Abstract
Cardiorenal syndrome involves disease and dysfunction of the heart that leads to progressive renal dysfunction. This study investigated the relationship between cardiac and renal disease in 91 aged chimpanzees at the Alamogordo Primate Facility by evaluation of the medical histories, metabolic parameters, functional measurements of the cardiovascular system, clinical pathology, and histopathology focused on the heart and kidney. Cardiac fibrosis was the most frequent microscopic finding in 82 of 91 animals (90%), followed by glomerulosclerosis with tubulointerstitial fibrosis in 63 of 91 (69%). Cardiac fibrosis with attendant glomerulosclerosis and tubulointerstitial fibrosis was observed in 58 of 91 animals (63%); there was a statistically significant association between the 2 conditions. As the severity of cardiac fibrosis increased, there was corresponding increase in severity of glomerulosclerosis with tubulointerstitial fibrosis. Altered metabolic, cardiovascular, and clinical pathology parameters indicative of heart and kidney failure were commonly associated with the moderate to severe microscopic changes, and concurrent heart and kidney failure were considered the cause of death. The constellation of findings in the chimpanzees were similar to cardiorenal syndrome in humans.
Keywords
As the population of captive chimpanzees in zoos, sanctuaries, and research facilities in the United States becomes geriatric, age-related diseases are becoming more apparent, and management of the chimpanzee colonies requires better understanding of these diseases. The most commonly reported age-related disease in captive chimpanzees leading to morbidity and death is chronic heart disease (CHD). Chronic renal disease, hypertension, and diabetes are diagnosed with increased frequency in aging captive chimpanzees; however, there is limited information regarding the association of heart disease with these other diseases. 5,15,16,43 We reviewed the medical and necropsy records and histopathologic changes of chimpanzees from the Alamogordo Primate Facility (APF) and identified a high prevalence of functional and microscopic changes in the heart and kidneys. These changes were suggestive of the cardiorenal syndrome described in geriatric humans, in which disease and dysfunction of the heart precede and lead to progressive kidney dysfunction. 11,47 As one organ progresses toward failure, the other becomes increasingly dysfunctional, contributing to decline in the first in a reciprocal process, frequently in conjunction with hypertension, arteriosclerosis, diabetes, and/or obesity. 47 Common changes described for the cardiorenal syndrome include chronic inflammation and increased extracellular matrix (ECM; fibrosis) that effaces heart and kidney parenchyma. The objective in this retrospective study was to characterize this cardiorenal-like syndrome in aged chimpanzees based on observed changes that were commonly identified in the animals at the APF.
Material and Methods
Body condition scores (BCSs), hematology, clinical chemistry, urinalysis, and clinical diagnosis of CHD and chronic kidney disease (CKD) were tabulated from the medical records of 93 chimpanzees housed at the APF from 2001 to 2015. Their necropsy reports were reviewed for diagnoses of cardiac fibrosis (CF), glomerulosclerosis with tubulointerstitial fibrosis (GS), and other diseases at the time of death. Their age, sex, terminal body weight, and ascribed causes of death were tabulated. Histologic sections of the kidney and heart were reviewed by one of the authors (J.C.) and assigned numerical severity grades based on the morphologic grade. Severity grades were correlated to numeric values of 1 (minimal changes), 2 (mild changes), 3 (moderate changes), and 4 (severe changes). CF was characterized by accumulation of ECM with progressive loss of cardiomyocytes and inflammation. GS was characterized by increased accumulation of ECM and progressive loss of renal corpuscles and tubules among fibrous tissue and inflammation. Arteriosclerosis was defined by thickened arterial tunica intima and/or media or atherosclerosis in one or multiple locations of the body, as identified in the necropsy report. Reference ranges for hematologic and serum clinical chemistry values had been published for adult chimpanzees in captivity and were utilized for evaluation of values from the animals at the APF. 26 Clinical pathology parameters evaluated included albumin, urea nitrogen, creatinine, phosphorus, calcium, hemoglobin, and hematocrit. CHD was diagnosed according to an abnormal rate or rhythm on auscultation of the heart during examination, conducted by a board certified veterinary cardiologist, and by conduction abnormalities on electrocardiography, abnormal chamber features on echocardiography, and cardiac enlargement on radiographs. CKD was defined by increased urea nitrogen and creatinine and by hypoalbuminemia. Diabetes was defined by increased fasting blood glucose and hemoglobin A1c based on published reference values. 39 Obesity was determined by the following BCSs: 1–2, underweight with palpable bony prominence; 3, ideal weight; 4–5, overweight with palpably increased subcutaneous body fat. Hypertension was defined with previously determined parameters separated by sex and, for females, by body weight. 17 Blood pressure (BP) was measured under anesthesia with an automatic oscillometry monitoring device (Passport 2 Patient Monitor; Datascope Corporation, Montvale, NJ, USA). For females with BCSs of 2–3, the mean systolic BP was 128 mm Hg (up to 144 mm Hg); for obese females with BCS ≥4, the mean systolic BP was 140 mm Hg (up to 258 mm Hg). For males, body weight had no impact on BP change, and the mean systolic BP was 147 mm Hg (up to 165 mm Hg). 16,17
Statistical significance of severity grades as paired numerical variables was determined with the Spearman rank coefficient with 95% confidence interval by Fisher z transformation. Associations and correlations of population parameters were statistically evaluated by odds ratio (OR) and risk ratio (RR) with chi-square analyses at 99% confidence interval.
Chimpanzees at the APF were maintained in strict accordance with the Guide for the Care and Use of Animals. 4 The facility and its program have been fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. The chimpanzees were maintained in same-sex group housing and fed a commercial primate diet (Purina Lab Fiber-Plus Monkey Diet Jumbo 5050). Same-sex housing was maintained to ensure the National Institutes of Health’s policy for breeding moratorium. Each animal received a complete physical examination and dental prophylaxis annually under anesthesia (tiletamine hydrochloride-zolazepam 3.0 mg/kg [50 mg/mL of tiletamine hydrochloride and zolazepam hydrochloride]), with evaluation by hematology, clinical chemistry, electrocardiogram, echocardiogram, abdominal ultrasound, tuberculosis testing, and BP measurement.
Results
Signalment
Medical records and necropsy reports for 60 males and 31 females contained sufficient information for evaluation. Remaining reports (2 animals) were disallowed for the study due to missing heart or kidney tissues. The median age in years at time of death was 30.5 (range, 10–53) for males and 26.5 (range, 16–51) for females, with 1 animal of unknown age.
Pathologic Findings
CF was identified in 56 of 60 males (93%) and 26 of 31 females (84%). CF ranged from minimal, characterized by focal to regional small foci of myocardial fibrosis, to severe with extensive loss of myocardium with replacement by fibrous matrix, variable numbers of inflammatory cell infiltrates, and disorganization of cardiomyocytes within areas of fibrosis (Figs. 1–3). GS was observed in 25 of 31 females (81%) and 38 of 60 males (63%). Severity ranged from minimal, characterized by multifocal annular fibrosis of renal corpuscles with little interstitial fibrosis and low-level inflammatory cell infiltrates, to severe with extensive loss of parenchyma, replacement by fibrous tissue, and large aggregates of inflammatory cells throughout the kidneys (Figs. 4–6). Concurrence of CF and GS was identified in 36 of 60 males (60%) and 22 of 31 females (58%), and this association as simultaneous events was significant (OR = 3.07, P < .001). In general, as the severity of CF increased, there was an increase in GS severity in animals with both conditions (Spearman rank correlation coefficient = 0.5, CI = 0.38–0.78). Animals with GS graded as minimal to mild had increased odds of death from heart failure as compared with animals without GS (OR = 1.4, P < .001). Arteriosclerosis occurred with both CF and GS in 23 of 31 (74%) of the animals (OR = 1.9, P < .001) and had significant association with hypertension (OR = 2.5, P < .001).
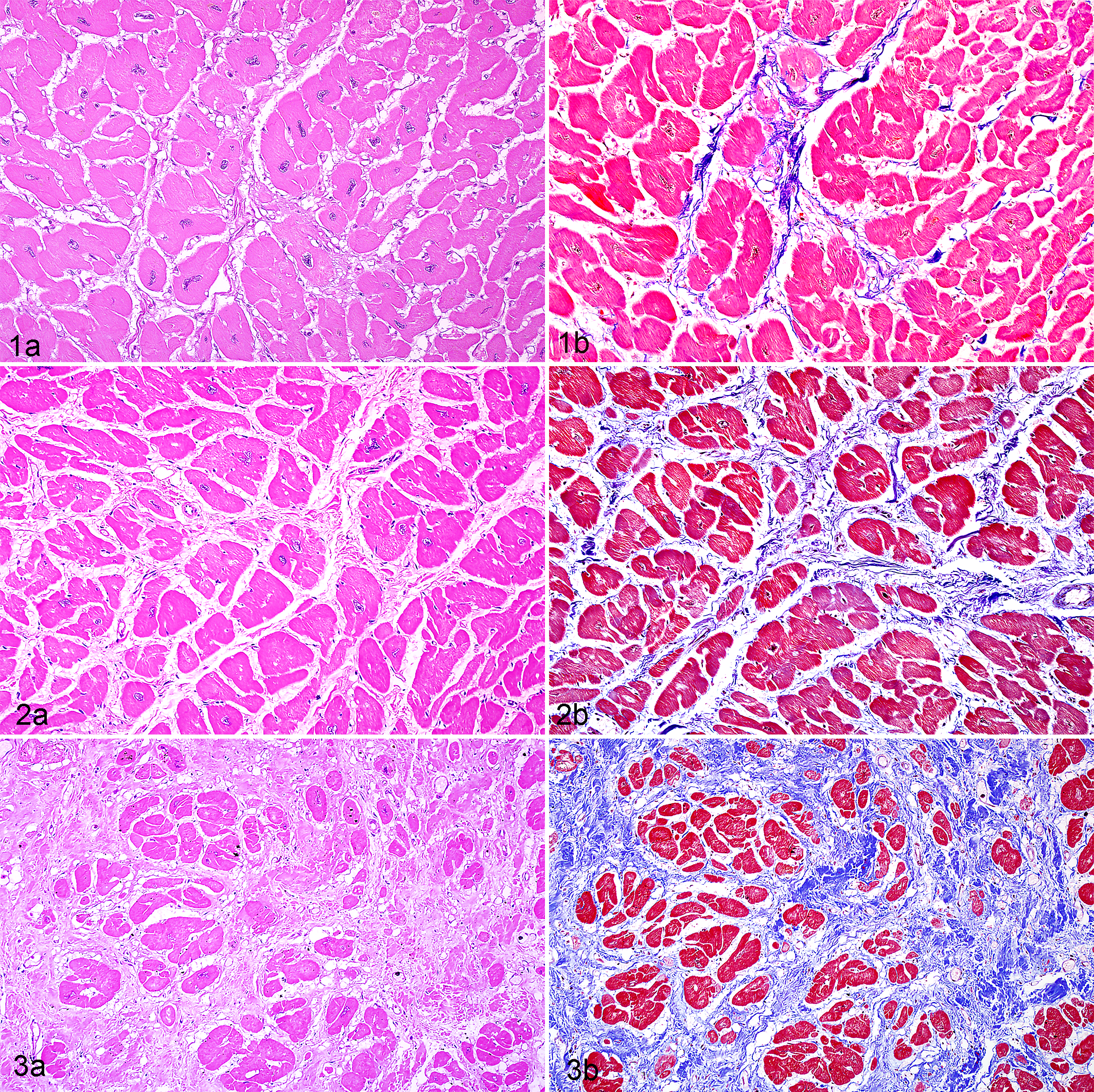
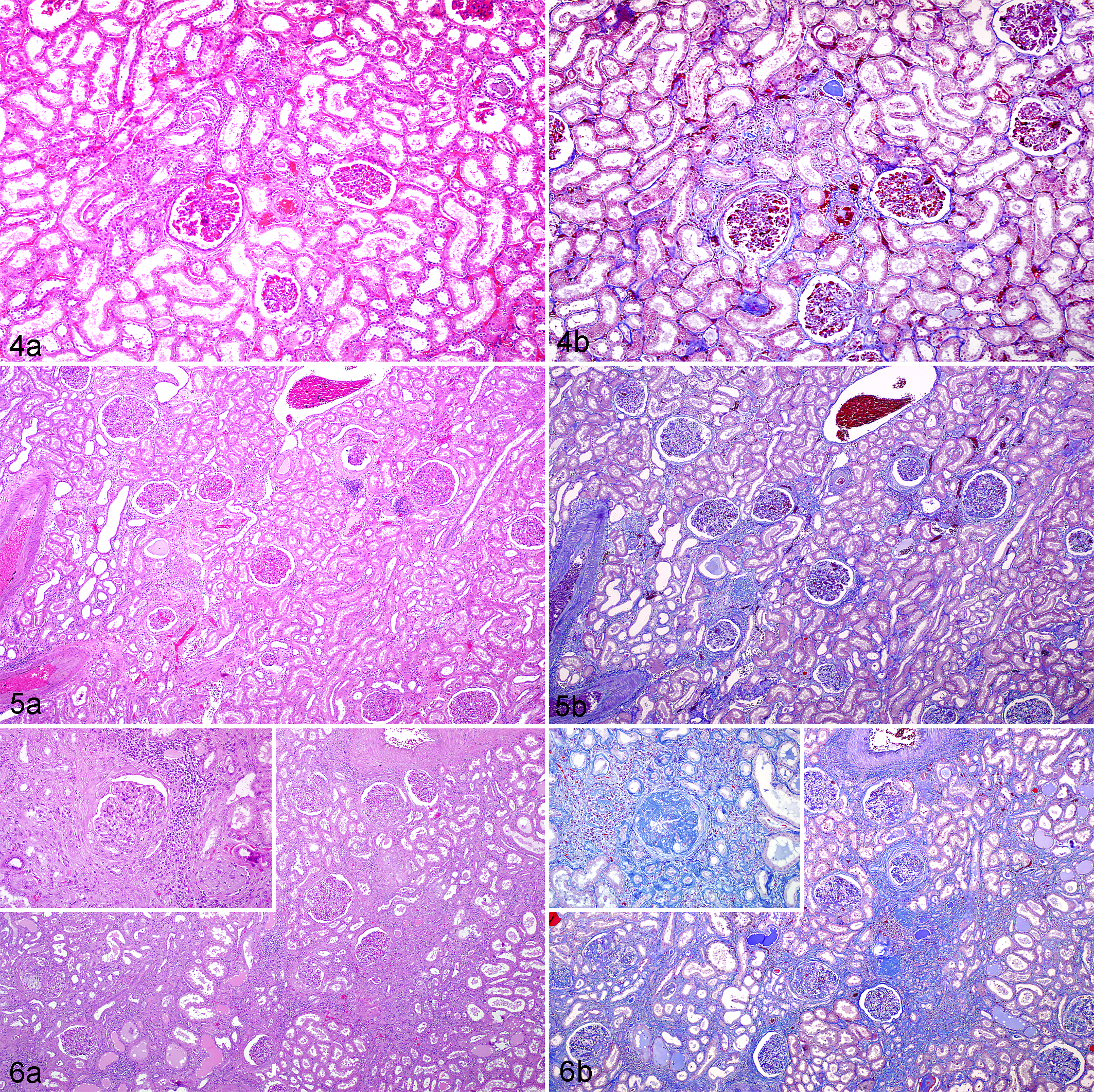
Clinical Findings
Many of the animals (40 of 91, 44%) died spontaneously or were euthanized primarily due to suspected or confirmed CHD. The combination of CHD and CKD, commonly with moderate to severe fibrosis in both organs histologically, was the second-most common cause of death (8 of 91, 9%). In some cases, animals with CHD without evidence of CKD (ie, normal serum urea nitrogen and creatinine) had microscopic fibrotic changes in the kidney at a statistically significant level (OR = 1.7, P < .001). Of the 5 animals (4 female, 1 male) recorded to have died from CKD without clinical CHD, 2 had CF microscopically. Females had statistically significantly increased risk of death from CKD as compared with males (RR = 3.8, P < .001).
Hypertension occurred in 12 animals, all of which had CF, GS, or both. Of these, 10 had both CF and GS; 5 died spontaneously of cardiac arrest; 1 was euthanized because of end stage CHD; and 3 died or were euthanized for both heart and kidney failure. The 5 females with hypertension had both CF and GS; of these, 3 had been diagnosed with metabolic syndrome prior to death based on BCS ≥4, fasting hyperglycemia, reduced levels of high-density lipoprotein, and hypertension. 43 A BSC of 4 (overweight) was identified in 6 of 31 females and 2 of 60 males, of which 5 females and 1 male had concurrent CF and GS. Three females that died or were euthanized due to concurrent CHD and CKD had moderate to severe CF and GS microscopically. All 3 had been diagnosed with nephritic syndrome antemortem, characterized by increases in urea nitrogen (>15 mg/dL), creatinine (>1.2 mg/dL), protein:creatinine ratio (>1.0 mg/dL), cholesterol (>190 mg/dL), triglycerides (>250 mg/dL), decreased serum albumin (≤3.8 g/dL), and tissue edema. 43
Clinical Pathology Findings
There were 74 animals (47 males and 27 females) that had hematology and clinical chemistry parameters available for evaluation within 4 months of death. Of those 74 animals, 16 (22%) had CF (12 males and 4 females); 3 (4%) had GS (1 male and 2 females); 52 (70%) had CF and GS (32 males and 20 females); and 3 (4%) had neither CF nor GS (2 males and 1 female).
Hypoalbuminemia was significantly correlated with CF, GS, and both (ORs = 2.4, 2.0, and 2.7, respectively, P < .001). Hypoalbuminemia was also significantly correlated with chronic inflammatory conditions other than GS or CF (OR = 54, P < .001). The severity of hypoalbuminemia was similar in chimpanzees with both CF and GS (mean, 2.3 g/dL) or only CF (mean, 2.2 g/dL); however, the risk of death from cardiac or renal disease was not associated with hypoalbuminemia (RR = 0.65, P < .001).
Minimally to markedly increased serum urea nitrogen and/or creatinine was present in 2 of 3 (67%) animals with GS, 10 of 16 (63%) animals with CF, and 17 of 52 (33%) animals with both CF and GS. In animals with CF and without microscopic changes of GS, the increased urea nitrogen and/or creatinine was considered to be due to reduced renal perfusion secondary to cardiac insufficiency.
Thirty-nine percent of animals had mild to moderate anemia characterized by decreased hemoglobin and/or hematocrit. Anemia was observed in 2 of 3 animals with GS (67%), 23 of 52 animals with both GS and CF (44%), and 3 of 16 animals with only CF (19%). Animals with GS were at increased risk of having anemia (RR = 1.4, P < .001). As GS increased in severity, risk of anemia increased (RR = 1.9, P < .001), but risk was not increased in animals with CF (RR = 0.62, P < .001), and anemia did not predict poor outcome from CHD or CKD as compared with animals without anemia (RR = 0.6, P < .001). Distribution of CF and GS by sex and associated conditions is presented in Table 1.
Percentage Population Distribution of Cardiac Fibrosis and Glomerulosclerosis With Associated Clinical Pathologic Changes and Diagnoses in a Colony of Captive Chimpanzees.a
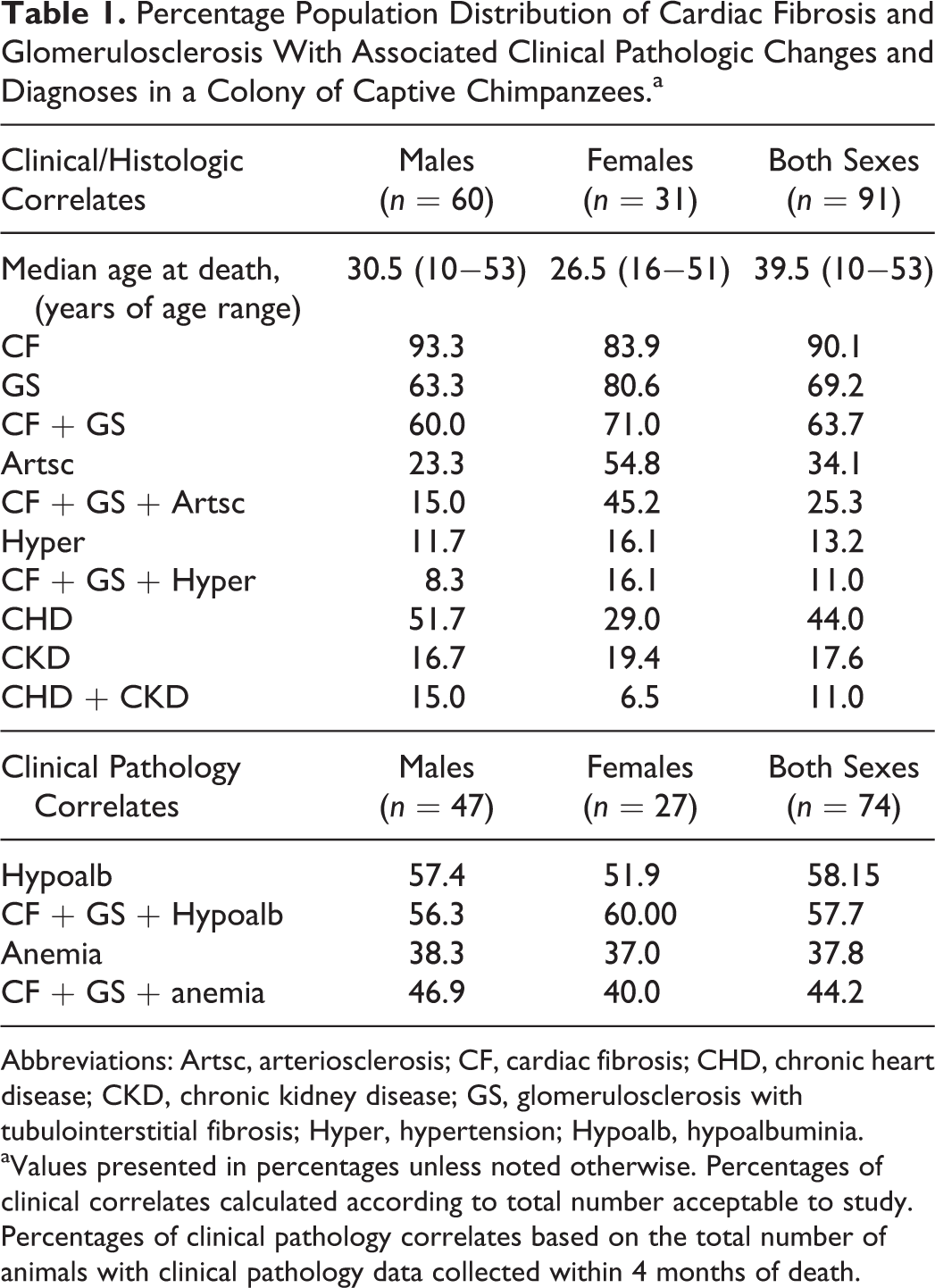
Abbreviations: Artsc, arteriosclerosis; CF, cardiac fibrosis; CHD, chronic heart disease; CKD, chronic kidney disease; GS, glomerulosclerosis with tubulointerstitial fibrosis; Hyper, hypertension; Hypoalb, hypoalbuminia.
aValues presented in percentages unless noted otherwise. Percentages of clinical correlates calculated according to total number acceptable to study. Percentages of clinical pathology correlates based on the total number of animals with clinical pathology data collected within 4 months of death.
Discussion
CF was the most common microscopic change, followed closely by GS in aged chimpanzees at APF, and these conditions often occurred concurrently. CHD was the primary cause of moribundity and death, followed by CKD or concomitant CKD and CHD. Chimpanzees have been reported to be prone to both CHD and CKD; however, the relationship between cardiac and renal disease in these animals has not been previously described. 5,43 Increased cardiac risk associated with CKD has been well documented in humans. 6,36,53 In human patients with mild kidney disease, the risk of cardiovascular events and death are increased relative to patients without kidney disease 33 ; the more severe the CKD, the greater the risk of heart failure. 23,24 Findings in our study were similar: as the severity of GS increased, there was increased severity of CF. Animals with GS had increased incidence of moderate to severe CF and increased risk of death from heart failure, as compared with animals without GS. Even animals with minimal to mild GS had increased risk of dying from heart failure versus animals without GS.
In humans, progression of disease associated with concomitant CKD and CHD is referred to as cardiorenal syndrome. It is characterized by progressive changes in both the heart and the kidney due to dysregulation of common hemodynamic, neurohormonal, biochemical, and immunologic feedback pathways. 42,47 One proposed explanation for the relationship between cardiac disease and kidney disease is that the reduction in cardiac output leads to reduced kidney perfusion, stimulation of renal baroreceptors, activation of the renin-angiotensin-aldosterone system (RAAS), and sodium retention with sequela of increased cardiac workload, fluid retention, and vascular expansion (increased BP and heart rate). This pathway results in release of profibrotic neurohormones with subsequent progressive myocardial and renal interstitial remodeling. 37,46 Other pathophysiologic changes considered important in the development and progression of the cardiorenal syndrome include changes in the transforming growth factor β (TGF-β) expression, activation of inflammatory mediators, and development of pressure overload and volume overload. 11,47
Angiotensin II, a product of the RAAS, binds G-protein-coupled receptors in kidney and heart, causing postglomerular vasoconstriction, which results in increased glomerular pressure, increased intravascular volume, increased cardiac workload, and cardiac muscle hypertrophy. Nonhemodynamic effects associated with changes in angiotensin II are important for the progression of diseases of the heart and kidneys via induction of reactive oxygen species, cytokine upregulation, induction of cell adhesion molecules, and increased profibrotic growth factor production. 7 Angiotensin II triggers upregulation of TGF-β expression, increased synthesis of ECM, stimulation of plasminogen activator inhibitor 1 production by endothelial and vascular smooth muscle cells, and macrophage activation/infiltration. The constellation of these changes results in progressive cardiac and renal dysregulation, remodeling, and dysfunction. Angiotensin II activates NADPH and NADH oxidases in vascular smooth muscle, cardiomyocytes, and renal tubular epithelial cells, generating reactive oxygen species such as superoxide, which contributes to tissue injury and promotes atherosclerosis. 21 Angiotensin II upregulates proinflammatory factors, including interleukin 6, monocyte chemotactic factor protein 1, and nuclear factor-kB in the kidney, all of which promote chronic inflammation. 46,49
TGF-β has been identified as a primary mediator of tissue fibrosis in the heart, kidney, and other organs. 19,35 Increased cardiovascular risk and progression of kidney disease have been demonstrated in humans with high serum levels of TGF-β1 and alterations in circulating markers of collagen metabolism, and increased serum TGF-β has been demonstrated in chimpanzees with cardiovascular and kidney disease. 15,19,32,35 Both increased angiotensin II and inflammatory cell infiltrates activate the TGF-β-related receptor, causing direct and indirect activation of profibrotic pathway, downstream profibrotic gene transcription, myofibroblast differentiation, and connective tissue remodeling. 45 The development of fibrosis in the kidney results in localized hypoxia, which contributes to progressive tubulointerstitial injury. 41 The TGF-β pathway is complex, with numerous ligands and signaling molecules involved in fibrotic reactions, and is likely central to progression of both CF and GS in aged chimpanzees.
Hypertension and arteriosclerosis were highly associated with each other and with CF and GS in the APF chimpanzees. Decreased cardiac output triggers the RAAS to release of aldosterone, which increases renal resistance to natriuretic peptides and increases sodium retention. Increased plasma sodium levels result in vascular expansion with volume overload, resulting in increased BP in humans and chimpanzees. 14 Both changes stimulate release of additional factors promoting cardiac muscle hypertrophy, progressive fibrosis, and exaggerated cardiomyocyte energy expenditure, cell death, and heart failure. 9,12,13,31,38 Volume overload also results in a RAAS-independent stimulation of cardiac hypertrophy. 30 Increased BP results in glomerular capillary hypertension with an associated reduction in GFR. 10 Volume overload may cause mesenteric venous congestion, which facilitates translocation of enteric gram-negative bacteria from intestines into portal blood, causing low-grade endotoxemia and subsequent inflammation. 10 Arteriosclerosis results in increased vascular stiffness and peripheral resistance, associated with renal dysfunction and CHD. 36 Additionally, inflammation within atherosclerotic plaques independently stimulates RAAS, promotes continued plaque formation, and induces vasoconstriction, which increases systemic and regional BP. 21 When compensatory mechanisms become inadequate, volume and pressure overload increase, resulting in progressive heart and kidney dysfunction. 22,37,40,46
Chronic inflammation was consistently present with both GS and CF in the aged chimpanzees at the APF. Even at minimal to mild severity grades, chronic inflammation is recognized as a factor promoting CKD and CHD. 1,10,36 Hypoalbuminemia was observed in a large portion of the animals with CF, GS, or both in our study and has been a frequent change in humans with CHD and CKD. 25,29 Decreased albumin synthesis is associated with systemic inflammation (negative acute-phase protein), cachexia, and poor nutrition and increased urinary protein loss in CKD. 20,25 In humans, hypoalbuminemia is associated with a poor prognosis in CHD and CKD. We found that hypoalbuminemia was not a prognostic indicator for the chimpanzees, as there was no significant association with animals that died of CHD and/or CKD as compared with those that died of other causes; however, the association may have been masked, as other causes of moribundity and death were primarily inflammatory conditions also resulting in low serum albumin. The proinflammatory cytokines tumor necrosis factor α (TNF-α) and interleukins 1 and 6 are increased following the loss of compensatory mechanisms associated with chronic renal failure and chronic heart failure and modulate ongoing inflammation associated with the cardiorenal syndrome. 44,50 Other mediators of inflammation that are commonly involved in CHD and CKD include cell adhesion molecules (E-selectin, ICAM-1, VCAM-1) and C-reactive protein (CRP). 10 CRP, which is also considered to be a prognostic marker for heart disease, 3 amplifies the inflammatory response, induces apoptosis, and promotes adverse ventricular remodeling. 52 Increased CRP is also independently associated with renal dysfunction (decreased GFR) and may play a role in the maintenance and progression of CKD. 51 Inflammatory cells such as monocytes/macrophages, dendritic cells, CD4+ T cells, and mast cells infiltrate the kidney in CKD and likely contribute to interstitial fibrosis. 19 Inflammation and its mediators CRP, VCAM-1, TNF-α, and interleukins 1 and 6 are also implicated in the genesis of hypertension by upregulation of RAAS, increasing vascular tone and aortic wall stiffness and reducing vascular wall resistance to oxidative insult through depletion of nitric oxide. 34
In our study, anemia was significantly associated with GS and present in many animals with concurrent CF. Anemia, primarily due to reduced production of erythropoietin by the diseased kidney, also develops frequently in humans with CHD and CKD and is referred to as cardiorenal anemia syndrome. 2,53 Decreases in circulating EPO may promote factors of cardiorenal syndrome, leading to further decline of heart and kidney function, and these factors may in turn interfere with receptor-mediated responsiveness to EPO, further contributing to the development of chronic anemia. 27 Experimentally, EPO has been shown to have a protective function by inhibiting apoptosis of cardiomyocytes and proximal tubular cells of the kidney, attenuating the loss of cardiovascular hemodynamic function, and promoting renal tubular epithelial regeneration and recovery after injury. 8,28,54 GS of aged chimpanzees was commonly associated with inflammatory cell infiltrates and, by inference, proinflammatory cytokines such as interleukin 1, TGF-β, and TNF-α, which are known inhibitors of EPO production. 18 In animal and in vitro models, administration of EPO was associated with decreased microscopic renal damage and decreased levels of IL-6, TNF-α, and CRP systemically. 18,20,54
Azotemia was significantly correlated to CF in the chimpanzees. In most cases, the animals had microscopic changes of GS. In animals with azotemia without GS, it was considered to be due to decreased glomerular perfusion (prerenal azotemia), secondary to reduced cardiac output from heart disease. 37
Several chimpanzees had ≥1 metabolic diseases known to be associated with cardiorenal syndrome in humans, including hypertension, diabetes, and obesity. 22 Diabetic nephropathy has been highly associated with cardiorenal syndrome in humans, and similar nephropathy has been demonstrated in chimpanzees with diabetes. 48 In our study, 2 females with diabetes had both CF and GS.
Conclusion
Concomitant CF and GS was a common finding in aged chimpanzees at the APF. Most of these animals died or were euthanized due to moribundity-associated CHD and CKD or from overt heart and kidney failure. The constellation of altered metabolic parameters, measures of cardiac and renal function, clinical pathologic changes, and histologic lesions of the heart and kidney in aged chimpanzees at the APF were similar to cardiorenal syndrome in humans.
Footnotes
Acknowledgements
The authors extend special thanks to Dr Anthony Comuzzie for his review of statistical methods and to Dr Jerrold Ward and Debbie Gillette for their help organizing color images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
