Abstract
Humans and guinea pigs are unable to produce vitamin C, with deficiency resulting in a well-known disorder of collagen synthesis. Pial basement membrane structure preservation is essential in the proper migration of neurons. In our study, intrauterine deprivation of vitamin C in guinea pig fetuses led to a collagen synthesis disorder, weakness, and finally a breach of pial basement membrane. We found excessive migration of the external germinal layer cells into the subarachnoid space of the cerebellum through defects in the pial basement membrane. The changes ranged from focal rupture of pial basement membranes to their complete disintegration. The loss of proper folia formation resulted in macroscopically visible flattening of the cerebellar surface. Different grades of dysplastic changes in the folia of the cerebellar cortex were observed in 2 experimental groups assigned different limits to mark the time of commencement and duration of vitamin C deprivation. The most severe form of dysplastic changes was characterized by marked irregularity of the cerebellar cortex similar to that in lissencephaly type II. Thus, prenatal vitamin C deficiency represents a novel animal model to study the effects of collagen synthesis on development of breaches in the pial basement membrane, disordered migration of neurons, dysplasia of cerebellar cortex, and the pathogenesis of lissencephaly.
Lissencephaly type II is a disease caused by alteration to the pial basement membrane (PBM) proteins, which causes it to weaken and become porous. This causes a deficiency of the glia limitans superficialis and radial glial cell connectivity. As a result, migrating neural cells pass through the pial gaps to form cell ectopia near the brain surface. This leads to loss of gyri and sulci, forming irregularities on the brain surface and a so-called cobbled formation, which is why lissencephaly type II is also sometimes called cobblestone lissencephaly. 9,34
Based on the fact that the structural integrity of the PBM is necessary for proper migration of the neurons, 2 main approaches have been established for experimental modeling of lissencephaly type II. First, animal models with targeted inactivation of genes involved in membrane protein synthesis have been studied. The lack of these proteins, such as collagen, laminin, and others, causes the PBM to weaken and finally rupture. 13,16,17,25,43 The second approach uses chemical destruction of meningeal cells over the developing cerebellum through exposure to 6-hydroxydopamine (6-OHDA). The loss of glio-pial membrane allows for overmigration of neurons through the pial breaches into the subarachnoid space. 1,29,30
Collagen is one of the key structural units of the basement membrane, and its synthesis is dependent on vitamin C. Humans, monkeys, and guinea pigs are unable to synthesize vitamin C because of a lack of L-gulono-γ-lactone oxidase. 27 It is well known that a deficiency of vitamin C in guinea pigs impairs the expression of type IV collagen, elastin, and laminin in blood vessels. 10,23
Guinea pigs are precocial rodents in that their brain development occurs predominantly during the intrauterine phase. 21 Our hypothesis was that the fetal vitamin C deprivation during cerebellar foliation would cause reduced collagen synthesis, consequentially weakening and rupturing the PBM to produce the dysplastic changes consistent with lissencephaly type II.
Materials and Methods
Animals and Environment
We studied albino guinea pigs (Cavia porcellus), aged between 4 and 6 months, obtained from the Department of Biochemistry, Faculty of Medicine, Novi Sad. The experiments with these animals were approved by the Ethics Committee of the University of Novi Sad (No: 01-160/6-2; EK: III-2013-02). Animals were kept in 400 (wide) ×1000 (long) × 300 (high)–mm plastic containers in a harem system: 3 or 4 females per male. Artificial cycles with 12 hours of light (08:00–20:00) and 12 hours of dark were provided. The ambient temperature was 23°C, and air was fully circulated 6 to 10 times per hour.
Inspection of the female vaginal introitus was performed on a daily basis. The presence of spermatozoids in a vaginal smear was designated the first day of gestation and at that time the dams were housed separately from males. Gestational length in guinea pigs (C. porcellus) can vary between 62 and 68 days. 26
The experimental study included 21 female animals that maintained pregnancy until they were euthanized. These were randomly divided in 3 groups: a control group, K (n = 7), and 2 experimental groups, E1 (n = 7) and E2 (n = 7). Experimental animals that were unable to achieve or maintain pregnancy during the experiment (0 in K, 1 in E1, and 9 in E2 group) were excluded from the study and replaced with new dams.
Experimental Protocol
All animals were sustained with a standard vitamin C–free commercial pellet diet. Control animals were supplemented with ad libitum water enriched with vitamin C (50 mg/100 ml water). Until the day of vitamin C restriction (see below), females in the experimental groups had the same diet as the females from the control group. Vitamin C deprivation began on the 20th day of gestation for group E1 and on the 10th day for group E2. Vitamin C deprivation continued for both E1 and E2 groups until the 50th day of pregnancy, when the animals were euthanized.
The experimental design was based on 2 facts. First, the justification for euthanasia on the 50th day of gestation was based on our pilot study in which the first signs of cerebellum foliation could be spotted on the 35th day of gestation and that the formation of all 10 cerebellum lobules were developed by the 45th day of gestation. Second, the duration of vitamin C deprivation was based on the fact that the first symptoms of deficiency in guinea pigs appeared after 3 to 4 weeks of vitamin C deprivation. 23,28
Perfusion Fixation
On the 50th day of gestation, fetuses were removed by caesarean section under urethane anesthesia. Perfusion fixation was performed through cannulation of the ascending aorta while simultaneously incising the right atrium to allow the egress of blood and perfusate. Then, 30 to 60 ml of buffer (0.27 mmol/l NaCl, 44 mmol/l dextrose, 47 mmol sucrose, 4.2 mmol/l CaCl2, 3.1 mmol/l Na-cacodylate, pH 7.4) was infused, followed by a ∼150-ml infusion of Zamboni’s fixative. Fetuses were weighed after perfusion. After fixation, the central nervous structure (cerebrum, cerebellum, medulla oblongata, and spinal cord), the eye, and the slice of muscle tissue from the upper limb were removed from the bodies and immersed in a fixative for 24 hours. For the purposes of this study, we are only presenting the data derived from the cerebellum.
Immunohistochemistry
For immunohistochemical analysis, we cut a 5-μm-thin midsagittal section from the region of the cerebellar vermis. After appropriate dehydration, the section was embedded in paraffin (Histowax, the Netherlands) and cut on a rotary microtome (Leica, Germany). Slides were routinely stained with hematoxylin and eosin (HE). Methods for immunohistochemical staining included primary antibodies: mouse anti–collagen IV Ab-3 in a 1:50 dilution (Lab Vision; Thermo Scientific, Rockford, IL), mouse anti–Ki-67 Ab-4 in a 1:100 dilution (Lab Vision; Thermo Scientific), and mouse anti-GFAP in a 1:100 dilution (Lab Vision; Thermo Scientific) using the appropriate visualization system: UltraVision LP Detection System HRP Polymer & AEC Chromogen (Lab Vision; Thermo Scientific) and UltraVision LP Detection System HRP Polymer & DAB Chromogen (Lab Vision; Thermo Scientific).
Ki-67 and GFAP immunostaining required antigen retrieval using citrate buffer (pH 6.0) in a microwave oven at 850 W for 20 minutes. Collagen IV immunostaining required antigen retrieval using protease XXV (Lab Vision; Thermo Scientific) for 10 minutes at 37°C. All the antibodies were applied for 30 minutes at room temperature. Visualization was performed using either AEC Chromogen (Lab Vision; Thermo Scientific) or DAB Chromogen (Lab Vision; Thermo Scientific). Mayer’s hematoxylin was used as a counterstain for immunohistochemistry followed by mounting and coverslipping (Bio-Optica, Italy) for slides. Prepared slides were analyzed using an Olympus BX-43 microscope (Olympus, Tokyo, Japan) and photographed on an Olympus DP 73 camera (Olympus).
Statistical Analysis
Body weight comparisons were performed using unpaired 2-tailed Student’s t-test. Data were reported as the mean ± standard deviation (SD). For the dysplastic changes comparison between 2 groups Mann-Whitney U test was employed. A difference was considered statistically significant for a P value less than .05 (P < 0.05).
Results
Effect of Vitamin C Deprivation on Pregnancy in Dams and Body Weight of Fetuses
During the experiment, spontaneous abortion was present in 0% of the control group and 14.23% and 52.95%, respectively, of pregnant dams in the E1 and E2 groups. None of dams developed clinical signs of scurvy during the study.
On the 50th day of gestation, fetal weights in group E1 (30.9 ± 3.16g, P < .002) and group E2 (27.21 ± 1.16g, P < .001) were both significantly reduced compared with the control group (33.80 ± 1.77g). In addition, some fetuses from the E2 group were significantly smaller than fetuses in the E1 and control groups, and the E2 group also had cases of talipes equinovarus (varus defomity or internal rotation at the carpometacarpal and tarsus) of the forelegs and hind legs with visible muscular atrophy.
Gross Lesions in Cerebellum of Vitamin C–Deprived Fetuses
The total number of fetuses (n = 60) was distributed as 20 in the control group, 21 in group E1, and 19 in group E2. Grossly, the cerebellar surface in animals from the control and E1 groups had normal development and anatomical arrangement of folia (Fig. 1). The absence of with smooth surface of the cerebellum was observed in the E2 group. These changes were most pronounced in the vermis with fused folia and the loss of borders between hemispheres and vermis (Fig. 2).
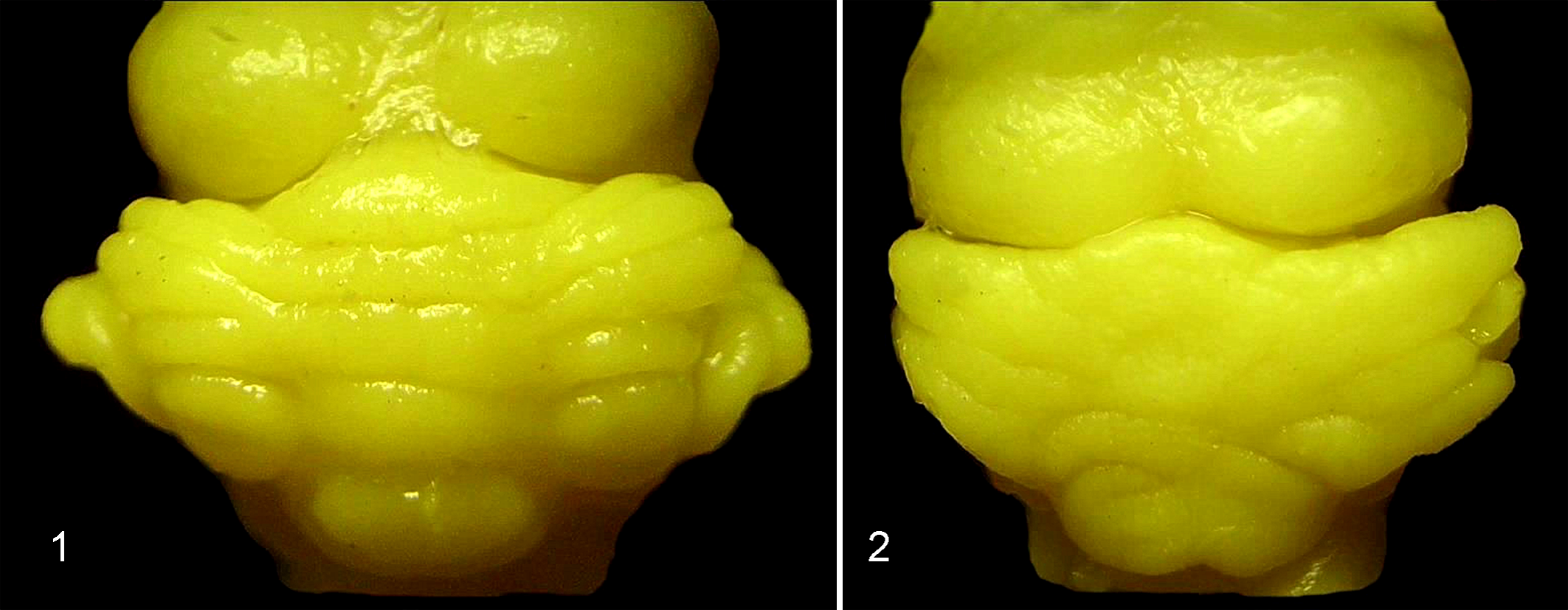
Cerebellum, fetal guinea pig, control group. Dorsal view of cerebellum shows regular arrangement of folia in cerebellar hemispheres and vermis. Zamboni’s fixation.
Qualitative Pathologic Changes of the Cerebellar Cortex in Vitamin C–Deprived Fetuses
Dysplastic changes of the cerebellar cortex were detected in fetuses in the E1 (25%) and E2 (100%) groups. These changes were not noticed in the control group of animals.
Descriptive scoring of dysplastic changes in the cerebellum was not evident in current literature, so we applied an ordinal method of classification regarding these changes following principles of histopathologic scoring.
7
Scoring (1–4) was based on the intensity of dysplastic changes in the cerebellar cortex and the number of affected folia: First stage: preserved structure of the cerebellar cortex without detectable lesions Second stage: focal ectopic clusters of external granular layer (EGL) cells in subarachnoid space of the fissure from one folium. The pial membrane has minor damage (breaches), but the structure of the adjacent cerebellar cortex was normal (Figs. 3, 4). Third stage: focal to complete obliteration of 1 or 2 fissures due to the fusion of opposing folia. In the affected areas, the cerebellar cortex had a loss of normal linear arrangement of Purkinje cells with a clear protrusion of the EGL and molecular layer into the subarachnoid space (Figs. 5, 6). Fourth stage: complete fusion of 3 or more fissures. This level of damage represents a serious disturbance of cerebellar cortex structure and Purkinje cell arrangement. The intensity of dysplastic changes in the cerebellum was the most intense, and it conformed to pathologic findings in lissencephaly type II (Figs. 7, 8).
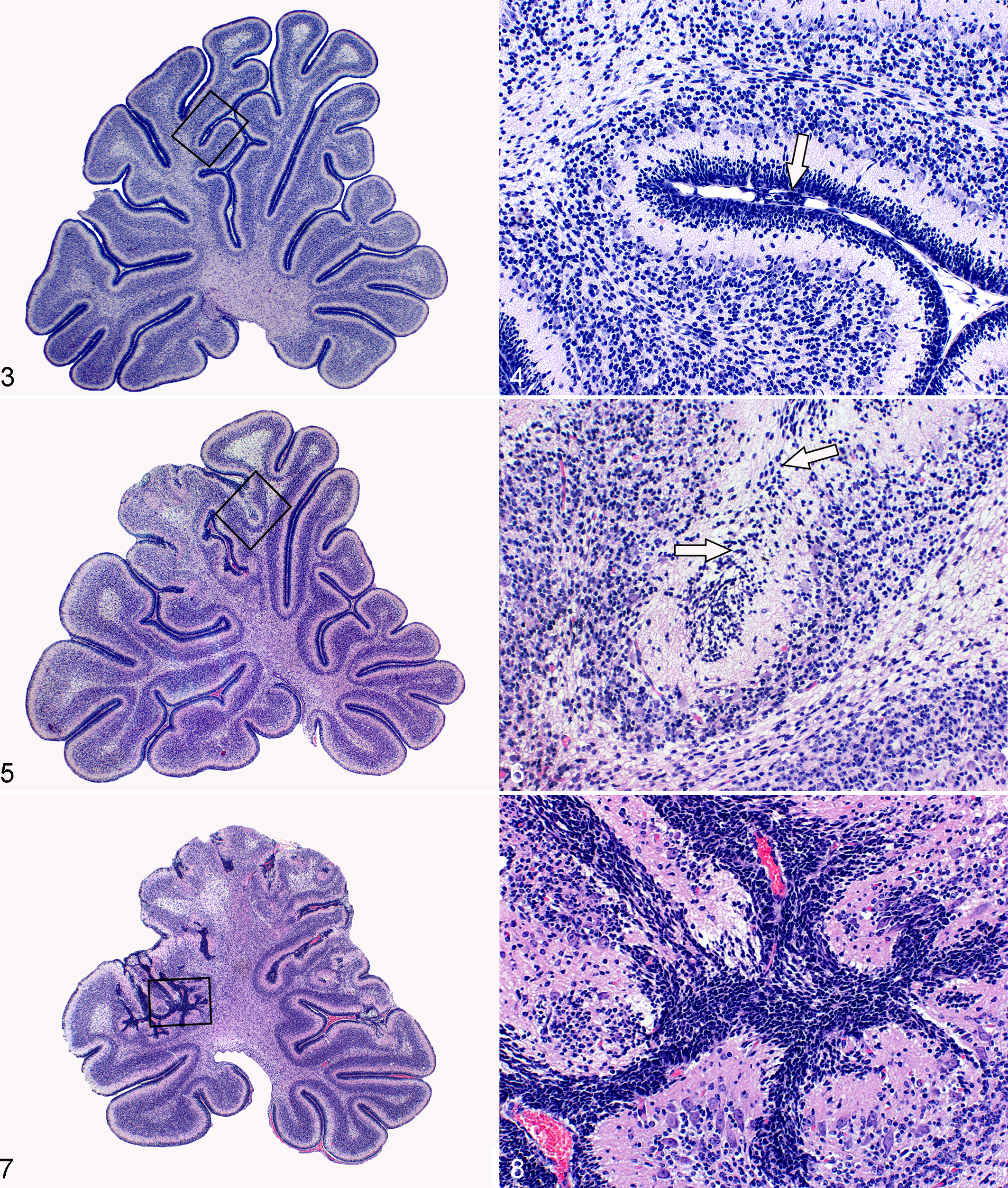
Cerebellum, guinea pig, E1 group (E1 = vitamin C deprivation after the 20th day of gestation). Low magnification of midsagittal section of vermis with visible second stage of dysplastic changes presented in black frame area. Hematoxylin and eosin (HE).
Anti–collagen IV immunohistochemical staining in the control group determined the baseline for PBM integrity (Fig. 9). PBM in animals from group E1 was mostly intact with rare signs of rupture, while the PBM structure in the E2 group was completely disrupted in the form of multiple ruptures and fragmentations. In most cases, individual fragments of pial cells and basal membrane were covered with ectopic content made up of the EGL cells. Due to the excessive migration of granular cells, pial membrane fragments were usually pushed deep into the molecular layer.
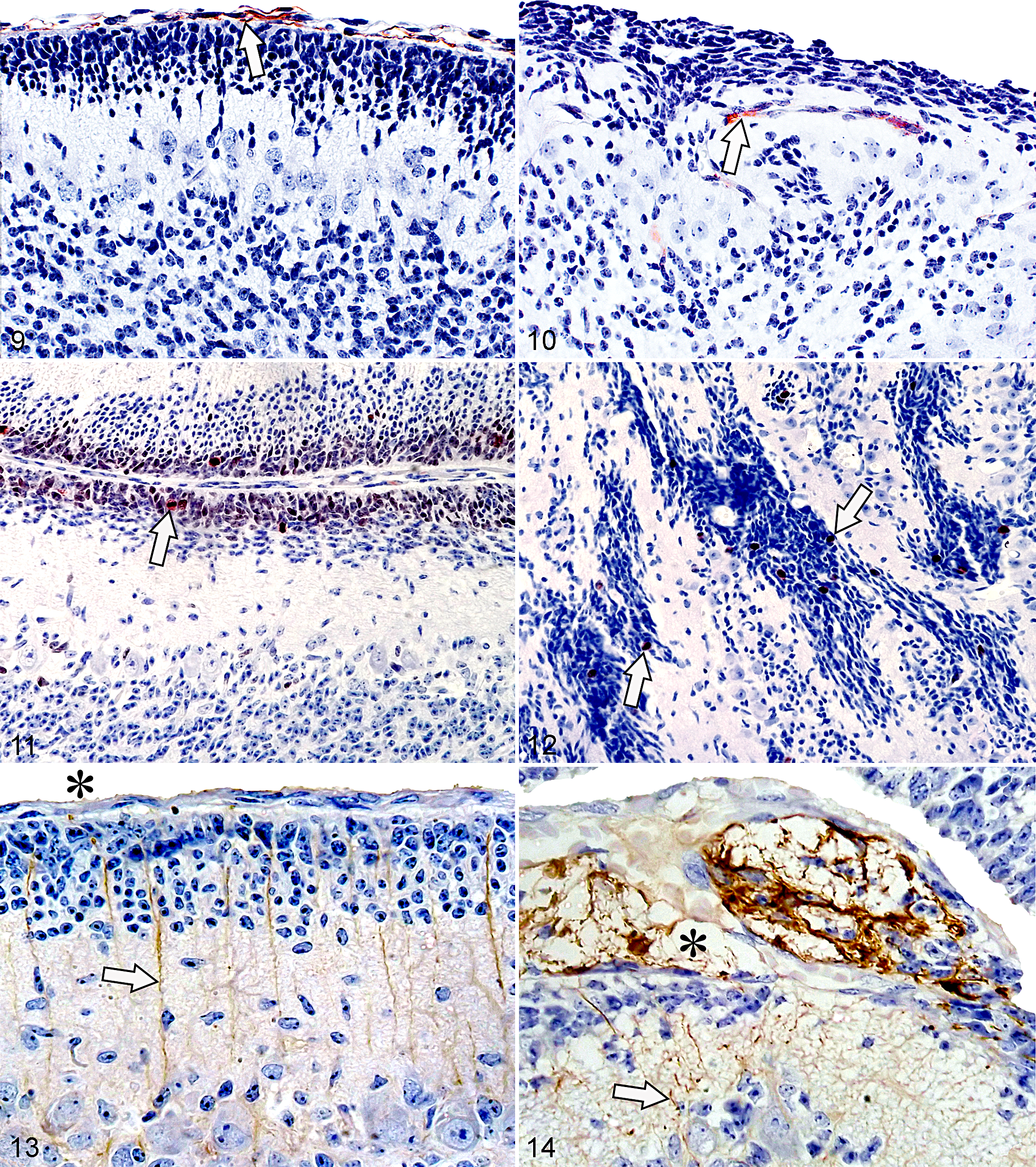
Cerebellum, fetal guinea pig, midsagittal section of vermis.
Despite dysplastic changes of the cerebellar cortex, ectopic cells of EGL retained anti–Ki-67 positivity (Fig. 12). Pronounced atrophy of the internal granular layer was observed in the areas of the cerebellar cortex with dysplastic changes.
Application of anti-GFAP staining in the control group showed Bergmann glial extensions spreading parallel through the molecular layer from the Purkinje cells to the PBM (Fig. 13). Pial membrane disruption in the E2 group resulted in complete alteration of the direction and structure of Bergmann glial cells. The ectopic mass of EGL cells in the subarachnoid space was very often permeated with intense gliosis (Fig. 14).
The most common occurrence of PBM rupture was observed in the depths of fissures with similar changes extending to proximal parts of the folia.
Quantification and Distribution of Pathologic Changes of the Cerebellar Cortex in Vitamin C–Deprived Fetuses
Dysplastic changes were not observed in the control group. In the E1 experimental group, 76% (16 of 21) were scored as first stage, 19% (4 of 21) were scored as second stage, and 5% (1 of 21) were scored as third stage dysplasia. In the E2 group, 11% (2 of 19) were scored as second stage, 42% (8 of 19) were scored as third stage, and 47% (9 of 19) were scored as fourth stage dysplasia. Statistical analyses of the dysplasia scoring are presented in Table 1.
Intensity Scores of Dysplastic Changes in Cerebellar Folia in Control, E1 and E2 Groups.
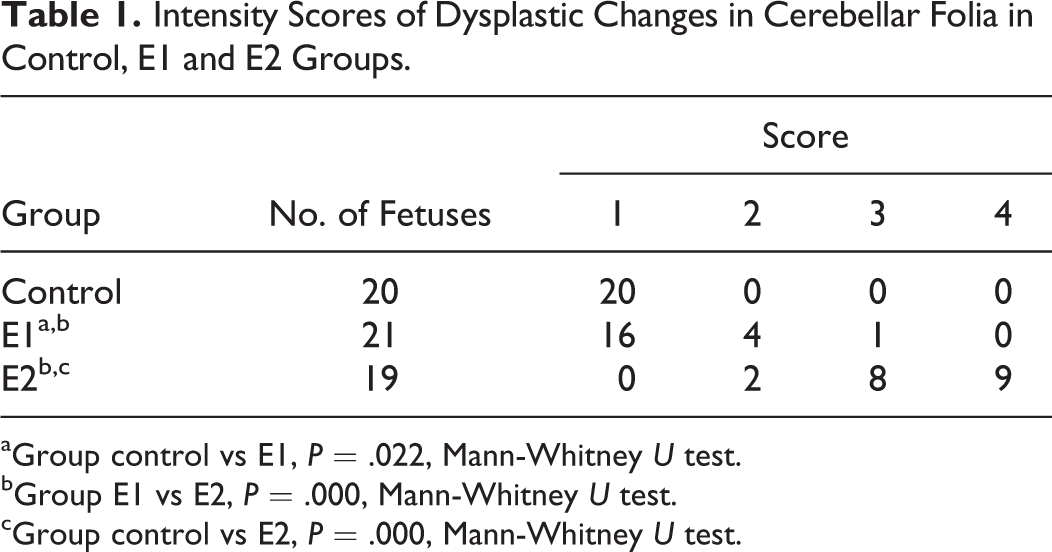
aGroup control vs E1, P = .022, Mann-Whitney U test.
bGroup E1 vs E2, P = .000, Mann-Whitney U test.
cGroup control vs E2, P = .000, Mann-Whitney U test.
In group E1 animals with dysplastic lesions (n = 5), the lesion was localized to the seventh folia in 5 animals, while 1 also had lesions extending into the fifth and sixth folia (Fig. 15). The fetuses from group E2 showed changes in all folia except the tenth. The highest incidence of dysplastic changes was observed in the folia belonging to the central lobe (VI, VII, and VIII folia) (Fig. 16).
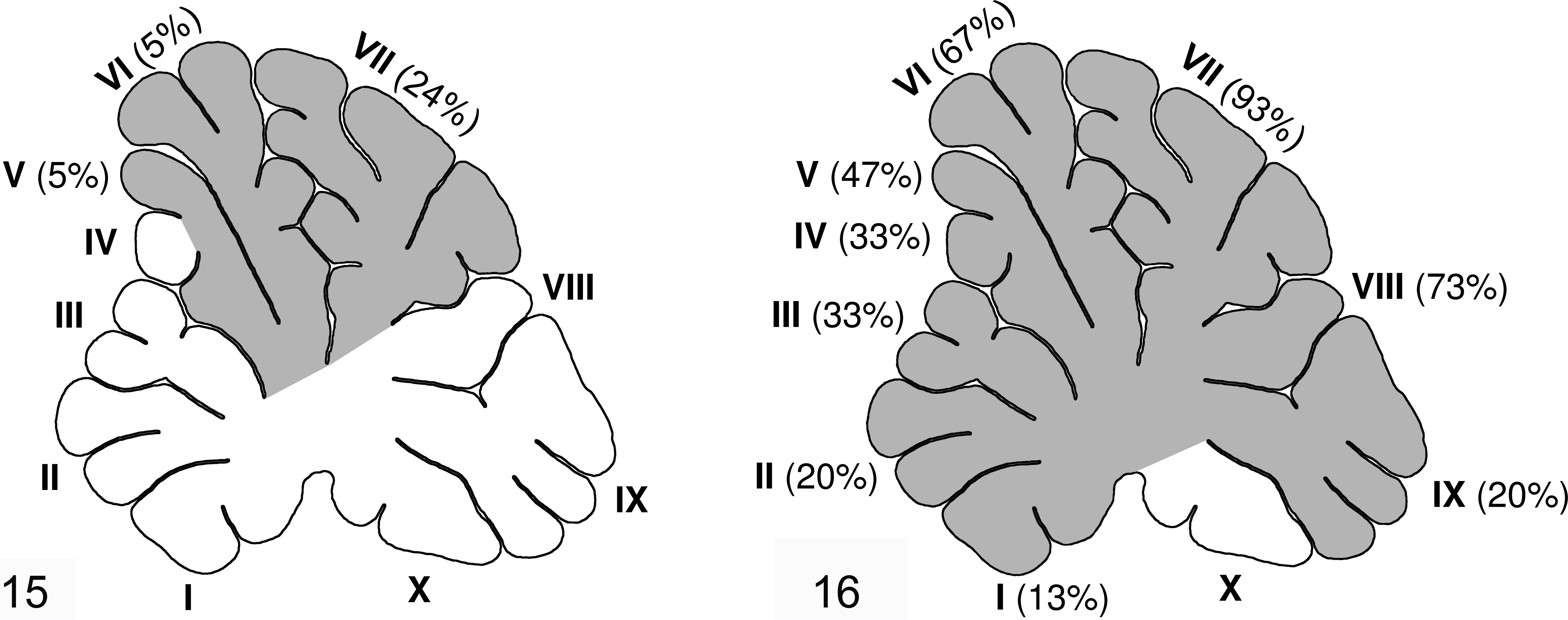
(E1 group) and
Discussion
Characteristics of Lissencephaly Type II
Lissencephalies are usually classified into type I and type II, both being characterized by the perturbation of neuronal migration but with different mechanisms of pathogenesis. 6 Lissencephaly type I is characterized by abnormal nucleokinesis causing aberrant cortical layer formation. 18 In contrast, lissencephaly type II is caused by disruption of basement membranes followed by overmigration of neurons into the subarachnoid space. 6 Lissencephaly type II is usually found in syndromes such as Fukuyama congenital muscular dystrophy (FCMD), muscle-eye-brain disease (MEB), and Walker-Warburg syndrome (WWS). These syndromes are characterized by the presence of muscular dystrophy and sometimes ocular abnormalities in addition to the pathologic dysplasia of cerebrum and cerebellum. Aberrant glycosylation of membrane glycoproteins such as dystroglycan appears to underlie the defect in all of these syndromes. 9 Dystroglycan represents a heavily glycosylated glycoprotein composed of 2 connected α and β subunits. β-Dystroglycan is a transmembrane protein interacting with both the cytoplasmic the cytoskeleton and extracellular α-dystroglycan. This is a peripheral membrane glycoprotein linking to extracellular molecules such are laminin, perlecan, agrin, and neurexin. 14 The deletion of brain dystroglycan in mice is characterized by disruptions of the glia limitans, which can lead to multiple abnormalities of cerebral and cerebellar development. It includes defects of cerebral lamination and the fusion of cerebellar folia with aberrant migration of granule cells, 24 which can also be seen in our study. Recent study of animal models with congenital muscular dystrophies (CMDs) such as WWS, FCMD, and MEB disease includes mutation of genes for encoding fukutin (FKTN), 35 POMGnT1 (O-mannose N-acetylglucosaminyltransferase 1), 44 POMT 1 (O-mannosyltransferase 1), 4 POMT 2 (O-mannosyltransferase 2), 39 and LARGE (like-glycosyltransferase). 15 All of those CMD animal models include disruption of the integrity of the PBM confirmed by electron microscopy and immunohistochemical analysis. Cerebellar defects include the fusion of opposing folia with disorganization of the external granular layer and Purkinje cells malposition. We have presented similar histologic findings in vitamin C–deprived guinea pig fetuses with a severe grade of cerebellar dysplasia.
Mouse embryos with a deletion of the Col4a1/2 locus and nidogen binding site for the laminin γ1 subunit have neuronal ectopia caused by local rupture of PBM with cells protruding into the surrounding mesenchyme. 25,42 Due to the lack of these essential components, the basement membrane changes in lungs and kidneys lead to intrauterine or postpartum death. It is important to note that postnatal development is possible in mice with a lack of β1-integrins 11 or the α1 subunit of laminin; 17 however, cerebellar fissuration and foliation disorder are also present. The loss of Purkinje cell polarity, ectopic EGL cells along the fusion line of 2 opposing folia, and atrophied internal granular layer are identical to those observed in our study. 11
Specificity of Chemically Induced Destruction of PBM as a Model of Lissencephaly Type II
The most comprehensive analysis of dysplastic changes in the development of the cerebellum was performed in studies on an animal model with an intracisternal injection of 6-OHDA. The first signs of damage in the cerebellar pial fibroblasts and meningeal cells occurred by 24 hours following application, and macrophage phagocytosis at the site of denuded of pia was present by 2 to 5 days. 29
The chemically induced destruction of meningeal cells destabilizes the basal lamina and glia limitans superficialis, further disorganizing the glial scaffold. 40 Sievers et al 30 described that the first changes can be observed when the Bergmann-glia endfeet lose contact with the PBM. Subsequently, the loss of integrity through PBM ruptures leads to the excessive growth of EGL cells, spreading from the Bergmann-glia endfeet into the subarachnoid spaces of cerebellar fissures. 29 As a consequence, this change influences neuronal migration as well as regular fissuration and foliation of the cerebellum. 31,32 Similarly, the weakening and rupturing of PBM in our investigation (Fig. 10) also caused disorganization in the glial scaffold of the cerebellar cortex (Fig. 14).
The use of light microscopy and transmission and scanning electron microscopy shows that the ectopic EGL cells develop into granule, stellate, and basket cells possessing a morphology identical to relative cells placed in situ. 22,30 We have also observed that ectopic cells of EGL continue to proliferate (Fig. 12) and form large colonies of granule and stellate cells in these regions. In addition, we have observed intense gliosis in ectopic areas (Fig. 14), suggesting that these ectopic cells usually disappear within 1 year.
Specifics of the Intensity and Distribution of Dysplastic Changes in Vitamin C–Deprived Guinea Pig Fetuses
Our pilot studies (see Materials and Methods) indicated that the first signs of cerebellar foliation can be observed in guinea pigs on the 35th day of intrauterine development. Formation of all 10 folia is completed by the 40th to 45th day of intrauterine development, and the specificity of differences in the distribution of dysplastic changes between the experimental groups can be explained using Larsell classification. 19
Using radioactive isotopes to mark mitotic active structures during the folia development in the cerebellum of rats, Joseph Altman 2 has identified 3 neurogenetic divisions. The first one is marked as early generated division and includes the formation of I, X, and a part of II folia. The second one, intermediate generated division, includes the formation of a part of II, III, IV, V, and IX folia. The third one, late-generated division, includes the formation of VI, VII, and VIII folia. On this basis, we can assume that the depletion of vitamin C in group E2, as well as the consequent collagen synthesis disorder and final weakening and rupture of PBM, took place in the early stage of cerebellar development. Folia that were developing at the time and all those that would develop in the future were affected by dysplastic changes. Because the early stage defect affected several folia, the presence of third and fourth stage dysplasia scores were common in the E2 group. The preservation of folia X can be attributed to the fact of its early formation.
In the E1 experimental group, dysplastic changes were identified only in folia VI, VII, and VIII. This clearly indicates that the critical moment of PBM rupture came during the period spanning E40 to E45 or in the period of late-generated division. Considering the relatively short period for progression of dysplastic changes leading to euthanasia, these cumulative observations clarify why this group had second or third stage dysplasia scores.
Dysplastic changes in the depth of the fissure were frequently identified in our study. This can be attributed to the mechanisms of folia formation that involve their stretching and lengthening to the bottom of the fissure. 20 It is probable that the fragile PBM is immediately affected by the pressure, which leads to its rupture.
Although we have analyzed static sections of immature cerebellar tissue, based on the statements by Sievers et al, 30 we can expect some of this dysplastic tissue to develop into mature tissue. Functional deficit will probably depend on the extent of folia involvement and the stage of cerebellar dysplasia.
For the fourth stage of cerebellar dysplasia, we can predict development of severe functional deficit with mental and motor retardation, which can be witnessed in some human syndromes. 9 Those with focal and mild dysplastic changes in the first stage of dysplasia will probably develop slight functional deficits or present with clinical features such as epilepsy, autism, or cognitive defects. 36,41
Significance of the Prenatal Vitamin C Deprivation Model in Understanding Brain Developmental Disorders
The pathogenesis of cerebellar damage in human lissencephaly is not well defined, but the progression of folial disease in our study might help explain the focal appearance in cortical dysplasia and the usually well-defined junction between normal and malformed brain tissue in humans. 9
Recent investigation of vitamin C deficiency in the period of prenatal 38 and early postnatal life 37 has detected a reduction of the hippocampal neurons in guinea pigs. However, the experimental groups of animals that received a diet low in vitamin C (100 mg/kg) were not as fully restricted as in our experiment. We speculate that lissencephaly was not diagnosed in that study because the collagen damage had insufficient time to develop in the vitamin C–deficient state.
The observation of talipes equinovarus with gross muscular atrophy in vitamin C–deprived guinea pig fetuses also gave an opportunity to use this animal model in the investigation of congenital muscular dystrophy associated with brain defects.
The disruptions of PBM were clearly present in collagen IV–deficient mouse embryos with early intrauterine lethality. 25 In our animal model, we were able to vary the intensity and distribution of dysplastic changes by modulating the initial time and duration of vitamin C deprivation.
Conclusions
Guinea pigs represent a unique animal model with regard to prenatal vitamin C deprivation and the consequential disturbance in collagen synthesis. The results of this study suggest that a vitamin C deficiency in the fetal guinea pig influences PBM rupture and the sequential development of dysplastic changes in the cerebellar cortex. The fact that neither humans nor guinea pigs are able to synthesize vitamin C creates an opportunity for further research of the impact of prenatal deprivation of vitamin C in the development of lissencephaly type II in humans.
Footnotes
Author Contribution
Conception or design: IC, NH, DL, NV, NS, JM, SS. Data acquisition, analysis, or interpretation: IC, NH, DL, NV, NS, JM, SS. Drafting the manuscript: IC, NH, DL, NV, NS, JM, SS. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding this study was supported by the Serbian Ministry of Science and Environmental Protection, Grant Number 175006/2011.
