Abstract
Pathologic and immunohistochemical changes caused by group I of the fowl adenovirus (FAV) serotype-1 99ZH strain, isolated from broiler chickens exhibiting gizzard erosion, were investigated in commercial broiler chickens. One hundred twenty-two chickens were inoculated with the strain by both oral and ocular routes at 1, 3, or 5 weeks of age and euthanatized for necropsy within 4–18 days of inoculation. Focal gizzard erosions were observed in the inoculated chickens of each age group. A histologically degenerative koilin layer, necrotic mucosa, intranuclear inclusion bodies in the glandular epithelial cells, inflammatory cell infiltrations in the lamina propria, submucosa, and a muscle layer were seen in the gizzards. Immunohistochemical staining showed evidence of FAV antigens in the intranuclear inclusion bodies. These findings were recognized regardless of their maternal antibody levels for FAV serotype-1. Gizzard lesions appeared later in the lower-dose-inoculated chickens than in the higher-dose–inoculated chickens. Numerous CD3-positive cells and IgY-positive plasma cells were seen in the gizzard lesions. In 5-week-old chickens the heterophil infiltrations in the lesions were milder than in younger chickens. Intranuclear inclusion bodies also were observed in the epithelial cells of the ileum or cecal tonsils of some chickens. Thus, this study shows that FAV-99ZH causes adenoviral gizzard erosion in broiler chickens without hepatic or pancreatic lesions and that cell infiltration is more severe than in dietary gizzard erosions.
Fowl adenovirus (FAV) is ubiquitous in chickens, as demonstrated by antibody surveys 11 , 41 and the high isolation rates of FAV in specimens taken from normal and sick birds. 3 , 19 FAV is associated with naturally occurring outbreaks of inclusion body hepatitis (IBH), 39 hydropericardium syndrome (HPS), 1 respiratory disease, 5 necrotizing pancreatitis, 32 or gizzard erosion.
Gizzard erosions and ulcers in chickens have been associated with diets that are deficient in vitamin B6 4 or with the ingestion of histamine, 14 gizzerosine, 26 and mycotoxins. 15 Gizzard erosions also have been observed at a high rate in the embryos and chicks of broilers. 36 However, several natural cases of gizzard erosion associated with FAV infection have been reported in layer chickens, broiler chickens, and quails in recent years. 2 , 10 , 25 , 37 Gizzard erosion has been observed in chickens inoculated with group I FAV serotype-8 and affected with hemorrhagic aplastic anemia syndrome. 12 In previous studies, however, the role played by FAV as a pathogen of gizzard erosion was not clearly established, because no viral examinations were conducted and because there were no adenoviral intranuclear inclusion bodies in the gizzard lesions. Some FAVs that had originally been isolated from either the proventriculus or a gastrointestinal pool of the tissues of broiler chickens caused gizzard erosions by experimental infection. 22 In our previous studies, outbreaks of adenoviral gizzard erosion (AGE) in slaughtered broiler chickens in Japan were examined, and an FAV serotype-1 named FAV-99ZH was isolated from the gizzard lesions with adenoviral intranuclear inclusion bodies; 31 also, AGE was experimentally reproduced in chickens inoculated with FAV-99ZH. 28 However, only a small number of specific-pathogen–free (SPF) white leghorn chickens were used, and no immunohistochemical examination was done during the experimental infection. There is thus insufficient information on the occurrence of gizzard and other organ lesions in chickens affected by this disease. We have been successful in reproducing AGE in broiler chickens inoculated with FAV-99ZH. 27 In this study, we describe in detail the histopathologic and immunohistochemical findings of 1-, 3-, and 5-week-old commercial broiler chickens inoculated with FAV-99ZH by both oral and ocular routes.
Materials and Methods
Chicken
One-day-old commercial broiler chickens were obtained from a commercial breeder. They were kept in isolated rooms in a negatively pressured house with filtered air. Noncontact control chickens also were maintained under the same conditions.
Virus
FAV-99ZH originally isolated from an outbreak of AGE in a commercial broiler chicken flock in Japan was used. 31
Experimental design
Twenty-six commercial broiler chickens were inoculated at 1 week of age with 103 tissue culture infective dose (50%) (TCID)50 per 0.05 ml of FAV-99ZH through both oral and ocular routes (group 1). In the same way, 26, 40, and 30 chickens were inoculated with 106TCID50/0.05 ml of FAV-99ZH at 1, 3, and 5 weeks of age, respectively (groups 2–4). The volume of inoculum was 0.1 ml orally and 0.02 ml as eye drops. All the chickens were observed daily for clinical signs and were euthanatized by cervical dislocation for necropsy within 4–18 days of inoculation. A total of 39 chickens were used as noninoculated controls. The preinoculated sera were examined by a virus neutralization test, 11 using FAV-99ZH for the FAV serotype-1 maternal antigen in all chickens. As the results show, the mean maternal antibody titers were 298, 550, and 14, ranging from 16 to more than 2,560, 32 to more than 2,560, and 2 to more than 64, in groups 1, 2, and 3, respectively. It was confirmed that all the chickens in group 4 did not have the maternal antibody. 27
Histology
After postmortem examination of the chickens, sections of the esophagus, proventriculus, gizzard, duodenum, jejunum, ileum, distal cecum, cecal tonsil, bursa of Fabricius, liver, pancreas, spleen, kidney, heart, lung, trachea, thymus, and brain were removed. The tissues were fixed in 20% buffered neutral formalin. The paraffin-embedded tissues were sectioned at 4 μm and stained with hematoxylin and eosin (HE).
Immunohistochemistry
The FAV-99ZH antigen in all sections, CD3-positive cells, and immunoglobulins IgA, IgM, and IgY in selected gizzards were detected in formalin-fixed, paraffin-embedded tissues by immunohistochemical staining. Rabbit anti-FAV 99ZH serum (prepared in the Zen-noh Institute of Animal Health, Chiba, Japan), affinity-isolated rabbit anti-human CD3 T-cell serum (Dako A/S, Copenhagen, Denmark), affinity-purified goat anti-chicken IgA-α chain–specific serum, affinity-purified goat anti-chicken IgM-μ chain–specific serum, and affinity-purified goat anti-chicken IgY Fab fragment serum (Bethyl Laboratories Inc., Texas, USA) were used as primary antibodies at dilutions of 1:12,000, 1:400, 1:4,000, 1:4,000, and 1:4,000, respectively, in 0. 01 M phosphate-buffered saline, pH 7.2. Based on a published procedure, the presence of FAV-99ZH antigen and CD3 was demonstrated by the EnVision+ Rabbit/HRP system (Dako, Copenhagen, Denmark). 30 , 38 Actinase E (Kaken Pharmaceutical Co. Ltd., Tokyo, Japan), 0.1% in phosphate-buffered saline for unmasking, and normal goat serum (Vector Laboratories Inc., California, USA) in 0.05 M Tris buffer (pH 7.6) as a blocking serum were used. The presence of IgA, IgM, and IgY was shown by the labeled streptavidin-biotin system. 13 Actinase E 0.1% in phosphate-buffered saline for unmasking, normal donkey serum (Chemicon International, Inc., California, USA) in 0.05 M Tris buffer (pH 7.6) as the blocking serum, biotinylated donkey anti-goat IgG antibody (Chemicon International) as the secondary antibody, and streptavidin peroxidase–labeled conjugate (Kirkegaard & Perry Laboratories Inc., Maryland, USA) were used. 3,3′-Diaminobenzidine-tetrahydrochloride served as the chromogen, and the sections were counterstained with methyl green.
Specificity of the polyclonal CD3 antiserum for chicken lymphoid tissues
The formalin-fixed, paraffin-embedded thymus, spleen, and bursa of Fabricius of SPF chickens were prepared for showing specificity of the polyclonal CD3 antiserum to human T cells. The CD3-positive cells were distributed in both the cortex and the medulla of the thymus and in the red pulp and periarteriolar lymphatic sheaths of the spleen. Negative controls in which normal rabbit sera were substituted for the primary antibody had no peroxidase staining.
Results
Clinical signs and gross lesions
The clinical signs and gross lesions were described in our previous report. 27 The prevalence of gizzard erosion in each inoculated group at various postinoculation (PI) days is shown in Fig. 1.
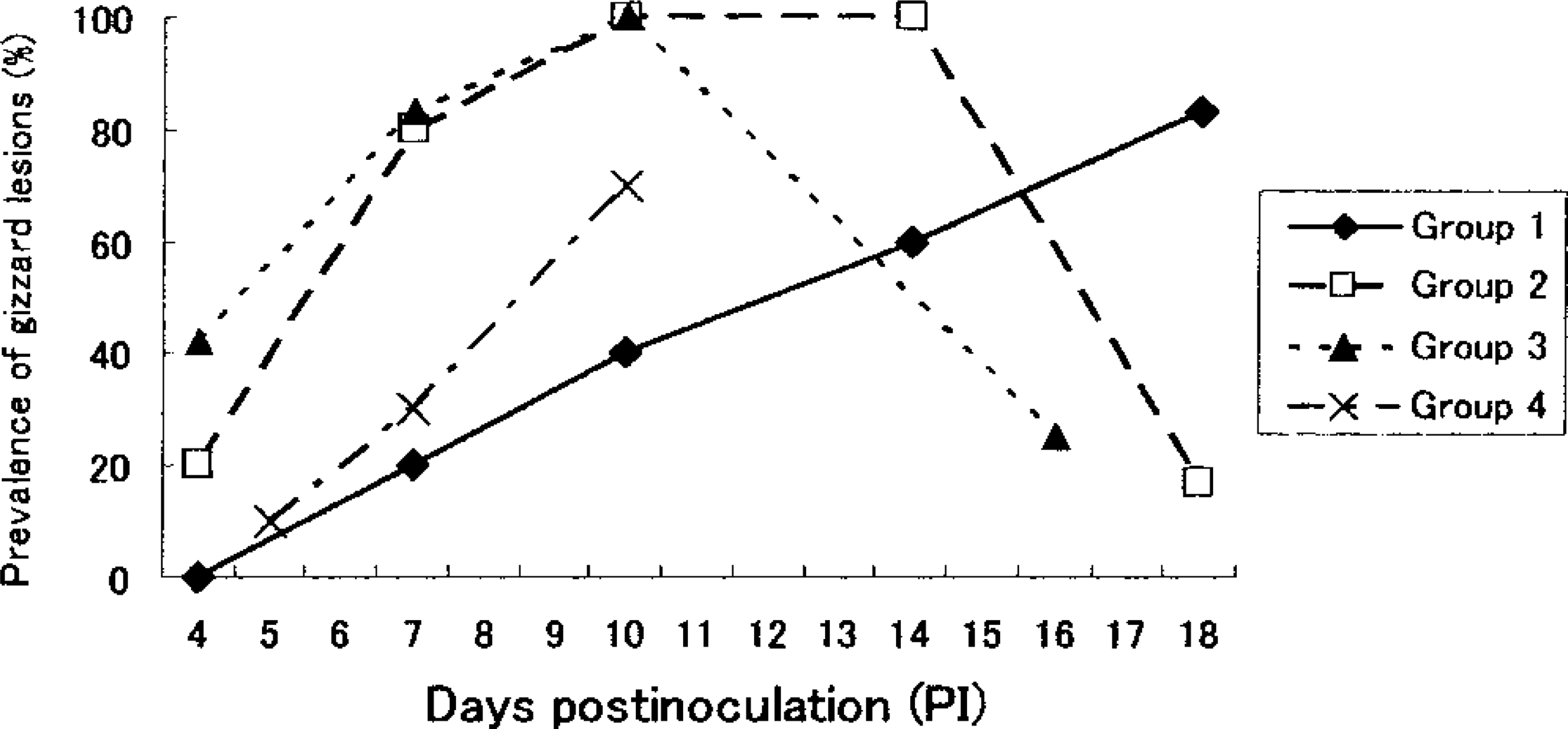
Prevalence of gizzard erosion grossly observed in chickens inoculated with FAV. (group 1: inoculated at 1 week of age with 103TCID50; group 2: inoculated at 1 week of age with 106TCID50; group 3: inoculated at 3 week of age with 106TCID50; group 4: inoculated at 5 week of age with 106TCID50). In group 1 the lesions appeared later than in group 2. In group 4 the prevalence of lesions was lower than in groups 2 and 3.
Histologic findings
The main microscopic lesions in gizzards that were similar to lesions of natural cases 2 , 31 and experimentally inoculated SPF white leghorn chickens 28 are summarized in Table 1. Degeneration of the koilin layer was observed from day 10 PI in group 1, day 7 PI in group 2, day 4 PI in group 3, and day 5 PI in group 4, with the peak of prevalence occurring on day 18 PI, days 10 and 14 PI, day 10 PI, and day 10 PI, respectively. The layer was thickened and loosened, and it contained variable numbers of desquamated cells. All the chickens on days 10 and 14 PI in group 2 and on day 10 PI in group 3 had degenerative koilin layers. Focal necrotic findings, for example, the degeneration, desquamation, and disappearance of glandular epithelial cells and the atrophy or disappearance of the column of koilin material in the lumen of the gland, were generally observed under the degenerative koilin layer (Fig. 2). However, on day 18 PI in group 2 and day 16 PI in group 3, the degenerative koilin layer was observed without epithelial necrosis. Proliferating glandular, epithelial cells in the bottom of the thickening mucosal membrane were observed on recovery. Large basophilic or amphophilic intranuclear inclusion bodies and, rarely, eosinophilic bodies, those with a halo in the glandular epithelial cells, were seen mainly in or around the necrotic area on day 4 PI and from days 10 to 18 PI in group 1, from days 7 to 10 PI in group 2, from days 4 to 10 PI in group 3, and from days 5 to 10 PI in group 4 (Fig. 3). Lamina propria of the necrotic area were edematous and contained inflammatory cell infiltrations consisting of mononuclear cells and heterophils. Mononuclear cell infiltration in the perivascular connective tissue in the submucosa and the muscle layer was observed from day 10 PI in group 1, day 7 PI in group 2, days 4 to 10 PI in group 3, and day 7 PI in group 4. Lymphoid nodule formation in lamina propria of the necrotic area was observed from day 14 PI in group 2 and from day 16 PI in group 3 (Fig. 4). No lymphoid nodule formation was observed in the gizzards of any of the chickens in groups 1 and 4.
Histologic and immunohistochemical findings in the gizzards of commercial broiler chickens in the treatment groups.
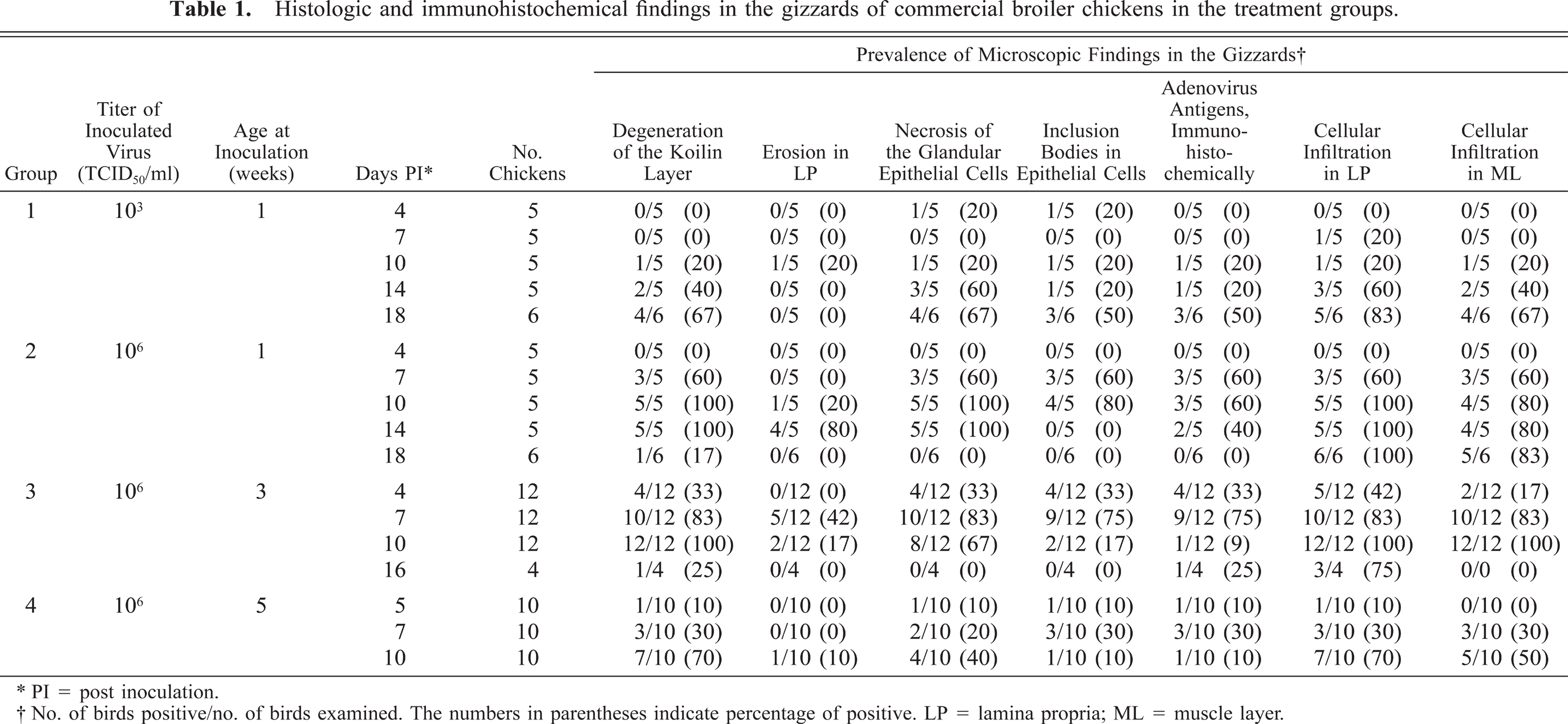
∗ PI = post inoculation.
† No. of birds positive/no. of birds examined. The numbers in parentheses indicate percentage of positive. LP = lamina propria; ML = muscle layer.
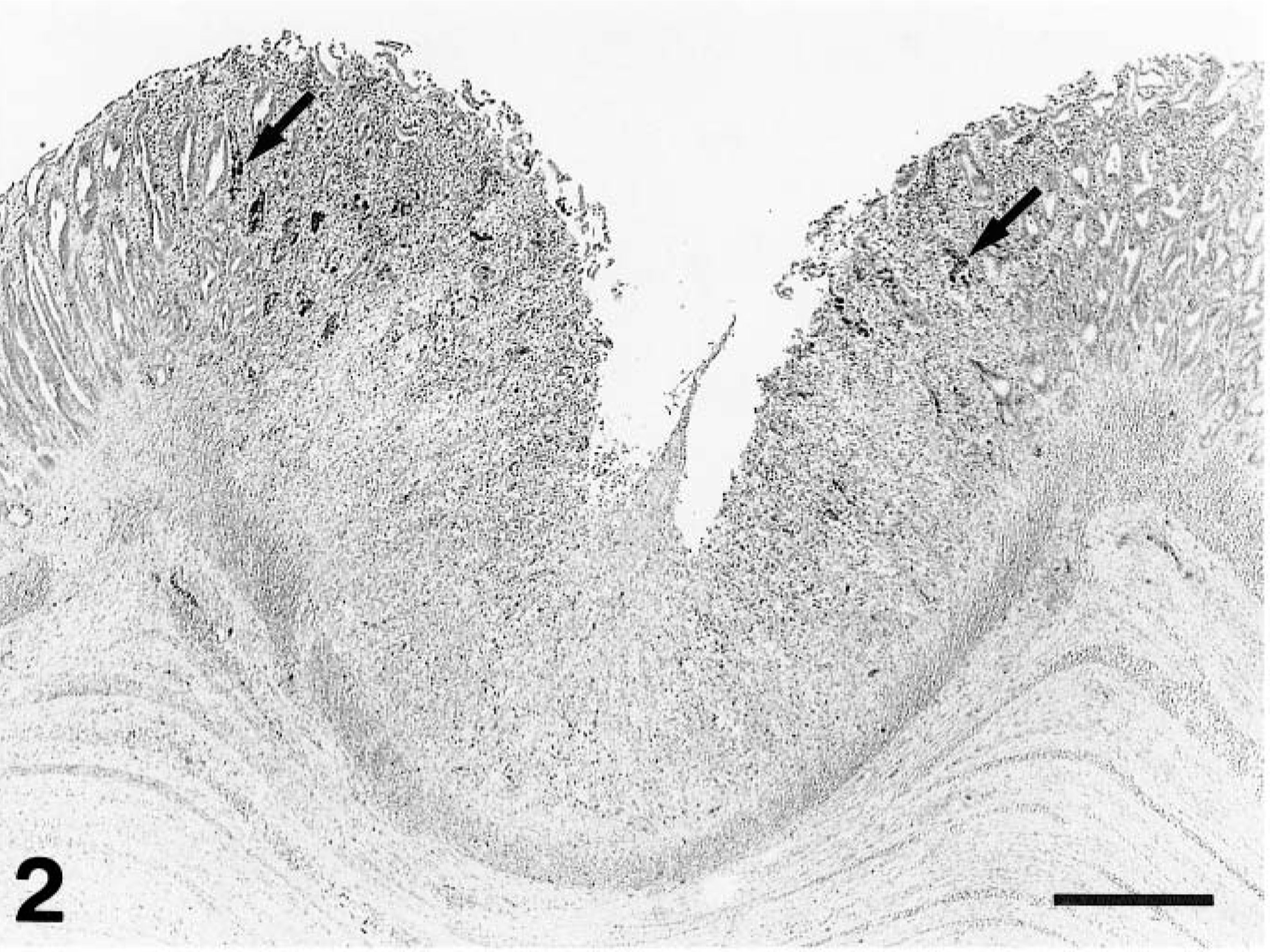
Gizzard; a chicken from group 2 on day 7 PI. Erosion, disappearance of epithelial cells, and inflammatory cell infiltration in focal necrotic mucosal membrane. Arrows point to positive-staining cells with intranuclear inclusion bodies, shown in higher magnification in Fig. 3. The koilin layer is artificially detached. Immunohistochemical staining for the FAV antigen, methyl green counterstain. Bar = 200 μm.
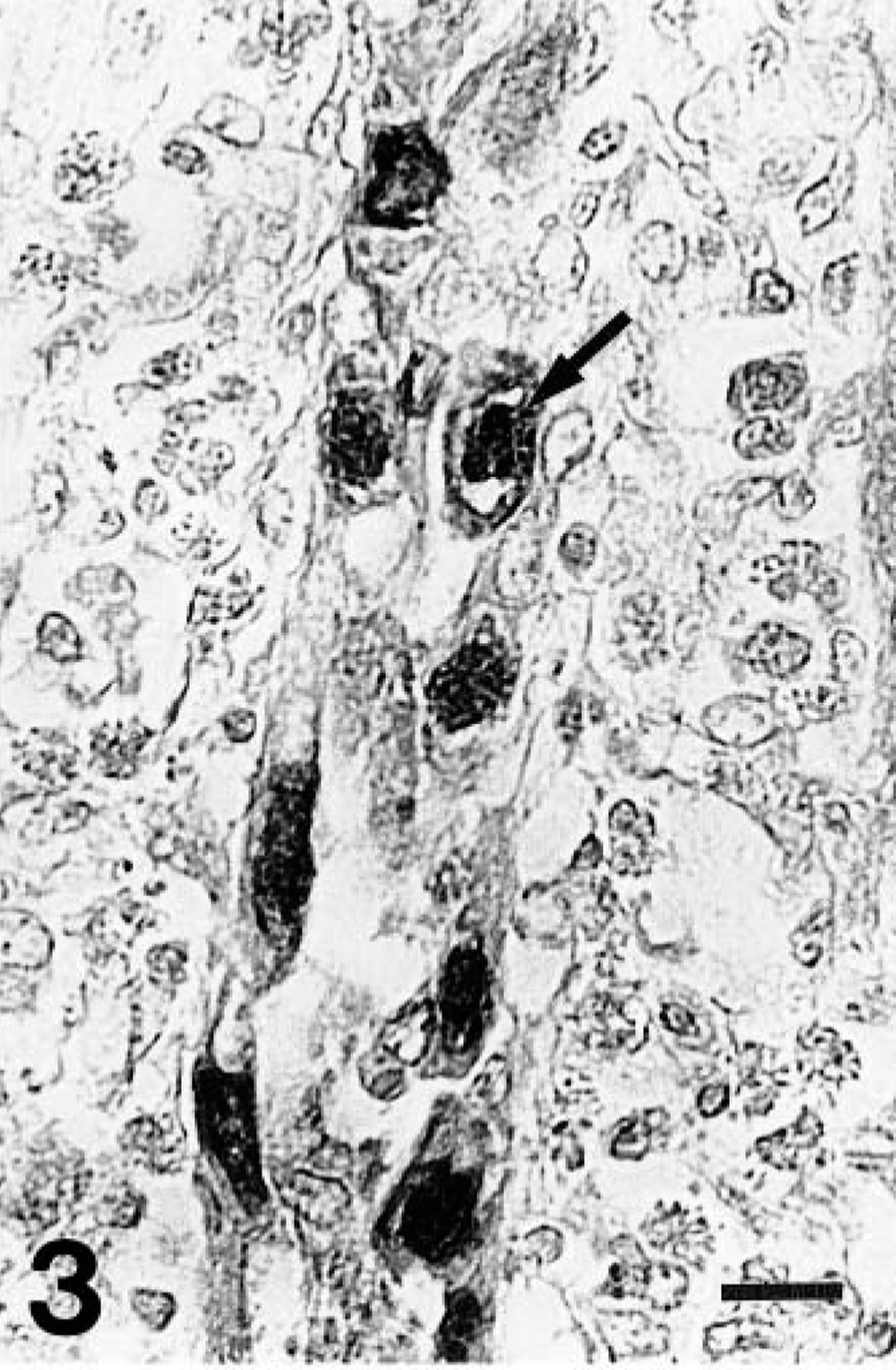
Gizzard; a chicken from group 1 on day 10 PI. Higher magnification of the FAV antigen–positive intranuclear inclusion bodies in the epithelial cells of an affected gizzard. Inclusions with a halo are seen (arrow). The lamina propria is edematous, and there are numerous inflammatory cells. Immunohistochemical staining for the FAV antigen, methyl green counterstain. Bar = 10 μm.
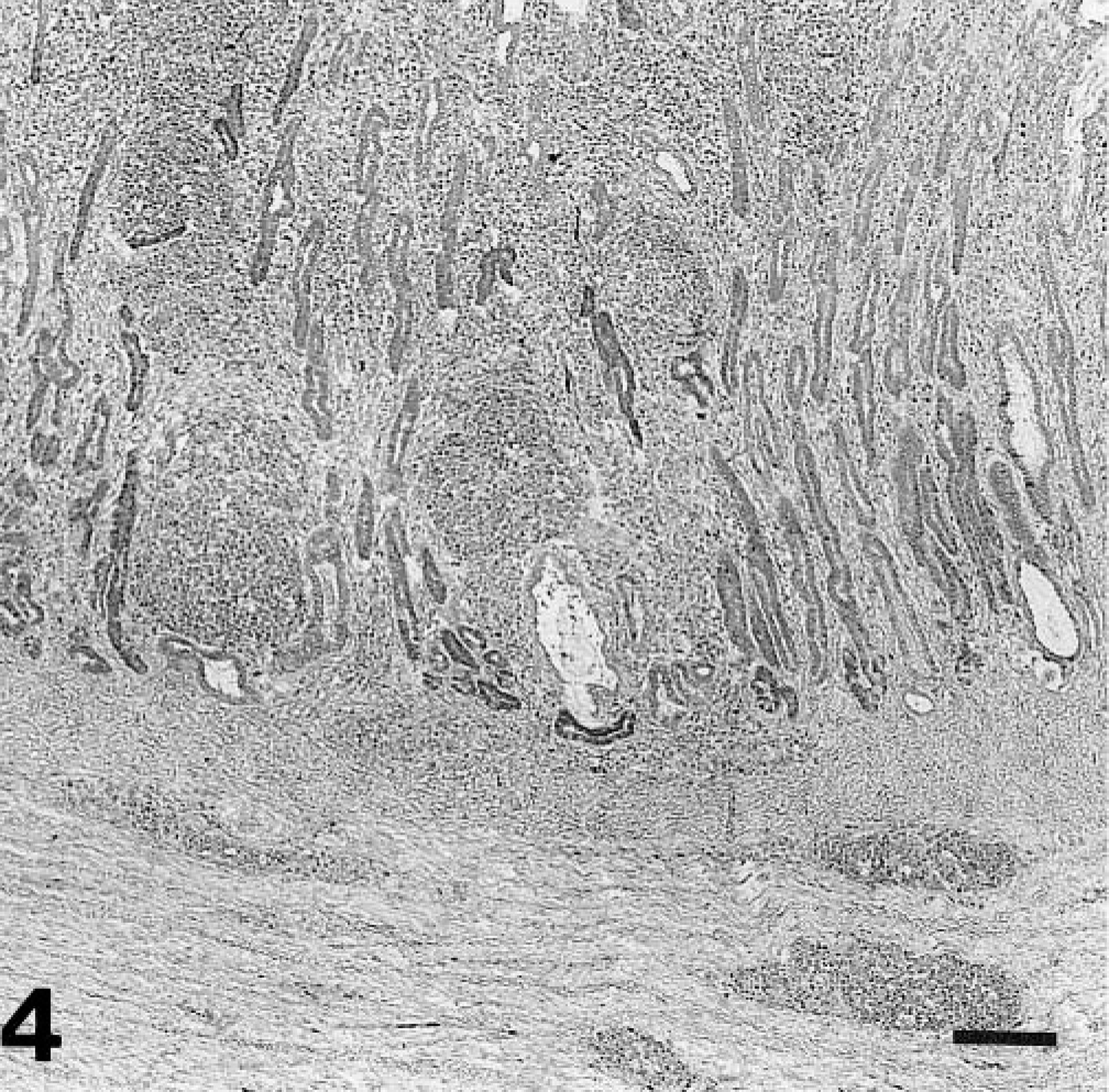
Gizzard; a chicken from group 2 on day 18 PI. Histologic findings in gizzard lesions during recovery. The glands are dilated and lined by hyperplasic epithelium, and the lamina propria is greatly expanded by mononuclear cells and lymphocytic nodules. HE. Bar = 100 μm.
There were several intranuclear inclusion bodies in epithelial cells of the ileum of one chicken on day 5 PI in group 4 and in the cecal tonsil of one chicken on day 14 PI in groups 1 and 4 and on day 4 PI in group 3 (Fig. 5). However, two of these chickens (the chicken on day 14 PI in group 1 and the chicken on day 4 PI in group 3) showed no gross lesions, and there was no histologic finding in the gizzards. Degenerative epithelial cells of the bile duct were observed in two of the 12 chickens on days 4 and 7 PI in group 3, respectively. No significant lesions were present in the other organs or in the noninoculated control chickens.
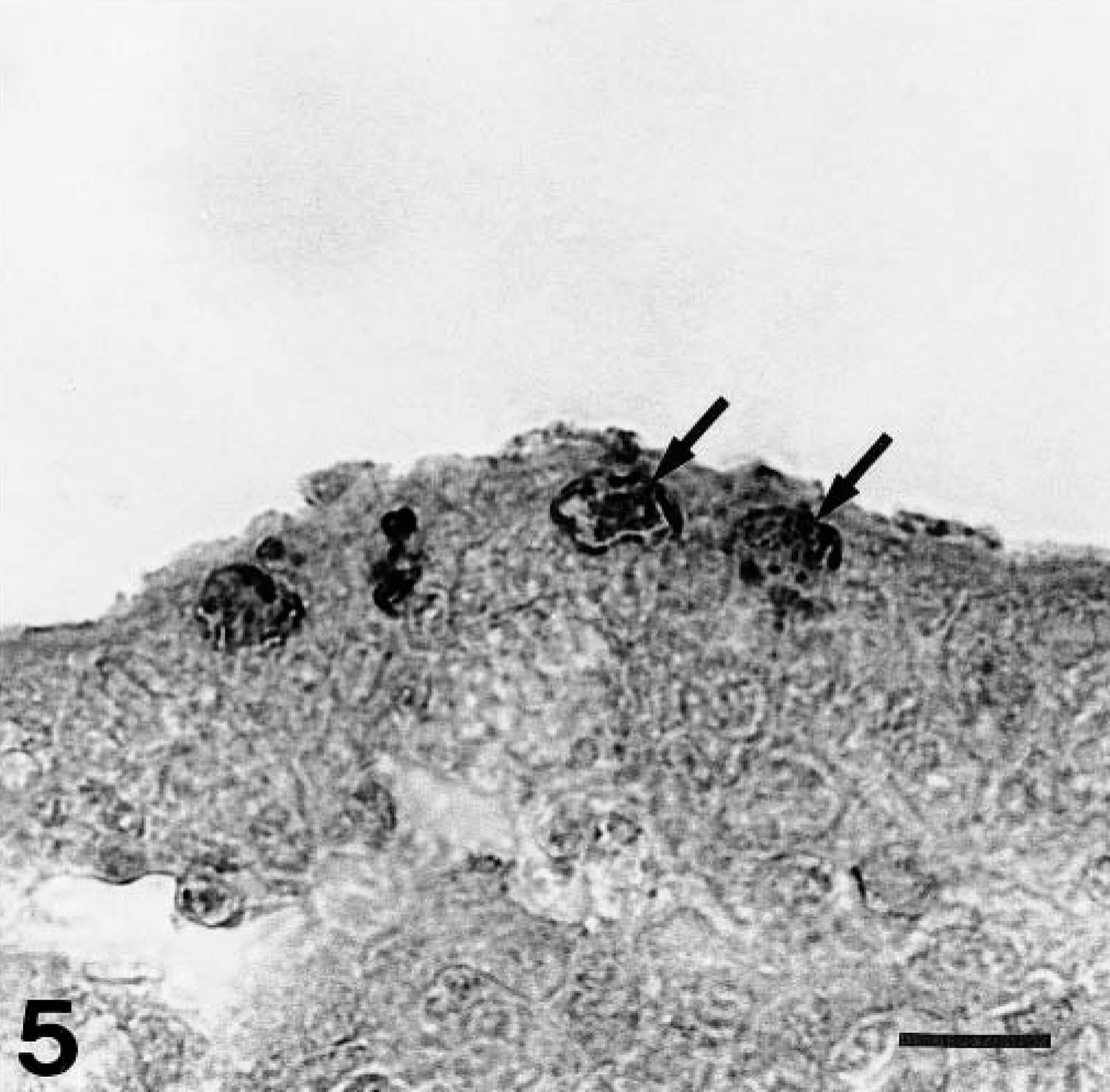
Cecal tonsil; a chicken from group 1 on day 14 PI. FAV antigen–positive intranuclear inclusion bodies are seen in the epithelial cells of the crypt (arrows). Immunohistochemical staining for the FAV antigen, methyl green counterstain. Bar = 5 μm.
Immunohistochemistry for FAV antigen
Intranuclear inclusion bodies of epithelial cells in the gizzard, ileum, and cecal tonsil were found to be positive for the FAV-99ZH antigen. In the gizzard, the degenerative epithelial cells and debris in the lumen of affected glands frequently showed positive reactions for the virus antigen.
Influence of maternal antibody
The gizzard lesions were recognized regardless of their maternal antibody titer levels. Macroscopic and microscopic lesions were observed in the inoculated chickens, even though they had received more than 1,280 maternal antibody titers. The chickens in group 3 were assigned to three subgroups according to their maternal antibody titer levels at 2 weeks of age. However, the characteristics of histologic and immunohistochemic findings, such as the FAV antigen–positive intranuclear inclusion bodies in the gizzard lesions, were observed in each subgroup as well, in all test periods and at high rates (Table 2). It was confirmed that all the chickens in group 4 did not have the maternal antibody. However, the gizzard lesions in group 4 were milder and of a lower prevalence than those in the other groups.
Influence of maternal antibody on histologic and immunohistochemical findings in the group three chickens.
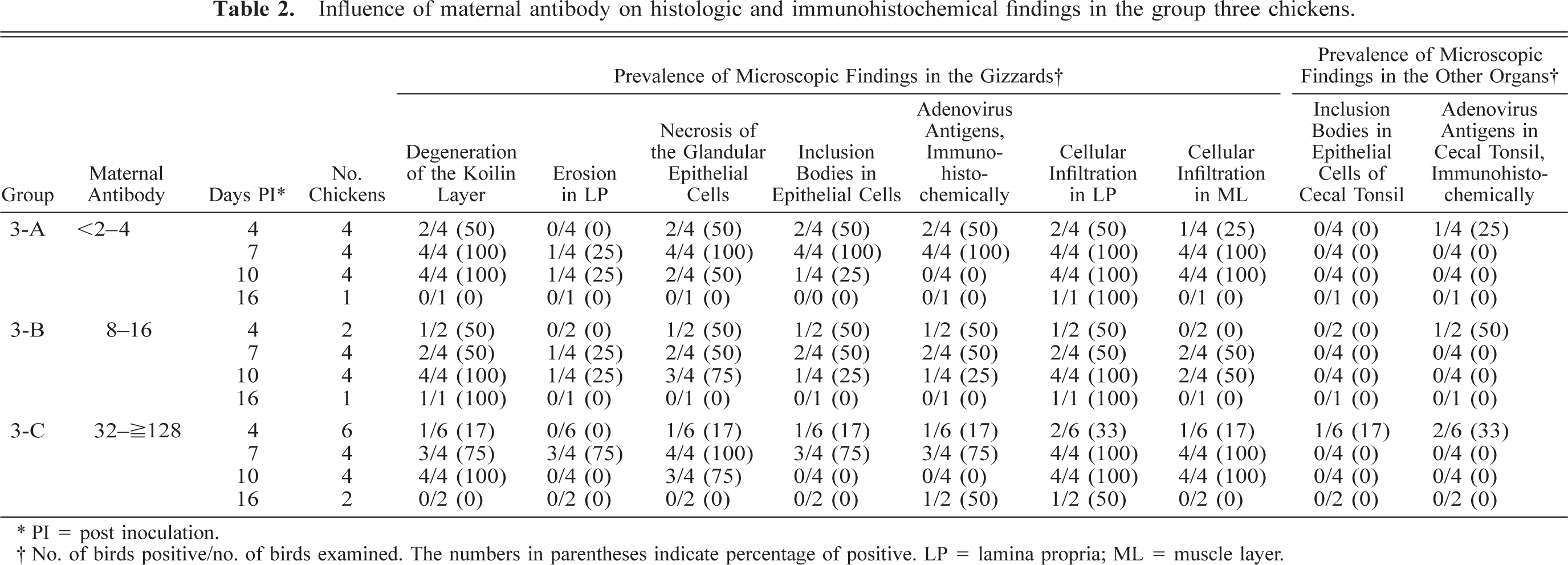
∗ PI = post inoculation.
† No. of birds positive/no. of birds examined. The numbers in parentheses indicate percentage of positive. LP = lamina propria; ML = muscle layer.
Identification of inflammatory cells in the gizzard lesions
Marked to severe infiltration of heterophils was observed in the necrotic lesions on days 10 and 18 PI in group 1, days 7 and 10 PI in group 2, and days 4 and 7 PI in group 3 by HE. In group 4, however, infiltration of the heterophils was milder (Table 3). Marked to severe infiltration of CD3-positive cells was observed in the necrotic or regenerative lesions from day 14 PI in group 1, day 7 PI in group 2, day 4 PI in group 3, and day 5 PI in group 4 (Fig. 6). CD3-positive cells were not observed in the intraepithelium. Infiltrated mononuclear cells on perivascular connective tissue in the submucosa or muscle layer were mostly CD3 positive. In the initial phase, in all groups except group 4, the necrotic lesions were occupied by heterophils, and CD3-positive cell infiltration was observed a little later. Lymphoid nodules in the lamina propria consisted of mostly CD3-negative cells. IgA-positive plasma cell infiltration in the necrotic or regenerative lesions was observed from day 10 PI in groups 1 and 2, day 4 PI in group 3, and day 5 PI in group 4. IgA-positive plasma cells in the lesions were grossly less even at the peak of the disease. Marked IgM-positive plasma cell infiltration in the necrotic or regenerative lesions were observed on day 18 PI in group 1, days 10 and 14 PI in group 2, and day 10 PI in group 3. IgY-positive plasma cells were detected in a larger number than IgA- or IgM-positive cells throughout the entire experimental period. IgA-, IgM-, or IgY-positive plasma cells were hardly observed in the histologically normal lamina propria of the gizzard in all the chickens examined. IgY-positive plasma cells also were observed in the perivascular connective tissue in the submucosa or muscle layer along with CD3-positive cells.
Identification of inflammatory cells in affected lamina propria of the gizzard.∗

∗− = Negative or a very few; + = slight; ++ = marked; +++ = severe.
† PI = post inoculation.
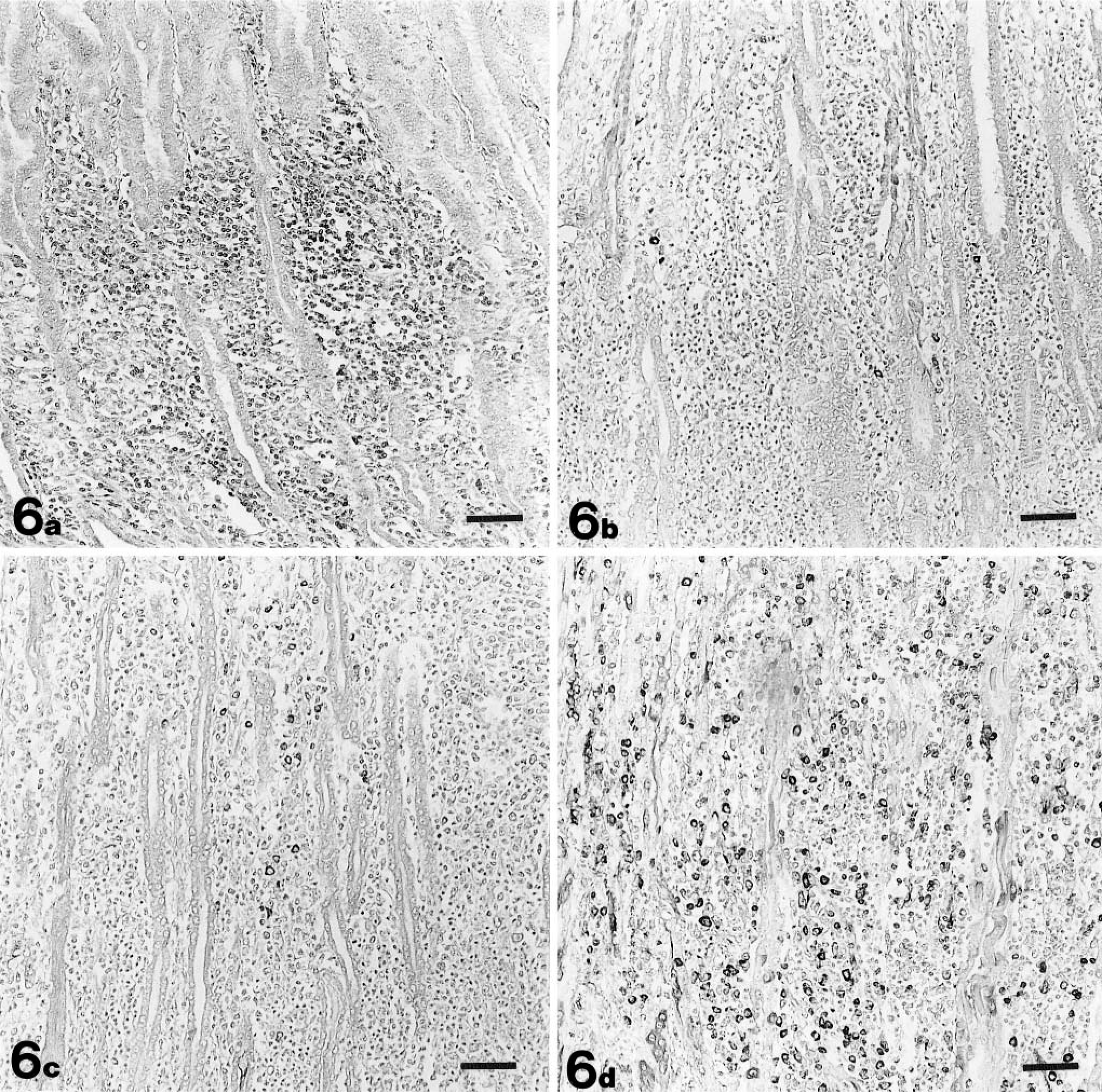
Gizzard; a chicken from group 3 on day 10 PI. Cells stained with CD3 ( Fig. 6a ), IgA ( Fig. 6b ), IgM ( Fig. 6c ), and IgY ( Fig. 6d ) in the lamina propria. Numerous CD3-positive cells are detected. IgA-positive plasma cells are recognized in only a few numbers. IgY-positive plasma cells are observed in larger numbers than IgA- and IgM-positive cells. Immunohistochemical staining for CD3, IgA, IgM, and IgY, methyl green counterstain. Bar = 50 μm.
Discussion
Experimental FAV-99ZH infection in 1-, 3-, and 5-week-old commercial broiler chickens resulted in gizzard erosion. Gross and histologic lesions in gizzards in the present experiment are similar to those of natural 2 , 31 or experimental cases using SPF white leghorn chickens. 28 However, infected broiler chickens tend to have more severe lesions, and the prevalence of lesions was higher than that in the SPF white leghorn chickens. The difference, to some extent, may be the result of differences in host susceptibility to FAV, or it may be due to the constitution of the birds.
Histologic changes in commercial broiler chickens by FAV-99ZH infection were revealed. Necrosis of the glandular epithelial cells, with intranuclear inclusion bodies that were immunohistochemically FAV antigen positive, was observed in the gizzard as an initial lesion. The inclusion in glandular epithelial cells of the gizzard is characteristic and is most useful for histopathologic diagnosis of the disease. However, the appearance term of the inclusion was of short duration, and even at climax, inclusion may not always be observed in the lesions. Marked cellular infiltration in the lamina propria of the gizzard was observed from the initial period, and the lesion lasted until the last day of the observation phase in each experimental group. Mononuclear cell infiltration in the perivascular connective tissue in the submucosa and muscle layer also was observed over the long term in each group. These findings were not reported for gizzard lesions produced by feeding diets containing fish meal, 16 high levels of histamine, 14 copper, 9 , 17 or mycotoxins. 15 , 23 Therefore, these findings may be available for presumptive diagnosis.
AGE was reproduced in all treatment groups in this study. However, there were some differences among the groups. Some experimental studies on IBH or HPS suggested that the appearance term or prevalence of lesions in the organs affected by FAV was influenced by various factors, such as the virulence or tissue tropism of the virus strain, the concentration of inoculum, and the inoculation route, age, or breed of the affected chickens. 7 , 18 , 24 Appearance of gizzard lesions occurred later in group 1 chickens than in the group 2 chickens that were inoculated with higher-dose inoculum at the same age. The delay of crisis caused by a low concentration of inoculum has been observed in other viral diseases. 8 , 24 The appearance of gizzard lesions occurred later in group 2 chickens than in the group 3 chickens that were inoculated at 3 weeks of age. However, the mechanism of the delay was not clear. The influence of maternal antibodies on infection with FAV serotype-1 was examined in this study. Gizzard lesions were observed in groups 2 and 3 chickens, irrespective of the maternal antibody titer. The chickens in group 2 that had 1,280 or more maternally derived neutralizing antibody titers at the time of viral exposure succumbed to FAV-99ZH infection. Several researchers have suggested the possibility of maternal antibodies preventing disease in chickens inoculated with FAVs that are associated with IBH. 7 , 12 , 33 Maternal antibody also influences the susceptibility of turkeys to infection with the hemorrhagic enteritis virus, which is classified as a group II avian adenovirus. 6 However, maternal antibody does not confer protection against some mucosal viral infections, such as infectious bronchitis (IB) or laryngotracheitis. 8 , 40 It is suggested that FAV-99ZH directly infected the gizzard mucosa and that the maternal antibodies in the blood were unable to prevent the mucosal lesion formation caused by FAV-99ZH. In group 4 the prevalence or severity of gizzard lesions was lower and the heterophil infiltration in the lesions milder than in the groups 2 and 3 chickens that were inoculated with the same titer of isolates. It is indicated that 5-week-old chickens have a higher resistance to FAV-99ZH infection than do younger chickens. However, because we did not continue our research in group 4 after day 10 PI, it is not clear whether this suggests a lower incidence or delay of the disease. We have not yet determined whether this result is related to the gradual maturation of immunologic competence.
Polyclonal CD3 is considered a highly specific pan–T-cell marker. 20 In this study polyclonal rabbit CD3 antiserum to human T cells was used for detection of CD3-positive cells in chicken gizzards after inoculation with FAV-99ZH. It has been confirmed that the anti-human CD3 T-cell serum can be used successfully in chicken and gives good results in formalin-fixed, paraffin-embedded tissue sections. 29 , 35 In this study numerous CD3-positive cells were observed in the gizzard lesions from the initial phase of the disease. This indicates the importance of local cell-mediated immunomechanisms in FAV-99ZH infection. It is well known that T cells mediate immunity against viral infection by a number of different mechanisms, including induction of cytotoxic activity, recognition of target antigens in conjunction with major histocompatibility complex, and production of lymphokines such as interferon-γ, interleukin-2, and tumor necrosis factor-β. An understanding of the differentiation of T-cell subsets is needed to gain insight into the cellular immune response in the gizzard lesion. It is not IgY but IgA that is considered critical in inducing resistance against most commonly occurring respiratory and intestinal infections. In this study, however, IgY-positive plasma cells were detected in a larger number than IgA-positive cells in gizzard lesions throughout the experimental period. The distribution of Ig-containing plasma cells in the mucosal lesion may be similar to the distribution in the respiratory tract of infectious bronchitis virus–infected chickens. 21 It is possible that IgY is more important than IgA in the gizzard lesion. The immune reaction in the gizzard mucous membrane of chickens is not clearly established.
Intranuclear inclusion bodies have been reported in epithelial cells of the digestive tract of chickens that were inoculated with FAV associated with HPS or IBH. 24 , 34 In this study, FAV antigen–positive intranuclear inclusion bodies were rarely observed in the ileum or cecal tonsils. However, chickens that are inoculated with FAV-99ZH have no typical HPS or IBH lesions in the liver, epicardium, or other organs. Under the conditions of this study, FAV-99ZH showed a tropism for and produced lesions largely confined to the gizzard or ventriculus mucosa. Thus, certain adenoviruses can be digestive pathogens even in the absence of adenoviral lesions in the liver, pancreas, or the development of hydropericardium.
