Abstract
Forty cases of equine penile disease were screened with polymerase chain reaction for the presence of papillomaviral DNA. Cases consisted of 20 squamous cell carcinomas (average age of horse, 23.9 years) and 20 non–squamous cell carcinoma diseases (average age of horse, 13.3 years). All horses but one originated from the Northeastern United States. Breeds were not recorded. As based on MY09/MY11 consensus primers, DNA sequences from equine papillomavirus type 2 were amplified from 9 of 20 horses (45%) with penile squamous cell carcinoma and only 1 of 20 horses (5%) with non–squamous cell carcinoma penile disease. Equine papillomavirus type 2 DNA was the only papillomaviral DNA amplified from any of the 40 horses. Tissues from the 10 horses in which papillomaviral DNA was detected by polymerase chain reaction were also screened with in situ hybridization and immunohistochemistry. The presence of papillomavirus was demonstrated in a subset of these by in situ hybridization (6 of 10) and immunohistochemistry (1 of 10). This report describes a possible association between equine penile squamous cell carcinomas and equine papillomavirus type 2. This study is also the first report of equine papillomavirus type 2 infection in North American horses.
Keywords
Squamous cell carcinomas (SCCs) constitute approximately 18% of all equine cutaneous neoplasms. 23 Of these, 45% are reported to involve the male external genitalia, 22 and SCCs are the most common neoplasm in this location. 24 Equine penile SCCs (PSCCs) commonly recur after treatment, and the long-term prognosis is poor.5,12 Whereas ultraviolet light is an accepted cause of SCC in sun-exposed skin in some species, ultraviolet overexposure may be a less likely cause of equine PSCCs because of the ventral location of the male genitalia. Although equine PSCCs have been associated with chronic inflammation and smegma accumulation, the role of these factors in neoplasm development is unclear.8,25
Approximately 5% of all human cancers and 50% of human PSCCs are caused by papillomavirus (PV) infection.2,16 The aim of this study is to investigate whether equine PSCCs are also associated with PV infection. The rate of detection of PVs in equine PSCCs is compared with the rate of detection in non-SCC equine penile lesions. If PVs are detected more frequently in PSCC than non-SCC lesions, this will provide evidence of a possible association between equine PSCC and PV infection.
Materials and Methods
Cases of equine penile disease were located by searching the archives of the Department of Biomedical Sciences, Cornell University, College of Veterinary Medicine, Ithaca, New York. Histologic diagnoses were confirmed for this study by one of the authors (J.P.). Ages were recorded, where available. Standard tests for normality were conducted using SAS 9.1. Differentiation between samples from geldings and stallions was not possible, because this information was not recorded.
For polymerase chain reaction (PCR), DNA was extracted from formalin-fixed paraffin-embedded tissues. Briefly, 10-μm sections were deparaffinized with xylene and then washed twice with ethanol. DNA was extracted with a DNeasy Blood & Tissue Kit (Qiagen GmbH, Hilden, Germany) according to manufacturer’s instructions. Two primer sets were used to amplify PV DNA from the samples by PCR. Both primer sets amplify conserved regions of the PV L1 gene.6,9 The primer set MY09/MY11 detects mucosal-adapted PVs, whereas the primer set FAP59/FAP64 detects cutaneous-adapted PVs.6,9 Both primer sets have been shown to detect PVs from a variety of animal species. 1 Amplification conditions for both primer sets were as previously published. 14 All PCR reactions were carried out in duplicate. DNA extracted from a bovine cutaneous fibropapilloma was used as a positive control, whereas template DNA was not included in the negative controls. Differences in PV detection rates were analyzed with Fisher exact test on a χ2 model, as performed with SPSS 15.0.
Amplicons were purified (Qiaex II 150 Gel Extraction Kit, Qiagen GmbH) and subjected to automatic dye-terminator cycle sequencing with BigDye Terminator 3.1 Ready Reaction Cycle Sequencing Kit and with an ABI3730 Genetic Analyzer (Applied Biosystems Inc, Foster City, CA). Results were compared with known sequences from GenBank (http://www.ncbi.nlm.nih.gov/Genbank/) using the basic local alignment search tool (BLAST; http://blast.ncbi.nlm.nih.gov/Blast.cgi).
In situ hybridization (ISH) for PV was performed using the Discovery automated slide-processing system (Ventana Medical Systems Inc, Tucson, AZ). The slides received pretreatment through mild cell conditioning using the citrate buffer–based RiboCC reagent (Ventana Medical Systems Inc) and enzyme pretreatment with Protease 3 for 12 minutes (Ventana Medical Systems Inc) as programmed. The sections were then incubated for hybridization with a generic antisense probe for PVs at 200 ng per slide for 1 hour at 37°C after a denaturing step for 4 minutes at 95°C (technique courtesy of Dr Matti Kiupel, of Michigan State University’s Department of Pathobiology and Diagnostic Investigation). Three stringency wash steps using 0.5× Ribo-Wash (Ventana Medical Systems Inc; equivalent to 0.5× saline sodium citrate) for 4 minutes at 42°C were followed by incubation of anti-rabbit anti-digoxigenin antibody (Sigma, St Louis, MO) for 32 minutes at 42°C. After streptavidin–alkaline phosphatase conjugate UMap anti-Rb AP (Ventana Medical Systems Inc) incubation for 16 minutes at 42°C, the signal was automatically detected with the BlueMap NBT/BCIP substrate kit (Ventana Medical Systems Inc) for 2 hours at 42°C. Finally, the sections were counterstained with the nuclear fast red–equivalent reagent Red Stain II (Ventana Medical Systems Inc) for 4 minutes before coverslipping.
Immunohistochemical (IHC) labeling was performed with an antibody that detects human PVs 1, 6, 11, 16, 18, and 31 (Lifespan Biosciences, Seattle, WA ). Briefly, IHC staining was performed with an automated staining system (Benchmark, Ventana Medical Systems Inc) that incorporated a commercial detection system (Enhanced V-Red Detection Kit, Ventana Medical Systems Inc). Antigen retrieval was achieved by incubating slides in a high-pH antigen retrieval solution (CC2 Std, Ventana Medical Systems Inc, Tucson, AZ) for 60 minutes. The primary antibody was applied at a concentration of 1:100 for 32 minutes. Sections were counterstained with hematoxylin. Positive control specimens included tissues known to be infected with canine PV. For negative control specimens, the primary antibody was replaced with Tris-buffer saline (Dako, Carpinteria, CA).
Results
Forty cases of equine penile disease were identified. These included 20 PSCCs and 20 cases of non-SCC penile disease (Table 1). The ages were available for 18 of 20 horses from the PSCC group and 14 of 20 horses from the non-SCC group. The average age of horses in the SCC group (23.9 years) was significantly higher than the average age of horses in the non-SCC group (13.3 years; P < .001). Age data for both groups followed a normal distribution. All horses in the study originated from the Northeastern United States with the exception of one, which was from Virginia.
Study Horses: Ages, Original Diagnoses, and EcPV-2 Amplification
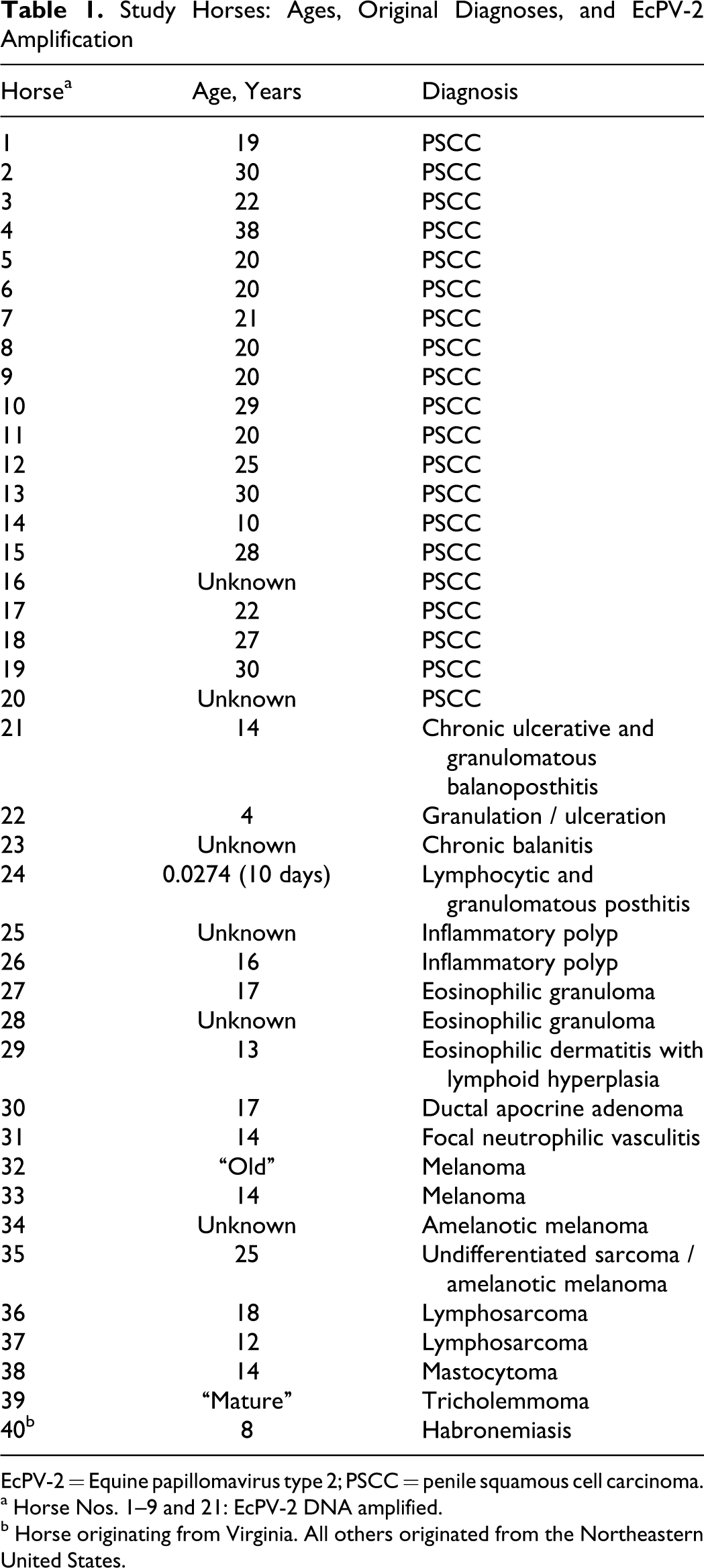
EcPV-2 = Equine papillomavirus type 2; PSCC = penile squamous cell carcinoma.
a Horse Nos. 1–9 and 21: EcPV-2 DNA amplified.
b Horse originating from Virginia. All others originated from the Northeastern United States.
As based on MY09/MY11 primers, PV DNA was amplified from 9 of 20 PSCCs. The PV DNA sequenced from all 9 was 98% to 100% similar to equine PV type 2 (EcPV-2; GenBank accession No. EU503122). The mismatching of small numbers of bases within the sequences is likely the result of errors in the sequencing process rather than evidence of multiple PV types. Based on MY09/MY11 primers, PV DNA was amplified from only 1 of 20 horses with non-SCC penile disease. The PV DNA amplified from this single case, a lesion of chronic ulcerative and granulomatous balanoposthitis, was 99% similar to EcPV-2 (GenBank accession No. EU503122). PV DNA was also amplified from the bovine fibropapilloma control according to MY09/MY11 primers. As based on the FAP59/FAP64 primer set, PV DNA was amplified from only the bovine cutaneous fibropapilloma control, suggesting that these primers have low affinity for EcPV-2.
PV DNA was detected significantly more frequently in lesions of equine PSCC than in lesions of non-SCC penile disease (P < .05). Because horses with PSCC in this study were significantly older than those with non-SCC disease, it was considered possible that older horses are more frequently infected by PVs. However, the average age of horses in which PV was detected (22.4 years) was compared with the average age of horses in which PV was not detected (17.9 years), and there was no significant difference (P = .1470).
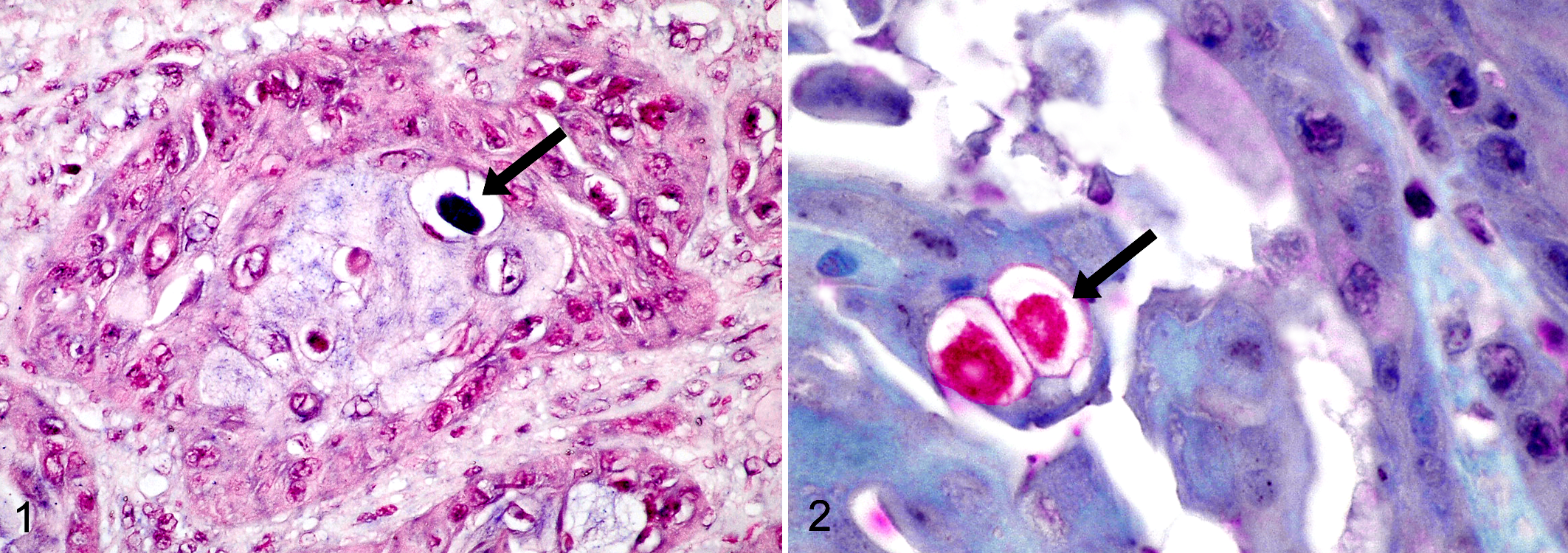
As based on ISH, PV DNA was detected in 6 of the 10 samples in which PV DNA had been detected by PCR. In these cases, PV DNA was detected in the nuclei of scattered neoplastic epithelial cells within lesions but not in adjacent, histologically normal tissue (Fig. 1). As based on IHC, PV antigen was detected in only 1 of the 10 samples in which PV DNA had been detected by PCR. In this case, antigen was detected in the nucleus of scattered neoplastic epithelial cells but not in adjacent, histologically normal tissue (Fig. 2).
Discussion
In this study PV DNA was detected by PCR significantly more frequently in equine PSCCs than in non-SCC penile diseases. In all 9 cases of PSCC in which PV DNA was detected, sequences from EcPV-2 were amplified. This report describes a possible association between PSCCs and EcPV-2, and is the first report of EcPV-2 infection in North American horses.
EcPV-2 is distinct from EcPV-1, which causes self-limiting cutaneous papillomas in young horses. 19 EcPV-1 has never been demonstrated in equine genital lesions, even those in which PV was visible immunohistochemically. 15 EcPV-2, the complete genome of which was characterized in 2008 (GenBank accession No. EU503122), is classified as a Dyoiotapapillomavirus, the only member of this genus. 3 In contrast, EcPV-1 belongs in the genus Zetapapillomavirus, and human PV 16, strongly associated with human penile cancer, is an Alphapapillomavirus.2,7
In humans, approximately half of PSCCs are associated with PV infection. For certain histologic subtypes, PV DNA can be identified in close to 100% of PSCCs.2,4,20 In horses, reports of an association between PVs and PSCCs are limited to the present study and a single conference presentation. 21
The significantly higher rate of detection of EcPV-2 in PSCCs in the current study suggests an association between this PV and equine genital neoplasia. However, given that PVs can asymptomatically infect skin, it is not possible to determine whether the PV is the cause of neoplasia in this study or simply an “innocent bystander.” 13 To prove that EcPV-2 causes equine PSCCs, further work is needed to investigate an oncogenic mechanism of the PV. Additionally, determining the incidence of EcPV-2 in horses and the mechanism of transmission may provide additional information on the relationship of this PV to equine PSCCs.
In addition to papillomas and invasive SCCs, a well-recognized lesion on the genitalia of old horses is the “precancerous plaque.” Precancerous plaques cannot be grossly distinguished from papillomas or SCCs and, because they frequently progress to invasive SCC, they are considered premalignant.5,10,12 Histologically, they consist of a dysplastic and hyperplastic epithelium, with or without koilocytes, overlying an intact basement membrane. 26 In a recent study, DNA of an unspecified PV was amplified by PCR from 10 of 10 penile and vulvar plaques. 26 These findings, along with the frequent detection of EcPV-2 in PSCCs in the current study, raise the possibility that the unspecified PV in precancerous plaques may be EcPV-2. In the current study, EcPV-2 DNA was amplified from 1 of 20 lesions of non-SCC disease. It is possible that this lesion, from a 14-year-old horse diagnosed with “chronic ulcerative and granulomatous balanoposthitis,” may have represented an early lesion of SCC, suggesting that PCR detection of EcPV-2 is warranted in cases of equine penile disease lacking a definitive diagnosis.
Interestingly, a 2007 study amplified a novel PV DNA sequence from 3 equine genital papillomas. 19 It is possible that the 2007 study actually amplified EcPV-2 DNA but could not classify it, because the EcPV-2 sequence was not added to GenBank until 2008. EcPV-2 sequences have recently been amplified from equine penile papillomas (C. G. Knight et al, unpublished data).
Several nonviral causes of equine PSCCs have been proposed. Because of the reported higher incidence of PSCCs in horses with lightly pigmented genital skin, ultraviolet radiation exposure has been suggested to promote neoplasm development.5,11 However, darkly pigmented horses also develop PSCCs.5,11 The accumulation of preputial smegma has been suggested as promoting equine PSCCs. While early studies reported that smegma was carcinogenic, 18 this finding is not universally accepted. 25 A possible role of smegma in development of equine PSCC was supported by the observation that geldings develop PSCC more frequently than do stallions, which tend to accumulate less smegma.5,8 This observation is not supported by all studies.10,12 In humans, chronic inflammation is associated with PSCC development. 4 Histologically, equine PSCCs often contain infiltrates of chronic inflammatory cells, but these are thought to be a response to the tumor, rather than a cause. 17
In the current study, tissues from the 10 horses in which PV DNA was detected by PCR were also screened using ISH and IHC. The presence of PV was demonstrated in a subset of these by ISH (6 of 10) and IHC (1 of 10), which suggests that PCR is a more sensitive test for the presence of EcPV-2 than either ISH or IHC; it also suggests that the virus is present in low quantities. In contrast to PCR or ISH, IHC detects structural capsid antigens and demonstrates these when replicating PV is present. Lack of detection of viral antigen by IHC in 9 of 10 horses in which PV DNA was amplified by PCR may indicate that there was no replicating PV or that the amount of antigen was reduced by prolonged formalin fixation.
In conclusion, the current study detected PV DNA by PCR significantly more frequently in equine PSCCs than in non-SCC lesions of the penis. The only PV amplified was EcPV-2. Although this study suggests that EcPV-2 is associated with a proportion of equine PSCCs, additional experiments are required to determine whether EcPV-2 is a cause of neoplasia.
Footnotes
Acknowledgment
We are deeply grateful to Dr Matti Kiupel, of Michigan State University’s Department of Pathobiology and Diagnostic Investigation, for performing in situ hybridization and immunohistochemistry in this study. We would also like to thank Dr Hendrik Nollens for providing the sequence of the papillomavirus probe.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
