Abstract
Enterococcal spondylitis (ES) is a disease of commercial broiler chickens, with a worldwide distribution. Symmetrical hind limb paralysis typical of ES results from infection of the free thoracic vertebra (FTV) by pathogenic strains of Enterococcus cecorum . To determine the pathogenesis of ES, birds with natural and experimental ES were studied over time. In natural disease, case birds (n = 150) from an affected farm and control birds (n = 100) from an unaffected farm were evaluated at weeks 1–6. In control birds, intestinal colonization by E. cecorum began at week 3. In case birds, E. cecorum was detected in intestine and spleen at week 1, followed by infection of the FTV beginning at week 3. E. cecorum isolates recovered from intestine, spleen, and FTV of case birds had matching genotypes, confirming that intestinal colonization with pathogenic strains precedes bacteremia and infection of the FTV. Clinical intestinal disease was not required for E. cecorum bacteremia. In 1- to 3-week-old case birds, pathogenic E. cecorum was observed within osteochondrosis dissecans (OCD) lesions in the FTV. To determine whether OCD of the FTV was a risk factor for ES, 214 birds were orally infected with E. cecorum, and the FTV was evaluated histologically at weeks 1–7. Birds without cartilage clefts of OCD in the FTV did not develop ES; while birds with OCD scores ≥3 were susceptible to lesion development. These findings suggest that intestinal colonization, bacteremia, and OCD of the FTV in early life are crucial to the pathogenesis of ES.
Keywords
Enterococcus cecorum is a gram-positive bacterial coccus that was first isolated from the intestinal tract of chickens as Streptococcus cecorum and later reclassified as Enterococcus. 15,38 Until recently, the established role of E. cecorum in health and disease was as a beneficial, lactic acid producing, enteric commensal of adult poultry. In an early definitive work characterizing the gut microbiota of poultry, Devriese et al reported that E. cecorum was first detectable in the gut of chickens at week 3 of life and progressed to become a dominant component of the gut microbiota of healthy chickens by 12 weeks of age. 15,17 However, in 2002, epidemics of osteomyelitis of the free thoracic vertebra (FTV) and femoral head due to the pathogenic strains of E. cecorum occurred in broiler and broiler breeder flocks (meat chickens). 13,39 Additionally, pathogenic E. cecorum causes septic disease in Pekin ducklings and racing pigeons. 23,24
The most common and recognizable clinical disease caused by pathogenic E. cecorum occurs in broiler chickens and is called enterococcal spondylitis (ES; also known as vertebral osteoarthritis [VOA] or “kinky back”). 1 –3,6,8,12,13,21,25,27,35,36,39 Unfortunately, one of the most frequently utilized common names for ES, kinky back, also refers to a developmental disease of chickens with a genetic basis, in which a malformation of the spinal column (spondylolisthesis) results in compression of the spinal cord and clinical signs that overlap with ES. 13,39 Outbreaks of ES have a typical presentation where symmetrical hind limb paraparesis or paralysis develops in affected birds at 5–8 weeks of age and results in 5%–15% mortality. 3,8,28 Paralysis results from compression of the thoracolumbar spinal cord by an inflammatory mass that develops due to uncontrolled infection of the FTV with pathogenic E. cecorum. 8,36 Some birds also have infection of the femoral head. 7,30 However, the most striking feature of ES is that the FTV and its articulations are the only location in the vertebral column where lesions develop.
The FTV has several unique anatomic features that may predispose this location to disease. For example, the FTV is the only vertebra in the thoracolumbar vertebral column of chickens that has weight-bearing articulations. 5,40 Cranial to the FTV is the notarium, which is a fusion of the thoracic vertebrae with synchondroses rather than the synovial articulations and intervertebral discs like those in mammals. Caudal to the FTV is the synsacrum which is a similar fusion of the caudal thoracic vertebra, lumbosacral vertebrae, and pelvic bones. The FTV is also under considerable mechanical stress as it bears and transfers the majority of the body weight to the hips and hind limbs. These unique anatomic features might predispose the FTV to infection with pathogenic E. cecorum. However, when birds are clinically affected by ES, the architecture of the FTV is effaced, and the early lesions cannot be studied. 8,36 Therefore, a primary goal of this work was to identify predisposing lesions in the FTV in the early, preclinical stages of infection.
In addition to host factors that influence susceptibility to disease, strains of E. cecorum causing outbreaks of ES are more virulent than commensal E. cecorum. Using molecular epidemiologic approaches, outbreaks of ES have been shown to be caused by certain genetically related strains of E. cecorum. 6 –8,25,37 Pathogenic genotypes of E. cecorum are significantly more virulent for chick embryos than commensal E. cecorum genotypes. 7 At the whole genome level, pathogenic E. cecorum have several unique and conserved features compared to commensal E. cecorum. Pathogenic E. cecorum are clonal at high resolution and have significantly smaller genomes with higher Guanine and Cytosine content. 9 Phylogenetic analysis of 5 conserved genes used to speciate enterococci revealed divergence between commensal and pathogenic E. cecorum genomes, suggesting pathogenic E. cecorum might be considered a subspecies. 9 Pathogenic E. cecorum contains a Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) and Cas (CRISPR-associated genes) locus that is similar to those of streptococci but unique among enterococci. 9 Finally, potential mediators of virulence were found to be conserved in pathogenic strains, including genes encoding predicted collagen-binding proteins, a capsular locus, and orthologs to the enterococcal polysaccharide antigen of E. faecalis and E. faecium. 9
Despite recent advances in understanding the genetic basis for increased virulence in pathogenic E. cecorum, little is known about critical steps in the pathogenesis of ES. The source of pathogenic E. cecorum is unknown. Vertical transmission of pathogenic strains from broiler breeder to broiler flocks has not been definitively demonstrated. 25,27,35 Environmental contamination by pathogenic E. cecorum may be responsible for repeated outbreaks on affected farms; however, persistence of E. cecorum in the environment between flocks is unproven. Regardless of the source of pathogenic E. cecorum, gut colonization and bacteremia are likely the key steps in the pathogenesis of ES; however, the timing of these steps in the preclinical period is unknown. In addition, several authors have speculated that clinical intestinal disease due to co-infection is required for bacteremia and ES. 1,2,28 Finally, it remains unclear why the FTV is the only location in the vertebral column that is affected in ES.
Therefore, the objectives of this study were to (1) assess the environment as a source of pathogenic E. cecorum, (2) document critical steps in the pathogenesis of ES including gut colonization and bacteremia, (3) identify lesions in either the intestines or FTV which are present prior to ES in natural disease, and (4) confirm the findings of natural infection in an experimental model of ES.
Materials and Methods
A commercial broiler chicken farm experiencing natural disease and a control farm were prospectively sampled over time. Additionally, broiler chickens were experimentally infected with pathogenic E. cecorum and the resulting disease was studied.
Natural Infection
Typically, outbreaks of ES occur sporadically in commercial broiler operations; however, some farms experience repeated outbreaks of ES. To minimize potential variability in nutritional or management practices among farms, a single North Carolina (NC) integrator assisted in identifying the case and control farms.
Identifying case and control farms
Industry partners were asked to identify candidate case/control farms (n = 3 each), with similar basic farm structure and management practices as follows: farms located within 2 hours of Raleigh, NC, that have similar feeders, watering systems, lighting, and stocking densities; same feed formulations provided by the integrator; and similar clean out and litter treatment protocols. Broiler chicks (Hubbard male crossed with Ross 708 female) for both case and control farms originated from company-owned breeder flocks and hatcheries with the same vaccination and coccidia control programs.
To confirm the case/control status, candidate farms were evaluated for ES when birds were 6–8 weeks of age using a combination of an owner-survey and a pre-study on-farm evaluation. For this prestudy on-farm evaluation, flocks in each house were examined individually by a board-certified clinical poultry veterinarian with expertise in ES (M.P.M.). Eight birds per farm (divided evenly among houses) with symmetrical paralysis consistent with ES were selected for necropsy. If symmetrical paralysis was not identified, birds with marked to severe lameness were selected. Necropsies were done by board-certified veterinary pathologists (L.B.B. and H.J.B.). Samples of spine surrounding the FTV were collected in 10% neutral-buffered formalin (NBF) for histopathology to confirm the presence or absence of microscopic lesions. If gross lesions were present in the spine, they were aseptically opened, sampled using sterile swabs, and cultures were taken using swabs to confirm the presence of E. cecorum. 8 Environmental samples including feed, water, litter, and swabs of dust on air vents were collected and cultured for E. cecorum: all recovered E. cecorum isolates were genotyped (see below).
From the 6 candidate farms, 1 case and 1 control farms were selected for longitudinal study based on a priori requirements: the selected case farm would have a history of repeated outbreaks of ES (as reported by the integrator) and have an active outbreak of ES confirmed at the time of the on-farm evaluation. In contrast, the control farm would never have experienced an outbreak of ES (as reported by the integrator) and would not have birds identified with lesions of ES in any house during the on-farm evaluation.
The case farm had 3 houses, which were routinely stocked with 12,400 broilers each. The broiler strain grown on this farm was a cross of Hubbard male and Ross 708 female. On the case farm, outbreaks of ES had occurred in the previous 3 flocks and an outbreak with characteristic clinical signs and lesions of ES was ongoing during the prestudy on-farm evaluation (Figs. 1 and 2). Pure cultures of pathogenic E. cecorum were isolated from lesions sampled during the confirmation trip.
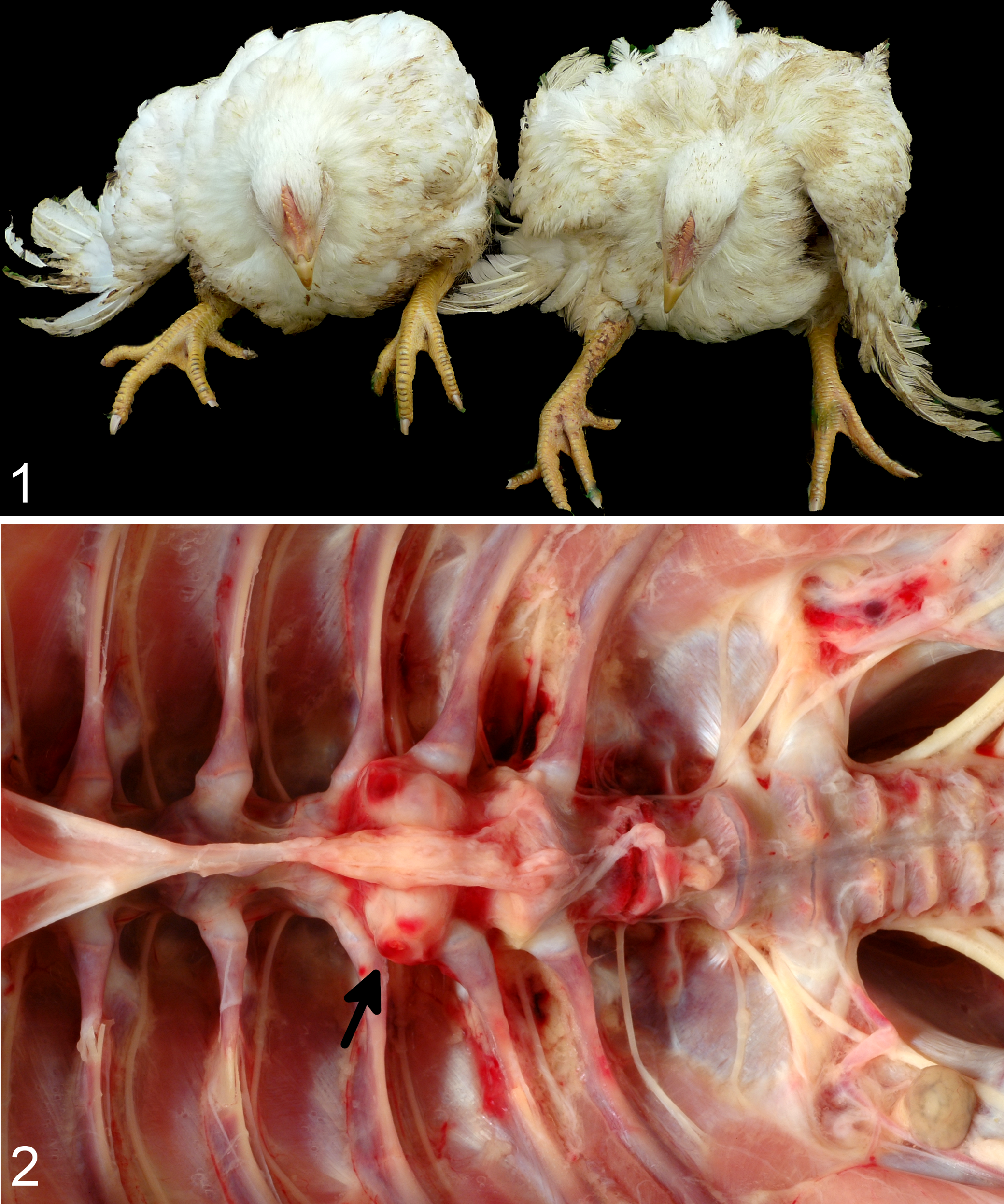
The control farm had 2 houses containing 16 500 birds each. The same broiler strain as the case farm (Hubbard male crossed with Ross 708 female) was grown on this farm. On the control farm, no ES was observed. Sporadic lameness was detected on the control farm due to vertebral column developmental anomalies (n = 1) and splay leg (n = 7).
Environmental sampling
To evaluate the internal broiler house environment as a potential source of pathogenic E. cecorum, samples of feed, water, litter, and air vent swabs from each house were cultured for pathogenic E. cecorum using the method described below. To ensure thorough sampling of the internal broiler house environment, each house was evenly divided into 6 separate sample areas. From each of the 6 areas, 3 tablespoons of litter were collected and pooled in a sterile Whirl-Pak bag (Fischer Scientific, Waltham, MA). In a similar manner, feed was collected from trays or automatic feeders. For water samples, a total of 5 mL of water was collected from 3 separate nipples in each of the 6 sections. Air vents (which) at both ends of the houses were swabbed inside the house (Stuart swabs; BD, Franklin Lakes, NJ). Environmental sampling was done 3 times during the study: (1) during prestudy on-farm evaluation, (2) prior to the placement of the study flocks, and (3) following the removal of case/control flocks for processing.
Longitudinal sampling
For both farms, birds were evaluated immediately prior to placement and weekly during weeks 1–4 of the growing period. For each time point, 30 case birds (10 birds from 3 houses) and 20 control birds (10 birds from 2 houses) with clinical signs of lameness were preferentially selected for evaluation. Samples were obtained for E. cecorum culture (intestines, spleens, and FTV lesions if present) and histopathology (intestines and FTV). Due to an ES outbreak on the case farm during this period, additional sampling for E. cecorum culture (intestines, spleens, and FTV lesions if present) was done at weeks 5 and 6. To confirm ES-negative status, the control birds were evaluated throughout the entire growing period (weeks 5–8) for clinical signs and gross lesions of ES.
Histopathology
For each bird, approximately 1 cm samples of duodenum, jejunum, ileum, ceca, and colon were collected into NBF. Following fixation, cross sections of intestinal samples were processed into paraffin, embedded together, and sections were stained with hematoxylin and eosin (HE).
All slides were randomized, recoded, and evaluated for lesions by a board-certified veterinary pathologist (L.B.B.) who was blinded to case/control groups. Intestinal sections of 150 case birds and 100 control birds were scored for villous blunting, inflammation, and presence of attaching bacteria or coccidia. Villous blunting was identified by death or degeneration of enterocytes at the villous tip or shortening of villi. Villous blunting was scored as follows: 0 = normal; 1 = minimal (less than 5% loss in villous height); 2 = mild (5%–10% loss in villous height); 3 = moderate (up to 25% loss in villous height); and 4 = severe (greater than 25% loss in villous height). Inflammatory infiltrates were scored as 0 = normal or minimal to mild lymphoplasmacytic infiltrates confined to the lamina propria; 1 = mildly increased numbers of lymphocytes, plasma cells, and scattered aggregates of 3–5 heterophils per 20× field; 2 = moderately increased lymphoplasmacytic infiltrates and frequent aggregates of >5 heterophils per 20× field confined to the lamina propria; 3 = marked lymphoplasmacytic and heterophilic inflammation that extended into the submucosa; and 4 = inflammation that extended into the muscular layers. Sections were evaluated for bacteria intimately associated with the microvillous brush border and scored for prevalence as 0 = no attached bacteria observed; 1 = rare (1–2) villi in 1 section with attached bacteria; 2 = a few (3–5) villi in 1 or more sections with attached bacteria; and 3 = numerous (>5) villi in 1 or more sections with attached bacteria. Presence of coccidia was scored as 0 = no coccidia observed; 1 = rare coccidia in a single section; 2 = few coccidia in multiple sections; and 3 = numerous coccidia in multiple sections.
Similarly, at each time point, the region including the FTV was collected to include the cranial and caudal articulations and approximately 0.5 cm of notarium and synsacrum. Following decalcification for 24–48hour in 10% formic acid (depending on the age of the bird), the vertebral column was trimmed in the sagittal plane to expose the articulations of the FTV and overlying spinal cord. Vertebral samples were processed into paraffin and 1 section per sample was stained with HE.
Sections from 145 case chickens and 96 control chickens were evaluated in a blinded manner as described above. For each spinal column, both FTV articular cartilages were scored and the highest overall single score recorded. Cases were excluded if histologic sections did not contain adequate representation of the FTV articular cartilages and adjacent articulating vertebrae.
Changes identified in the articulations of the FTV were consistent with osteochondrosis dissecans (OCD)-spectrum lesions. To compare OCD-spectrum lesions between the case and control birds, a scoring system was developed using the natural progression of OCD lesions as follows: 0 = normal; 1 = osteochondrosis latens (focal or multifocal areas of chondrocyte necrosis and eosinophilia that do not perturb the growth plate or result in cartilage clefting); 2 = osteochondrosis manifesta (perturbation of the growth plate with areas of retained cartilage and lagging endochondral ossification); 3 = OCD (visible clefts within the articular cartilage that do not communicate with the joint space); 4 = OCD with communication with the joint space; and 5 = complete disruption of the articular cartilages and effacement of the joint by inflammatory infiltrates.
Bacterial isolation and culture
In a prior study, we reported that pathogenic E. cecorum exhibit erythromycin and tetracycline resistance. 8 We exploited the natural resistance of pathogenic E. cecorum to these antibiotics and amikacin to enrich and select for pathogenic strains. All environmental and intestinal samples were cultured in both antibiotic-free Todd Hewitt broth with 1.0% yeast extract (THBY) and THBY supplemented with a final concentration of 64 µg/mL amikacin, 8 µg/mL tetracycline, and 4 µg/mL erythromycin (THBY-ATE). For feed and litter samples, 250 mL of sterile buffered peptone water (BPW) was thoroughly combined with the dry material, then 2 mL aliquots were added to 18 mL of THBY or THBY-ATE. Similarly 2 mL of each water sample was added to 18 mL of THBY or THBY-ATE. The 2 air vent swabs were combined by rinsing in 10 mL BPW and then 2 mL aliquots were transferred to 18 mL of THBY or THBY-ATE.
For each bird, the entire intestinal tract (except the ∼1 cm segment collected into NBF for histopathology) was placed into a sterile Whirl-Pak bag. Intestinal samples were macerated in the bag, and sterile swabs dipped into the macerated intestines were used to directly inoculate Columbia agar with colistin–nalidixic acid (CNA) and 5% sheep blood (BD). Separate swabs from the intestinal samples were used to inoculate THBY-ATE broth. For culture of spinal lesions, inflammatory masses at the FTV were sprayed with 70% ethanol before they were opened, incised with a sterile scalpel blade, and the central exudate was sampled with a Stuart swab and cultured on CNA plates.
All cultures, either broth or agar, were incubated overnight at 37°C with 5% CO2. For broth cultures, following overnight incubation a 10-µL loop broth was streaked onto the CNA agar, which was again incubated overnight at 37°C with 5% CO2. From CNA plates, up to 4 individual small, gray, mucoid, nonhemolytic or slightly α-hemolytic colonies were isolated and identified as E. cecorum using a previously described multiplex polymerase chain reaction for identification of enterococci. 22 Isolates identified as E. cecorum were cryopreserved at −80°C for genotyping.
Genotyping
Representative E. cecorum isolates from case and control chickens were selected for genotyping using pulsed field gel electrophoresis (PFGE). For case birds, isolates were selected to represent all 3 houses and included intestinal isolates (n = 33) obtained during weeks 1–4; spleen isolates (n = 15) obtained during weeks 1–4; and isolates from FTV lesions (n = 9). For control birds, all intestinal isolates (n = 17) obtained from birds in both houses weeks 1–4 were selected. In addition, isolates recovered from FTV (n = 8) collected from affected birds during the previous outbreak on the case farm were selected for comparison.
E. cecorum isolates were genotyped by PFGE using SmaI as previously described. 8 For accurate comparison of gel images, Salmonella H9812 serotype Braenderup (ATCC BAA-664, American Type Culture Collection, Manassas, VA) digested with XbaI was included in 3 lanes of each gel. A position tolerance of 1% was used for band matching.
Data analysis
Descriptive statistics were used to express culture results. The OCD scores between case and control birds were compared using Statistica 12 software (Statsoft Inc., Tulsa, OK) to perform the nonparametric Mann-Whitney U-test. Significance was set at P < .05 for all statistical tests. To compare genotypes, a dendrogram was created with Bionumerics 4.6 software (Applied Maths, Austin, TX), using a similarity matrix of Dice coefficients and the unweighted pair group method with arithmetic mean (UPGMA) algorithm. A cutoff of 95% similarity was used to determine the clusters.
Experimental Infection
In the longitudinal study of natural infection, OCD of the FTV was identified as a potential risk factor for ES. To determine the possible association between OCD in the FTV and ES lesion development, an experimental study was conducted. Experimental use of birds was approved by the Animal Care and Use Committee and conducted in compliance with the Guidelines for Care and Use of Laboratory Animals at North Carolina State University.
For experimental infection a commercial strain of broiler chicken (Cobb 500) was used. One-day-old chicks (n = 234) were obtained from industry collaborators. Prior to exposure, 20 birds were euthanized, and spleens and intestines were E. cecorum cultured to confirm their E. cecorum-negative status. The remaining chicks were inoculated orally with 1 × 107 colony-forming unit (CFU) in 100 µL of a previously characterized and sequenced pathogenic field strain of E. cecorum (SA3; GenBank accession number CP010064). Strain SA3, recovered from an ES lesion during an outbreak investigation, represents the largest genotype clade of pathogenic E. cecorum isolated to date, was included in the comparative genetic sequencing (CGS) analysis, and has been shown to be virulent in a chick embryo lethality assay. 7 –9 SA3 was grown overnight at 42°C with 5% CO2 in THBY extract, washed twice in phosphate-buffered saline and adjusted to the desired concentration. Concentration of the inoculum was verified with plate counts. Spleens were cultured at weeks 1–4 to confirm infection.
Birds were housed in an animal biosafety level 2 facility with an antechamber for donning and doffing protective equipment. In this facility, each room was isolated from all other rooms and external environment with positive pressure ventilation and a filtered air handling system. Birds were housed this way to prevent exposure to pathogens and to prevent the spread of pathogenic E. cecorum into the environment. Birds were grown in isolation for 7 weeks using industry standards for temperature and lighting. Birds were evaluated daily for clinical signs by veterinarians with expertise in poultry diseases (M.P.M., L.B.B., and H.J.B.). No evidence of concurrent infection was observed at any time point either clinically or at necropsy. Birds were fed ad libitum least-cost diets containing corn, soybean meal, dried distiller’s grains, and wheat. Diets were ground at 400 μm, giving more importance to pellet quality than particle size. Nicarbazin added to control coccidia in the starter diet was the only feed additive.
Twenty birds/week were sampled at weeks 1–4 and week 4.5, 40 birds/week were sampled at weeks 5 and 6, and the remaining birds (n = 34) were sampled at week 7 when the experiment ended. At each time point, sampled birds were euthanized by cervical dislocation, weighed, and necropsied by board-certified pathologists (L.B.B. and H.J.B.). The sex of each bird was recorded at necropsy. Spine, including the FTV and articulating vertebrae, was collected from all birds as described above.
Histopathology
For each bird, the articular cartilages comprising the FTV joints were scored for OCD and the highest overall single score recorded (as described above). The FTV was also evaluated histologically for characteristic features of ES including the presence or absence of inflammatory changes and intralesional bacteria consistent with E. cecorum. Based on presence or absence of these histologic findings, birds were categorized as ES-positive or ES-negative. Birds were excluded if the histologic sections did not contain adequate representation of the articular cartilages of the FTV and articulating vertebrae. The final number of vertebral columns scored for OCD and evaluated for ES status was 209.
Data analysis
Descriptive statistics were used to express culture results, and Fisher’s exact test was used to compare intestinal colonization at weeks 1–4 in E. cecorum-positive and E. cecorum-negative birds. The OCD scores, body weight, and sex were correlated with E. cecorum-positive or E. cecorum-negative status to determine if they were risk factors for disease. The nominal logistic fit feature of JMP Pro v. 11.0 (SAS Institute Inc., Cary, NC) was used to test the association of histologic evidence of ES with OCD scores; body weight (nested with week) and sex were included in the model as factors. The ordinal logistic fit feature of JMP Pro v. 11.0 (SAS Institute Inc.) was used to test the association of OCD scores with body weight (nested with week) and sex in the model as factors. Body weight/week was compared between ES-positive and ES-negative birds using a Student’s t-test or Mann-Whitney U-test if conditions of normality or heterogeneity of variance were not met. Significance was set at P < .05 for all statistical tests.
Results
Natural Disease
During the sampling period, the case farm developed an outbreak of ES. Overall mortality for the case farm was 4.8% in house 1, 5.5% in house 2, and 4.5% in house 3 (Fig. 3). Week 1 mortality on the case farm was predominantly due to peritonitis and yolk sacculitis caused by Escherichia coli and Pseudomonas aeruginosa (data not shown). Increased mortality at weeks 4–6 in all houses was due to ES, which caused an initial spike in mortality around week 4, and steadily increased from week 6 until processing. The control flock did not experience an outbreak of ES and had typical overall mortality (1%–2%).
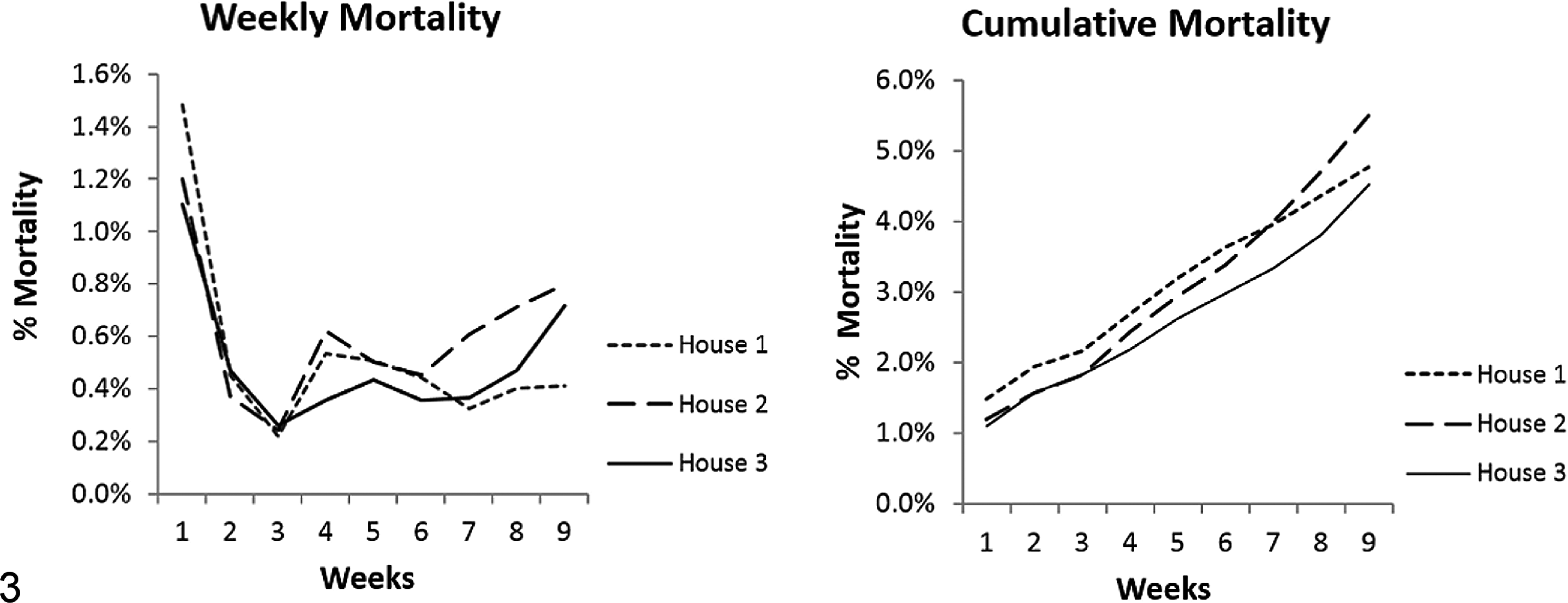
Case farm weekly and cumulative mortality curves. Overall mortality for the case farm was 4.8% in house 1, 5.5% in house 2, and 4.5% in house 3. The case farm had high week 1 mortality, which was predominantly due to peritonitis and yolk sacculitis caused by Escherichia coli and Pseudomonas aeruginosa. Increased mortality at weeks 4–6 in all houses was due to enterococcal spondylitis (ES), which caused an initial spike in mortality at week 4 and steady increases in mortality from week 6 until the flock was processed.
Environmental sampling
The environment was sampled during the pre-study on-farm evaluation and both prior to and following placement of the case and control flocks. E. cecorum was not isolated from any environmental samples.
Intestinal colonization
Culture of case and control bird intestines prior to placement (day 0) were negative for E. cecorum. In control birds, E. cecorum colonized the intestinal tract beginning in week 3, whereas intestinal colonization began as early as the first week in the case birds (Fig. 4). Intestinal colonization with E. cecorum in weeks 1–4 was significantly different between case and control birds (P < .001, Fisher’s exact test, n = 120 case and 80 control birds); 79 (66%) of 120 case birds were positive compared to 17 (22%) of 80 control birds.
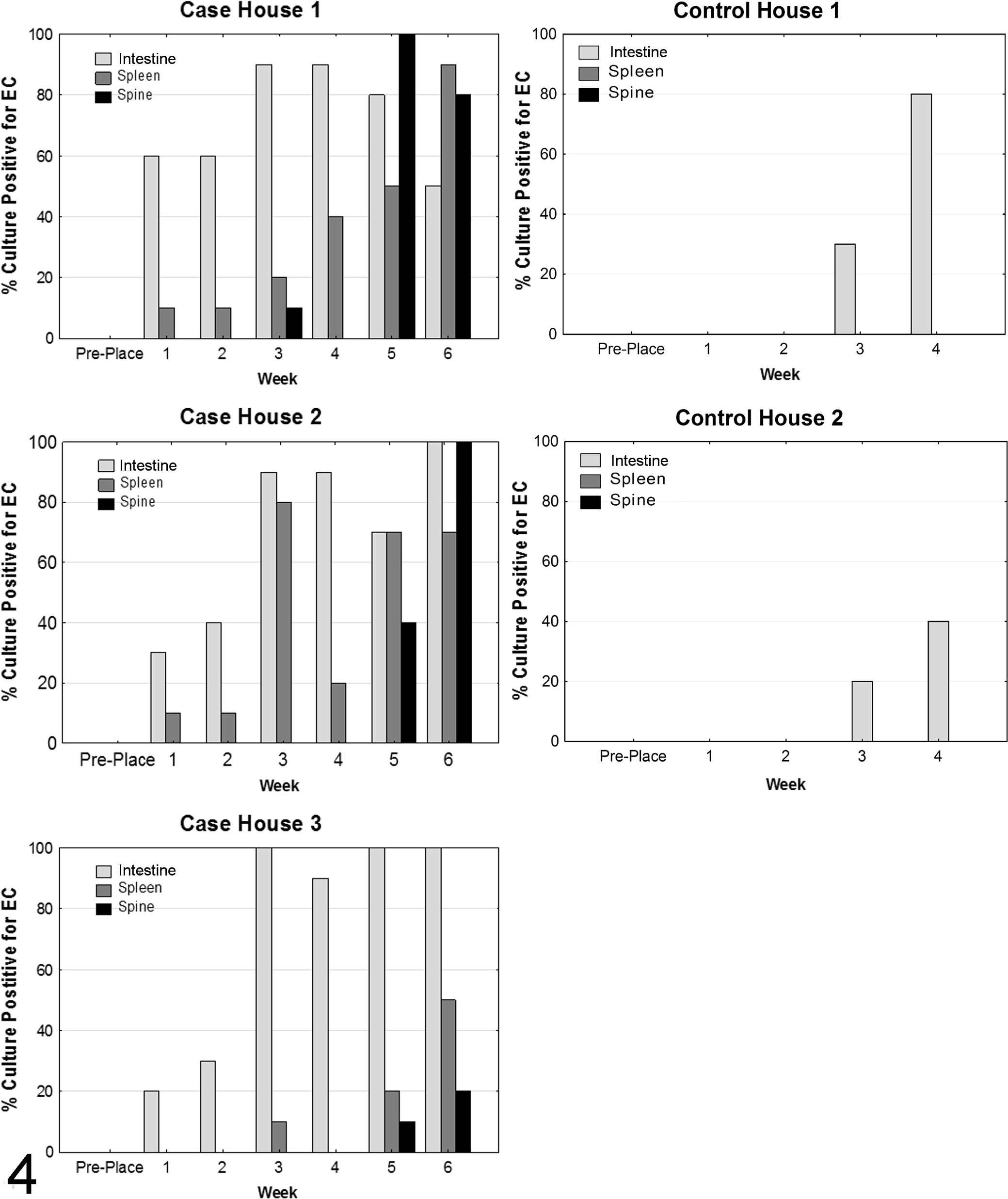
Enterococcus cecorum (E. cecorum) culture results for case and control farms. On the control farm, E. cecorum was not detected until week 3 in either house. In contrast, E. cecorum was recovered in high numbers from the intestines and spleens of case farm birds during weeks 1–3. On the case farm, intestinal colonization and bacteremia (evidenced by splenic infection) preceded infection of the free thoracic vertebra (spine) which was first identified at week 3 in house 1 but peaked during weeks 5 and 6 in all houses.
Bacterial isolates from spleens
E. cecorum was not recovered from spleens of control birds at any time during the experiment. However, case birds had positive splenic cultures, an indicator of bacteremia, beginning at week 1 in houses 1 and 2 and at week 3 in house 3 (Fig. 4). The number of case bird spleens positive for E. cecorum increased throughout the 6-week sampling period in all houses, with house 1 having the highest overall prevalence of isolates from spleen.
Genotype comparison
Consistent with previous observations and despite a wide diversity of E. cecorum genotypes, E. cecorum strains isolated from lesions formed a genetically distinct and highly clonal cluster (Fig. 5). 8 At a cutoff of 70% similarity, a cluster containing only isolates from birds affected by ES was observed. This cluster contained 15 (45%) of 33 intestinal isolates, all spleen isolates (n = 15), and all FTV isolates (n = 17) from the case farm. At a similarity of 95%, E. cecorum genotypes within this cluster separated into 3 smaller clusters. Of these, 1 cluster contained only E. cecorum recovered from FTV lesions during the prestudy on-farm investigation of the case farm. The other 2 clusters represented isolates from the FTV, spleen, and intestine of case birds sampled during the study period (Fig. 5).
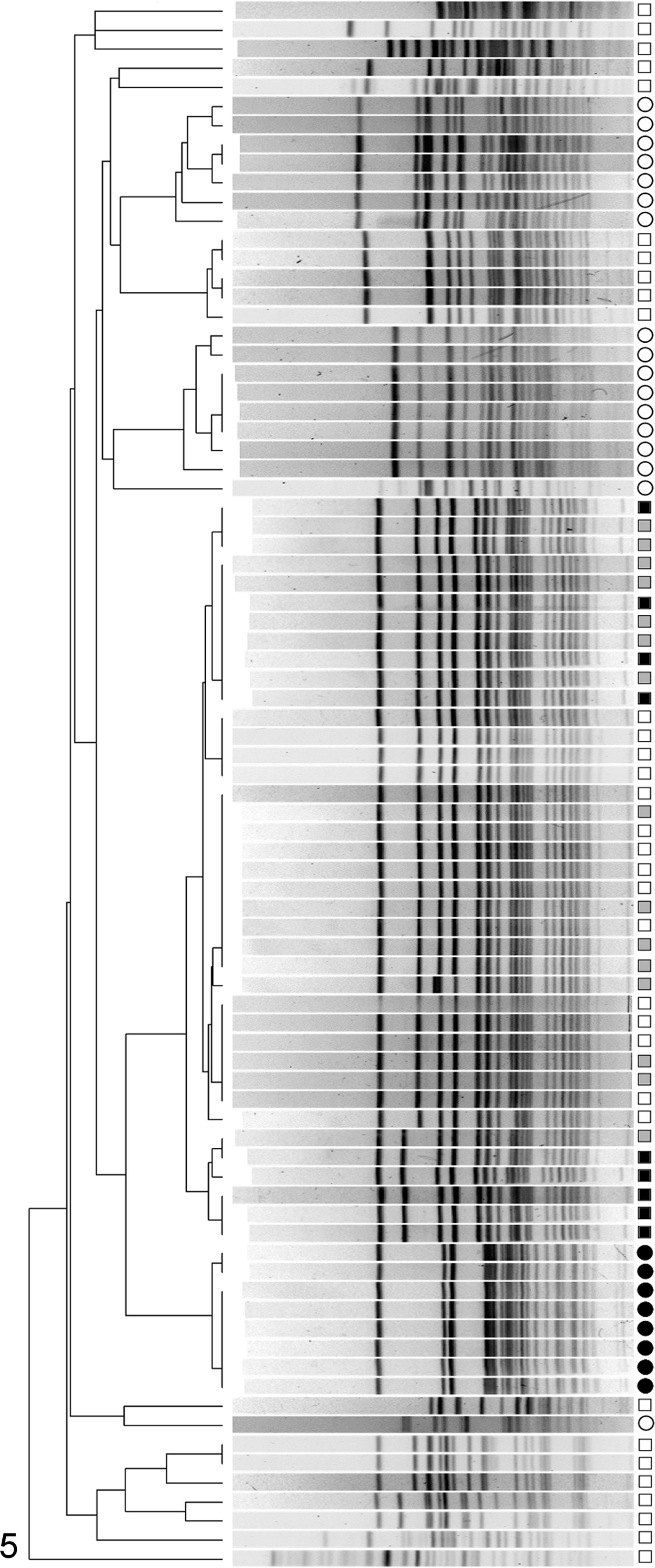
Dendrogram of genotypes produced by pulsed field gel electrophoresis of Enterococcus cecorum isolates. Control bird intestinal isolates are denoted by an open circle (^). For case birds, intestinal isolates are indicated by an open square (□), splenic isolates are indicated by a closed gray square (▪), and isolates from the free thoracic vertebra (FTV) are indicated by a solid black square (▪). Isolates recovered from free thoracic vertebra (FTV) lesions of birds during the prestudy on-farm evaluation of the case farm are indicated by a solid black circle (•). At 70% similarity, pathogenic E. cecorum (which includes isolates from spleen and intestine from case birds) form a cluster distinct from commensal E. cecorum (all isolates from control birds and some intestinal isolates from case birds). Within the pathogenic E. cecorum cluster, at 95% similarity, 3 smaller clusters are observed. One of these smaller clusters represents isolates recovered from free thoracic vertebral lesions sampled during the confirmation trip to the case farm immediately preceding the longitudinally sampled case flock. The other 2 smaller clusters represent 2 pathogenic genotypes, which were circulating in the longitudinally sampled case flock.
Intestinal histopathology
In case and control flocks, changes identified in the intestine were minimal to mild and included villous blunting, lymphoplasmacytic and heterophilic inflammatory infiltrates in the lamina propria, bacteria intimately associated with the microvillus border, and rare intraepithelial coccidia at several life stages (Supplemental Table 1). Compared to the case flock, birds from the control flock had significantly higher histopathology scores for inflammation, attaching bacteria, and coccidia (P < .001 for each parameter, Mann-Whitney, n = 150 case birds, n = 100 control birds; Fig. 6). However, the overall scores were low, and no clinical evidence or significant gross evidence of intestinal disease was noted on either farm. These findings are consistent with the subclinical or mild intestinal disease that was more apparent in the control flock.
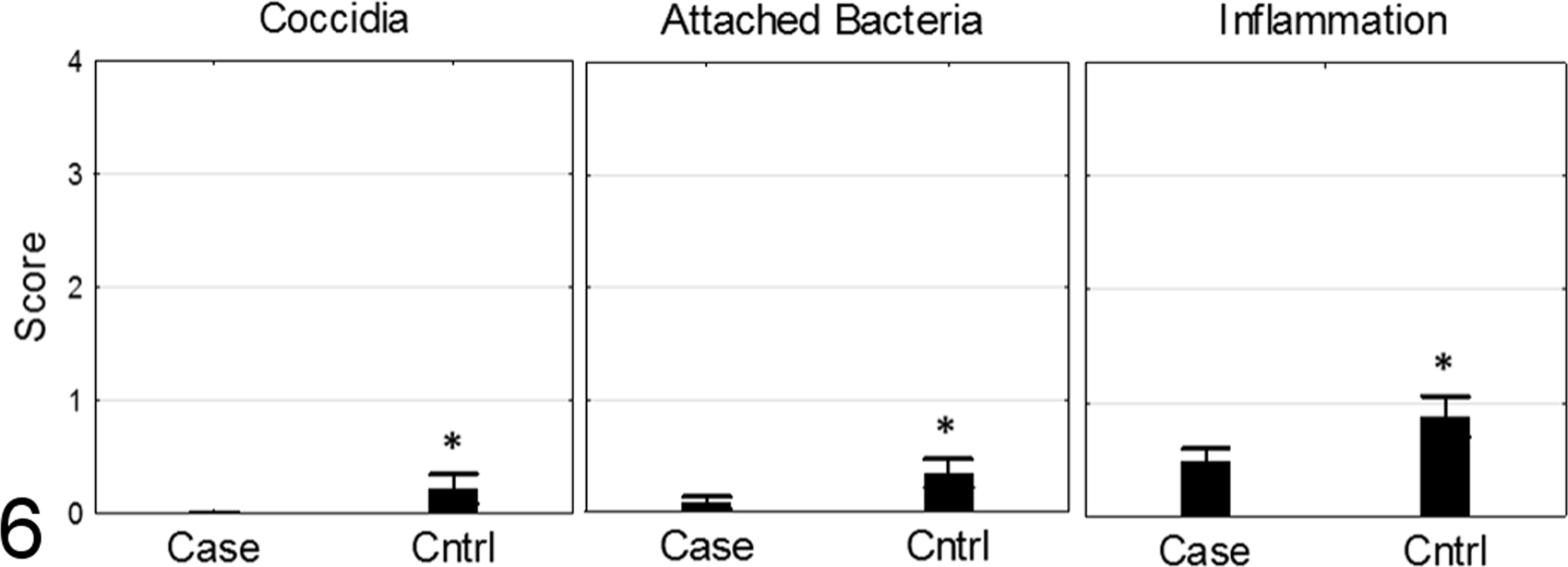
Intestinal histopathology scores for birds from case and control farms. Control farm birds had significantly higher scores for coccidia, attaching bacteria, and inflammation. Overall scores for both farms were low, consistent with subclinical intestinal disease.
Spinal histopathology
Evaluation of weekly samples revealed a stereotypical lesion development (Figs. 7–9) in the articular cartilages and primary spongiosa of the FTV and its articulations. Histologic lesions were consistent with OCD, and all developmental stages (i.e., latens, manifesta, and dissecans) were observed in weeks 1–4. The defining lesion of OCD, the variably large cartilaginous clefts that are formed, was identified in birds as early as week 1. These clefts were often partially to completely filled with thrombocytes, fibrin, and erythrocytes (Fig. 7). In some birds, cocci consistent with E. cecorum colonized and lined these clefts (Fig. 8). Beginning week 3, inflammation dominated by heterophils and macrophages was observed targeting cartilage clefts with intralesional bacteria. Inflammation was accompanied by bone remodeling and compression of the overlying spinal cord forming the characteristic ES lesion (Fig. 9).
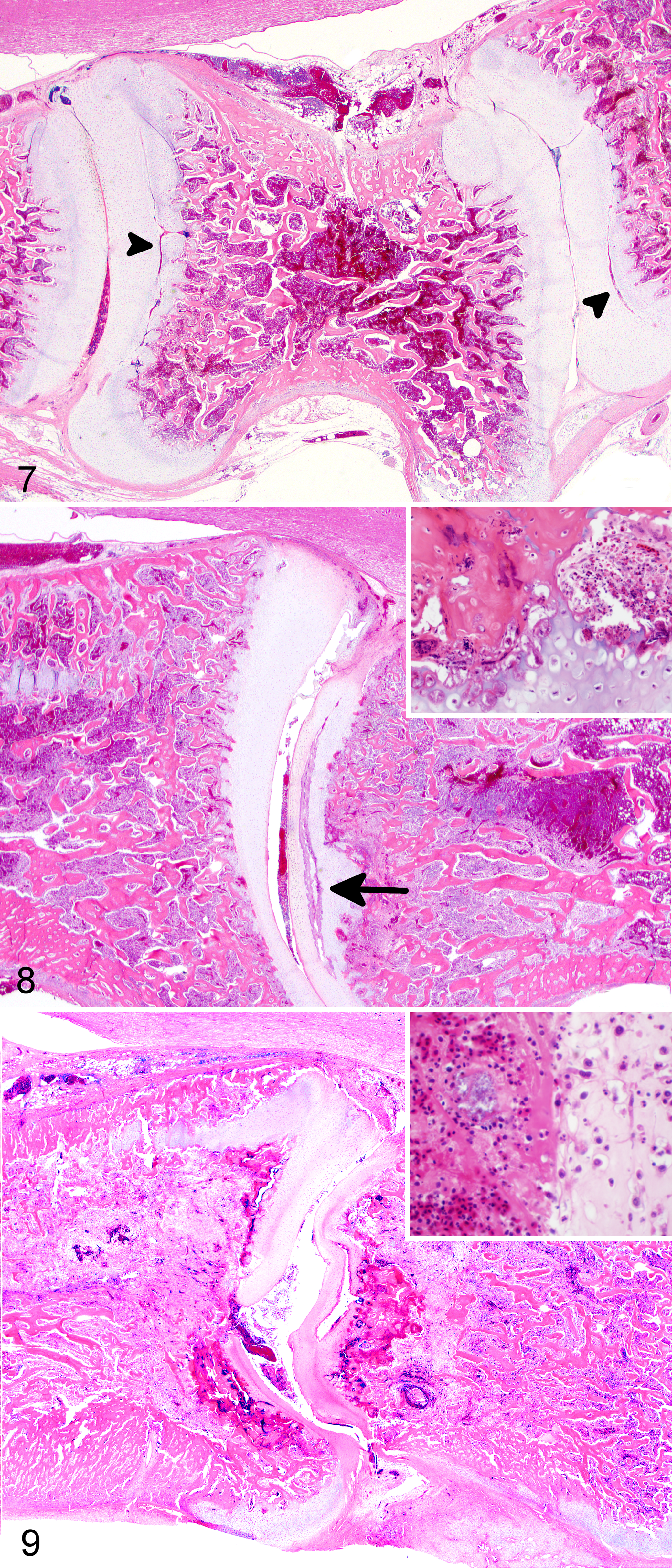
Enterococcal spondylitis Free thoracic vertebra (FTV), 3- to 4-week-old chickens.
As OCD of the FTV appeared to be important for ES lesion development, a histologic scoring system for OCD severity was developed following the natural progression of OCD-spectrum lesions (Figs. 10–15). Overall, the OCD lesions increased in frequency and severity throughout weeks 1–4 on both case and control farms (Fig. 16). Lesions of OCD were identified in 78 (54%) of 145 case birds including 41 (28%) of 145 birds with scores ≥3. Lesions of OCD were identified in 45 (47%) of 96 control birds including 25 (26%) of 96 birds with scores ≥3. There was no significant difference when the mean OCD scores were compared between case and control farms (p = .37, Mann-Whitney n = 145 case birds, n = 96 control birds).
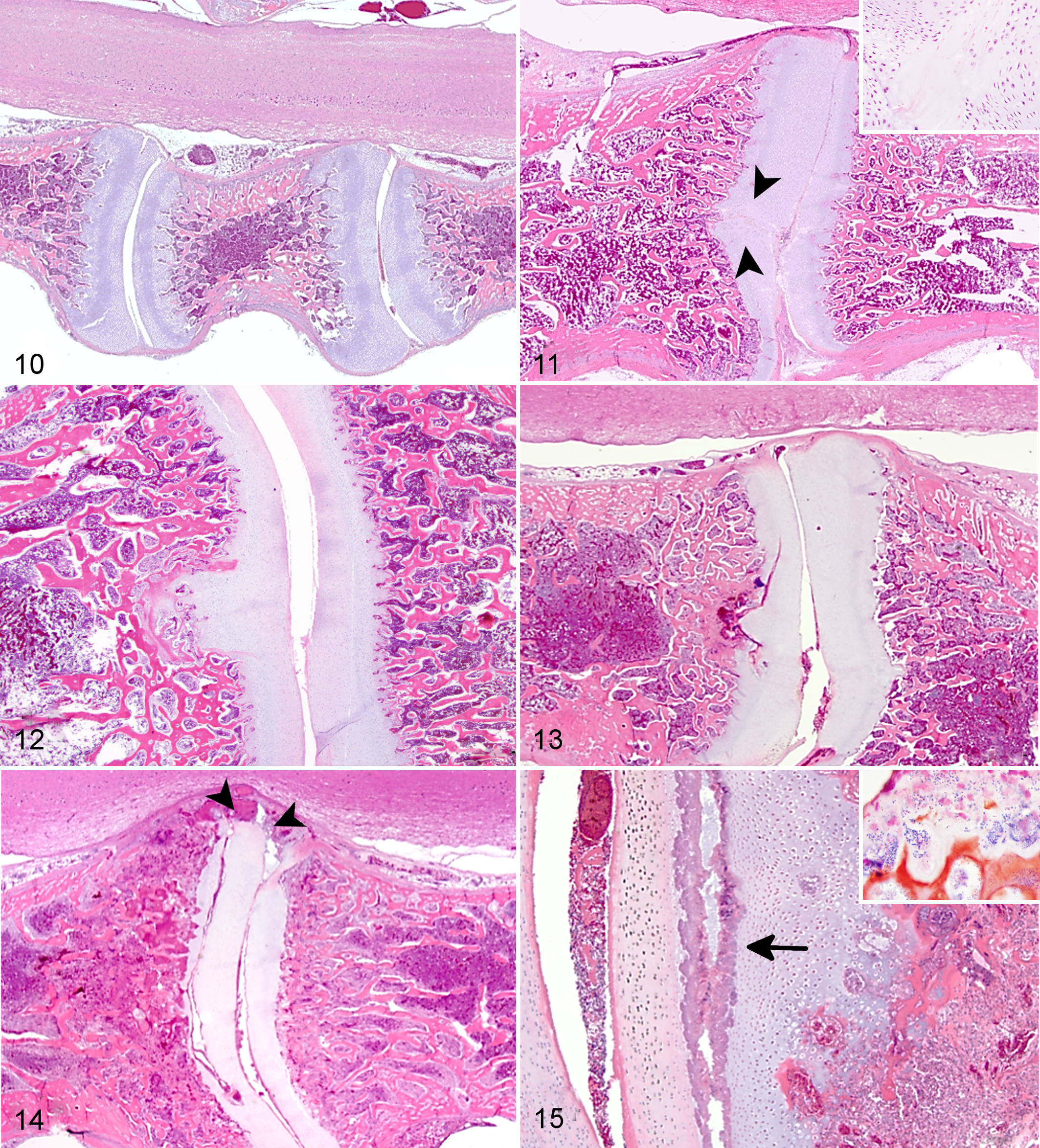
Osteochondrosis dissecans (OCD) scoring system, free thoracic vertebra (FTV). Hematoxylin and eosin.
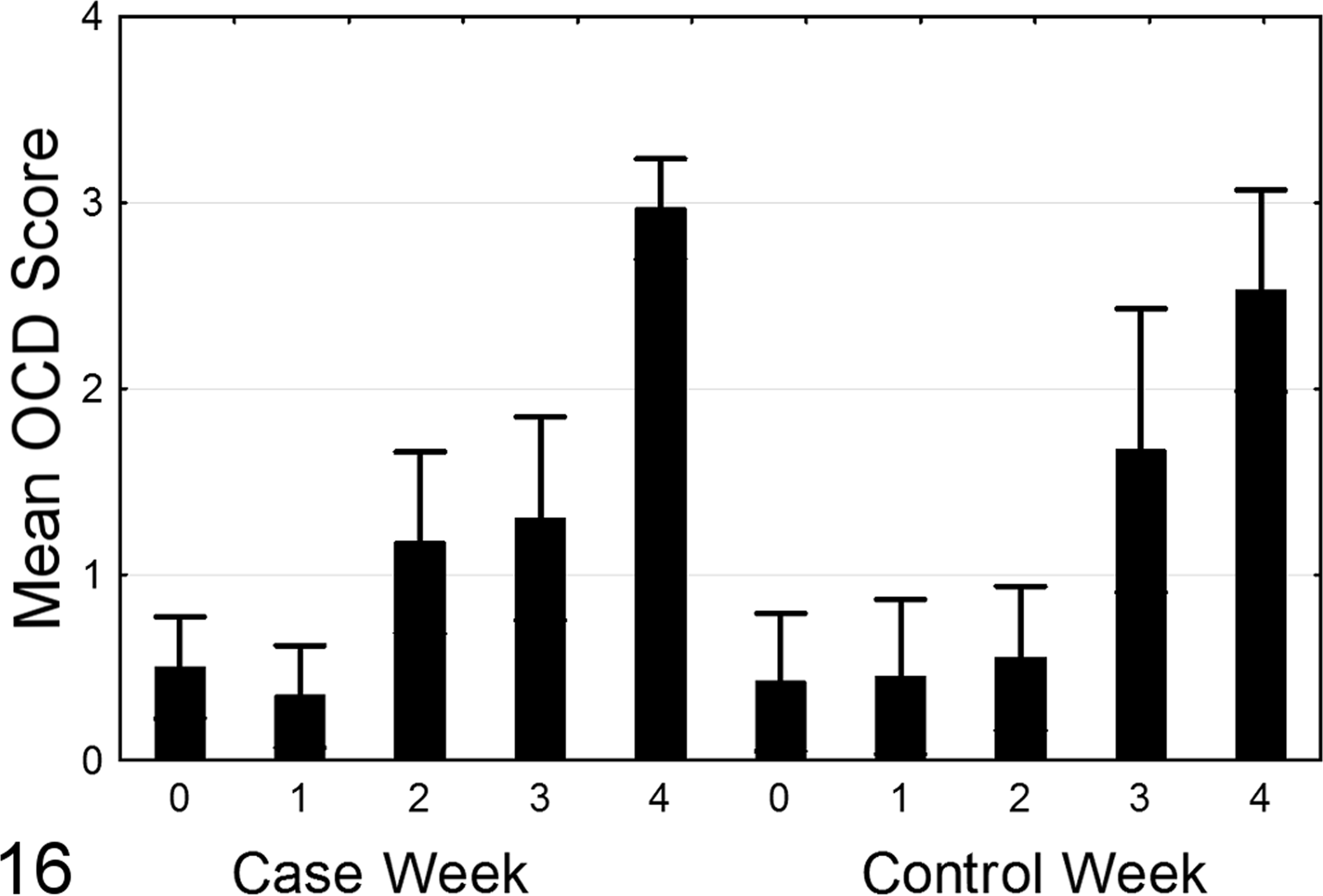
Osteochondrosis dissecans (OCD) scores (mean ± 95% confidence interval) per week for case and control flocks. Note increasing scores at weeks 1–4. No significant differences in OCD scores were observed between case and control farms.
Experimental Disease
Experimental infection with E. cecorum resulted in characteristic clinical signs of ES, which peaked between weeks 5 and 6, closely mirroring the course of disease observed in natural infection. However, in the experimental infection, the overall prevalence of ES lesions was 62 (29.7%) of 209.
Splenic infection
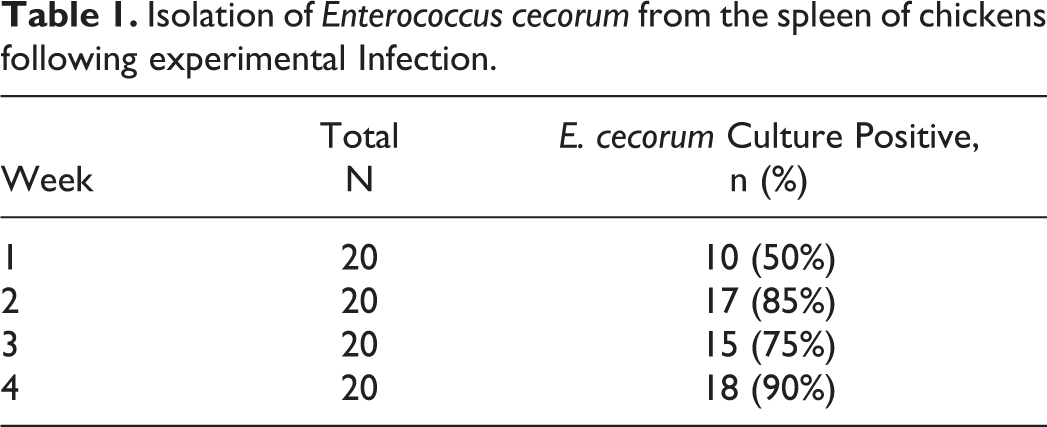
After oral infection with 107 CFU pathogenic E. cecorum strain SA3, E. cecorum was isolated from the spleen of 10 (50%) of 20 birds at week 1, which increased to 18 (90%) of 20 at week 4 (Table 1).
Isolation of Enterococcus cecorum from the spleen of chickens following experimental Infection.
OCD scores in ES-positive and ES-negative birds
Individual bird data for weekly OCD scores and body weight are provided in Supplemental Table 2. The OCD scores >1 were present in 185 (88.5%) of 209 birds and OCD scores ≥3 were present in 158 (75.6%) of 209 birds (Table 2). All ES-positive birds had OCD scores ≥3, while the ES-negative birds had a range of OCD scores from 0 to 4. The ES-positive status was significantly associated with OCD score (P < .0001, nominal logistic fit, n = 209 birds) and with decreasing body weight/week (P = .020, nominal logistic fit, n = 209 birds; Fig. 17). While a trend toward increased ES in male birds was observed, the association of sex with ES was not significant (P = .19, nominal logistic fit, n = 209 birds). Likewise, increasing OCD scores (regardless of ES status) were significantly associated with decreasing body weight/week (P < .0001, ordinal logistic fit, n = 209 birds) but not with male sex (P = .66, ordinal logistic fit, n = 209 birds).
Effect of Naturally Occurring Osteochondrosis Dissecans on the Outcome of Experimental Infection With Enterococcus cecorum.a
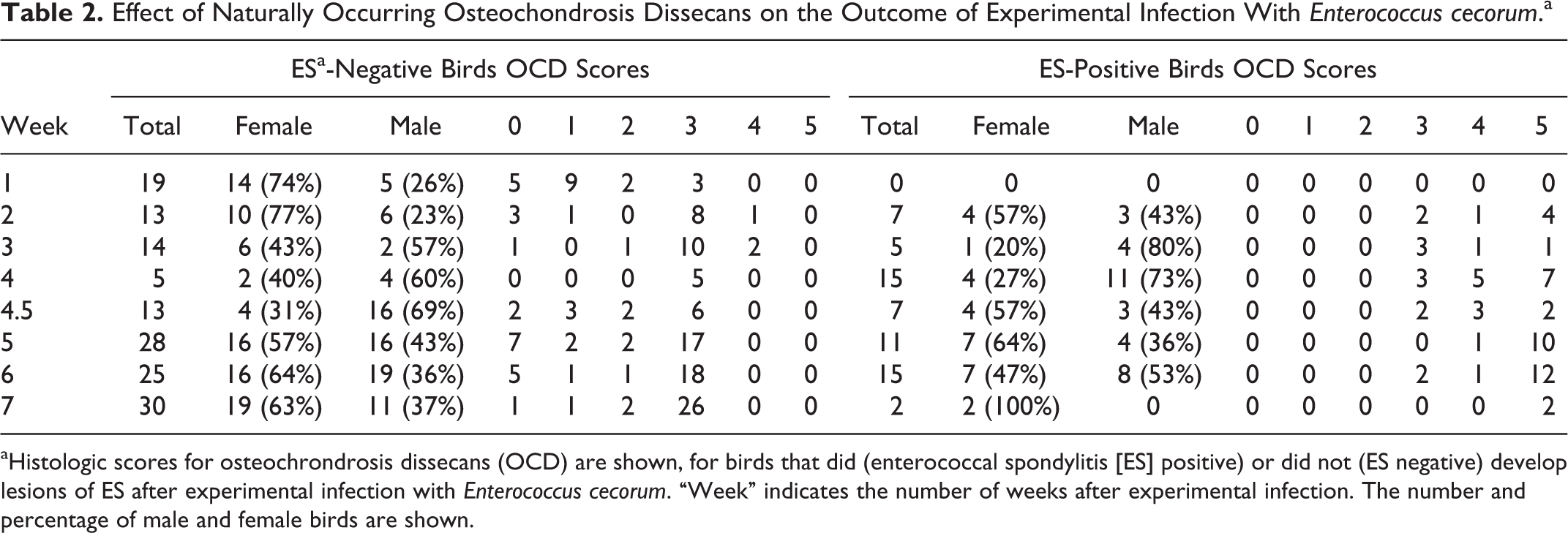
aHistologic scores for osteochrondrosis dissecans (OCD) are shown, for birds that did (enterococcal spondylitis [ES] positive) or did not (ES negative) develop lesions of ES after experimental infection with Enterococcus cecorum. “Week” indicates the number of weeks after experimental infection. The number and percentage of male and female birds are shown.
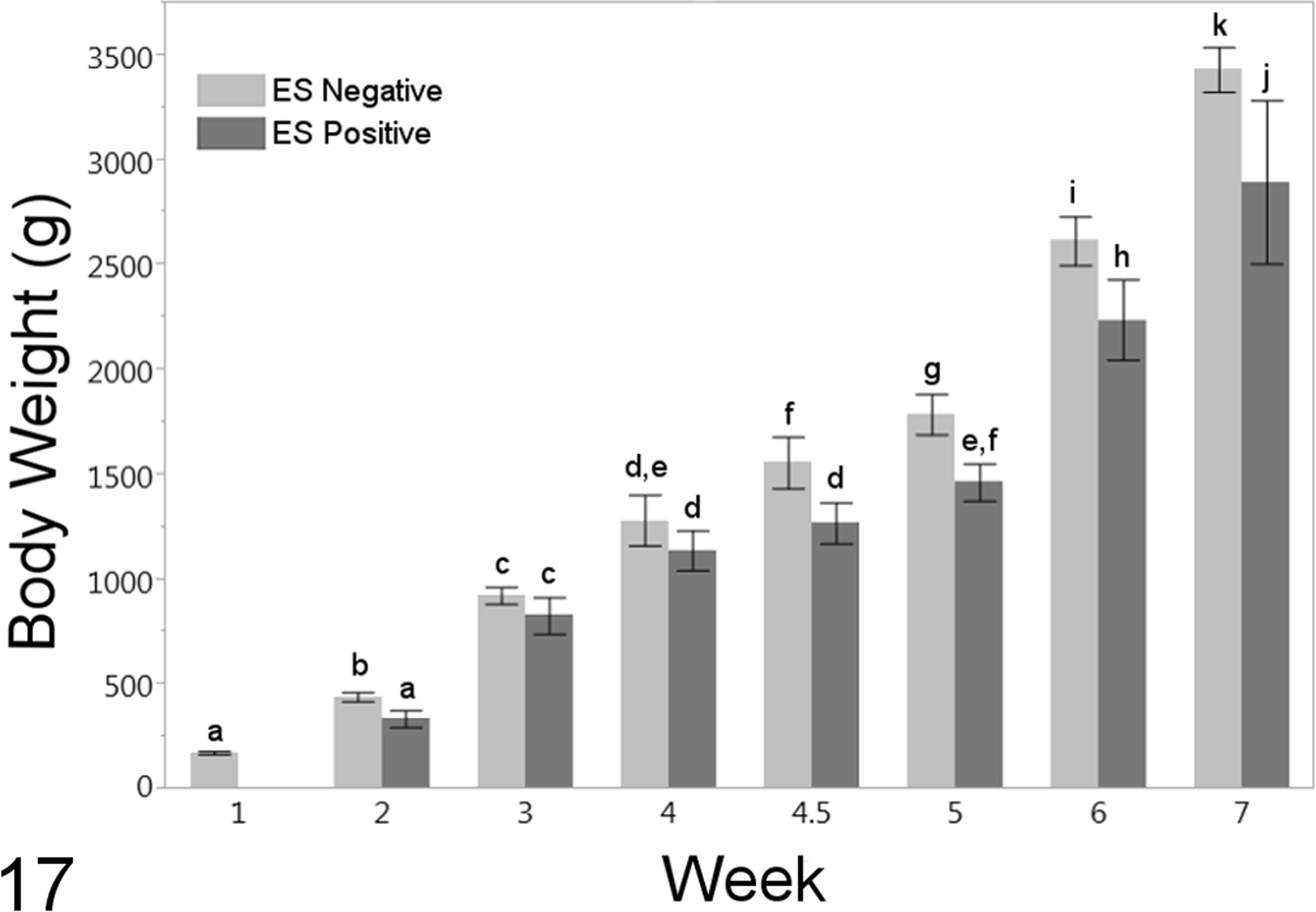
Body weights of birds affected with enterococcal spondylitis (ES positive) and nonaffected birds (ES negative). Columns represent means ± 95% confidence intervals. Columns that are labeled with the same letters (a–j) are not significantly different. Body weight as a function of week was negatively associated with ES positive status.
Discussion
In this study, the farm environment as a source of pathogenic E. cecorum was investigated, and the critical steps in the pathogenesis of ES including timing of intestinal colonization and bacteremia were determined. To date, ES studies have focused on characterization of clinical disease and pathogen properties; however, little is known regarding early steps of infection. By the time affected birds show clinical signs of ES in weeks 4–6, spinal lesions are well developed and have histologic changes consistent with chronic infection. As such, the early life period (weeks 1–4) was the focus of this study.
Important steps in the predicted pathogenesis of ES were defined in this study. Intestinal colonization with E. cecorum in broilers had been reported to begin at week 3. 17 However, in our study, E. cecorum with pathogenic genotypes were identified in naturally infected birds as early as week 1. Pathogenic E. cecorum appear to pioneer the gut flora in contrast to commensal E. cecorum strains that did not appear until week 3 in control farm birds. This ability to colonize the gut early in life may provide pathogenic E. cecorum strains with a competitive advantage and potentiate dissemination throughout a flock.
Anecdotal evidence from ES outbreak investigations has led to speculation that concurrent intestinal disease is needed for pathogenic E. cecorum to escape the gut and cause systemic infection. 1,2,28 However, in this study, quantifiable changes in the intestine of case birds were minimal to mild and significantly lower than those in control farm birds. Despite the lack of clinical intestinal disease and minimal to mild microscopic changes in the intestine, E. cecorum with the same genotypes as were detected in isolates from spinal lesions were frequently detected in the spleen of case birds. On the case farm, E. cecorum-positive spleens were found as early as week 1 in house 1 and during weeks 2 and 3 in houses 2 and 3 respectively. The timing of splenic infection was recapitulated in the onset of clinical signs of ES, which began first in house 1 (week 4) followed by house 2 (week 5) and house 3 (week 6). In general, bacteremia (measured as splenic infection) preceded clinical signs by 2–3 weeks.
These findings indicate that detection of gut colonization and bacteremia is possible at week 1 and clinical intestinal disease is not a prerequisite for E. cecorum bacteremia. However, further study of intestinal barrier dysfunction that includes both structure and function is needed in this early life period. For example, histologically normal intestines could have functional deficits that could result in a more permeable mucosal barrier and potentiate E. cecorum bacteremia. Likewise, disruption of intestinal structure by infectious, mechanical, or toxic irritants could also potentiate E. cecorum bacteremia, even though these were not present in the case flock. Despite these knowledge gaps, the ability to detect pathogenic E. cecorum in subclinical birds as early as week 1 could enable therapeutic interventions prior to the onset of clinical signs. Such an examination would be of greatest value for flocks being placed on farms with a history of ES outbreaks. It is worth noting that both critical steps in the pathogenesis of ES occur during the period when maternal antibodies are active. Use of an inactivated vaccine (bacterin) administered to broiler breeder hens may provide passive immunity to chicks during this critical early, 3-week period.
As in our previous studies, the genotype profiles of pathogenic E. cecorum (i.e., isolates recovered from spinal lesions) are distinct from commensal isolates and formed a large single cluster at a cutoff of 70% similarity. The observation that intestinal and splenic isolates recovered in the preclinical period have genotypes that cluster tightly with isolates recovered from the FTV confirms that intestinal colonization and bacteremia precede infection of the FTV. In addition, genotype profiles of isolates recovered from the gut, spleen, and spine identified 2 pathogenic strains responsible for the case farm outbreak. These 2 genotypes were different from the genotype of isolates recovered from the ES outbreak observed during the prestudy on-farm evaluation of the case farm. These findings, coupled with our inability to detect E. cecorum in the environment prior to or following the growing period, suggest pathogenic E. cecorum do not persist in the farm environment. However, the source of pathogenic E. cecorum for these and similar outbreaks remains unclear. Vertical transmission from breeder stock has not been demonstrated and horizontal transmission between farms in the vertically integrated broiler system seems unlikely. 25,35 E. cecorum has a wide host range including chickens, ducks, pigeons, canaries, pigs, calves, horses, cats, and dogs. 4,14,16 –18 As such, a biologic reservoir cannot be ruled out. While the source of infection remains unknown, identification of multiple strains causing the case farm outbreak suggests that any vaccination strategy would need to account for multiple, and potentially antigenically diverse, strains of pathogenic E. cecorum. Further work is clearly needed to assess the antigenic and genetic diversity of pathogenic E. cecorum causing outbreaks of ES in broilers worldwide.
The OCD-spectrum lesions were found to be critical in the pathogenesis of ES in both natural and experimental disease. The OCD-spectrum lesions were identified in the articular cartilages of the FTV in both naturally and experimentally infected birds, suggesting this is a common background lesion in modern broiler chickens. In natural infection, identification of pathogenic E. cecorum within characteristic OCD cartilage clefts in lesions with scores ≥3 led to further investigation of OCD and E. cecorum infection as cofactors for disease.
Experimental infection of commercial broilers was done to determine if increasing OCD scores were a risk factor for ES. While direct comparison of E. cecorum infection in birds with high and low OCD scores would have been desirable, currently there is no antemortem screen that allows for accurate categorization of birds by OCD score. Likewise, OCD is multifactorial and difficult to induce. Because of these limitations, we chose to experimentally infect a relatively large group of chickens and subsequently classify them as ES-positive or ES-negative based on clinical signs and microscopic lesions. The OCD scores from ES-positive and ES-negative birds were compared statistically. Using this approach, we observed that increasing OCD scores were significantly associated with positive ES status. In fact, birds with OCD scores <3 were completely resistant to ES and all ES-positive birds had OCD scores ≥3.
Development of the cleft in the articular cartilage of the FTV (OCD score 3) is likely required in the pathogenesis of ES. However, the mechanisms that explain why OCD predisposes to E. cecorum infection remain unclear and require further study. It could be that hemorrhage into these clefts lined by devitalized cartilage bring circulating pathogenic E. cecorum to this site. Once the bacteria gain access to the site, they could persist through binding to exposed, devitalized cartilage and evading opsonophagocytosis. In a previous CGS analysis, we observed that several surface-expressed extracellular matrix-binding proteins were conserved in pathogenic E. cecorum. 9 These proteins may mediate binding to the exposed necrotic cartilage within OCD lesions. Likewise, conserved capsular polysaccharide and surface polysaccharide antigens identified by CGS may play a role in evading opsonophagocytosis and killing by macrophages or heterophils.
Interestingly, the prevalence of birds with grades 3 and 4 OCD was greater in the experimental infection than in the natural infection groups. While a specific cause for this increased OCD prevalence in the experimental birds is unclear, this population also exhibited a much higher (∼30%) prevalence of ES than typically seen in naturally occurring outbreaks. However, direct comparisons of the natural and experimental infection studies are confounded by several uncontrolled factors including variations in dose of pathogenic E. cecorum, vaccination status for infectious agents other than E. cecorum, parasite control programs, exposure to additional pathogens, variations in gut flora, feed composition, genetic background, and husbandry procedures. All of these factors could play a role in the increased susceptibility observed in experimentally infected birds.
Regardless of the underlying causes, OCD-spectrum lesions have a well-characterized stereotypical progression, which is thought to be driven by vascular disturbances or increased type II collagen synthesis. 26,30,31,41 Morphologic features of OCD result from localized failure of endochondral ossification. Early changes in cartilage consist of a poorly defined area of eosinophilia and hypocellularity in the articular cartilage (osteochondrosis latens), which may extend into subchondral bone. This change can remain stable or progress to areas of cartilage retention in the ossification zone (osteochondrosis manifesta), which can again become stable or progress to an OCD lesion. Fulminant OCD lesions are characterized by clefts in the hyaline cartilage, which may or may not communicate with the joint space, or extend into subchondral bone where they combine with microfractures of the primary spongiosa. All stages of OCD lesion development were observed in the broilers in these studies and were used to develop the histologic scoring system for OCD severity.
The OCD lesions occur in several species including humans, pigs, horses, dogs, and chickens. In chickens, the FTV is uniquely susceptible to OCD. In the cervical spine, articulations between vertebrae have joint capsules and disks. 10 They form in much the same way as vertebral joints in mammals and other joints in birds. In contrast, FTV joints have neither a joint capsule nor an intervertebral disk. They appear to develop initially as a synchondrosis along with the other intervertebral joints in the thoracolumbar spine. At some point, FTV joints form by mechanical separation of the synchondrosis. Since the weight-bearing FTV articulates through epiphyseal cartilage making it susceptible to trauma, this is the only articulation in the vertebral column where OCD lesions occur. The OCD lesions in the FTV of layer hens were sufficient to cause paralysis in affected birds, with a prevalence approaching 16%. 11 In broilers, OCD associated with lameness most commonly affects the FTV and femoral trochanter. 11,19,20,29,34 In outbreaks of ES, some birds also have lesions in the femoral head. 7,30 While it is likely that OCD lesions in the femoral head were also present in birds in our study, the focus of this study was on the primary lesion in the FTV. Further study correlating OCD of the femoral head and infection of this location with pathogenic E. cecorum is needed.
The OCD lesion development is multifactorial in domestic animals which includes genetics, trauma, anatomic conformation, rapid growth, male sex, large breeds, and dietary components. 26,18,24 In our study, experimentally infected Cobb 500 birds developed higher rates of OCD and ES than naturally infected Hubbard/Ross708 crosses. As this observation could be due to environmental, nutritional or management factors, additional controlled experimental infections of various commercial strains of broiler are needed to assess the possible role of genetic backgrounds in ES susceptibility. Interestingly, in our study OCD severity was significantly associated with decreased body weight, which may have resulted from the impaired ability of affected birds to access feed. Similarly, no association between OCD and male sex was found. This observation is in contrast to the clinical case reports in which there has been a predisposition of male birds to ES. 1 –3,12 The ES was first described, and continues to be reported, in male broiler breeders providing further support for this association. It may be that the association of sex with ES is a weak one, which requires the numbers present in commercial operations to be identified. The precise genetic basis or impact of dietary components on development of the OCD in the chicken is unknown. Chickens may have a unique risk factor for developing OCD: temperature of egg incubation. Incubation temperatures are often not uniform in commercial incubators and low early–high late (LH) incubation temperature profiles are known to exacerbate musculoskeletal lesions by altering growth and development of the skeleton. 32,33 However, the impact of LH incubation temperature on OCD development is unknown.
In conclusion, our findings indicate that intestinal colonization and bacteremia due to pathogenic E. cecorum occur during the first 3 weeks of life and that OCD lesions in the FTV, which are common in broiler chickens during this same period, predispose infected birds to ES. These findings provide significant information for detecting and controlling this important emerging disease of broiler chickens.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was funded by US Poultry and Egg Association Grant #682.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
