Abstract
Pododermatitis is frequent in captive flamingos worldwide, but little is known about the associated histopathologic lesions. Involvement of a papillomavirus or herpesvirus has been suspected. Histopathologic evaluation and viral assessment of biopsies from 19 live and 10 dead captive greater flamingos were performed. Selected samples were further examined by transmission electron microscopy and immunohistochemistry. Feet from 10 dead free-ranging greater flamingos were also evaluated. The histologic appearance of lesions of flamingos of increasing age was interpreted as the progression of pododermatitis. Mild histologic lesions were seen in a 3-week-old flamingo chick with no macroscopic lesions, and these were characterized by Micrococcus-like bacteria in the stratum corneum associated with exocytosis of heterophils. The inflammation associated with these bacteria may lead to further histologic changes: irregular columnar proliferations, papillary squirting, and dyskeratosis. In more chronic lesions, hydropic degeneration of keratinocytes, epidermal hyperplasia, and dyskeratosis were seen at the epidermis, as well as proliferation of new blood vessels and increased intercellular matrix in the dermis. Papillomavirus DNA was not identified in any of the samples, while herpesvirus DNA was seen only in a few cases; therefore, these viruses were not thought to be the cause of the lesions. Poor skin health through suboptimal husbandry may weaken the epidermal barrier and predispose the skin to invasion of Micrococcus-like bacteria. Histologic lesions were identified in very young flamingos with no macroscopic lesions; this is likely to be an early stage lesion that may progress to macroscopic lesions.
Greater flamingos (Phoenicopterus ruber roseus) are kept in captivity worldwide, but especially in Europe (65% of 139 International Species Inventory System [ISIS] member institutions), with a total of approximately 5500 birds. 18 Pododermatitis was the reason for euthanasia or a secondary finding in 95% of the flamingos (18/19) necropsied between 2000 and 2011 at the Institute of Animal Pathology, University of Bern, Switzerland (F. Wyss, unpublished survey, 2011). Corroborating these findings, a survey of the European and North American flamingo population showed varying degrees of gross plantar foot lesions in up to 100% of captive flamingos examined. 28
Many types of birds, including raptors, penguins, water fowl, and cockatiels, develop pododermatitis. 12,14 Wild birds are rarely affected. 9,16,20 This was supported by the evaluation of free-ranging greater flamingos in the Camargue, France, which did not show any foot lesions. 41 Factors influencing the level of pododermatitis have been evaluated mainly from poultry farm data, where nutrition 6,26 and substrate 2,24,25,27 were identified as contributing factors. The types of flooring in water ponds and dietary zinc availability have been identified as possible factors in development of pododermatitis in flamingos. 41 –43
Pododermatitis in turkeys is characterized by the presence of hyperkeratosis and erosion or ulceration of the sole. 3 Excess pressure on the feet of pet birds may result in similar lesions. 32 In most of the avian patients, the classic bumblefoot lesion (nodular lesion with central ulceration) is called pododermatitis, but additional types of foot lesions were described in flamingos: hyperkeratosis (slight overgrowth/marked overgrowth), fissures (superficial/deep), nodular lesions (without ulceration/with ulceration), and papillomatous growths (finger-like/cauliflower-like). 28,41
Previous evaluation of papillomatous lesions revealed some similarities to virus-induced papillomata of dogs and cats (N. Robert, personal communication, 2009), and thus papillomavirus involvement was suspected. 34 Supporting this hypothesis, virus-like particles were found in a Northern gannet (Morus bassanus) with proliferative pododermatitis 4 and in multiple waterfowl species with verrucous hyperkeratotic lesions. 44 Proliferative foot lesions caused by herpesviruses were found in macaws and cockatoos as well as in a duck with ulcerative foot lesions. 36,40 Mycotic pododermatitis was found in turkey poults. 33 Several etiologic agents have been associated with avian pododermatitis, but their involvement in the lesions in flamingos is unknown.
Flamingos have been kept in Zoo Basel since 1933. Currently, the group consists of around 100 adult greater flamingos (P. ruber roseus) and the offspring (20–25 per year), which are usually transported to other institutions in winter. Foot problems have been recognized for over 10 years, and lesions may have become more severe in recent years (A. Studer-Thiersch, personal communication, 2009). Bacteria were isolated from skin scrapings of several flamingos in the group, identified as Arsenicicoccus dermatophilus, a novel Micrococcus-like bacterium, similar to Dermatophilus sp. 10
The aim of this study was to investigate the histologic lesions of flamingos affected and unaffected with macroscopic pododermatitis using samples from captive and free-ranging birds. Molecular biologic tests for viruses, immunohistochemistry, and transmission electron microscopy were performed on individual samples to identify infectious agents.
Materials and Methods
Sample Collection
Feet of 19 different greater flamingos (14 adult, 1 subadult, 4 juvenile) from Zoo Basel were biopsied during annual routine health checks and diagnostic examinations. Punch biopsies (4 mm in diameter; Biopsy Punch, Stiefel Laboratories, Medical Solution Gmbh, Hünenberg, Switzerland) were collected from 9 flamingos with lesions, generally papillomatous. Bleeding was stopped by closing the wound with absorbable suture material (Vicryl Plus 4-0; Ethicon, Johnson & Johnson, St-Stevens-Woluwe, Belgium). Bleeding was considerable in some individuals, so only finger-like proliferations were sampled from a further 10 cases and bleeding was very mild. Samples were halved longitudinally, so that each sample consisted of all epidermal and dermal layers sampled; half was frozen at –80°C for virological assessment (n = 19) and the rest immediately fixed in formalin 4% (n = 17) (Formafix, Hittnau, Switzerland).
The research institute Tour du Valat (Arles, France) collected 20 feet from 10 adult free-ranging greater flamingos found dead in the Camargue (South France). The feet were stored frozen at –20°C and sent to the first author (F.W.) in February 2011 with the import permit (no 504/11) and CITES permits issued in Switzerland (no 455/11) and France (no FR1101300014-E). The feet were thawed and fixed in 4% formalin.
Postmortem tissues from the feet of 10 juvenile captive greater flamingos (Table 1) that died or were euthanized in 2010 and 2011 were sampled (Zoo Basel, n = 9; Tierpark Dählhölzli, Bern, n = 1). Compared with adult flamingos, 4 had no gross lesions and 6 had mild lesions only. All samples were fixed in 4% formalin, and 1 sample from Zoo Basel was stored at –80°C for virological assessment.
Gross Lesions and Scoring of Defined Histologic Changes in the Feet of Dead Juvenile Captive Flamingos From 2 Institutions in Switzerland, 2010–2011.
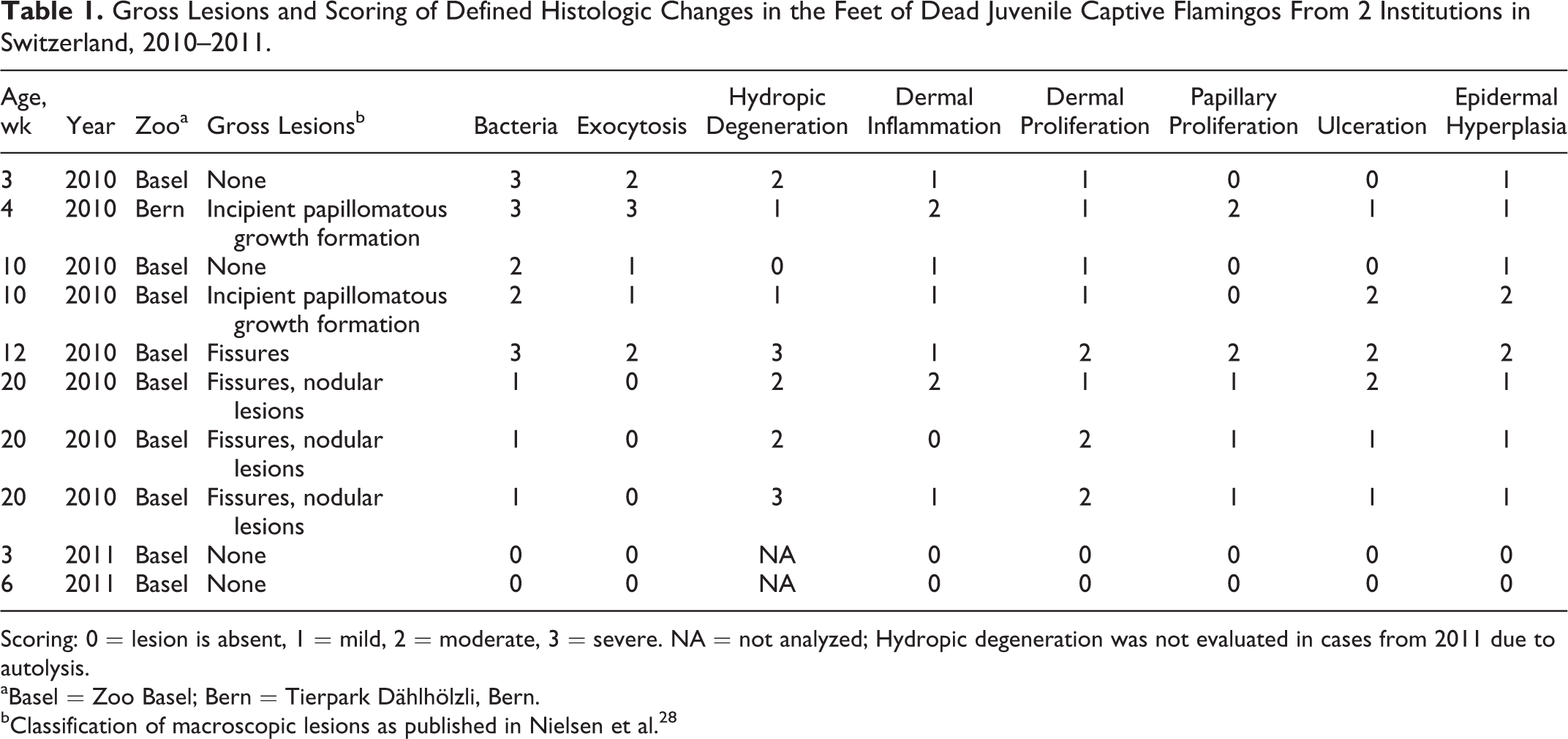
Scoring: 0 = lesion is absent, 1 = mild, 2 = moderate, 3 = severe. NA = not analyzed; Hydropic degeneration was not evaluated in cases from 2011 due to autolysis.
aBasel = Zoo Basel; Bern = Tierpark Dählhölzli, Bern.
bClassification of macroscopic lesions as published in Nielsen et al. 28
Histopathology
All histopathologic samples (biopsies of live captive animals and samples of free-ranging and captive flamingo carcasses) were embedded in paraffin according to standard protocols. From feet with lesions, the plantar skin at the base (tarsometatarso-phalangeal joint), as well as second, third, and fourth digit, was sampled from each foot separately (8 slides in total), with at least 1 piece from the proximal and 1 from the distal region of the toe. From feet without lesions, the plantar skin at the base (tarsometatarso-phalangeal joint), as well as second, third, and fourth digit from both sides, was sampled together (4 slides in total) (the prevalence of lesions was previously shown to be similar on the right and left foot). 28 Sections of a thickness of 5 μm were cut and stained with hematoxylin and eosin. For further examination, sections were also stained with periodic acid–Schiff and Gram stain (Stirling’s modification of Gram stain).
Histologic lesions from juvenile dead flamingos were scored from 0 (absent) to 3 (severe) (Table 1). Samples were assessed for the number of affected cells and number of bacteria, but also for the number of affected locations throughout the 4 or 8 slides (depending on the presence or absence of macroscopic lesions). Slides were scored independently by 2 authors (V.S. and F.W.).
Virological Assessment
To assess the presence of viral DNA, biopsy samples of 19 live flamingos taken in December 2009 and 1 dead flamingo were tested at the Institute of Virology (Vetsuisse Faculty, University of Zurich, Zurich, Switzerland). Total DNA was extracted using a QIAamp DNA Mini Kit (Qiagen, Hombrechtikon, Switzerland) following the manufacturer’s tissue protocol. Three different polymerase chain reaction (PCR) assays using broad range primers were used to test for the presence of papillomavirus DNA: FAP59/FAP64, CP4/CP5, and AR-E1F2/AR-E1R9. 8,17,21,23 A pan-herpes herpesvirus PCR using degenerate primers targeting a highly conserved region of the DNA polymerase gene was conducted to test for herpesvirus DNA. 7 PCR products were analyzed on 2% agarose gels. Amplicons in a size range between 200 and 300 bp were excised from the gel using the QIAquick Gel Extraction Kit (Qiagen). The extracted DNA fragments were also PCR amplified with specific primers. 37 The PCR products were purified with the QIAquick PCR Purification Kit (Qiagen) and sequenced to determine their characteristics (Microsynth, Balgach, Switzerland) by cycle sequencing using an ABI 377 sequencer (Applied Biosystems/Invitrogen, Basel, Switzerland). The resulting sequences were compared with published sequences from Genbank by the NCBI Basic Local Alignment search tool (NCBI database, http://blast.ncbi.nlm.nih.gov/Blast.cgi).
Transmission Electron Microscopy
Four biopsy samples from live flamingos at Zoo Basel were examined by transmission electron microscopy to screen suspicious areas for viral particles. Three samples were from papillomatous lesions, and the last was taken near a fissure. The areas of interest were identified from paraffin sections. Using the paraffin slides for reference, corresponding tissue regions were identified on the cut surface of the paraffin blocks, and appropriate samples were excised. The collected tissue samples were deparaffinized by melting for 1 hour at 60°C and washed twice with Histoclear (Chemie Brunschwig, Basel, Switzerland) and twice with 100% ethanol for 15 minutes each. Samples were rehydrated through a descending ethanol series (94%, 90%, 80%, 70%, 50%, and distilled water for 15 minutes each) and transferred to 0.1 M cacodylate buffer (pH 7.4; 3 times for 10 minutes each). Tissue samples were postfixed with 1% OsO4 (Chemie Brunschwig) in 0.1 M cacodylate buffer overnight at 4°C and embedded in Epon (FLUKA, Buchs, Switzerland) using acetone as a transitional solvent. Epon was polymerized for 3 days at 60°C. Semithin sections 1 μm thick were produced with diamond knives (Diatome, Biel, Switzerland) and stained with toluidine blue (Merck Chemicals, Zug, Switzerland). Resin blocks were trimmed to narrow down the regions of interest, and ultrathin sections exhibiting silver interference (50–60 nm in thickness) were produced with diamond knives (Diatome) on a Reichert-Jung Ultracut E (Leica, Heerbrugg, Switzerland). Sections were collected on collodion-coated 200-mesh copper grids (Electron Microscopy Sciences, Hatfield, PA). Sections were double stained with 0.5% uranyl acetate for 30 minutes at 40°C (Sigma-Aldrich, Steinheim, Germany) and 3% lead citrate for 10 minutes at 20°C (Laurylab, Saint Fons, France) in an Ultrastain (Leica, Vienna, Austria) and examined in a Philips CM12 transmission electron microscope (FEI, Eindhoven, Holland) at an accelerating voltage of 80 kV. Micrographs were captured with a Mega View III camera using the iTEM software (version 5.2; Olympus Soft Imaging Solutions GmbH, Münster, Germany).
Immunohistochemistry
Five samples were tested for the presence of papillomavirus antigen by immunohistochemistry using a polyclonal rabbit antiserum against bovine papillomavirus type 1 on paraffin cut sections (Prairie Diagnostics Services, Saskatoon, Canada). Four of the samples were obtained by biopsy from cauliflower-like papillomatous growths, and one was taken from the foot of a dead flamingo with fissures. Canine cutaneous papillomas were used as positive controls.
Results
Histology
Histologic but not macroscopic lesions were detected in a 3-week-old flamingo chick from Zoo Basel. The youngest animal examined showing macroscopic lesions was a 4-week-old flamingo chick from Tierpark Dählhölzli. There was a good consensus between the 2 examiners (V.S. and F.W.) for scoring the histologic lesions, with only a few minor discrepancies (±1 point).
Lesions of juvenile dead flamingos were characterized by Micrococcus-like bacteria invading the stratum corneum. Microorganisms exhibited zoospores and segmented and branching pseudohyphae (PAS negative, Gram positive). Epidermal lesions of exocytosis (mainly heterophils), serocellular crust formation (collections of degenerated heterophils and proteinaceous fluid in the stratum corneum), and hydropic degeneration of keratinocytes, mainly in the stratum granulosum, were observed in association with the bacteria. Epidermal hyperplasia, orthokeratotic dyskeratosis, and ulceration were not directly associated with bacteria but seen in most of the samples. Dermal lesions included accumulation of heterophils and lymphocytes, neovascularization, and increased intercellular matrix and were also noticed in areas not directly associated with bacterial invasion. In addition, irregular columnar proliferations of the epidermis and dermis with papillary squirting and dyskeratosis were seen. Examples of these observations and the scoring of some defined lesions in relation to age and macroscopic lesions are shown in Figures 1 to 5 and summarized in Table 1, respectively.
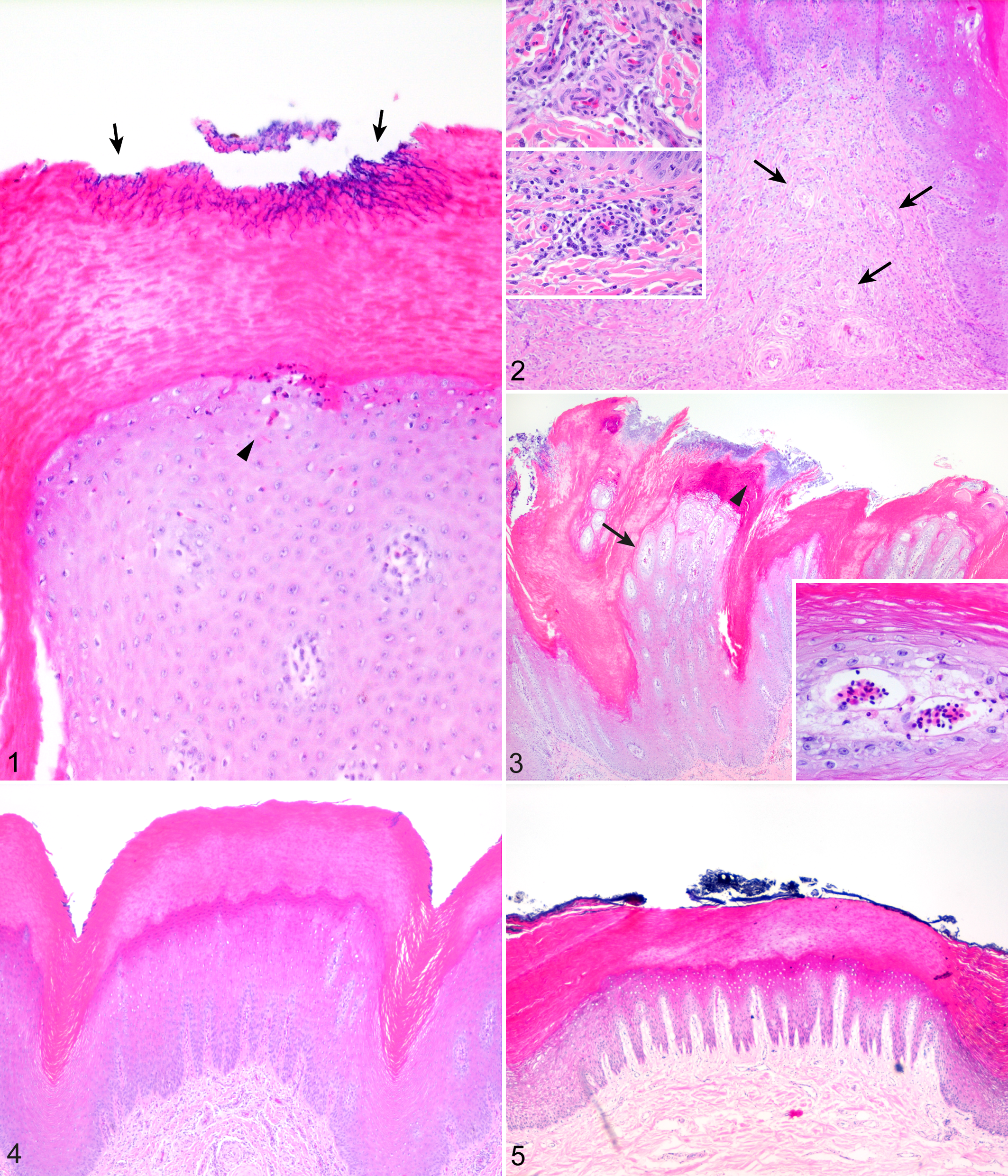
Pododermatitis, captive juvenile greater flamingo, epidermis and/or dermis of the plantar foot.
Biopsies taken from older live flamingos displayed the histologic appearance of various macroscopic lesions. 28 Micrococcus-like bacteria were very rare in biopsies taken from adult animals. Cauliflower-like papillomatous growths (Fig. 6) were composed of multiple columnar proliferations of epidermis and dermis with degenerated heterophils or serum lakes at the surface (Fig. 7).
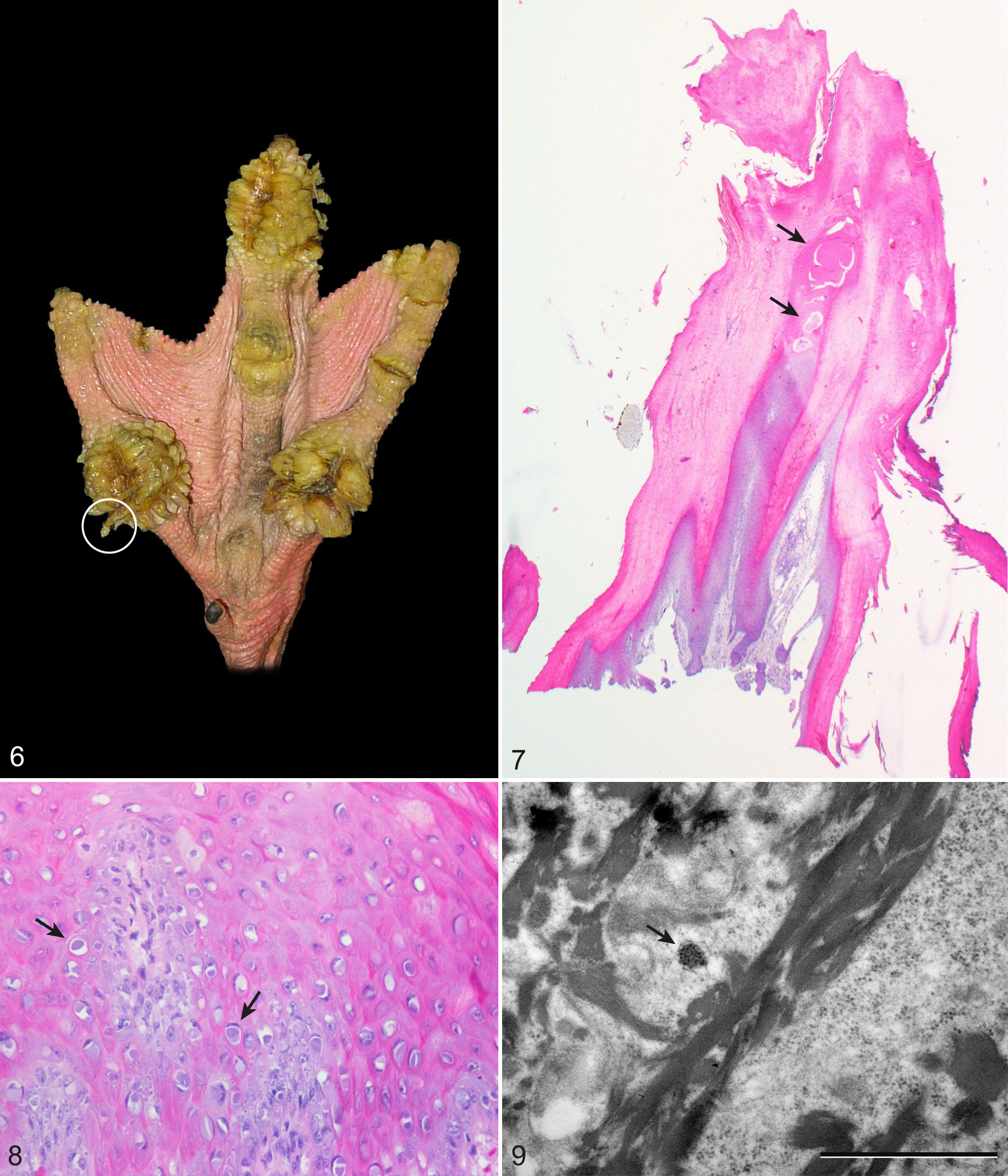
Pododermatitis, captive greater flamingo.
In three 5-month-old dead flamingos from Zoo Basel, multiple basophilic intracytoplasmic inclusion-like particles were detected in keratinocytes with hydropic degeneration (Fig. 8). However, electron microscopic evaluation of these areas revealed no evidence for any viral particles.
No macroscopic lesions were detected in the feet of free-ranging flamingos from the Camargue, in which a thinner epidermis but a similar thickness of the stratum corneum (Fig. 5) compared with flamingos from Zoo Basel was noted (Fig. 4).
Viral Assessment
PCR assays for herpesvirus and papillomavirus DNA were performed in 20 samples obtained from feet of flamingos from Zoo Basel. No evidence for the presence of papillomaviruses was found. However, herpesvirus DNA of an unknown strain was amplified in 3 samples: a 3- and a 9-year-old flamingo with a cauliflower-like papillomatous growth and a 5-year-old flamingo with several fissures. The herpesvirus DNA polymerase was 69% identical to gaviid herpesvirus 1. 29
Transmission Electron Microscopy
Four samples of flamingos from Zoo Basel were examined using transmission electron microscopy. Three were biopsies of cauliflower-like papillomatous growths (4, 19, and 54 years old), and one was collected at necropsy from a 4-year-old with fissures. Spherical intracytoplasmic inclusions (120–140 nm in diameter) were observed in occasional keratinocytes from 2 cases with papillomatous growths (4 and 54 years old). The cytoplasm contained about 20 or more regularly spaced round granules of 20 nm in diameter (Fig. 9).
Immunohistochemistry
Five adult flamingos with papillomatous lesions from Zoo Basel were tested negative for bovine papillomavirus type 1 using immunohistochemistry.
Discussion
This is the first detailed histopathologic characterization of pododermatitis in greater flamingos. Foot lesions were apparent in captive but not in free-ranging individuals. Our results suggest that histologic lesions develop in very young flamingos, when the skin still appears macroscopically unaffected. Bacterial invasion and inflammation were seen particularly in young birds, with development to more degenerative and proliferative lesions with age. Biopsies of live adult flamingos taken from macroscopically visible papillomatous lesions showed even more pronounced columnar proliferations mostly with serum lakes at the top (Fig. 9). The hypothesis that a papillomavirus is responsible for the papillomatous lesions could not be confirmed.
Small sample size and wide age range preempted statistical analysis. Samples from the 2 flamingo chicks examined in 2011 were autolyzed because they had been predated and their bodies not recovered for several days postmortem. Nevertheless, no invading bacteria and an intact epidermis were noted. Samples obtained from free-ranging flamingos from the Camargue were stored frozen before fixation in formalin, causing freezing artifacts. However, the thickness of the stratum corneum and the epidermis could be compared with the feet of captive flamingos.
Micrococcus-like bacteria invading the stratum corneum and exocytosis of heterophils into the epidermis were detected in all juvenile flamingos examined, except the 2 from 2011. Exocytosis was suspected to be a direct acute reaction to bacterial invasion. Bacterial invasion and exocytosis appear to be early histologic signs of pododermatitis. The number of bacteria and the number of affected locations decreased with the age of the animals, while hydropic degeneration of keratinocytes and dermal inflammation with heterophils and lymphocytes, neovascularization, and increased intercellular matrix were more prominent with age and may reflect chronicity. Negligible numbers of Micrococcus-like bacteria and exocytosis were seen in the samples of the three 20-week-old flamingos. Dermatophilus congolensis in mammals is associated with moist environments and damaged skin, 38 but free-ranging flamingos in the Camargue are rarely seen on dry substrate. Desiccation of the skin may alter the skin barrier and may predispose captive animals to foot lesions. Suboptimal flooring or insufficient dietary zinc could be other possible predisposing factors for bacterial invasion of the feet of flamingos in this study. 41–43 Histologic examination of the feet of 2 flamingo chicks in 2011 showed neither bacteria nor an epidermal disruption. However, the bacterium A. dermatophilus was isolated from skin scrapings of all 20 chicks in 2011 at 12 weeks of age in the course of a bacteriologic study. 10 None had macroscopic foot lesions at that time point. These chicks grew up in an enclosure with water ponds filled with fine-granular sand, 42 while in 2010, the same enclosure had no sand cover on the concrete flooring. Furthermore, a different diet was fed to the chicks from 2010 and from 2011, which led to a difference in plasma zinc levels of this 2 groups, with lower levels in the juveniles in 2010. 43 The change in flooring and/or diet (higher plasma zinc levels) may have improved skin health, so while bacteria were still isolated, they were not able to invade the epidermis. This hypothesis is supported by the pathogenesis of closely related Dermatophilus sp, which are known as opportunistic bacteria and not able to infect normal healthy skin. 38
Basophilic inclusion body-like particles were found in the cytoplasm of keratinocytes of three 20-week-old flamingos. Papillomavirus particles are 45 to 50 nm in diameter, have no envelope, and are intranuclear. 31 In our samples, clusters (120 nm in diameter) of granules (20 nm in diameter) were detected; the size of these granules was not compatible with previous descriptions of papillomavirus inclusions. No evidence of papillomavirus infection was found using other methods. No viral DNA was amplified with any of the broad-range PCRs, which have previously been used to detect papillomavirus DNA in various species. 11,22 Also immunohistochemistry, which had successfully been applied to detect papillomavirus antigen in mammal and bird species before, failed to detect papillomavirus group-specific antigen in the tested samples. 5,19,39 A novel herpesvirus related to gaviid herpesvirus 1 was found in 3 of 20 samples in this study. Avian herpesviruses are ubiquitous and have been isolated from a wide variety of species. Most herpesvirus strains are host adapted and cause only mild, subclinical, or latent disease. 30 If severe disease occurs (eg, Marek’s disease in poultry, Pacheco’s disease in psittacines, or duck plague in waterfowl), signs of depression, respiratory or neurologic signs, and unexpected deaths are the most common clinical presentations. 13 Herpesviruses have been documented in proliferative foot lesions of macaws and cockatoos with species-specific variations 36 and in ulcerative lesions of a duck. 40 Low infectivity of the herpesvirus in groups of macaws and cockatoos is suspected, 30 and it seems unlikely that a herpesvirus is responsible for the infection of a whole group of flamingos. Herpesviruses form intranuclear inclusion bodies 120 to 200 nm in diameter, arranged in paracrystalline arrays. 40 Clusters detected by transmission electron microscopy in foot samples in this study were 120 nm in diameter but were intracytoplasmic and granular, not paracrystalline.
Hyperplasia of epithelial cells associated with vacuolar degeneration and cytoplasmic inclusion bodies have been found in juvenile flamingos with suspected pox virus infection. In contrast to the inclusions in our study, poxvirus inclusions were eosinophilic and twice as large (250–300 nm in diameter). 1 Location of poxvirus in flamingos was mostly confined to the face and beak. 1,35
Location or size of the inclusion bodies or clinical presentations of papilloma-, herpes-, or poxvirus are not in accordance with our findings, and therefore, these particles were suspected to represent proteinaceous material.
The larger dimension of the epidermal layers, except the stratum corneum in captive compared with free-ranging flamingos (Figs. 4, 5), may indicate epidermal hyperplasia and could be due to a compensatory mechanism to the chronic stimulus of a suboptimal substrate. 15
In raptors, it is thought that pododermatitis develops following trauma to the feet. 12 In our study, histologic lesions were detected in macroscopically normal-appearing feet. We hypothesize that proliferative lesions are reactions to the inflammation caused by A. dermatophilus 10 being able to invade a weakened skin barrier, possibly caused by factors such as flooring 41,42 and nutritional deficiencies. 43
Footnotes
Acknowledgements
We thank the technicians from the Institute of Animal Pathology, the Institute of Virology, and the Division of Veterinary Anatomy for their assistance in preparing the specimens; Dr Monika Welle for her help with the evaluation of the histologic lesions; Michelle Aimée Oesch for her help with the illustrations; and Dr Kathryn Perrin for review of the manuscript.
Author Contribution
Conception or design: FW, CW, SH, ME, MS, NR. Data acquisition, analysis, or interpretation: FW, VS, CW, SH, SG, AA, ME, MF, CL, MS, RN. Drafting the manuscript: FW, VS, ME, CL, MS. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
