Abstract
Cancer immunosuppression that facilitates tumor progression and metastasis evolves by development of an immunosuppressive network. The aim of this study was to assess this network in dogs with benign or malignant tumors with or without confirmed metastasis. The authors showed that the number of various T cell subpopulations was constant during tumor development; however the number of regulatory T cells (Tregs) was significantly higher in tumor-bearing dogs than in healthy individuals. The number of myeloid-derived suppressor cells (MDSCs) and their p-STAT3 expression (which is a negative regulator of hematopoiesis and regulates VEGF expression) were higher in cancer patients than in control dogs, however their number increased significantly in late-stage cancer patients. Canine mammary carcinomas with confirmed metastases to either lymph nodes or internal organs had greater MDSCs and Treg infiltration than benign mammary tumors or malignant mammary tumors for which metastases had not been detected. Similarly, expression of p-STAT3 and VEGF-C was the highest in tumors with confirmed metastases. This research shows changes occurring in the blood (n = 30 patients) and tumor tissue of patients (n = 100) during canine mammary tumor development. The findings should be considered preliminary because of the small number of samples. Nonetheless, the findings suggest that a high level of Tregs and MDSCs as well as high expression of p-STAT3 and VEGF-C may significantly contribute to mammary tumor progression and metastasis in dogs.
Keywords
Cancer immunosuppression that facilitates tumor progression and metastasis evolves by development of an immunosuppressive network. Particularly important players of this network are T lymphocytes: CD4+ T helper cells which initiate antitumor immune response and CD8+ cytotoxic T cells which show tumor killing activity. 12,16 However, these killing functions can be influenced by regulatory T cells (Treg) which induce peripheral immune tolerance and promote tumor progression and development. This network has been an area of particular interest to develop novel therapies against tumors. 12,27 So far, several tumor-derived factors, such as interleukin-10 (IL-10), chemokine C-C motif ligand 2 (CCL-2), or vascular endothelial growth factor (VEGF) have been characterized as important compounds that facilitate tumor spread from the primary site to secondary organs. 31,32 Tumor-derived VEGF, the strongest angiogenic factor, acts as a chemoattractant that induces recruitment of myeloid cells from the bone marrow into peripheral vessels, and to the tumor site. 15 On the other hand, these recruited immature myeloid cells (eg, myeloid derived suppressor cells, MDSC) promote angiogenesis by activation of signal transducer and activator of transcription 3 (STAT3) which increases VEGF secretion and in turn is a negative regulator of hematopoiesis. It blocks immune cell maturation and increases their number in blood vessels and tumor mass. Moreover, impaired clearance of apoptotic cells, by tumor-associated macrophages observed within the tumor, induces antibodies targeting its own antigens and occurrence of pseudo-autoimmune status. 13,17,22 The increased concentration of autoantibodies as well as accumulation of MDSCs in tumor tissue also increase infiltration of the Tregs. 16
The main aim of the present study was to assess immunosuppression in dogs at various stages of mammary tumor development. We have assessed (1) the number of cells belonging to various T subpopulations, Tregs and MDSCs in blood of healthy and tumor-bearing dogs to estimate immunosuppression in dogs with benign and malignant mammary tumors with or without confirmed metastasis; (2) number of Tregs and MDSCs in tumor tissues as well as p-STAT3 and VEGF expression which are important regulators of tumor-related immunosuppression to draw a full picture of immune status of patients with different types of mammary gland malignancy.
Materials and Methods
Patients
Mammary-tumor-bearing dogs were divided into 3 groups: dogs with benign tumors, dogs with malignant tumors without detected metastasis, and dogs with tumors that metastasized to lymph nodes or other distant organs. Tumor masses were measured using external calipers. The greatest longitudinal diameter (length) and the greatest transverse diameter (width) were determined. The clinical examination of lymph nodes was performed and histological diagnosis was made to confirm the type of the tumor. In our study we have examined blood of 26 healthy dogs and 30 mammary-tumor-bearing dogs. We were able to obtain tissue samples of only 18 of these tumor-bearing dogs. Thus, for immunohistochemistry we used mammary cancer tissue samples from 18 of these dogs and 82 samples from our archives (in total, N = 100). The dogs’ owners gave written or verbal permission for the use of their animals’ blood and tissues for this work. Samples were obtained within study approved by the III Local Ethical Committee (approval no. 8/2012, 17 January 2012) of the Warsaw University of Life Sciences.
Blood Cell Analysis
Anticoagulated whole blood was collected for diagnostic purposes from healthy dogs (n = 26) or mammary-tumor-bearing dogs (n = 30) during routine veterinary procedures (Supplemental Table 1). Then, it was immediately subjected to mononuclear cell separation using Accuspin System-Histopaque 1077 (Sigma-Aldrich), according to the manufacturer’s protocol. For this study, blood from mammary-tumor-bearing dogs was used. Isolated mononuclear blood cells were divided into 3 tubes containing various antibody cocktails: (1) BD Pharmingen Dog T Lymphocyte cocktail, (2) anti-CD4 (APC conjugated), anti-FoxP3 (PE conjugated), anti-CD25 FITC (all obtained from eBiosciences), and (3) anti-Gr1 (RPE conjugated), anti-CD11b (FITC conjugated) and anti-CD33 either anti-p-STAT3 (APC conjugated) antibodies (all obtained from Thermo Scientific, USA), according to the manufacturer’s instructions. Leukocytes were initially identified and gated on the basis of morphological criteria (SSC vs FSC cytogram) using FACSAria II (BD Biosciences, USA). Subsequently, (1) CD3+/CD4+ and CD3+/CD8+; (2) CD4+/CD25+/FoxP3+ and (3) Gr1+/CD11b+/CD33+ or Gr1+/CD11b+ expressing p-STAT3 cells were gated and counted. 2,4,5,23
Immunohistochemical (IHC) Examination
Canine mammary tumor tissue sections were derived from the archives of the Department of Pathology and Veterinary Diagnostics, Faculty of Veterinary Medicine, Warsaw University of Life Sciences (Poland) and Instituto Zooprofilattico Sperimentale del Umbria e delle Marche (Italy). Samples were surgically collected during mastectomy from 100 female dogs of various breeds (including n = 18 samples obtained from dogs subjected to the blood analysis). Tissue samples were fixed in 8% neutral buffered formalin and routinely embedded in paraffin. Tumor samples included those diagnosed as benign (n = 30), malignant with no detectable metastases (n = 40), and malignant with local lymph-node, distal lymph-node, or internal organ or bone marrow metastases (n = 30). The presence of neoplastic cells in each metastatic site was histologically confirmed using hematoxylin and eosin (HE)-stained slides (in case of local metastases primary tumor was surgically removed with lymph node during mastectomy; in case of distant metastases, primary tumor and its metastatic site were removed after animal euthanasia). The tumors were diagnosed according to the World Health Organization Histological Classification and Mammary Tumors of the Dog and Cat classification with further modifications (Supplemental Table 2). 10,11,21
Sections of 4 µm were taken from paraffin tumor tissue blocks and were incubated overnight at 37°C. After dewaxing in xylene and rehydration in ethanol, antigens were retrieved by placing slides in 0.02 M citrate buffer (pH 6.0) and boiling in a decloaking chamber.
Samples were incubated in a peroxidase blocking reagent (Dako, Denmark) for 10 min at room temperature prior to 30 min incubation in 5% bovine serum albumin (Sigma Aldrich, Germany). Then, the primary anti-canine antibodies anti-G1 (1:100 dilution; Thermo Scientific), anti-FoxP3 (1:100 dilution; Thermo Scientific), anti-p-STAT3 (1:100 dilution; Thermo Scientific), anti-VEGF-C (dilution 1:100; Thermo Scientific) were diluted in 1% bovine serum and were applied to cells overnight at 4°C. Subsequently, an Envision kit comprising labeled polymers of secondary anti-rabbit/mouse antibodies conjugated with the HRP enzyme complex (Dako) was used. The 3,3′-diaminobenzidine (Dako) substrate was used to develop colored products. Finally, nuclei were counterstained with hematoxylin. Each IHC experiment was controlled by omitting primary antibodies on representative paraffin slides.
Three consecutive tissue sections were analyzed. Pictures of each slide were taken (10–20 depending on the sample size) using an Olympus BX60 microscope. Colorimetric intensities of IHC-stained antigen spots (brown precipitate reflecting antigen expression) were counted using a computer-assisted image analyzer (Olympus Microimage™ Image Analysis version 4.0 software for Windows, Center Valley, PA, USA). The results are presented as mean pixel IOD (Integrated Optical Density) related to color intensities of each p-STAT3 and VEGF-C antigen spot or the number of Gr1 either FoxP3-expressing cell.
Statistical Analysis
Statistical analyses were performed using Prism version 5.00 software (GraphPad Software, La Jolla, CA, USA). One-way analysis of variance (ANOVA) with Tukey’s honest significant difference post hoc test were applied, and differences were considered significant when
Results
Number of Selected Leukocytes in the Blood of Healthy and Tumor-Bearing Dogs
Flow cytometric analysis showed that the number of Th lymphocytes (expressing CD3 and CD4 antigens) in the blood of healthy dogs was 58.79% (SD = 15.03). In dogs with benign tumor their number was 59.2% (SD = 6.77), in dogs with malignant tumors without confirmed metastasis: 59.77% (SD = 7.48), and in dogs with tumors that gave local or distant metastases: 51.68% (SD = 12.05). No significant differences were observed between these groups (Fig. 1a).
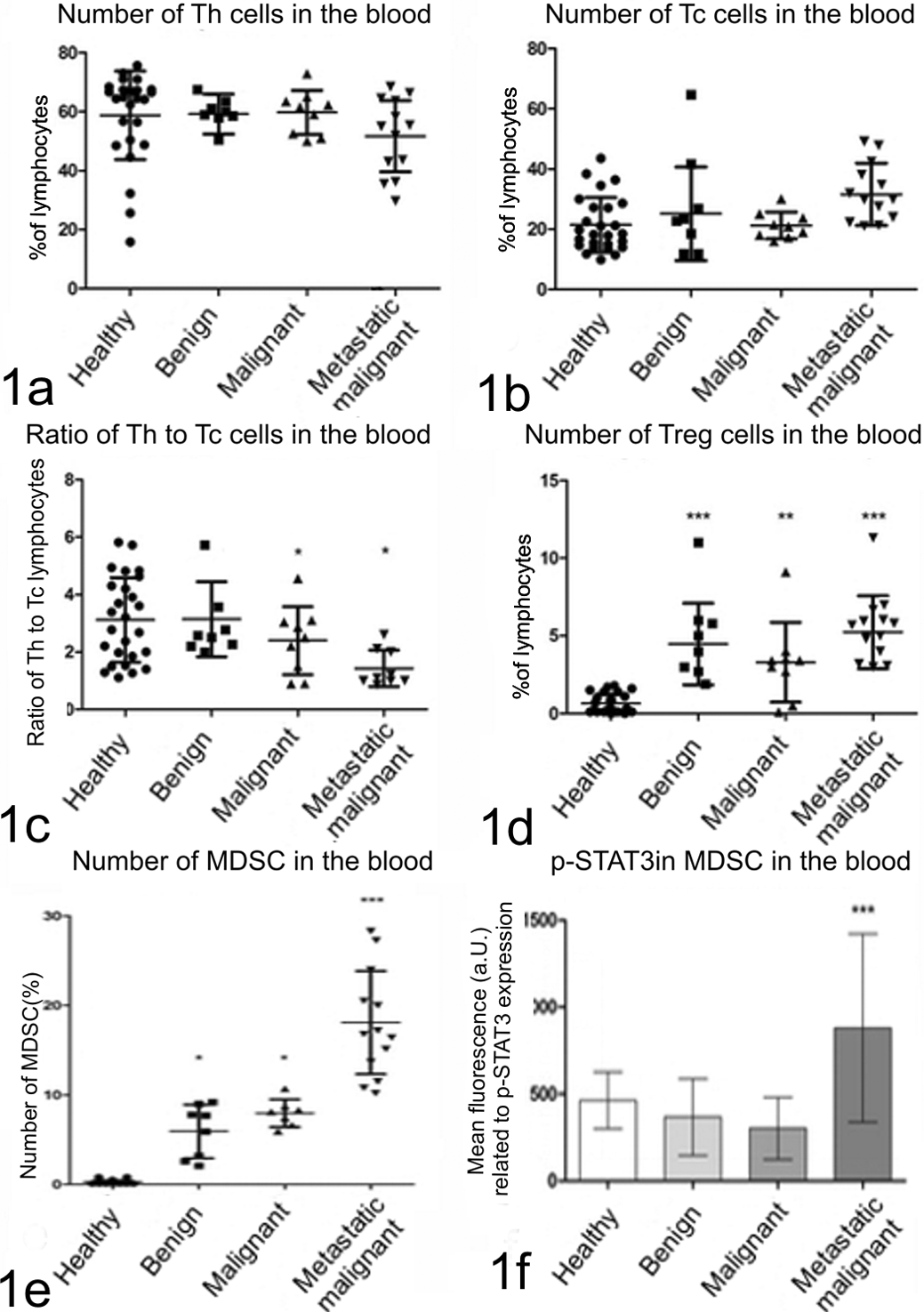
Graphs showing number of lymphocytes (1a) T helper (Th); (1b) T cytotoxic (Tc); (1c) Th to Tc ratio; (1d) T regulatory (Treg); (1e) myeloid-derived suppressor cells (MDSC) circulating in the blood; and (1f) p-STAT3 expression in MDSCs assessed using flow cytometry. Healthy dogs are compared with dogs with benign, malignant (without confirmed metastasis) and malignant (with confirmed metastasis) mammary tumors. The findings of decreased ratio of Th to Tc cells in dogs with malignant tumors (with and without confirmed metastasis), and increased number of Treg and MDSC cells in dogs with mammary tumors indicated immunosuppression. However, the level was the highest in dogs with malignant tumors with confirmed metastases (the highest number of MDSC and the highest p-STAT3 expression in these cells). One-way ANOVA + Tukey post hoc tests have been applied using GraphPad 5.0 (Prism). *
Similarly, no significant differences were observed in the numbers of Tc lymphocytes (expressing CD3 and CD8 antigen) between patient groups. Tc numbers in healthy dogs, and dogs with benign, malignant, and metastatic tumors were as follows (respectively): 21.44% (SD = 9.11), 25.16% (SD = 15.57), 21.26% (SD = 4.42), and 31.61% (SD = 10.39) (Fig. 1b).
Despite no differences having been found in the numbers of Th and Tc lymphocyte between patients at various stages of tumor development, the ratio of Th/Tc lymphocytes significantly differed (
The number of blood Treg cells (expressing CD4, CD25, and FoxP3 antigens) differed in all tumor-bearing dogs compared to healthy animals. In healthy dogs the number of Treg was 0.64% (SD = 0.61) of all lymphocytes, whereas in dogs with benign tumors, malignant tumors without confirmed metastasis and malignant tumors with metastases their numbers differed significantly and were as follows: 4.47% (SD = 2.36,
Number of MDSCs (CD11b+/Gr1+/CD33+) in the blood leukocyte population of healthy dogs was 0.25% (SD = 0.19). In dogs with benign and malignant non-metastatic mammary tumors, the MDSC number was significantly higher: 5.93% (SD = 2.99) and 7.97% (SD = 1.58;
Number of Gr1+, FoxP3+ Cells and Level of p-STAT3 and VEGF-C Expression in Canine Mammary Tissues
Microscopic analysis showed that the number of Gr1-positive cells was significantly higher (
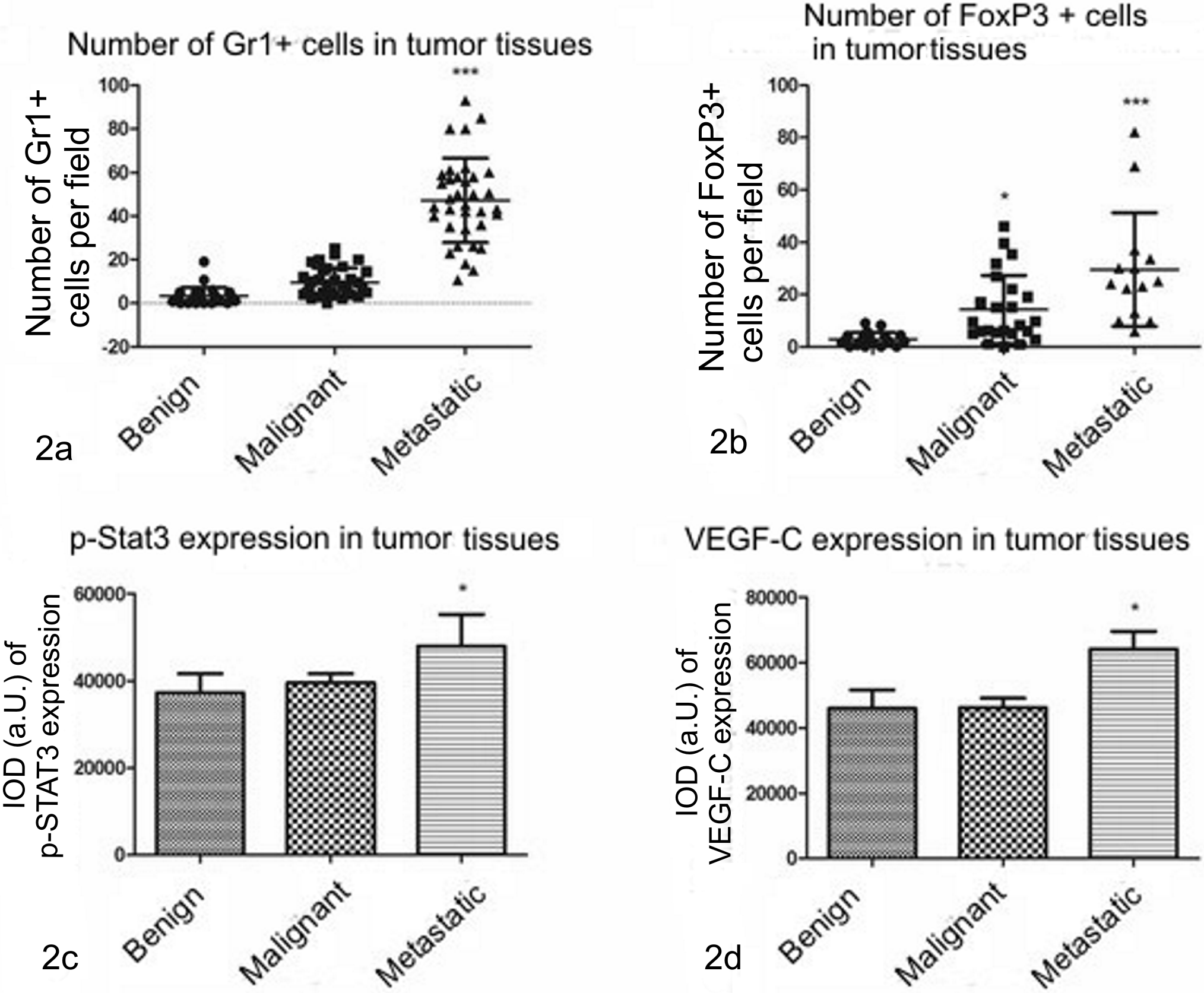
Graphs showing number of Gr1-positive cells (2a), FoxP3-positive cells (2b), p-STAT3 (phosphorylated Signal Transducer and Activator of Transcription 3) (2c), and VEGF-C (Vascular Endothelial Growth Factor C) (2d) expression in canine mammary tumors (benign n = 30, malignant nonmetastatic n = 40 and malignant metastatic n = 30). The given values represent number of cells per field (2a and 2b) or Integrated Optical Density of antigen staining (2c and 2d). In dogs with malignant tumors with confirmed metastases, the number of Gr1-positive and FoxP3-positive cells as well as p-STAT3 and VEGF-C expression are significantly higher than in benign or malignant nonmetastatic tissues. One-way ANOVA + Tukey post hoc tests are applied (GraphPad Prism 5.0). *
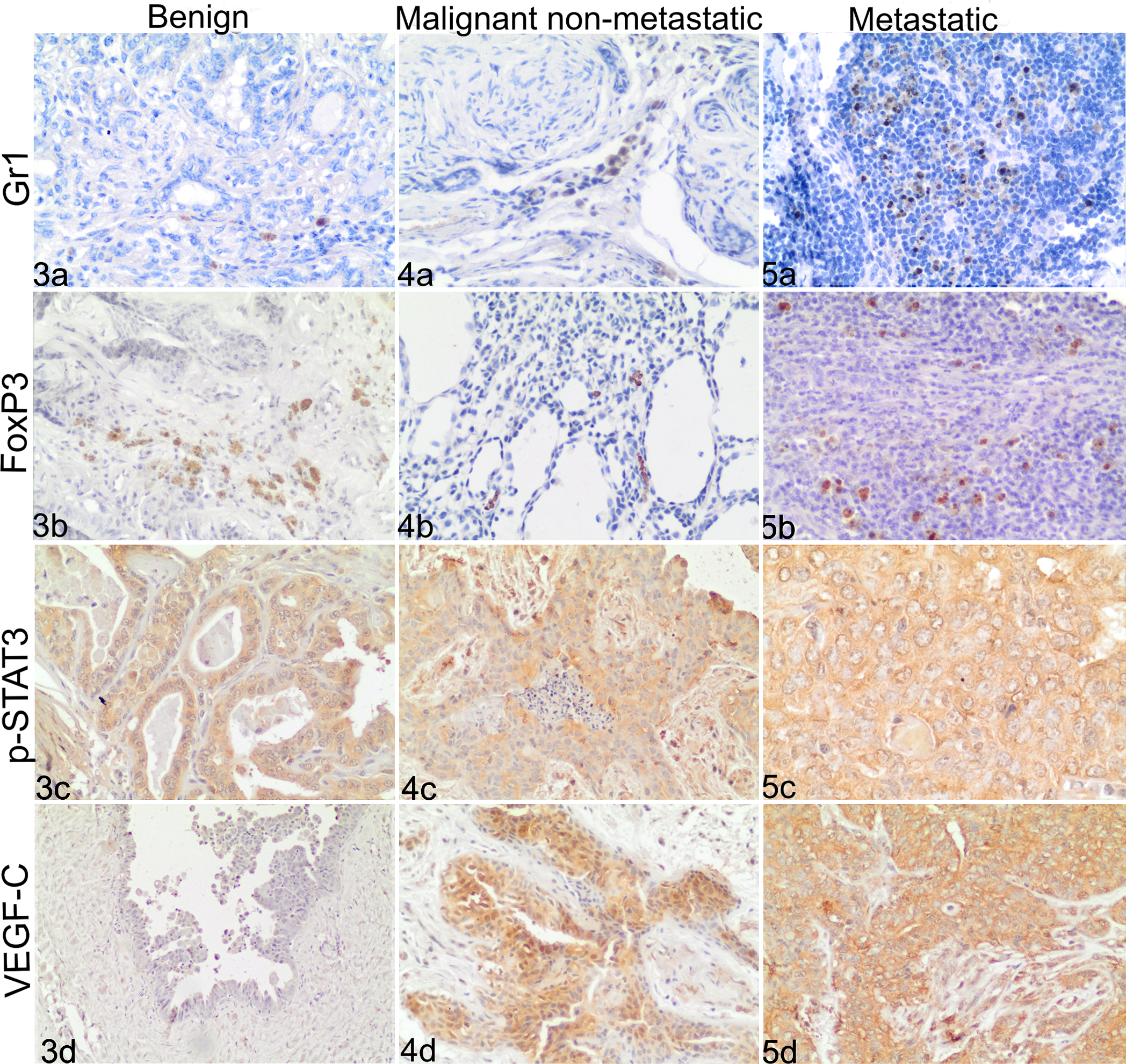
IHC staining, Canine mammary tumors: a = Gr1, b = FoxP3, c = p-STAT3, d = VEGF-C .
Number of FoxP3-expressing cells in benign mammary tumors was 2.893 cells per field (SD = 0.5801), in malignant nonmetastatic tumors it was significantly higher (
The expression of p-STAT3 was found in cancer cells and also in hematopoietic cells but not in the other stromal cells. Significantly higher expression (
Expression of VEGF-C in benign tumors was 45,993 a.U. (SD = 5,639), in malignant nonmetastatic tumors IOD was 46,217 (SD = 2,887), however it was the highest in malignant metastatic tumors (
In 18 dogs we analyzed the level of correlation between number of MDSC and Treg cells in blood and mammary cancer tissues (defined in tissues as Gr1+ and Foxp3+ cells, respectively) (Table 1). We found significant correlation between number of MDSC in blood and tissues of patients with benign tumors (
Correlation Between Number of MDSC (Defined in Tissues as Gr1+) and Treg (Defined in Tissues as Foxp3+) Cells in Blood and Cancer Tissue of Mammary-Tumor-Bearing Dogs (n = 18/30).

MDSC, myeloid-derived suppressor cell.
Discussion
The present study is the first attempt to assess the number of selected subpopulations of peripheral blood leukocytes in dogs at various stages of mammary cancer development. Our findings should be considered preliminary due to the small number of blood samples from patients with mammary cancer. 18,19 This is an unexplored field of veterinary sciences, and even the reference ranges of some subpopulations of blood cells are not arbitrary standardized for healthy animals. Moreover, significant differences exist between published reports which are very likely related to the various antibodies used and different cell gating during the analysis. Some authors indicated that the percentage of peripheral blood immune cells in dogs depends on the breed, age, and sex. According to the published data, in healthy dogs the number of Th (CD3+/CD4+) lymphocytes is 30.9%–55.1% and Tc (CD3+/CD8+) lymphocytes is 15.9%–28.9% . 9,29,30 In our study the values of Th and Tc lymphocytes in healthy dogs were similar to these published by other authors (58.78% and 21.44%). Surprisingly, we found that the values of Th and Tc cells did not significantly change between healthy individuals and tumor-bearing dogs. However, it is important to note that we did not assess the activity of these lymphocytes (which may vary between examined groups) but their numbers only.
The ratio of CD4 to CD8 lymphocytes may be an indicator of immunosuppression and in healthy dogs is usually between 1.7 and 2.8. Results of our study showed value 3.11 (SD = 1.47) in healthy animals and we did not observe any significant changes between healthy dogs and patients with benign and malignant tumors that had not metastasized. Interestingly, although the number of CD4 and CD8 lymphocytes did not differ between various groups of dogs, we observed decreased CD4/CD8 ratio (1.43) in animals with malignant tumors that had local or distant metastasis. Similar results were described
26
in breast cancer patients. The average CD4/CD8 ratio gradually decreased in various patient groups from healthy donors, to dogs with advanced tumors. It can be explained as the immunosuppressive effect of Treg lymphocytes. The published numbers of Tregs in dogs are different between various authors (mainly due to various antigens that are used for their identification).
9,28,29,30
Risetto
24
showed that in healthy dogs the number of cells expressing FoxP3/CD25/CD4 antigens (we used the same antibodies in our study) is below 1%, similar to our findings (0.64%, SD = 0.61). In tumor-bearing animals, the number of Tregs was significantly increased at all stages of mammary tumor development. Moreover, their numbers in blood of animals with tumors with local/distal lymph node metastases or metastases to internal organs or bone marrow significantly correlated with their numbers in cancer tissues (
Treg infiltration in tissues has been shown by previous authors to statistically correlate with high histological grade of canine mammary tissues and evidence of lymphatic invasion. 12 Tumor-specific Treg frequently accumulate in tumors, inhibiting immune function of other inflammatory cells infiltrating the tumor mass in a similar way as they act in the blood. 3,6,14 Thus, they are negative regulators of T cell immune responses and they can suppress the proliferation and activity of both CD4 and CD8 cells inhibiting cytokine secretion. Also Treg interactions with MDSC are very important in tumor progression. 14,20 Our results (partially published and discussed before) 20 showed that the number of circulating MDSCs was significantly higher in dogs with benign mammary tumor and malignant tumors compared with healthy dogs; however, their number was the highest in dogs with mammary cancer with confirmed metastasis to lymph nodes or distant organs. 20 Moreover, their number in blood of patients with advanced tumors significantly correlated with their increased infiltration in mammary tumors with local/distal lymph node metastases or metastases to internal organs or bone marrow.
Interestingly, we showed that in patients with late-stage tumor (malignant tumor with confirmed metastasis) expression of p-STAT3 in circulating MDSCs significantly increased compared to healthy dogs or dogs with benign tumors or malignant tumor without confirmed metastasis. Similarly, in cancer tissues the number of Gr1+ cells and their p-STAT3 expression were significantly higher in tumors with local/distal lymph node metastases or metastases to internal organs or bone marrow than in benign or malignant tumors with no detectable metastases. STAT3 is a negative regulator of hematopoiesis 25 and its higher expression may lead to spread of MDSC in cancer tissue. 14 These tumor-infiltrating myeloid precursors enhance angiogenesis by secretion of various angiogenic factors (mainly VEGF) which is mediated by p-STAT3. 14 On the other hand, VEGF promotes rapid phosphorylation of STAT3. 1,14 In our study we showed similar tendency in expression of STAT3 and VEGF—higher in tumors with local/distal lymph node metastases or metastases to internal organs or bone marrow than in benign tumors or tumors with no detectable metastases.
The other mechanism by which MDSCs contribute to cancer progression is immunosuppression via inhibition of T cell activity in a similar way as Treg. 7 Although there are no published results in this field in canine mammary cancer, it has been shown that in tumors with local/distal lymph node metastases or metastases to internal organs or bone marrow, infiltration of T lymphocytes was significantly lower than in tumors with no detectable metastases. 8
To summarize, our findings suggest that MDSCs and Treg contribute to cancer progression by induction of immunosuppression and increase of angiogenesis. Both mechanisms are regulated by STAT3 and VEGF.
Footnotes
Acknowledgements
We would like to thank Mrs Małgorzata Bulkowska for her technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant UMO-2013/09/D/NZ5/02496 from the National Science Centre. Some of the preliminary results have been previously published; 14,20 however experiments conducted within present study included more or different samples.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
