scientific literature, there are only 4 reports of spontaneous primary lung tumors in rabbits, all malignant. 3,11,39,41,43,48 Primary lung tumors present in this series were small, benign, and incidental and therefore potentially easily missed during routine necropsy. Although Shope papillomavirus (kappapapillomavirus-2) is used in this institution, only a single rabbit in this study was exposed to that agent and the rabbit did not develop a lung tumor. In contrast to the pulmonary fibropapillomas of rabbits described in this study, papillary lung tumors of mice and rats (papilloma of terminal bronchiole and papillary bronchioloalveolar adenoma) have characteristically delicate stroma. 40 Papillary adenomas (bronchiolar papillomas) of man may have focal sclerosis but are not generally diffusely sclerotic. 16 By immunohistochemistry, those tumors are positive for thyroid transcription factor-1 and surfactant apoproteins A and B. Because the tumors in this series were so small and lost on recuts, immunohistochemistry was not performed.
In summary, the goal of this study was to document and describe common spontaneous background lesions of aging rabbits. Those utilizing rabbits in chronic research studies must be cognizant of these age-related lesions in order to avoid inadvertently attributing these lesions to experimental manipulation.
Footnotes
Acknowledgments
Dr Milena Bogunovic translated the Grebenskaya paper. Thanks to Ellen Mullady and Gretchen Snavely for histologic specimen preparation. We are grateful for assistance from Roland Myers with electron microscopy specimen preparation and photography. Dr Richard R. Fox and Karla K. Balogh provided historical information for the audiogenic strain.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Rabbit breeding colonies supported by NIH grant 2R01CA047622 (ND Christensen). Dr. Chroneos is supported by NIH grant HL128746.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
