Abstract
A juvenile dwarf rabbit (Oryctolagus cuniculus) with clinical signs of dyspnea and suspected ascites was submitted for necropsy. The main macroscopic findings were a watery red pleural effusion and some whitish striated foci in the lungs. In addition, there were multifocal scars in the cortex of the kidneys. The histologic examination of the lungs showed a severe granulomatous pneumonia with detection of acid-fast bacilli, in the kidneys, an interstitial chronic lymphoplasmacellular nephritis with interstitial fibrosis, and in the brain, a multifocal granulomatous and partly necrotizing encephalitis with detection of spores, suggestive of encephalitozoonosis. In the lungs, Mycobacterium genavense was verified by polymerase chain reaction and 16S ribosomal RNA gene sequencing. To our knowledge, this is the first report of an M. genavense infection in a rabbit, with the lungs being the only affected organ. Therefore, an aerogen infection seems to be the most contemplable way of infection.
Mycobacterium genavense is a member of the so-called mycobacteria other than tuberculosis (MOTT), and has been named after the source of the first isolate (Geneva/Switzerland, lat. Genava). 2,3,8 These mycobacteria are small, acid-fast coccobacilli, that do not grow on solid medium, and show fastidious growth on some liquid media (e.g., BACTEC 13A, Middlebrook 7H12). 2
Mycobacterium genavense has been identified in 1992 (taxonomic identification in 1993) as the causative agent of disseminated mycobacteriosis in patients with human immunodeficiency virus (HIV). The main clinical findings in infected human patients are fever, diarrhea, and massive weight loss. 2,3,7,8 At present, infections in animals have been described in many birds, 1 dog, 1 cat, 2 aged ferrets, and 1 monkey. 9–11,13,14,16–18
The affected organs in humans and animals are the intestine, liver, spleen, lymph nodes, bone marrow, and sometimes also the skin, kidneys, lungs, and brain. Histologically, foamy macrophages and epithelioid cells, containing acid-fast intracytoplasmic bacilli, are detected in different organs (discussed previously). In a few cases, the appearance of multinucleated giant cells has been described. 2,3,7–11,13,14,16–18 This case report describes a dwarf rabbit with granulomatous pneumonia, caused by M. genavense, with many epithelioid and multinucleated giant cells in the lungs.
A juvenile male dwarf rabbit was assigned for necropsy to the Institute of Veterinary Pathology, Ludwig-Maximilians-University Munich, exhibiting clinical signs of dyspnea and suspected ascites. No information about the housing, health status, or medication has been submitted. Specimens of various organs were embedded in paraffin or plastic, and stained with hematoxylin and eosin, Giemsa (paraffin and plastic), and Ziehl-Neelsen (paraffin). Paraffin-embedded organs, showing granulomatous inflammation, were analyzed using polymerase chain reaction (PCR). After dewaxing with Auto-dewaxer (Invitrogen, Karlsruhe, Germany) and extraction of total genomic DNA using the QiaAmp Mini Kit (Qiagen, Hilden, Germany), a 510-bp fragment at the 5′-end of the 16S ribosomal RNA gene was amplified with Mycobacterium genus-specific primers. 4 The sequence of the amplified fragment was determined on an Applied Biosystems 310 Genetic analyzer using BigDye v1.1 sequencing kit and the obtained 16S recombinant DNA sequence data were analyzed with the SmartGene IDNS system (http://www.smartgene.com, SmartGene Services, Lausanne, Switzerland).
The main macroscopic findings were a watery red pleural effusion and some whitish striated foci in the lungs (Fig. 1). Furthermore, multifocal scars were diagnosed in the cortex of the kidneys.

Lung; dwarf rabbit. Granulomatous pneumonia: multifocal light-colored striated foci all over the lung (image of the right lobe).
Histologically, the lungs showed severe intra-alveolar infiltration with foamy macrophages and multinucleated giant cells, accompanied by mild-to-moderate lymphocellular and plasmacellular interstitial infiltration (Fig. 2). Using Ziehl-Neelsen staining, a few acid-fast bacilli were visible in the cytoplasm of macrophages, giant cells, and in the interstitium (Fig. 3). These acid-fast bacilli were identified as M. genavense, using PCR. Database comparison on the SmartGene IDNS System (Smart Gene Services) clearly identified the sequence as belonging to M. genavense with complete sequence identity to GenBank accession number X60070, except for 2 nucleotide ambiguities present in the corresponding record of the public GenBank database.

Lung; dwarf rabbit. Granulomatous pneumonia: severe intra-alveolar infiltration with inflammatory cells (foamy macrophages and multinucleated giant cells of Langhans-type), only a few partially ventilated alveolae; fibromuscular thickening of the alveolar septa and alveolar lining cell hyperplasia, mild interstitial lymphocellular and plasmacellular infiltration. HE. Bar = 100 µm.
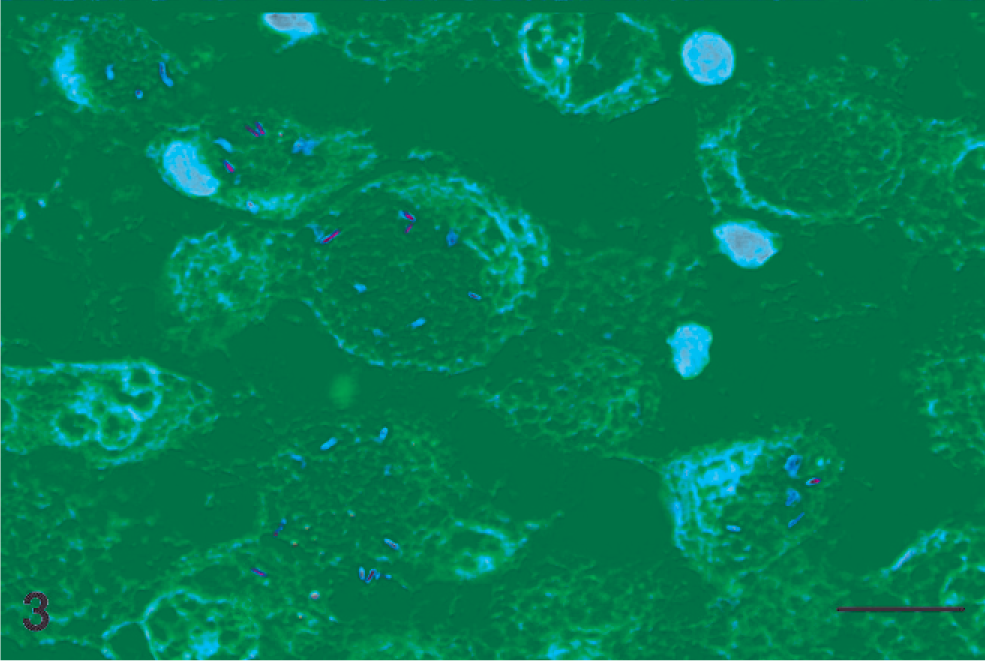
Lung; dwarf rabbit. Few acid-fast bacilli in the cytoplasm of macrophages, epithelioid cells, and giant cells; in addition, some lymphocytes. Ziehl-Neelsen. Bar = 10 µm.
In addition, a moderate interstitial lymphocellular and plasmacellular nephritis with interstitial fibrosis (acid-fast negative) and a granulomatous and partly necrotizing encephalitis with detection of spores were diagnosed, and interpreted as Encephalitozoon cuniculi-associated lesions. A negative acid-fast stain of both organs, and a negative PCR of the brain, excluding a disseminated form of mycobacteriosis, supported this assumption. Furthermore, the type of inflammation in the kidneys was chronic lymphocellular and plasmacellular without any epithelioid or multinucleated cells, which are typical for a mycobacteriosis, especially the M. genavense infection. Therefore, a possible disseminated form of mycobacteriosis has been excluded.
Mycobacterium infections are reported to be very rare in rabbits, and naturally occurring mycobacterium infections include M. bovis, M. avium, and, in juvenile animals, M. tuberculosis. 1,15 Furthermore, rabbits are used as animal model for human infectious diseases caused by these mycobacterium species. 5,6 To our knowledge, this is the first report of a M. genavense infection in a rabbit. A M. genavense infection is described in the literature as a disseminated mycobacteriosis in humans and animals with granulomatous inflammation of several organs (intestine, liver, spleen, lymph nodes, bone marrow, skin, kidneys, lungs, and brain). 2,3,7–11,13,14,16–18 However, the lungs were the only affected organs in our case, whereas the abdominal organs and lymphatic tissues were without pathologic findings, consistent with M. genavense infection. Besides disseminated forms of nontuberculous mycobacterial infections, exclusively chronic pulmonary infections are described, commonly associated with M. kansasii or M. xenopi. Affected individuals show clinical signs of coughing, but also fever, weight loss, and, as in the present case, dyspnea. 12 In comparison with other cases, the amount of acid-fast bacilli was rather low in this dwarf rabbit. 3,7–11,13,14,16–18
The route of infection with M. genavense may vary. M. genavense seems to be a ubiquitous-occurring pathogen, and infections in animals by drinking water are mentioned in the literature. 16 In the present case, the granulomatous pneumonia makes an aerogen infection the most contemplable way of infection. Unfortunately, we have no information about the history of the rabbit, including other diseases with impaired immune response or use of immunosuppressive drugs, which could have promoted a mycobacteriosis. The question remains, whether or not the encephalitozoonosis might have weakened the rabbit's immune system, and promoted the observed M. genavense infection. In humans, cases of mycobacteriosis in immunocompromised hosts (e.g., patients with HIV, patients with lung pathology or certain body habiti) are described, but it is mentioned also that pulmonary mycobacteriosis typically affects patients who are not severely immunocompromised or even are immunocompetent. 12 Last, the potential zoonotic risk should be kept in mind. Infected pets may provide an environmental source for immunocompromised hosts, especially patients with AIDS and patients on immunosuppressive therapy or, as mentioned previously, immunocompetent people. Infections from man-to-animal should be considered, and cannot be completely excluded in the present case, because of the unknown health status of the rabbit's owner.
In conclusion, an M. genavense infection should be included in the differential diagnosis of disseminated or organ-specific granulomatous inflammation, and immunocompromised hosts and immunocompetent individuals are not aware of this disease.
Footnotes
Acknowledgement
We are grateful to Dr. Nadja Herbach for manuscript revision.
