Abstract
In humans, periostin plays a critical role in the enhancement and chronicity of allergic skin inflammation; however, whether it is involved in the pathogenesis of canine dermatitis remains unknown. The aim of this study was to examine the expression patterns of periostin in healthy, atopic, and nonatopic chronically inflamed canine skin. Biopsy specimens from 47 dogs with skin disease and normal skin tissue from 5 adult beagles were examined by light microscopy, immunohistochemistry, and in situ hybridization. In normal skin, periostin was localized just beneath the epidermis and around the hair follicles. In chronically inflamed skin, periostin expression was most intense in the dermis with inflammatory cell infiltrates. In contrast, low levels of periostin were detected in acutely inflamed and noninflamed skin. Conversely, all canine atopic dermatitis tissues characteristically showed the most intense expression of periostin in the superficial dermis, particularly at the epidermal–dermal junction. In situ hybridization showed that periostin mRNA was broadly expressed in the basal epidermal keratinocytes, outer root sheath cells, and dermal fibroblasts in normal dog skin. High expression of periostin mRNA was observed in fibroblasts in dog skin with chronically inflamed dermatitis. Moreover, in some chronically inflamed skin specimens, periostin mRNA expression was increased in basal keratinocytes. The severity score of chronic pathologic changes and CD3+ cell number in the dermis were correlated with distribution pattern of periostin in the atopic skin. These data suggest that periostin could play a role in the pathophysiology of chronic dermatitis, including atopic dermatitis, in dogs.
Periostin is an extracellular matrix protein that belongs to the fasciclin family. 10,15,23 Periostin modulates cell function by binding to integrin molecules on the cell surface, thus providing signals for tissue development and remodeling. 3,15,23,24 The production of periostin is induced by IL-4 and IL-13, which are signature cytokines of the T helper 2 (Th2)–type immune response in bronchial asthma, suggesting the involvement of periostin in allergic inflammation. 4,19 In humans, periostin is highly expressed in the skin of atopic dermatitis (AD) patients and is correlated with disease severity. 9 Mice lacking periostin are more resistant to sensitization to inhaled allergens. 9 In AD lesions, it is suspected that fibroblasts are the main source of periostin, although this has not been confirmed by in vivo analysis. 9,17,20 Periostin binds to αv-integrin on mouse keratinocytes in vitro, which results in the induction of proinflammatory cytokines, including thymic stromal lymphopoietin (TSLP). 9 Thus, periostin may play a critical role in the enhancement and chronicity of allergic skin inflammation.
Canine AD is a common, pruritic, allergic skin disease of dogs that shares several clinicopathologic features with human AD, including age of onset, skin area affected, severe pruritus, and an immunopathologic mechanism. 8,12,18 Histologically, the skin lesion is characterized by inflammation in the superficial dermis (SD). 13,18 In addition, both human and canine AD are characterized by allergic inflammation due to the release of various cytokines, mainly Th2 cytokines such as IL-4 and IL-13. 12,16,18 Klukowska-Rötzler et al 7 reported increased expression of the TSLP gene in the skin of dogs with AD, similar to that observed in human AD. However, only 1 report described a significant alteration of the gene expression of POSTN, which encodes periostin in atopic skin compared with healthy skin in dogs. 22
The aim of this study was to examine the expression patterns of the periostin protein in healthy, atopic, and nonatopic inflamed skin in dogs. Furthermore, we examined the localization of periostin mRNA by in situ hybridization (ISH) in these skin tissues.
Materials and Methods
The clinical data and pathologic diagnoses of 47 dogs of various breeds included in the present study are listed in Supplemental Table 1. Their ages ranged from 4 months to 17 years, and the cohort included 27 males, 19 females, and 1 dog for which the sex was not documented. A total of 62 biopsies were taken from the 47 dogs with skin diseases. Skin punch biopsies were fixed in 10% neutral-buffered formalin, embedded in paraffin, cut, and stained with hematoxylin and eosin. The skin diseases included in the present study were divided into 3 groups: chronic dermatitis (inflammatory skin disease with acanthosis and dermal fibrosis; case Nos. 1–41), acute dermatitis (case Nos. 42 and 43), and noninflamed skin disease (case Nos. 44–47). Chronic skin diseases included AD (n = 9), sebaceous adenitis (n = 4), superficial pyoderma (n = 2), pemphigus foliaceus (n = 2), deep pyoderma (n = 2), and sterile pyogranuloma (n = 2). The pathologic diagnosis was based on clinical and light microscopic features. The diagnosis of canine AD was based on the clinical criteria proposed by Terada et al. 21 The skin tissues from 5 adult beagles (2 males and 3 females, aged 1–7 years) were used as controls.
Total RNA was extracted from normal skin tissue using the TRIzol reagent (Invitrogen Life Technologies, Carlsbad, CA, USA). Extracted total RNA was treated with RNase-free DNase (Promega, Madison, WI, USA) and used in reverse transcription polymerase chain reaction (RT-PCR) employing the SuperScript III First-Strand Synthesis System (Invitrogen Life Technologies). Primer sequences were obtained by determining the predicted mRNA sequences of Canis lupus familiaris periostin (NCBI database accession No. XM_003433308). The primer sequences were as follows: canine periostin forward, 5′CAGCTCAGAGTCTTTGTCTATCGTACAG3′; reverse, 5′CTCCTGGTGTTAGGTGATAAAGGATG3′. PCR reaction with KOD FX Neo (Toyobo, Osaka, Japan) was performed as follows: denaturation at 94°C for 5 minutes; 30 cycles of denaturation at 98°C for 10 seconds, annealing at 66°C for 30 seconds, and extension at 68°C for 30 seconds; and a final extension step at 68°C for 30 seconds. After confirmation using agarose gel electrophoresis, the gene fragment was cloned into the pTA2 vector (Toyobo) using a TArget clone-Plus-TA cloning kit (Toyobo). The partial sequences of POSTN were cloned into One Shot TOP10 Chemically Competent Escherichia coli (Invitrogen Life Technologies) and sequenced using a 3730xl DNA Analyzer (Applied Biosystems, Foster City, CA, USA).
Immunohistochemistry (IHC) was performed using the immunoenzyme polymer method and the primary polyclonal or monoclonal antibodies shown in Supplemental Table 2. After incubation with 4% BlockAce (Yukijirushi, Sapporo, Japan) for 10 minutes at room temperature, dewaxed sections were incubated overnight with the antibodies against periostin at 4°C. Peroxidase-conjugated anti-mouse (Histofine Simple Stain MAX-PO (M), Nichirei, Tokyo, Japan) or anti-rabbit (EnVision+TM, Dako Corp, Glostrup, Denmark) immunoglobulin G (IgG) was used as secondary antibody. After the immunoreaction, the sections were colorized with diaminobenzidine and counterstained with Mayer’s hematoxylin. Nonimmune mouse or rabbit IgG was used in place of the primary antibody as a negative control.
Immunofluorescence assay was performed using anti-type IV collagen, anti-laminin, and anti-periostin antibodies. This assay used FITC-conjugated goat anti-rabbit IgG (EY Laboratories, San Mateo, CA, USA). Immunofluorescence was analyzed using an FSX100 fluorescence microscope (Olympus, Tokyo, Japan).
For quantitative morphometric analysis, the localization and distribution of periostin were assessed within 4 areas of the skin: the epidermal–dermal junction (EDJ), which constitutes an anatomic functional unit containing the basement membrane 1 ; the perifollicular area (PF), including the follicular basement membrane and the connective tissue follicle; the SD, the top layer of the dermis normally composed of thin collagen fibers; and the deep dermis (DD), the thin bottom layer of the dermis normally composed of thick collagen fibers. Within each area, the expression of periostin was assessed semiquantitatively and scored as follows: 0 = no expression, 1+ = weak expression, 2+ = moderate expression, 3+ = high expression. In canine AD cases, the 3 distribution patterns of periostin in the skin were defined as follows (compared with the normal control, which was assigned a normal pattern): basement membrane pattern (weak to moderate expression in EDJ and PF), superficial pattern (high expression in EDJ or weak expression in SD), diffuse pattern (moderate expression in SD or expression in DD), as shown in Table 1. Epidermal thickness was measured; the number of CD3-positive cells (T cells), CD20-positive cells (B cells), and mast cells in the SD were counted in 10 randomly selected high-power fields for each sample, and their average values were calculated. Dermal fibrosis was semiquantitatively assessed to evaluate the severity of chronic pathologic changes as follows: no (0), mild (1+), moderate (2+), and severe (3+).
Distribution Pattern and Expression Intensitya of Periostin in the Skin From Atopic Dogs.
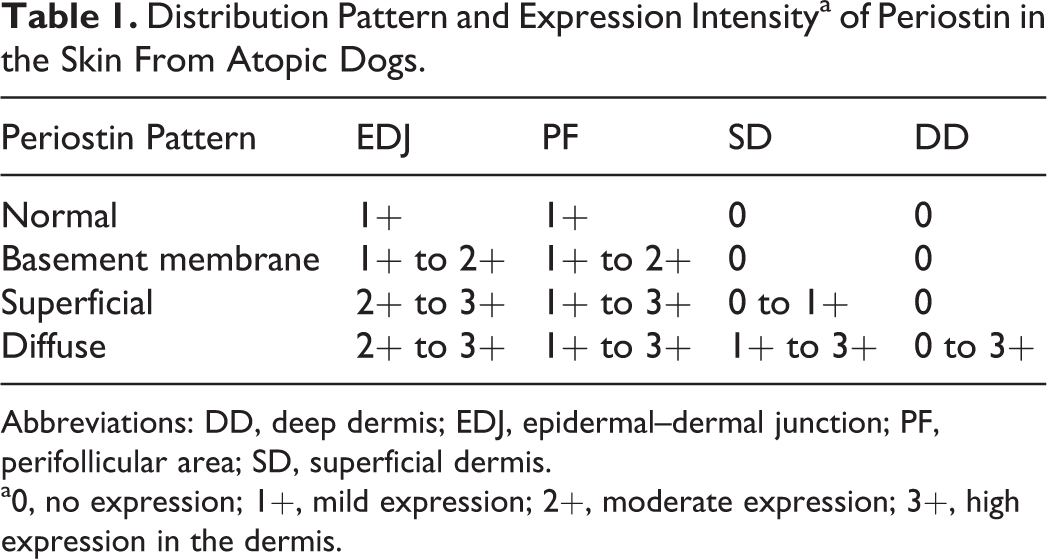
Abbreviations: DD, deep dermis; EDJ, epidermal–dermal junction; PF, perifollicular area; SD, superficial dermis.
a0, no expression; 1+, mild expression; 2+, moderate expression; 3+, high expression in the dermis.
ISH was performed on samples from 21 dogs (case Nos. 1–7, 10, 11, 15, 17, 18, 21, 22, 24, 31, 32, 36, 37, 42, and 43) using the QuantiGene ViewRNA ISH Tissue Assay (Affymetrix, Santa Clara, CA, USA) according to the manufacturer’s protocol. Gene-specific probe sets for canine periostin mRNA consisting of an average of 20 different probe pairs were custom designed and synthesized by Affymetrix. The signal for periostin mRNA was visualized with fast red and counterstained with Gill’s hematoxylin. A canine periostin mRNA sense probe or an ISH reaction solution without the probes was used as a negative control, according to Affymetrix’s recommendations.
Statistical analyses of distribution patterns of periostin and histologic scores were performed using GraphPad Prism (version 5.0, GraphPad Software, La Jolla, CA, USA). One-way analysis of variance was performed to analyze the difference between distribution patterns of periostin and histologic scores, and significant differences were identified using the Tukey–Kramer test. P values <.05 were considered statistically significant.
Results
Expression of the Periostin Gene in Normal Skin Tissues
RT-PCR was performed to confirm the expression of the periostin gene. The mRNA encoding periostin (346 base pairs) was present in normal skin tissue (data not shown). The sequence was registered in the DNA Data Bank of Japan (accession No. LC008358). Sequence analysis of the amplification product showed homology to the partial sequences of predicted canine periostin mRNA (100%) and human periostin mRNA (92%).
Expression and Localization of Periostin mRNA and Protein in Normal Dog Skin
ISH revealed that periostin mRNA-positive cells were located in the outer root sheath, particularly in the outer cells along the basement membrane, whereas there was a small number of signals in the outer root sheath inner cells in normal dog skin (Fig. 1a). The periostin protein was located at the basement membrane of hair follicles (score 1+; Fig. 1b). In the epidermis, periostin mRNA was expressed only in the basal epidermal keratinocytes (Fig. 2a). The periostin protein was detected at low level in the EDJ (score 1+; Fig. 2b) but not in DD (score 0). Periostin protein was not detected in the cells. The signals were not detected in tissues that underwent the ISH procedure using the sense probe or the ISH solution without the probe (Fig. 2c). No staining was observed with nonimmune rabbit IgG (Fig. 2d).
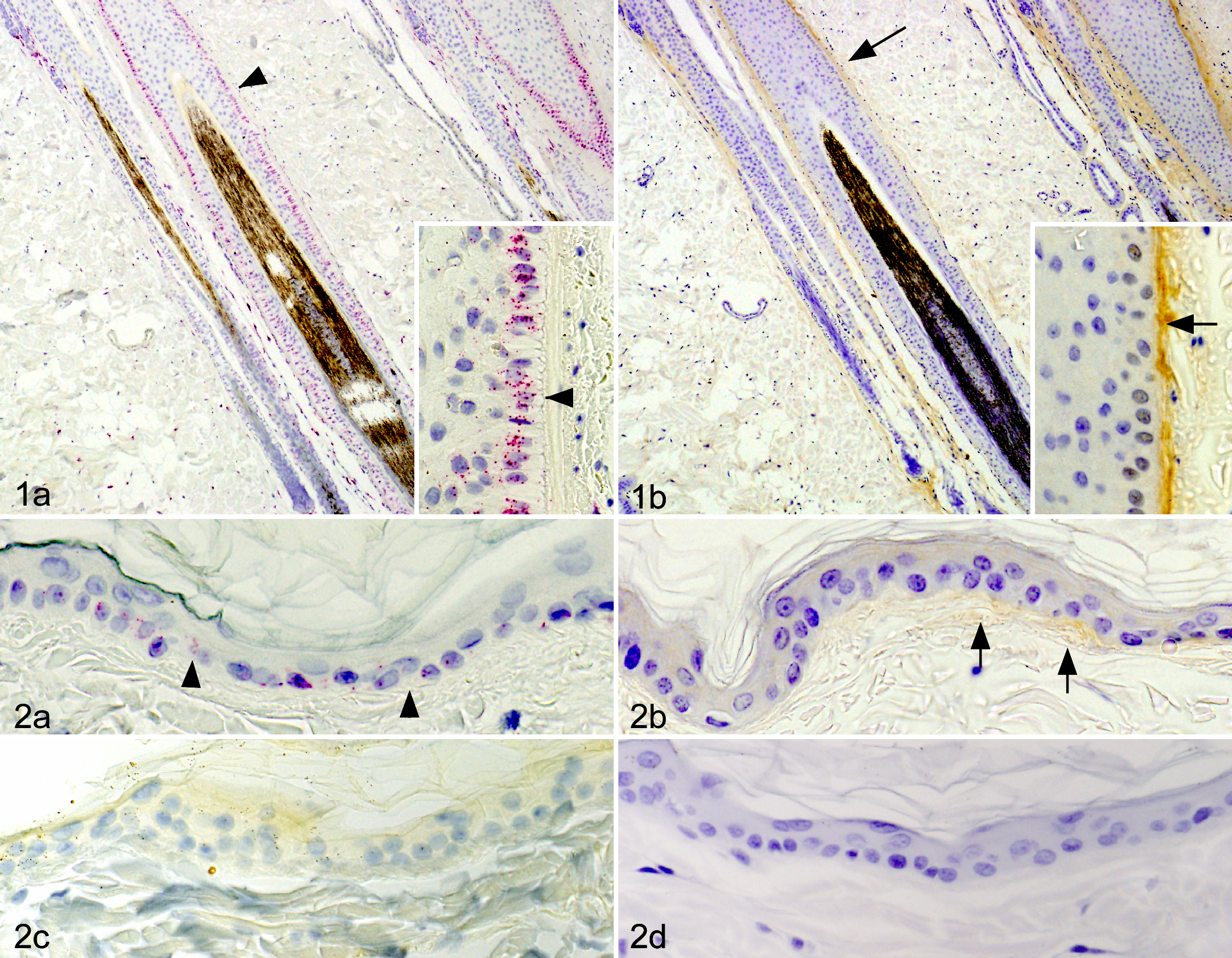
Expression and Localization of Periostin mRNA and Protein in Skin Tissues of Dogs With Chronic Inflammatory Disease
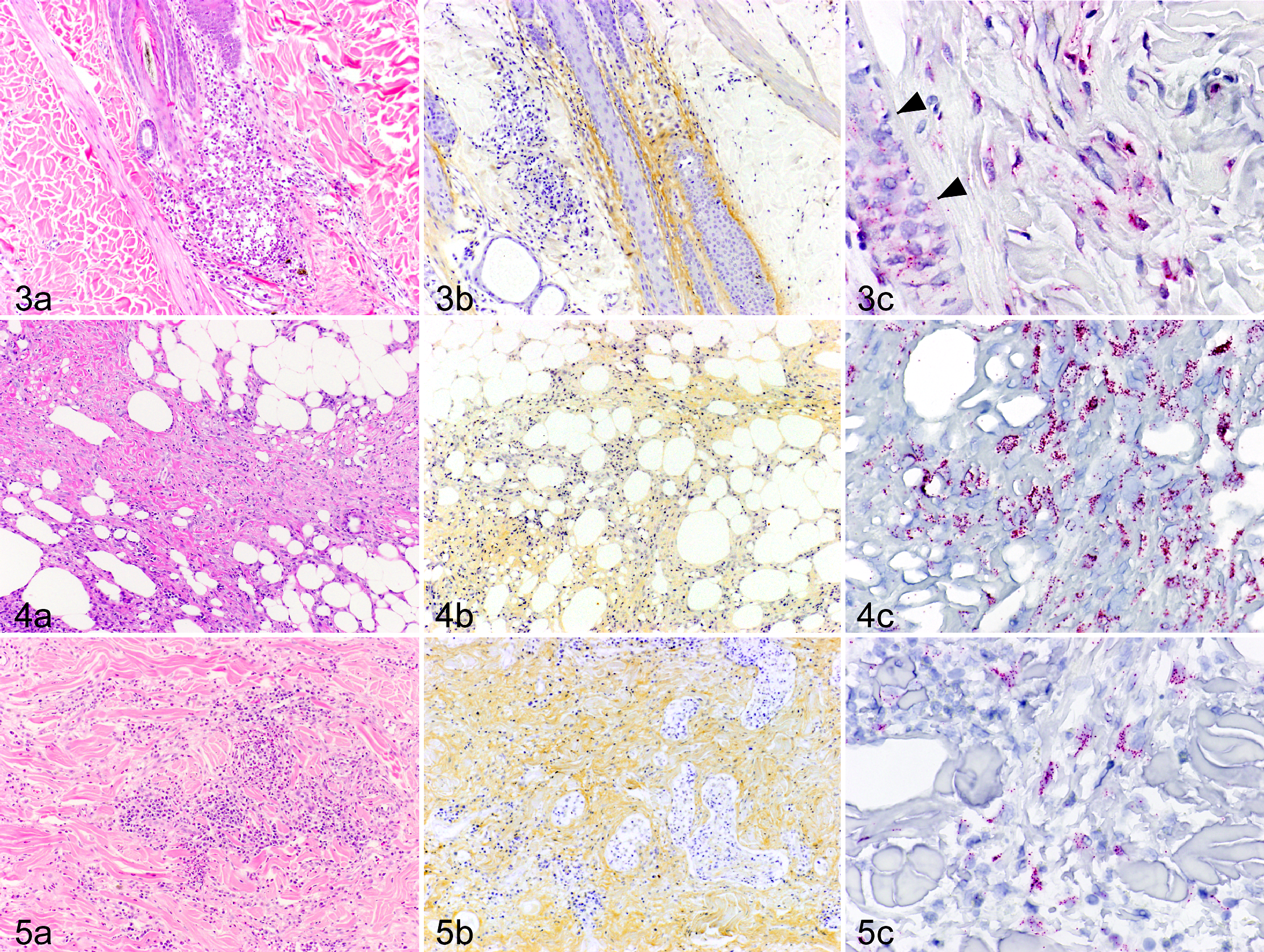
The results of the immunohistochemical examination of skin diseases are summarized in Supplemental Table 1. Periostin protein expression in chronically inflamed skin was most intense in the dermis with inflammatory cell infiltrates (scores 1+, 2+ or 3+; Figs. 3a, b, 4a, b, 5a, b), whereas low levels of periostin were detected in acutely inflamed and noninflamed skin diseases. Sebaceous adenitis was characterized by the destruction of the sebaceous gland and associated with an intense expression of periostin in the SD, particularly at PF. In diffuse dermatitis, periostin protein expression was intense throughout the dermis. In sterile pyogranuloma, both inflammatory cells and periostin protein expression were observed in DD and subcutaneous tissue. Among the inflammatory skin diseases, all canine AD tissues characteristically showed the most intense expression of periostin in the SD, particularly at EDJ (2+ or 3+; Fig. 6a). Expression of type IV collagen and laminin was detected at the EDJ and vascular basement membrane (Fig. 7a, b). Periostin was more prominently expressed at the EDJ and detected faintly at the vascular basement membrane (Fig. 7c). No staining was observed with nonimmune rabbit IgG (Fig. 6c).
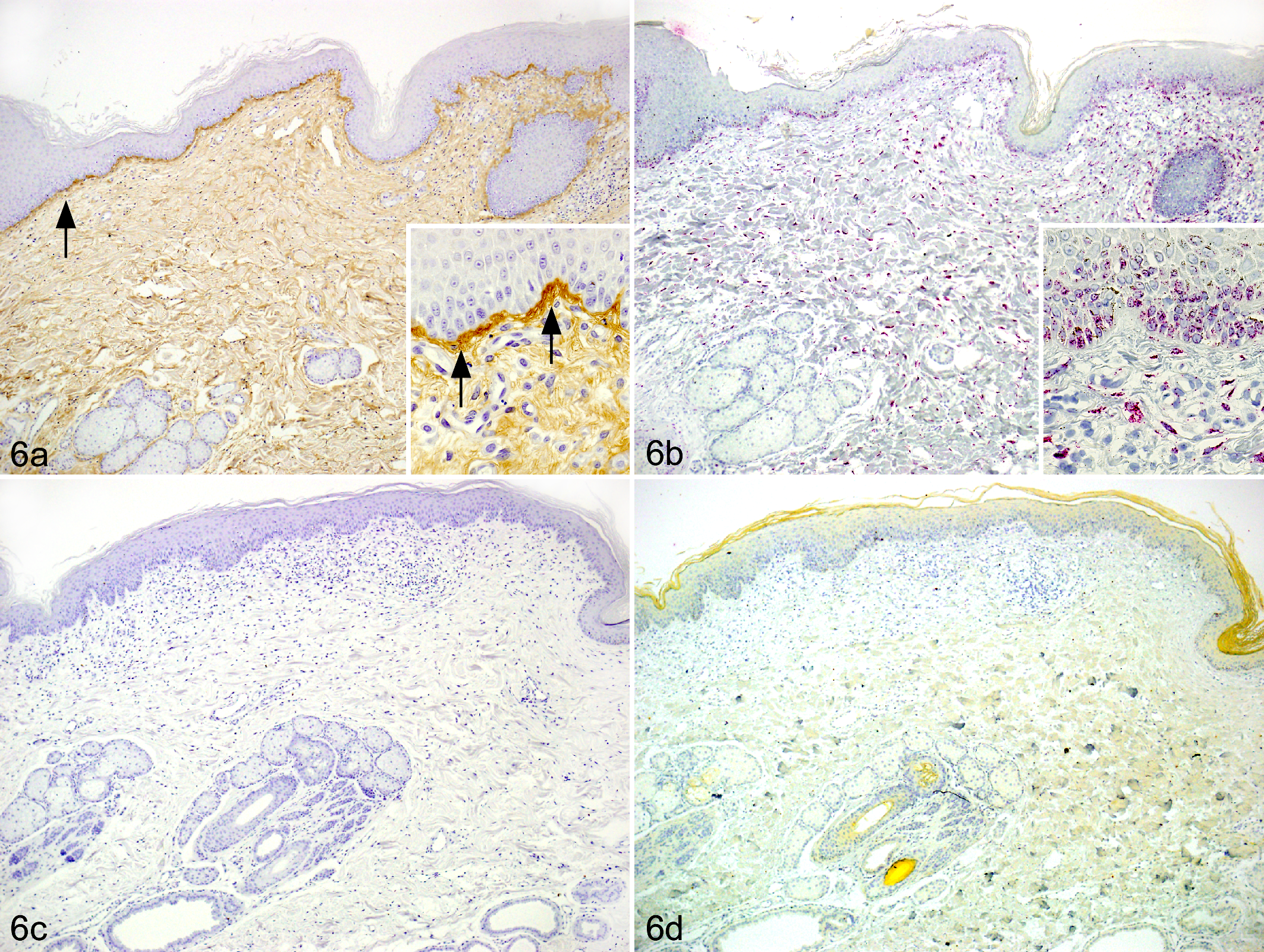
Atopic dermatitis, skin, dog, case No. 5. (a) The expression of periostin was significantly elevated in the superficial dermis, particularly at the epidermal–dermal junction (arrows). Inset: High-power magnification view of the epidermis and superficial dermis, IHC for periostin. (b) The periostin mRNA was more prominently expressed in keratinocytes. Inset: High-power magnification view of the epidermis and superficial dermis. In situ hybridization (ISH) for periostin. (c) No staining was observed with nonimmune rabbit immunoglobulin G. IHC. (d) No signal was observed with the periostin mRNA sense probe. ISH for periostin (sense probe).
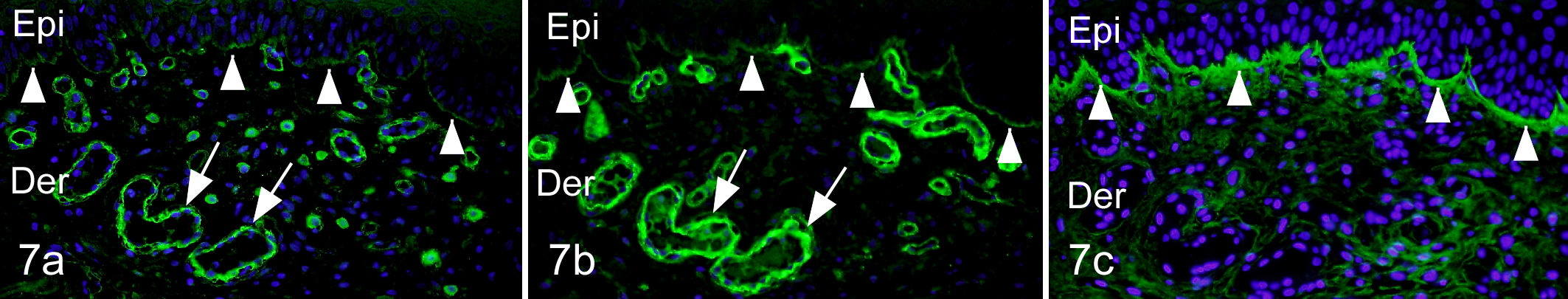
Atopic dermatitis, skin, dog, case No. 5. Epi, epidermis; Der, dermis. (a) Expression of type IV collagen was detected at the epidermal–dermal junction (EDJ; arrowheads) and vascular basement membrane (arrows). Immunofluorescence staining (IF) for type IV collagen. (b) Expression of laminin was detected at the EDJ (arrowheads) and vascular basement membrane (arrows). IF for laminin. (c) Periostin was more prominently expressed at the EDJ (arrowheads) and detected faintly at the vascular basement membrane. IF for periostin.
Positive signals for periostin mRNA were detected in spindle-shaped stromal cells in the inflammatory areas (Figs. 3c, 4c, 5c), together with protein expression. These stromal cells were positive for vimentin and negative for factor VIII–related antigen and smooth muscle actin, indicating that most of the stromal cells were fibroblasts. In addition, the periostin mRNA was expressed in some adipocytes. In 6 of 21 dogs (case Nos. 3, 5, 10, 15, 24, 32), increased periostin mRNA expression was observed more clearly in epidermal keratinocytes (Fig. 6b). No signals were detected in tissues that underwent the procedure using the sense probe or the ISH solution without the probe (Fig. 6d).
The distribution patterns of periostin were correlated with chronic pathologic changes and the number of CD3+ cells in the skin tissues of atopic dogs. Chronic pathologic changes (epidermal thickening and fibrosis) and the number of CD3+ cells in the skin were significantly different between some distribution patterns of periostin, especially normal and diffuse patterns, although there were no significant differences in the number of CD20+ cells and mast cells between the different distribution patterns of periostin (Table 2, Fig. 8).
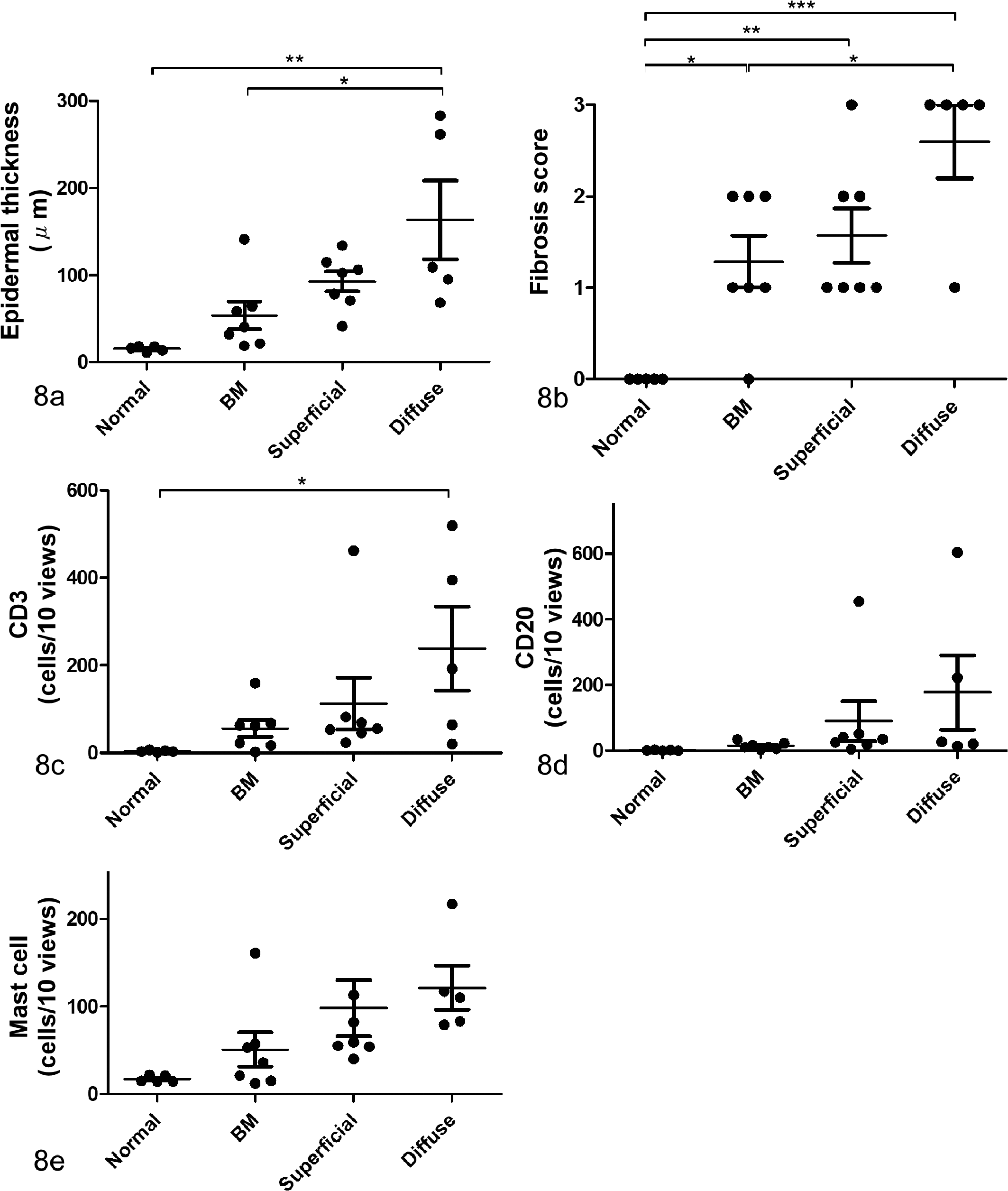
Correlation between distribution patterns of periostin and histologic severity in the skin tissues of atopic dogs. Three distribution patterns of periostin of atopic dogs were as follows: basement membrane (BM) pattern, superficial pattern, and diffuse pattern (compared with the healthy control, which was assigned a normal pattern). The number of inflammatory cells (CD3+, CD20+, and mast cells) in the dermis was counted in 10 high-power fields for each sample. Chronic pathologic changes (epidermal thickening and fibrosis) and the number of CD3+ cells in the skin were correlated between some distribution patterns of periostin, especially between normal and diffuse pattern. *P < .05. **P < .01. ***P < .001. One-way analysis of variance. (a) Epidermal thickness was measured in 10 high-power fields for each sample, and a significant positive correlation was observed between epidermal thickness and periostin patterns. (b) Correlation between periostin patterns and fibrosis scores. (c) Correlation between periostin patterns and the number of CD3-positive cells (T cells). (d) Correlation between periostin patterns and the number of CD20-positive cells (B cells). (e) Correlation between periostin patterns and the number of mast cells.
Histologic Findings and Periostin Immunolabeling of Tissue Samples From Atopic Dogs.
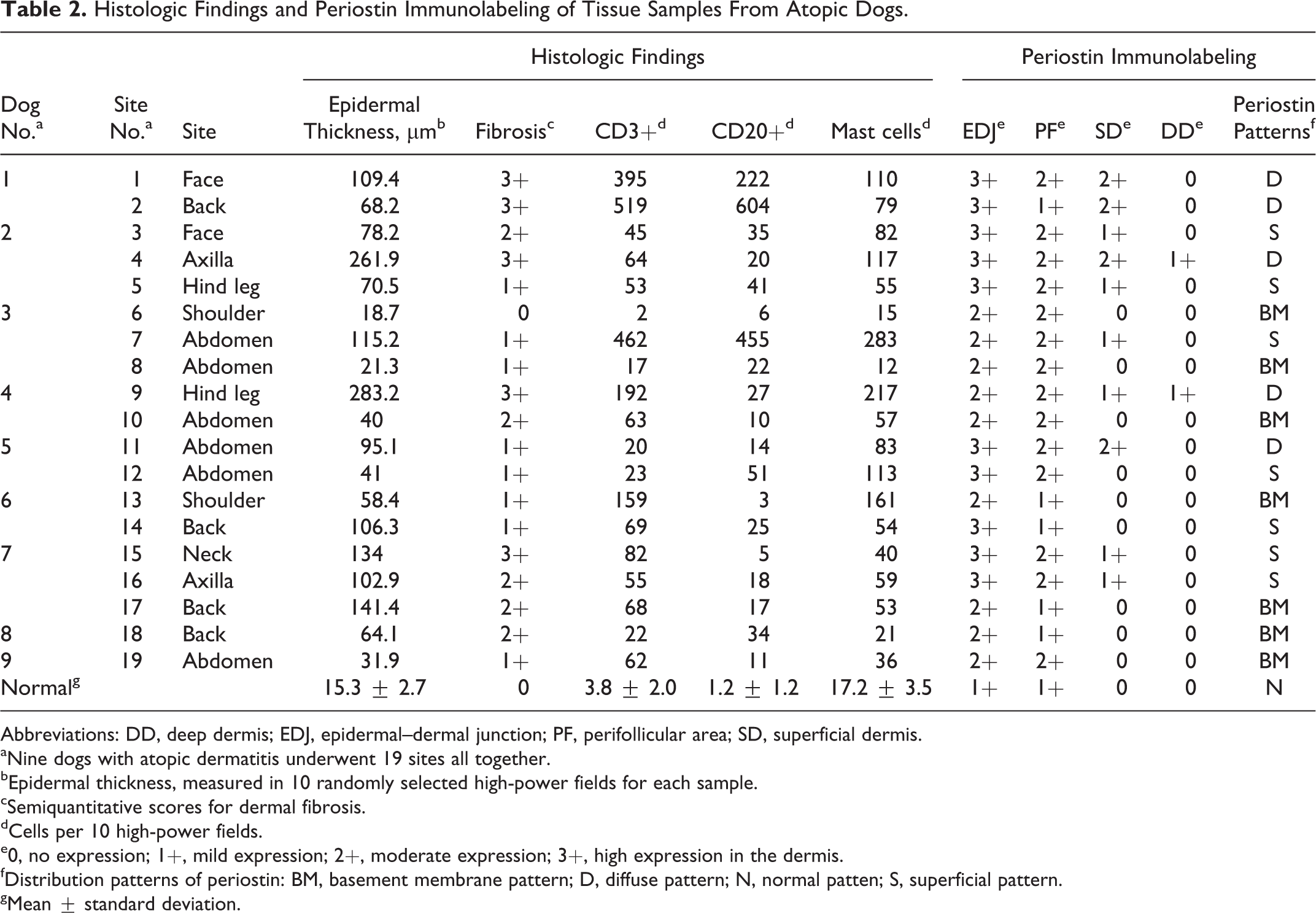
Abbreviations: DD, deep dermis; EDJ, epidermal–dermal junction; PF, perifollicular area; SD, superficial dermis.
aNine dogs with atopic dermatitis underwent 19 sites all together.
bEpidermal thickness, measured in 10 randomly selected high-power fields for each sample.
cSemiquantitative scores for dermal fibrosis.
dCells per 10 high-power fields.
e0, no expression; 1+, mild expression; 2+, moderate expression; 3+, high expression in the dermis.
fDistribution patterns of periostin: BM, basement membrane pattern; D, diffuse pattern; N, normal patten; S, superficial pattern.
gMean ± standard deviation.
Discussion
To the best of our knowledge, this report is the first to show the expression patterns of periostin mRNA and protein in various spontaneous skin diseases of dogs. We found that in normal canine skin, similar to human and murine skin, periostin mRNA was expressed only in the basal epidermal keratinocytes, outer root sheath cells, and dermal fibroblasts and that periostin was deposited just beneath the epidermis and around the hair follicles. 11 Furthermore, the distribution pattern of periostin mRNA and protein in normal and inflamed skin appeared to be significantly different. An increased expression of periostin was observed in chronically inflamed skin compared with normal, noninflamed, and acutely inflamed skin.
In contrast, real-time RT-PCR using total RNA from sections of 5 lesion sites in 4 atopic dogs showed no significant difference in the ratio of periostin:GAPDH mRNA expression between atopic and control skin groups (supplemental material). By real-time RT-PCR using frozen sections, it was impossible to determine the distribution pattern of periostin mRNA. As the number of periostin-nonproducing cells (eg, lymphocytes) was increased in atopic skin compared with control skin, it was difficult to compare periostin gene expression.
In humans and experimental animal models, it has been shown that periostin may play a critical role in the chronicity of allergic inflammation.4,9,17,19,23 Recently, using periostin-deficient mice, Masuoka et al 9 reported that a lack of periostin results in failed development of allergic skin inflammation caused by sensitization to inhaled allergens. Furthermore, in vitro analyses revealed that periostin promoted the production of TSLP through integrins and the NF-κB pathway in keratinocytes. 9 Increased periostin expression in the skin of dogs with AD and other types of chronic dermatitis, as observed in the present study, supports the proposed involvement of proinflammatory cytokines in the pathogenesis of human AD and experimental AD models.
Previous in vitro analyses showed that dermal fibroblasts produce and secrete periostin, which is dependent on cytokine stimulation via integrin signaling. 9,17,20,23 Our ISH data strongly suggest that fibroblasts are the main source of periostin in inflamed dermatitis in dogs. Conversely, in some inflamed canine skin, periostin mRNA was notably expressed in keratinocytes. Nishiyama et al 11 showed that periostin mRNA was temporarily expressed not only in fibroblasts but also in keratinocytes in hair follicles during wound healing. To the best of our knowledge, no reports have focused on periostin production by epidermal cells in inflamed skin.
In the present study, we confirmed that canine AD is characterized by the expression of periostin in the SD, particularly at EDJ. Periostin was partially colocalized with other extracellular matrix, such as type IV collagen and laminin components of the basement membrane. Moreover, the severity of chronic pathologic changes and infiltration of CD3+ cells in the dermis were different between distribution patterns of periostin, especially between normal and diffuse patterns. In atopic dogs, T cells are major components of the inflammatory cell infiltrate in the skin. 6 Th2 cytokines play a critical role in inducing allergic inflammation in addition to promoting IgE class switching. 12,16,18 Previous studies showed that canine AD is associated with overproduction of Th2 cytokines. 5 Therefore, we suspect that Th2 cytokines may induce periostin production in keratinocytes and that periostin plays a critical role in the chronicity of allergic inflammation via an autocrine or paracrine mechanism. Several articles have recently shown that canine AD is a chronic and recurrent skin disease with a Th2-type response. 5,7,8,12,16,18 Despite treatment with inhaled glucocorticoids or cyclosporin, some dogs continue to have uncontrolled AD that requires more intensive therapy. 14 Periostin may contribute to the enhancement and chronicity of allergic skin inflammation by activating keratinocytes and fibroblasts in the absence of environmental allergens. Corren et al 2 examined patients with poorly controlled asthma treated with lebrikizumab, which is a monoclonal antibody to IL-13. Patients with high pretreatment levels of serum periostin had greater clinical improvement than patients with low periostin levels. Furthermore, the serum levels of periostin were significantly elevated in AD patients compared with controls. 9 We suspect that periostin may play an important role in the pathogenesis of dermatitis in dogs. In vitro analyses using cultured canine keratinocytes and fibroblasts may provide valuable information regarding stimulatory factors and the functional roles of periostin.
Footnotes
Acknowledgements
We thank Dr Yoko Kakinuma at Azabu University for her excellent assistance. We also thank Dr Sadatoshi Maeda, Department of Veterinary Internal Medicine, Gifu University, for his valuable advice regarding quantitative polymerase chain reaction methods. This study was supported by many veterinary clinicians, and we are especially thankful to Dr Tamio Ohmuro, Ohmuro Veterinary Clinic, and Dr Fumio Suetsugu, Maria Pet Clinic.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author was supported by the graduate student research project grant 2014(DV1206) awarded by Azabu University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
