Abstract
Myocarditis can cause death or permanent heart damage. As epidemiologic and etiopathologic data for canine myocarditis are lacking, we performed a retrospective study using nucleic acid extracted from archived (2007 to 2015) tissues from myocarditis cases and control dogs without myocardial lesions. Heart tissue from pediatric/juvenile and adult dogs was tested with a comprehensive panel of conventional and real-time polymerase chain reaction (PCR) assays targeting recognized agents of canine myocarditis based on a literature review and informed by the comparative epidemiology of human myocarditis. The PCR screen, which included canine parvovirus 2 (CPV-2), canine distemper virus, canine herpesvirus, Borrelia spp, West Nile virus, adenovirus, parainfluenza virus, pneumovirus, respiratory coronavirus, influenza virus, Bartonella spp, Rickettsia spp, Mycoplasma spp, and Neospora caninum, did not detect agents in 35 of 66 cases (53%; 95% confidence interval [CI], 41%–65%) and was frequently negative in adults (21/26); by comparison, agents were not detected in 27 of 57 controls (47%; 95% CI, 35%–60%). Canine distemper virus, herpesvirus, adenovirus, coronavirus, parainfluenza virus, Mycoplasma haemocanis, and N. caninum were occasionally detected in both cases and controls; thus, PCR detection was not considered to indicate causation. We previously reported that CPV-2 continues to be associated with myocarditis in young dogs despite widespread vaccination; in adults, CPV-2 was detected in 2 of 26 cases and 4 of 22 controls. As several agents were similarly detected in cases and controls, it is unclear if these are cardiopathogenic, incidental, or latent. West Nile virus was detected at the analytic limit in 1 adult case. We did not detect Borrelia spp, Bartonella spp, Rickettsia spp, or influenza A virus in the myocarditis cases. These data demonstrate the limitations of current targeted diagnostic tests and the need for additional research to identify unknown agents and develop testing strategies for canine myocarditis.
Keywords
Although myocarditis is a devastating disease resulting in permanent cardiac damage or death, the cause is not known for most veterinary cases. Systemic viral, bacterial, or protozoal infections are implicated, but in practice, the underlying cause is often not identified as there are few confirmatory clinical tests for canine myocarditis. Diagnosis is most frequently made at postmortem, and published investigations are largely limited to necropsy case reports and case series. Screening for all of the reported causes of canine myocarditis in individual clinical cases is cost prohibitive and often unrewarding, and thus targeted testing strategies are needed.
Myocarditis is characterized by nonspecific clinical signs, including acute weakness, syncope, exercise intolerance, respiratory difficulties, and/or abdominal distension; arrhythmias or left- or right-sided congestive heart failure are potentially fatal complications. 91 A presumptive diagnosis is based on a combination of patient history, clinical signs, disease course, physical examination findings, echocardiographic and electrocardiographic abnormalities, and elevated circulating cardiac troponin I (cTnI) levels, which is unfortunately not specific to myocarditis. 20,43,49 Treatment is largely supportive.
The substantial deficiencies in clinical diagnosis and treatment of canine myocarditis are related to a paucity of epidemiologic data. In humans, viral infections are associated with the majority (69%) of myocarditis cases. 40 In contrast, the prevalence of viral myocarditis in the dog is undetermined. Autopsy series have been invaluable in understanding the epidemiology of human myocarditis and have informed specific molecular testing recommendations that have improved myocarditis diagnosis. 13,22,23,40,67,72,97,108
In the dog, there have been limited molecular investigations of myocarditis. Maxson et al 81 tested for a small number of viral agents (canine parvovirus 2 [CPV-2], canine adenovirus [CAV], canine herpesvirus [CHV]) not detected in their small cohort of canine myocarditis cases (n = 9). Our previous retrospective study demonstrated a significant association of CPV-2 with myocarditis/myocardial fibrosis in young dogs 2 years of age or less; however, the cause of myocarditis remained unknown in the majority of cases, and the role of CPV-2 in adult myocarditis was not investigated. 44 As there have been few advances in our ability to diagnose and treat myocarditis in companion animals, more information is needed to elucidate the underlying causes.
The goal of this study was to direct testing strategies, improve disease diagnosis, and contribute to the understanding of the etiopathogenesis of this devastating disease. We first performed a literature review for primary reports of natural causes of canine myocarditis. Based on this review and the human literature, we hypothesized that viral infections are frequent causes of canine myocarditis, with different agents affecting pediatric/juvenile and adult dogs. We then performed a retrospective case-control study to test this hypothesis on archived tissue from a cohort of dogs representing different age classes. Myocardial samples were tested with a comprehensive panel of validated polymerase chain reaction (PCR) assays to screen for possible causes of canine myocarditis in our region (northeastern United States), encompassing reported causes of canine (CPV-2, canine distemper virus [CDV], CHV, Borrelia burgdorferi, West Nile virus [WNV], Bartonella spp, Rickettsia spp, and Neospora caninum) and human myocarditis (adenovirus, herpesviruses, and influenza virus), as well other agents common in the canine population. 46
Materials and Methods
Literature Review
A literature review was performed using the Medline Database from 1970 to April 2017 searching for primary reports of natural causes of canine myocarditis using the search terms canine and dog, in conjunction with myocarditis and myocardial inflammation. For causes of myocarditis cited in textbooks, we attempted to identify a primary resource. 24,80 Reports were excluded if they described experimental induction of disease, described an unconfirmed/unknown agent (ie, not identified by culture, molecular methods, other ancillary testing, or specific histologic characteristics), lacked description of pathologic features, did not identify the causal agent in the heart, or were learning exercises (ie, “Pathology in Practice” manuscripts). Reports describing endocarditis, pericarditis, or epicarditis without extension into the myocardium were excluded. The search was focused on causes, demographic information, and pathologic features.
Retrospective Case Selection
Formalin-fixed, paraffin-embedded (FFPE) tissue from the Cornell Animal Health Diagnostic Center (AHDC) pathology archives was used to evaluate dogs with myocarditis and/or myocardial fibrosis for infectious agents. A search of archived cases from June 2007 to November 2015 was performed to identify adult dogs 2 or more years of age of any breed or sex using similar diagnostic terms consisting of myocardial fibrosis, myocarditis, cardiac fibrosis, chronic myocarditis, fibrosing cardiomyopathy, cardiac failure, restrictive cardiomyopathy, or end-stage heart. Controls were identified from a similar age distribution and time period using the aforementioned keywords as exclusion criteria to identify adult dogs with ostensibly healthy hearts. Our previously described cohort of pediatric/juvenile cases and controls that were 2 years of age or less was also tested. 44
Histopathology was evaluated by a veterinary pathologist (K.K.) blinded to disease status. Cardiac tissue was scored histologically as previously described to assess the severity of necrosis, inflammation, and fibrosis on a scale of 0 to 3 (0 = absent, 1 = mild [up to 10%], 2 = moderate [between 10% and 25%], 3 = severe [over 25%]). 44 Myocardial fibrosis was evaluated by Masson trichrome stain. Gram stain, Gomori-methenamine silver, periodic–acid Schiff, and/or Warthin-Starry histochemical stains were performed on a subset of cases to assist in detection of organisms. Histochemical stains were performed by the AHDC Histopathology Laboratory.
Nucleic Acid Extraction
Nucleic acid was extracted from two 10-μm sections of archived FFPE cardiac tissue sectioned into sterile Eppendorf tubes. The microtome blade was changed between samples and the microtome face was cleaned with 70% ethanol to minimize cross-contamination. Samples were incubated overnight in lysis buffer with proteinase K at 60°C. After incubation, the tubes were briefly spun down and allowed to cool to room temperature. Lysates (200 μL) were transferred to a 96-well plate for extraction using the Biomek 4000 automated workstation (Beckman Coulter, Indianapolis, IN) Total Nucleic Acid preset program with the Agencourt Formapure Kit (Beckman Coulter) according to manufacturer instructions. Nucleic acid was quantified with a Nanodrop ND-1000 spectrophotometer (Thermo-Fisher, Waltham, MA); the average nucleic acid concentration was 336 ng/μl.
PCR
All PCR experiments were run with appropriate controls, including a no-template reaction as well as a confirmed positive for each pathogen. The 95% confidence intervals (CIs) of total proportions with PCR detection were calculated using the modified Wald method and GraphPad QuickCalcs (GraphPad Software, La Jolla, CA).
Conventional PCR
Conventional PCR was performed using housekeeping gene RPS19 primers and CPV-2 VP1-VP2 primers as previously described. 15,44,84 Conventional PCR for Neospora canis was performed as previously described with the following alterations to PCR conditions: denature (94°C; 1 minute), annealing (50°C; 1 minute), and primer extension (74°C; 3.5 minutes) repeated for 40 cycles. 130 PCR reaction mixtures were separated on a 1.5% agarose gel (Thermo Scientific, Waltham, MA) and run at 125 V for 45 minutes. Amplicon sequencing was performed by the Cornell University Biotechnology Resource Center and analyzed using a BLAST search of GenBank.
Real-Time PCR
Samples were diluted to 50 ng of DNA/μl. All real-time PCR (rtPCR) testing was performed semiquantitatively with platform-specific cycle threshold (Ct) values interpreted according to interquartile ranges based on serial dilutions of positive controls across the dynamic range of the assay. The Ct value is the number of cycles when amplification is detected and is inversely proportional to the amount of target nucleic acid in the sample.
rtPCR testing for WNV was performed at the microliter scale on the StepOne platform (Applied Biosystems, Foster City, CA). 38 To avoid false negatives, WNV PCR was not performed on cases/controls submitted during low-risk periods for WNV exposure (ie, 3 weeks after the first hard frost until an ambient temperature supporting mosquito activity). rtPCR was performed using primers and probe to amplify the CPV-2 VP2 gene as previously described. 44 rtPCR for CHV-1 was performed on the 7500-FAST platform (Applied Biosystems) using published primer/probes with the following PCR conditions: 10 minutes at 48°C, then 3 minutes at 95°C, followed by 40 cycles of 15 seconds at 95°C, then 60 seconds at 60°C. 70 Reactions were carried out in a 25-μl volume (4 μl of nucleic acid per reaction) using PathID multiplex 1-step real-time PCR master mix (Applied Biosystems). rtPCR for Mycoplasma haemocanis was performed as previously described. 8,96
Nanoliter-Scale rtPCR
Reverse transcription and preamplification were performed together using a pool of 18 primer sets (for either respiratory or tick-borne pathogens) and the TaqMan FastVirus 1-step master mix (Applied Biosystems). Preamplified products were loaded onto custom nanoscale PCR plates (OpenArray; Applied Biosystems) printed in a 18 × 3 format. 46 The pathogens targeted included canine respiratory coronavirus (CRCoV, betacoronavirus), CDV, CAV, canine parainfluenza virus (CPiV), influenza A, Mycoplasma cynos, and Streptococcus zooepidemicus. The CAV assay is optimized for detection of CAV-2. The tick-borne pathogens targeted included Bartonella spp, B. burgdorferi, Borrelia mayonii, Borrelia miyamotoi, and Rickettsia spp. To avoid false negatives, tick-borne pathogen testing was not performed on puppies less than 6 weeks of age. Nanoscale panels were run at the AHDC.
Immunohistochemistry and In Situ Hybridization
Immunohistochemistry (IHC) for CDV (mouse monoclonal gift from James A. Baker Institute of Cornell University, Ithaca, NY), CPV-1 (rabbit polyclonal gift from James A. Baker Institute Carmichael Laboratory Cornell University), adenovirus (MAB8052; Millipore, Temecula, CA), WNV (clone 7H2; VRL, Gaithersburg, MD), and coronavirus (FIPV3-70; Custom Monoclonals International Sacramento, CA), was performed on a subset of cases by the AHDC Histopathology Laboratory. Tissue with confirmed infection and sections incubated without primary antibody served as positive and negative controls. In situ hybridization for CHV-1 and CPV-2 was performed as previously described with appropriate controls. 44,56
Results
Literature Review
According to search criteria, the causes and characteristics of natural, nonexperimental myocarditis in dogs described in 45 reports are summarized in Table 1 (and detailed in Suppl. Table S1). There were 13 reports of viral myocarditis, including WNV, 18,25,74 CPV-2, 1,11,27,44,50,52,71,88,102,111 and CPV-1/minute virus. 51 There were 15 reports of protozoal myocarditis, including Leishmania infantum, 103,121 N. caninum, 6,58,68,82,85,90,98 Trypanosoma cruzi (Chagas disease), 31,64,127,128 Hepatozoon americanum, 9 and Toxoplasma gondii. 53 There were 9 reports of bacterial myocarditis, including Bartonella spp, 14,35,41,109 B. burgdorferi, 33,57,73 Citrobacter, 28 Clostridium piliforme, 131 and Staphylococcus spp. 57 There were 6 reports of fungal myocarditis, including Inonotus tropicalis, 101 Coccidioides immitis, 16,100 Aspergillus spp, 21,61 and Blastomyces dermatitidis, 107 and a report of algal myocarditis caused by Prototheca spp. 115 Case reports and case series represented the vast majority of reports (43/45). Ten reports described features of myocarditis in dogs of various ages; 30 reports included pediatric/juvenile cases, and 26 reports described features of adult cases. Ten reports described CPV-2 myocarditis in pediatric/juvenile dogs and not in adults. Fungal and algal cases were mostly described in adults.
Summary of Features of Natural Myocarditis Previously Reported in the Dog.
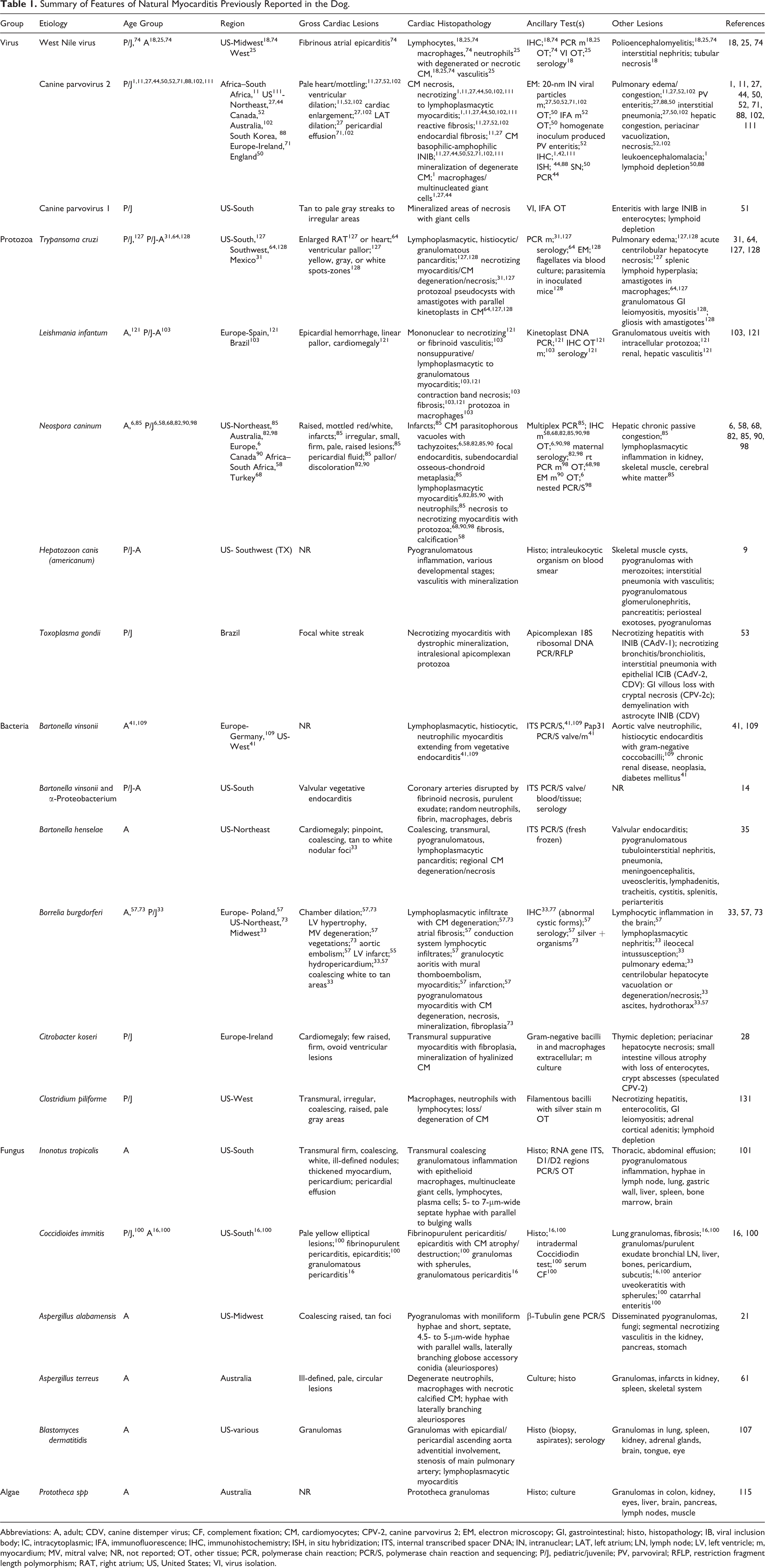
Abbreviations: A, adult; CDV, canine distemper virus; CF, complement fixation; CM, cardiomyocytes; CPV-2, canine parvovirus 2; EM, electron microscopy; GI, gastrointestinal; histo, histopathology; IB, viral inclusion body; IC, intracytoplasmic; IFA, immunofluorescence; IHC, immunohistochemistry; ISH, in situ hybridization; ITS, internal transcribed spacer DNA; IN, intranuclear; LAT, left atrium; LN, lymph node; LV, left ventricle; m, myocardium; MV, mitral valve; NR, not reported; OT, other tissue; PCR, polymerase chain reaction; PCR/S, polymerase chain reaction and sequencing; P/J, pediatric/juvenile; PV, parvoviral; RFLP, restriction fragment length polymorphism; RAT, right atrium; US, United States; VI, virus isolation.
Case Demographics
A search of the AHDC archives (June 2007 to November 2015) identified 1911 canine cases from the search period. Twenty-seven adult cases and 22 controls were identified using search criteria. PCR was performed on 26 cases with available tissue (Suppl. Table S2). The demographics of pediatric/juvenile cases and controls was previously reported. 44 Cases and controls were primarily from the northeastern United States.
The median age (minimum, maximum) of adult myocarditis/myocardial fibrosis cases was 6 years (2, 14). There were 5 castrated males, 4 intact males, 12 spayed females, and 5 intact females. Case breeds included mixed breeds (2), Maltese (2), Staffordshire Bull Terrier (2), Labrador Retriever (6), Beagle Hound (2), and 1 each of Greyhound, Shetland Sheepdog, French Spaniel, Golden Retriever, Rottweiler, Bichon Frise, Scottish Terrier, Pomeranian, Siberian Husky, Welsh Corgi, Weimaraner, and Puli. The median age of control cases was 9 years (2, 13). The controls included 6 castrated males, 3 intact males, 8 castrated females, and 5 intact females. Control breeds included Labrador Retriever (1), German Shepard (2), Staffordshire Bull Terrier (2), French Bulldog (2), Rat Terrier (2), Mixed Breed (2), Cocker Spaniel (2), and 1 each of Chihuahua, Spaniel, Coton De Tulear, Bulldog, Boston Terrier, Australian Shepard, Beagle Hound, Wheaton Terrier, and Cane Corso. The distribution of myocarditis cases by submission month is shown in Figure 1. Adult myocarditis cases occurred at a low frequency throughout the calendar year while pediatric/juvenile cases were most numerous in the summer (June/July) and fall (October/November).
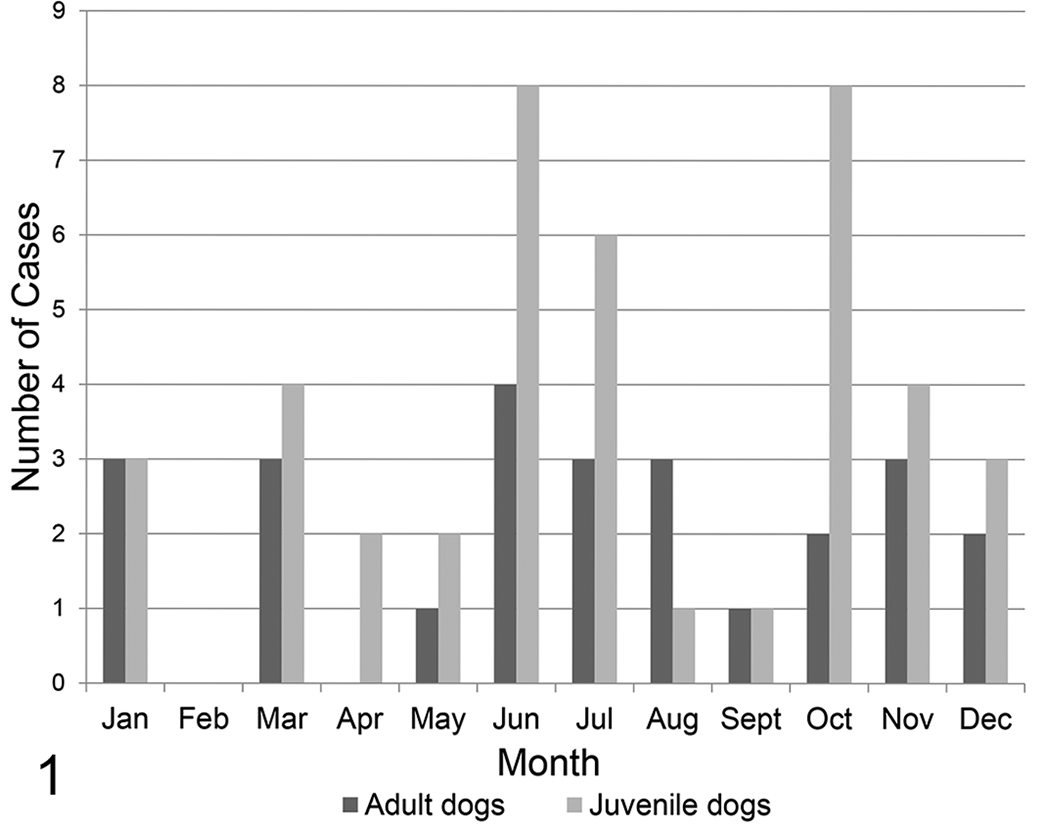
Canine myocarditis submissions for necropsy from June 2007 to November 2015 grouped by month and age.
The summary results of PCR testing are shown in Table 2. Not all PCR tests were performed for all cases/controls due to the aforementioned exclusion criteria. Nucleic acid from 12 different pathogens was detected by PCR in 14 pediatric/juvenile cases and 19 pediatric/juvenile controls, as well as in 5 adult cases and 11 adult controls. More than 1 agent was detected in the myocardium of 4 pediatric/juvenile cases and 4 pediatric/juvenile controls, as well as in 2 adult cases and 4 adult controls. The PCR testing did not detect any agents in 35 of 66 cases (53%; 95% CI, 41%–65%), including 14 of 40 pediatric/juvenile and 21 of 26 adult cases. No agents were detected in 27 of 57 controls (47%; 95% CI, 35%–60%).
Polymerase Chain Reaction Detection of Infectious Agents in Canine Myocarditis Cases and Controls by Age Class.
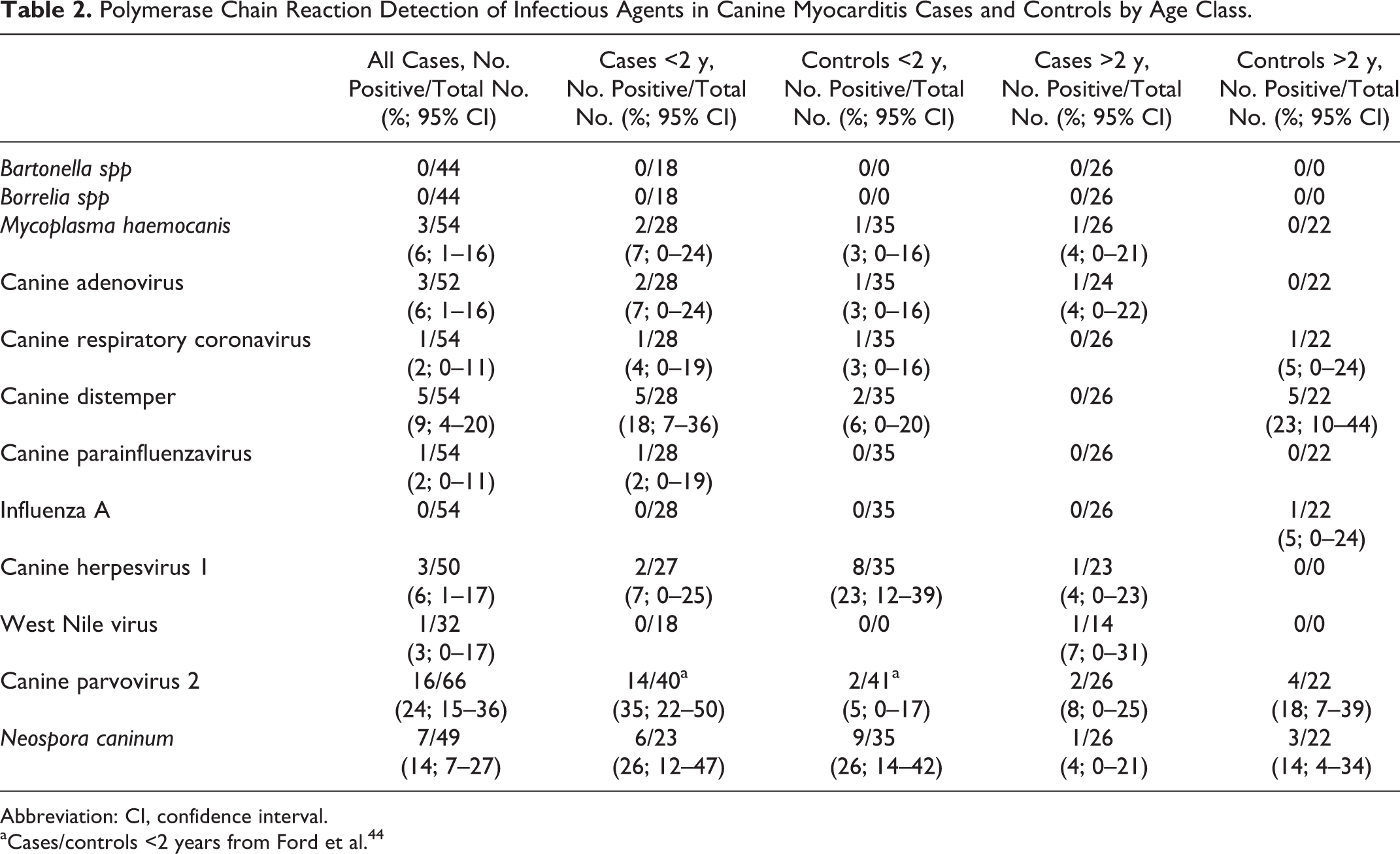
Abbreviation: CI, confidence interval.
aCases/controls <2 years from Ford et al. 44
M. haemocanis was detected by rtPCR in pediatric/juvenile myocarditis cases, pediatric/juvenile controls, and adult myocarditis cases but not in adult controls. In pediatric/juveniles, M. haemocanis was detected with M. cynos. The rtPCR-positive cases and controls were in the low-positive range of the assay (Ct values 21.2–30.0), at or below the analytic detection limit, and attempted amplicon sequencing was unsuccessful.
CAV was detected by rtPCR in pediatric/juvenile cases and pediatric/juvenile controls, as well as in an adult case but not in adult controls. Although CAV rtPCR-positive cases and controls were in the moderate Ct range for the nanoscale platform (11.86–13.19), adenoviral inclusions and antigen were not detected in the myocardium of cases or controls by histopathology and IHC, respectively.
CRCoV was detected by rtPCR in a single pediatric/juvenile case, pediatric/juvenile control, and adult control but not in adult cases. Although CRCoV PCR-positive cases and controls had moderate Ct values (range, 15.05–19.37), CV antigen was not detected in the myocardium of cases by IHC.
CDV was detected by rtPCR in pediatric/juvenile cases, pediatric/juvenile control, and adult controls but not in adult cases. The median Ct value for pediatric/juvenile cases was 13.74 (range, 7.61–15.86); the Ct values for pediatric/juvenile controls were 16.99 and 17.05. The 7.61 value was considered high positive, and all others were moderate. CDV antigen was not convincingly detected by IHC in the myocardium of PCR-positive cases or controls. In pediatric/juvenile cases and controls, CDV nucleic acid was codetected with nucleic acid of other agents, including CAV (2/5 cases, 1/2 controls), M. cynos (1/5 cases), CPiV (1/5 cases), CPV-2 (2/5 cases), and Neospora (2/5 cases). The median CDV Ct value for adult controls was 18.13 (range, 13.30–19.30) and in the moderate range similar to pediatric/juvenile controls.
CPiV was detected by PCR in 1 pediatric/juvenile case (moderate Ct value 17.99) but not in pediatric/juvenile controls, adult cases, or adult controls.
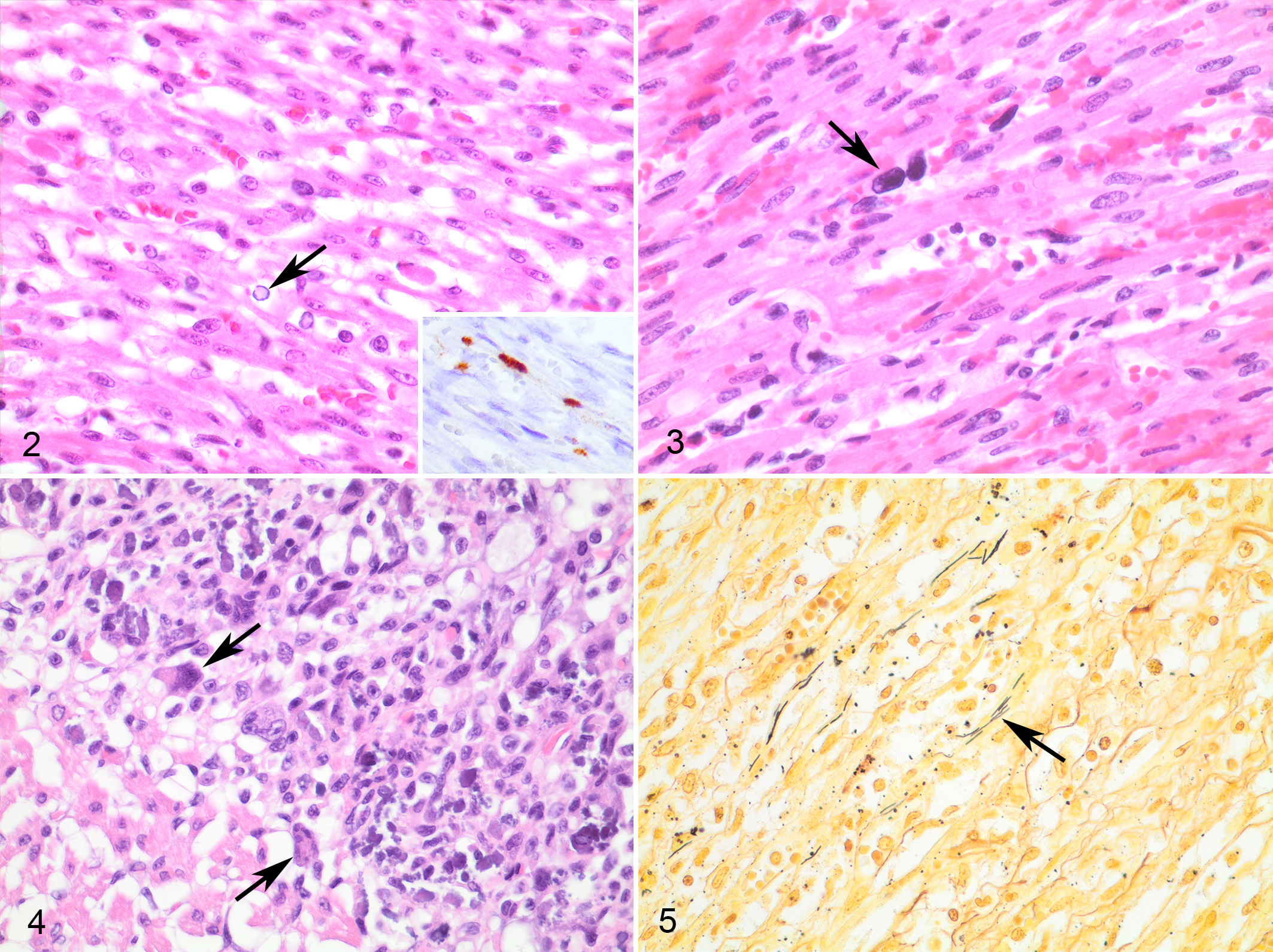
CHV-1 was detected by PCR in pediatric/juvenile cases, pediatric/juvenile controls, and 1 adult case. The Ct range for pediatric/juvenile cases was 31.23 to 38.44 (median 37.94 Ct) or approximately 1 viral genome copy per microliter (range, 25.64–39.91). CHV-1 was detected in the myocardium of an 8-day-old PCR+ case using in situ hybridization (ISH) with signal localized to endothelial cells (case 40, Fig. 2) along with endothelial inclusions, cardiomyocyte degeneration, mineralization, hemorrhage, and mild neutrophilic inflammation considered secondary to myocardial damage. Herpesviral inclusions were not observed in other cases or controls.
WNV was detected at or below the analytic detection limit and was thus considered low positive (Ct value 37.7) in 1 adult case but not detected in pediatric/juvenile cases, pediatric/juvenile controls, or adult controls. In the low-positive case, WNV antigen was not detected in the myocardium via IHC.
Neospora nucleic acid was detected in pediatric/juvenile cases, pediatric/juvenile controls, an adult case, and adult controls. Amplicon sequences from 2 of 7 PCR-positive pediatric/juvenile cases matched N. caninum sequences in Genbank (top 3 matches KX683873.1, LN714476.1, and KF6498848.1) with high (95%–96%) identity. Sequencing of other cases failed due to signal noise. Neospora PCR detection occurred without histological evidence of characteristic protozoa.
CPV-2 was detected in adult cases and controls in the low-positive range. The Ct range for adult cases was 27.69 to 31.33, and the median Ct value for adult controls was 31.01 (Ct range, 29.61–32.60); with standard curve interpolation, the range of PCR-positive adult cases was 8.35 × 101 and 1.15 × 103 copies and the median quantity (minimum, maximum) of controls was 1.05 × 102 copies (3.35 × 101, 2.88 × 102). CPV-2 was not detected in the myocardium of adult cases using ISH.
The Lyme exposure status was unknown for the majority of cases except for 2 adult cases with medical histories of positive serology, but B. burgdorferi was not detected in cardiac tissue in any cases or controls by rtPCR. B. miyamotoi is also present in the northeastern US region but was not detected in any samples. We did not detect Bartonella spp, Rickettsia spp, or S. zooepidemicus in any of the cases or controls regardless of age.
Cardiac histologic features were not associated with detection of agents by PCR. Demographic, histologic, and tissue detection characteristics of cases with PCR detection are presented in Table 3. In the adult cohort of 26 cases, 18 had some evidence of cardiomyocyte necrosis, 20 had inflammation, and 9 had some degree of myocardial fibrosis; 11 cases had a combination of necrosis, inflammation, and fibrosis. Five cases had predominantly necrosis, 9 cases had a combination of necrosis and inflammation, 9 cases were predominately inflammatory, and 3 cases had predominantly fibroplasia or fibrosis. A review of case histories and pathology reports revealed systemic disease in 15 cases, including 10 cases with systemic inflammation consistent with sepsis. Significant lesions were limited to the myocardium in 7 cases, including 5 cases that were predominately inflammatory. There were also 4 cases of mild cardiac necrosis, inflammation, or fibrosis, which most likely represented incidental changes. One pediatric case had multifocal to regional cardiomyocyte necrosis with dark, inclusion-like material in endothelial nuclei (case 18, Fig. 3), and 1 pediatric case had multifocal cardiomyocyte necrosis with mineralization and multinucleate cells also not immunoreactive for CPV-1 (case 22, Fig. 4). 44 CPV-1 was not detected by IHC in the subset of pediatric/juvenile cases with suggestive myocardial necrosis, mineralization, and giant cells.
Demographic, Histologic, and Tissue Detection Characteristics of Cases With Agent(s) Detected by PCR Screen.
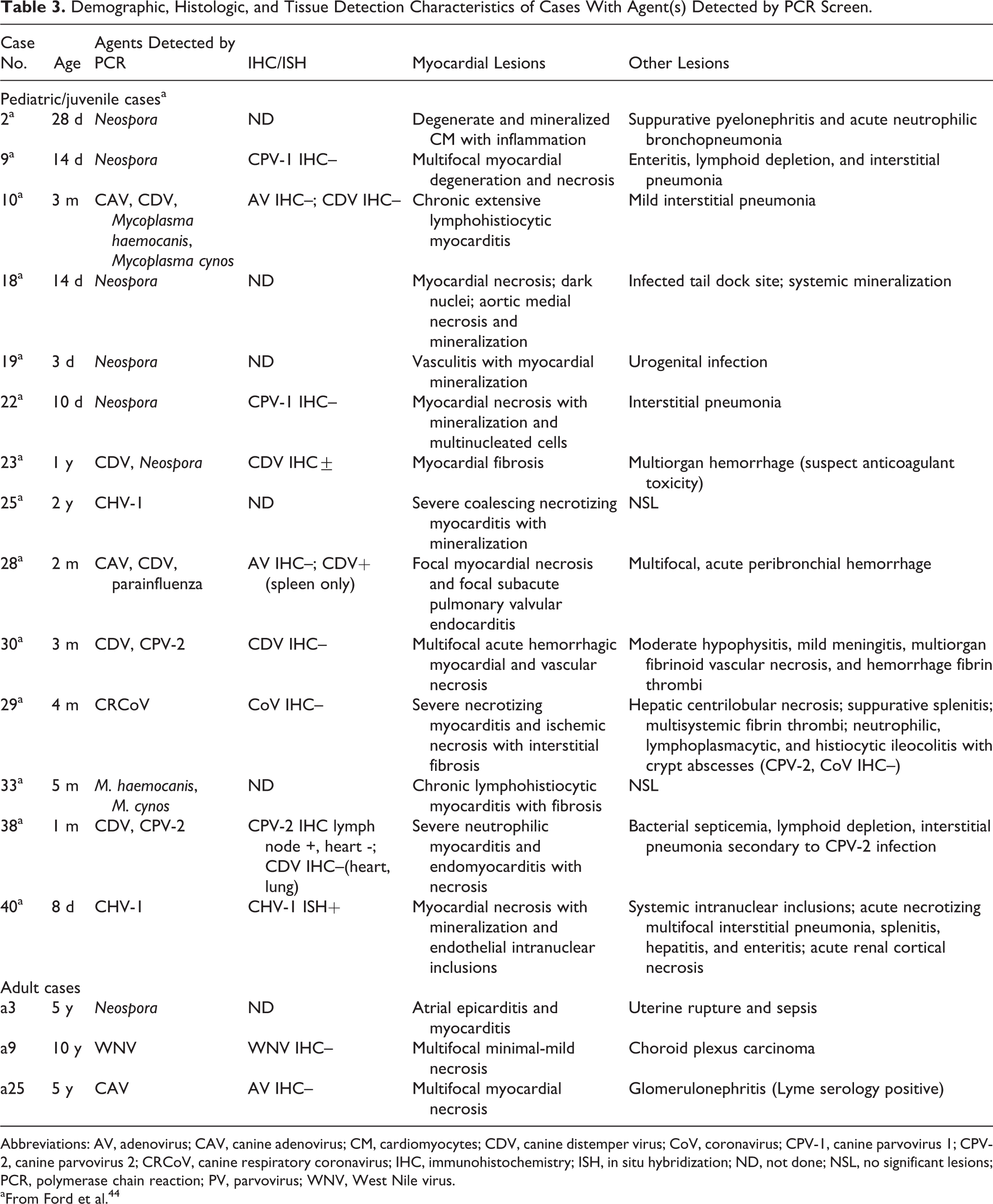
Abbreviations: AV, adenovirus; CAV, canine adenovirus; CM, cardiomyocytes; CDV, canine distemper virus; CoV, coronavirus; CPV-1, canine parvovirus 1; CPV-2, canine parvovirus 2; CRCoV, canine respiratory coronavirus; IHC, immunohistochemistry; ISH, in situ hybridization; ND, not done; NSL, no significant lesions; PCR, polymerase chain reaction; PV, parvovirus; WNV, West Nile virus.
aFrom Ford et al. 44
Aerobic bacterial culture isolated Enterococcus spp (1/3) and Escherichia coli (2/3) from myocardial or other tissues of 3 pediatric/juvenile cases, but intralesional bacteria were not evident in examined myocardial sections. Bacterial culture was not performed on tissues from adult cases. Gram or modified Steiner stain was performed on 8 pediatric/juvenile cases with bacteria identified in 3. Bacteria morphologically consistent with C. piliforme were identified in 1 pediatric case (case 31, Fig. 5) with silver histochemical stain.
Discussion
Despite a comprehensive PCR screen, the cause of myocardial lesions remained undetermined for a large proportion of cases in our study. In particular, the cause of myocarditis was not known for the majority of adult cases, demonstrating the limitations of available molecular tests. Furthermore, agent detection by PCR did not imply causation: M. haemocanis, CAV, CRCoV, CPiV, CDV, and CHV were not associated with myocarditis as detection occurred with equivalent frequency in myocarditis cases and controls. Several agents of myocarditis (T. cruzi, L. infantum, H. americanum, C. immitis) were not identified in our cohort but would not occur in our region (northeastern United States) without travel history to the respective endemic areas.
Our research builds on previous research of canine myocarditis, which has been largely limited to case reports and case series reporting Bartonella spp, 14,35,41,109 Borrelia burdorferii, 33,57,73 Citrobacter koseri, 28 C. piliforme, 131 Staphylococcus spp, 57 L. infantum, 103,121 I. tropicalis, 101 N. caninum, 6,58,82,85,90,98 T. cruzi (Chagas disease), 31,64,127,128 WNV, 18,25,74 CPV-2, 1,11,27,44,50,52,88,102 CPV-1/minute virus, 51 H. americanum, 9 T. gondii, 53 C. immitis, 16,100 Aspergillus spp, 17,21,61 B. dermatitidis, 107 and Prototheca spp 115 as natural causes. There are reports of cardiac involvement with CHV 94 and CDV 55 experimental infection. The frequency and current prevalence of canine myocarditis agents are unknown except for areas with endemic Chagas disease or Leishmania infection. 64,103,127
Viral Agents
The viral agents tested for by the PCR panel were CPV-2, CRCoV, CDV, CAV, CPiV, influenza A, CHV-1, and WNV; most agents were occasionally detected in cases and controls.
The majority of reports of canine myocarditis describe CPV-2 myocarditis. Features were reviewed in our previous retrospective study, which demonstrated a significant association between CPV-2 and myocarditis/myocardial fibrosis in young dogs. 44 CPV-2 is cardiotropic and causes myocarditis in susceptible puppies but also contributes to canine myocarditis due to gastrointestinal (GI) barrier disruption, immunosuppression, and bacterial myocarditis. 28,53,123 In this study, CPV-2 was detected in several adult myocarditis cases; however, the viral quantity was low and not detected by ISH, and detection frequency was similar in adult controls. Considering the replication requirements of parvoviruses for mitotically active S-phase cells, these results suggest PCR detection of latent or subclinical infection. 92,93 Available data do not support CPV-2 as a primary cause of myocarditis in adult dogs. 81,87
The related CPV-1 (minute virus of canines) is a cause of neonatal enteritis with large intranuclear inclusions, lymphoid depletion, and less frequent focal to diffuse areas of myocardial necrosis with mineralization and giant cells. 51 CPV-1 was not detected in selected cases in this cohort by IHC and appears to be an infrequent cause of canine myocarditis in neonates only.
CDV-associated necrosis and calcification of the myocardium of suckling pups is described in textbooks. 60 Nevertheless, primary descriptions of myocardial degeneration, necrosis, mineralization, and CDV inclusions are limited to experimental studies using gnotobiotic neonatal Beagles inoculated with virulent CDV at 5 to 7 days but not in pups inoculated at 10 to 21 days of age. 55 A literature search did not identify reports of naturally occurring canine myocarditis with CDV as a single agent, which suggests the role of CDV in myocarditis is primarily as an immunosuppressive agent. 53,125
WNV was detected at the analytic limit in 1 adult case in our study. Myocarditis, along with meningoencephalitis and multifocal hemorrhage, is a prominent lesion in native and exotic birds infected with vector-borne West Nile virus; comparatively, myocardial involvement is rare in nonavian species. 114 Clinical WNV disease is rare in dogs, but necrotizing variably lymphocytic and neutrophilic myocarditis with vasculitis has been reported in dogs that also have WNV polioencephalomyelitis. 18,25,74
The test panel used here was informed in part by the comparative epidemiology of human myocarditis. In humans, the most frequent agents associated with myocarditis include adenovirus, herpesviruses (cytomegalovirus, Epstein-Barr virus, human herpesvirus 6, herpes simplex viruses 1 and 2), enterovirus, hepatitis C virus, influenza viruses A and B, parvovirus B19, and rhinovirus. 12,40,67,72,97 Other causes (including parainfluenza, group A Streptococci, and Mycoplasma spp) have rarely been detected via serology or culture in patients with clinical features of myocarditis. 63
CAV has not been reported as a cause of canine myocarditis. In our cohort, CAV was infrequently detected by PCR in cases and controls; however, adenoviral antigen was not detected by IHC. CAV-1 (infectious canine hepatitis) infects hepatocytes and endothelial cells. 32 Clinical disease is infrequent due to widespread vaccination and most often occurs in inappropriately vaccinated dogs under 1 year of age often in concert with stress, overcrowding, and/or coinfection. 118 CAV-2, the agent of infectious tracheobronchitis, causes either inapparent infection or respiratory disease often as a coinfection with a variety of viruses and bacteria, where dogs are cohoused. 117 Available data do not support a role for CAV in canine myocarditis.
CHV-1 infection is associated with fatal systemic disease in neonatal puppies (recently reviewed in Jager et al 56 ). In neonates, characteristic necrosis and hemorrhage can be observed in most organs; myocardial degeneration with or without necrosis can be a feature of systemic infection. 32,94 Latent infection is established in ganglia, which may remain subclinical or manifest as mild, recurrent genital, respiratory, or ocular disease. 32,56 Available data do not support a role for CHV-1 in myocarditis in adult dogs.
Acute myocarditis can be a complication of influenza virus infections in humans. Canine influenza virus infection can result in respiratory disease, most frequently in group-housed dogs. 99 CRCoV and CPiV both frequently contribute to canine respiratory disease with asymptomatic infection or mild respiratory illness; both viruses were detected in a single pediatric/juvenile myocarditis case. CPiV replicates in epithelial cells of the upper respiratory tract but is occasionally associated with viremia. 39 Strains of canine enteric CoV (distinct from CRCoV) have been associated with severe systemic disease, and highly pathogenic enteric CoV is an emerging cause of systemic vasculitis in dogs. 19,132 Available data do not support a role for influenza A, CPiV, or CRCoV in canine myocarditis.
Bacterial Agents
In our cohort, bacteria were identified in myocardial sections in 3 cases; unfortunately, bacterial culture was not routinely performed in cases. Bacteria, particularly Streptococcus, Staphylococcus, and Bartonella spp, frequently cause mural and valvular infective endocarditis, which has been reviewed by others. 69,78,119,120 Reported causes of bacterial myocarditis include Bartonella spp, 14,35,41,78,109 B. burdorferii, 33,57,73 C. koseri, 28 C. piliforme, 131 Rickettsia rickettsii, 104 Staphylococcus spp, 57 and anaerobic infection 45 .
In the dog and other species, C. piliforme can cause necrotizing enterocolitis, hepatic necrosis, and occasionally necrotizing myocarditis. 131 As C. piliforme is difficult to culture, diagnosis is based on observation of characteristic microscopic lesions in young animals with intracellular bacilli (demonstrated by silver histochemical stains) in cells at the periphery of areas of necrosis; serologic tests and PCR are also available.
The northeastern United States is a Lyme disease endemic area with high rates of B. burgdorferi infection in dogs. While infection is frequent, clinical disease associated with canine B. burgdorferi infection is infrequent and generally nonspecific, including fever, arthritis, anorexia, lymphadenopathy, glomerulonephritis, neurologic signs, and rarely myocarditis. Lyme carditis is rare in humans and usually presents as a self-limiting atrioventricular block during acute infection, although a broad range of cardiac involvement has been reported. 42,67 Lyme carditis may be a direct inflammatory reaction to spirochete infection or may be immune mediated, similar to Lyme nephritis. 20,36,42 There are intriguing studies associating B. burgdorferi with human dilated cardiomyopathy, but epidemiologic data are lacking. 66,95,113 The European Concerted Action on Lyme Borreliosis (EUCALB) has proposed a clinical case definition for diagnosis of Lyme carditis in humans, which includes acute-onset atrioventricular conduction or other rhythm disturbance, myocarditis or pancarditis, and the detection of specific serum antibodies, supported by the detection of Borrelia spp (culture and/or PCR) in endomyocardial biopsy. 112 Cardiologic manifestations are not well documented in dogs, and there are few reports associating Borrelia spp with canine myocarditis, usually via IHC, and no specific criterion for the diagnosis of cardiac borreliosis in dogs. 33,57,73,76 In dogs, descriptions vary from pyogranulomatous 33 to chronic lymphoplasmacytic and histiocytic myocarditis with fibrosis, 57,73 the latter similar to human Lyme carditis. 42 We did not detect Borrelia spp by PCR in our cohort of cases from a Lyme endemic area, but tissue detection in experimentally infected dogs can be sporadic. 2,116 These data indicate Borrelia spp are not frequently associated with lesions of canine myocarditis; however, the lack of detection by PCR does not rule out Lyme carditis in humans. Furthermore, the host-bacterium interactions responsible for manifestations of Lyme disease are not well defined.
In dogs, pyogranulomatous lymphoplasmacytic endocarditis and myocarditis are associated with infection with several species of the Bartonella genus. 14,35,41,78,105,109 In our cohort, we did not detect Bartonella spp by PCR in the hearts of cases or controls regardless of age; however, PCR was performed on nucleic acid from FFPE tissue, which has been demonstrated to affect the sensitivity of Bartonella detection. 35
M. haemocanis, the agent of canine hemotropic mycoplasmosis, was detected by rtPCR in occasional cases and controls. Hemotropic mycoplasma is transmitted primarily by tick bite, transfer of infected blood during aggressive contact, and possibly perinatally. 26,65,86 Clinical disease (ie, hemolytic anemia) is rare in nonsplenectomized, immunocompetent dogs. We attribute the detection of M. haemocanis secondary to immune changes related to systemic disease.
Protozoal Agents
Protozoal agents were not observed histologically in our cohort, but we detected N. caninum by PCR in both cases and controls. N. caninum infection is associated with progressive hind limb paresis, ataxia, and ascending paralysis most severe in congenitally infected puppies. Encephalitis, myositis, myocarditis, pneumonia, and dermatitis have also been reported. 6,58 Infection is often disseminated with Neospora zoites in multiple tissues, but lesions and organisms are occasionally confined to the heart. 90 The prevalence of N. caninum infection and frequency of subclinical infection in the canine population are unclear; seroprevalence has been reported in up to 37.8%, with higher prevalence in farm dogs in rural environments compared to urban settings (reviewed elsewhere). 10,36,75 Subclinically infected dams can transmit the organism transplacentally, and successive litters may be born infected. 36 Variable vertical transmission and subclinical infection have been suggested by serosurvey of breeding dogs: N. caninum seroreactivity was reported in up to 52% (32/61) of pups born to seropositive dams while 22 of 88 (25%) pups demonstrated clinical signs of neosporosis. 7 In our study, the clinical relevance of detection of N. caninum by PCR in the absence of Neospora zoites was difficult to determine; thus, subclinical infection and contamination must be considered. 126 In dogs, T. gondii myocarditis has been reported with viral immunosuppression. 53
T. cruzi, the hemoflagellate protozoan agent of Chagas disease (American trypanosomiasis), causes chronic pancarditis in dogs and humans. Chagas disease is limited geographically due to the range of the triatomine insect vector (reduviid or “kissing” bugs), which is present in the southern United States, Mexico, Central America, and South America. Seroprevalence, up to 23.6%, varies according to location, with higher seroprevalence in sporting and working breeds. 31,47,64 Intracellular amastigotes within pseudocysts are present primarily in cardiac muscle. Acute infection is associated with granulomatous and necrotizing myocarditis and numerous T. cruzi pseudocysts. Organisms are less frequent (or absent) with chronicity and are accompanied by lymphoplasmacytic infiltrate and fibrosis. 106,127 During acute infection, low serum antibodies may result in false-negative results via serum immunofluorescence testing. 127
The domestic dog is the intermediate host for 2 species of Hepatozoon, an arthropod-borne protozoal infection. Old World hepatozoonosis (Hepatozoon canis) occurs worldwide and has been reported in Asia, southern Europe, Africa, South America, and the southern United States. H. canis meronts, which have a unique wheel-spoke arrangement of micromerozoites, are reported to be very plentiful in the myocardium and other tissues of wild African carnivores. Conversely, they do not frequently involve the myocardium of South African domestic dogs and are not associated with myocardial inflammation. Dogs with severe infections can have multiorgan involvement with splenomegaly, hepatomegaly, and lymphadenomegaly. 4,5,48,83 New World hepatozoonosis is primarily found throughout the southeastern United States and Gulf coast of Texas. H. americanum parasites are frequently found in skeletal and cardiac muscle, which can cause pyogranulomatous myositis. Inflammation is scant around “onion skin” cysts or developing schizonts but robust in response to released merozoites with pyogranulomatous vasculitis and vascular mineralization. 3,9,48
Protozoa of the genus Leishmania cause disease in dogs in the Mediterranean region and parts of Africa, Asia, and the Americas. Lesions are most commonly found in the skin and hemolymphatic organs, but granulomatous and lymphoplasmacytic myocarditis with cardiomyocyte loss, vasculitis, fibrosis, and amastigotes within myocardial macrophages has also been reported in dogs. 103,121
Fungal Agents
None of the cases in our cohort had evidence of fungal myocarditis.
In endemic areas of the southwestern United States, C. immitis causes restrictive pericarditis, epicarditis, and myocarditis as part of disseminated coccidioidomycosis. 16,59,79,100,110 Most dogs present with pulmonary coccidioidomycosis following inhalation of infective arthrospores. Young, large breed, hunting, working, or sporting dogs are most at risk. Disseminated infection frequently involves bone, joint, and lymph node, but pericardial involvement can lead to heart failure, arrhythmia, syncope, and sudden death. 95 Pericardial infection can be diagnosed based on intralesional spherules in biopsy specimens from pericardectomy samples. 54 Intradermal coccidioidin test and serum coccidioidomycosis complement fixation tests are prone to false negatives. 100
Granulomatous myocarditis can be observed as a component of systemic mycotic infection with identification based on fungal histomorphology, culture, serology, and/or PCR. 17,21,61,101 Myocardial and great vessel involvement have been reported in cases of canine systemic blastomyocosis. 107
Algal Agents
Myocardial involvement is frequent in dogs with disseminated protothecosis. Prototheca zopfii and Prototheca wickerhamii have been associated with often abundant myocardial organisms. 115 None of the cases in our cohort had evidence of protothecosis.
Agents With Secondary Myocardial Effects
Several infections are associated with myocardial lesions, but available literature does not indicate direct cardiac involvement. Myocardial involvement is observed in dogs with systemic vasculitis due to R. rickettsii, possibly related to disseminated endothelial infection or secondary immune-mediated mononuclear inflammatory reaction. Myocardial vasculitis and hemorrhage can be accompanied by small vascular thrombi, focal ischemic necrosis, and left atrial and mitral valve endocarditis, but infection of cardiomyocytes has not been demonstrated. 104
Dogs with babesiosis and clinical anemia are reported to have cardiac arrhythmia and nonspecific cardiac lesions, including fibrin microthrombi, lymphoplasmacytic and variably neutrophilic infiltrates, and necrotic foci. 37 Furthermore, cTnI is elevated in dogs with immune-mediated hemolytic anemia or complicated infection, although infection of cardiomyocytes has not been reported. 77 Cardiac changes may the result of cardiac infection or secondary to clinicopathologic abnormalities caused by Babesia (eg, anemia, disseminated intravascular coagulation, electrolyte alterations, renal insufficiency). Similarly, cTnI elevations and cardiac arrhythmia reported with acute natural Ehrlichia canis infection were correlated with the severity of anemia and signs of systemic illness (ie, tachycardia, fever, leukocytosis); infection of cardiomyocytes has not been reported. 34 African horse sickness occasionally causes disease in dogs, usually associated with consumption of uncooked, infected horse meat. 89,122,124 Necropsy findings in dogs include respiratory (“dunkop”-like) lesions with immunoreactivity for African horse sickness primarily labeling pulmonary microvascular endothelial cells similar to infected horses. In the myocardium of infected horses, immunoreactivity is localized to endothelial cells, circulating leukocytes, and infrequently red blood cells but not cardiomyocytes; subendocardial myocardial hyaline degeneration and necrosis with possible myocardial IHC labeling have been reported in 1 dog. 30,124
Conclusion and Testing Recommendations
Our study demonstrates the challenges of determining the causes of canine myocarditis. A comprehensive PCR screen did not determine the cause of myocarditis in most canine cases in our study; furthermore, agents detected by PCR were often not considered to be causative. Regardless, our study and the data available to date inform some testing recommendations.
Although CPV-2 is well recognized as a cause of myocarditis in susceptible puppies, PCR detection was similar in adult cases and controls in our study and CPV-2 myocarditis has not been reported in adult dogs; thus, CPV-2 is not considered associated with myocarditis in adult dogs.
Many of the PCR-detected pathogens (CDV, CHV, and CAV) are associated with subclinical, latent, or otherwise inapparent infection, particularly in adult animals. As immunization with modified live vaccine (CDV, CPV-2, CAV-2, and CPiV) is recommended for dogs beginning as early as 6 weeks of age, PCR may be detecting vaccine virus and not field strains. The codetection of multiple agents infrequently associated with disease (due to widespread vaccination) suggests detection of vaccine virus. PCR detection at least represents evidence of exposure to an infectious agent; thus, ultimately, it is unclear if PCR-detected agents are cardiopathogenic, incidental, or latent as clinical recognition of myocarditis usually occurs as viral titers are waning.
Evidence indicating age restriction of some agents supports initial development of age-specific testing recommendations, primarily based on excluding tests for agents that have not been documented as a cause of myocarditis in an age group, although screening of larger cohorts of myocarditis cases is needed. Based on available data and pathogenesis of individual agents, we suggest testing to include CPV-2 and CHV in neonatal and pediatric/juvenile patients and CPV-1 and CDV only in cases of neonatal myocarditis. Data available to date do not support testing for CDV, CHV, or CPV in cases of adult myocarditis. 81 WNV and B. burgdorferi appear to be infrequent causes of myocarditis, and the decision to test should be made on a case-by-case basis. Despite the prevalence in human myocarditis, it does not appear that CAV or influenza is associated with canine myocarditis. Bacterial culture (augmented by special histochemical stains) and virus isolation are recommended for all myocarditis cases as unrecognized/novel pathogens may be identified. Fungal, protozoal, and algal pathogens can often be detected via histologic examination, although the number of organisms can vary according to chronicity and previous treatment. The occurrence and incidence of immune-mediated myocarditis, a diagnosis made by exclusion in humans based on IHC criteria in the absence of PCR pathogen detection, are unknown in the dog.
In humans, endomyocardial biopsy paired with molecular testing (PCR for viral genomes and leukocyte IHC) is the gold standard for diagnosis. Unfortunately, ancillary testing to identify microbial agents of canine myocarditis is usually not elected because screening for all of the reported causes of myocarditis is cost prohibitive; furthermore, when testing is performed, it is after death and does not always reveal an underlying cause. 29,62,129 Current diagnostic modalities are insufficient to identify the causative agents in most cases. There is a need to identify agents and establish criteria for the diagnosis of immune-mediated myocarditis, particularly in adult dogs. New approaches are needed to identify emerging, unrecognized, or novel pathogens. This study establishes groundwork to stimulate the development of novel testing strategies for the diagnosis and thus investigation of the etiopathogenesis of canine myocarditis.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819839241 - The Causes of Canine Myocarditis and Myocardial Fibrosis Are Elusive by Targeted Molecular Testing: Retrospective Analysis and Literature Review
Supplemental Material, DS1_VET_10.1177_0300985819839241 for The Causes of Canine Myocarditis and Myocardial Fibrosis Are Elusive by Targeted Molecular Testing: Retrospective Analysis and Literature Review by Alex Molesan, Laura Goodman, Jordan Ford, Samantha J. Lovering and Kathleen Kelly in Veterinary Pathology
Footnotes
Acknowledgements
We are grateful for the expert technical assistance of Renee Anderson, Brittany Chilson, Melissa Laverack, and the Animal Health Diagnostic Center Histology Laboratory staff for technical support. We thank Edward Dubovi for manuscript review and the Cornell University library for assistance obtaining primary source material. Student financial support was provided by NIH Training Grant T35OD010941 and the AVMA-AVMF Second Opportunity Research Scholarship.
Declaration of Conflicting Interests
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This data was presented at the American College of Veterinary Internal Medicine Forum 2019.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
