Abstract
Rainbow trout gastroenteritis (RTGE) is an emerging syndrome linked to the presence of large numbers of the segmented filamentous bacterium “Candidatus arthromitus” within the intestine. The present study examined the histopathological changes of the digestive tract of 152 trout with gross lesions typical of RTGE. Histopathology showed that 129 of 152 fish (85%) affected with RTGE had segmented filamentous bacteria in the distal intestine and/or pyloric caeca. The presence and number of segmented filamentous bacteria were always significantly higher (P < .001) in pyloric caeca, thereby suggesting the preferred site for these bacteria. Histopathological changes included enterocyte detachment and congestion of the lamina propria and adventitial layers. Samples from 6 RTGE-affected trout were examined using scanning and transmission electron microscopy, revealing a close interaction of segmented filamentous bacteria with the mucosa of distal intestine and pyloric caeca, with the presence of bacterial attachment sites, and with associated morphological changes of the apical membrane of enterocytes. Despite these interactions, segmented filamentous bacteria were not always adjacent to the areas with pathological changes, suggesting that if these organisms play a role in the pathogenesis of RTGE, extracellular products may be involved. Ultrastructural changes included loss of microvillar structure, membrane blebbing, hydropic mitochondrial damage, and basal hydropic degeneration of enterocytes, which frequently resulted in disruption of tight junctions and enterocyte detachment. The resulting exposure of large areas of lamina propria probably resulted in the compromise of the host osmotic balance and the facilitation of the entry of secondary pathogens.
Keywords
Rainbow trout gastroenteritis (RTGE) was suggested in 1999 to describe a specific syndrome of rainbow trout, Oncorhynchus mykiss Walbaum. 9 This condition has been reported in Croatia, France, Italy, Spain, and the United Kingdom during the summer. 9,26,36,37 The economic impact of RTGE is significant, and daily mortalities of 0.5% to 1.0% were common during outbreaks. 9,26
The presentation of RTGE includes severe enteritis with massive accumulation of segmented filamentous bacteria (SFB) within the lower digestive system. 9,26,37 These SFB have been classified as “Candidatus arthromitus,” a group of bacteria closely related phylogenetically to Clostridium. 35,37 Trout SFB have been suggested as the etiologic agent for RTGE, although this possibility has not been confirmed, in part because of the current inability to culture SFB in vitro. 5,9,26,37 The gross presentation of RTGE has been described in several studies to include abdominal dilation (externally), and severe distal enteritis with congestion and edema of the intestinal wall has been found (internally). 9,26,37 The intestine contains a yellow and viscous fluid in which large numbers of SFB can be seen under light microscopy. 9,26,37 Histopathological changes include severe enterocyte detachment, congestion, and SFB accumulation in distal intestine and pyloric caeca. 9,26,37 Previous reports on the preferred location of SFB in the digestive system of enteritic trout vary; Michel et al 26 reported their presence throughout the digestive system, whereas Branson 9 observed SFB preferentially in the distal intestine.
Enteritides other than RTGE have been described in rainbow trout, including bacterial, parasitic, fungal, and viral conditions 6,14,31,39 although isolated enteritis is infrequent in fish and most often part of systemic disease. 14 Of all the enteritides of rainbow trout, RTGE is the only one that is reported to be associated with SFB, suggesting that this condition is a distinct disease entity despite the generic term used to describe it.
There are several reports on the pathological changes accompanying RTGE at both the histopathological level and the ultrastructural level, but most of these focused on the detection and description of SFB rather than on the host responses. No numerical data have been provided in any report to support observations of where SFB are preferentially found in RTGE-affected (RTGE+) fish. 9,26 This study aimed to contribute to these areas by further examination of SFB histopathological presentation and the use of scanning and transmission electron microscopy to complement previous ultrastructural studies on the presentation of RTGE.
Materials and Methods
RTGE Gross Case Definition
All the RTGE+ fish in this study were identified by a case definition created from previous literature and based on the gross presentation. 9,26,37 The case definition was as follows: RTGE is a condition of rainbow trout, observed in units with daily cumulative mortalities of 0.5% or more and present during the summer. Affected fish present a distended abdomen externally, whereas internally, their lower intestine is dilated, congested, and oedematous, containing a yellow viscous substance.
Fish Sampling and Sample Processing
The rainbow trout in this study were sampled from 1 June to 31 September, 2006, at 11 UK sites with RTGE presence. At each site visited, 2 groups of fish were sampled from a single productive unit (ie, cage, pond, or raceway) during an RTGE outbreak: first, as many moribund fish showing external signs of RTGE as practically possible (moribund RTGE+) and, second, 30 randomly sampled apparently healthy fish. The sample size of the latter group was chosen to enable disease detection with 95% confidence if the prevalence was at 10% or more. This strategy enabled the random detection of cohabiting fish consistent with the case definition, which were processed and included in the analysis as “subclinical” RTGE+ fish (“subclinical” RTGE+).
All fish were sampled on-site and euthanized before sampling with benzocaine (SIGMA E1501) at 250 mg/liter 3 ; furthermore, fish origin, fork length, weight, and the presence of any external or internal gross signs were recorded. Samples for histopathology were taken from 3 locations in the digestive tract: the pyloric end of the stomach (one 1-cm tubular section), proximal pyloric caeca (10 to 15 whole caeca), and distal intestine (one 1-cm tubular section). All the digestive tissues sampled were sectioned longitudinally before placement in fixative to allow rapid penetration, with the aim of minimizing artifacts caused by autolysis. 14 All histology samples were placed in 10% buffered formalin and left for at least 24 hours before processing using standard protocols and embedding in individual paraffin wax blocks. 31 Finally 5-μm sections from these blocks were cut, placed on slides, and stained with hematoxylin and eosin. Sections positive for SFB were also Gram-stained following standard protocols.
SFB Histological Presence
The digestive tissues of all fish consistent with the case definition for RTGE were assessed for the presence of SFB and histopathological changes. All the histology slides were read “blind” by the same person, and SFB were identified as bacteria approximately 0.6 to 1.2 μm in diameter and up to 60 μm in length, with apparent segmentation every 1.2 to 1.6 μm. 37 SFB presence was noted in each tissue after scanning the entire section at objective 10×, and if present, the number of SFB was recorded as the mean number of SFB counted in 3 microscopic fields at 20× (area = 0.95 mm2). The microscopic fields were systematically chosen for SFB abundance, and a value of 100 was recorded for SFB numbers equal to or exceeding 100 bacteria per field. Additionally, any histopathological changes observed were noted. Finally, the statistical differences regarding histological presence and quantity of SFB were assessed with respect to the organ (ie, distal intestine or pyloric caeca) and clinical status (ie, moribund or “subclinical”). The statistical tests included Pearson chi-square test (Pχ2) and Fisher exact (FE), where appropriate, for SFB presence and Kruskal–Wallis (KW) for SFB numbers, 12 with significance assumed under P = .05. All the tests were conducted using EpiInfo software package (Centers for Disease Control and Prevention, Atlanta, GA).
Ultrastructural Description of RTGE
Samples from 6 moribund rainbow trout with gross presentations consistent with RTGE were also processed for scanning and transmission electron microscopy (cases 1–6). These fish were sampled from a single site at 1 point, and only pyloric caeca and distal intestine samples were taken, which were incised to allow rapid fixation. The samples were processed for scanning and transmission electron microscopy following previously described protocols. 8,17
Results
In sum, 464 rainbow trout were sampled. Of these, 134 were moribund fish consistent with the case definition for RTGE (moribund RTGE+) and 330 were randomly sampled fish from the same units. During postmortem examination, consistency with RTGE case definition was recorded in 18 of the randomly sampled fish, and these fish were included in the analyses as “subclinical” RTGE+ fish, which resulted in a total number of 152 RTGE+ fish (“subclinical” + moribund) for which the analyses were performed. The remaining 312 fish were not consistent with RTGE case definition and were not included in this study.
Externally, “subclinical” RTGE+ fish presented with milder dilation of the abdomen but, internally, with all gross signs included in RTGE case definition. In 22% of the “subclinical” RTGE+ fish, the yellow viscous intestinal content was mixed with feed.
SFB Histological Presence
The mean weight of the RTGE+ fish was 218 g ±90.5 (SD). All these fish had pathological changes consistent with the case definition of RTGE both internally and externally (Figs. 1, 2 ). Microscopic examination of the yellow viscous fluid from the vent revealed large quantities of sporulating SFB in most fish (Fig. 3).
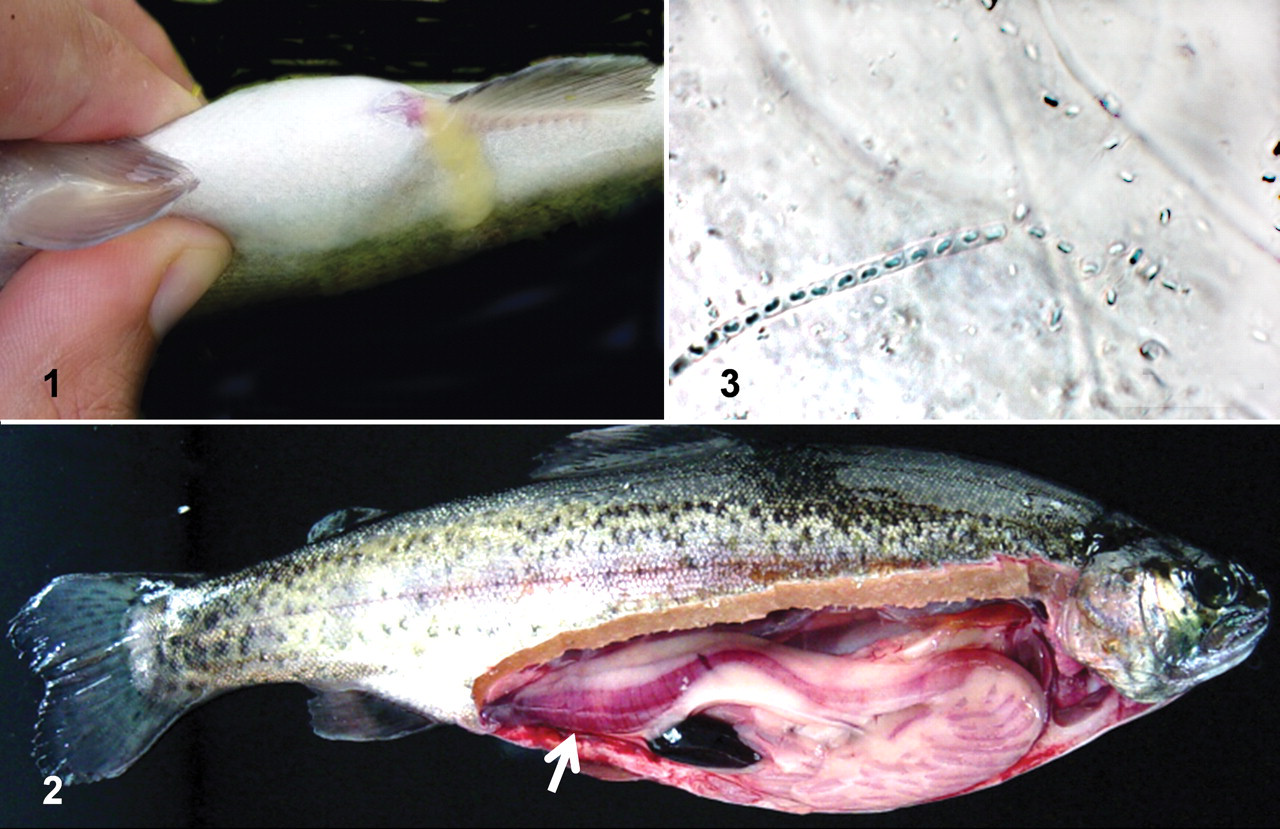
Case 1: Gross and microscopic changes in trout with rainbow trout gastroenteritis.
Histologically, it was possible to observe SFB in the pyloric caeca and/or distal intestine of 129 RTGE+ fish (of 152 [85%]), both attached to the mucosa and free within the lumen (Figs. 4, 5 ). The SFB were never observed in any of the sections of pyloric stomach. No significant differences were observed between moribund and apparently healthy RTGE+ fish in the presence of SFB (P = .70; FE) or in the SFB numbers observed (P = .40; KW). Marked differences were observed, however, depending on the organ, and SFB detection was significantly more frequent in pyloric caeca than in distal intestine of all RTGE+ fish (P < .001; Pχ2). The average number of detectable SFB per 20× field was significantly higher in pyloric caeca than in distal intestine (P < .001; KW), with SFB counts of 45.9 ± 2.9 (SE) SFB per field in the former and 6.5 ± 1.3 (SE) SFB per field in the latter. Moreover, apparent degradation of SFB was observed in the distal intestine of 10 RTGE+ fish, as evidenced by reduced stain retention and disruption of the filamentous structure (Figs. 6, 7). Gram variability of SFB was observed in the Gram-stained sections, although most SFB were Gram-positive.
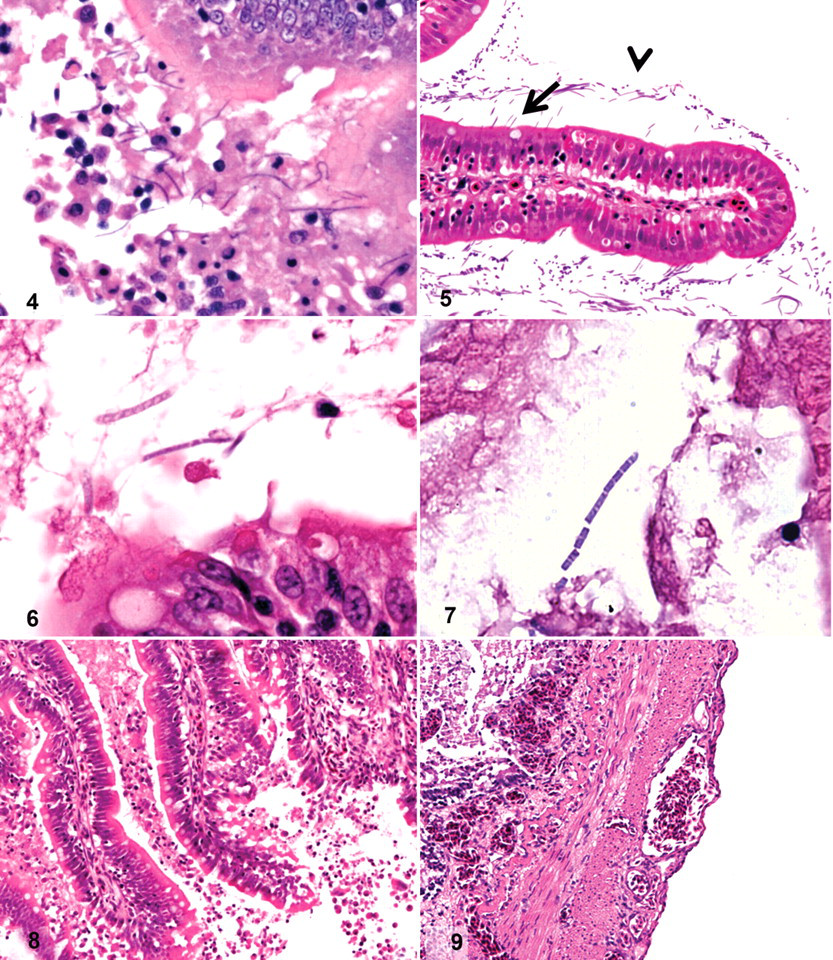
The histopathological changes observed in RTGE-positive fish included generalized enterocyte detachment (Fig. 8) and congestion of the adventitial layers and the lamina propria in distal intestine and/or pyloric caeca (Fig. 8, 9). Histopathological changes were significantly more frequent in distal intestine (P < .001; Pχ2) than in pyloric caeca.
Case 1: Histological presentation of rainbow trout gastroenteritis.
Ultrastructural Description of RTGE
Scanning electron microscopy
Large numbers of SFB were always present in distal intestine and/or pyloric caeca of the 6 RTGE+ fish sampled for electron microscopy (cases 1 to 6). These bacteria could be seen free-floating in the lumen and attached to the intestinal mucosa in both these locations (Figs. 10, 11), although epithelial-associated SFB were not ubiquitous and appeared to be restricted to specific locations. All SFB were approximately 1 μm wide, of variable length, and clearly segmented (Fig. 12). In the distal intestine, SFB occasionally had structures consistent with propagation by budding at the distal end (Fig. 13). Enterocytes frequently interacted with SFB, and these interactions resulted in changes in their apical membrane, which folded around sections of SFB and presented apparent “trails” suggesting that engulfment of SFB by enterocytes had taken place (Figs. 14, 15).
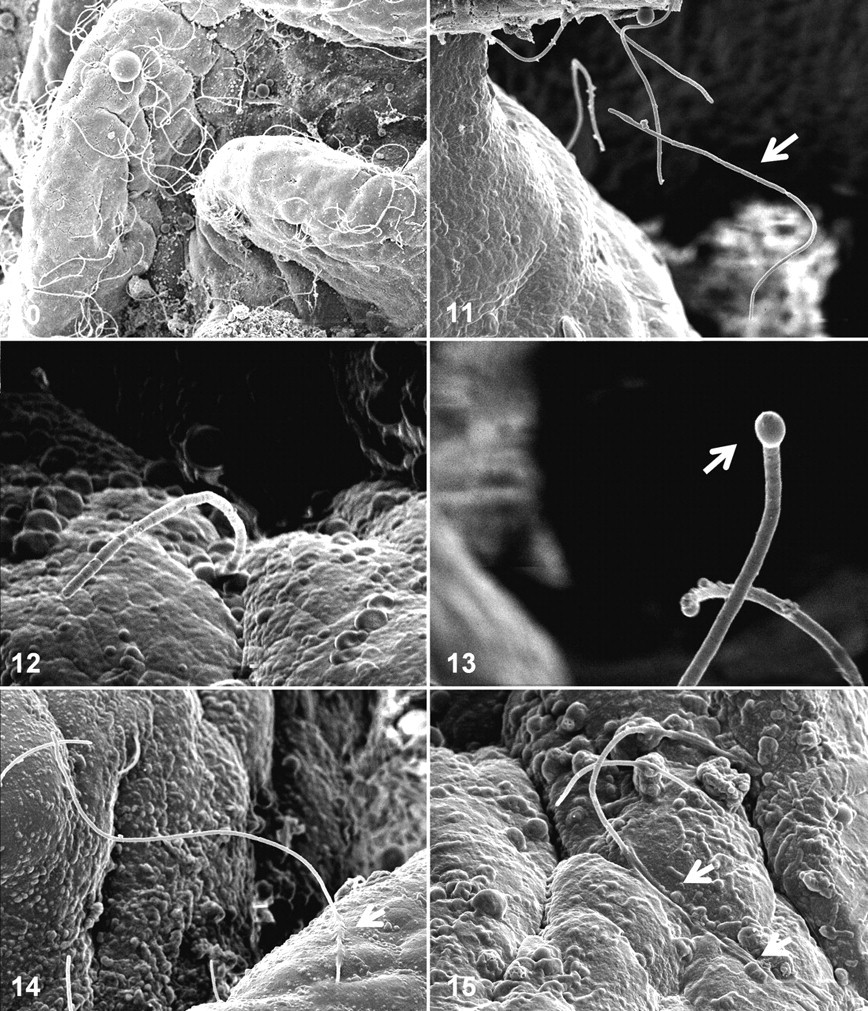
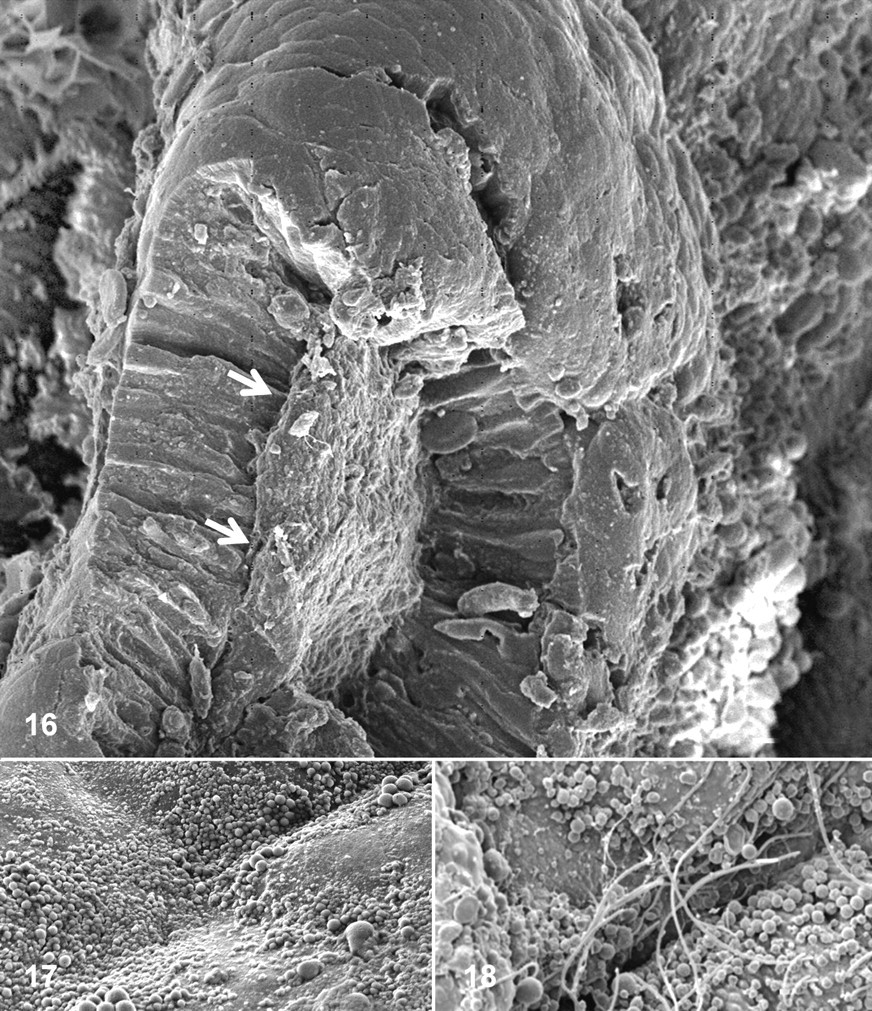
Pathological changes observed included multifocal detachment of the mucosal layer and pronounced apical blebbing of enterocytes (Figs. 16–18). The former always resulted in direct exposure of the lamina propria to the digestive lumen, whereas the latter could be associated or not with SFB proximity (Figs. 17, 18).
Transmission electron microscopy
Different developmental stages were observed within single SFB filaments, including vegetative, dividing, and sporulating stages (Figs. 19, 20 ). Loss of cellular structure resulted in spore release (Fig. 21). The proximal segment at SFB attachment sites presented a pear-shaped appearance (Fig. 22). These segments were always extracellular and surrounded by an electron-dense area within the cytoplasm of the adjacent enterocytes (Fig. 23). No other cellular reaction was observed, and no direct evidence of SFB phagocytosis was observed in transmission electron microscopy.
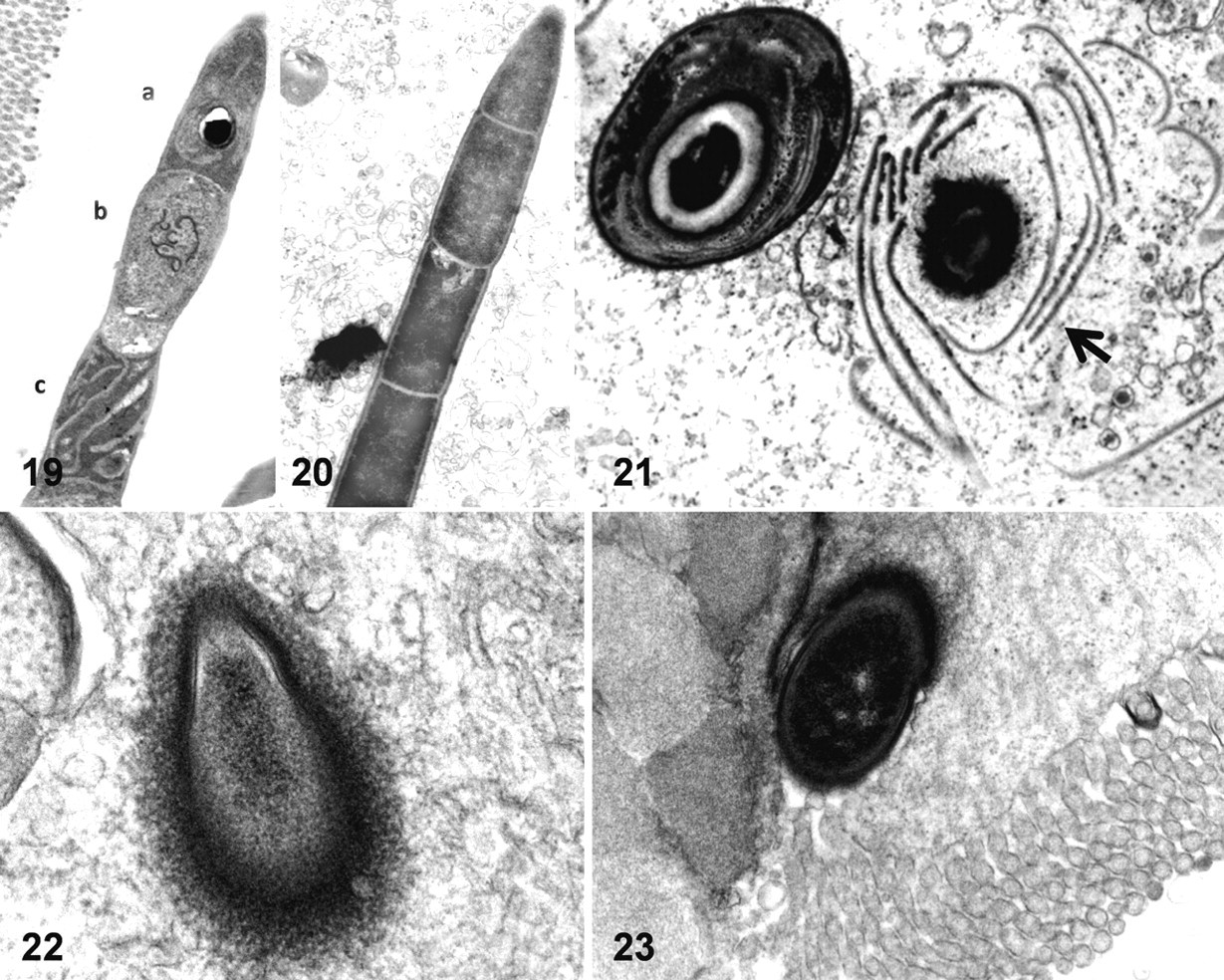
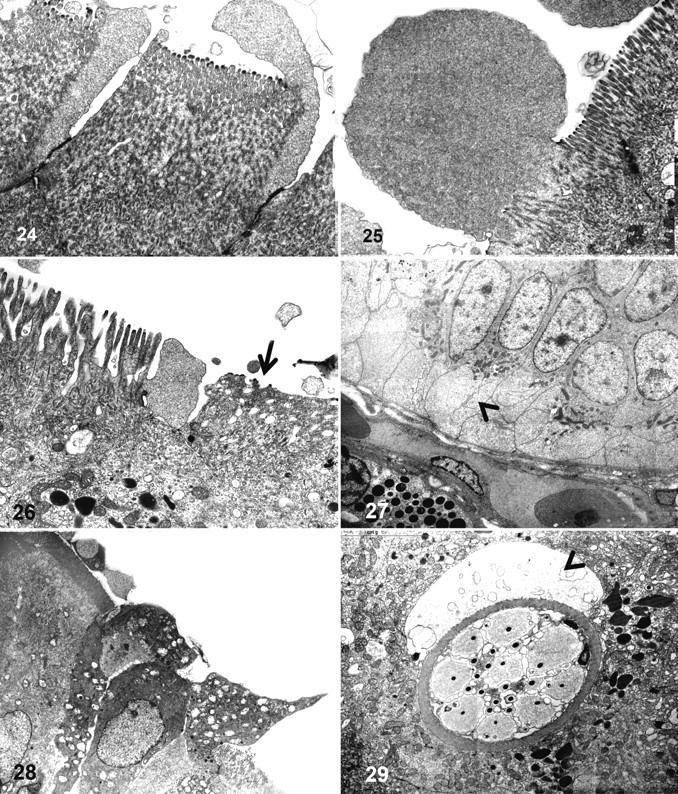
Ultrastructural changes indicative of osmotic imbalance and cytoskeletal damage were frequently observed at the apical and basal poles of enterocytes in all cases. At the apical pole, these changes included cytoplasmic membrane blebbing and loss of microvilli structure (Figs. 24–27). The presentation of membrane blebbing suggested that this occurred initially close to tight junctions and progressed to the rest of the apical pole, whereas some blebs appeared to cast off from the cell. Hydropic degeneration with cytoplasmic dilution was seen at the basal pole, and the cytoplasmic membrane appeared intact (Fig. 28). Other enterocyte lesions included hydropic degeneration of mitochondria. A large number of enterocytes had lost the tight junction integrity and were detaching from the enteric mucosa (Fig. 29). Changes in other cell types were also seen; for example, rodlet cells were abundant and always present adjacent to an area of fluid accumulation, suggesting that they may have been actively secreting (Fig. 30).
Discussion
Accumulation of large numbers of “C arthromitus” have been observed in the lower digestive tract of rainbow trout with RTGE. 9,26,37 In this study, most RTGE+ fish presented with large numbers of SFB in their lower digestive tract, detectable by histopathology or electron microscopy. By contrast with other reports, however, 26 the presence of SFB was never confirmed in any of the pyloric stomach sections examined in this study. Although the histopathological presence of SFB was significantly associated with RTGE+ fish, these microorganisms were observed in several apparently healthy controls from the same units (data not shown). This was probably a result of a generalized increase of SFB load within affected units or subclinical infection, although all these fish were actively feeding and did not present any of the gross changes associated with RTGE. No significant differences were observed in the presence and quantity of SFB between moribund and “subclinical” RTGE+ fish, but SFB were more commonly observed and in higher numbers in pyloric caeca than in distal intestine. Both these observations suggest that the pyloric caeca are the preferred site for histological detection of SFB in RTGE+ fish. Previous reports on the common location of trout SFB do vary: Michel et al 26 reported their presence throughout the digestive system, whereas Branson 9 observed SFB more frequently in the distal intestine. It is not possible to contrast our findings with these reports, because none of them included numerical data. The differences between organs were significant in our study, and it is possible that the conditions in pyloric caeca are more favorable for SFB. This possibility is supported by the observation of reduced staining and loss of structure (degeneration) of SFB noted in several distal intestines from RTGE+ fish, an observation consistent with SFB membrane impairment as previously reported. 26 Congestion and enterocyte detachment were more frequent in distal intestine, and it is possible to hypothesize that SFB degradation in the distal intestine could have coincided with sporulation, cellular toxicity, or autolysis during fixation. Also, this may have been the reason for the failure to detect these organisms histologically in 15% of RTGE+ fish. The degradation of SFB could also have a bearing on RTGE pathogenesis, given that “C arthromitus” are closely related to Clostridium 34,37 and may produce endotoxin, which would be released after cytoplasmic membrane damage. 26
All analytical methods used in this study revealed SFB both free within the lumen and attached to the digestive mucosa. On the mucosal surface, SFB appeared to concentrate in specific areas, whereas other locations were devoid of these organisms. Morphological differences between mucosal areas with and without SFB were not noted in this study, although they may exist and several authors have noted a tropism of SFB for lymphoid tissues and specific locations in other animals. Preferred locations include the ileum of mice, 11 the ileum and caecum of the chicken, 21 and the ileal Peyer’s patches of several species. 19,23,25,28,32,33 All the SFB observed in this study had an appearance consistent with previous reports, both in size, segmentation, and sporulation and in the presence of different developmental stages within a single filament. 9,26,37 There was no evidence for spore germination in either intestine or pyloric caeca, as previously observed. 26 This observation suggests that these spores may constitute the form of dispersal between hosts, as has been shown for SFB in mice. 21 There was a close interaction of SFB with the digestive mucosa of RTGE+ rainbow trout and SFB attachment sites, and apparent engulfment of SFB by enterocytes was observed. Attachment sites were similar to those described for SFB in mice and poultry, with an electron-dense area resulting from actin accumulation, implying a cell metabolic response, 19 although the nipple-like appendages reported in other species 10,11,19,40 were not observed in the distal segments of trout SFB, which presented a pearlike shape. It is possible that this absence resulted from the plane of the section, although morphological differences have been observed between SFB in different animal species, 2,16,18,22,23,32,33 and it is possible that trout SFB do not present this feature. Apparent engulfment of SFB by adjacent enterocytes was suggested by observations based on scanning electron microscope, although this was not directly confirmed by transmission electron microscopy. It is not possible to discard this possibility, and intracellular bodies structurally similar to SFB were occasionally observed in transmission electron microscopy (data not shown), but phagocytosis was never observed directly. Phagocytosis of SFB has been reported in poultry and has been related to their role in the activation of the mucosal immune system. 40
The role of SFB in the etiology of RTGE is unclear, 9,26,37 and this study failed to demonstrate a role. None of the pathological changes observed in the digestive system of RTGE+ fish were exclusively associated with closely apposed SFB, suggesting that if SFB are indeed the cause of RTGE, they are inducing pathological changes without direct contact—by toxin release, for example. 26 An etiological role of SFB has never been demonstrated in other species, and although accumulation of large numbers of SFB were associated with stunting syndrome in turkey poults, this syndrome was subsequently shown to be caused by a virus. 1,4 No inclusion bodies or viral particles were seen in the digestive system of RTGE+ fish in this or other studies.
Severe pathological changes were observed in the digestive mucosa of rainbow trout affected with RTGE, including loss of microvilli, apical blebbing, hydropic change to mitochondria, and basal hydropic degeneration. All these changes suggest cytoskeleton damage and severe osmotic imbalance at the enterocyte level. The blebs observed were consistent with zeosis; that is, their matrix was devoid of cytoplasmic organelles and filled with ribosomes only. 15 The cytoplasmic blebbing of enterocytes has been reported in other species to be associated with local ischaemia, with the presence of enterotoxin and apoptosis, 20,24,27,38 and can precede enterocyte detachment. 7,38 In RTGE+ fish, large numbers of affected enterocytes lost the integrity of their tight junctions and were shed to the enteric lumen. Mucosal loss was extensive and resulted in direct exposure of a large area of the lamina propria to the digestive lumen. This probably would have compromised osmotic balance and facilitated the entry of opportunistic pathogens, given that the digestive osmotic barrier is located at the tight junctions and is part of the passive immune system. 13 Finally, the frequent presence of rodlet cells with adjacent fluid accumulation suggests that these cells were being recruited and were actively secreting. Rodlet cells are exclusive to teleosts, and it is believed that they have a role as part of the host response to tissue damage, stress, and noxious agents, including parasites, toxic metals, and acid exposure. 29,30 It is therefore possible that rodlet cell recruitment and activity may be a common feature of fish enteritis, although there are no reports in this respect. Degranulation of eosinophilic granular cells was not seen in RTGE fish, in line with reports of an independent action of these two cell types. 29
Footnotes
Acknowledgements
We would like to acknowledge the managers of the sites involved for their time and help and Lynton Brown for the processing of electron microscopy samples.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
This research was supported by the Scottish Aquaculture Research Forum; the Department of Environment, Food and Rural Affairs; and the British Trout Association.
