Abstract
Diabetic human patients have increased risk of heart failure compared to healthy subjects. The underlying mechanisms for this are not fully understood, and to help develop improved treatment strategies, well-characterized animal models are essential. To investigate cardiac dysfunction in diabetes, this study evaluated myocardial changes in 10 aging rhesus monkeys with and without diabetes. Based on evaluation of plasma glycosylated hemoglobin and glucose, 7 of 10 rhesus macaques had diabetes for a minimum of 11 months, while 3 of 10 were categorized as nondiabetic. A detailed histological examination of formalin-fixed left ventricular myocardial samples was followed by a semiquantitative evaluation of myocardial fibrosis and fat infiltration; digital quantifications of myocardial collagen, lipofuscin, and nuclear area fractions; and measurements of cardiomyocyte diameter. Histological myocardial evaluation revealed the presence of lipofuscin; large nuclei; interstitial, replacement, and vascular fibrosis; adipocyte infiltration; and vacuolar degeneration with atrophy of cardiomyocytes and fibrosis. However, there were no differences between groups for semiquantitative fat infiltration, fibrosis, cardiomyocyte size, collagen, or nuclear and lipofuscin area fraction. Lipofuscin area fraction correlated with plasma insulin, triglyceride, total cholesterol, and high-density lipoprotein cholesterol concentrations. In conclusion, myocardial pathological changes were found in left ventricular myocardium in aged rhesus macaques, independent of the stage of diabetes. The duration of diabetes might have been too short to cause differences between groups.
Keywords
The risk of heart failure is 2 to 5 times greater in human diabetic patients than in nondiabetic control subjects independent of age, obesity, dyslipidemia, and coronary artery disease. 24,28 This association has been recognized for several decades, 18 and diabetic cardiomyopathy is now accepted as a separate clinical form of myocardial disease manifested as decreased cardiac performance in the absence of macrovascular changes such as hypertension and atherosclerosis. 15 Diastolic dysfunction of the left ventricle is usually the first sign of diabetic cardiomyopathy and cardiac changes. Later, diastolic dysfunction can progress into heart failure first with preserved and then with reduced ejection fraction, the latter due to additional systolic dysfunction. 15 The myocardial changes are believed to be induced by hyperglycemia and myocardial insulin resistance. 14 The mechanisms of myocardial injury include increased oxidative stress, lipotoxicity, apoptosis, and cell signaling impairments. 15 Furthermore, structural alterations of cardiomyocyte hypertrophy and myocardial fibrosis are reported. The exact underlying mechanisms and sequence of diabetes-induced myocardial changes are not yet known.
Different animal models have been used to study diabetic myocardial changes. However, few animal models develop diabetes spontaneously. Therefore, chemically induced type 1 diabetes mellitus or genetically modified animals with type 2 diabetes mellitus are typically used. 14,20,22 However, spontaneous type 2 diabetes mellitus has been described in nonhuman primates (NHPs) with clinical features similar to humans. As age and body weight increase, so do obesity, dyslipidemia, insulin resistance, and hyperglycemia. In addition, comorbidities occurring in humans such as atherosclerosis, nephropathy, retinopathy. and neuropathy have also been observed in diabetic NHPs. 3,7 For example, left ventricular function evaluated by echocardiography was shown to be impaired in diabetic cynomolgus monkeys. 9 This suggests that NHPs may be a model for human diabetic cardiomyopathy, but knowledge regarding myocardial morphological changes of diabetic NHPs is sparse. 9 Through histological analyses, we aimed to evaluate myocardial lesions in tissue from rhesus monkeys with spontaneously developed diabetes. We hypothesized that aging rhesus monkeys with spontaneous diabetes would show increased myocardial fibrosis, fat infiltration, and cardiomyocyte hypertrophy compared to nondiabetic age-matched NHPs.
Materials and Methods
Animals, Housing and Feeding
Animal experiments were carried out in accordance with the Danish Act on Experiments on Animals, the Appendix A of ETS 123, and the EU Directive. A project license was issued by the national authority. All animal experiments were performed in accordance with relevant guidelines and regulations and were approved by the Novo Nordisk Ethical Review Council. The animals were socially housed under environmental-enriched species-specific conditions in cages constructed to ensure high welfare during experimental studies.
In this study, left ventricular myocardial tissue from 10 rhesus monkeys (Macaca mulatta) was obtained from tissue archives at Novo Nordisk A/S. The monkeys (male, n = 6; female, n = 4) were derived from a colony (BioTest A/S, Konárovice, Czech Republic). Animals were housed in male and female pairs in standard double-sized cages (1.8 × 2.0 × 2.0 m) and had free access to outdoor facilities with environmental enrichments such as shelves, ropes for swinging, and changeable plastic toys. All animals were fed a pelleted standard monkey diet: Feed NOE OW (Racio, Břeclav, Czech Republic). Food was provided ad libitum and served in the morning in portions of 350 g/d/animal. Feeding was supplemented with fruit and vegetables. Housing and procedures followed Novo Nordisk A/S ethical guidelines. Animals were not included in studies for 4 to 5 years prior to euthanasia, except for one 3-week pharmacologic study with an antidiabetic drug 9 months before euthanasia. However, after this time point, none of the animals were given drugs affecting glucose or lipid metabolism.
Diagnosis of Diabetes
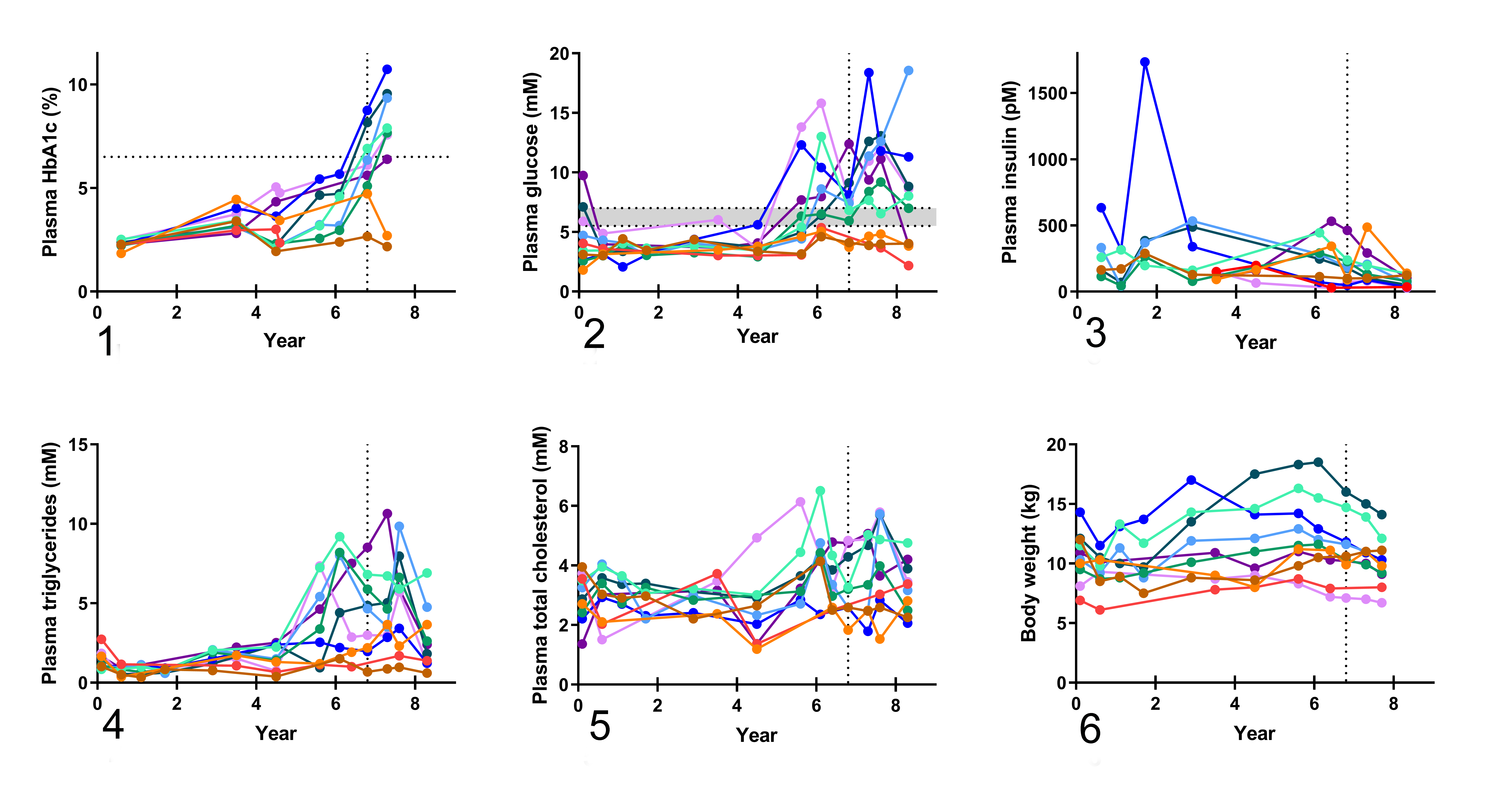
Measurements of plasma glycated hemoglobin (HbA1c), glucose, insulin, triglycerides, total cholesterol, and body weight from 2002 to 2010 are presented in Figs. 1 to 6. Diabetes was diagnosed when HbA1c levels (Fig. 1) were above 6.5%, 33 which in December 2009 allocated 2 monkeys (brown and orange lines) to the nondiabetic group and 6 animals to the diabetic group (i.e., animals were diabetic at least 11 months prior to euthanasia). One animal (Fig. 1, dark purple line) with a prediabetic HbA1c level 33 was allocated to the diabetic group because of a fasting plasma glucose concentration above the human cutoff value of 7 mM (126.1 mg/dl) 1 on 5 consecutive measurements over 2 years (Fig. 2). Finally, 1 female NHP (Fig. 1, red line) was pregnant during 2008 to 2009, and blood samples were therefore not collected. However, since plasma glucose levels (Fig. 2) did not exceed 7 mM and insulin levels (Fig. 3) remained low for the entire sampling period, 13 this female NHP was included in the nondiabetic group. All blood analyses were conducted on a Hitachi 912 autoanalyzer (Boehringer Mannheim, Mannheim, Germany) and a Biosen autoanalyzer (EKF Diagnostics, Cardiff, UK) at Apigenex (formerly RE&D VUFB, Prague, Czech Republic), except for the final measurements, which were measured in house at Novo Nordisk A/S (Måløv, Denmark) as described below. For values below or above the detection range of the assay, concentrations were set as lower or upper detection range values, respectively.
Blood was collected yearly using a vacutainer system (B. Braun Melsungen AG, Melsungen, Germany) from the saphenous vein in awake, fixated animals that had been fasted overnight. For measurements of plasma glucose, triglycerides, total cholesterol, and high-density lipoprotein cholesterol (HDL-C), blood was collected into lithium heparin vials (Vacuette LH; Greiner Bio-One, Kremsmünster, Austria). Blood for measurement of plasma insulin and C-peptide was collected in EDTA-coated vials (Vacuette K3E EDTA; Greiner Bio-One, Kremsmünster, Austria). Finally, 10 μl blood for HbA1c analysis was collected from the needle by capillary micropipettes (Drummond Scientific Company, Broomall, PA) into Eppendorf tubes containing 1 ml hemolyzing solution (Hemolysing Reagent cat. 148-8457; Roche Diagnostics, Hvidovre, Denmark) and then transferred into Hitachi cups. Vials were inverted gently several times to ensure mixing of blood and anticoagulant. Samples were then kept on wet ice until centrifugation. All samples were centrifuged for 10 minutes at 4000 g and 4°C. Upon centrifugation, samples of lithium heparin plasma were transferred to Hitachi cups, and EDTA plasma was transferred into Eppendorf tubes. All samples were stored at –80°C until further analysis.
Plasma glucose, total cholesterol, HDL-C, triglyceride, and HbAc1 were measured by an automatic analyzer Hitachi 912 (Boehringer Mannheim, Mannheim, Germany) using commercial kits (cat. 11447521216, 11489437216, 03030067122, 1188872216, and 11822098216, respectively; Boehringer Mannheim). For determination of plasma insulin and C-peptide concentrations, commercial ELISA kits were used (cat. 10-1113-01 and 10-1136-01, respectively; Mercodia AB, Uppsala, Sweden).
Necropsy and Tissue Processing
Animals were deeply anesthetized with a mixture of medetomidine (0.2 mg/kg) and ketamine (10 mg/kg) for exsanguination by cardiac puncture (where blood was collected for processing) followed by a lethal intravenous injection of 1 g/kg T-61 (Merck Animal Health, Madison, NJ) into the saphenous vein. Blood was collected as 100 ml into EDTA tubes (cat. 366643; Becton Dickinson A/S, Lyngby, Denmark) and 100 ml into 10-ml heparin tubes (cat. 367880; Becton Dickinson A/S) and processed as described above. Plasma samples were analyzed as described for historic samples. Following euthanasia, animals were placed in supine position and organs and tissues were removed. Following collection of tissue for other purposes, full-thickness transmyocardial samples from the left ventricle free wall were collected.
The samples were fixed in 10% neutral buffered formalin. After approximately 24 hours, the tissue was processed in a tissue processor (Leica APS300 S) and paraffin wax embedded. Three consecutive tissue sections of 4-µm thickness were cut on a microtome and mounted on separate Superfrost Plus slides (Thermo Scientific, Karlsruhe, Germany). Sections were stained with hematoxylin and eosin (HE), picrosirius red (cat. ab150681; Abcam, Cambridge, UK), Weigtert’s hematoxylin (PSR), or periodic acid–Schiff (PAS) (cat. 1.09033.0500 and AMPQ00254.500; VWR International, Søborg, Denmark). All tissue sections were digitalized with a NanoZoomer XR (Hamamatsu Photonics, Hamamatsu, Japan) scanner at 40× magnification.
All tissue analyses were carried out on digitalized slides using the NDP.view 2 (Hamamatsu Photonics). Each section was first examined histologically and lesions were described. This was followed by quantifications as described below.
Semiquantitative and Quantitative Analysis of Tissues
Histologically, semiquantitative analysis of interstitial fibrosis, replacement fibrosis, and perivascular fibrosis was performed on each myocardial section. Furthermore, epicardial, interstitial, and perivascular fat infiltrations were evaluated. All parameters were scored on a scale 0/1+/2+/3+ (modified after van Hoeven and Factor 29 ) by an observer blinded to animal identity. An average for vascular parameters was calculated for each tissue section. Only arterial vessels with a diameter above 100 µm were included. Of the included vessels, lumen diameter was additionally measured and section mean lumen/artery ratio was calculated.
Image analysis software from Visiopharm for Windows 7, version 7.0.0.2899 (Visiopharm A/S, Hørsholm, Denmark) was used to estimate area fractions of myocardial collagen on PSR-stained tissue sections, myocyte nuclear size on HE-stained sections, and lipofuscin (as positive-stained granules) on PAS-stained tissue sections. An algorithm was designed to automatically detect myocardial tissue and create regions of interest (ROIs) that excluded epicardium and endocardium. ROIs were manually adjusted where applicable. Other algorithms were designed to detect and calculate myocytes, nuclei, lipofuscin, and areas without any tissue components, and area fractions were calculated.
Cardiomyocyte diameter was measured on HE-stained sections at 400× magnification. On each section, a total of 100 cardiomyocytes were measured and a mean value was calculated for each animal. The smallest diameter was measured on cross-sectionally cut cardiomyocytes holding a visible, central nucleus as described by Baandrup and coworkers. 2 Cells were measured in clusters of 10, distributed on 10 different locations. Intraobserver variation was investigated on 3 sections that were each evaluated 6 times.
Statistical Methods
Statistical analysis was performed using SAS Enterprise Guide 7.1 (SAS Institute, Cary, NC). Summary statistics were tabulated using univariate analysis and are reported as median and range. For statistical analysis of body weight and blood parameters, only the last obtained measurement was used. Male and female animals were pooled into 1 diabetic and 1 nondiabetic group after nonparametric Kruskal-Wallis test showed only a significant difference between sexes for body weight (P = .03) and a tendency for plasma glucose concentration (P = .07). Because of small sample size, the nonparametric Kruskal-Wallis test for continuous variables and Fisher exact test for categorical variables were used to compare groups. Spearman correlation test was used to evaluate relationships between body weight, age, plasma glucose, HbA1c, insulin, C-peptide, triglycerides, total cholesterol, and HDL-C and histological myocardial findings of cardiomyocyte size and area fraction of collagen, myocyte nuclear size, and lipofuscin staining. In addition, a test for correlation between cardiomyocyte size and nuclei area fraction was performed. Differences were considered significant when P < .05. The data analyzed in this study are not available.
Results
Basic Characteristics
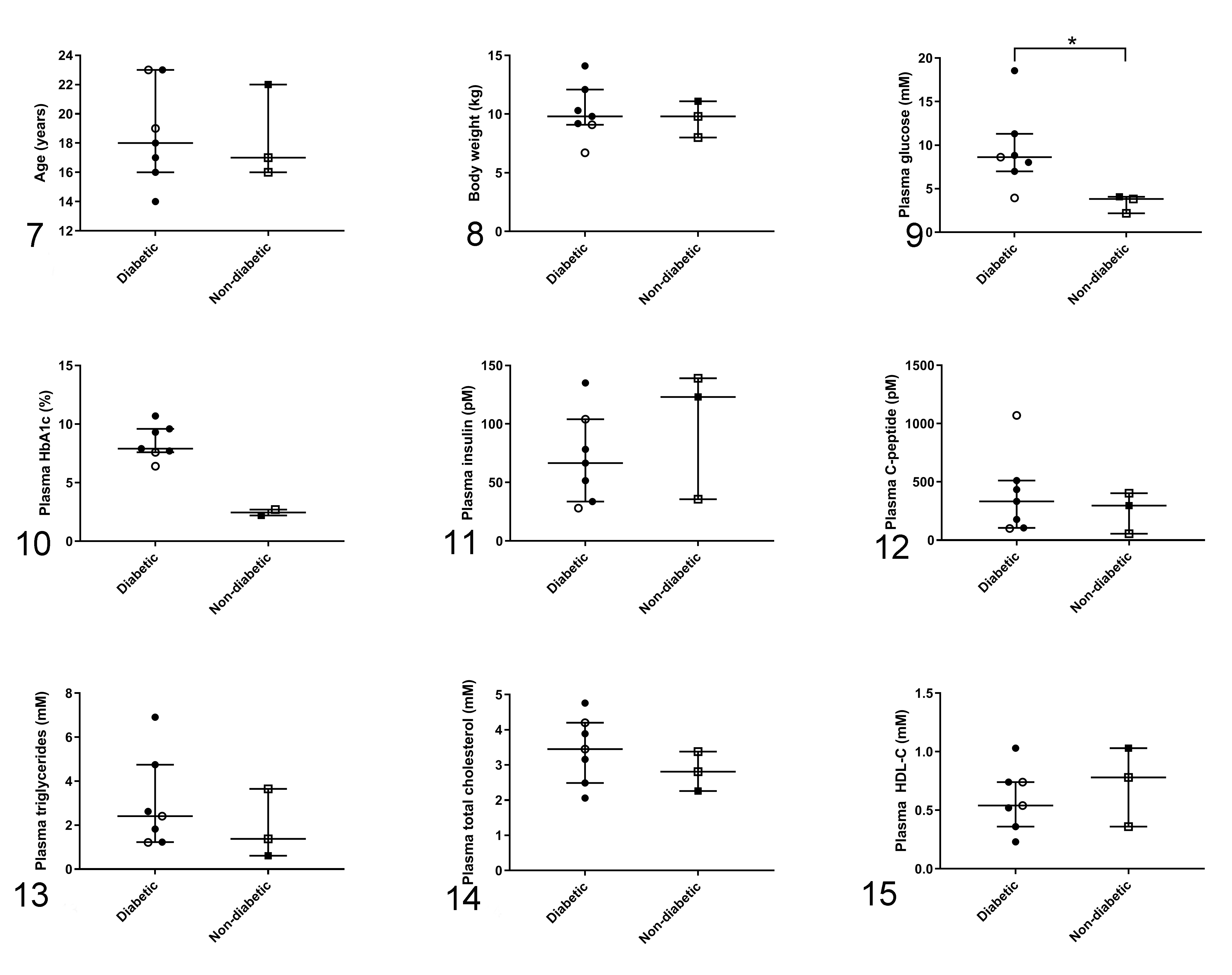
Basic characteristics are presented in Figs. 7 –15. Statistical analysis showed no difference in age and body weight between groups (P = .82, Fig. 7; P = .91, Fig. 8, respectively). At the time of euthanasia, plasma glucose levels were significantly higher in the diabetic group compared to the nondiabetic group (P = .04; Fig. 9). The HbA1c levels were also higher, although this did not reach statistical significance (P = .057; Fig. 10). No differences were found between the 2 groups for plasma insulin (P = .36, Fig. 11), C-peptide (P = .49; Fig. 12), triglycerides (P = .49; Fig. 13), total cholesterol (P = .36; Fig. 14), and HDL-C (P = .49; Fig. 15) concentrations.
Histological Evaluation
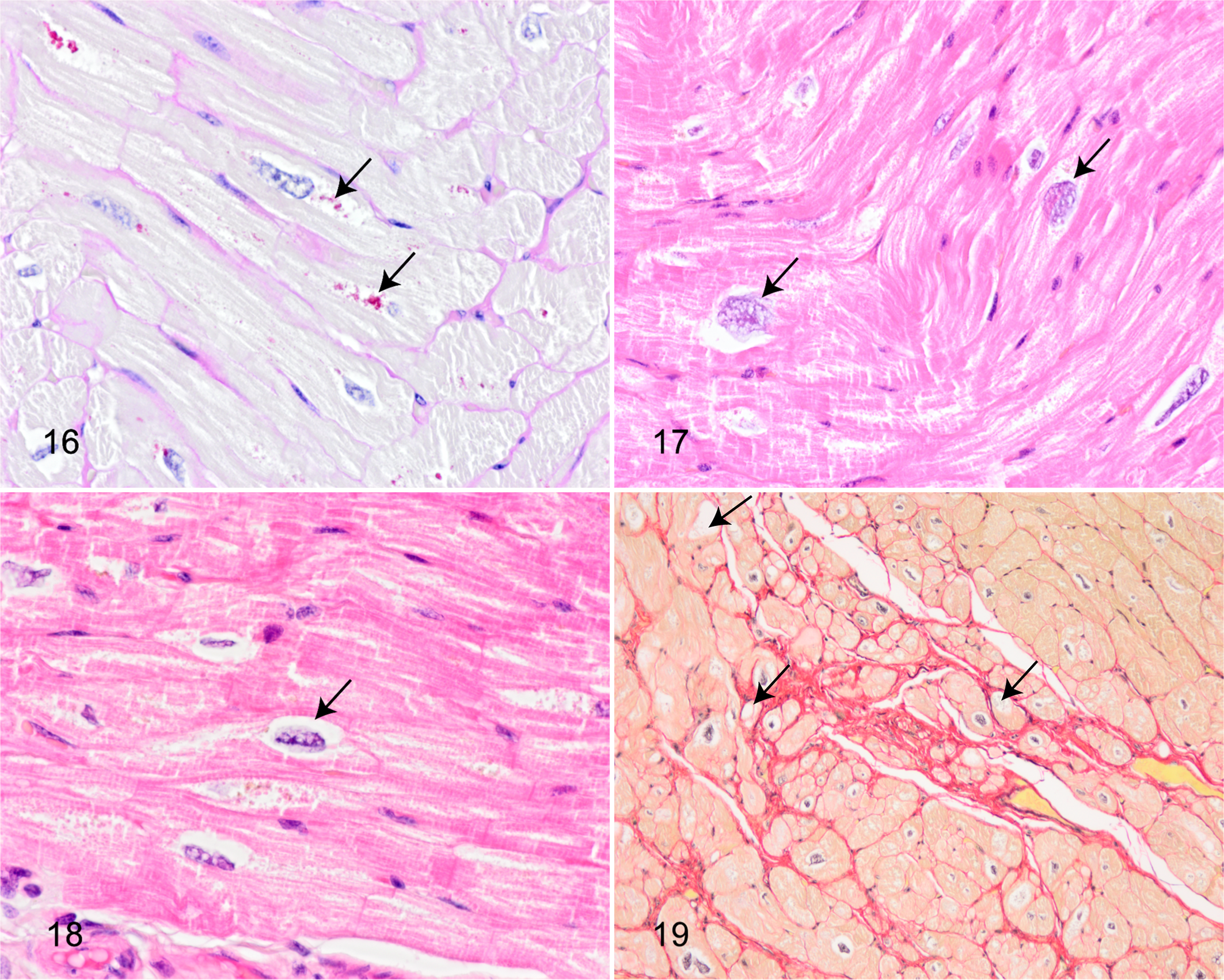
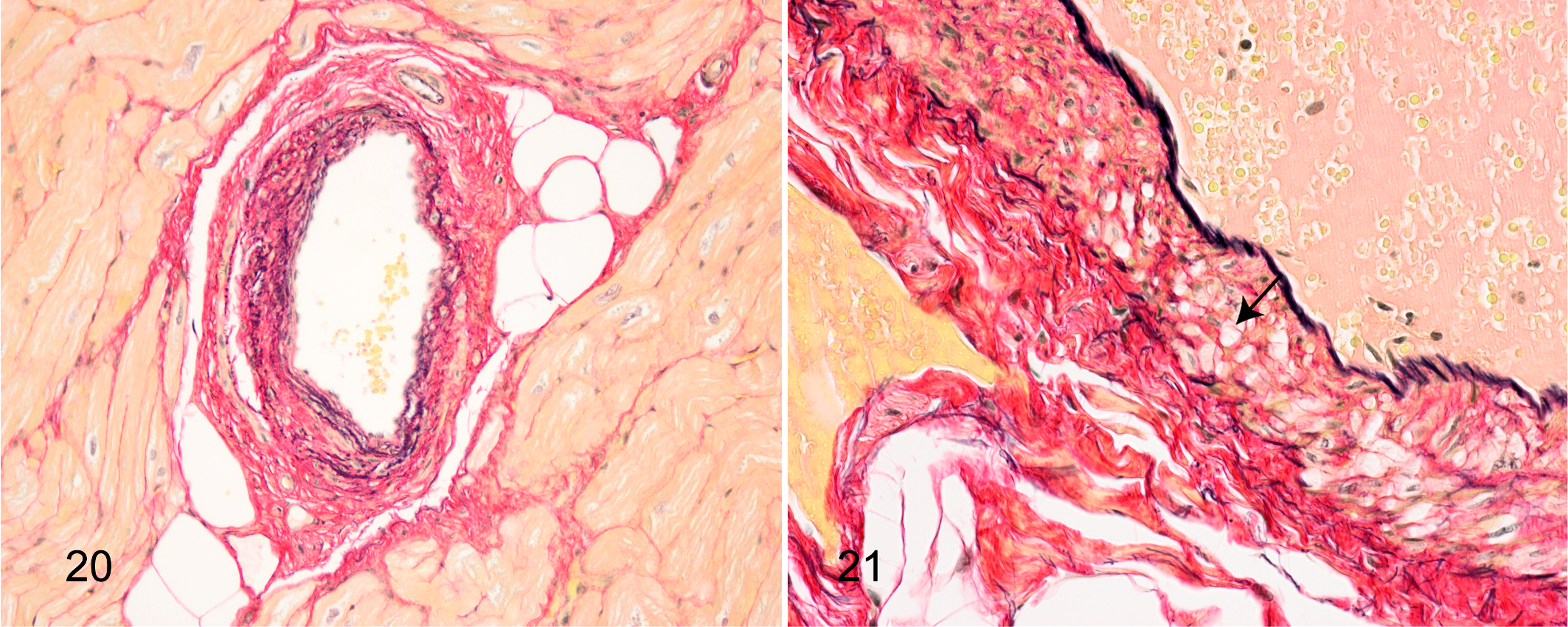
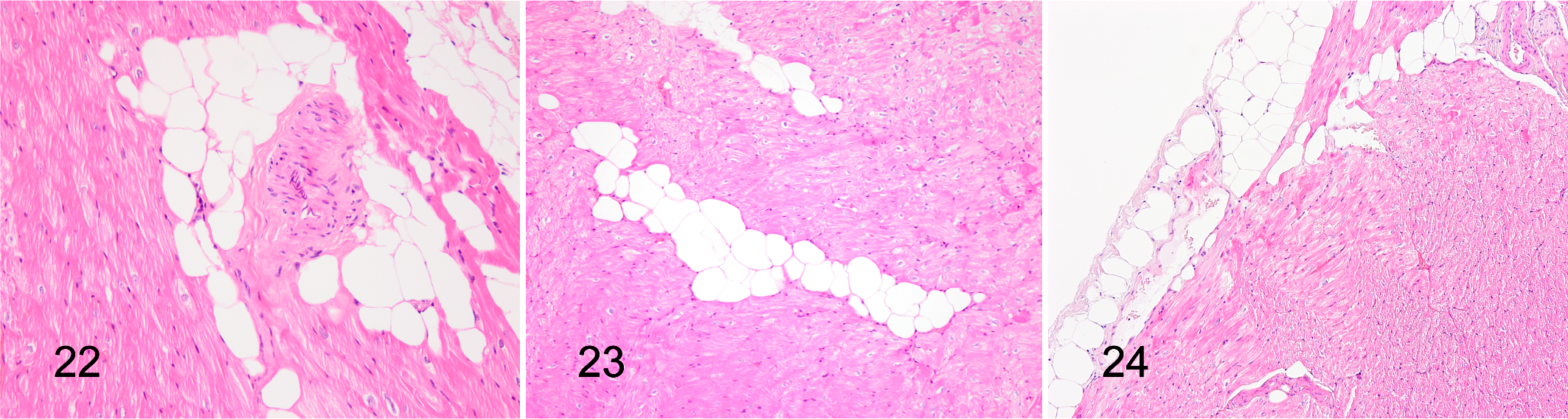
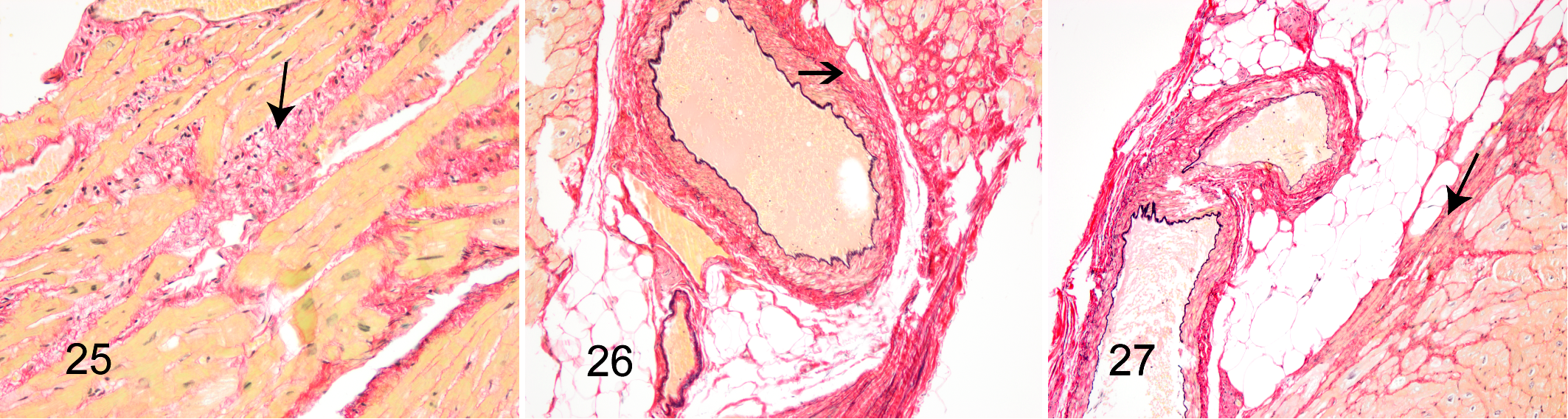
Histological examinations revealed pigmentation at the poles of cardiomyocyte nuclei across all tissue sections; the pigment was PAS positive and interpreted as lipofuscin (Fig. 16). In addition, large variation in cardiomyocyte nuclei size was observed, and a moderate number of nuclei appeared very large. Indeed, nuclei with a diameter >10 µm were observed in all animals (Fig. 17). Two of the diabetic animals had single cells with nuclei resembling Anichkov cells (Fig. 18). Vacuolar degeneration of cardiomyocytes was seen in 5 of 7 diabetic and 2 of 3 nondiabetic animals. The vacuolar degeneration was accompanied by cardiomyocyte atrophy and interstitial and replacement fibrosis (Fig. 19). Intramural arterial changes were observed in 2 of 7 diabetic animals. One animal had 2 small muscular arteries with tunica media predominantly composed of elastic and collagen fibers with a limited area having retained smooth muscle cells (Fig. 20). The other animal had a medium-sized muscular artery with increased collagen content and vacuolar degenerative changes of smooth muscle cells in tunica media (Fig. 21). Findings of perivascular fat deposition ranged from moderate to severe for the 2 animals. Minimal to moderate perivascular fatty infiltration was found in 3 of 3 nondiabetic and 3 of 7 diabetic animals (Fig. 22), while minimal multifocal interstitial fat infiltration was found in only 2 of 7 diabetic animals (Fig. 23). Epicardial fat deposition was minimal to moderate in the diabetic group and absent to minimal in the nondiabetic group (Fig. 24). Multifocal interstitial fibrosis (Fig. 25) was observed in 2 of 7 diabetic and 1 of 3 nondiabetic animals, and perivascular fibrosis was seen in 1 animal from each group (Fig. 26). In addition, in 2 diabetic animals, fibrosis was present in the epicardium encroaching on the cardiomyocyte layer, replacing cardiomyocytes and thus representing replacement fibrosis (Fig. 27). Focal inflammation was present in 2 diabetic animals and compromised approximately 60 and 80 mononuclear cells (Fig. 28).
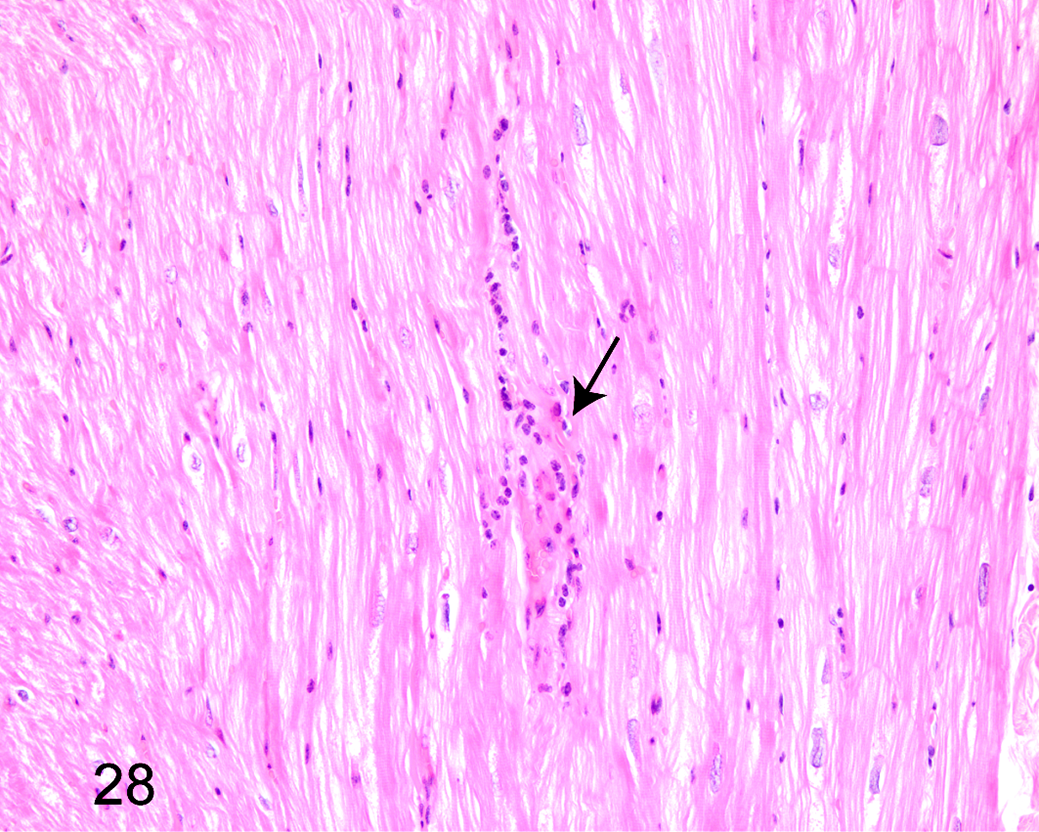
Diabetic animal, myocardium, rhesus macaque. Focal inflammation with mononuclear cells. Hematoxylin and eosin.
Semiquantitative and Quantitative Analyses of Tissues
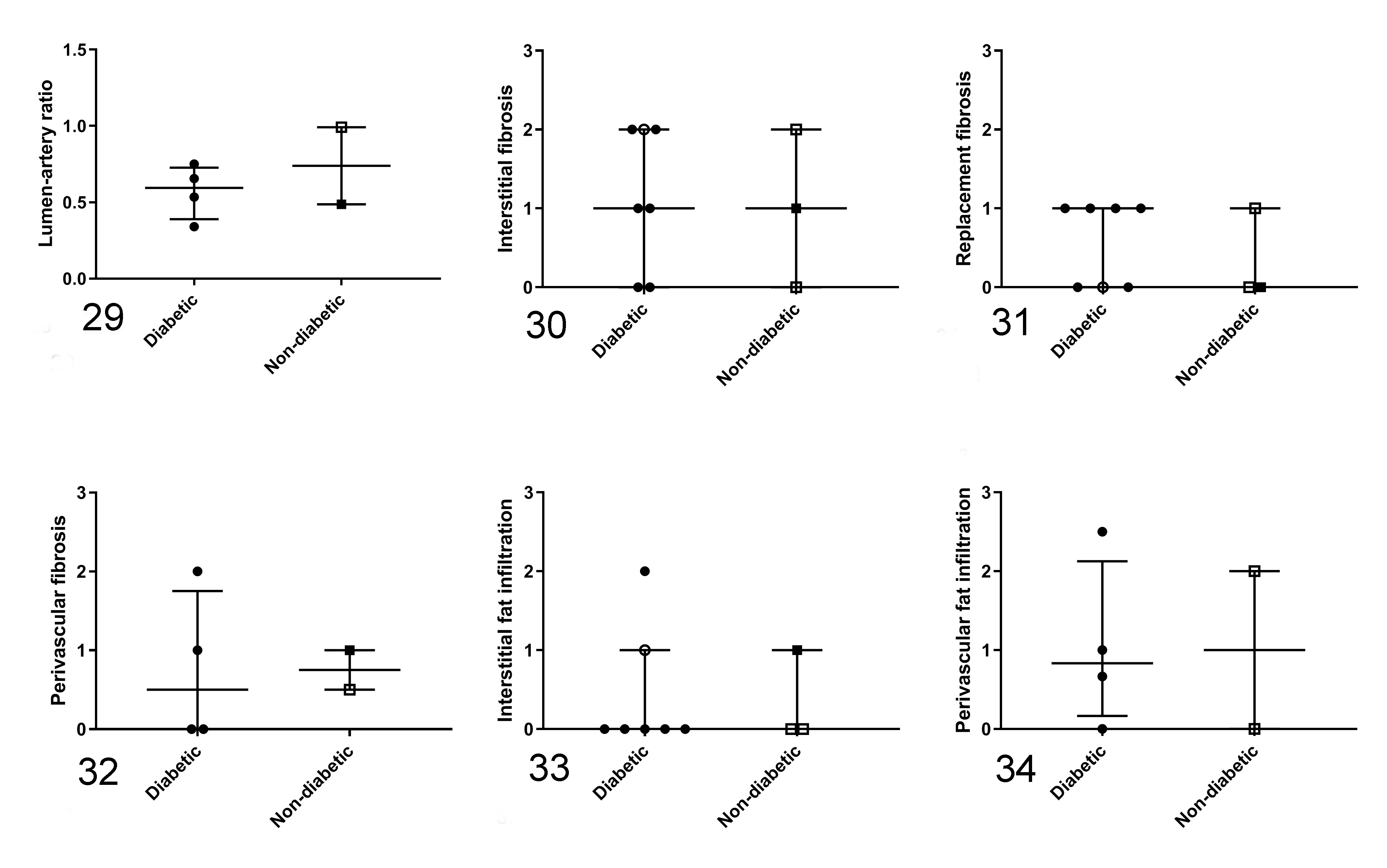
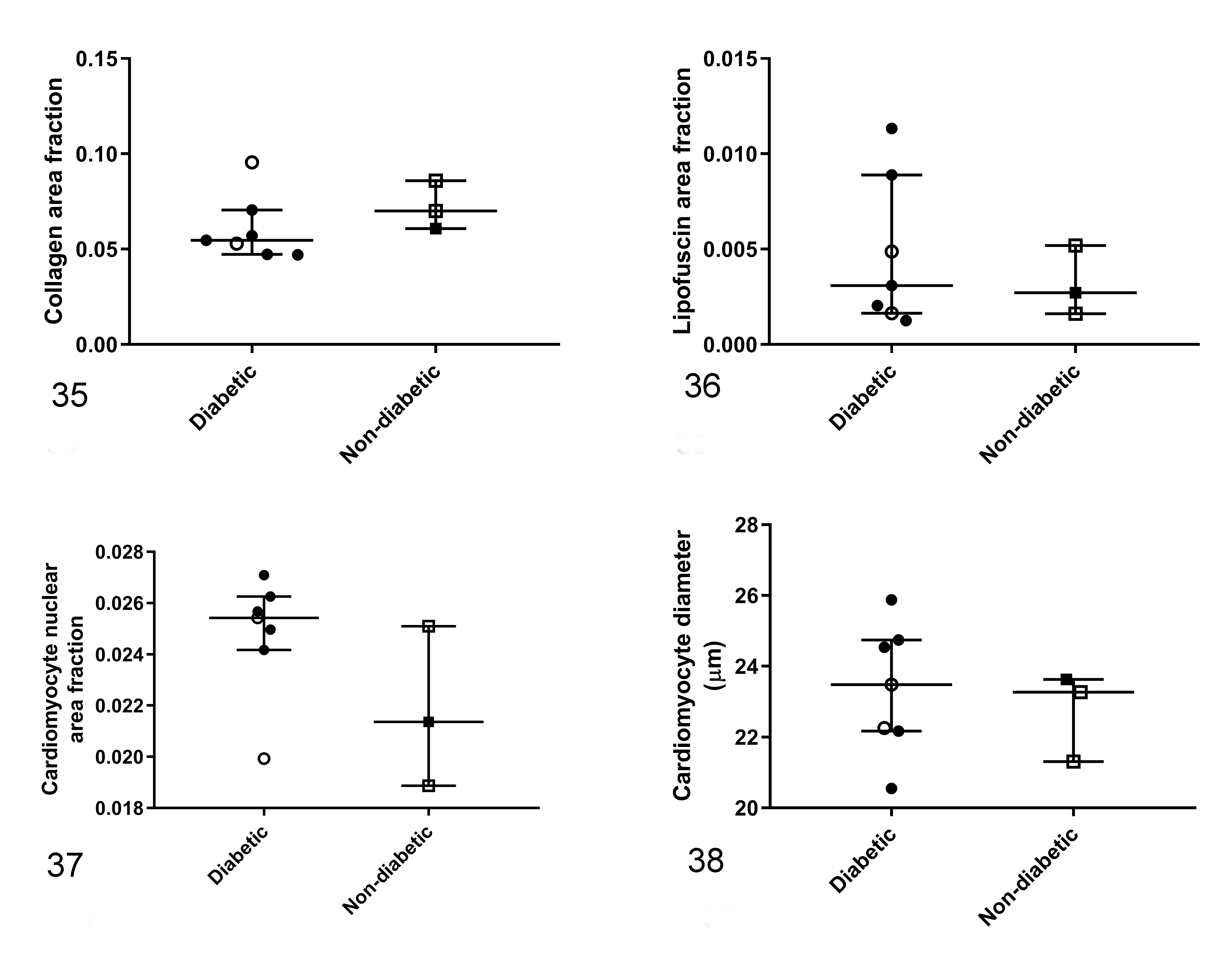
Vascular parameters were measured in 4 of 7 diabetic and 2 of 3 nondiabetic animals. There was no difference between groups for lumen/artery ratio (P = .82; Fig. 29) or interstitial (P = 1.00; Fig. 30), replacement (P = 1.00; Fig. 31), or perivascular (P = .64; Fig. 32) fibrosis. Likewise, interstitial (P = .65; Fig. 33), perivascular (P = 1.00; Fig. 34), and epicardial (P = 1.00) fat infiltrations were not different between groups. No differences were found between the diabetic and nondiabetic groups for myocardial collagen (P = .26; Fig. 35), lipofuscin (P = .82; Fig. 36), and nuclear (P = .14; Fig. 37) area fraction determined by image analysis. The cardiomyocyte size measured by cell diameter was not different between the diabetic and nondiabetic animals (P = .65; Fig. 38). Coefficient of intraobserver variance was 7.67%.
Correlation Analysis
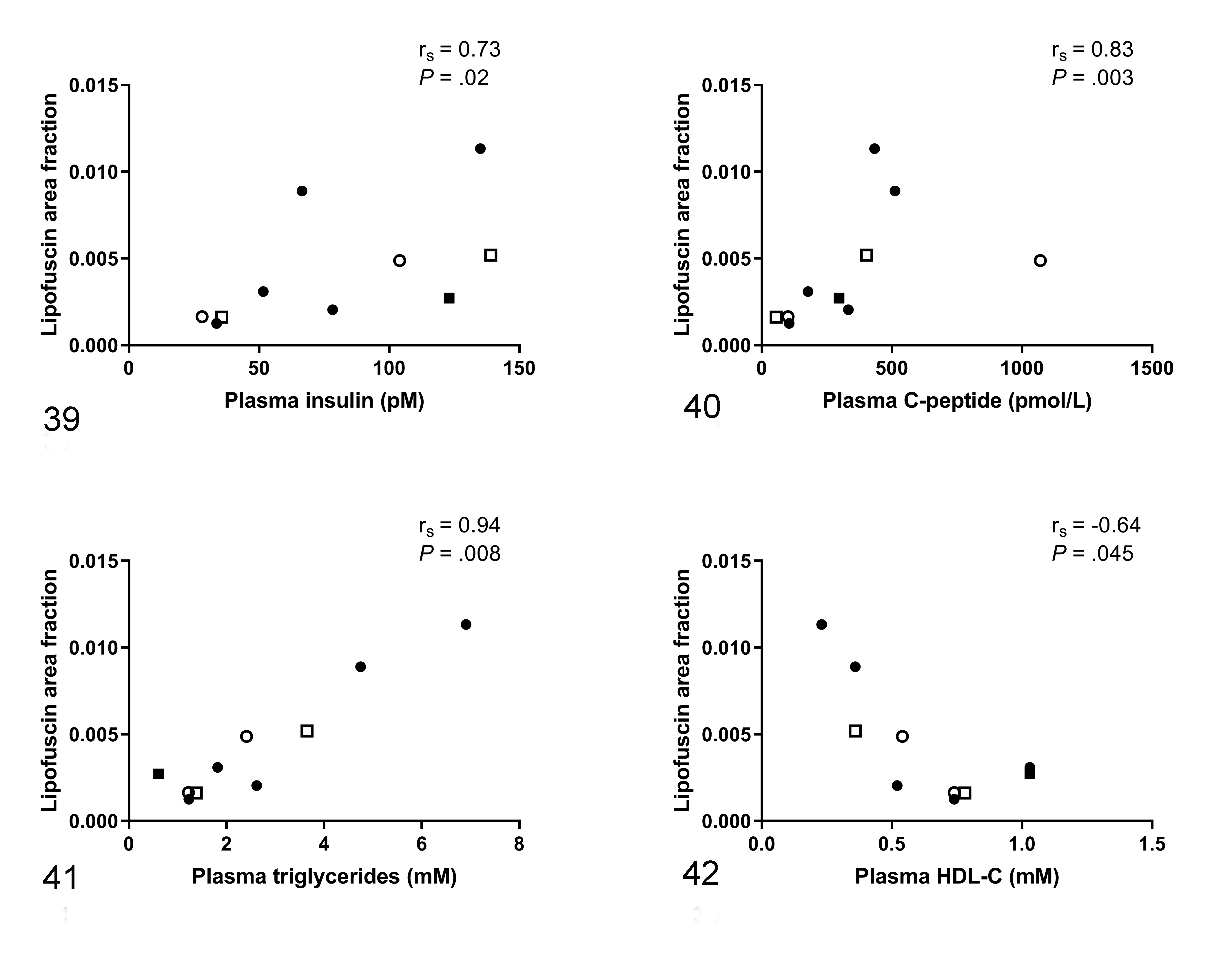
Spearman’s correlation test demonstrated that lipofuscin fraction area was positively correlated to plasma insulin (r s = .73, P = .02; Fig. 39), C-peptide (r s = .83, P = .003; Fig. 40), and triglycerides (r s = .94, P = .008; Fig. 41) and was negatively correlated with HDL-C (r s = .64, P = .045; Fig. 42). Nuclear area fraction was negatively correlated with HDL-C (r s = –.74, P = .01). Furthermore, cardiomyocyte size tended to be positively correlated with plasma glucose concentrations (r s = .59, P = .07) and nuclear area fraction with plasma triglyceride concentrations (r s = .61, P = .06). No correlation was found between nuclear area fraction and cardiomyocyte size (r s = .15, P = .68).
Discussion
This study is to our knowledge the first to specifically evaluate myocardial pathological changes in spontaneously diabetic and nondiabetic rhesus monkeys. Although limited in its conclusions due to a low number of animals, the study provides insight into unique data. In contrast to our hypothesis, fibrosis, fat infiltration, and cardiomyocyte hypertrophy were not different in diabetic and nondiabetic animals. Furthermore, although showing no difference between groups, the amount of lipofuscin in myocardial tissue correlated positively with plasma concentrations of insulin, triglycerides, and total cholesterol and negatively with HDL-C. In the present study, diabetes was diagnosed approximately 1 year prior to euthanasia in 7 of 10 animals based on measurement of plasma HbA1c and glucose concentrations (Figs. 1, 2). The definitions and categorizations are in line with what has previously been described for NHPs, 9,12,13 although normal fasting plasma glucose concentrations in NHPs are around 1.7 mM (30.6 mg/dl) lower than in humans, 31 arguing for an even lower cutoff value than what is defined here (∼5.8 mM [104.5 mg/dl]). In addition, the high levels of plasma triglycerides preceding the time point of group segmentation may reflect increased body fat mass and/or indicate early glucose metabolism dysfunction prior to the onset of diabetes. The subsequent decreasing body weight, probably due to wasting, supports that the diabetic state had progressed. 13 The NHPs of this study spontaneously developed hyperglycemia and overt diabetes over a period of several years from ad libitum food administration. Human type 1 diabetes mellitus involves β-cell destruction that can be autoimmune or idiopathic, usually leading to absolute insulin deficiency. 1 Meanwhile, type 2 diabetes mellitus involves hyperglycemia that may develop over time, after a period of decreased insulin sensitivity, hyperinsulinemia, and dyslipidemia. The NHPs of this study may be a model of the human type 2 diabetic condition, but further studies are needed to clarify the pathogenesis of spontaneous diabetes mellitus in NHPs.
There were variance and overlap in plasma parameters, which could contribute to the lack of statistically significant differences between groups in myocardial parameters. Histological characteristics of diabetic NHP myocardium have previously been described. In a study by Gu and coworkers, 9 localized inflammation was reported near myocardial edema and intramural vasculature after autopsy of 2 spontaneously dead cynomolgus monkeys. In addition, cardiomyocyte fragmentation, atrophy, and brown granulation at the cardiomyocyte nuclear poles were observed, which is in line with the findings of the present study. In addition, we demonstrated that the PAS-positive brown granulation, which likely is lipofuscin, was correlated with plasma insulin and lipid concentrations. This fits the notion that the presence of lipofuscin is linked to oxidative stress, 10 which is expected to be increased with high lipid availability and oxidation, 30 as was likely the case in the NHPs of this study. Hence, high plasma lipid concentrations may have been an indicator of cardiomyocyte metabolic stress.
Moreover, the study by Gu and coworkers 9 compared in vivo cardiac function assessed by echocardiography in healthy, prediabetic, and diabetic animals. Using pulsed-wave Doppler, they demonstrated decreased left ventricular diastolic function by a decreased E-wave and E/A ratio in the diabetic and prediabetic group compared to the controls, which could be indicative of diabetic cardiomyopathy. It could be speculated whether the diabetic rhesus monkeys in the present study also had decreased cardiac function. If so, it could serve as a model of early diabetic cardiomyopathy, in which myocardial structural changes are not yet prominent. Campbell and coworkers 5 proposed that fibrosis and cardiomyocyte hypertrophy are a consequence rather than a cause of diabetic heart disease. Human investigations often rely on cardiac tissue from presurgery heart patients or autopsies, in which diabetes likely has been present for several years, and thus represent late-stage diabetic cardiomyopathy. Decreased myocardial performance is associated with increased age and diabetes duration in type 1 diabetes mellitus patients. However, systolic and diastolic dysfunction was present in only 1.7% and 14.4% of patients without previously known heart disease, HbA1c concentrations of 8.2%, and duration of diabetes for 25.5 years. 17 Thus, the lack of differences in myocardial changes between the diabetic and nondiabetic groups in the present study might be because 1 year of diabetes is insufficient time for the animals to develop such changes. In addition, as cardiovascular complications only manifest in a fraction of the human diabetic population, 27,28 the same could be expected in an NHP model. However, it cannot be excluded that a real difference between the 2 groups exists since post hoc sample size calculations indicate that the present study is underpowered. For example, at least 12 animals in each group are required to detect a 20% difference in myocardial fraction area (power = .80; α = .05).
Fibrosis and fat infiltration, vacuolar degeneration, and large cardiomyocyte nuclei determined from the nuclear area fraction were found in both diabetic and nondiabetic animals with no clear differences between groups. This suggests that the changes might be normal and perhaps related to aging and/or obesity. However, none of the myocardial observations showed a relationship with age, body, or plasma parameters in the correlation analyses. Likewise, we found no association between cardiomyocyte diameter and nuclear size in the present study, suggesting that the large nuclei were not related to cardiomyocyte hypertrophy. The lack of relation could, however, be caused by different approaches for assessment of cardiomyocyte size and nuclear size, respectively. In a study by Koda and coworkers, 19 cardiomyocyte nuclear diameter of normal human hearts and hypertrophied hearts from patients with systolic dysfunction were 4.56 ± 0.56 µm and 7.37 ± 0.90 µm (mean ± SD), respectively. This is smaller than some of the large nuclei observed in the present study. However, these investigators also found a significant correlation between cardiomyocyte hypertrophy and nuclear hypertrophy, indicating that mechanisms other than cardiac hypertrophy might cause the variation in nuclear size in the present study. Nuclear size of cardiomyocytes has been related to enhanced biosynthesis activity, including DNA repair/synthesis, transcription, and translation efficiency. 19 Perhaps this explains the inverse relationship between plasma HDL-C concentrations and cardiomyocyte nuclear size found in the present study. As HDL-C is a potential inhibitor of cell stress and death, 11 increased nuclear size might reflect increased synthesis due to cellular stress or damage.
The greatest advantage of using NHPs as models for human diseases is the high similarity in anatomy and physiology. 25 Other species have also been studied as models of diabetic cardiomyopathy. Both db/db mice and Zucker (diabetic) fatty rats show cardiac abnormalities comparable to what is observed in human diabetic patients, including in vivo assessed decreased left ventricle diastolic function, increased left ventricle mass, and increased lipid and fat oxidation evaluated ex vivo. 4 Increased myocardial collagen content has likewise been found in some diabetic rodent models 16,26,32 but not in others. 6,8 In terms of efficacy and safety, rodents are considered less translational. 25 Recent studies on cardiac structure and function in obese and/or diabetic porcine models, which yet again are more similar to humans in terms of anatomy and physiology, indicate that some porcine breeds are more prone to develop functional and myocardial structural changes than others, 21,23 although differences in study designs might have influenced the findings. Thus, in addition to the species, choice of strain and breed might be important considerations. The study has limitations. The number of animals and sample material (blood and tissue) was limited. Moreover, in vivo measurements of cardiac function were not performed. Measurements of tibia length, waist circumference, or height that could have been used to index heart weight were not measured. Finally, the animals were treated with a drug for a 3-week period 9 months prior to euthanasia. We cannot exclude that this treatment may have influenced our results, but we find it unlikely due to the long washout period.
In conclusion, no differences in myocardial pathological changes where found between aging diabetic and nondiabetic rhesus monkeys. The duration of diabetes might have been too short for differences to develop between groups.
Footnotes
Acknowledgements
We thank Betina Gjedsted Andersen and Elisabeth Wairimu Petersen from the University of Copenhagen; Bettina Brandrup, Camilla Ingvorsen, Jonas Kildegaard, and Susanne Juul Rasmussen from Novo Nordisk A/S for skilled technical assistance on tissue preparation and analyses; and Hanne Hoffmann Frølund Refsgaard from Novo Nordisk A/S for assistance on statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The rhesus monkeys, from which tissue and data were obtained, were bought and owned by Novo Nordisk A/S, although animals were housed at BioTest A/S. T.P.L., R.K.K., K.L., and E.M.W. were employees at Novo Nordisk A/S. H.D.P. was employed at Ellegaard Göttingen Minipigs A/S.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Novo Nordisk A/S and LIFEPHARM (Novo Nordisk—LIFE In Vivo Pharmacology Centre) (PhD grant for S.K.).
