Abstract
An organism reported in the early literature to be a rare cause of cervical lymphadenitis in guinea pigs, Streptobacillus moniliformis, has been reclassified as Caviibacter abscessus. We describe a series of sequential cases of abscesses in guinea pigs that were presented to our clinic from which the only agent isolated was a unique, serum-requiring bacterium. Discrete colonies were not detected in 6.5% CO2 or anaerobically on routine primary isolation media containing up to 5% whole sheep blood, with and without cysteine, vitamin K, and hemin supplementation after 7 days of incubation at 37°C. Based on subsequently determined growth requirements, the organisms were best described as serum-requiring, aerotolerant anaerobes. Colonies were detectable within 24 h at 37°C in an anaerobic atmosphere on a mycoplasma agar–based medium containing 10% pig serum and reached 3 mm in diameter within 3–5 days. Microscopic appearance consisted of small gram-negative rods and coccobacilli with occasional filaments. However, in direct smears from clinical specimens and from weak or dysgonic growth on plates incubated under suboptimal growth conditions (e.g., in 6.5% CO2), irregular rods with occasional small bulbous forms or numerous long wavy filaments were observed. All of the isolates generated unique spectral profiles similar to that of C. abscessus when examined by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Phylogenetic analysis of partial 16S rRNA gene sequences showed that the isolates were identical to each other and shared 99.9% sequence identity with C. abscessus.
Keywords
Cervical lymphadenitis and abscesses are common in domestic guinea pigs (Cavia porcellus).15,17 Complete surgical excision is recommended rather than surgical drainage because of the thickness of pus within the abscesses (Donnelly T. Guinea pigs. The Merck Veterinary Manual; 2015. Available at: http://goo.gl/DXfDpg; Ritchie B. Abscesses in guinea pigs. Pet Place: Small Animals; 2014. Available at: http://goo.gl/d3VG1N). As a result of the empirical surgical approach to therapy and the inherent sensitivity of guinea pigs to treatments with some antibacterial drugs, bacterial etiologies and in vitro susceptibilities are seldom determined in clinical cases. Streptococcus equi subsp. zooepidemicus is listed in veterinary medical references as a usual cause of suppurative lymphadenitis, but other organisms, often associated with the oral cavity and upper respiratory tract, may also be found (http://goo.gl/DXfDpg). 11 It has also been stated that “cultures from abscesses are frequently negative” (http://goo.gl/d3VG1N). In 1941, a causal agent was described as “difficult to grow and sometimes suggestive of a filamentous organism.” 18 Subsequently, Streptobacillus moniliformis, an organism matching the 1941 morphologic description, was named as a cause of cervical abscesses in guinea pigs.1,10 An isolate of S. moniliformis was identified in a similar manner from a case of granulomatous pneumonia in a guinea pig. 14 It was suggested that guinea pig isolates differed from S. moniliformis isolates from rats as, unlike rat isolates, guinea pig isolates grew optimally in anaerobic conditions and did not ferment carbohydrates. 14 Some authors suggested that the guinea pig abscess agent was more like Fusobacterium necrophorum. 17 Because of the known zoonotic disease potential of S. moniliformis isolates from rats and the commensal existence of the organism in the oral cavities of rats, S. moniliformis was listed as an agent for routine monitoring of laboratory rodents. 16 Because of the difficulties in culturing S. moniliformis from oral samples, various immunoassays and polymerase chain reaction (PCR) assays were developed for such monitoring.3,4,13 It was later demonstrated that guinea pigs were resistant to infection with S. moniliformis isolated from rats 5 and that Leptotrichia spp. may produce false-positive results in one of the earlier S. moniliformis PCR assays. 6 During the course of our investigations into the causes of abscesses in a series of pet guinea pigs, a European study using an archived isolate originally deposited as S. moniliformis from an abscess in a guinea pig in Sweden and an isolate obtained in 2015 from a similar lesion in a guinea pig from Germany reclassified both organisms as Caviibacter abscessus. 8
Our report describes the clinical isolation and identification of C. abscessus in a series of 9 pet guinea pigs with abscesses. Each patient was presented to the avian and exotic animal clinical service at the University of Tennessee (UT), Veterinary Medical Center (Knoxville, Tennessee) between 2014 and 2016 and had a clinical diagnosis of abscessation, with most located in the region of the cervical lymph nodes. All but 1 patient was owned by a single guinea pig–rescue enthusiast. However, the abscesses appeared to have originated in multiple unrelated colonies. A summary of patient histories is presented in Table 1. The abscesses were surgically excised en bloc, if possible, and then placed into a sterile petri dish and submitted for aerobic and anaerobic cultures.
Summary of American breed pet guinea pigs from which Caviibacter abscessus was isolated.*
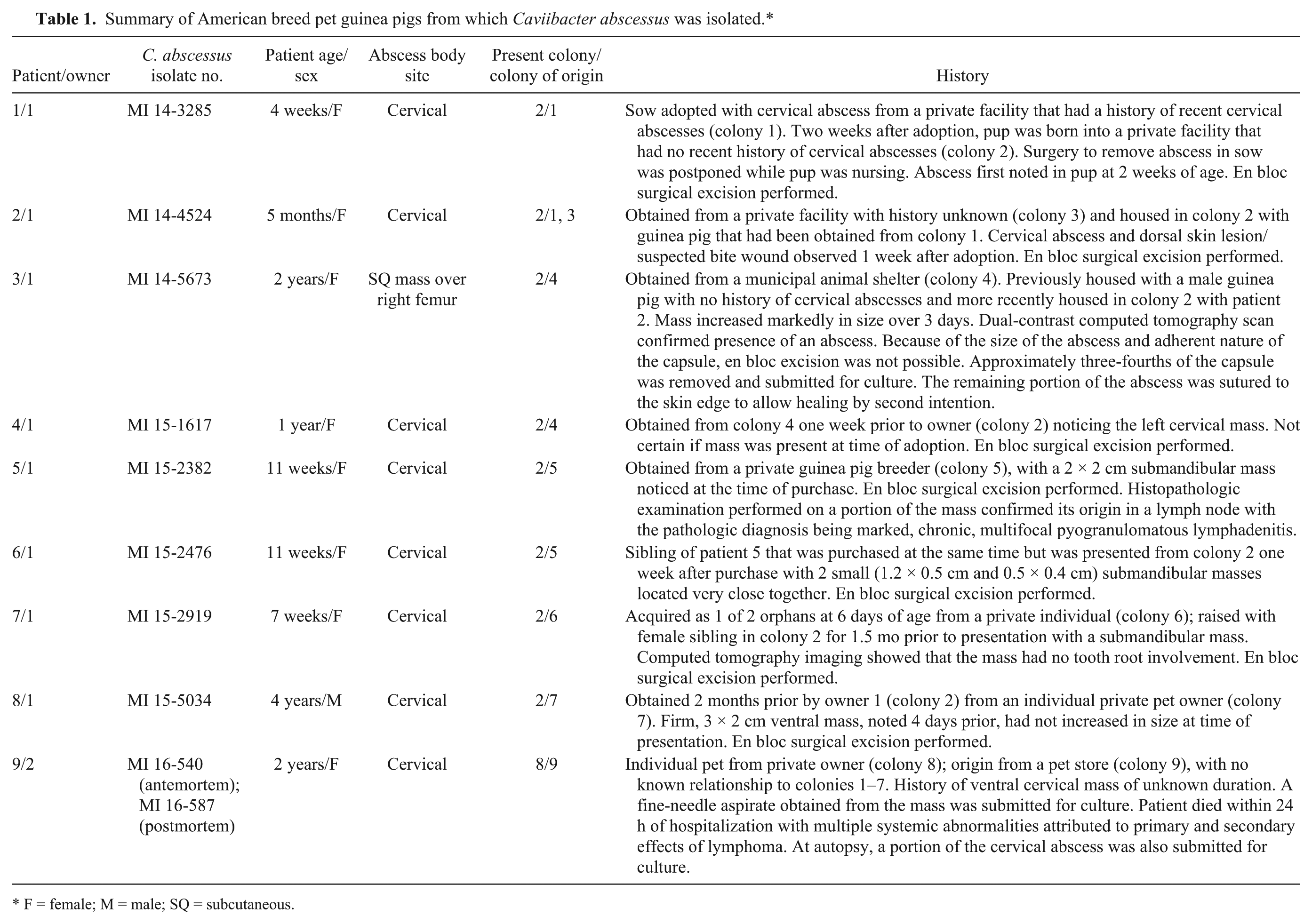
F = female; M = male; SQ = subcutaneous.
Abscesses received intact were aseptically opened; purulent material from the inner capsular walls of each abscess was processed by routine culture procedures at the UT College of Veterinary Medicine, Clinical Bacteriology and Mycology Laboratory (Knoxville, TN). Media and growth conditions included MacConkey agar a and fluid thioglycollate broth a incubated aerobically at 37°C; Columbia agar a and colistin–nalidixic acid agar a plates each containing 5% sterile sheep blood b incubated in 6.5% CO2 at 37°C; and phenylethyl alcohol agar a and CDC anaerobic blood agar a plates each supplemented with 5% sterile defibrinated sheep blood b incubated in an atmosphere containing 5% H2, 10% CO2, and 85% N2 at 37°C.
A direct Gram stain from patient 1 revealed irregular, gram-variable or incompletely decolorized rods, some with bulbous terminal swellings or beaded staining appearance (Fig. 1). Growth was not detected in or on any of the inoculated primary isolation media after incubation for 24 h. Retrospectively, it was decided to inoculate media that might be more suitable for isolation of the previously described putative guinea pig “S. moniliformis.” Inoculating loops were used to transfer portions of the initial inoculum, seen as a dull film in the first quadrant of the 24-h aerobic Columbia blood agar streak plate, onto nonselective mycoplasma agar plates (PPLO [pleuropneumonia-like organism] agar a containing 0.3% yeast extract, c 0.01% calf thymus DNA, d and 10% pig serum e ). One plate was incubated in 6.5% CO2 at 37°C, and 1 plate was incubated in an anaerobic atmosphere at 37°C. A Gram stain of this secondary inoculum revealed organisms similar to those seen in the direct Gram stain but with a uniform gram-negative staining reaction. After incubation for 24 h on the mycoplasma agar plates, minute, pinpoint (<0.5 mm) colonies were observed on each plate. After incubation for 72 h, off-white, umbonate, 1.5–2.0 mm diameter colonies were observed on the anaerobic plate (Fig. 2), whereas colonies observed on the plate incubated in CO2 did not increase in size. All media were incubated for 7 days, and no other organisms were detected. When the mycoplasma agar was included as a primary plating medium, samples from patients 2–9 yielded high levels (>100 to >1000 colonies) of an identical gram-negative anaerobic rod in pure culture. Isolates were preserved in a cryoprotectant that contained sucrose, potassium phosphate, glutamate, and bovine albumin fraction V 7 at −80°C and were satisfactorily retrieved on several occasions for subsequent testing.
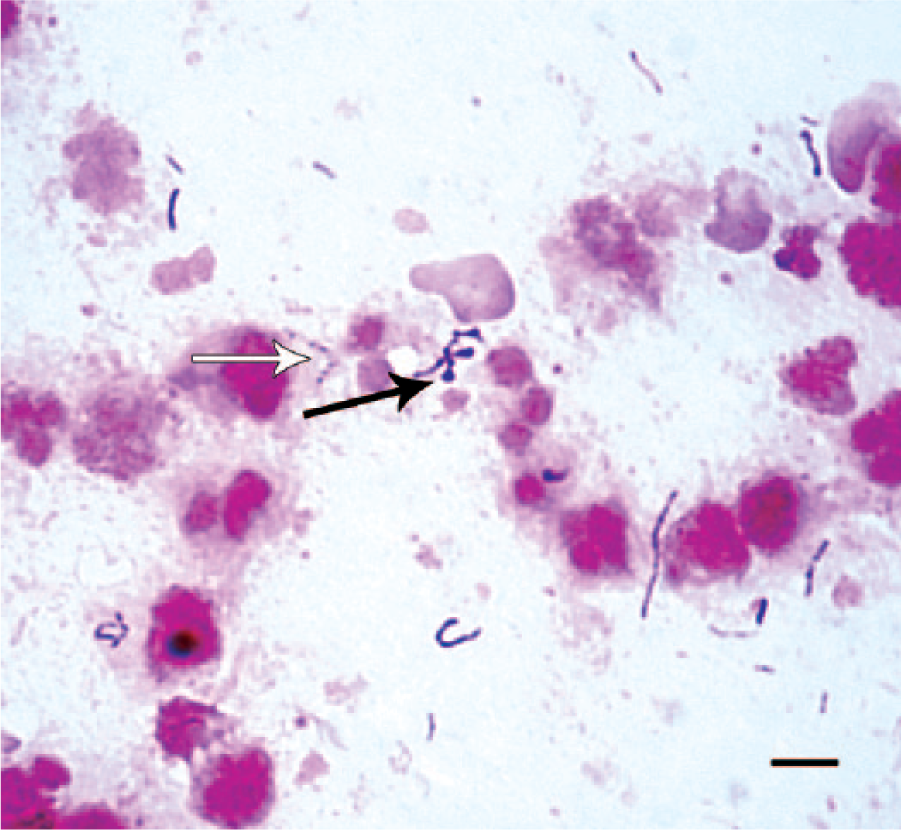
Gram stain containing gram-variable (incompletely decolorized), irregularly shaped rods with occasional bulbous swellings (black arrow) and beaded staining appearance (white arrow) in a smear of culture material obtained from within a cervical abscess. Bar = 5 µm.
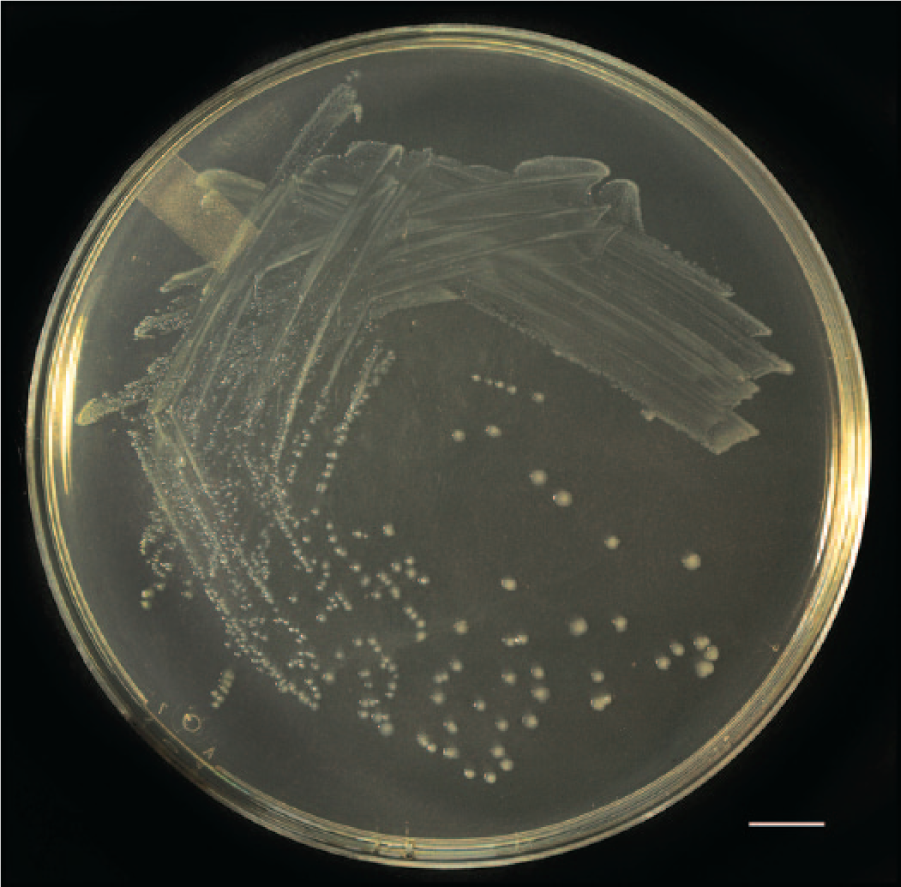
Colonies of Caviibacter abscessus, from a cervical abscess in a guinea pig, grown on nonselective mycoplasma agar in an anaerobic atmosphere at 37°C for 3 days. Bar = 14 mm.
Upon anaerobic subculture on the mycoplasma agar–based medium with 10% pig serum, each isolate produced smooth, off-white colonies with raised centers that reached 4 mm within 5 days’ incubation at 37°C. On the same medium incubated in 6.5% CO2, weak growth with pinpoint colonies (<0.5 mm) was observed after incubation at 37°C for 5 d. Weak confluent growth without detectable colonies was observed in the first quadrants of streak plates (Columbia, colistin–nalidixic acid, and CDC anaerobic agars) containing 5% sheep blood after incubation for 5 days at 37°C in capnophilic and anaerobic atmospheres. The isolates did not grow on chocolate agar
a
under any of the incubation conditions. In mycoplasma broth medium
a
with 10% pig serum, growth appeared as sediment at the bottom of the tube within 24 h of static anaerobic incubation at 37°C. Suspensions made from such growth formed aggregated “bread crumb”–like clumps (Fig. 3). Cells had gram-negative staining reactions and showed variable morphologies. Cells grown anaerobically with mycoplasma-based media containing 10% pig serum appeared as relatively uniform rods (0.5 × 3–5 µm) or coccobacilli with few short chains or filaments (0.5 × <200 µm; Fig. 4a). Cells grown with the same medium incubated in 6.5% CO2 consisted entirely of long, wavy filaments (Fig. 4b). Based on their relative growth on different blood product–containing media and in different atmospheres on subculture (Table 2), the isolates were best described as blood or serum-requiring, aerotolerant anaerobes. Catalase, oxidase, and indole production were not detected. The isolates were nonhemolytic on a modified version of the mycoplasma agar that contained 5% pig serum and 5% defibrinated sheep blood. In a carbohydrate test medium
a
with 10% pig serum, the isolates fermented glucose, maltose, trehalose, and salicin. By conventional test methods,
a
the isolates were positive for hippurate hydrolysis and negative for esculin hydrolysis. Preformed enzyme activity was determined in an off-label use fashion with either all or portions of 2 commercial test kits.f,g Inocula were prepared as heavy suspensions from 3-d cultures grown anaerobically at 37°C on mycoplasma agar. Positive reactions included β-glucuronidase, α-

Growth of Caviibacter abscessus suspended from bottom of tube after incubation in mycoplasma broth in an anaerobic atmosphere at 37°C for 24 h.
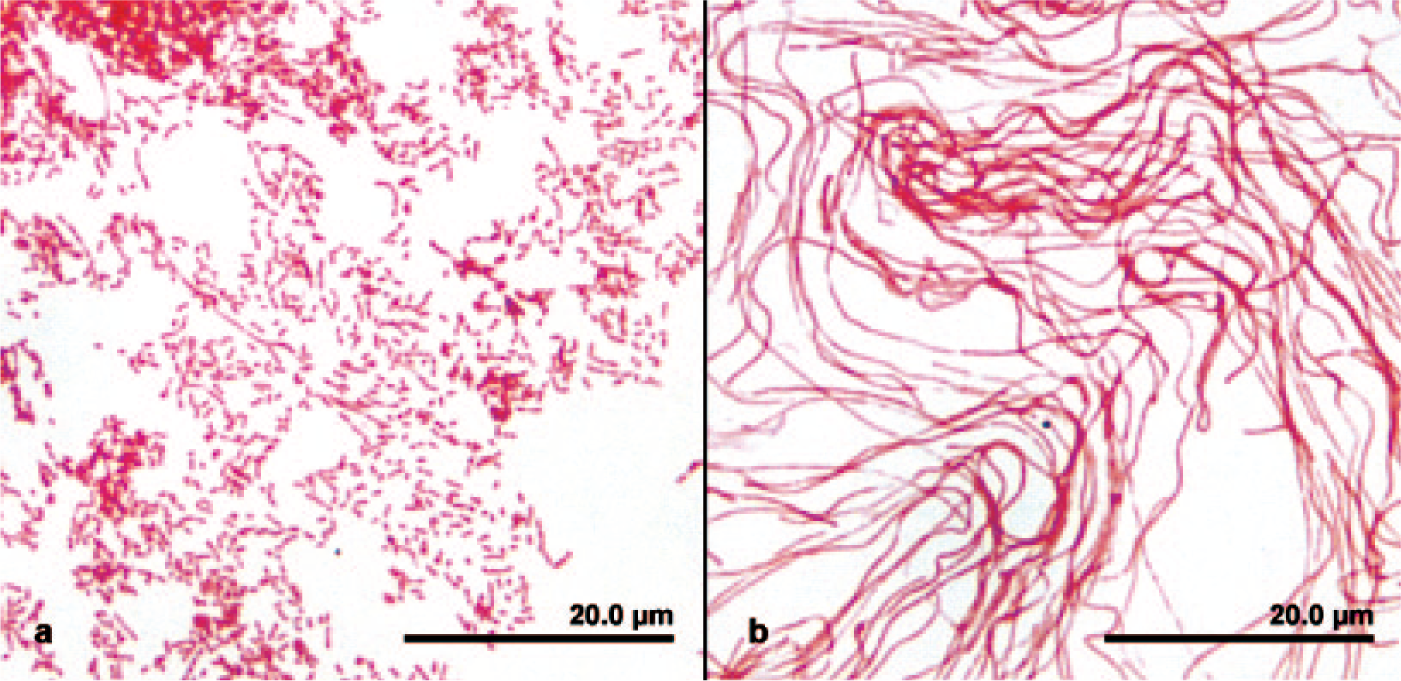
Gram stain of Caviibacter abscessus, from a cervical abscess in a guinea pig, grown on nonselective mycoplasma agar for 3 days.
Growth patterns of guinea pig Caviibacter abscessus strain MI 14-3285 on different media and under different atmospheric conditions after 5 days at 37°C.
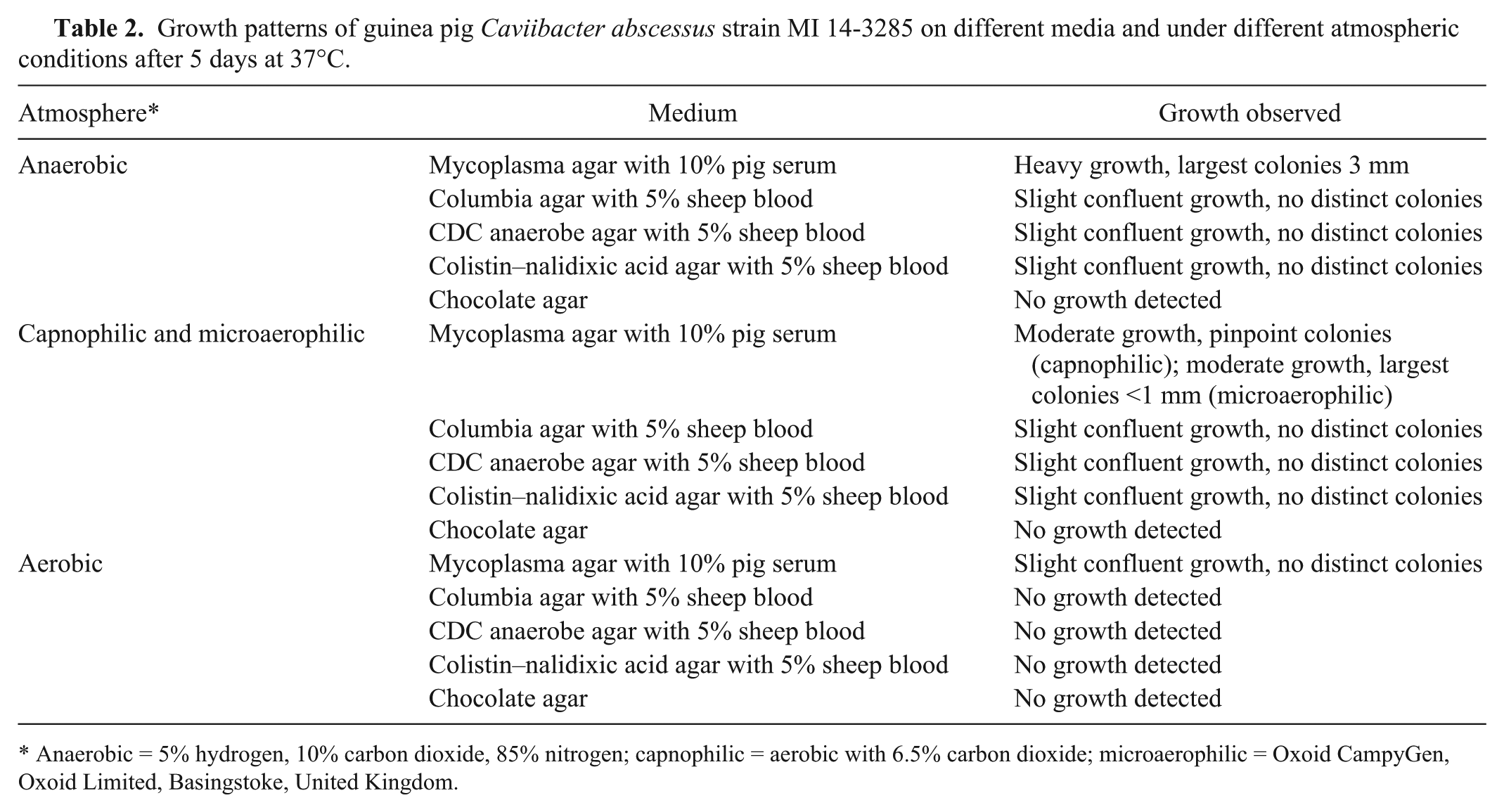
Anaerobic = 5% hydrogen, 10% carbon dioxide, 85% nitrogen; capnophilic = aerobic with 6.5% carbon dioxide; microaerophilic = Oxoid CampyGen, Oxoid Limited, Basingstoke, United Kingdom.
Amplification products were not detected from lysates of any of the 9 guinea pig isolates when a previously described 16S ribosomal (r)DNA–based Streptobacillus-specific PCR assay was performed as previously described.9,13 Species identification was pursued with each isolate by partial 16S rRNA gene sequencing. DNA was extracted from individual colonies by physical disruption with glass beads h and subjected to PCR with universal eubacterial primers designed to amplify a portion of the 16S rRNA gene. 19 The primers, Broad 1 (GCGGATCCTGCAGAGTTTGATCCTGGCTCAG) and Broad 2 (GGCTCGACCGGGTTACCTTGTTACGACTT), correspond to bases 8–27 and 1492–1510 of the Escherichia coli small ribosomal subunit gene. The PCR assays were performed with a commercial Taq polymerase premix i containing reaction buffer and nucleotides. The thermocycler conditions included 1 cycle of denaturation and enzyme activation at 95°C for 1.5 min, 30 cycles of annealing at 50°C for 30 s, extension at 72°C for 2.5 min, and denaturation for 1 min at 94°C. This was followed by a final cycle of annealing at 50°C for 2 min, extension at 72°C for 5 min, and holding at 4°C. The PCR amplification products were subjected to electrophoresis in 1% agarose gel containing 1 µg/mL ethidium bromide and were visualized by ultraviolet transillumination. The PCR products were of expected size, and single-strand DNA was removed by exonuclease treatment j before submitting to the UT Genomics Core Facility (Knoxville, TN) for bidirectional sequencing with the PCR primers.
Nucleotide sequence data were analyzed using a commercial software program. k Alignment of the sequences obtained from each isolate showed that they shared 100% nucleotide sequence identity with each other. The sequence (1,402 nucleotides) of a representative isolate, strain MI 14-3285, was submitted to GenBank and assigned accession KR612328. The partial 16S rRNA gene sequences of the isolates were compared to sequences in GenBank using the BLASTn algorithm. 2 The sequences were shown to have 99.9% gene sequence similarities with those of the 2 strains of C. abscessus. 8
Isolate MI 14-3285 was further examined by obtaining partial sequences from the groEL, gyrB, and recA genes. DNA was extracted with a commercial kit, l and PCR amplicons for direct sequencing were obtained using primers and a procedure that were previously described. 20 The partial groEL, gyrB, and recA sequences from isolate MI 14-3285 were submitted to GenBank and assigned accessions KU195695, KU195696, and KU195697, respectively. Comparisons of the partial groEL, gyrB, and recA sequences of strain MI 14-3285 with those deposited in GenBank from the genome of C. abscessus type strain CCUG39713 revealed 99%, 100%, and 100% nucleotide sequence similarities, respectively.
Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) was performed on isolates from patients 1–7 using the ethanol–formic acid extraction method for better resolution, following the manufacturer’s instructions, m and after manual inclusion of spectra of the type strains of C. abscessus (CCUG 39713), Sneathia sanguinegens (CCUG 41628), and Sneathia amnii (CCUG 52976) to the database (MBT DB-5627 species list). The 7 guinea pig isolates generated similar spectral profiles to that of C. abscessus with scores above 2.1, and were not related to either of the 2 added Sneathia species or to the existing spectra covering S. moniliformis, Leptotrichia trevisanii, Leptotrichia wadei, and an uncharacterized Leptotrichia sp.
The abscesses examined in our report appear to have originated while patients were housed in multiple, unrelated colonies. Because C. abscessus was also isolated from guinea pigs in 2 other countries, 8 it is likely that the organism occurs commonly in guinea pigs. However, the full extent of the distribution and transmission of C. abscessus among guinea pigs and other animals is not known. In our case, C. abscessus was recovered from all 9 pet guinea pigs that were presented with abscesses over a 2-year period, with no other guinea pigs being presented for similar conditions during this time. Whether isolation from cervical abscesses in guinea pigs is a consistent feature of C. abscessus awaits further confirmation. A prior investigation into the high incidence of cervical abscesses in stock guinea pigs at several laboratories reported that “S. moniliformis” was isolated in pure culture from every guinea pig examined (identification based on morphologic and growth characteristics; total numbers not disclosed). 10 It was interesting to note that, during the present study period, cervical abscesses were observed in 2 sow–pup pairs that belonged to owner 1 (neither were cultured), patient 2 had an apparent antecedent skin wound, and patient 4 was not housed in direct physical contact with other guinea pigs (although it was not certain if the abscess in patient 4 originated after adoption). Collectively, these observations suggest that transplacental, oral contact, and aerosol modes of transmission may each have been possible, as is known for other members of the family Leptotrichiaceae.12,20,21
Our report suggests that C. abscessus is a significant bacterial pathogen of guinea pigs, particularly in association with cervical abscesses. The isolates were readily cultivated in pure culture on a mycoplasma agar containing 10% pig serum incubated in an anaerobic environment. Other investigators isolated putative S. moniliformis from guinea pigs on complex mycoplasma-like media formulations containing up to 30% horse serum.1,10,14 Minimum nutrient requirements and optimal isolation media have yet to be determined for recovery of C. abscessus from guinea pigs. Phenotypic properties of our isolates were consistent with those described for C. abscessus, 8 with minor exceptions (e.g., extent of growth on primary agar plates containing 5% blood or on chocolate agar, and production of glucuronidase). However, phenotypic characteristics alone do not readily discriminate C. abscessus from other serum-requiring members of family Leptotrichiaceae.
As previously observed, 8 MALDI-TOF MS provides a rapid means for identification of this organism. Although C. abscessus appears to be susceptible to several antimicrobial drugs, 8 in vitro susceptibility testing methods and interpretations have not been standardized for serum-requiring anaerobes such as C. abscessus. Using standard concentration disks and anaerobic incubation on mycoplasma agar containing 10% pig serum, the resultant zone diameters of growth inhibition suggested a trend of greater in vitro inhibitory activity for ceftiofur (30 mm), chloramphenicol (23 mm), and doxycycline (30 mm) than for ciprofloxacin (19 mm), enrofloxacin (15 mm), gentamicin (11 mm), and trimethoprim–sulfamethoxazole (6 mm).
In the 6 months preceding our study, prior to the inclusion of anaerobically incubated mycoplasma agar containing 10% pig serum, aerobic and anaerobic cultures of purulent material from similar abscesses in 2 guinea pigs were reported as “no growth detected.” Another potential limitation was that those samples were submitted only on mini-tip swabs rather than an entire abscess. We suggest that C. abscessus may be missed in routine aerobic and anaerobic primary isolation procedures used in many veterinary laboratories because of its fastidiousness, variable morphologies, gram-negative staining reaction, as well as insufficient sampling for detection in direct Gram stains. Additional isolations and further study of this novel organism is required to determine its full range of distribution and pathogenicity.
Footnotes
Acknowledgements
We thank Dr. Patrick Sullivan for assistance with sample collection, Dr. William Shek for providing a reference Streptobacillus moniliformis isolate for polymerase chain reaction testing, Rupal Brahmbhatt and Matthew Riley for assistance with nucleic acid assays, Alexandra Collaud for assistance with matrix-assisted laser desorption/ionization time-of-flight mass spectrometry, Joy Chambers for assistance with graphic illustration, and Phil Snow and Robert Donnell for photography.
Authors’ contributions
DA Bemis contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. BH Johnson and RD Jones contributed to conception of the study; contributed to acquisition, analysis, and interpretation of data; and gave final approval. MJ Bryant, BV McCleery, and CB Greenacre contributed to conception of the study; contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; and gave final approval. V Perreten and SA Kania contributed to design of the study; contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; and gave final approval.
a.
BBL, BD Diagnostic Systems, Sparks, MD.
b.
Hemostat Laboratories, Dixon, CA.
c.
Hardy Diagnostics, Santa Maria, CA.
d.
DNA from calf thymus, Sigma-Aldrich, St. Louis, MO.
e.
Porcine serum, Gibco-Life Technologies, Grand Island, NY.
f.
RapidID ANAII, Remel Inc., Lenexa, KS.
g.
ID 32 STAPH, bioMérieux Inc., Durham, NC.
h.
BioSpec Products Inc., Bartlesville, OK.
i.
Premix Taq, Takara Bio Inc., Otsu, Shiga, Japan.
j.
ExoSAP-IT, USB Corp., Cleveland, OH
k.
Lasergene v7.2, DNASTAR Inc., Madison, WI.
l.
Ultraclean, MO BIO Laboratories Inc., Carlsbad, CA.
m.
Application Note, MT-80, Microflex LT; Bruker Daltonics GmbH, Bremen, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
