Abstract
The great apes (chimpanzees, bonobos, gorillas, and orangutans) are our closest relatives. Despite the many similarities, there are significant differences in aging among apes, including the human ape. Common to all are dental attrition, periodontitis, tooth loss, osteopenia, and arthritis, although gout is uniquely human and spondyloarthropathy is more prevalent in apes than humans. Humans are more prone to frailty, sarcopenia, osteoporosis, longevity past reproductive senescence, loss of brain volume, and Alzheimer dementia. Cerebral vascular disease occurs in both humans and apes. Cardiovascular disease mortality increases in aging humans and apes, but coronary atherosclerosis is the most significant type in humans. In captive apes, idiopathic myocardial fibrosis and cardiomyopathy predominate, with arteriosclerosis of intramural coronary arteries. Similar cardiac lesions are occasionally seen in wild apes. Vascular changes in heart and kidneys and aortic dissections in gorillas and bonobos suggest that hypertension may be involved in pathogenesis. Chronic kidney disease is common in elderly humans and some aging apes and is linked with cardiovascular disease in orangutans. Neoplasms common to aging humans and apes include uterine leiomyomas in chimpanzees, but other tumors of elderly humans, such as breast, prostate, lung, and colorectal cancers, are uncommon in apes. Among the apes, chimpanzees have been best studied in laboratory settings, and more comparative research is needed into the pathology of geriatric zoo-housed and wild apes. Increasing longevity of humans and apes makes understanding aging processes and diseases imperative for optimizing quality of life in all the ape species.
The term great apes generally describes 3 genera composed of the bonobo or pygmy chimpanzee (Pan paniscus), the common chimpanzees (Pan troglodytes ssp), the western and eastern gorillas (Gorilla gorilla ssp and Gorilla beringei ssp), and the Bornean and Sumatran orangutans (Pongo pygmaeus and Pongo abelii). According to many anthropologists, there is a fourth ape genus with 1 extant species: Homo sapiens. Since these apes are our closest living “relatives,” an understanding of great ape health, disease, and aging may shed light on human conditions. 73 Yet the apes are not just furry humans, and considerable differences in lifestyle and life history are present among the great ape species as well as between apes and humans. For example, when compared with other apes (and humans), orangutans have a slower metabolism, reproduce at a later age, and have a longer interbirth interval and prolonged infant dependency, yet orangutans do not have a greater maximum life span than do the other apes. 196,280,281
Aging in all species is thought to be the result of insults acquired throughout life, leading to necessary adaptations (allostasis) to approximate homeostasis. The cumulative load (allostatic load) of these insults and attempted adaptations eventually leads to progressive decline in organ function associated with aging, and allostatic overload hastens aging. 56,157,198,249 The numbers and kinds of insults demanding allostasis are legion—with disease, inflammation, and attendant oxidative damage (so-called inflammaging) considered by some to be particularly deleterious. 28,75,76,172,241,283 The ability to adapt to these insults also declines with age, limiting capacity for allostasis. Depletion of stem cells in various tissues, mitochondrial dysfunction, and decline in immune system functions (immunosenescence) contribute to weakened allostatic potential. 28,145 Mitigating effects promoting “successful aging” include diet and psychosocial parameters, such as social networks and “happiness.” 111 These factors may also play a role in successful aging of apes, as “happy” orangutans seem to live longer. 277
Because of genetics and life events both positive and negative, aging is an individual process without a set timetable. Chronologic age does not always match with physiologic or biological age, making pinpointing of the age at which an individual human or animal is considered elderly or geriatric a difficult prospect. 172 Indeed, what constitutes ape old age has not been entirely agreed on and may need reevaluation in this age of changing demographics. 76,125
The World Health Organization definition of an elderly person differs depending on geographic circumstances: chronologic age >60 or 65 years (y) for developed countries and 50 y for Africa (Definition of an older or elderly person. http://www.who.int/healthinfo/survey/ageingdefnolder/en/index.html. Published 2010. Accessed January 23, 2015). In developed countries, octogenarians and nonagenarians are much more common than ever before, and the maximum recorded life span is 122 y, which is almost twice that of the oldest recorded chimpanzee.
Thus, humans have evolved with a potential maximal life span greater than that of any other primate. 76 Reasons for this are unclear. Despite individual-based data from longitudinal studies of several primate species, no relationship to phylogeny could be identified in the risk of death at the onset of adulthood (initial adult mortality rate) or in the rate of aging (rate of increase in age-specific mortalities with advancing adult age). In contrast, initial mortality rate and rate of aging varied according to habitat and social factors, among and within species, suggesting that mortality was shaped more by environment than phylogeny. 37
The hypothesis that more rapid telomere shortening in apes could account for shorter life span seems to have been disproven. 250 The oxidative stress theory of aging predicts that longer-lived species should have less oxidative stress or better coping mechanisms. Analyses of fibroblasts from 13 species of primates for markers of oxidative stress and mitochondrial function gave conflicting results, and the authors concluded that further studies were warranted before conclusions could be drawn. 57 Another comparative study looked at plasmalogen levels to test the hypothesis that decreased levels of plasmalogens and alterations in lipid metabolism are associated with cardiovascular disease and aging. 176 Although differences were observed between humans and apes, interpretation was open to question. In contrast, in support of the oxidative theory, clear evidence was found that healthy young adult male chimpanzees (n = 10, aged 11–16 y) had higher levels of markers of oxidative stress, higher levels of markers of cardiovascular disease, decreased antioxidant levels, and decreased cardioprotective markers, when compared with healthy young adult human males (n = 10, aged 22–30 y). 265
So, exactly how old is an “old” ape? Chimpanzees in the wild have a reported life expectancy at birth of between 13 and 25 y, depending on the study. 78,118 Rare individuals, all females, have been reported to live into their early 50s. 37 In captivity, the average life span (from birth) for males is 25 y and for females, 34 y, with estimated maximum life spans of 57 and 75 y, respectively. 118,179 (Chimpanzee Species Survival Plan. http://www.lpzoo.org/conservation-science/projects/chimpanzee-ssp. Accessed March 25, 2015). Various studies have considered chimpanzees to be “old” at 30 or 35 y, and 1 clinical study suggested that 25 y for males and 30 y for females are more realistic. 264 Bonobos are relatively rare in the wild and in zoos, and life expectancy and maximum life span are not well established. Maximum life expectancy in the wild is thought to be about 40 y, while the oldest known-aged captive bonobo was 60 y 57 (Bonobo Species Survival Plan, http://www.bonoboconservation.com/bonobos, accessed March 25, 2015). Gorillas in the wild seldom live into their 40s, with median age at death (after surviving infancy) being 37 y. 37 The oldest known-aged mountain gorillas in the Mountain Gorilla Veterinary Project pathology database were females 42 and 43 y old (yo; L.J.L., Gorilla Doctors, unpublished). Old age is thought to commence at 35 y in captive western lowland gorillas, and the maximum age recorded in captivity is 57 y (a female). 160 Wild male orangutans can live up to 58 y and females, to 53 y, and captive maximum life span is similar (58 y for males and 57 y for females).280,281 Orangutans are considered to be “older adults” at 35 y in some studies and 40 y in others. 9,155,233,280
Physiologic and pathologic states associated with aging in humans and animals include the following: hair loss or graying, generalized frailty, loss of bone (osteopenia and osteoporosis), loss of muscle mass and strength (sarcopenia), arthritis, dental attrition and tooth loss, reproductive senescence, loss of cognitive function and other neurodegenerative conditions (including sensory impairment: hearing, smell, taste, and vision [eg, cataracts, macular degeneration]), cardiovascular disease (including hypertension), endocrinopathies (eg, type II diabetes/metabolic syndrome), renal disease, decline of immune functions, and increased occurrence of many types of neoplasms. 13,125,283 Any one individual will not demonstrate all facets of the aging phenotype but will rather be a mosaic of various aspects of molecular, cellular, and organ changes unique to that aged individual. 271
Evidence for similar changes in aging nonhuman great apes is reviewed in this article, drawing on published literature, personal experience, and pathology databases for the apes. Great apes in North American zoos accredited by the Association of Zoos and Aquariums are managed according to animal care guidelines within Species Survival Plans (SSPs), which also facilitate movement of animals among zoos for preservation of genetic diversity (https://www.aza.org/species-survival-plan-program/ and https://www.aza.org/uploadedFiles/Animal_Care_and_Management/Animal_Programs/Animal_Programs_Database/Animal_Care_Manuals/ChimpanzeeCareManual2010R.pdf). Husbandry and care of apes in zoos have evolved over time in response to improved science-based understanding of species needs. A small subset of bonobos, gorillas, and orangutans in the SSP pathology archives lived all, or part of, their lives in primate research facilities accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International and were managed in cooperation with the SSPs. Thus, an individual ape may have experienced diverse diets and environments during its life, which might influence aging. SSPs have clinical veterinary and pathology advisors charged with developing health standards and pathology databases to help inform management decisions. We are all SSP pathology advisors (L.J.L.: gorillas, orangutans, bonobos; R.M.: orangutans; K.A.T.: chimpanzees). All SSP gorillas are western lowland (G. g. gorilla); SSP orangutans include Bornean and Sumatran species and hybrids of the 2; the ancestry of SSP chimpanzees is uncertain, and the population is managed as a single species (Pan troglodytes); and there are no subspecies of bonobo. For wild apes, a pathology database for mountain and eastern lowland gorillas (G. b. beringei and G. b. graueri) is maintained by the Mountain Gorilla Veterinary Project (Gorilla Doctors; L.J.L., pathologist), and several long-term field studies on chimpanzees have necropsy programs (eg, Jane Goodall Institute’s Gombe Stream Research Centre and the Gombe Ecohealth Project; K.A.T., pathologist). Information on the life histories, diet, behavior, and other aspects of wild eastern gorillas and chimpanzees can be found in the primatology literature. 80,95,203 All ape cases that we examined and reported in this article were natural deaths or were euthanized for humane reasons.
External Aspects of Aging in Apes
Hair Loss and Graying
Can one tell from outward appearances if an ape is old? Balding or thinning hair and gray hair are commonly associated with aging in humans. In wild chimpanzees, characteristics used to identify older females include dull, thin, brownish, or graying hair coat with thinning most marked on head, shoulders, and back; tooth loss; sagging, wrinkled faces; and slower, more deliberate movement. 24,118,179 Hair loss in nonhuman primates may be due to parasitism, atopy, self-plucking (trichotillomania), or excessive allogrooming and by itself should not be assumed to be age related. 34,187 Gorilla adult males develop silvering of the hair in a saddle pattern on the back and upper limbs (extending more distally in western lowland gorillas) at about 13 y, but this does not necessarily increase with age.
Frailty and Sarcopenia
Frailty is a phenotype of aging humans and is defined as having ≥3 of the following 5 signs: slowness of gait, loss of hand grip strength, self-reported exhaustion, low energy levels, and unintended weight loss. 51 Frailty is often accompanied by sarcopenia, which is defined as loss of muscle mass, strength, and quality, principally as a result of old age (but also occurring in disease-associated cachexia). 7,47 Sarcopenia is defined histologically by decline in the number and size of myofibers, especially type II fast-twitch fibers, and by fatty infiltration of muscles (steatosis). It is thought to be due to a complex interplay of nutrition, decreased exercise, decreased anabolic hormones, increased oxidative damage, abnormal mitochondrial function, and altered autophagy and myostatin-dependent signaling. 217 It is not known if frailty and sarcopenia occur in elderly apes. Studies of wild and captive chimpanzees have noted decreased or slow locomotion in older animals, which was attributed to arthritis. 24 The few necropsied wild chimpanzees at Gombe with clinical histories of frailty and histologic evidence of sarcopenia have had underlying infections, commonly chimpanzee simian immunodeficiency virus (SIVcpz) with AIDS-like immunopathology. 253 Trackers for the wild mountain gorillas have noted that geriatric animals often move more slowly and are separated from their groups. Generalized wasting is also seen; usually, this is associated with specific lesions at necropsy, such as infections, traumatic wounds, and fractures (L.J.L., Gorilla Doctors, unpublished data). Of the elderly zoo apes that we examined postmortem, most had impressive retention of muscle mass and tone despite chronic debilitating conditions. Therefore, establishing whether old age–related frailty exists in apes, in the absence of non-age-related comorbidities, requires further research.
Osteopenia and Osteoporosis
Bone loss is a significant cause of morbidity leading to fractures in elderly humans, especially women. Osteoporosis is estimated to affect about 15% of 50 to 59 yo women and 70% of 80 yo women. 238 The most widely used nonhuman primate models for osteoporosis are ovariectomized Old World monkeys, mainly macaques (Macaca sp), baboons (Papio sp), and African green monkeys (Chlorocebus sp), and loss of bone density is reported to occur naturally in semicaptive rhesus macaques (M. mulatta). 46,235 Skeletal remains can be used to evaluate bone density and skeletal lesions in apes and are a valuable and underutilized resource. 96 Such studies have identified loss of bone density in wild chimpanzees from 2 study populations. 106,175,246 In the first study, femoral necks from Gombe chimpanzees were analyzed by single-photon absorptiometric scanning, and 5 levels of the diaphysis were analyzed by that method as well as by computerized axial tomography scan. 246 The female chimpanzees >40 yo had a 40% smaller bone mineral index than did females <25 yo, with the greatest changes being in the middiaphysis rather than the femoral neck, as is common in elderly women. Morphologically, this corresponded to bone loss at the endosteal surface of the cortex, leading to a larger medullary cavity. Another study examined the long bones from a set of skeletal remains from 15 Gombe chimpanzees, with plain radiography, single-photon absorptiometry, and computed tomography, and found the greatest loss of bone mineral density in the midshaft of humeri and femurs. 175 Mass of the long bones also decreased. A third report looked at femoral neck and vertebral bone density by dual-energy x-ray absorptiometry in skeletal remains from 4 captive female chimpanzees and 4 wild ones from the Mahale Mountains. 106 The bone density in 1 “old” female was significantly lower than the mean of the other 7 and met the criterion for human osteoporosis. Age-related bone density changes in other apes need to be investigated.
Arthritis
Types of Nonseptic Arthritis
Arthritis increases with age in all species examined to date. It is estimated that over half of human adults >65 yo have joint pain and/or overt arthritis. 149,286 Sorting out the comparative aspects of arthritis can be difficult because of confusing and changeable nomenclature and probable differences among species. 3,54,211 In humans and domestic species, refining the diagnoses of arthritis relies on clinical symptoms and laboratory tests, along with imaging studies (radiography and magnetic resonance imaging [MRI]), joint effusion cytology, and, occasionally, synovial biopsy.
The most common type of old age, “wear and tear” arthritis is often called degenerative joint disease (DJD), a term used interchangeably with osteoarthritis and osteoarthrosis (OA). 228 The term osteoarthritis implies that inflammation is present, while osteoarthrosis is a broader term. Which term is used often depends on the user’s bias or understanding of pathogenesis. OA is not an inevitable result of human aging but likely occurs due to accumulated insults and predisposing factors, such as obesity, female sex, genetic/racial background, macrotrauma (eg, sports injuries), and underlying problems with joint mechanics (eg, angular deformity, laxity, or dysplasia). 228,286 These lead to mechanical microtrauma and resulting damage to cartilage, joint capsule, supporting tendons and ligaments, and subchondral microvasculature. Although inflammation can occur as a result of degradation of joint components by mechanical forces, this is not primarily an inflammatory arthritis (IA), although some recent literature contests this. 284 Articular cartilage has little potential for spontaneous repair even in younger individuals, and, as in all organ systems, repair mechanisms and cellular and matrical plasticity decrease with age. 220 The term chondrosenescence has been coined to highlight the changes. 173
In contrast, IA can begin at any age, although disability is marked in the elderly. Two main types of IA in humans include (1) autoimmune erosive rheumatoid arthritis (RA), in which patients’ serum is positive for rheumatoid factor, and (2) “seronegative,” “nonerosive,” or “reactive” arthritis, which is secondary to inflammatory processes elsewhere in the body (eg, inflammatory bowel disease, bacterial enteritis, urinary tract infections, psoriasis). 30,226 Some authors consider seronegative arthritis in humans to be an erosive disease, which confuses the nomenclature further. 211 The presence of predisposing disease processes is a major criterion for a clinical diagnosis of IA. Since seronegative arthritis often involves the spine, the term spondyloarthropathy (SA) has been used by some authors to refer collectively to these inflammatory syndromes. 210,211 Humans with SA commonly but not invariably carry the major histocompatibility complex class I allele HLA B27. 226
Diffuse idiopathic skeletal hyperostosis (DISH) is a symptomatic or asymptomatic disease of the spine and entheses of peripheral joints, especially in elderly men. 182 There is confusion in the literature about when to apply this term and how to differentiate it from other forms of spinal arthritis with bony proliferation. Criteria have been used to differentiate DISH from OA and SA—such as anterior vertebral body fusion/bridging in areas of intact disc space, absence of sacroiliac disease, and calcification peripheral to the joints. A review of degenerative skeletal diseases of primates stated that DISH is likely underdiagnosed in nonhuman primates and that nonhuman primate spinal lesions have been commonly, possibly erroneously, lumped under degenerative disc disease. 3
Another type of arthritis that occurs in older humans is crystal deposition disease, which includes deposition of uric acid crystals (ie, articular gout) or calcium pyrophosphate deposition (CPPD; pseudogout). 151 Among primates, gout appears to be a uniquely human malady. CPPD, however, has been reported in a variety of nonhuman primates. 204 Both conditions are increased in older and elderly humans, with a mean age of 69 y for gout and 73 y for CPPD. 151 Men are more affected than women (82% of gout cases, 63% of CPPD). CPPD can present as a primary acute mono- or pauciarticular IA, but calcium pyrophosphate crystals are also common in joints affected by OA, IA, infections, and joint instability. 1,209 Mechanical compression of cartilage causes shedding of crystalline CPP into the joint; the crystals then cause an inflammatory reaction that must be differentiated from immune-mediated IA. 108
There is overlap in the distribution of joint involvement in the types of arthritis. DJD/OA in humans involves ≥1 joints and is usually asymmetrical. The most affected joints are those of hands, knees, hips, and shoulders. DJD/OA can also involve the articular facets of the spine (ie, apophysial or zygapophyseal joints) and the vertebral bodies, especially in the neck and lumbar spine in humans. 21 It is often associated with disc space collapse due to the age-related desiccation of the nucleus pulposus, termed degenerative disk disease (DDD). Spinal OA secondary to DDD can lead to osteophytic “lipping” or complete bridging between vertebral bodies (“spondylosis”) and osteophytosis of apophyseal joints. 21 DISH begins as ossification of the ventral ligaments and tendons of the lumbar and cervical spine (enthesophytosis) and can lead to bridging and fusion of vertebral bodies. 3 DISH is not predisposed to by DDD. Spondyloarthropathy/IA also causes bridging and fusion of vertebral bodies due to formation of syndesmophytes. Sacroiliac joint fusion is commonly present as well, with or without involvement of ≥1 joints of the appendicular skeleton (generally <5; ie, pauciarticular). 210 Ankylosing spondylitis is a subtype of SA in which the combination of damage to intervertebral joints and fusion of vertebral bodies is the main phenotype. 215 In RA, there is symmetrical distribution, involving >5 joints, often beginning in distal “small joints” (eg, wrists, ankles, hands, and feet). CPPD can involve any joint but is usually in the appendicular skeleton, and it can occur concurrently in joints affected by OA and SA. 209
Morphologically, DJD/OA is differentiated from SA, particularly in synovial joints, by sclerosis rather than erosion of the subchondral plate. 211 Cartilage changes in OA include grooving (score lines), pitting, and focal or diffuse full-thickness loss of articular cartilage with eburnation of underlying exposed bone. In SA, erosions are primarily marginal, and there is often adjacent new bone formation in response to the erosions. In OA, there is often remodeling of articular surface contours with osteophyte production at the junction between articular cartilage and bone. The latter is in contrast to the mineralization and bone formation in the joint capsule insertions and attached ligaments and tendons (ie, entheseal bone formation, enthesophytes) that occur in SA. DISH also causes ligamentous enthesophyte formation but begins in the ventral spinous ligament, whereas the bone formation in SA usually involves only the margins of the annulus fibrosis (ie, syndesmophytosis). 182 Radiographically, “flowing” calcification causing fusion of vertebral bodies and calcification of ligaments of the paraspinous processes is more suggestive of DISH. 3 Confusingly, bone proliferation in SA can also cause vertebral body bridging (due to syndesmophytosis) and fusion of the articular facets (due to apophyseal enthesophytosis). In CPPD, crystalline material can be seen grossly coating or embedded in cartilage or soft tissues of synovial (diarthrodial) joints.
Arthritis in Wild Apes
Most information in the literature on arthritis in ape species comes from studies of museum collections of defleshed skeletons from wild apes killed in the early part of the 20th century and exhumed skeletal remains of apes from long-term field studies, such as those initiated by Jane Goodall (chimpanzees) and Dian Fossey (mountain gorillas). Classification of the type of arthritis from these specimens is problematic, since helpful soft tissue changes, clinical laboratory data, and postmortem diagnosis of concurrent diseases that support or refute the diagnosis of IA are typically lacking. Lesions in such specimens often represent “end stage” arthritis and may have characteristics of different processes. Additionally, in most cases, the exact age at death is unknown, and few of the skeletons are likely to have come from truly geriatric individuals. Comparison among studies is further complicated by the use of varying methodologies and terminologies (see above).
By examining museum collection skeletal remains of wild adult bonobos and 2 subspecies of common chimpanzee (P. troglodytes troglodytes, the “central” chimpanzee; P. t. schweinfurthii, the “eastern” chimpanzee), the authors of 1 study concluded that SA, rather than OA, was the predominant type of arthritis in chimpanzees. 213 OA was found in 1 of 34 (3%) bonobos and 6 of 79 (7.5%) central and 1 of 26 (3.8%) eastern chimpanzees, while SA was seen in 21% of bonobos and 28% of both chimpanzee subspecies. In animals characterized as having SA, sacroiliac involvement was present in 100% of bonobos, 71% of eastern chimps, and 18% of central chimps. In contrast, another study of wild African ape skeletons—including eastern chimpanzees (from Gombe, Tanzania), central chimpanzees, bonobos, and lowland gorillas (from museum collections)—found only rare spinal lesions (confusingly designated “vertebral osteophyte production” if involving the vertebral bodies and OA if involving the articular facets). 130 OA of peripheral joints was also rare. OA of the shoulder joints was seen in only 2 elderly bonobos, and unilateral knee OA was present in 1 bonobo. Two more recent studies investigated remains of age-known chimpanzees in Kibale National Park in Uganda and Gombe National Park in Tanzania using the same methodology.44,136 In both these studies, degenerative joint lesions were more common in the appendicular skeleton than spine, although in the Gombe chimps the lumbosacral spine was affected in some animals. At Kibale, lesions were noted in 75% of individuals. Except for 2 old females, cases of severe arthritis were associated with evidence of past trauma. 44 At Gombe, OA was positively correlated with age such that 84% of chimpanzees >15 y of age had moderate or severe OA. Arthropathy was most common in the elbow, hip, and knee joints. 136
Arthritis is found in wild gorillas, and evidence of SA was more prevalent than OA in adult skeletons from both western lowland gorillas and eastern gorillas (mountain and Grauer’s subspecies were lumped together). 212,216 OA was found in 1 of 38 (3%) eastern gorillas and 8 of 99 (8%) western gorillas, while SA was seen in 8 of 38 (21%) eastern gorillas and 20 of 99 (20%) western lowland gorillas. SA was male predominant. 212 Another study agreed that joint disease was most often spinal in mountain gorillas, mostly in lumbar spine. 153
In a study examining 55 adult orangutan skeletons from museum collections (identified as the Bornean orangutan, Pongo pygmaeus pygmaeus), 89% of specimens were from wild animals. 214 No evidence of OA was found, but 11% had hallmarks of inflammatory or erosive arthritis (9% males and 15% females). The authors concluded that the pattern in orangutans was similar to that of other great apes (“which clearly have spondyloarthropathy”), except for involvement of statistically fewer metacarpal phalangeal joints, the complete absence of new bone growth at margins of erosions, and a trend toward less sacroiliac joint involvement.
Arthritis in Captive Apes
OA can be classified as primary (when no underlying condition is identified) or secondary (with previous trauma the most common predisposition in humans). 90 Wild apes often show evidence of concurrent trauma, complicating subclassification. In contrast, trauma is less common in captive apes, but other risk factors may contribute to their arthritis (exhibit design, obesity, etc). In our experience, common sites include the knees, hips, elbows, and lower spine.
The literature contains a case report of clinically diagnosed bilateral knee DJD/OA in a 25 yo male laboratory-housed chimpanzee and a report in which 3 geriatric chimpanzees (2 males 40 and 45 yo and a female 40 yo) were treated with acupuncture for OA of the knees (stifle joints). 158,266 No published reports on arthritis in captive bonobos were found, and the only case in the Bonobo SSP pathology database with a diagnosis of arthritis was a 54 yo female with severe OA of the right hip and mild degenerative changes in the right knee; she died of cardiomyopathy. Since the next-oldest animals in the database are only in their 30s, it may be that OA is a late life event in this species.
The prevalence of arthritis in the SSP population of gorillas (deceased and still living) has not been determined. Initial reports of “rheumatoid arthritis” in several infant to young adult gorillas were associated with intra-articular mycoplasma infection and lymphoplasmacytic synovitis. 40 However, the described asymmetrical pattern of joint involvement is more suggestive of either septic or reactive arthritis rather than true RA. 212 It is unknown if these gorillas suffered clinically from arthritis as they aged. Several additional reports of appendicular and spinal arthritis, dating back to the 18th century, suggest that both DJD and OA are present in old gorillas. 35 A 33 yo male gorilla was euthanized because of severe bilateral OA of the hips. 16 A study of skeletons from 3 male (36, 36, 37 yo) and 2 female (23–25, 27 yo) zoo-housed lowland gorillas found slight changes of “lipping” in only 8% of forelimb joints but severe changes in 25% of the lower limb joints, with the males more affected. 184 “Lipping” of vertebral “centra” (bodies) was present in 70% of vertebrae in males and 68% in females. Although changes were present in cervical, thoracic, and lumbar vertebrae, all affected animals (males and females) had lesions in lumbar vertebrae, and there was fusion of lumbar vertebrae (spondylosis) in all the males.
Two case reports in older adult female gorillas also highlight spinal changes. Traumatic herniation of a lumbar disc necessitating diskectomy was reported in a 36 yo female zoo-housed eastern lowland gorilla (erroneously called a mountain gorilla). 15 Similarly, a 42 yo female gorilla from another zoo developed spinal stenosis requiring laminectomy. 282 Radiographs in both these papers show abundant syndesmophytes and early vertebral body bridging and, in the case of the first animal, unilateral partial fusion of the sacroiliac joint, all suggestive of SA.
Additional reports suggesting SA in gorillas include an adult female gorilla who developed spinal lesions resembling ankylosing spondylitis and peripheral arthritis when she was 26 yo and underwent bilateral hip replacement. 2,192 OA was present in spine and other joints when she died at 47 yo (Gorilla SSP data). Her son developed acute IA of the wrist. 2 Another adult female (27 yo) developed acute IA postpartum. 123 Three cases of acute peripheral arthritis resembling reactive arthritis have been diagnosed in juvenile gorillas following shigellosis.183,200 Whether these animals develop more severe degenerative changes as they age remains to be seen. These cases prompted examination of gorillas for a major histocompatibility complex class I marker similar to HLA B27, which predisposes humans to ankylosing spondylitis and other spondyloarthopathies. 2,123,256 Leukocytes from several gorillas examined clinically via human diagnostic reagents were reactive for HLA B27. 2 More sophisticated studies of gorilla leukocyte antigens determined that a class one allele identical to HLA B27 does not exist in gorillas. 258 However, molecular reconstruction of a gorilla class one protein, GoGo-B01, showed binding domains similar to HLA B27, and functional studies showed the ability of GoGo-B01 to bind proteins bound by HLA B27. The role of GoGo-B01 in IA in gorillas needs further investigation, as both affected and unaffected gorillas have this allele.
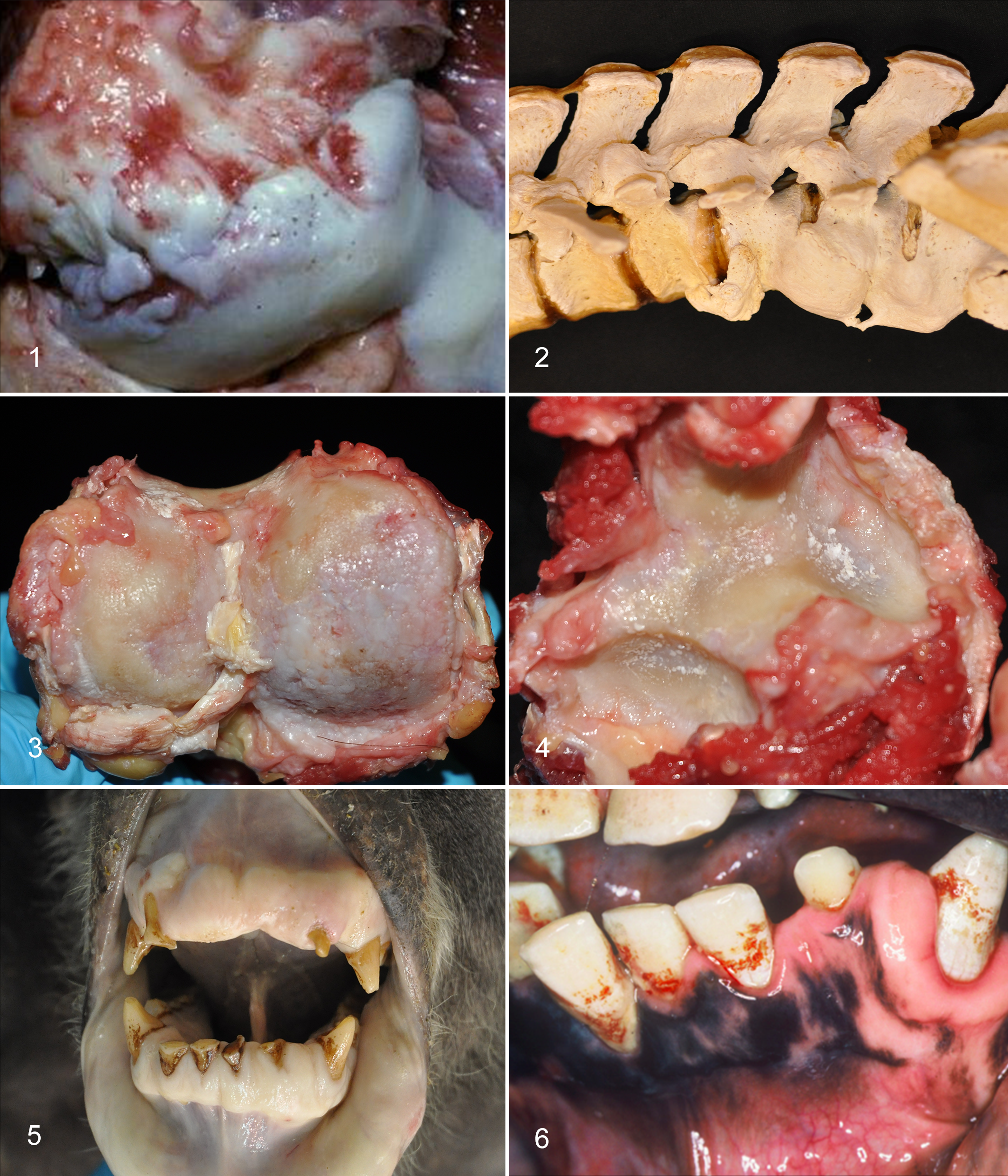
We could find no published accounts highlighting arthritis in zoo orangutans, but images of a radiograph and a 3-dimensional reconstruction from the spine of an orangutan (age not given) were used to illustrate DISH in a review chapter on skeletal diseases of primates. 3 In contrast to the literature, in our experience, arthritis is common in older adult orangutans. In our cases, the pattern has been confusing, sometimes more suggestive of OA than IA, with eburnation as well as erosion of cartilage and with osteophytes as well as enthesophytes. Spinal involvement has had features of SA and DISH as well as DDD and OA. Severe arthritis of the knees was seen in Sumatran orangutan males 34 and 41 yo (Fig. 1) but not in a 28 yo Sumatran orangutan male who had spinal arthritis with osteophytes, syndesmophytes, and partial fusion in the lumbar spine (Fig. 2) or a 39 yo male Sumatran orangutan who had only OA of the right elbow. A 56 yo Sumatran orangutan female (maximum SSP captive life span record) had arthritis in nearly every joint: the left shoulder, both sternoclavicular joints, both elbows, the right carpus, both knees, and both tarsal joints with a pattern most suggestive of OA (Fig. 3). She also had DDD, dorsal disc protrusions throughout the spine, and some ventral syndesmophytosis, especially in the lumbar area. Interestingly, crystals—confirmed to be calcium pyrophosphate by polarized microscopy as well as hydroxyapatite by chemical analysis (courtesy of Dr Bruce Rothschild, University of Kansas School of Medicine)—were seen in the arthritic left shoulder and the elbows (Fig. 4). This is the only case of CPPD in any of the apes of which we are aware.
In summary, while arthritis seems to be common in captive apes, a more thorough retrospective analysis of pathology reports, clinical histories, and radiographic findings from the SSP populations of known-aged apes is clearly needed to better categorize the type of arthritis and determine prevalence and epidemiology. Prospectively, more detailed examination of all joints at necropsy or during physical examinations of wild and captive apes would also facilitate understanding patterns of arthritis in aging apes.
Diseases of the Oral Cavity: Dental Attrition, Tooth Loss, Periodontitis
In humans, age-related dental lesions include dental attrition (occlusal wear), tooth loss, and increased frequency of periodontitis. Dental attrition and tooth loss are common in humans, with up to 50% of elderly individuals >65 yo being partially or completely edentulous. 201 A landmark Centers for Disease Control and Prevention study involving full periodontal assessment of 3742 adult humans reported that 70.1% of adults >65 yo had periodontitis. 66 Moderate or severe periodontitis was noted in 64% of those adults; rates were higher in men (56.4%) than in women (38.4%). Periodontal disease is the predominant factor influencing tooth loss, with tooth trauma as a minor factor. There is also a clear association between periodontitis and diabetes in humans. Detailed mechanisms are incompletely understood but involve aspects of innate and adaptive immune function and blood sugar levels, with periodontal inflammation negatively affecting glycemic control. 199 A cause-and-effect relationship has not been proven, but some studies also report associations between periodontal disease and cardiovascular disease. 5 Underlying common mechanisms of inflammation and bacterial involvement are proposed as contributing factors. 259 Periodontal pathogens such as Aggregatibacter actinomycetemcomitans, Tannerella forsythia, Porphyromonas gingivalis, and Prevotella intermedia have been found in carotid artery atheromatous plaques. 5
Periodontal disease and tooth loss are also common among elderly apes in the wild and in captivity. Gross lesions have been documented through evaluation of exhumed or museum specimens. One study examined 15 complete and 6 partial skulls from exhumed remains of adult mountain gorillas (ages unknown). 153 Most (19 of 21) had moderate enamel wear, with severe wear in 3 of these, likely associated with the abrasive nature of many plants eaten by these apes. All adults had resorption of alveolar bone, primarily in maxillary posterior teeth, presumably due to periodontal disease. Evidence of periodontal abscesses was seen in 11 of 21 gorillas and periapical abscesses in 4. Five adults had antemortem tooth loss, and 1 male had lost 8 teeth. In a study of remains of 10 adult wild chimpanzees from Gombe (known or estimated ages 13–43 yo), enamel wear was common in all but was most severe in the 3 oldest animals (39–43 yo), all of which had antemortem tooth loss. 134 Evidence of a single apical abscess was found in a 19 yo female, while the 4 oldest chimpanzees (35–43 yo) had 3 to 9 abscesses each. Older wild chimpanzees have also been observed, while alive, to have lost or broken teeth. 24 In a comparison of adult African ape skulls—based on collections of mountain, Grauer’s, and western lowland gorillas, bonobos, and eastern and central chimpanzees—differences were found in dental attrition, “dental disease” (which included caries, abscesses, excessive calculus, and root resorption), and tooth loss. 67 Mountain gorillas, bonobos, and lowland gorillas had much more dental disease (45%, 33%, and 27%) than did Grauer’s gorillas or eastern chimps (8% and 6%).
A study of 123 skulls from Bornean orangutans collected in the late 19th century identified age as the greatest risk factor of dental pathology. 244 In animals judged to be old based on criteria of tooth wear, 26% had evidence of periodontal abscesses, 23% had periodontal bone loss, but only 3% had antemortem tooth loss. Female orangutan skulls had significantly more evidence of abscesses and bone loss than did males.
Periodontitis, tooth loss, and bone resorption were frequently seen in geriatric captive zoo apes that we examined (Figs. 5, 6), but caries are rare in our experience. 166,167 In a small sample of skeletons from known-aged captive western lowland gorillas (3 males 36, 36, and 37 yo; 2 females 23–25 and 27 yo), all animals had some evidence of occlusal enamel attrition, which was more extensive in the older males. 184 No mention was made of other dental lesions or tooth loss in those 5 animals.
Factors exacerbating dental attrition and periodontitis include age-related wear, poor diets (high sugar) during early life in some animals, and acid erosion (noted in some gorillas with the behavioral habit called “regurgitation and reingestion”). 166 Genetics and oral microbiota may also play a role, as in humans, but these factors require additional study. In captive living animals, several individual case reports have been published involving periodontal and surgical procedures and proposed standardized dental charts for apes.121,166 One study involved periodontal evaluation of orangutans up to 20 y of age, and it found microbiota similar to that of humans. 243 Another study documented localized periodontitis in an adult male gorilla (diagnosed at 30 y), with culture and serologic evidence of A. actinomycetemcomitans and depressed neutrophil chemotaxis in comparison with that of humans. 167
To date, no large systematic studies of the oral pathology of elderly captive apes have been published. Understanding potential systemic contributing factors, such as diabetes mellitus (see metabolic syndrome) and cardiovascular disease (see later in this article), requires additional comparative study. As humans and apes live longer, deeper understanding of factors that will optimize nutritional status and systemic health and maximize tooth preservation becomes increasingly important.
Reproductive Senescence and Reproductive Tract Lesions
Female Reproductive Senescence and Menopause
A long period of life after reproductive senescence and menopause seems to be fairly unique to humans among the primates. 6,70,72,112,115,272 In wild chimpanzees, reproductive senescence occurs only near the end of life. 70 While only a small percentage of wild female chimpanzees live past 40 y, more than half of those gave birth to at least 1 offspring. The oldest known-aged individual giving birth was 55 yo. Some studies of captive chimpanzees provide data that support late-in-life menopause, with menstrual cycles in females as old as 50 yo.112,115,138 In contrast, other studies of captive chimpanzees found hormonal profiles and interbirth interval patterns suggesting that reproduction may terminate well before death, possibly due to greater longevity than that of their wild counterparts. 97 Intervals between menstrual cycles were reported to increase with age between 19 and 39 yo, in a pattern suggestive of perimenopause. 92 Changes in reproductive hormones indicative of perimenopause (at 30–35 yo) and menopause (35–40+ yo) have also been recorded. 261,262 Hormone profiles in one >40 yo bonobo suggested that menopause had occurred in that individual, and histology of her ovaries revealed depletion of primary oocytes and developing follicles, as well as increased connective tissue. 97
Data from zoo-housed gorillas in North America suggest that menopause does occur and that it is preceded by a period of irregular cycling and low fertility similar to human perimenopause. 17 , 160 Reproductive senescence has been less well studied in the other ape species. In wild mountain gorillas, one 42 yo female was pregnant with a near-term fetus when she was found dead with lesions suggestive of right-sided heart failure (L.J.L., Gorilla Doctors, unpublished). Similarly, female orangutans seem to be able to reproduce to the end of their life spans. 233 Note that gynecologic or pathologic evaluation of the reproductive tracts of the senescent female apes is absent from these studies. Pathologic changes resulting in infertility might give a false impression if senescence is defined only by the absence of births. Given the importance of human menopause in determining other age-related health conditions, further investigation is needed into fertility and hormonal changes in aging apes, especially those in zoo and the wild. 13,104
Female Reproductive Tract Lesions
Morphologic correlates of female reproductive senescence include diminution of ovarian primary oocyte numbers and accumulation of proliferative lesions such as endometrial polyps, endometrial hyperplasia, adenomyosis, uterine neoplasms (eg, fibroids [leiomyomas], and endometrial cancers). 18,91,98 Depletion of primary oocytes has been clearly demonstrated in histology of old chimpanzees and older female mountain gorillas, although a 31 yo female still had abundant primary oocytes (L.J.L., Gorilla Doctors, unpublished). 115
Adenomyosis has been reported in 2 chimpanzees 32 and 41 yo and in a 48 yo orangutan. 26,100 Adenomyosis was diagnosed in a 31 yo mountain gorilla with a history of repeated abortions (L.J.L., Gorilla Doctors, unpublished). Endometriosis was diagnosed in a nulliparous 28 yo gorilla with abdominal effusion who died under anesthesia. 65 Endometrial polyp formation with infarction and adenomyosis were associated with persistent vaginal bleeding in a 24 yo orangutan examined by one of the authors (L.J.L.).
Reproductive tract tumors, especially leiomyomas (analogous to “fibroids” of women), are reported to occur in 28% of female laboratory-housed chimpanzees.39,109,267 Increasing age (>30 y) was associated with increased prevalence. Ovarian neoplasms in chimpanzees include “fibrothecomas” in 2 animals, 39 and 48 yo. 97 A comprehensive look at the disease of the reproductive tracts of aging chimpanzees is covered in this issue of Veterinary Pathology. 48
Leiomyomas have been infrequently reported in other apes, with a single report in a captive lowland gorilla and none in the mountain gorilla pathology database. 181 The Contraceptive Reproductive Health Surveillance program of the Association of Zoos and Aquariums’ Wildlife Contraception Center has found leiomyomas in 1 of 9 gorillas, 3 of 7 Sumatran orangutans, and 0 of 6 Bornean orangutans in addition to 4 of 10 chimpanzees whose reproductive tracts were examined (Dr Anneke Moresco, personal communication)
Malignancies of the female reproductive tract are infrequent in chimpanzees; a recent review found only 3. 40 However, there are reports in the literature of cervical, uterine, and ovarian adenocarcinomas in captive lowland gorillas. 122,191,242 Affected individuals were 35, 36, 39, and 50 yo. A 46 yo western lowland gorilla had a uterine leiomyosarcoma causing urine stasis, pyelonephritis, and fatal sepsis. 42
Male Reproductive Senescence and Reproductive Tract Lesions
Male reproductive senescence (“andropause”) and lesions of the aging human male reproductive tract have been less well studied. New evidence from studies of “primitive” societies suggests that postreproductive longevity happens in men as well as women. 218,268 Hypogonadism and lower levels of androgens can contribute to relative loss of muscle mass (sarcopenia), osteopenia, and other aspects of a geriatric phenotype. 218 Human testicular atrophy is associated with decreased number and volume of Leydig (interstitial) cells. 27 Testicular atrophy due to age alone may be difficult to assess because of the presence of other disease conditions and ill health, which could influence testosterone levels and decrease spermatogenesis.
Among cases that we examined, testicular atrophy and hypospermatogenesis were noted in a 56 yo male zoo-housed chimpanzee with chronic heart failure and two 33 yo Sumatran orangutans, one with acute cholecystitis and heart failure and the other with chronic laryngeal air sacculitis and pneumonia. Adult male gorillas normally have relatively small testes with apparent hypospermatogenesis, but they also have increased interstitial cells, as opposed to the decrease noted in adult men. 71,81,110 This situation may be normal and cyclic, and although it is commonly seen in older male mountain gorillas, it is difficult to attribute to aging alone.
Testicular tumors in humans, especially seminomas, are not necessarily associated with advancing age, although other types of tumor may be. A mixed Sertoli-Leydig tumor was reported in a 38 yo chimpanzee, and interstitial cell adenomas were reported in a 33 yo gorilla. 99,129
Benign prostatic hyperplasia, a bane of human males as they age, is reported to develop in “late middle age” (25–29 y) in chimpanzees. 239 Benign prostatic hyperplasia has not yet been reported in the other apes, perhaps because accessory sex glands are not always examined postmortem.
Obesity, Metabolic Syndrome, and Type II Diabetes
In humans, body weight tends to increase with age and then decrease in the very elderly. 163 Nonetheless, about 42% of adults 65 to 74 yo and 28% of adults >75 yo in the United States are considered obese. 74 Obesity is often coupled with metabolic syndrome, which is defined by abdominal obesity, elevated fasting blood glucose, elevated triglycerides, low levels of high-density lipoproteins, and elevated blood pressure. 163 Metabolic syndrome prevalence increases with age, especially in women.
Obesity is also of concern in captive apes in zoos, sanctuaries, and laboratory settings. 102,148,188 Although it would seem obvious to tell subjectively when an ape is obese, objective criteria of obesity have been defined only in chimpanzees. Two groups have tackled this task. One found that skin fold thickness, not absolute body weight or body mass index, served as a good biomarker in females but not males. 263 The other group found that waist circumference was a biomarker in both males and females. 11 Female chimpanzees were more likely than males to be overweight or obese (body score 4 or 5 of 5). In 16 chimpanzee females >35 yo, almost 44% met the criteria for metabolic syndrome. 188 Obesity has been correlated with hypertension in female chimpanzees. 69 Two accounts of “morbidly obese” adult orangutans in their 30s have associated obesity with hypothyroidism, hypertensive heart disease, and stroke. 245,276
Metabolic syndrome in humans is often associated with insulin resistance and type II diabetes, initially with elevated levels of insulin but later with insulin deficiency. In chimpanzees, waist circumference was correlated with a higher calculated estimate of insulin resistance in females but not males. 11 Published reports of type II diabetes in apes are rare. One report in chimpanzees describes 2 females that were not obese when diagnosed in their 20s. 207 Another study found a <1% overall incidence of type II diabetes in a colony of 260 chimpanzees and 3.7% in those >30 yo. 169 Three older adult chimpanzees (30–33 yo at time of diagnosis) with type II diabetes were all females.
Type II diabetes has been diagnosed in orangutans in the SSP population. 278 Fasting insulin and glucose and glucose tolerance testing of 30 orangutans identified 2 diabetic animals, a male and a female (both 30 yo), and 2 prediabetic animals (a 40 yo female and an 18 yo male). 102 All were overweight, although weight alone or age did not correlate with fasting insulin and glucose levels. The authors concluded that captive orangutans may be prone to glucose intolerance and diabetes. This is interesting in light of the fact that orangutans have the slowest metabolism of any primate studied to date. They are largely frugivorous and are adapted to cycles of low protein and caloric content in their diets in the wild.196,270 Captive management may not reflect this feeding ecology.
While type II diabetes in older macaques, mangabeys, and baboons is associated with amyloidosis of islets of Langerhans, thus far this does not seem to be the case in chimpanzees120,128 There is mention of 1 orangutan with islet amyloidosis, but information about metabolic status was not given. 59 Additional studies of diabetic apes will be needed to define histopathologic correlates of diabetes in these species.
Liver Disease
As humans age, they become more susceptible to liver diseases, and the liver becomes less able to respond to injury. 119 Cellular and morphologic correlates of liver senescence in humans include a generalized decline in hepatocyte size, accumulation of lipofuscin, and “large cell change,” which is characterized by karyomegaly (polyploidy). 140 Polyploidy reaches 27% in elderly >85 yo, as compared with 6% in people in their 20s. Clinical chemistries of laboratory-housed chimpanzees revealed decreasing albumin with age, suggestive of declining liver function. 264 There have been no systematic reviews of liver changes with age in any of the apes.
Hepatocellular polyploidy has been seen in about 15% of wild mountain gorillas and a higher proportion of eastern lowland gorilla adults. 131,178 In some but not all individuals, other evidence of liver disease was present, such as necrosis or bridging fibrosis. Since polyploidy can also signal hepatocyte reaction to a variety of injuries, including plant alkaloids, it is uncertain whether the nuclear changes in these animals are part of the “normal aging process” or a reflection of toxicity. 131 Mild anisokaryosis with polyploidy and lipofuscinosis was seen in a 48 yo chimpanzee (Fig. 7), while marked anisokaryosis with polyploidy was identified in 1 geriatric (46 yo) zoo orangutan with chronic passive congestion examined by one of us (L.J.L.).
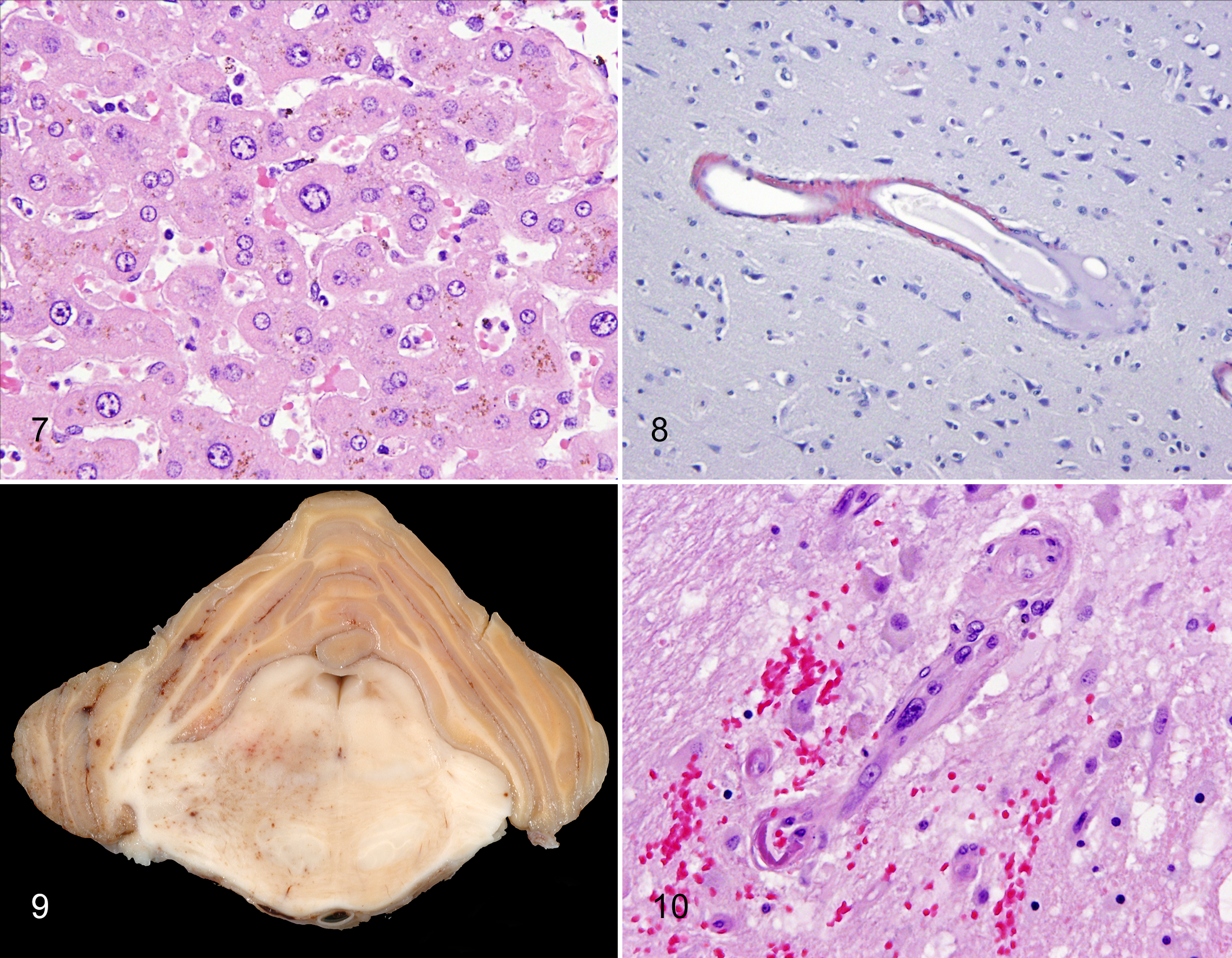
Lipofuscin accumulation in centrilobular and midzonal hepatocytes is common in wild mountain gorilla adults beginning in adolescence. 178 In contrast, in adult captive lowland gorillas, centrilobular lipofuscinosis appears to be rare, while hepatocellular hemosiderosis is common. 156 Hepatocellular lipofuscinosis was present in all adult orangutans >28 yo examined by one of us (L.J.L.), while hepatocellular hemosiderosis was in only 1 adult with chronic respiratory disease and cachexia. Obviously, additional studies are needed to determine if liver lesions and decline in liver function are common in geriatric apes.
Nervous System Disorders
Cognitive decline and neurodegenerative diseases are significant concerns in the aging human population, and there is a huge interest in spontaneous and derived animal models for understanding pathogenesis and developing therapeutic interventions. 45,77,78 Decline in cognitive function in aging humans ranges from mild forgetfulness and slower data processing to severe dementia. Dementia is a syndrome in which thinking skills are impaired to the point that the individual can no longer perform daily functions. There are many causes of dementia in humans, the main ones being Alzheimer disease (67.7%) and vascular dementia (17.4%; secondary to strokes or transient ischemic accidents). 38,137 The remaining cases of dementia are either idiopathic or due to neurodegenerative diseases (eg, Parkinson disease, Lewy body dementia). The prevalence of all dementias in the United States increases with age, with estimates of 32.7% to 50% of people >90 yo.
There is evidence that cognitive decline occurs in great apes.9,10,139,252 In aged chimpanzees and orangutans, only certain types of tasks were affected—such as social cognition, some motor skills, and, in the case of orangutans and gorillas, relative numerousness judgment.
Pathologic correlates of human brain aging, such as the senile plaques of Alzheimer disease and cerebral vascular disease, are reported in nonhuman primates, including chimpanzees, orangutans, and gorillas.87–89,135,193,206 Other hallmarks, such as dystrophic neurites and tau-neurofibrillary tangles, are less common, leading some authors to believe that true Alzheimer disease is likely unique to the human ape. 77 In orangutans (28, 31, and 36 yo), low numbers of amyloid plaques were found in cingulate gyrus and temporal lobes. 88 In 3 chimpanzees (56 and 59 yo females and a 45 yo male), amyloid-containing senile plaques were especially prominent in the older female in cortex and hippocampus, as well as amyloid deposition in meningeal and cortical vessels of both females. The plaques were immunoreactive to beta-amyloid 4 and apolipoprotein E. 87,88 A 41 yo female chimpanzee that suffered a left cerebral stroke involving temporal, parietal, and occipital lobes was found to have tau protein accumulation in neurons, abnormal neurites, and tau plaques as well as neurite plaques and neurofibrillary tangles. 206 The tau lesions were most frequent in the frontal and temporal lobes. Cerebral amyloid angiopathy (CAA) and senile plaques containing sparse beta-amyloid were also present.
Tau protein, in addition to amyloid plaques, has been detected with immunohistochemistry and immunofluorescence in the brains of older lowland gorillas from zoos. 193 The study examined frontal cortex and/or hippocampus from female gorillas 32, 35, 50, and 55 yo and males 13, 22, 42, 43, and 49 yo, using a panel of mono- and polyclonal antibodies directed against various beta-amyloid and tau epitopes, as well neurofilament and glial proteins. Amyloid plaques and vascular amyloidosis were noted in the prefrontal and frontal cortices of the 3 oldest males and 2 older females. Plaques were more frequent in females and vascular lesions more prevalent in the males. The finding of vascular amyloidosis and fewer plaques in the older males was different from the 1 previous report of a 44 yo male gorilla in which there were plaques but no vascular amyloidosis. 135 Plaques were also present in the hippocampus and dentate gyrus of the 2 oldest females and the 42 yo male. Plaque density appeared to increase with age, being 2.5 times greater in the 55 yo female than the 50 yo female. Plaques lacked a central core and were not associated with dystrophic neurites but had mild associated astrogliosis. Tau protein elements were found in fibrils in cortex and hippocampus and a few neurons in the oldest animals without associated lesions. Thus, the lesion in the geriatric gorillas had some but not the full suite of Alzheimer disease lesions, nor was cognitive dysfunction noted in the histories of any of the gorillas.
Strokes and transient ischemic accidents are significant aging phenomena in human elderly, associated with underlying cerebrovascular disease such as atherosclerosis, arteriosclerosis, and CAA. 103,137 Significant cerebrovascular changes occur in conjunction with hypertension, and hypertension is one of the greatest risk factors for strokes (see below). Similar vascular lesions, especially arteriosclerosis, arteriolosclerosis, and CAA, have been seen in the apes. 87,88 Strokes have been well documented in chimpanzees, in both laboratory and zoo settings; the 12 cases reported in 1 study ranged in age from 25 to 55 y, with most of them being >30 yo and all but 1 being female. 127 Vascular lesions, both atherosclerosis and arteriosclerosis, were identified in some but not all of the cases. We are aware of strokes in 2 bonobos (a 38 yo female and a 25 yo male; Dr Victoria Clyde, Bonobo SSP, personal communication), a 44 yo female gorilla, a 32 yo male Bornean orangutan, and a 55 yo female Sumatran orangutan. 274 In cases that we examined, CAA was seen in a 48 yo female chimpanzee without other neuropathology (Fig. 8), and severe cerebrovascular arteriosclerosis and arteriolosclerosis associated with microhemorrhage, edema, and malacia were seen in the brain of a 54 yo female orangutan with a history of hemiparesis (Figs. 9, 10).
Loss of brain volume, particularly in the frontal lobe cortex and hippocampus, is another characteristic of the aging human brain. 202 Examination of brains from chimpanzees have yielded conflicting results. A study that examined chimpanzee postmortem brain weights demonstrated a moderated loss of about 0.79 g/y. 116 A study based on MRI detected loss of gray and white matter volume. 50 The statistics were driven by a few very old chimps (>50 yo); however, gray matter volume decline in female chimps was still significant even when very old chimps were removed from the analysis. Loss of white matter occurs in humans beginning in their 50s before the onset of “old age,” and in the chimpanzee study white matter loss worsened only after 44 y, long after the suggested onset of old age (30–35 yo) and much closer to the maximum life span than in humans. 50 MRI of 69 living chimpanzees plus formalin-fixed brains from an additional 30 chimpanzees found no significant change in brain volume from ages 10 to 45 y and no decline in white matter volume. 232 A larger study consisting of MRI scans from 219 chimpanzees also showed little or no loss of volume and only mild loss of gray matter with aging. 20 Thus far, it seems that among the great apes, humans are unique in their capacity for brain shrinkage.
Neuronal lipofuscinosis is another aging change in the brain of humans and other animals. Although neuronal lipofuscinosis is a universal aging change in neurons of most species, 1 study stated that the pattern of lipofuscin accumulation, especially the relative scarcity in Purkinje cells of the cerebellum, is uniquely characteristic of humans and chimpanzees. 94 In that study, brains from 6 chimps (of which only 2 were geriatric at 36 and 45 yo), 2 gorillas (nongeriatric adults, 10 and 30 yo) and 1 young adult orangutan (13 yo) were examined and compared with humans, Old World monkeys (Papio, Erythrocebus, and Macaca, none of which were geriatric), and prosimians (Microcebus and Cheirogaleus, of which 2 were old adults) with attention directed only toward the cerebellum. The “control” human brains examined were from individuals 64 to 100 yo. Given the low sample number and relatively younger ages of the apes and other nonhuman primates, additional research is needed before the conclusions of that study can be supported.
Deterioration of Special Senses
Loss of vision and hearing can be devastating and socially isolating consequences of aging in humans. Age-related hearing loss (ARHL) begins in the fifth or sixth decade of life, and the prevalence increases with age, although estimates of prevalence vary widely. Depending on the study, ≥40% of people >75 yo are affected. 208 The prevalence in men is greater than that of women. ARHL is generally referable to changes in the inner ear structure, such as loss of hair cells in the organ of Corti, followed by neuron loss; however, primary loss of auditory neurons and changes in the auditory cortex (central hearing loss) can occur concurrently or separately. We found no reports of ARHL in any of the apes.
Presbyopia or farsightedness due to decreased lens accommodation is a common complaint of aging humans. Observations of increased focus distances of a geriatric wild female chimpanzee (estimated age 50 yo) during allogrooming led observers to suspect that she was farsighted. 82 Far more serious ocular aging changes in elderly humans, such as macular degeneration, can lead to partial or complete blindness. Age is the most significant risk factor for macular degeneration and cataracts. 194 Vascular-based retinopathies, which can lead to loss of vision, include those associated with hypertension and type II diabetes, both of which increase in the elderly.
Nonhuman primates have a macula and retinal anatomy similar to that of humans. 285 but there is only 1 published report of suspected macular degeneration in apes. A 31 yo “geriatric” female lowland gorilla with OA and severe periodontal disease was noted to have problems with near, but not distance, vision. 240 Ophthalmologic examination revealed depigmented spots in the maculae similar to drusen bodies, leading to a diagnosis of the dry form of macular degeneration.
Cataracts are not uncommon in aging apes, including chimpanzees, orangutans, and gorillas. Although most often age related, cataracts have been reported in young gorillas and orangutans 6 and 14 yo. 61,147,174 In other reports, zoo-housed apes with cataracts have included gorillas aged 37, 39, and 42 y and a chimpanzee aged 29 y. 61,147,150,174 Cataracts have also been identified in wild mountain gorillas in the Mountain Gorilla Veterinary Project database (Gorilla Doctors, L.J.L. unpublished data). All animals were adults. Those of known age ranged from 21.5 to 37 y. In most cases, the lesions were mild, but one 24 yo female had a mature mineralized cataract that would have impaired vision, and another female, with estimated age of 37 yo, had extensive bilateral cortical cataracts.
Retinal disease due to vascular lesions was described in a 34 yo female gorilla with a history of hypertension (based on measurements taken during anesthetic events). 185 She developed acute blindness due to bilateral retinal detachment and intraocular hemorrhage. Cataracts were also present. Retinal vascular arteriolosclerosis characteristic of hypertension was found.
Ophthalmologic pathology of the apes is yet another area needing further investigation.
Cardiovascular Disease
In humans, even in the absence of negative lifestyle choices, such as smoking and unhealthy eating habits, structural and functional changes take place in the aging cardiovascular system. 58,162,279 “Baseline” or “intrinsic” aging changes include an increase in left ventricular wall thickness in the absence of hypertension (particularly in males), decreased numbers of myocytes, increased myocyte size and polyploidy (compensatory hypertrophy), increased interstitial connective tissue and matrix, intimal and median thickening of arteries and loss of elasticity (vascular stiffening), increased left atrial size, and altered matrix in heart valves. 19, 58, 60, 79, 162, 231 In humans, myocardial remodeling also includes genesis of cardiomyocytes from cardiac stem cells. 227
Polyploidy is considered a normal part of myofiber growth in the postnatal growing heart. 8 Polyploidy increases due to stress and strain on the heart and in situations of decreased energy availability to the cardiomyocytes, therefore indicating the presence of a pathologic process. 8,140,227,269 Likewise, the accumulation of lipofuscin in the perikaryon of cardiac myocytes is thought to be a normal aging change that increases during myocardial remodeling due to oxidative stress and altered autophagy. 62 Autophagy is essential for cardiac homeostasis. 254 Autophagy is protective and antiapoptotic, but the accumulation of excessive lipofuscin prevents normal autophagy and is proapoptotic in the aging heart. 62
Heart Disease in Apes Compared With Humans
Heart disease is the leading cause of death in older humans in the United States, accounting for about 25% of all deaths in individuals >64 yo. 117 Based on reviews of the SSP populations and laboratory colonies, cardiovascular disease is also the leading cause of death in captive great apes: 45% of bonobos (SSP data), 41% of gorillas, 38% of zoo chimpanzees, >50% of colony chimpanzees, and 20% of orangutans.* The prevalence of cardiovascular disease increases with age in apes as in humans, although death can occur in nongeriatric adults.
One of the major causes of heart disease in older humans is atherosclerotic coronary arterial disease (CAD) with subsequent cardiac ischemia or infarction causing sudden death or congestive heart failure. 230 In contrast, in all the apes, an idiopathic condition variously termed fibrosing cardiomyopathy, interstitial myocardial fibrosis, or idiopathic cardiomyopathy is the most commonly diagnosed entity. 142,168,220,260 This condition resembles idiopathic myocardial fibrosis in humans, which occurs in the absence of arterial obstruction and has been recognized and debated about for decades. 222 It also resembles the lesions of human idiopathic cardiomyopathy, hypertensive cardiomyopathy, and experimentally induced catecholamine cardiomyopathy in rhesus macaques. 93,133,229 Sudden deaths (hypothesized to be due to arrhythmias associated with conduction system fibrosis) and congestive heart failure both occur in affected apes. 144,168,171 In contrast to CAD, the vessels, primarily endomyocardial (intrinsic) arteries and arterioles, in affected great apes have varying degrees of median hypertrophy and hyalinization (arteriosclerosis and arteriolosclerosis; Figs. 11, 12). However, especially in chimpanzees, fibrosis can occur without detected arterial changes. 142,253,260 Gross and microscopic changes in the hearts of aging apes are similar to “baseline” aging, but those with cardiac deaths often have exaggerated changes, especially left ventricular hypertrophy, interstitial fibrosis, and polyploidy. Polyploid nuclei in the apes (and Old World monkeys) are often convoluted, in contrast to the “box car” nuclei described in humans. 229
Cardiac disease increases with age in apes. In gorillas, echocardiography revealed increasing left ventricular wall thickness from 1.6 cm in young adult males to >2.1 cm in males >31 yo, while left ventricular chamber size decreased. 180 In that study, female gorillas showed little difference in cardiac measurements with advancing age, with left ventricular wall thickness ranging from a minimum of 0.9 cm to a maximum of 1.4 cm. All of the males between 31 and 40 yo in the study were considered to have cardiomyopathy. Increased body weight/obesity correlated with cardiomyopathy. Although males are more often affected, our postmortem examination of hearts showed that females may also develop marked myocardial fibrosis, even in the absence of left ventricular hypertrophy.
Echocardiography of normal adult chimpanzees comparing animals of different ages (10–49 yo) did not find significant differences in left ventricular wall thickness of either males or females with age. 234 However, left ventricular hypertrophy is seen in some but not all chimpanzees with myocardial fibrosis (Figs. 13, 14). 142
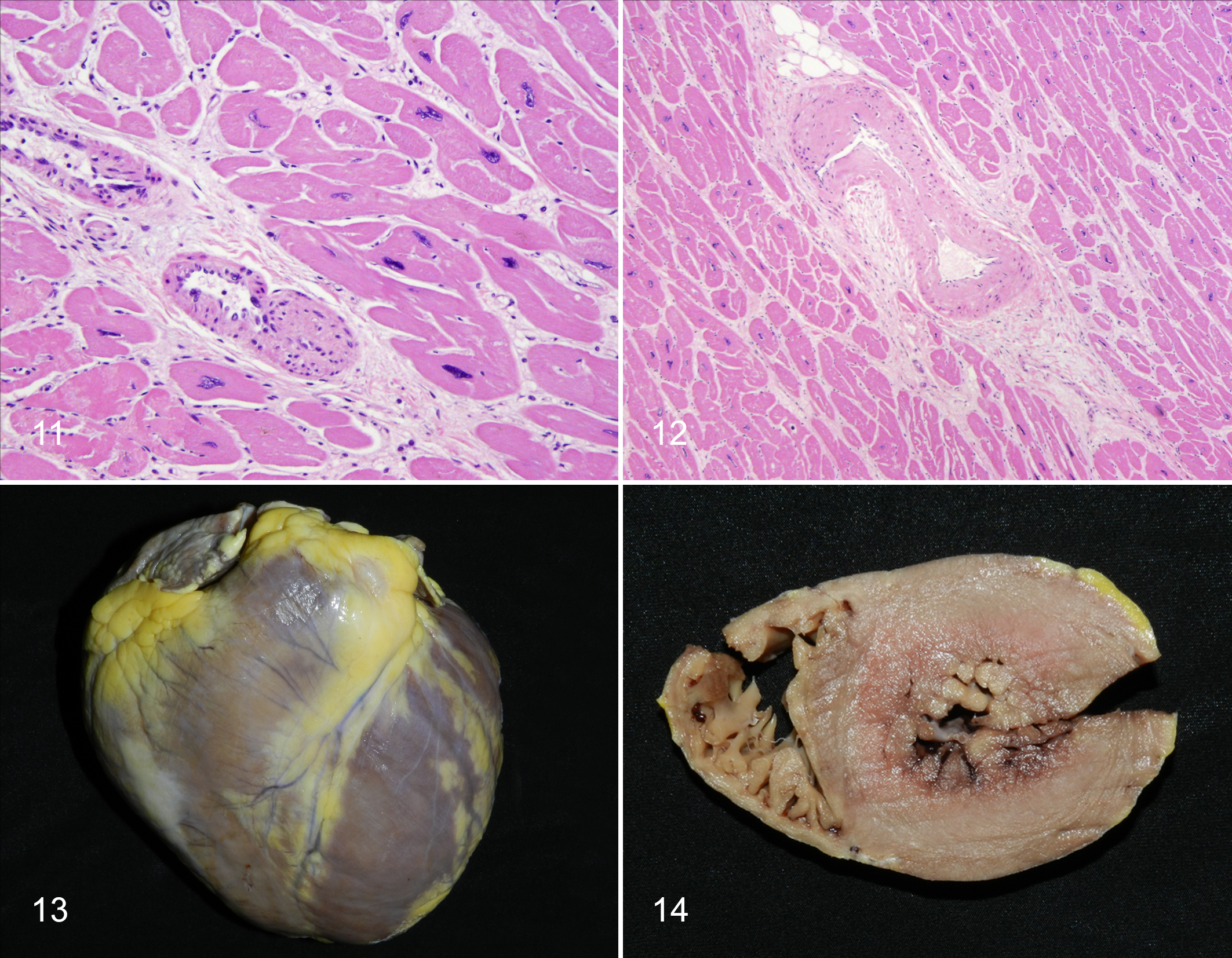
Left ventricular hypertrophy, as well as arteriosclerosis of intrinsic coronary arteries, suggests the possibility of hypertension as an underlying pathogenesis for fibrosing cardiomyopathy in apes. 221,248,276
Hypertension
Hypertension in humans increases with age, and it is estimated that >60% of individuals >65 yo are hypertensive. Increasing arterial stiffness is thought to be important in the pathogenesis. 247,248 Metabolic syndrome, which increases with age (see above), also contributes to vascular stiffness and hypertension. Other factors include chronic inflammation (“inflammaging”), abnormal sympathetic nervous system activity, psychosocial/life stress, and chronic renal disease. 32,43,64 Salt sensitivity increases with age (likely due to changes in the renal interstitium and associated changes in renin/angiotensin), contributing to hypertension. 152
As mentioned above, heart lesions in the apes are suggestive of hypertension, and several lines of evidence support this hypothesis. First is the echocardiographic pattern and gross morphology of the ape hearts with generally concentric left ventricular hypertrophy. The histologic changes are also compatible: polyploid hypertrophied myocytes and dissecting interstitial fibrosis, rather than large foci suggestive of true infarction. 222,230,248 There are also changes in intramural (intrinsic) coronary arteries suggestive of hypertension, at least in gorillas and orangutans. 64 Changes in small-caliber vessels are those of mural thickening and luminal narrowing, brought about by either “eutrophic inward remodeling” or smooth muscle cell hypertrophy. 113 Altered character and amount of extracellular matrix and mural hyalinization suggestive of leakage of plasma into the wall due to endothelial dysfunction also contribute to the appearance of hyaline arteriosclerosis. 36, 84, 162 The arterial change is often segmental within the intramural (intrinsic) coronary arteries and arterioles. In contrast, the larger epicardial coronary vessels are usually unaffected, similar to “small vessel disease” in humans. 177
Definitive proof of the relationship between hypertension and ape heart disease relies on antemortem demonstration of hypertension. Developing antemortem normal blood pressure reference ranges and documenting hypertension has been difficult in the apes because compliance with awake blood pressure monitoring requires extensive training and validation of methodology (documenting peripheral vs central pressures). Most reports of blood pressure monitoring are in sedated or anesthetized animals, and anesthetic protocols can affect these values. Blood pressure reference ranges have been reported for chimpanzees. 68 Diastolic pressure was documented to increase with obesity in females but not in males. However, males in general had higher diastolic pressures than females. Age was a factor in elevated diastolic blood pressure but not systolic. 69 Experimental dietary studies in chimpanzees have shown individual variability with regard to “salt sensitivity” and development of hypertension, but the presence of concurrent renal disease or other confounding factors was not investigated. 63
Hypertension was diagnosed antemortem in 2 bonobos, a mother and son, who both died of stroke (Dr Victoria Clyde, Bonobo SSP, personal communication). The male had massive cardiomegaly and an aortic dissection. Both had histologic vascular changes of hypertension, and both had myocardial fibrosis, although only the male’s heart was enlarged. Hypertension was diagnosed antemortem in 2 gorillas (based on blood pressure measurements during anesthetic events). In a 34 yo female, there was marked left ventricular hypertrophy (free wall 2.6 cm thick) and median hypertrophy with “onion skin fibrosis” in small arteries in the retina, choroid, and meninges of the brain, kidney, and heart. 185 In a 28 yo male, hypertension was documented over a 2 y period. 171 He developed congestive heart failure and died under anesthesia. The heart was dilated with marked interstitial fibrosis and more mild fatty infiltration in the left ventricle, as well as mural thickening of small arterioles. Hypertension was suspected to be the etiology of left ventricular hypertrophy, cardiogenic death, and historical stroke in a 32 yo orangutan. 276 Renal changes compatible with hypertension were also documented.
Aortic Dissections: Further Evidence for Hypertension?
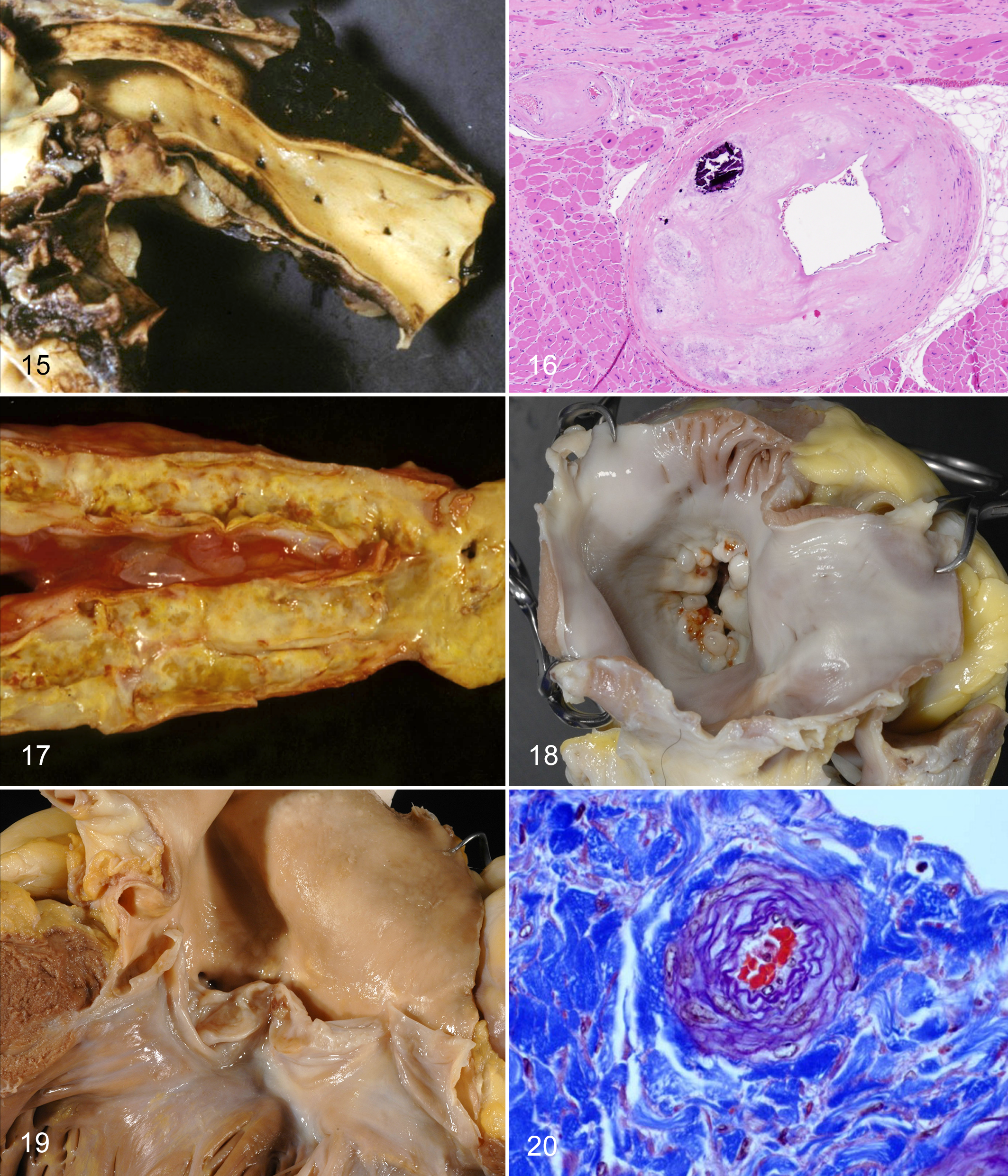
In humans, the mean age of aortic dissections is 63 y, and males account for 65% of cases. 186 The most common risk factors are hypertension and connective tissue disorders such as Marfan syndrome. Aortic dissections have been identified as a major problem causing sudden death in gorillas. 132 Of 8 affected gorillas in that report, 6 were males. Ages ranged from 16 to 43 yo (mean age 31 y). All dissections were in the ascending aorta (DeBakey type II, Stanford type A) with rupture of the aorta occurring just distal to the aortic valves or within an aortic sinus (Fig. 15). It is the second-most common cardiovascular problem in gorillas. Similar dissections have caused death in 4 male bonobos (13.7–25.7 yo; SSP database) and 1 chimpanzee >35 yo. 85 These data help support the contention that hypertension is a plausible explanation for “fibrosing cardiomyopathy.” However, no dissections have been identified, to our knowledge, in orangutans, which also suffer from “fibrosing cardiomyopathy.” Since myocardial fibrosis is an “end point” with many possible pathogeneses, it would actually be surprising if hypertension were the entire “answer” to all ape heart disease. 58,141
Atherosclerosis
Although the pathology data do not support coronary atherosclerosis as a cause of chimpanzee heart disease, it still warrants examination as a possible aging change in apes. 260 In humans, atherosclerosis increases with age, and atherosclerosis of coronary arteries is the most common cause of cardiac deaths and congestive heart failure among elderly humans. 273,274 The pathogenesis of atherosclerosis is still not completely elucidated. Current understanding is that it is the result of complex processes involving endothelial damage, inflammation, dyslipidemias, and changes in arterial intercellular matrix. 230,273,274 Cardiovascular risk factors such as hypertension and dyslipidemias contribute to the initial endothelial damage as well as to subsequent events in the progression of the disease.
Does atherosclerosis occur in captive or wild apes? Literature from the 1960s and 1970s stated that chimpanzees had the highest “rate” of spontaneous atherosclerosis of any nonhuman primate species, and chimpanzees have been used as animal models for atherosclerosis induced by “atherogenic” diets. 165 More recent studies of laboratory-housed chimpanzees found that only 3 of 29 chimpanzees with myocardial fibrosis had mild atherosclerosis. 142 Another study identified 8 of 87 chimps with arteriosclerosis/atherosclerosis with a mean age of 33 yo, which was significantly older than those without arterial disease. 225 Affected vessels were in brain (n = 4), aorta (n = 3), kidney (n = 1), heart (n = 1), and adrenal (n = 1), confirming that atherosclerosis was not typically associated with heart disease in chimpanzees. In zoo chimpanzees examined by one of us (L.J.L.), aortic atherosclerosis was seen in a 34 yo female and a 56 yo male, both without coronary atherosclerosis. Clinical studies in chimpanzees have suggested that hypercholesterolemia and dyslipidemias are not predictive of atherosclerosis. 260
In gorillas, plaques/grade I atheromas were demonstrated by transesophageal echocardiography in the ascending and descending aorta in gorillas 10, 26, 27, and 36 yo and grade II atheroma in a 40 yo male, suggesting that severity increases with age. 22 Plaques were correlated with decreased high-density lipoprotein, increased cholesterol:high-density lipoprotein ratio and increased ApoB/ApoA1 but not total cholesterol alone. In our experience, coronary artery atherosclerosis is rare in gorillas, although in the past CAD was reported to have caused myocardial infarction in gorillas. 101 We have seen atherosclerosis involving small-caliber intrinsic coronary arterioles in the absence of involvement of the larger epicardial vessels in only 2 gorillas: a 34 yo male with chronic heart failure and a 39 yo female with hypertension and stroke (Fig. 16).
Atherosclerosis in zoo orangutans is less well described. A 20 yo female died with myocardial infarction associated with left anterior descending coronary artery atherosclerosis. 223 She had a history of high total cholesterol values (329 and 341 mg/dl). In adult orangutans examined by one of us (L.J.L.), including 5 males (12, 28, 33, 34, and 39 yo) and 4 females (12, 15, 45 and 56 yo), atherosclerosis has been confined to the aorta, with the most severe lesions in the caudal abdominal aorta and internal iliac arteries in the older males (Fig. 17).
Based on the above, under current management practices, we agree with the contention that coronary arterial atherosclerosis is rare, not only in chimpanzees, but in all the apes. 260 The significance and pathophysiology of atherosclerosis in the aorta of great apes, particularly orangutans, bear further investigation.
Cardiac Valvular Disease
The 2 main forms of valvular disease in aging humans are myxomatous degeneration of the mitral valve (well known to veterinary pathologists because of the similar condition in dogs) and sclerosis and calcification of the aortic valve. 19,231 Aging changes in the human mitral valve include increase in polyglycans, increase and remodeling of collagen, and decrease in elastin. Myxomatous degeneration in humans is not considered to be part of normal aging, but when it occurs, it becomes clinically significant in the sixth decade of life and is associated with either mitral insufficiency or mitral stenosis. Aortic valve sclerosis and calcification is a disease of elderly humans and is characterized by stiffening of aortic valve margins and nodular deposition of fibrous tissue with calcification and, occasionally, bone formation. 257
Other than infective valvular endocarditis, valvular disease has not been well described in the apes. Isolated valvular lesions were very rare in echocardiograms of 99 western lowland gorillas, although mild mitral regurgitation was been noted. 180 In the apes that we examined, infective valvular disease was seen in a few mountain gorillas, and myxomatous degeneration of mitral valve was diagnosed in three >30 yo mountain gorilla females, a 42 yo male orangutan, and a 48 yo female zoo chimpanzee (Fig. 18). A single case of aortic valve fibrosis and mineralization was diagnosed clinically and confirmed on cardiac necropsy in a 47 yo male zoo gorilla (Fig. 19).
The high prevalence of heart disease in captive apes is cause for great concern in the zoo and laboratory communities. Much work is needed to better define the lesions and pathogenesis. To this end, the Great Ape Heart Project (GAHP), based at Zoo Atlanta, has developed standardized protocols for ante- and postmortem evaluation of ape hearts and a relational database that will allow analysis of clinical and necropsy data to better understand, diagnose, treat, and ideally prevent great ape heart disease (see http://greatapeheartproject.org/).
Cardiovascular Disease in Wild Apes
An important and not yet completely answered question is whether cardiovascular disease, particularly “fibrosing cardiomyopathy,” is found in wild apes or if it is the result of captive management. In a limited study of wild chimpanzees, myofiber hypertrophy as evidenced by karyomegaly (polyploidy) was seen even in immature animals, but fibrosis was present in only 2 individuals: an aged female (44 yo) and an adult male (28 yo). 253 Arteriosclerosis was not noted in those animals. In a review of causes of mortality in mountain gorillas, cardiovascular disease was considered to be the cause of death in only 3% of animals. 189 Both male and females were involved. More recently a higher prevalence was noted in eastern lowland gorillas (G. b. graueri) from an isolated population on Mt Tshiaberimu in the Democratic Republic of Congo. 131 Lesions included myocardial fibrosis and arteriosclerosis in intrinsic coronary arteries, along with marked myofiber hypertrophy as described in zoo-housed lowland gorillas. Atherosclerosis has not been identified in coronary arteries of wild mountain gorillas, but fatty streaks and plaques have been seen in the aortas of 2 geriatric females, 35 and 39 yo, both of whom were cachectic and had advanced cancers. Additional necropsies with detailed examination of the heart, based on protocols developed by the GAHP, are needed to determine the nature and extent of cardiovascular disease in wild apes.
Renal Aging
Renal senescence is a common but not universal occurrence in elderly humans. This term encompasses decline in renal function as well as decreased ability to respond to acute injuries. Renal aging is viewed as a complex process with multiple etiologies; the pathophysiology has been the subject of several excellent reviews of the aging human kidney and rodent models. 31,159,161,275 In humans, it is influenced by sex (males > females), race (African Americans > Caucasians), genetics, and the presence of cardiovascular disease. Genomic studies have shown kidney aging to be associated with differential expression in as many as 500 genes, including increased expression of immune and inflammatory proteins, increased synthesis and turnover of interstitial matrix, increase in oxidative stress markers, and decreased expression of genes associated with collagen degradation and metabolic processes. 31 For example, expression of the klotho gene appears to be particularly important in suppressing age-related changes in kidney and other organs. 31 In addition, upregulation of acute- and chronic-phase reactants provides important markers of microinflammation, which contributes to injury and aging in kidney and heart. 251
Aging is accompanied by functional as well as morphologic changes. Several studies stress that creatinine clearance and glomerular filtration rate (GFR) decrease up to 55% in individuals >80 yo, when compared with maximum GFR of people in their 20s or 30s (<60 vs 90–120 ml/min/1.73 m2). 159 Interestingly, decline in GFR is not always accompanied by increases above the reference intervals for blood urea nitrogen (BUN) and creatinine, possibly due to altered dynamics of creatinine production due to sarcopenia and diet in the elderly. It was suggested by some authors that the reference range for normal creatinine should be lowered for the elderly. It was also suggested that low GFR not be used to diagnose renal dysfunction in the absence of proteinuria or other evidence of renal disease. 31
Another important functional change is decreased renal blood flow, including actual flow and that in relation to total cardiac output. 161 Morphologic correlates in kidneys of elderly humans and rodent models include vascular changes such as arteriosclerosis and arteriolosclerosis, median smooth muscle hypertrophy, and intimal fibrosis, which are similar to changes seen in younger adults afflicted with hypertension and diabetes. 31, 161 Eventually, altered blood flow leads to glomerular sclerosis and obsolescence in the outer cortex, with associated tubular atrophy and interstitial fibrosis in both cortex and medulla. In addition to collagen accumulation, accumulation of normal matrix constituents (eg, hyaluronan) and abnormal constituents (eg, advanced glycation end products) contributes to increased extracellular matrix in the glomerular mesangium and in the tubulointerstitium. 31,152,161 In the medulla, abnormal interstitium can significantly alter fluid and sodium balance and may account for the “salt sensitivity” of the elderly. 152 Other histologic changes in elderly kidneys may include epithelial tubular lipofuscinosis and nonspecific infiltration of the interstitium by inflammatory cells, primarily lymphocytes and macrophages, in both cortex and medulla. This change is consistent with chronic interstitial nephritis and is similar to changes seen in older dogs and cats. The diagnosis of “chronic interstitial nephritis” in humans is usually confined to specific inflammatory conditions, while in veterinary medicine it has a much broader connotation. Nonetheless, interstitial inflammation is considered an important contribution to chronic renal disease in humans. 205
Clinically, declining renal function appears to be common in aging laboratory-housed chimpanzees. 264 This is evidenced by increasing levels of BUN and creatinine in male chimpanzees beginning in the 25 to 30 yo range and in females in the 30- to 35 yo range. Increased urine protein:creatinine ratios in chimps >24.5 yo revealed protein leakage suggestive of abnormal glomerular function. 143 In a clinical study of 16 female chimps >35 yo, renal disease was present in 31.5% and characterized as nephrotic syndrome in 3 animals, hydronephrosis in one and “unclassified renal disease” in another. 188
In contrast to the above laboratory chimpanzee studies, it must be noted that renal diagnoses in zoo-housed apes derive from compiled necropsy reports, which are typically based on light microscopy alone and not always supported by clinical renal data. However, the accurate diagnosis of glomerular disease depends on the use of light microscopy, electron microscopic, and immunofluorescent techniques. All 3 modalities are routinely used in human nephropathology and have recently been used, along with clinical data, in dogs to diagnose glomerular disease. 53 To date, such detailed analyses have not been performed in zoo animals, but complementary retrospective and prospective renal studies are planned activities of the GAHP, as part of ape cardiovascular disease investigation (see above).
In mortality reviews of zoo chimpanzees, chronic renal disease was considered the cause of death in 3 of 24 (12.5%) adults 10 to 35 yo but in none of the 11 animals >35 yo. 85 Severe renal lesions consisting of membranous glomerulopathy with multifocal global sclerosis and tubular protein were seen in 2 older adult wild chimpanzees (K.A.T.), but overall prevalence of renal disease in wild chimpanzees is yet to be determined. Renal lesions in laboratory chimpanzees and the connection between cardiac and renal disease are covered in another paper in this issue of Veterinary Pathology. 52
In other apes, renal vascular lesions and interstitial fibrosis were seen in the 2 hypertensive bonobos, highlighted above in the Hypertension section. Renal disease was considered to be the cause of death in 26% of the 15 orangutans >40 yo but in only 16% to 18% of the 95 younger adults 20 to 40 yo. 155 Renal disease was a comorbidity in an additional 20 younger adults (21%) and 5 older adults (33%). The most common diagnoses were “chronic interstitial nephritis,” “chronic glomerulonephritis,” and “end-stage kidney.” The etiology is uncertain and likely not the same in all cases. In some older males, interstitial nephritis was suspected to be associated with previous bouts of cystitis and ascending infection. Hypertension may be playing a role in renal disease in some orangutans; in at least 1 older male that we examined, there were glomerular and renal vascular lesions suggestive of underlying hypertension. Additional support for this possibility comes from the orangutan SSP mortality review. 155 For 122 of 168 orangutans >15 yo (subadult males, adults/old adults of both sexes), cardiovascular disease, respiratory infections, and renal disease were the most frequent primary, secondary, or tertiary causes of death. In animals with >1 of these diagnoses, a statistical association was noted between cardiovascular and renal disease but not cardiovascular and respiratory disease or renal and respiratory disease (SSP, unpublished data).
An interesting morphologic change observed in 3 adult male orangutans was dense accumulation of tubular epithelial pigment (lipofuscin)—periodic acid–Schiff positive, Perl’s negative—that was grossly visible as greenish-brown discoloration of the outer stripe of the medulla. This change was not seen in the kidneys of any other apes that we examined.
In contrast to orangutans, renal disease was considered the cause of death in only 1 adult female lowland gorilla and 1 wild mountain gorilla, both of which had pyelonephritis. 170,189 However, renal disease as a comorbidity was not investigated in either of these gorilla studies. Incidental aging changes that we observed in captive and wild gorilla kidneys include medullary interstitial hyalinization, due to an increase in extracellular matrix positive for Masson’s trichrome and periodic acid–Schiff, and mild multifocal cortical interstitial and periglomerular fibrosis. In 1 male gorilla dying of aortic dissection, hypertrophic changes in small renal arterioles were suggestive of hypertension (Fig. 20). More detailed investigation of gorilla renal disease is needed.
Immunosenescence and Anemia
Based on studies in humans and laboratory rodents, the innate and adaptive immune systems show declining function and dysregulation with age (immunosenescence). 4,104 Although numbers of neutrophils, monocytes, and natural killer cells do not decline with age, they are functionally less robust for parameters such as intracellular killing, cytotoxicity, and macrophage clearance of infections. B cells diminish in diversity of response to known and novel antigens. T cells are also less diverse with reduced function, and T-cell counts in peripheral blood tend to decline. Immune system dysregulation leads to a chronic proinflammatory state with elevated levels of markers of inflammation, such as C-reactive protein, that may contribute to accelerated aging in some individuals (so-called inflammaging). 28,283 Vitamin E and zinc deficiencies may also play a role in immune function senescence.
Immunosenescence has been examined in baboons but not in aging apes. 126 Examination of causes of mortality in captive ape populations subjectively showed no increase in infections with advanced age. 85,155,170 Infections were common among all age classes of free-ranging mountain gorillas and chimpanzees that we reviewed (L.J.L., K.A.T.). Changes in the hemograms of male and female chimpanzees from young adulthood through old age were reviewed. 264 Although there was no decrease in total white blood cell counts, lymphocyte counts were not addressed specifically.
Red cell parameters (total red blood cell count, hemoglobin, and hematocrit) were decreased especially in older adult male chimpanzees >30 yo; they approximated or were lower than those of the same-age adult females (in which these same parameters increased with age). 264 Anemia in elderly humans is common and multifactorial, with most frequently reported causes being chronic inflammatory diseases, iron deficiency (usually due to blood loss), chronic renal disease, vitamin B12 (cobalamine) deficiency (ie, pernicious anemia), and decrease in testosterone and other androgenic steroids in men. 12,218 During reproductive years, the hematocrit of female mammals is generally lower than that of males due to suppressive effects of estrogen and the positive influence of androgenic hormones. In the chimpanzee study, although red cell parameters decreased in males, none of the animals were considered to be anemic. 264 Also, although renal parameters (BUN and creatinine) increased with age in male and female chimps, values were not indicative of renal failure
Whether immunosenescence occurs in the nonhuman apes is yet another area for further investigation.
Neoplasia
Other than a few notable childhood tumors (eg, embryonal nephroma and some hemolymphatic malignancies), neoplasia in most species is associated with aging. 195 Malignancies are the second cause of death in humans >64 yo in the United States, after heart disease, and they surpass heart disease in the 65 to 74 yo age group. 117 The prevalence of cancers in apes appears to be much lower than in humans and Old World monkeys, based on reviews and case reports in the literature. 29,154,236 Possible explanations have included the facts that (1) the longer human life span allows more time for accrual of genetic and epigenetic events central to carcinogenesis, (2) immunosenescence causes decline in tumor surveillance, and (3) humans are likely exposed to a greater variety of potentially carcinogenic agents. Yet other explanations may be the relatively fewer necropsies that have been performed on apes compared with laboratory-housed Old World monkeys and humans and the fact that apes in the wild and, historically, those in captivity seldom reached “old age.” However, a biological basis is hinted at in a study that found lower expression of proapoptotic genes in humans versus chimpanzees, which, though surely not the entire “answer,” would favor carcinogenesis and cancer progression in humans. 14
A review of neoplasms in 2 populations of captive laboratory-held chimpanzees and in the published literature found 102 naturally occurring and 12 experimentally induced neoplasms. 39 The average age at diagnosis of benign tumors was 32 yo and malignancies, 25 yo. Benign neoplasms were almost 6 times more frequent than malignancies. As mentioned above, uterine leiomyomas were one of the most frequent tumors in older female chimpanzees. Hepatocellular neoplasms were common as well, often, though not exclusively, occurring in animals that had been experimentally inoculated with hepatitis B virus. No neoplasms were identified in 12 wild chimpanzees necropsied at Gombe Tanzania. 253
In the SSP population of chimpanzees, neoplasia was a cause for mortality in 2 of 24 adults 10 to 35 yo (8.3%) and 1 of 11 old adults >35 yo (9%). 85 In bonobos, only a single report of neoplasia has been published. 92 A 1994 review of causes of death or euthanasia in zoo-housed western lowland gorillas found neoplasms in 1 of 13 adult males 9 to 30 yo and 1 of 7 adult females >30 yo. 170 A 2008 review of mortality in zoo-housed orangutans found malignancies in 5 of 170 animals (of which 110 were adults or old adults). 155 These studies undoubtedly underrepresent the number of neoplasms in apes, as only neoplasms causing mortality were reported.
Table 1 presents spontaneous neoplasms diagnosed in apes in SSP reviews, the published literature, and proceedings abstracts and those that we diagnosed, including those in free-ranging mountain gorillas. Benign tumors of the skin or mucus membranes (eg, lipomas, fibromas, papillomas) were excluded. Neoplasms of the female reproductive tract feature prominently, especially in chimpanzees and gorillas, as do hemolymphatic malignancies in orangutans and gorillas. Several endocrine tumors, mostly adenomas, are also identified. Most were clinically silent except for a pituitary prolactinoma in a gorilla. 49 Notably infrequent were several of the neoplasms found in elderly humans, including mammary, colorectal, and bladder cancers. Interestingly, neoplasia, including malignancies, has been found in wild mountain gorillas.
Spontaneous Neoplasms Identified in Great Apes.
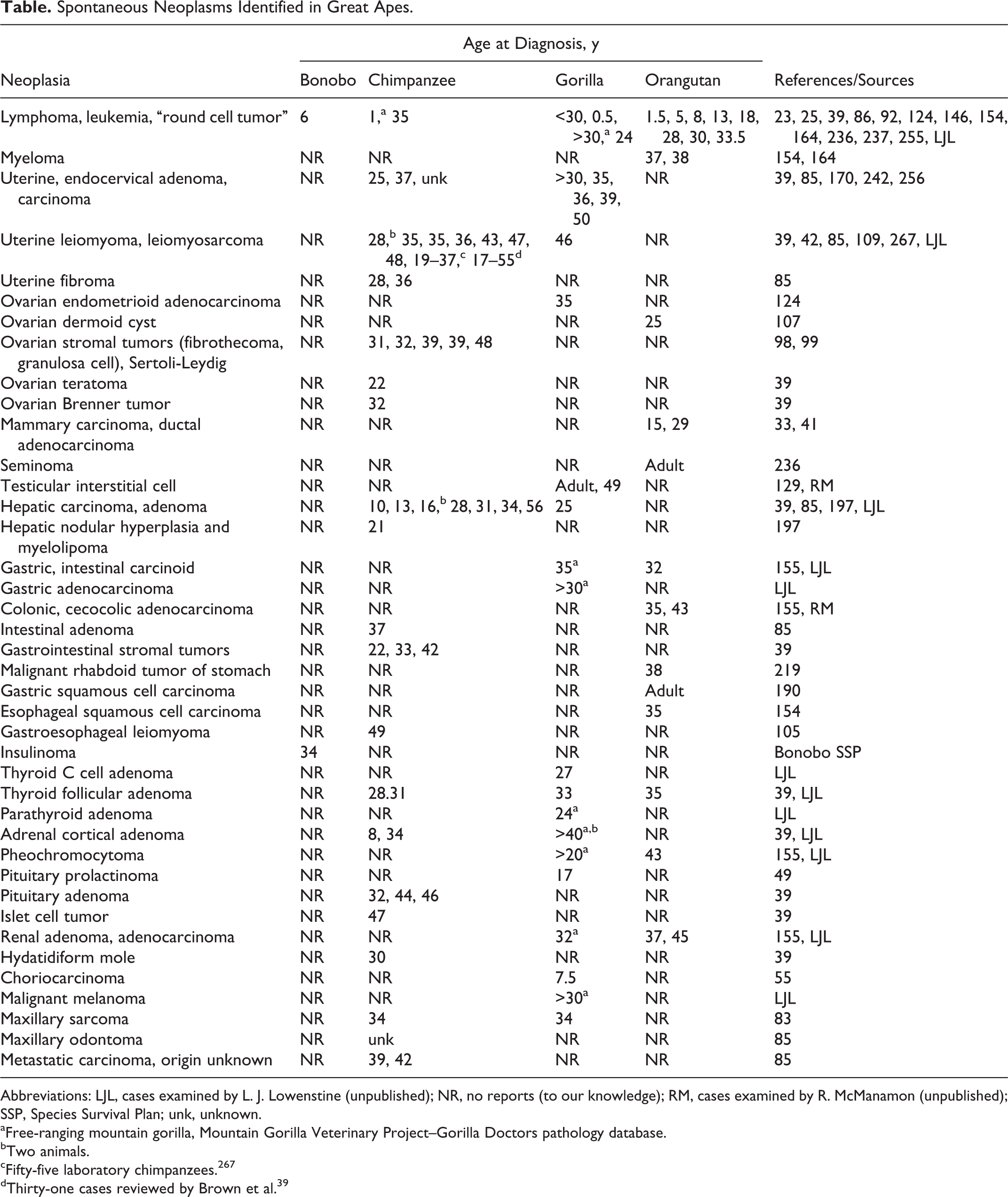
Abbreviations: LJL, cases examined by L. J. Lowenstine (unpublished); NR, no reports (to our knowledge); RM, cases examined by R. McManamon (unpublished); SSP, Species Survival Plan; unk, unknown.
aFree-ranging mountain gorilla, Mountain Gorilla Veterinary Project–Gorilla Doctors pathology database.
bTwo animals.
cFifty-five laboratory chimpanzees. 267
dThirty-one cases reviewed by Brown et al. 39
Conclusions
From all of the above, it is clear that great ape gerontology is in its infancy as compared with the understanding of aging processes in the human ape. Many of the changes found in aging apes are consistent across species, with some notable differences. Arthritis is more common in gorillas than the other apes, and inflammatory spondyloarthropathy is relatively more common in all the apes than in humans. Renal disease is more commonly a cause of mortality in aging orangutans and chimpanzees than in gorillas. The prevalence of neoplasia appears lower in apes than in humans, and the types of neoplasms differ among the apes. Benign uterine leiomyomas are more frequent in chimpanzees, while malignancies of the female reproductive tract are more common in gorillas. Hemolymphatic malignancies are more common in orangutans and gorillas than in chimpanzees. Like the human population, populations of apes in captivity are aging, providing challenges to end-of-life care. Retrospective “mining” of existing pathology databases for zoo-housed and free-living apes and the development of additional pathology programs to study diseases of wild apes in their range countries will help us gain insights into the hominid aging process. Prospective studies such as the GAHP will help us better define the pathology of ape senescence, helping inform management decisions. As is the case for improving health in aging in humans, it is hoped that further understanding of the pathology of aging in apes will provide information for improving their health throughout their life spans. 224
Footnotes
Acknowledgements
We thank past and current Species Survival Plan management groups, especially the coordinators for bonobos, chimpanzees, gorillas, and orangutans, for entrusting us over the years with the pathology databases and all zoo veterinarians who have submitted cases to us for necropsy and histopathology. We thank our pathology colleagues at our home institutions, zoos, diagnostic laboratories, and Yerkes National Primate Research Center who performed some of the necropsies and shared their data with us. We also thank our valued collaborators, the past and present Species Survival Plan veterinary advisors, especially Dr Victoria Clyde (bonobos); Drs Kay Backues and Kathryn Gamble (chimpanzees); Drs Pam Dennis, Tom Meehan, and Hayley Murphy (gorillas); and Drs Chris Bonar, Nancy Lung, and Joe Smith (orangutans). We also thank the present and past directors of the Mountain Gorilla Veterinary Project, especially current codirectors Drs Mike Cranfield and Kirsten Gilardi, and all the Gorilla Doctors, field veterinarians, and regional veterinary managers who have contributed cases to the Mountain Gorilla Veterinary Project pathology database. Cases submitted by Karisoke Research Institute (Diane Fossey Gorilla Fund International) are also acknowledged.
Inclusion of pathology data from wild gorillas and chimpanzees would not be possible without the consent and valuable cooperation of the in-country authorities, and we especially acknowledge Dr Antoine Mudakikwa and Elizabeth Nyirakaragire of the Rwanda Development Board, the Institut Congolais pour la Conservation de la Nature in the Democratic Republic of Congo, and the Uganda Wildlife Authority for the gorilla cases. For chimpanzee cases, we thank Drs Elizabeth Lonsdorf, Iddi Lipende, Michael Kinsel, and Jane Raphael, Jane Goodall Institute, and the responsible Tanzanian authorities for continued support of Gombe Stream Research Centre.
We also thank Dr Laura D. Braswell for review of the oral disease portion of the manuscript, Dr Cathy A. Brown for helpful comments on the renal disease section, Dr Brian Murphy for review of the section on arthritis, and Dr Wilson Yau for invaluable help with the image plates.
All wild gorilla and chimpanzee samples were imported with proper CITES and USPHS permits.
Notes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Portions of this work were supported in part by the Yerkes National Primate Research Center (base grant P51 OD11132) awarded by the National Center for Research Resources of the National Institutes of Health. The wild chimpanzee pathology was funded by the Leo S. Gutham Foundation and by a grant from the National Institutes of Allergy and Infectious Diseases (R01158715A).
