Abstract
Four cases of neoplasms in the heads of old dogs were studied. All the dogs showed both carcinomatous and sarcomatous malignant components in an admixed growing pattern. Histologic analysis of the tumors showed that the carcinomatous cells resembled squamous cell carcinoma in all dogs except one, where an adenoid arrangement of the neoplastic cells was also observed. The sarcomatous component showed osteoid matrix produced by pleomorphic poorly differentiated cells, which is regarded as a typical feature of osteosarcomas. Immunohistochemical analysis demonstrated that the components were positive only for cytokeratin (carcinomatous component) or vimentin (sarcomatous component). This observation led us to classify the neoplasms as true carcinosarcomas, thus providing evidence of a new preferential location for this unusual tumor in dog.
Carcinosarcomas have been defined by the World Health Organization 14 as neoplasms composed of intimately admixed malignant epithelial and mesenchymal elements. These tumors are uncommon in veterinary medicine, although, in dogs, they have been reported in the mammary gland, 8 thyroid gland, 7 mandibular salivary gland, 10 lumbosacral region, 9 and lung. 11 In other species, they are even rarer: for example, one case has been described in a feline lung 6 and another in the skin of a horse. 1 In human medicine, this type of tumor is also uncommon but has been found in several anatomical sites: the female genital tract, skin, gastrointestinal tract, hepatobiliary system, head, neck, respiratory system, and urinary tract. 14 These tumors usually occur in older individuals and display aggressive biologic behavior. 2 Their histogenesis is not clearly understood, and even their nomenclature is a matter of discussion.
Four neoplasms in old dogs (8–13 years) were studied. All related with osseous structures in the head. The breed, age, sex of the dogs, and location of the tumors are summarized in Table 1. No data about gross morphology were supplied by the clinicians. In addition, in the necropsy of dog 4, samples were taken from the regional lymph nodes, lung, liver, and spleen. Tissue samples for light microscopy were fixed overnight in 10% neutral formalin, paraffin embedded, and stained with hematoxylin and eosin (HE). Selected 4-µm histologic sections were stained with Massson's trichrome or by the avidin-biotin-peroxidase complex method (ABC) with several primary antibodies, whose clone, dilution, and pretreatment for antigen retrieval are summarized in Table 2. All the antibodies were purchased from DAKO Corp. (Carpinteria, CA). Counterstaining involved Mayer's hematoxylin, while both positive- and negative-control sections were included for each antibody.
Characteristics of the dogs and location of tumors.

Characteristics of the primary antibodies used in this study.
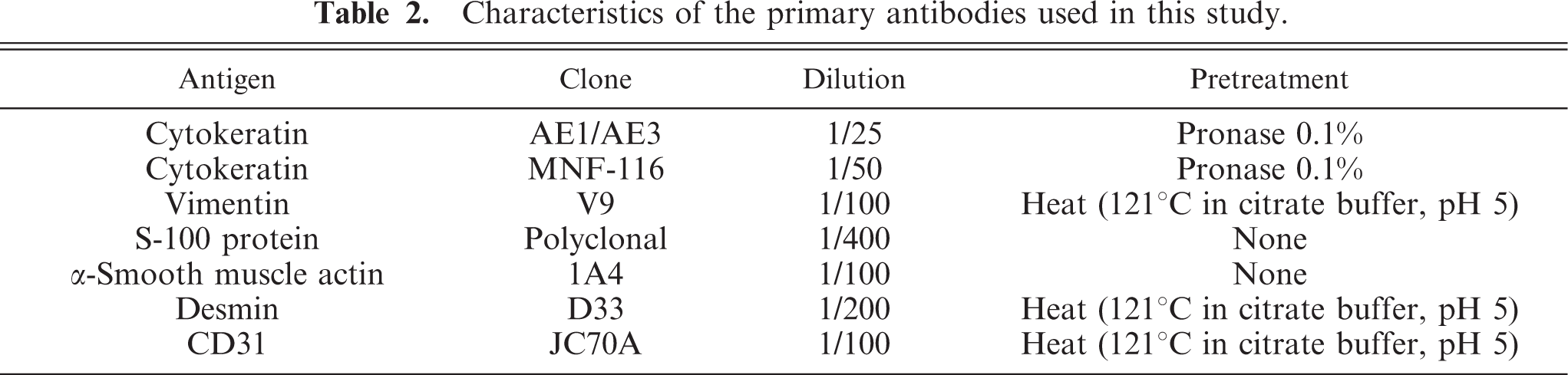
In dogs 1 and 2, a tumor mass with an infiltrative pattern of growing was observed. No lymphatic or blood vessel invasion could be observed in the periphery of the tumors. Differently sized islands of polyhedral cells showing ovoid or vesicular nuclei with prominent nuleoli and abundant lightly acidophilic cytoplasm resembling epithelial cells were observed. Among these groups of cells, keratin could be observed (Fig. 1a, b). In dog 2, a well-differentiated osseous tissue was found admixed with epithelial cells and keratin. The presence of some inflammatory cells in these areas suggested an osteolytic process. In both dogs, the epithelial cells and keratin showed strong positive immunolabeling for both anticytokeratin monoclonal antibodies (Fig. 1c), although no immunolabeling was observed with the other antibodies used in this study. The islands were surrounded by a heterogenous cell population consisting of cells with scant cytoplasm and elongated or oval nuclei containing inconspicuous nucleoli and ovoid or polygonal cells with large round or oval nuclei and prominent nucleoli (Fig. 1a, b). Few mitotic figures were observed (<1 figure/field at 400×). Among these cells, osteoid material was observed, as thin strands of hyaline and acidophilic material, which stained blue with Masson's trichrome. The heterogeneous cell population showed positive immunolabeling for antivimentin antibody (Fig. 1d), and a small number of spindle cells also stained positive for anti-α-actin antibody. In dog 1, scarce cells positive for cytokeratin labeling were admixed with the heterogenous mesenchymal cells, but no coexpression of vimentin and cytokeratin could be observed. In dog 2, anastomotic cords of small polyhedral cells showing positive immunolabeling for anticytokeratin were observed among the heterogeneous mesenchymal cells; adjacent sections showed that these cells were negative for antivimentin labeling.
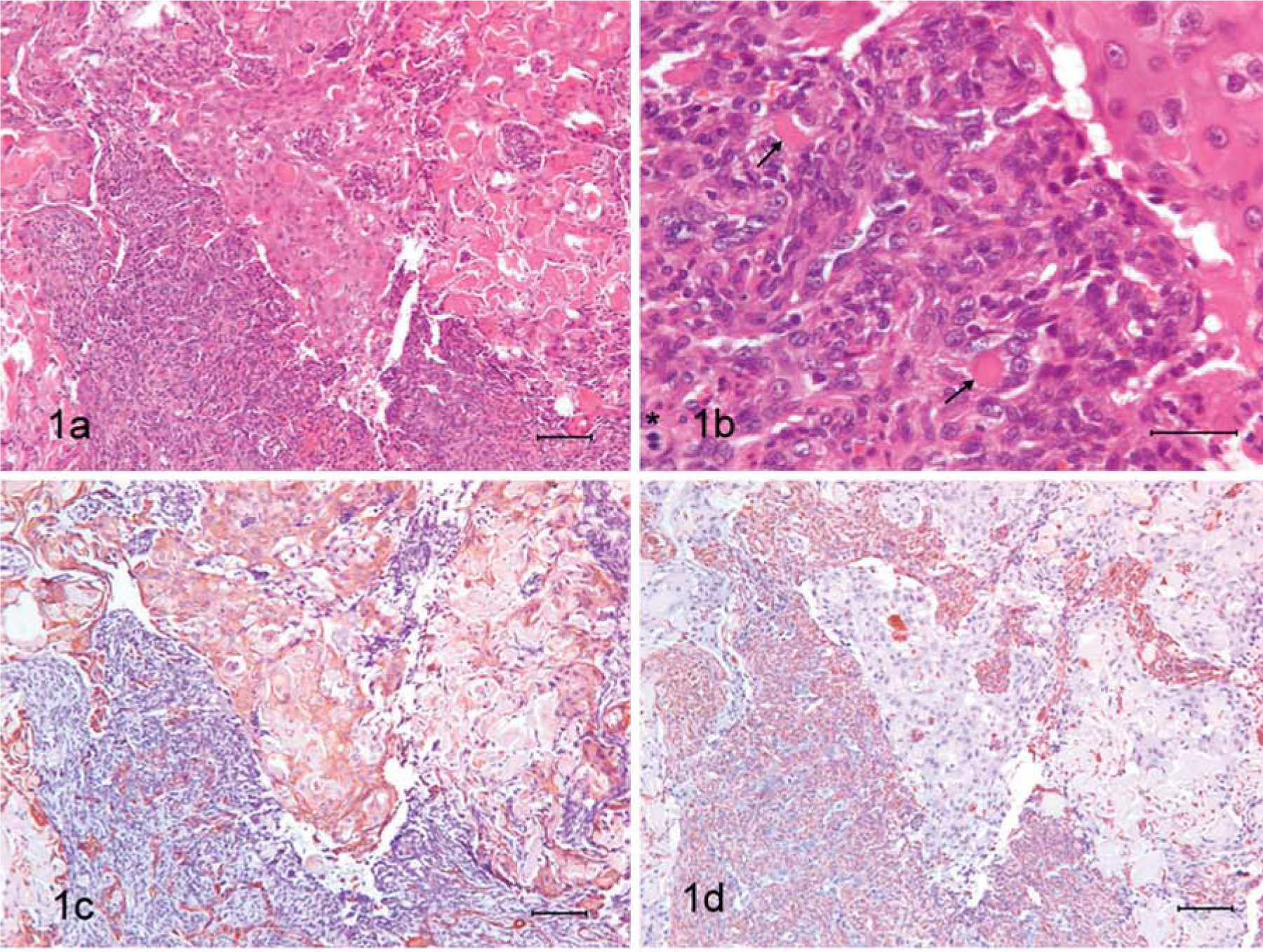
Dog 2. Area of tumor where admixed proliferation of mesenchymal and epithelial neoplastic cells is shown. Keratin is abundant among epithelial cells. HE. Bar = 100 µm.
A tumor mass with invasion of adjacent tissues was observed in dog 3. Invasion of lymphatic or blood vessels could be not observed. Neoplastic cells were located in large nests or follicles with wide trabeculae of well-differentiated connective and osseous tissue between them. The neoplastic cells were polyhedral or rounded, with a high nucleus/cytoplasm rate; the scant cytoplasm was basophilic, and the nuclei were rounded or irregularly shaped, euchromatic, with prominent nucleoli and significant anisocariosis. A high mitotic index (3–4 figures/field at 400×) was also observed. Apoptotic figures were usually found in the center of the nests. Within the nests, small groups of polyhedral cells showing vesicular nuclei and abundant eosinophilic cytoplasms were observed (Fig. 2a,b). In other areas of the neoplasm, the tumor cells showed a lack of follicle organization and a diffuse growth was observed with osteoid material among the cells. Irregular groups of cells with squamous differentiation were observed in these areas. Immunolabeling demonstrated that the abundant eosinophilic cytoplasm cells in the nests and the cells with squamous differentiation were positive for anticytokeratin antibodies (Fig. 2c), while most of the cells in the follicles or nests and the surrounding stroma were positive for antivimentin antibody (Fig. 2d). No coexpression of both antigens was found. No immunolabeling was observed for the other antibodies used in this study.
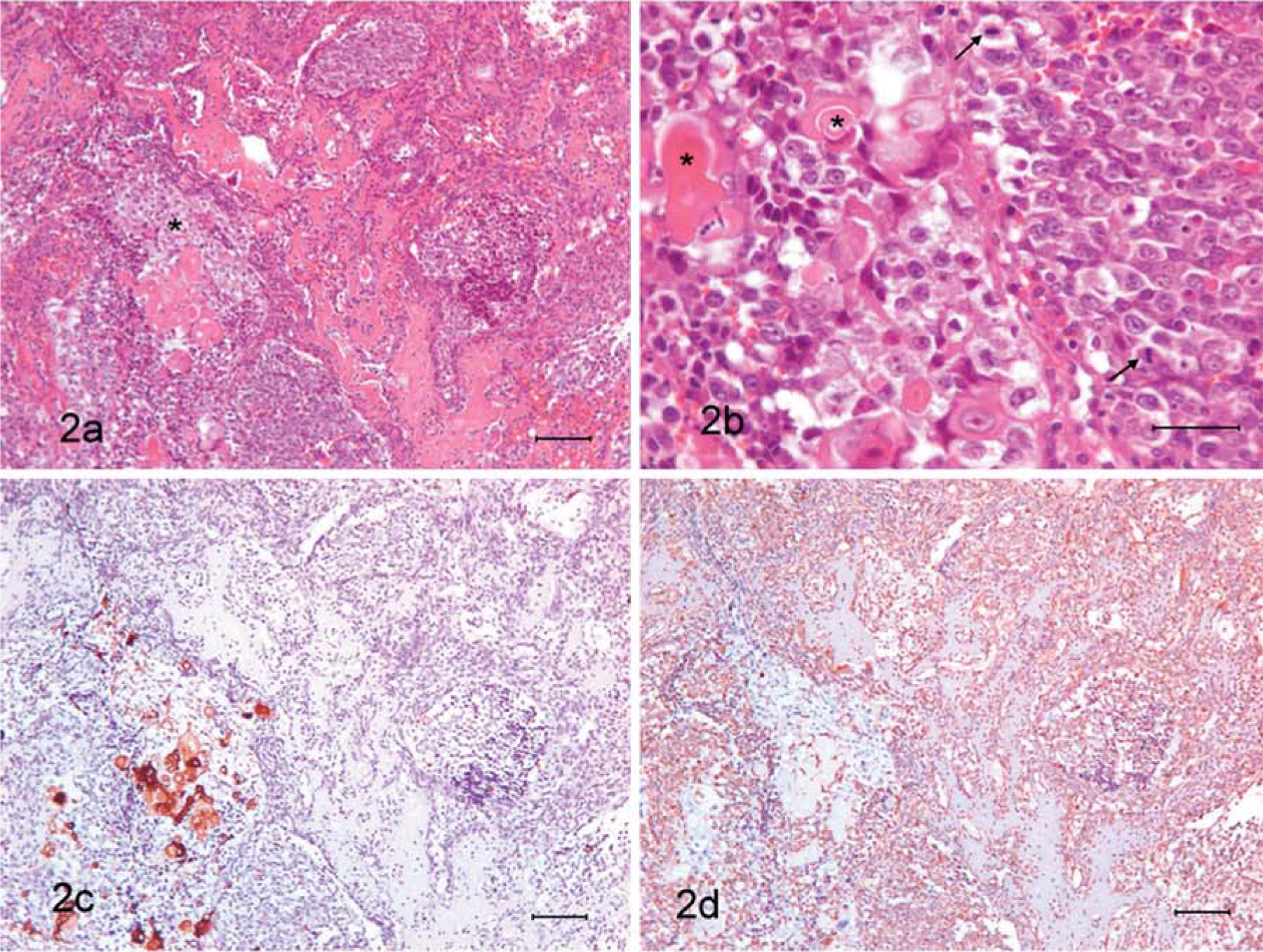
Dog 3.
The tumor of dog 4 displayed two clearly defined areas. The area was separated in some places by a connective stroma (Fig. 3a). Both areas were joined to the adjacent osseous tissue and osteolytic processes were observed in the peripheric areas. In one of them, most of the tumoral cells resembled a neoplastic epithelial proliferation with papillar and acinar organization. The papillae were lined by 3–4 layers of polyhedral cells with scant to moderate basophilic cytoplasm containing rounded to oval euchromatic nuclei with prominent nucleoli. The most superficial cells in the papillae were larger and showed an abundant eosinophilic cytoplasm (Fig. 3b). Mitotic figures were scarce (<1/field at 400×). Necrosis and hemorrhages were found.
Dog 4.
In the another area, most of the cells were mesenchymal-like with a fusiform, ovoid, rounded, or reticulated morphology, high nucleus/cytoplasm ratio, scant acidophilic cytoplasm and irregular nuclei with 1–2 prominent nucleoli (Fig. 3c). Moderate mitotic index (2/field at 400×). Among these cells, well-differentiated osseous trabeculae and foci of calcifying and ossifying cartilage were observed. Small islands of cells with squamous differentiation were observed within this sarcomatous area.
Immunohistochemical analysis confirmed the epithelial nature of the first described area of the tumor because cells in the papillae and acini showed positive immunolabeling for anticytokeratin antibodies (Fig. 3d). Infiltrations of mesenchymal-like cells were observed near the border with the area. The mesenchymal-like cells showed strong positive immunolabeling for antivimentin antibody (Fig. 3e) and negative for anticytokeratin antibodies; cartilaginous foci were positive for anti-S-100 protein antibody.
The four neoplasms included in this study contained a mixed cell population, and the malignant proliferation of both epithelial- and mesenchymal-like cells could be observed. Malignant epithelial proliferation could be diagnosed as squamous cell carcinomas, except in one case, where a putative adenocarcinomatomous area was found together with the squamous cell carcinoma. Malignant mesenchymal proliferation could be diagnosed as osteosarcomas because, in all of them, the typical lacelike structures of osteoid matrix could be observed between the pleomorphic neoplastic cells. Immunoshistochemical analysis confirmed the histological findings: carcinomatous areas were positive for cytokeratin while sarcomatous areas were positive for vimentin. In the malignant mesenchymal areas, there was a low expression of α-actin and, in dog 4, positive immunolabeling for protein S-100 in the chondroid foci. The existence of chondroid S-100-positive tissue in osteosarcomas has been described. 12 Neither the coexpression nor the expression of the other antigens studied was found in any area of the neoplasms. Thus, histologic and immunohistochemical analysis led us to define the four neoplasms as carcinosarcomas, thus suggesting a new preferential location for this unusual tumor in the dog.
Immunohistochemical analysis was necessary for the accurate diagnosis of the studied carcinosarcomas. Dogs 1 and 2 showed a poorly discernible carcinomatous component, comprised of small polyhedral cells between sarcomatoid pleomorphic cells; in dog 3, the nest-type distribution and the characteristics of the cell cytoplasm suggested an epithelial origin, but immunohistochemistry pointed to a mesenchymal component.
The histogenesis of carcinosarcomas remains uncertain and there are several hypotheses to explain their origin. The convergent or multiclonal theory suggests separate mesenchymal and epithelial origins from two or more stem cells; thus, this theory points to the idea that carcinosarcomas might be the result of true collision tumors. Dog 4, where malignant epithelial and mesenchymal populations are relatively well separated, could be an example of this multiclonal theory. However, the most likely hypothesis is the divergent or monoclonal theory, which describes tumors as developing as a result of the ability of totipotential neoplastic cells to undergo multiple pathways of terminal differentiation into histologically recognizable epithelial and mesenchymal elements. In the study by Thompson et al., 13 the authors evaluated clonality in both malignant epithelial and mesenchymal elements using the X-chromosome inactivation technique. In all six dogs studied, the tumor cells from both tumor components showed monoclonality and clonal identity. The closely admixed growth of both malignant populations in dogs 1–3 of our study suggests a monoclonal histogenesis. However, immunohistochemistry is not enough to confirm the histogenesis of the studied neoplasms. A third way that might be considered is sarcomatous transformation induced in the stroma by a carcinoma. 3, 5 This phenomenon could start as a desmoplasia but would lead to the development of a mesenchymal malignant proliferation. The coexpression of cytokeratin and vimentin has been observed in some tumor populations of this type, but this fact could not be demonstrated in any of the carcinosarcomas in our study.
Mammary gland carcinosarcomas are malignant but not very aggressive neoplasms and postsurgical survival is relatively long: mean 18 months. 8 Metastases are of mixed, sarcomatous or carcinomatous type and, in some studies, have been reported as being very frequent. 4 Extramammary carcinosarcomas are very rare, but metastases have been reported in lumbosacral carcinosarcomas, 9 lung carcinosarcomas, 11 and thyroid carcinosarcomas, 7 which confirms the highly malignant nature of these tumors. In our study, no data were available concerning the clinical evolution of the tumors, except in dog 4, where the very aggressive and rapid growth led to the owners to ask for the animal to be put down 4 weeks after diagnosis. After necropsy, no metastases were found in the regional lymph node or the other organs.
In the differential diagnosis, it is necessary to include the mixed apocrine tumor, characterized by a malignant proliferation of the epithelial and/or myoepithelial populations, with chondroid or osseous metaplasia from myopepithelial cells; in this case, both populations are positive for cytokeratin, while, in addition, myoepithelial cells are also positive for vimentin and α-actin. The pseudosarcomatous stroma of these tumors show minimal reactive-type atypia; atypical mitotic figures are absent and there is none of the lacelike osteoid matrix seen in malignant bone tumors. The spindle cell carcinoma is also morphologically similar to carcinosarcomas, although in this case, the neoplastic cell population is positive for cytokeratin. 8 Finally, teratomas are derived from all embryological layers and the presence of abundant histologically benign epidermoid elements with dermal adnexal structures is usually a distintive feature. This pattern was not observed in the carcinosarcomas.
In conclusion, our study suggests a new preferential location for carcinosarcomas in old dogs beside the well-known location in the mammary gland. Our study also demonstrates that an immunohistochemical analysis is always necessary to establish an accurate diagnosis.
Footnotes
Acknowledgements
We thank Juan Sanchez and the technical staff of Histovet (St Quirze del Valles, Barcelona) for their excellent assistance.
