Abstract
Chronic kidney disease is common in geriatric cats, but most cases have nonspecific renal lesions, and few studies have correlated these lesions with clinicopathological markers of renal dysfunction. The aim of this study was to identify the lesions best correlated with renal function and likely mediators of disease progression in cats with chronic kidney disease. Cats were recruited through 2 first-opinion practices between 1992 and 2010. When postmortem examinations were authorized, renal tissues were preserved in formalin. Sections were evaluated by a pathologist masked to all clinicopathological data. They were scored semiquantitatively for the severity of glomerulosclerosis, interstitial inflammation, and fibrosis. Glomerular volume was measured using image analysis; the percentage of glomeruli that were obsolescent was recorded. Sections were assessed for hyperplastic arteriolosclerosis and tubular mineralization. Kidneys from 80 cats with plasma biochemical data from the last 2 months of life were included in the study. Multivariable linear regression (
Chronic kidney disease (CKD) is common in geriatric cats, affecting up to 49% of cats older than 15 years, 23 but most cases have nonspecific renal lesions. In a postmortem study of 64 cats with CKD, 8 specific renal diseases, including neoplasia, chronic pyelonephritis, polycystic kidney disease, and amyloidosis, were identified in approximately half the cats. In another study of renal biopsy specimens from 47 azotemic cats, 19 approximately one-third had specific renal diseases. Reports of feline chronic renal disease have been mainly descriptive. A 1968 publication 16 described in detail the histologic features of feline CKD, which included interstitial fibrosis and inflammation, tubular atrophy, glomerulosclerosis, and arteriolosclerosis.
Although the lesions in feline CKD have been well-described, they have generally not been quantified, and few studies have attempted to relate these lesions to clinicopathological data. The first aim of this study was to improve understanding of the pathogenesis of feline CKD by quantifying renal lesions. The second aim was to determine which lesions were correlated with the severity of azotemia and clinicopathological abnormalities predicting progression of azotemia, such as hyperphosphatemia, anemia, or proteinuria. 7
Materials and Methods
Cats with CKD and geriatric cats (>9 years of age) with nonrenal diseases were recruited as part of a routine geriatric health screening program. Clinics were run between 1992 and 2010 at 2 London first-opinion practices: the People’s Dispensary for Sick Animals, Canning Town/Bow, and the Beaumont Sainsbury Animal’s Hospital, Camden. CKD was diagnosed only in cats with plasma creatinine concentration >2.0 mg/dl and with history and clinical signs compatible with chronic, rather than acute, kidney disease. Postmortem examinations were offered to all clients having cats euthanized at the clinics. Only cats with postmortem data and a plasma biochemical profile during the last 2 months of life were included in the study. Cases with a clinical diagnosis of CKD were staged using modified International Renal Interest Society (IRIS) classifications according to the last plasma creatinine concentration (2.0–2.8 mg/dl for stage 2, 2.9–5.0 mg/dl for stage 3, and >5.0 mg/dl for stage 4). Azotemic cats were also classified according to urine protein to creatinine ratio (UPC) for IRIS proteinuria status (<0.2, nonproteinuric; 0.2–0.4, borderline proteinuric; and >0.4, proteinuric). Azotemic cats with concentrated urine (urine-specific gravity >1.035) were excluded from the study because these cats may have had prerenal azotemia, in which case plasma creatinine concentration would not be expected to reflect renal lesions. All nonazotemic cats were grouped for descriptive purposes.
Clinicopathological Data
Urine samples were collected by cystocentesis; UPC from cats with bacteriuria or gross hematuria were excluded. Blood samples were obtained by jugular venipuncture in heparinized tubes. Spun urine supernatant and plasma were stored at –80°C until analysis. UPC and plasma biochemical data were obtained from Idexx Laboratories (Wetherby, UK). Urine protein was measured using a colorimetric pyrogallol red method; creatinine was measured using a colorimetric picric acid method. Systolic arterial blood pressure was measured using the Doppler method and the mean of 5 readings calculated for each visit. Fundic examinations were undertaken on all cats with mean systolic blood pressure >160 mm Hg. Hypertension was diagnosed if blood pressure measurements exceeded 170 mm Hg on 2 consecutive visits or on 1 visit in association with hypertensive choroidoretinopathy. All hypertensive cats were started on amlodipine therapy at a daily dose of 0.625 mg, which was increased to 1.25 mg (or exceptionally up to 2.5 mg) if blood pressure remained >160 mm Hg. Fundic examinations were not routinely undertaken after commencement of amlodipine therapy. Time-averaged systolic blood pressure (SBPOT) was calculated by plotting systolic blood pressure against time, then dividing the area under the curve by the interval between the first and last blood pressure measurements. The reported method 14 was modified to include pretreatment blood pressure measurements in the calculations. Although all other data were obtained shortly before death, SBPOT (rather than the last available blood pressure measurement) was used in analyses because a single blood pressure value is more likely to be affected by “white-coat” hypertension or hypovolemia. All azotemic cats were offered a commercial renal diet as standard care. In normotensive cats, this was started immediately after diagnosis, and in hypertensive cats, after the blood pressure had been stabilized with amlodipine. Other treatments were dispensed according to the needs of the individual cat. Plasma total thyroxine concentration (TT4) was measured in cases in which hyperthyroidism was suspected based on history and clinical signs; cats with TT4 >55 nmol/l were diagnosed with the condition. Cats with uncontrolled hyperthyroidism (TT4 >40 nmol/l) at the last sampling were excluded from the study.
Histopathological Data
At necropsy, each kidney was transected longitudinally and transversely and then fixed in 10% neutral-buffered formalin. Fixed tissue, selected randomly from either the right or left kidney, was trimmed for histologic processing to include the cortex and medulla in the paraffin block. For each case, 2 transverse sections, 3 μm thick, were evaluated by a renal pathologist (C.A.B.) masked to all clinicopathological data. One section, stained with hematoxylin and eosin (HE), was used for histologic evaluation; the second section, stained with hematoxylin and periodic acid-Schiff (PASH), was used for morphometric analyses. Electron microscopy was used as necessary to evaluate glomeruli for lesions of glomerulonephritis. Four histologic parameters were assessed: cortical interstitial fibrosis, cortical interstitial inflammation, glomerulosclerosis, and glomerular volume. Tissues were also evaluated for vascular abnormalities that have been associated with hypertension (hyperplastic arteriolosclerosis, hyaline arteriolosclerosis, and fibrinoid vascular necrosis). Hyperplastic arteriolosclerosis was defined as arteriolar concentric medial smooth muscle hyperplasia and basement membrane duplication (Fig. 1). The accumulation of hyaline material in the vascular wall without necrosis (hyaline arteriolosclerosis), or with evidence of necrosis (fibrinoid vascular necrosis), was also assessed. One additional section for each case was evaluated with Von Kossa’s stain (by S.C.) to assess tubular mineralization, defined as mineralization of the tubular basement membranes and/or tubular epithelial cells (Fig. 2). Tissues were scored for interstitial fibrosis and interstitial inflammation on a scale of 0 to 3 with 0.5 intervals as follows: 0 = no fibrosis/inflammation or rare small foci, 1 = mild or scattered multifocal areas of fibrosis/inflammation affecting <5% of the section, 2 = moderate fibrosis/inflammation affecting 25% to 50% of the section, 3 = diffuse or coalescing fibrosis/inflammation (Fig. 3).
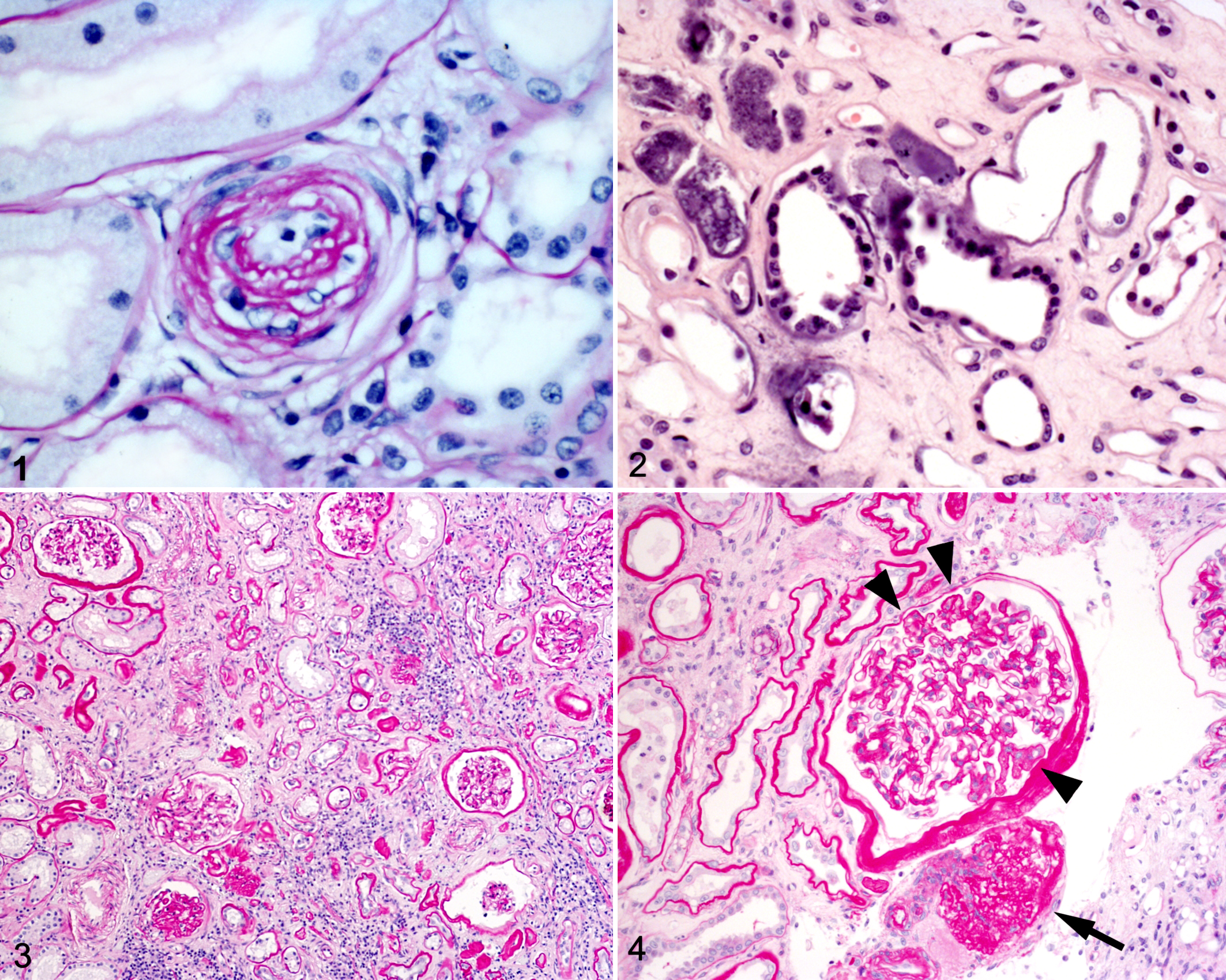
Glomerulosclerosis was evaluated by scoring 25 glomeruli for the degree of mesangial matrix expansion and deriving the mean score, as described. 11 These scores were on a scale of 0 to 3 (only integers were used) as follows: 0 = normal (matrix could encircle no more than 1 nucleus), 1 = mild (matrix could surround several nuclei but not extend to peripheral capillary loops), 2 = moderate (matrix expansion involving peripheral capillary loops and affecting <50% of the glomerulus), and 3 = severe (matrix expansion affecting >50% of the glomerulus). In addition, an obsolescence index (%) was determined by counting all glomeruli in two 70-mm2 fields and recording the number that was obsolescent (global matrix expansion with loss of capillary lumina; Fig. 4). Obsolescent glomeruli were assigned a score of 4, and the obsolescence index was combined with the mean glomerular score using the following formula: combined glomerular score = 0.04 × obsolescence index + mean glomerular score × (1 – obsolescence index/100). The cross-sectional area of 20 randomly selected nonobsolescent outer cortical glomerular capillary tufts was measured using Image Pro Plus 4.1. software as described. 6 Glomerular volumes were calculated using the formula V = βκ(area)3/2, where β = 1.38 (the shape coefficient for spheres), κ = 1.1 (the size distribution coefficient for glomerular profiles), and “area” is the mean glomerular profile planar area.
Statistics
Associations between histologic variables were assessed using Spearman’s rank correlation coefficient, the Mann Whitney
Results
Renal histologic data were obtained from 80 cats: 75 nonpedigreed domestic cats, 2 Burmese, 2 Persian, and 1 Siamese. Fifty cats were male (all but 2 had been neutered); 28 were neutered female cats. Eighteen (23%) of the cats had a history of hyperthyroidism. Thirteen (16%) of the cats had renal lesions of a specific disease (Table 1). The most common of these was lymphoma, affecting 5 male, nonpedigreed domestic cats, aged 15 (12, 16) years at death. Of the 5 cats with lymphoma, 2 reached IRIS stage 4, whereas 1 was in stage 3 and the other 2 were nonazotemic at the last blood sample. Only 1 cat (IRIS stage 4) was recorded as having palpably enlarged kidneys, but both the nonazotemic cats had evidence of lymphoma in other organs. No cat was treated for lymphoma. A cat with papillary necrosis (case 1) had been treated with meloxicam for 55 days prior to death, but the azotemia was stable and preceded meloxicam therapy. A cat with renal dysplasia (case 2) had a littermate (excluded from the study) with the same condition. Of the cats with azotemic CKD, those with specific renal diseases died at 11 (6, 15) years of age; those with nonspecific renal lesions died at 16 (13, 18) years of age. The nonazotemic cats died at 15 (14, 19) years of age. Nine nonazotemic cats were hypertensive (reflecting active recruitment of hypertensive cats to the clinics), 9 were normotensive, and 2 had unknown blood pressure. Palpable renal abnormalities (renomegaly in 2 cats, asymmetry in 1, and small kidneys in 6) were noted in the clinical records of 5 normotensive and 4 hypertensive nonazotemic cats. None of the cats with clinically palpable renal abnormalities had a specific renal disease diagnosed histologically. The last blood sample for cats in the study was taken 21 (3, 36) days before death and the last urine sample 7 (0, 34) days before death.
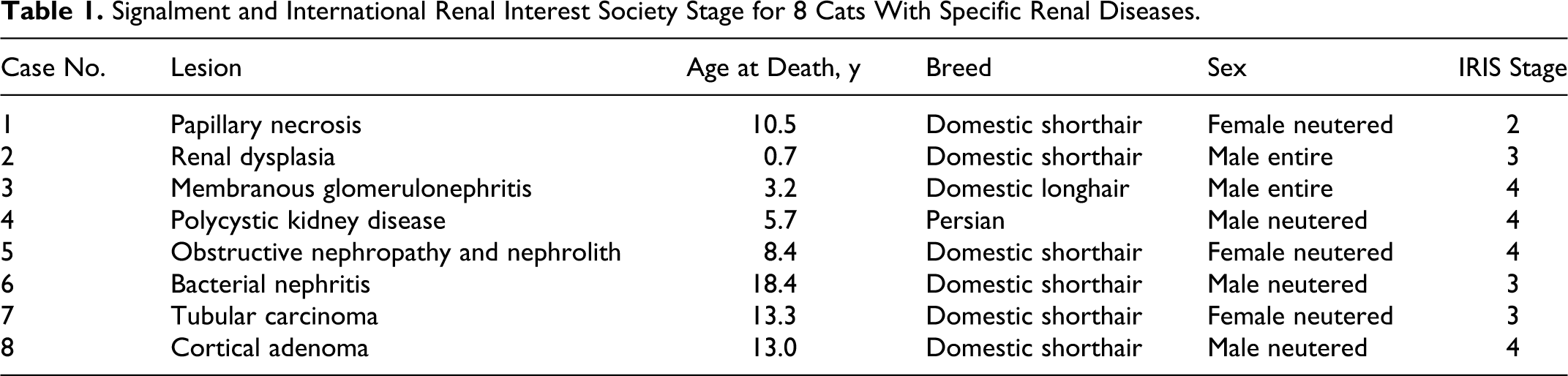
Signalment and International Renal Interest Society Stage for 8 Cats With Specific Renal Diseases.
The cortical interstitial fibrosis score was moderately correlated with the cortical interstitial inflammation score (
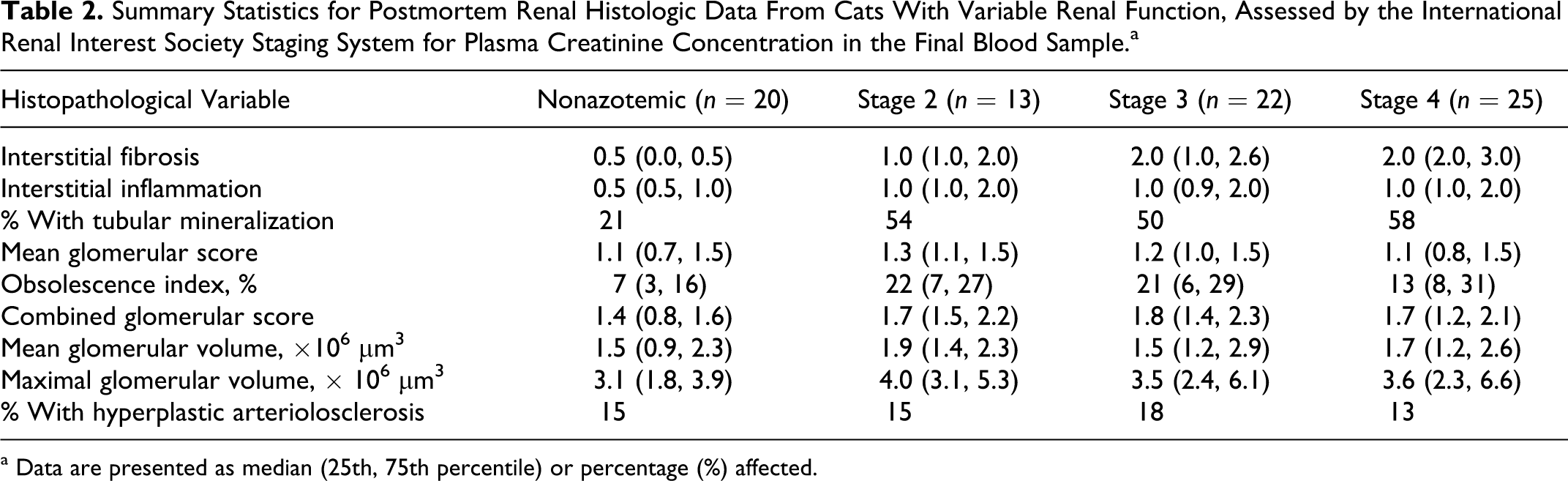
Twenty (25%) cats were nonazotemic at the last blood sample, 13 (16%) were in IRIS stage 2, 22 (28%) were in stage 3, and 25 (31%) were in stage 4. The histologic data for cats in each IRIS stage are in Table 2; the univariate analysis of lesions associated with final clinicopathological data is in Table 3. The multivariable model for lesions associated with plasma creatinine concentration contained only the interstitial fibrosis score (
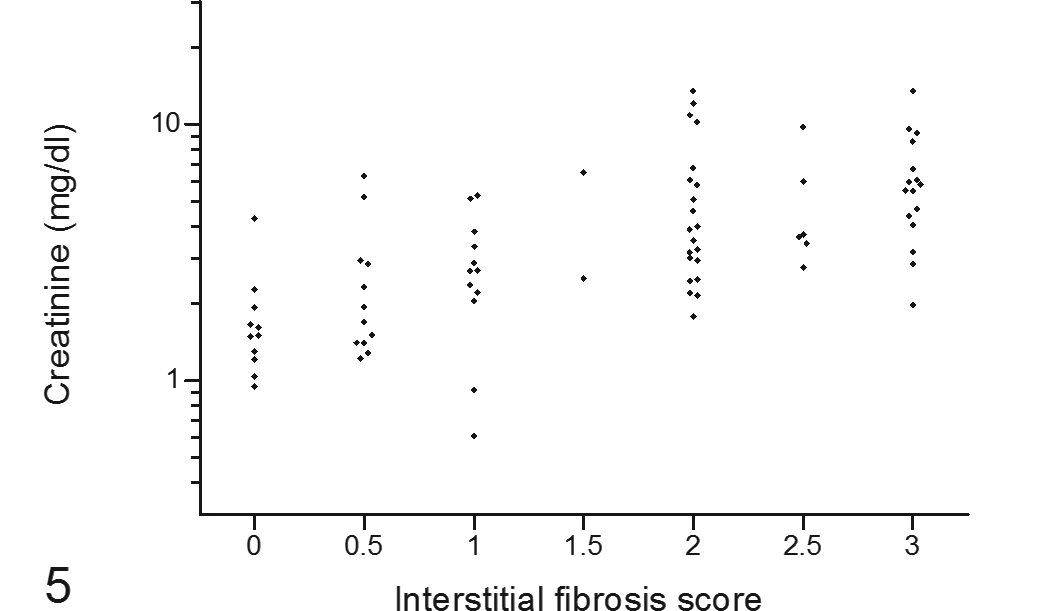
Positive correlation of plasma creatinine concentration from the final blood sample with postmortem renal interstitial fibrosis score in cats with variable renal function.
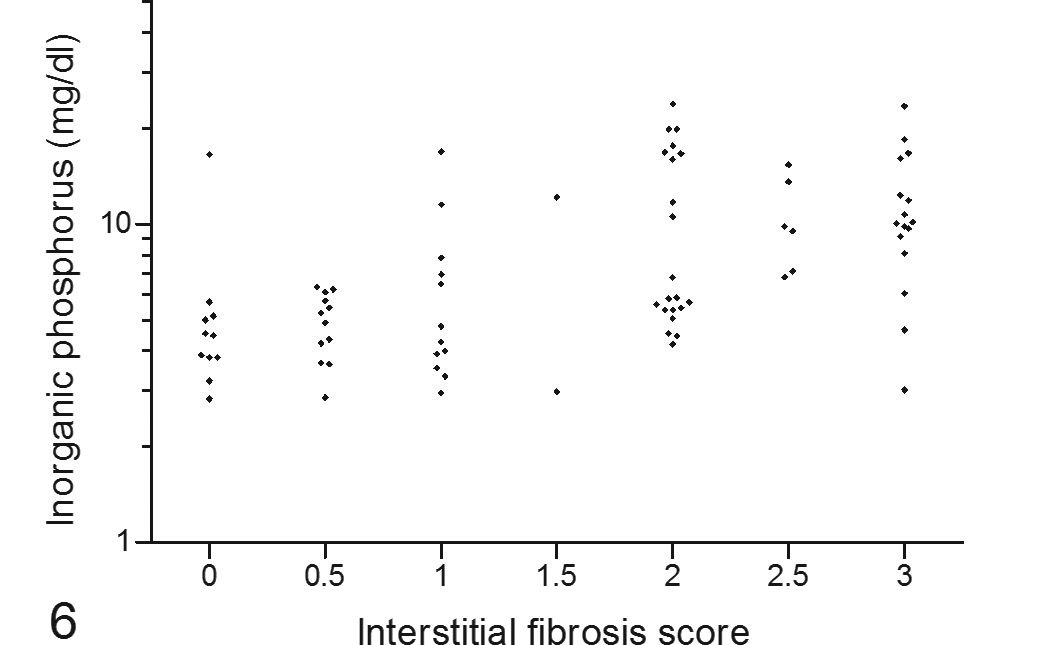
Positive correlation of plasma inorganic phosphorus concentration from the final blood sample with postmortem renal interstitial fibrosis score in cats with variable renal function.
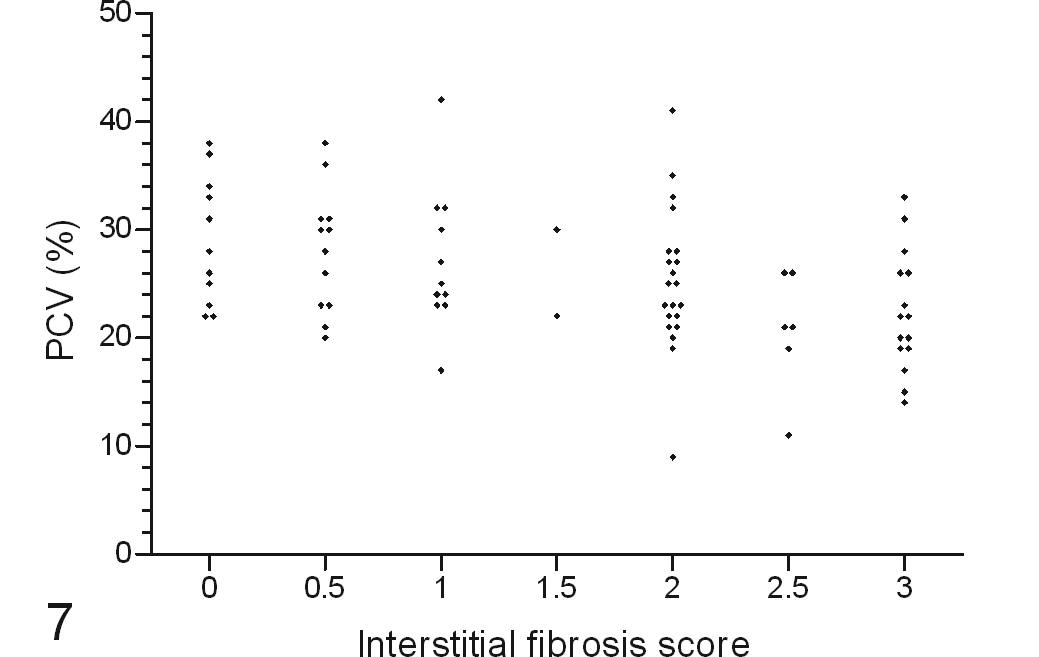
Inverse correlation of final packed cell volume with postmortem renal interstitial fibrosis score in cats with variable renal function.
Summary Statistics for Postmortem Renal Histologic Data From Cats With Variable Renal Function, Assessed by the International Renal Interest Society Staging System for Plasma Creatinine Concentration in the Final Blood Sample.a
a Data are presented as median (25th, 75th percentile) or percentage (%) affected.
Univariate Analysis of Association of Renal Histologic Variables With Packed Cell Volume (PCV), Time-Averaged Systolic Blood Pressure (SBPOT), Log10-Transformed Urine Protein to Creatinine Ratio (UPC), Plasma Creatinine, and Inorganic Phosphorus Concentration in Cats.a
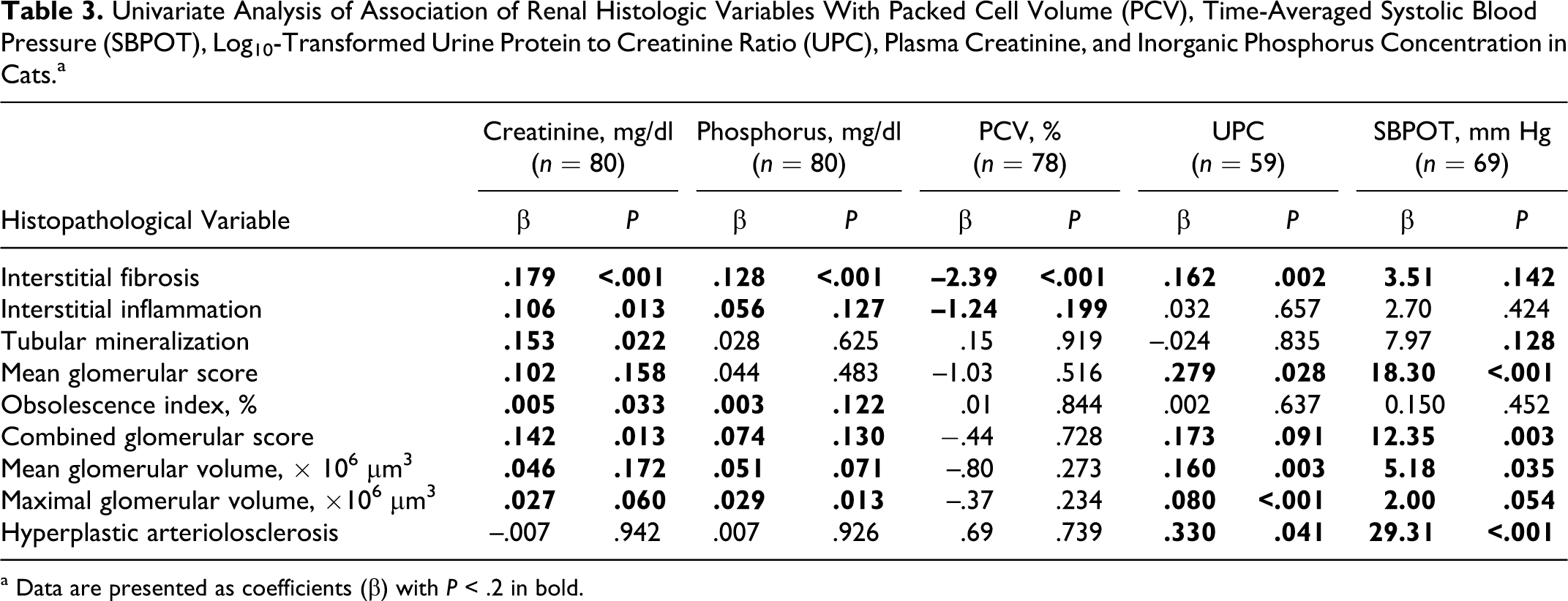
a Data are presented as coefficients (β) with
Of the 59 cases with suitable urine samples, 45 had azotemic CKD; 6 were nonproteinuric, 5 were borderline proteinuric, and 34 were proteinuric. The variables remaining in the multivariable model predicting proteinuria (adjusted
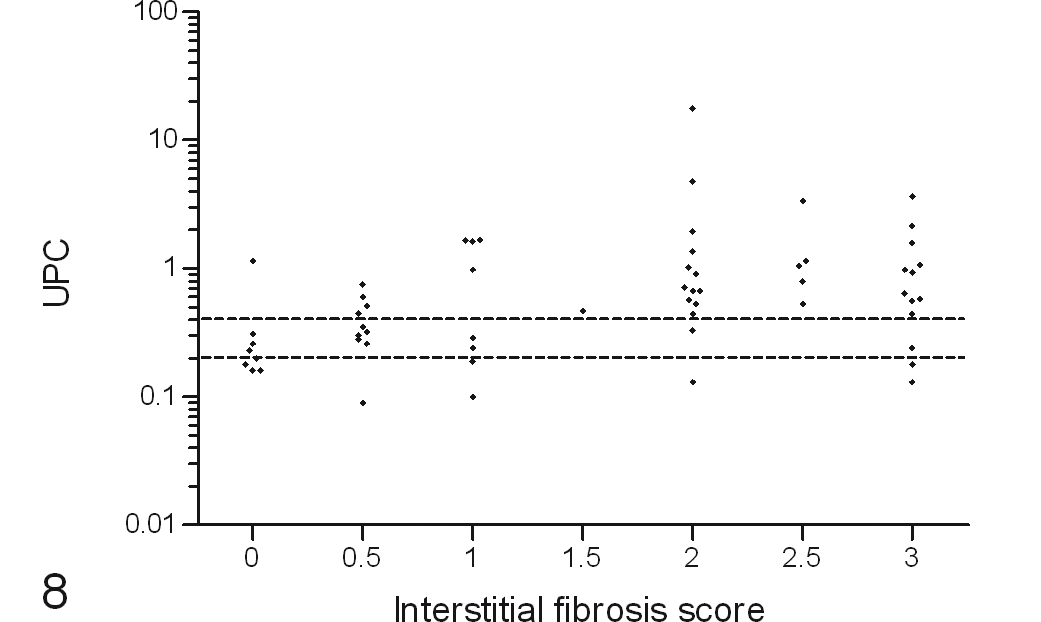
Positive correlation of urine protein to creatinine ratio (UPC) from the final urine sample with postmortem renal interstitial fibrosis score in cats with variable renal function. The dotted lines indicate the boundaries for International Renal Interest Society borderline proteinuria status; <0.2 nonproteinuric, 0.2 to 0.4 borderline proteinuric, and >0.4 proteinuric.
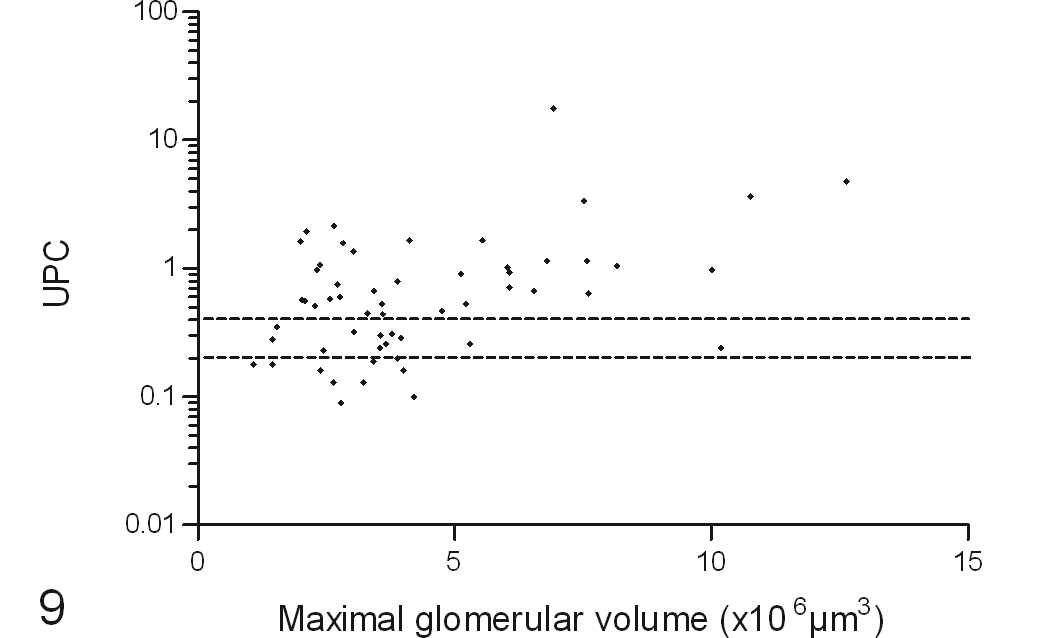
Positive correlation of urine protein to creatinine ratio (UPC) from the final urine sample with postmortem maximal glomerular volume in cats with variable renal function. The dotted lines indicate the boundaries for International Renal Interest Society borderline proteinuria status; <0.2 nonproteinuric, 0.2 to 0.4 borderline proteinuric, and >0.4 proteinuric.
Blood pressure data were available for 69 cases, including 35 normotensive and 34 hypertensive cats, with SBPOT of 136 (119, 146) mm Hg and 159 (144, 168) mm Hg, respectively. The hypertensive cats had received amlodipine therapy for 284 (138, 574) days before death. Vascular lesions in arterioles (<100 μm in diameter) consisted almost exclusively of hyperplastic arteriolosclerosis. One cat had hyaline arteriolosclerosis in addition to hyperplastic arteriolosclerosis, but vascular fibrinoid necrosis was not observed in any cat. In the multivariable model (adjusted
The hypertensive cats with hyperplastic arteriolosclerosis had systolic blood pressure of 211 (198, 226) mm Hg before treatment, received amlodipine therapy for 307 (42, 576) days, and had SBPOT of 171 (160, 178) mm Hg. In contrast, the hypertensive cats without such vascular lesions had systolic blood pressure of 194 (179, 209) mm Hg before treatment, received amlodipine therapy for 284 (171, 647) days, and had SBPOT of 152 (142, 159) mm Hg. Only 1 normotensive cat had hyperplastic arteriolosclerosis; this cat had SBPOT of 146 mm Hg, based on measurements spanning the last 1085 days before death. Hyperplastic arteriolosclerosis was detected in 32% (7/22) of the hypertensive cats with retinal lesions and 25% (3/12) of those with without retinal lesions documented. Figure 10 illustrates the difference in SBPOT between cats with and without hyperplastic arteriolosclerosis. Figure 11 shows the relationship between mean glomerular score and SBPOT.
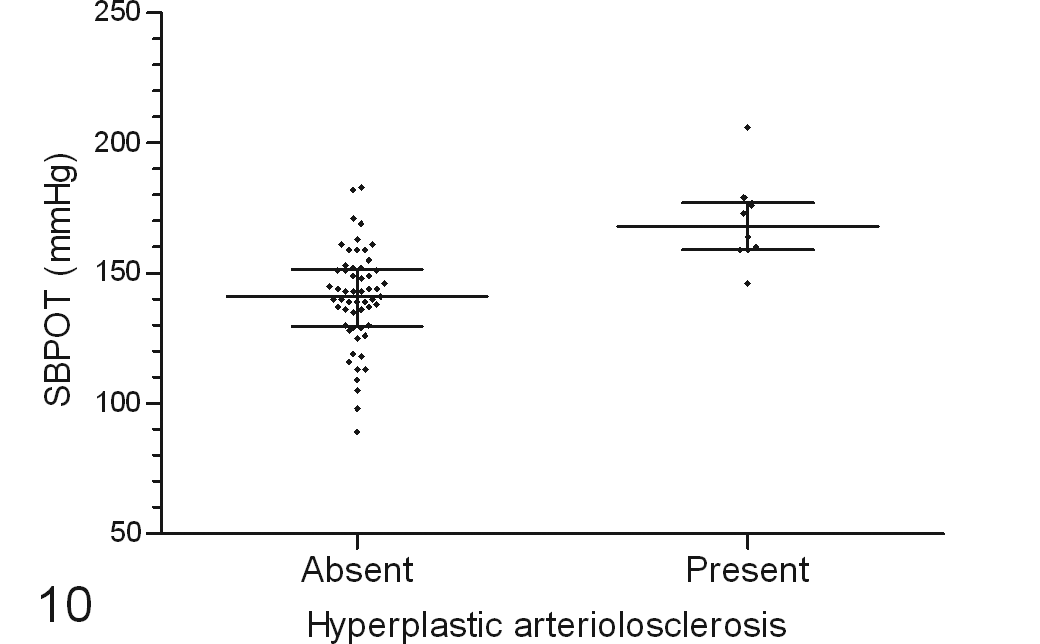
Association of postmortem renal hyperplastic arteriolosclerosis with higher time-averaged systolic blood pressure (SBPOT). The horizontal lines indicate median values and interquartile ranges.
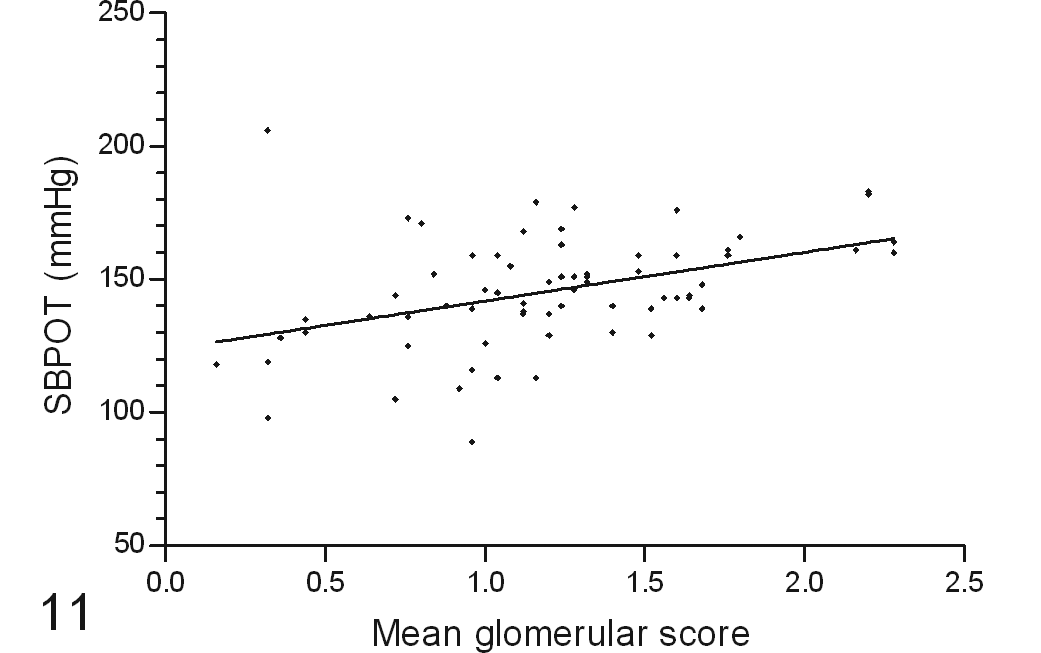
Positive correlation of time-averaged systolic blood pressure (SBPOT) with mean postmortem glomerulosclerosis score in cats with variable renal function. The line of best-fit is drawn on the graph (
Discussion
Most cats with CKD had tubulointerstitial disease, and primary glomerular disease was detected in only 1 cat. Lesions of glomerulosclerosis were relatively mild and overshadowed by tubulointerstitial lesions. Compared with previous studies, 8 specific renal diseases were identified in only a small proportion of cats (18% of the azotemic cats). This might reflect the chronicity of renal disease in the study population. Notably, the cats with specific renal diseases in this study were younger at death than the cats with nonspecific renal lesions. Previous studies have also found cats with nonspecific renal lesions to be older,15,19 suggesting these lesions might represent a disease process that progresses more slowly. Chronic inflammation is often implicated in the development of fibrosis via mechanisms such as macrophage activation, so the correlation between interstitial inflammation and fibrosis scores is noteworthy.
In 2 studies with fewer cats, interstitial fibrosis, but not glomerular lesions, correlated with renal function.25,27 In the present study, most histomorphometric variables were associated with renal function, but interstitial fibrosis was the strongest predictor of plasma creatinine concentration. In human CKD, the severity of tubulointerstitial lesions is better correlated with renal function than that of glomerular lesions. This is true even in patients with primary glomerular diseases.1,9,26 Interstitial fibrosis in human renal biopsy specimens indicates a poor prognosis 10 and is believed to represent the common final pathway of CKD progression.
Because of the limitations of creatinine as a precise marker of renal function, data were also described based on IRIS staging for CKD in which bands of plasma creatinine are used to categorize the stage of CKD. Fibrosis was the only lesion for which scores increased with IRIS stage (Table 2), reinforcing the conclusion that the degree of fibrosis best predicts the functional consequences of renal disease. The nonazotemic cats were grouped together in Table 2. It might have been possible to define some of these cats as IRIS CKD stage 1 based on clinical findings suggestive of CKD (such as palpably abnormal kidneys). However, the requisite renal imaging was not practical for nonazotemic cats in the primary care practice setting in which the cases were recruited.
Hyperphosphatemia was also associated with greater interstitial fibrosis in the present study. Plasma phosphate concentration increases in renal disease because renal clearance of phosphate is a function of glomerular filtration rate (GFR). Hyperphosphatemia could thus be a marker of reduced renal function in cats with greater interstitial fibrosis. In cats with surgically reduced renal mass, those fed a high-phosphorus diet had greater renal mineralization, fibrosis, and inflammation than those fed a low-phosphorus diet. 22 These structural abnormalities were accompanied by higher serum phosphorus and parathyroid hormone concentrations. Although parathyroid hormone concentrations were not measured in the present study, the plasma phosphate concentration from the last blood sample was not associated with tubular mineralization. This suggests the basis of the association between plasma phosphate concentration and interstitial fibrosis does not involve tissue mineralization.
Anemia may contribute to interstitial fibrosis by exacerbating renal hypoxia. 20 However, there are alternative explanations for the observed relationship between PCV and renal fibrosis. Myofibroblasts secrete extracellular matrix and are abundant in the interstitium of fibrotic kidneys. They might originate from resident peritubular fibroblasts or through the process of epithelial-mesenchymal transition. Erythropoietin, produced in peritubular fibroblasts,4,21 regulates erythropoiesis and inhibits apoptosis. 24 Myofibroblasts, in contrast, produce minimal erythropoietin. 3 Relative erythropoietin deficiency is believed to be the main cause of anemia in human patients with CKD, 17 and conversion of fibroblasts to myofibroblasts during active fibrosis could be responsible for this deficiency. 3 Further studies might determine whether the presence of renal interstitial myofibroblasts is related to the severity of anemia.
Glomerular hypertrophy is an adaptive response to the loss of nephrons and has been documented in cats with experimental partial renal ablation2,6 but has not been measured in naturally occurring feline CKD. Cats with stage 2 CKD had greater glomerular volume than normal cats, but subsequently, glomerular volume was closer to normal. Once mesangial matrix expansion reaches a certain threshold, glomerular volume begins to diminish as the capillary lumen is lost. The lower glomerular volume in late CKD could be explained if glomerular hypertrophy eventually becomes maladaptive and leads to sclerosis and obsolescence. However, the severity of glomerulosclerosis was mild in cats with all stages of CKD. This suggests an alternative mechanism, such as ischemia, leads directly to obsolescence.
Higher UPC was associated with increased glomerular volume in the present study. The increase in glomerular volume can be attributed mainly to greater capillary loop volume because the severity of glomerulosclerosis (mesangial matrix expansion) did not remain in the multivariable model predicting UPC. With glomerular hypertrophy, podocytes must cover a larger surface area, which might cause structural changes that compromise the glomerular filtration barrier. In a study using Munich-Wistar-Frömter rats, glomerular hypertrophy preceded albuminuria. 13 The albuminuria in that model was associated with effacement of podocyte foot processes, increased desmin, and reduced podoplanin expression in podocytes. Furthermore, a rat model of adriamycin-induced nephropathy showed that glomerular hypertrophy was associated with detachment of podocytes and proteinuria. 18 In another study, markers of podocyte injury in cats with CKD were not associated with the degree of renal dysfunction. 12 Podocyte loss can lead to adhesions of the capillary tuft to Bowman’s capsule, but such adhesions were rare in the present study population.
Proteinuria could lead to progression of kidney disease via overload of proximal tubular epithelial cells and release of cytokines. The severity of proteinuria is associated with tubulointerstitial changes in some human diseases such as lupus nephritis. 9 Proteinuria was not associated with interstitial inflammation in the present study but was associated with greater interstitial fibrosis independently of glomerular size. Interstitial fibrosis represents loss of tubules, and if a disease process is causing tubular loss, the same process might cause malfunction of remaining tubules. Filtered proteins are normally reabsorbed in the proximal tubules. Therefore, proteinuria could be a marker of tubular dysfunction concurrent with interstitial fibrosis. Alternatively, proteinuria might cause interstitial fibrosis. A third possibility is that loss of tubules and nephrons could result in glomerular hypertension and hence an increase in filtration pressure, which contributes to proteinuria. Glomerular hypertension might also lead to glomerular hypertrophy, further exacerbating proteinuria.
In the multivariable model, cats with higher SBPOT had greater glomerulosclerosis, and approximately one-third of the hypertensive cats had hyperplastic arteriolosclerosis. In human patients, 2 key mechanisms are believed to mediate hypertensive nephrosclerosis. Direct transmission of hypertension to the glomerulus and loss of renal autoregulation could cause glomerular capillary damage. 5 In addition, arteriolosclerosis could lead to renal ischemia and subsequent tubulointerstitial disease. 20 SBPOT was better correlated with the mean glomerular score than with the combined glomerular score. This indicates hypertension was associated with mild glomerulosclerosis, characterized by mild mesangial expansion with only rare involvement of peripheral capillary loops but not obsolescence. SBPOT was not associated with interstitial fibrosis or inflammation in the multivariable model, suggesting systemic hypertension was not a primary progression factor in the study cats. The treatment of the hypertensive cats with amlodipine must be considered as a confounding factor; if the cats had remained untreated, hypertension might have led to more severe lesions. 14
A limitation of the present study was that renal function data were based on test results that antedated tissue collection for histologic evaluation by up to 2 months. Although simultaneous collection of renal tissue and blood samples would have facilitated the correlation of histologic and functional parameters, renal biopsy is not standard clinical practice for cats with CKD because it is unlikely (except in cases of protein-losing nephropathy or renomegaly) to yield information that would change the case management. Nevertheless, the availability of whole kidneys, rather than biopsy specimens, for histologic sections strengthens the study. Although blood was not collected at euthanasia, the exclusion of cats with no blood sample in the last 2 months of life lessened the probability of major renal functional changes in the interval between blood and tissue collection.
Although the degree of azotemia correlates with renal function, it can be confounded, especially by muscle wastage because creatinine is derived from the breakdown of creatine phosphate in muscle. Muscle wastage is common in anorexic end-stage CKD patients and in the general geriatric feline population due to other conditions, such as osteoarthritis. Direct measurement of GFR is the gold standard for renal function, but current methods are impractical for routine clinical use.
In conclusion, cortical interstitial fibrosis is the renal lesion best correlated with the severity of azotemia, hyperphosphatemia, and anemia in cats with CKD. Proteinuria is associated with both interstitial fibrosis and glomerular hypertrophy; systolic hypertension is associated with glomerulosclerosis and renal vascular lesions.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Vetoquinol as part of a PhD studentship.
