Abstract
Equine mammary tumors are uncommon, and relatively sparse histopathologic and molecular data exist. The present study describes the histopathologic features of 7 such tumors, which exhibited infiltrative growth, intermediate to high mitotic rates, and focally extensive necrosis. The tumors exhibited variably strong staining for vimentin and cytokeratin 14, as well as frequently weak cytoplasmic staining for pan-cytokeratin. E-cadherin expression was strong. Interestingly, a subgroup of the tumors exhibited strong nuclear staining for estrogen receptor α. Three of 7 tumors exhibited nuclear expression of the transcription factor STAT3, suggesting that STAT3 was transcriptionally active. Rare to absent nuclear STAT3 expression was observed in carcinomas exhibiting moderate to intense staining for cytokeratin 14. This investigation confirms previous investigators’ assertions that equine mammary tumors have a malignant phenotype. A subset of the equine mammary tumors exhibited estrogen receptor α expression, suggesting that these tumors may potentially have similar molecular characteristics to their feline and canine counterparts.
Domestic herbivores develop mammary tumors at a much lower frequency than do domestic carnivores. Mammary tumors in mares are rare, despite the fact that horses in the United Kingdom generally represent a companion animal group rather than a production animal species and as such often reach an advanced age. In general, equine mammary tumors are carcinomas. They tend to be locally aggressive and metastasize to the regional lymph nodes and other organs. 2,3,8,9,11 The scarcity of equine mammary tumors is exemplified by the fact that these tumors have mainly been documented by means of case reports 2,3,8 and small case series. 9,11 Molecular characterization, which is gaining increasing prominence in small animal mammary oncology, 13 has so far been limited, although staining for estrogen receptor and progesterone receptor has been attempted 11 and successfully demonstrated in one mare. 2
The purpose of the current study was to characterize the morphology of equine mammary tumors in conjunction with the expression of selected intermediate filaments and the adhesion molecule E-cadherin. Estrogen receptor α (ERα) status was assessed in view of the previously reported ER-positive case 2 and the possible prognostic implications. Finally, nuclear expression of STAT3 in equine mammary tumors was interrogated as a potential future prognostic marker. The signal transducers and activators of transcription (STATs) are transcription factors that influence cellular differentiation, proliferation, survival, and death. STAT3 is constitutively activated in approximately 50% of primary human breast tumors 1 and may predict a poor prognosis in ERα-positive cases. 7 STATs are generally activated by transient phosphorylation prior to dimerization and translocation to the nucleus to control transcription, and thus nuclear localization may indicate transcriptional activity.
Methods
A search of the histopathology and necropsy databases at Beaufort Cottage Laboratories from 2006 to 2013 (totaling 15 789 equine submissions) identified 6 cases of equine mammary carcinoma and no cases of equine mammary hyperplasia or adenomas. A further equine mammary carcinoma was identified from the histopathology submission records of Bridge Pathology Ltd, which comprised 470 equine histopathology submissions received between 2008 and 2013. Representative histologic sections were examined and the molecular characteristics of the tumors were investigated.
Three-micrometer sections of paraffin-embedded material were mounted on positively charged slides (Snowcoat; Surgipath Europe Ltd, Peterborough, UK). Sections were stained with hematoxylin and eosin, and immunohistochemical staining for pan-cytokeratin, cytokeratin 14, and vimentin was carried out using a routine protocol employing an automated immunohistochemistry system (Dako Autostainer; Dako UK, Ely, UK). Diaminobenzidine solution was used to demonstrate peroxidase activity, and slides were counterstained with hematoxylin. Diluent was used as a negative control. Immunohistochemical staining for nuclear STAT3 expression (horse Nos. 1–4) was carried out manually. Immunofluorescence staining for nuclear localization of STAT3 (horse Nos. 5–7), ERα, and E-cadherin was performed manually. Standard protocols were followed and are described elsewhere. 4 The antibodies and dilutions employed are detailed in Supplemental Table 1. Appropriate species- and isotype-matched immunoglobulins were used as negative controls for manual staining. Quiescent mammary tissue removed from a mare at necropsy provided positive control tissue for pan-cytokeratin, cytokeratin 14, vimentin, and E-cadherin staining.
Clinical and Histopathologic Findings
Limited signalment and clinical data were available. For 5 of the 7 mares, the age range was 10 to 20 years (median, 11 years). Two were cobs, and one was a Thoroughbred; for the others, breed was not known. Information on parity and lactation status was not available. In some cases, the mass had been present for some time (horse Nos. 1, 2). In 2 cases (horse Nos. 1, 5), the lesion was originally diagnosed and treated as mastitis. In one instance (horse No. 3), unilateral mastectomy was performed, but generally treatment was limited or not recorded. Euthanasia was frequently undertaken following the results of biopsy. In one case (horse No. 2), necropsy was carried out, and metastatic spread to the sublumbar, prefemoral, and caudal cervical lymph nodes, lung, spleen, and liver was identified and confirmed histologically.
For each case, histopathologic and immmunohistochemical/immunofluorescent findings are detailed in Supplemental Table 2. Representative images are displayed in Figs. 1 to 10. The 7 equine mammary tumors exhibited a range of histologic appearances predominantly characterized by a tubulopapillary pattern (Fig. 1), a solid pattern, or a combination of the two. Two tumors (horse Nos. 1, 6) exhibited a prominent comedone pattern of necrosis (Figs. 2, 6; horse No. 1) similar to that previously recorded. 8 All tumors exhibited an infiltrative phenotype similar to that already documented. 3,8,9,11 Infiltration of the tumors with lymphocytes, plasma cells, macrophages, and neutrophils was frequent. The aggressive phenotype of the tumors was reflected by an intermediate to high mitotic rate (Suppl. Table 2). In addition to horse No. 2, where metastatic spread was confirmed at necropsy, in horse No. 3, aggregates of neoplastic cells were clearly visible within lymphatic vessels adjacent to the main mass (Fig. 3).
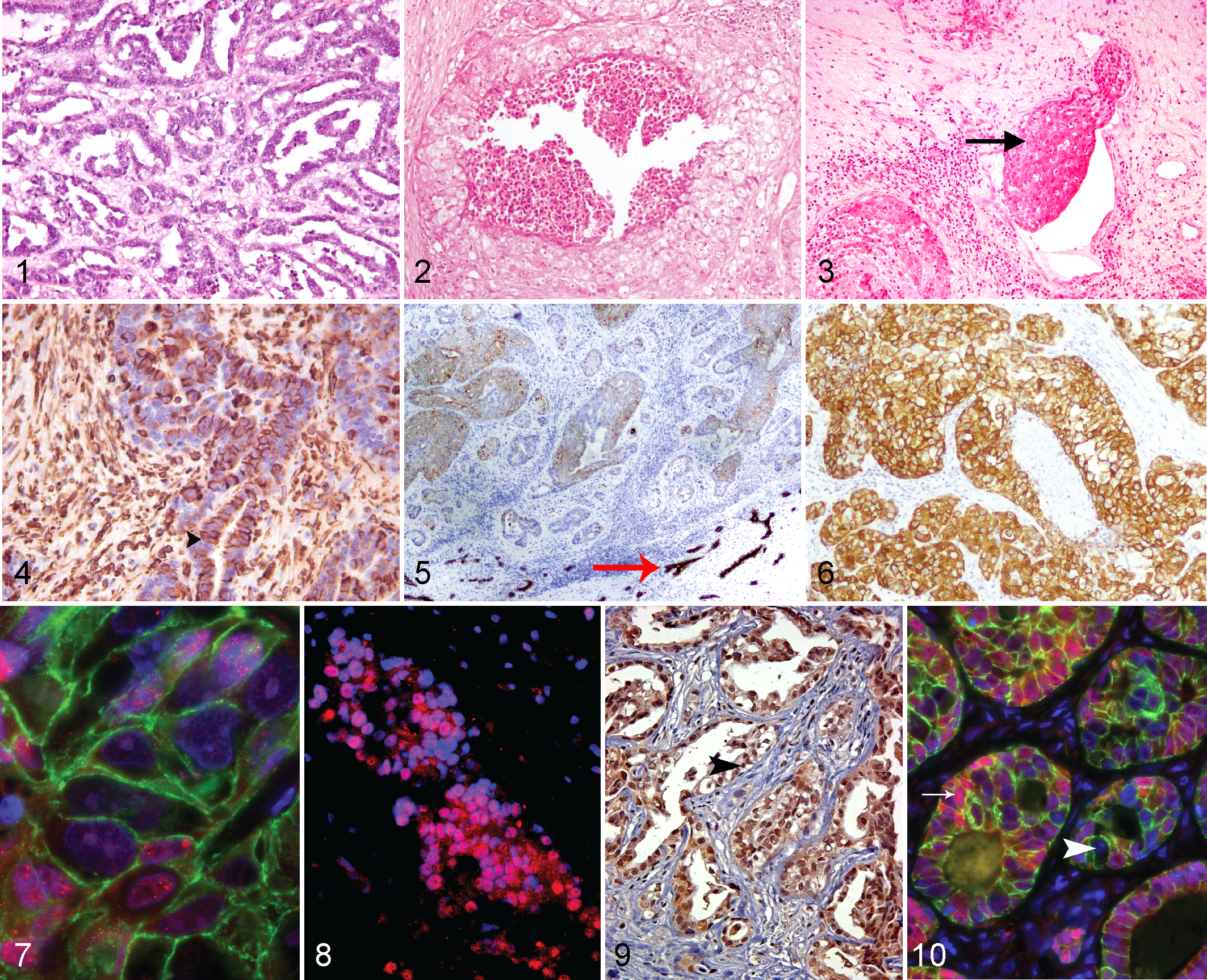
Mammary carcinoma, horse, mammary gland. Figures 1–3. Hematoxylin and eosin (HE).
Moderate to intense vimentin staining of the majority of the neoplastic cells was observed in 4 of the cases examined (Fig. 4). Pan-cytokeratin staining was generally mild to moderate in intensity, despite strong staining in adjacent nonneoplastic mammary tissue (Fig. 5). It was noted that in control quiescent mammary tissue, the pan-cytokeratin antibody employed preferentially stained luminal epithelial cells, with weaker or absent staining observed in cells with location and morphology consistent with basal epithelial cells (Suppl. Fig. 1). Consequently, a specific basal cytokeratin 14 stain was also employed to determine if the tumors exhibited a basal phenotype (Suppl. Fig. 2 and Fig. 6). Two of 7 cases (Nos. 1 and 6) exhibited intense staining for cytokeratin 14 (Fig. 6), while less than 20% of the neoplastic cells examined exhibited moderate levels of cytokeratin 14 staining in horse Nos. 3 and 7.
Three of 7 cases (horse Nos. 1, 4, and 5) exhibited strong nuclear staining for ERα (Figs. 7, 8), while 3 of the 7 equine mammary carcinomas (horse Nos. 4, 5, and 7) exhibited strong nuclear expression of STAT3 (Figs. 9, 10). Expression of the intercellular adhesion molecule E-cadherin was strong in all 7 cases (Figs. 7, 10).
Discussion
Where available, clinical history suggested that the mammary masses had frequently been present for some time, and 2 cases (horse Nos. 1 and 5) had clinical characteristics suggestive of mastitis. A presenting clinical suspicion of mastitis has been described previously, 3 and mastitis may be a secondary complication of a mammary tumor. 11
A previous case report has demonstrated staining for estrogen receptor and progesterone receptor in an invasive micropapillary carcinoma in a mare. 2 In the present report, we use immunofluorescence to demonstrate notable levels of nuclear ERα staining in 3 of the 7 carcinomas examined. Assessment of estrogen receptor positivity is fundamental to breast cancer diagnosis and prognostication in humans, and expression has been confirmed in both canine mammary tumors and their feline counterparts. The demonstration of nuclear ERα expression in equine mammary tumors is exciting as it suggests that hormonal influences may also potentially play a role in the pathogenesis of a subset of mammary tumors in herbivorous domestic species, despite their differing clinical characteristics compared with their canine counterparts.
ERα expression status did not correlate with a particular histopathologic pattern in the present case series. However, although it is not possible to draw firm conclusions from analysis of 7 cases, it is interesting that ERα-positive staining tended to be observed in tumors with weak or absent vimentin staining. This correlates with recent evidence suggesting that in humans, vimentin expression, which tends to be associated with epithelial-mesenchymal transition (EMT), is higher in triple-negative breast cancers than in other subtypes such as those expressing estrogen receptor. 12
Pan-cytokeratin staining in these tumors was generally mild to moderate in intensity, which is in contrast to a previous report where equine mammary ductal carcinoma cells were strongly positive for pan-cytokeratin (clone Lu-5). 3 Notably even in horse No. 6, in which approximately 60% of the neoplastic cells exhibited mild to moderate cytoplasmic expression of cytokeratin, staining was considerably weaker in the neoplastic cells than in adjacent nonneoplastic mammary tissue. In nonneoplastic tissue, luminal epithelia exhibited intense staining with the pan-cytokeratin antibody, while weaker staining was noted in cells with location and morphology consistent with basal epithelia. Consequently, a specific basal cytokeratin 14 stain was also employed. Two mammary carcinomas (horse Nos. 1, 6) exhibited intense staining with cytokeratin 14, while horse No. 3 exhibited a moderate degree of staining, suggesting a basal origin for these tumors. In 2 cases (horse Nos. 3, 6), cytokeratin 14 expression correlated with an absence of ERα expression, as would be expected. However, curiously, in horse No. 1, intense cytokeratin 14 expression coincided with ERα positivity. Although uncommon, a small proportion of human basal-like breast cancers are reported to exhibit ERα positivity, determined by gene expression profiling. 5
Membranous expression of E-cadherin was strong in all 7 cases; in the one carcinoma that was known to have metastasized (horse No. 2), expression remained strong in both the primary tumor and pulmonary metastases. This is consistent with the human breast cancer literature, where it has been demonstrated that E-cadherin expression in metastatic ductal carcinomas may be the same intensity or stronger than in the equivalent primary masses. 6 It is traditionally considered most likely that E-cadherin expression is downregulated during the process of EMT during metastasis, with subsequent reexpression in metastatic foci. However, it has recently been suggested that in some instances of human inflammatory breast carcinoma, E-cadherin may also be upregulated in tumor microemboli, favoring intravasation. 10 The immunofluorescence findings in the present study could be compatible with either hypothesis.
Interestingly, strong nuclear localization of STAT3 was detected in 3 of the 7 equine mammary carcinomas (horse Nos. 4, 5, and 7), suggesting a potential role for STAT3 activity in a subset of equine mammary carcinomas. These cases exhibited a tubulopapillary to solid growth pattern and in 2 cases exhibited convincing ERα expression, tentatively suggesting that a more luminal phenotype might be associated with nuclear STAT3 expression. The 3 cases that exhibited moderate to intense positive staining for cytokeratin 14 (horse Nos. 1, 3, and 6), indicating a more basal phenotype, exhibited rare to absent nuclear STAT3 expression, potentially further supporting this interpretation. In human breast cancer, some authors have suggested a correlation between STAT3 expression and the luminal subtype, 7 although this is not consistently demonstrable. 1
A major limitation of this study is that the small number of available cases precludes any meaningful correlations or prognostic assertions. Given the rarity of equine mammary tumors, wide-ranging multicenter collaborations are required to build adequate case material for any such prognostic or molecular relationships to be adequately interrogated. Such investigations would also require comprehensive clinical follow-up. Although all of the cases for which outcome data were available were euthanized following diagnosis, the records available do not indicate whether this decision was based on clinical condition of the mare or prognostic advice from the attendant veterinary surgeon.
Taken together, our histopathologic data confirm previous reports that equine mammary tumors have an invasive and malignant phenotype. Importantly, the histologic phenotype and molecular characteristics appear to be heterogeneous, with variable expression of intermediate filaments, ERα, and nuclear STAT3. The role of STAT3 activity in equine mammary tumors merits further investigation, particularly to confirm or refute the potential inverse correlation with cytokeratin 14 expression. Our findings also confirm that a subset of equine mammary tumors exhibit ERα expression, which suggests that these tumors may have similar molecular characteristics to their feline and canine counterparts.
Footnotes
Acknowledgements
Some of the data detailed in this manuscript were presented as a short communication at the 2013 ACVP Annual Meeting, Montreal, Canada, November 16–20, 2013 (presentation: November 19). We thank the referring veterinary surgeons for submission of these cases, as well as M. Fordham, L. Webster, and T. M. Hoather of the Department of Veterinary Medicine, University of Cambridge, for technical assistance in preparation of tissue sections and automated immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Histology costs were supported by an RCVS Trust Small Project Grant (SG10 1185).
