Abstract
Although there have been several studies on the use of immunohistochemical biomarkers of canine mammary tumors (CMTs), the results are difficult to compare. This article provides guidelines on the most useful immunohistochemical markers to standardize their use and understand how outcomes are measured, thus ensuring reproducibility of results. We have reviewed the biomarkers of canine mammary epithelial and myoepithelial cells and identified those biomarkers that are most useful and those biomarkers for invasion and lymph node micrometastatic disease. A 10% threshold for positive reaction for most of these markers is recommended. Guidelines on immunolabeling for HER2, estrogen receptors (ERs), and progesterone receptors (PRs) are provided along with the specific recommendations for interpretation of the results for each of these biomarkers in CMTs. Only 3+ HER2-positive tumors should be considered positive, as found in human breast cancer. The lack of any known response to adjuvant endocrine therapy of ER- and PR-positive CMTs prevents the use of the biological positive/negative threshold used in human breast cancer. Immunohistochemistry results of ER and PR in CMTs should be reported as the sum of the percentage of positive cells and the intensity of immunolabeling (Allred score). Incorporation of these recommendations in future studies, either prospective or retrospective, will provide a mechanism for the direct comparison of studies and will help to determine whether these biomarkers have prognostic significance. Finally, these biomarkers may ascertain the most appropriate treatment(s) for canine malignant mammary neoplasms.
Keywords
In the intact adult female dog, spontaneous mammary tumors are the most common neoplasm; malignant tumors account for up to 50% of cases. 109,112 Thus, canine mammary tumors (CMTs) have been the focus of intense research by veterinary oncologists and pathologists for the past decades.
Immunohistochemistry (IHC) has, in parallel with conventional histopathology, played an increasing role as a diagnostic tool for the identification of neoplasms and pathogens. 29,34,66 In human breast pathology, IHC is routinely used to assist with the prognosis and to determine the specific treatment for patients. 85 In dogs with mammary tumors, IHC is not routinely used, although there have been an increasing number of studies looking for reliable diagnostic and/or prognostic IHC biomarkers during the past 15 years. However, these studies cannot be compared because of the differences in how these biomarkers have been used to label mammary neoplasms and because of differences in how they have been evaluated.
In this article, we provide an overview of the most useful and promising immunohistochemical markers in canine mammary tumor pathology and address practical questions related to the methodology. By standardizing the methods used for labeling and interpreting these IHC markers, we aim to ensure consistency and reproducibility in future study results.
Anatomy, Histology, and Immunohistochemistry of the Normal Canine Mammary Gland
The mammary gland is found exclusively in mammals and forms the glandular tissue of a mamma. 17,113 Dogs usually have 10 mammae, each characterized by a corpus mammae (made up of 8–14 lobules, connective tissue, and skin) and a papilla mammae (nipple). 17 The mammary gland is a modified apocrine sweat gland, characterized by a tubuloalveolar structure. 113,123 Its full development only occurs in the adult female, during pregnancy, at which time there is ductal epithelial cell proliferation and lobuloalveolar differentiation. At parturition, the mammary gland consists of a complex ductular-lobular-alveolar structure, associated with alveolar secretion. 113
The epithelium throughout the ductal-lobular system is composed of a dual-cell population of luminal epithelial and basal myoepithelial cells, juxtaposed to a continuous basement membrane. 113 Larger ducts are lined by a bilayered cuboidal epithelium, whereas the smaller ducts have a single layer of cuboidal epithelium, and all ducts are surrounded by fusiform myoepithelial cells. 113 Nonsecretory alveoli are similar to the small ducts, whereas secretory alveoli have a cuboidal to columnar luminal epithelium, with variable numbers of intracellular fat droplets surrounded by star-shaped myoepithelial cells. 113
It is important to understand that the canine mammary gland is a hormone-dependent organ whose cyclic activity is associated with consecutive development and regression phases that differ between individual glands and also within each gland. 93,100 For example, during diestrus, there is complete mammary lobuloalveolar and secretory differentiation, 93,100 which is due to the long-lasting and functional corpus luteum that produces high progesterone levels independent of pregnancy. 100
A canine mamma is the homologue of a single human breast, 17 which has similar developmental characteristics. 16 The majority of breast development occurs during puberty, but terminal differentiation is induced only by pregnancy. 16 Recent evidence suggests that in the human breast, a common cell of origin exists for both epithelial and myoepithelial cells; these progenitor cells are suprabasally located between the luminal and basal layer. 11–13
Normal canine mammary gland cells have a distinct immunoprofile (Figs. 1–6): luminal epithelial cells are characterized by the expression of type I acidic keratins K18 and K19 (previously called cytokeratins) 103 and type II basic keratins K7 and K8. Basal/myoepithelial cells express type I acidic keratins K14 and K17 and type II basic keratins K5 and K6. Myoepithelial cells also express other markers such as p63, vimentin, P-cadherin, CD10, epidermal growth factor receptor (EGFR), maspin, and 14-3-3 sigma protein.23, 27, 35, 35, 38–40, 46, 52, 99, 113, 114, 123 In addition, due to its contractile phenotype, myoepithelial cells also express smooth muscle–specific proteins such as smooth muscle actin (SMA) and calponin (Table 1). 23,28,35,40,123
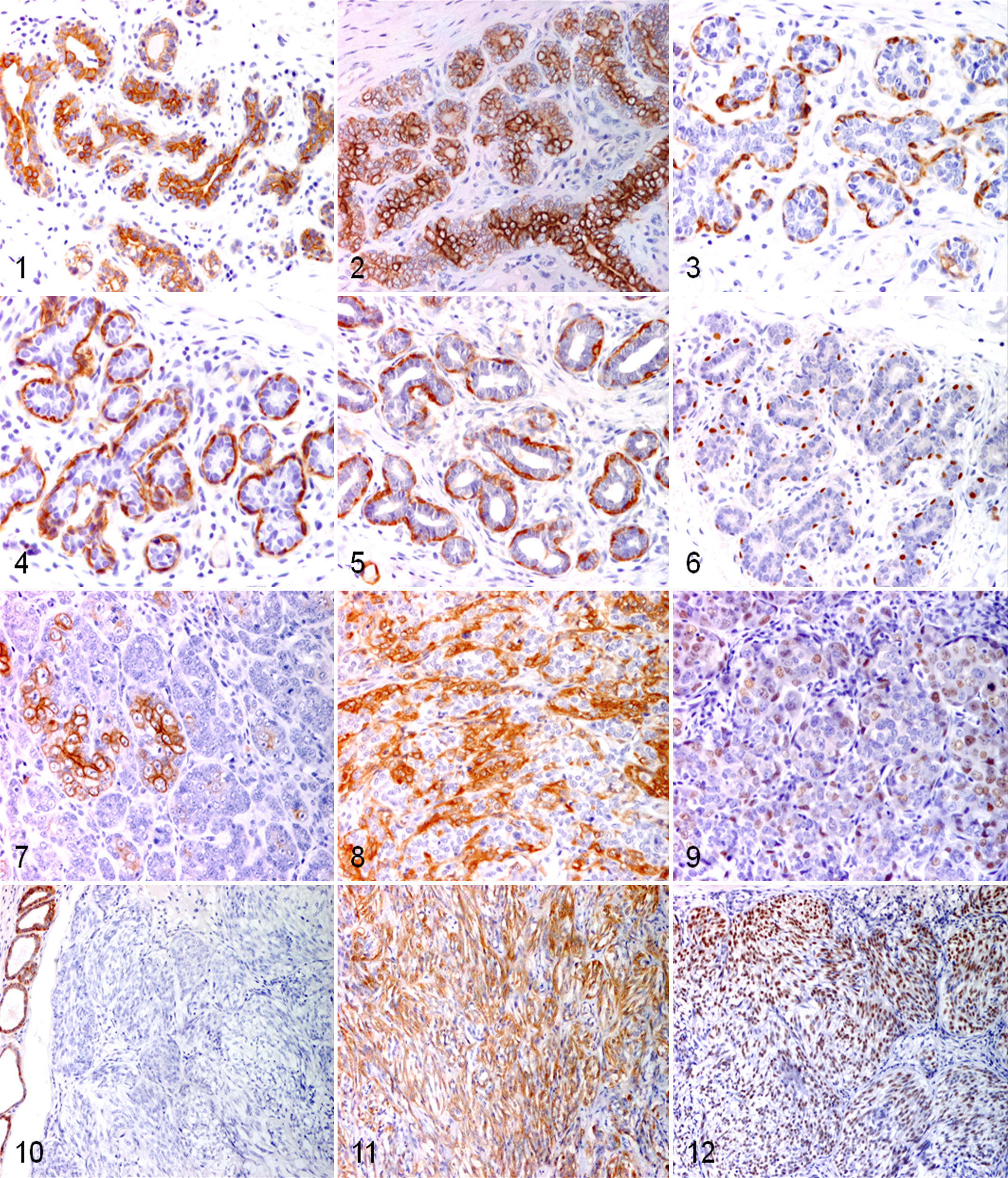
Normal mammary gland; dog. Immunohistochemistry for CKAE1-AE3 showing positive staining of the luminal cells and basal/myoepithelial (less intensely stained) cells. CKAE1AE3, ABC method, counterstained with hematoxylin.
Immunophenotypic Characteristics of Mammary Epithelial Cells.

K, keratin; SMA, smooth muscle actin.
aMyoepithelial cells exhibit a diffuse CD10 cytoplasmic labeling, whereas luminal cells exhibit a “Golgi-like” cytoplasmic labeling.
Human breast luminal epithelial and myoepithelial cells have similar immunohistochemical features. 16 The different steps of cell differentiation have not been precisely defined. 11 Using immunofluorescence, Bocker et al 12 demonstrated that in both human luminal and myoepithelial cell lineages, the cells seem to exist at intermediate stages of maturation, expressing various combinations of markers. A small population of cells that are K5 positive but negative for K8, K18, K19, and SMA have been identified. These cells have morphological features of stem cells that have the capacity to differentiate toward either the glandular or the basal phenotype. 12
Cell Differentiation Markers for the Diagnosis of Canine Mammary Tumors
During mammary tumorigenesis, cell-specific differentiation markers are usually retained. Morphologic evaluation in conjunction with these immunophenotypic differences can be used to help provide a definitive diagnosis. 54,113 The diagnosis of canine mammary tumors is one of the more frequent tasks of veterinary pathologists in countries where prepubertal or pubertal ovariohysterectomy is not routinely performed in the female dog, since mammary tumors are a very common finding. 6,112
CMTs are characterized by a varied morphology, forming simple (epithelial luminal or myoepithelial cells), complex (epithelial luminal and myoepithelial cells), mixed (epithelial luminal and/or myoepithelial cells, and osseous/cartilaginous metaplastic tissue), and mesenchymal tumors; tumors characterized by myoepithelial proliferation are also frequently observed. 44,77,78 Benign mixed tumors are very common and are formed by the proliferation of benign glandular epithelial and myoepithelial cells with mesenchymal metaplastic elements, primarily cartilage and bone. 44,77,78
Carcinomas are the most common malignant CMTs and are classified according to their morphological features. In simple carcinomas, there is the proliferation of only 1 cell type (luminal epithelial or myoepithelial cells), whereas complex carcinomas are characterized by a dual-cell proliferation (luminal epithelial and myoepithelial cells). 77 In simple carcinomas, there is increasing malignancy from tubulopapillary to solid to anaplastic subtypes. Complex carcinomas are usually associated with a better outcome. 77 True malignant mixed tumors (carcinosarcomas) and sarcomas are uncommon. 53,77 On routine diagnosis, the complex morphology of CMTs may make it difficult to identify the different types of proliferating cells, which is particularly important because of the different prognoses associated with the different histologic subtypes. 78 Although the CMTs classification published by the World Health Organization (WHO) in 1999 may still be applied in routine veterinary diagnostic pathology, 78 a new histological classification scheme was recently published, including several new histological types: cribriform type of simple carcinoma, comedocarcinoma, micropapillary carcinoma, and carcinoma and malignant myoepithelioma. 44 Based on the new classification for CMTs, IHC may be required for a definitive diagnosis. 44 Although myoepithelial cells can be easily recognized in some tumors, in others it is more difficult to identify these cells on routine histopathological evaluation. Cell differentiation markers used in studies of CMTs include markers of epithelial cells, markers of myoepithelial cells, and markers of mesenchymal cells.10, 23, 27, 28, 35, 36, 40, 46, 52, 71, 91, 92, 98, 99, 123
Historically, cell differentiation markers have been used to study the histogenesis of canine mammary tumors, primarily the role of myoepithelial cells in the genesis of mixed tumors based on their immunophenotype. 28,35,113 Several theories have been proposed for the origin of the osseous and cartilaginous metaplastic elements based on IHC studies of spontaneous canine mammary tumors: metaplasia from the epithelial cells, metaplasia from the stromal connective tissue, or metaplasia from the basal/myoepithelial cells. 28,78,113 A number of IHC studies support a myoepithelial cell histogenesis based on immunophenotype characteristics 10, 23, 28, 35, 40, 46, 91, 117 and have found that myoepithelial cells can undergo metaplastic transformation to mesenchymal cells by mechanisms that are not completely understood. Whereas suprabasal myoepithelial cells retain their immunoprofile, interstitial myoepithelial cells lose the expression of epithelial markers (keratins) and acquire a fibroblastic-like phenotype with increased vimentin expression. 10,40 These immunohistochemical studies support the epithelial-mesenchymal transition (EMT) hypothesis involving myoepithelial cells, which will undergo a myoepithelial-mesenchymal transition (MMT). 10 However, experimental and gene expression–based studies might favor another explanation, including the origin of mesenchymal components from mammary stem cells. 53,128,129
Several myoepithelial cell markers are available that have an exclusive or preferential myoepithelial expression pattern (Table 2). However, these antibodies display differences in terms of sensitivity and specificity. 10,54 Smooth muscle cell markers (calponin, SMA) are sensitive, but in addition to myoepithelial cells, they also label stromal myofibroblasts and vascular smooth muscle cells (in human breast tissues, calponin stains stromal myofibroblasts less strongly than SMA). 22,24,80 P63 is a highly sensitive and specific nuclear myoepithelial marker, and one of its advantages is that it is not expressed by myofibroblasts and vascular smooth muscle cells. 24 Basal keratins (K5 and K14) label myoepithelial cells and do not label stromal fibroblasts, but they have a low specificity because some epithelial cells (normal and neoplastic) will express basal-type keratins. 24,28,35,71
Antibodies Commonly Used to Identify Mammary Myoepithelial Cells (MECs) in Canine Mammary Tumors.
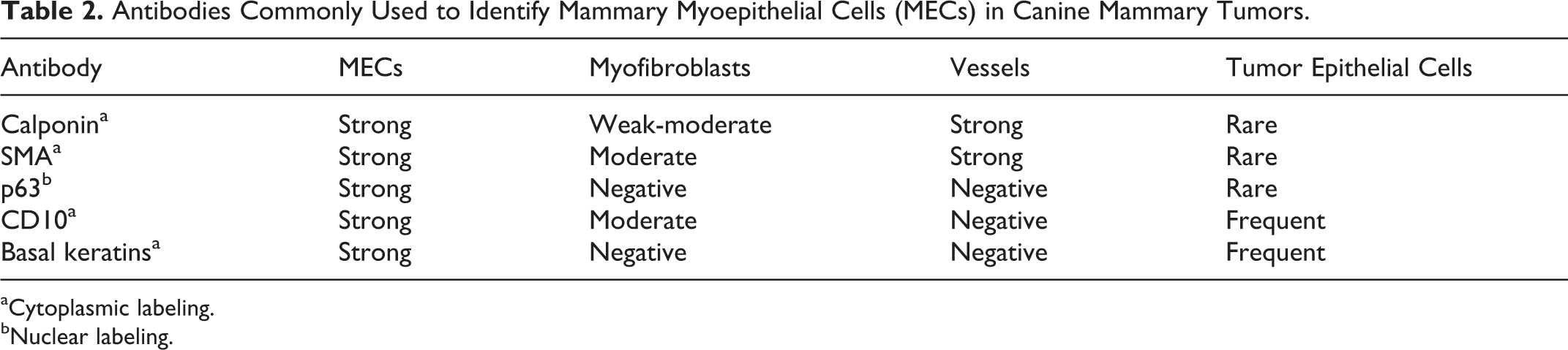
aCytoplasmic labeling.
bNuclear labeling.
The frequent participation of myoepithelial cells in CMTs and the variable morphology they can adopt often increases the difficulty of providing an accurate morphologic diagnosis. Four myoepithelial cell morphologies have been observed in complex and mixed canine mammary tumors: resting and proliferating suprabasal myoepithelial cells, as well as interstitial spindle and stellate myoepithelial cells. 10,28,35,117 A basal/myoepithelial cell phenotype has been described in a subset of neoplasms, traditionally considered simple carcinomas, using immunohistochemical myoepithelial cell markers. 23,35,37,46,71
Carcinoma and malignant myoepithelioma, a recently described entity, consists of a dual population of luminal epithelial cells and myoepithelial cells. 44 The myoepithelial cells may show marked differences in their morphology ranging from spindle cells to round cells to polygonal cells. There is moderate to marked positive staining with myoepithelial cell markers and variability in the proportion of cells that stain positive. 92 Thus, the type of expression might be influenced by the level of differentiation of the proliferating myoepithelial cells. Carcinoma and malignant myoepithelioma requires IHC to identify the dual population of luminal epithelial cell and myoepithelial cells (Figs. 7–9). 44
In humans, fibroadenomas are the most frequent benign tumors, whereas the most common type of breast carcinoma is the so-called invasive ductal carcinoma not otherwise specified (IDC-NOS) or of no special type (IDC-NST). 118 IDC-NST is a diagnosis of exclusion and comprises those carcinomas that fail to exhibit sufficient characteristics to warrant their classification as a special type of carcinoma. 127 Breast cancer special types account for up to 25% of all breast cancers. The WHO classification recognizes the existence of several distinct histological special types: lobular (5%–15%), tubular (2%), cribriform (0.8%–3.5%), medullary (1%–7%), papillary (1%–2%), and micropapillary (2%) carcinoma. 118 In contrast to dogs, myoepithelial proliferation is uncommon in human breast cancer. Tumors showing differentiation to myoepithelial cells, such as adenomyoepithelioma or pure malignant myoepithelioma, are rare and usually have a predominantly spindle cell growth pattern. 33,90
Although biphasic tumors are associated with decreased malignancy in dogs 77,131 and humans, 33 malignant myoepithe-liomas have a distinct clinical behavior and a poorer prognosis in humans. 33 Tsuda et al 121 showed that high-grade human breast tumors exhibiting myoepithelial differentiation have an aggressive behavior. Additional studies are needed to understand the role of myoepithelial cells in the development of canine mammary carcinomas and their prognostic significance in these neoplasms. 71 In human studies, the importance of myoepithelial cells in breast tumor progression has always been underestimated; however, it is now believed that these cells appear to have a dual function, acting both as tumor suppressors and promoters. Myoepithelial cells are considered natural tumor/invasion suppressors by the secretion of various anti-angiogenic and anti-invasive factors such as maspin or TIMP1. However, myoepithelial cells associated with ductal carcinoma in situ show abnormal behavior, lose their normal tumor suppressor functions, and promote stromal invasion (by secreting various molecules implicated in the degradation of the basement membrane), hypoxia, and inflammation. Yet, the underlying molecular mechanisms of myoepithelial cell functions during tumor progression are still unclear. 84
Human breast cancers have been extensively studied by IHC—namely, the distribution of cytoskeletal proteins such as keratins. 1 Most carcinomas exhibit positive labeling for luminal keratins, indicating a differentiated glandular phenotype. 1 However, it has become increasingly clear that some breast cancers (such as IDC-NST carcinomas) show partial or complete basal/myoepithelial differentiation on immunohistochemical evaluation 90 and are characterized by the expression of molecules normally seen in the basal/myoepithelial compartment of the normal breast. 90,121 This “basal” differentiation has raised the attention of pathologists since these tumors have an aggressive biological behavior and poor patient prognosis (see below). 121
Routinely, one of the applications of cell differentiation IHC markers relies on the classification and differential diagnosis of unusual human breast lesions. 54 Myoepithelial markers can be useful not only to address stromal invasion (see below) but (1) to differentiate radial scars (a benign lesion characterized by a fibroelastotic core with entrapped ducts demonstrating a dual myoepithelial and epithelial layer, with surrounding radiating ducts and lobules) 62 from tubular carcinomas (due to myoepithelial cells attenuation in the central parts of the radial scars), (2) for the diagnosis of breast papillary lesions (in conventional intraduct papillomas, a complete layer of myoepithelial cells is present, whereas in solid papillary carcinoma, myoepithelial cells are absent), (3) in the classification of (adeno)myoepithelial cell tumors, and (4) for the diagnosis of adenoid cystic carcinoma in core biopsies (positive for SMA and p63 but negative for calponin). 24,124
Recommended Guidelines
According to standardization in human IHC, cell differentiation marker IHC assays are considered class I tests, meaning that their interpretation is carried out in the context of the tumor’s histomorphologic characteristics, and the results are used only by pathologists and should be interpreted as negative or positive. 120 In general, in humans, in class I tests, positive IHC labeling is quantitatively defined as the presence of more than 10% of immunoreactive cells and is used in conjunction with the cellular localization of the evaluated antigen. 120 A threshold of 10% of positive cells has been used for many antibodies in several studies of CMTs, and thus this should be considered the standard parameter for the evaluation of these markers.
Class II IHC tests are used in humans for prognostic or predictive purposes, and therefore their immunohistochemical interpretation follows more rigid guidelines. 120 HER2 (human epidermal growth factor receptor 2), estrogen receptor (ER), and progesterone receptor (PR) represent class II IHC markers, and they are interpreted differently as they consider the evaluation of the intensity of the immunoreaction together with the percentage of IHC-labeled cells 120 (see below).
We recommend the use of an antibody panel, including IHC markers, for luminal (K8, K18, and K19) and myoepithelial (p63, calponin) cells. Similarly, to confirm the diagnosis of malignant myoepithelioma, a rare spindle cell neoplasm characterized by the proliferation of myoepithelial cells, an immunohistochemical approach is mandatory to differentiate it from fibrosarcomas and other spindle cell neoplasms, both epithelial and mesenchymal. To confirm the presence of myoepithelial cells in all these situations, the use of an immunohistochemical panel of epithelial (pan-keratin), mesenchymal (vimentin), and myoepithelial cell markers is recommended. The positive labeling for pan-keratin will confirm the epithelial nature of the neoplasm; the positivity for at least 1 myoepithelial marker (p63, calponin, SMA) confirms the diagnosis of malignant myoepithelioma (Figs. 10–12). Care must be taken in the choice of antibodies for the IHC identification of myoepithelial cells and the interpretation of the results, keeping in mind that neoplastic myoepithelial cells can lose their myoepithelial traits. In our opinion, it is advisable not to rely on only 1 antibody to identify myoepithelial differentiation: at least 2 myoepithelial markers should be used. Although all of the above-mentioned antibodies may be used, at present, the most suitable markers are p63 and calponin.
Assessment of Stromal Invasion
In situ carcinoma is defined in human breast cancer as a malignant epithelial proliferation confined to the mammary ductal system that is surrounded by an outer layer of myoepithelial cells and the basement membrane, whereas invasive carcinoma infiltrates and grows beyond the basement membrane. 135 This morphologic distinction is often difficult and can be challenging in routine histological sections. 54 In humans, myoepithelial cell markers are commonly used to assess stromal invasion. By identifying the loss of the myoepithelial cell layer, invasive carcinomas can be differentiated from in situ carcinomas. 134
In CMTs, however, the loss of the myoepithelial cell layer integrity does not appear to identify in situ carcinoma, especially in complex tumors with myoepithelial interstitial proliferation (Figs. 13–15). 98,113 Therefore, this IHC application might not be relevant in dogs as myoepithelial markers would not help to distinguish in situ from invasive carcinomas. 98,113 P63 may produce a discontinuous nuclear labeling pattern, giving the impression that myoepithelial cells are absent. 24
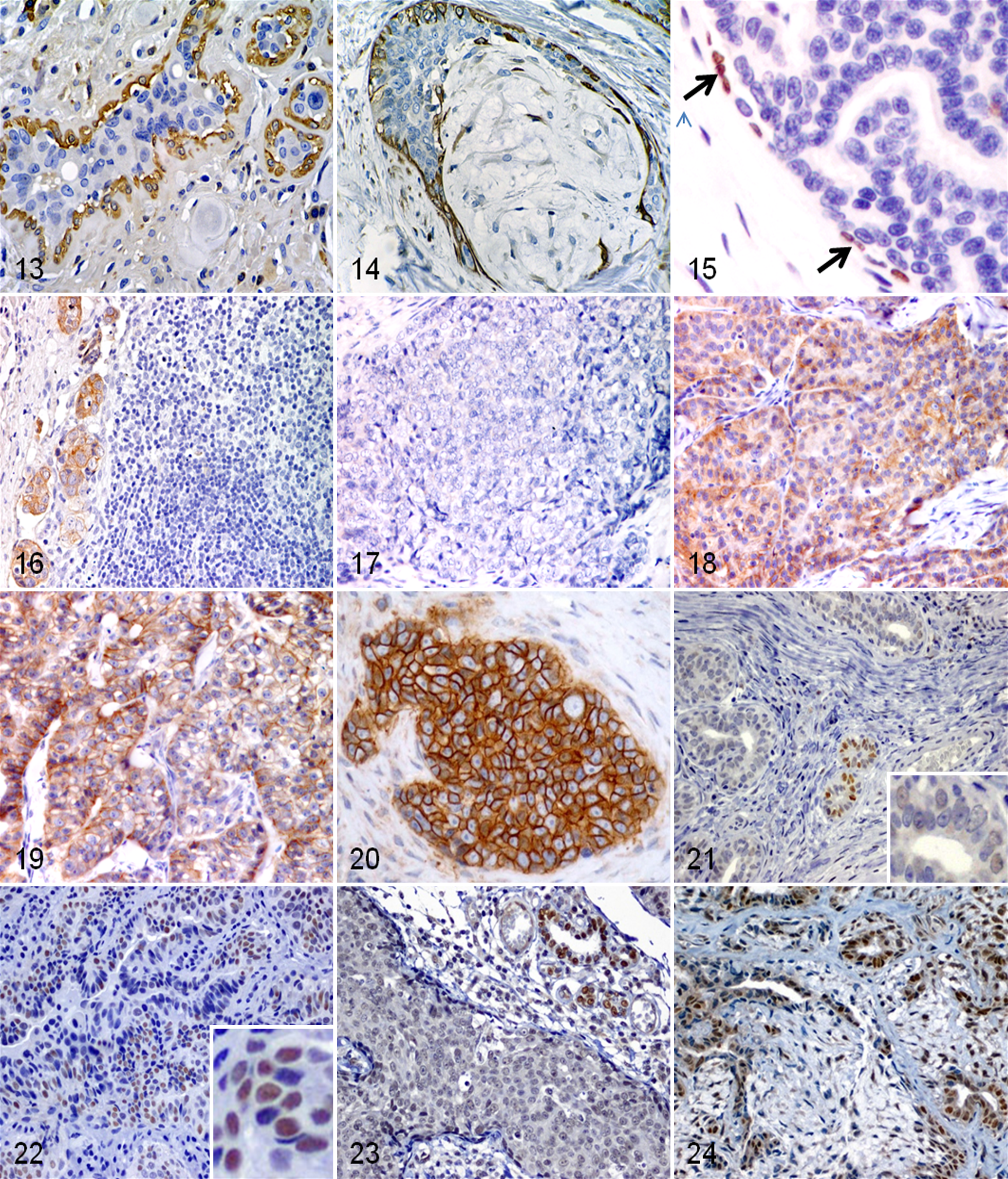
Mammary gland, carcinoma mixed type; dog. Immunohistochemistry for calponin. Irregular nests of atypical luminal negative epithelial-type cells surrounded by a complete layer of hypertrophic calponin-positive cells, which documents the integrity of the myoepithelial cell layer. Calponin, ABC method, counterstained with hematoxylin.
Recommended Guidelines
Although myoepithelial cell layer integrity is not definitive for the diagnosis of malignancy in canine mammary tumors, 98 in simple carcinomas, in which there is no myoepithelial proliferation, a combination of p63 (nuclear reactivity) and calponin or basal keratins (K5, K14) (cytoplasmic reactivity) is recommended to determine whether there is stromal infiltration as they label different cellular compartments of the myoepithelial cell, thereby complementing one another. 54,98
Lymph Node Micrometastasis Detection and Circulating Tumor Cells
Although used in human pathology, the identification of occult metastatic disease by IHC is controversial, and the clinical significance of such findings is unclear. 54,113,126 Recent data found there was no clinical advantage of the IHC evaluation of negative sentinel nodes for micrometastases in women with breast cancer. 126 It is important to recognize that in human medicine, isolated tumor cells (ITCs) or tumor cell aggregates less than 0.2 mm in diameter are considered as N0 rather than as true metastases. 124 Recently, Matos et al 72 detected micrometastases in about 9% of the cases of hematoxylin and eosin (HE) morphologically negative lymph nodes from dogs with malignant mammary tumors. In another study conducted in dogs with mammary carcinomas, a significant difference in survival was found between bitches without metastases and bitches with lymph node macrometastases greater than 2 mm, whereas there was no significant difference when the micrometastases were less than 2 mm. 116 The significance of detecting isolated tumor cells in canine lymph nodes is unknown.
Although not described in canine species, benign glandular inclusions constitute a well-documented uncommon finding that can mimic metastatic carcinoma when observed in axillary nodes from human patients undergoing breast surgery. 19 These inclusions are often located in the capsule of the node and consist of uniform cells that form glandular spaces that could contain cilia and may also be surrounded by myoepithelial cells. 19,54
In human breast cancer research, several studies have focused on the development of sensitive assays that allow detection of circulating tumor cells (CTCs) in the peripheral blood of cancer patients, allowing the detection of early metastatic spread. 3 CTCs are rare events and constitute a prognostic factor for survival: the identification of CTCs ≥5 cells per 7.5 ml of blood seems to be a reliable identifier of human patients at higher risk for disease progression and decreased survival. 51,115 Recently, a few studies have described potential markers for the detection of CTCs in CMTs, but further studies are needed to evaluate the predictive potential of CTCs for the disease outcome in dogs. 20,21
Recommended Guidelines
Immunohistochemistry for keratins (using an anti–pan-keratin antibody, such as clone AE1/AE3) is a valuable tool for the detection of carcinoma micrometastases or ITCs in lymph nodes considered negative on routinely stained sections (Fig. 16). To reduce the probability of missing macrometastases larger than 2 mm, we suggest that multiple sections of the regional lymph nodes, at approximately 2-mm intervals, are embedded and sectioned, even when there is no gross disease (according to recommendations for pathologic evaluation of sentinel lymph nodes in human breast cancer 125 ). IHC on morphologically negative sections may be useful in detecting subtle metastatic involvement, such as the presence of single neoplastic cells. 54 Prospective studies designed to study the clinical relevance of micrometastases detected by IHC in CMTs require further investigation.
HER2 (Human Epidermal Growth Factor Receptor 2)
In human breast cancer, HER2 expression is routinely used as a prognostic and predictive factor. 85 HER2 overexpression is found in up to 30% of human breast carcinomas and is associated with gene amplification in 85% to 90% of cases. 55,60 In humans, HER2 overexpression correlates with aggressive clinicopathological features, such as the development of metastases, sensitivity to specific systemic therapies, and drug resistance. 15,95,108,132
In dogs, the significance of HER2 overexpression is unclear in mammary tumors. Although there are an increasing number of IHC studies on this topic, there is great variability in the results. In 1996, Ahern and coworkers 2 determined that benign CMTs showed no HER2 mRNA levels, in contrast to most malignant CMTs, which were positive, implying that HER2 may play a role in the malignant progression of these neoplasms. However, subsequent IHC studies on HER2 expression have not been consensual, with overexpression rates in malignant tumors ranging from 17.6% to 48%. 37,58,70,97,101 Furthermore, there is no agreement on its prognostic value. 26,37,56,70,94
Recommended Guidelines
In our opinion, these discrepancies are, in part, related to variations in IHC protocols and evaluation systems used, which hamper any definitive conclusions on the role of HER2 in CMTs.
To overcome interlaboratory discordances, HER2 IHC testing has been standardized in human breast cancer. The HercepTest (DAKO, Glostrup, Denmark) was the first IHC method approved by the US Food and Drug Administration (FDA) to determine HER2 expression, and an internationally accepted evaluation system for HER2 IHC has also been recommended (HercepTest scoring). 130 According to recent guidelines proposed by the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP), 130 IHC for HER2 should be scored as 0, 1+, 2+, and 3+ (Figs. 17–20), and only 3+ tumors are considered positive (Table 3). Until recently, most CMT studies have not followed this standardized protocol. Although the evaluation system used was frequently the HercepTest scoring, both 2+ and 3+ cases were considered positive. 26,37,56,58,70,94,101 In an attempt to standardize CMT HER2 IHC, we therefore recommend the use of approved IHC methods, following the recent human guidelines. 130
Interpretation Criteria for HER2 Immunohistochemistry (IHC) in Canine Mammary Tumors: A Proposal Based on a Human Breast Cancer System.

aAccording to the guidelines proposed by the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP). Only 3+ tumors are considered positive. 130
Preanalytical, analytical, and postanalytical (interpretation) factors must be addressed: 130 HER2 testing requires fixation in 10% neutral buffered formalin, optimal fixation times should be 6 to 48 hours, IHC should be evaluated only in invasive carcinomas or in its invasive component, interpretation criteria should be standardized, and a tumor should be considered positive when more than 30% of tumor cells show an intense and uniform circumferential membrane labeling (chicken-wire pattern). If cytoplasmic labeling obscures the membrane labeling, the assay should be repeated. Exclusion criteria include the use of fixatives other than 10% neutral buffered formalin, inadequate fixation times, tissues with strong membrane labeling of internal normal ducts or lobules, and tissues where the controls produce unexpected results. Commercially available control slides may be used to validate a HER2 IHC protocol; these slides contain 4 spots of human breast cancer cells, with respective HER2 reactivity 0, 1+, 2+, and 3+.
We must stress, however, that the best immunohistochemical scoring method for HER2 is controversial in human breast cancer, with the new ASCO/CAP guidelines still not universally accepted in human medicine. Several studies refer to the classical HercepTest scoring system with a 10% cutoff, 43,61,96 despite a number of investigations demonstrating a greater concordance between fluorescent in situ hybridization (FISH) and immunohistochemical analysis with the 30% criterion. 14,48,68 Liu et al 68 showed that immunohistochemical analysis according to the ASCO/CAP criterion provided better specificity and accuracy for the detection of HER2 status in breast cancer than the previously used FDA 10% criterion, providing evidence to support the new ASCO/CAP guidelines for the assessment of HER2 status. This criterion reduces false-positive rates, but some 2+ immunohistochemical scores may represent false-negative cases. 68,105
The presence of uniformity of staining should also be addressed in canine mammary carcinomas, as intratumor genetic heterogeneity has been reported for human breast cancer, mainly associated with cases with equivocal IHC staining (2+). 67 HER2 intratumoral heterogeneity represents subclonal diversity within the tumor. 122 This finding has been linked to the observed variations in therapeutic responses and the conflicting data on the prognostic and predictive role of HER2 status in subsets of human breast cancer patients. 50,104 A recent study revealed that women with HER2 heterogeneity have decreased disease-free survival, suggesting that genetic instability, and hence aberrant HER2 amplification in subclones of such tumors, may be associated with breast cancer progression. 104 This phenomenon should be taken into account by veterinary researchers, as it may increase subjectivity in HER2 interpretation; thus, its evaluation should be preferentially performed on representative, whole tumor samples. 104
We anticipate that the application of the proposed guidelines for HER2 in future studies will standardize CMT investigations and establish the value of HER2 as a potential prognostic or predictive factor.
Estrogen and Progesterone Receptors
The detection of hormone receptors in formalin-fixed, paraffin-embedded (FFPE) samples of CMTs by IHC started at the end of the 1990s and permitted the identification of positive cells by microscopic observation of labeled nuclei. 42,45,82 Thereafter, many studies on the hormone receptor status of CMTs were conducted, focusing on ERα (referred to throughout as ER) and PR, their clinical and pathologic associations, and, to a lesser extent, prognostic value. Studies evaluating the usefulness of ER and PR as prognostic factors in dogs with mammary tumors are uncommon and include 2 retrospective 18,74 and 2 prospective ones. 69,82 Moreover, their results are inconclusive: malignant CMTs with ER expression had a better prognosis in 1 prospective multivariate study 82 but not in another, 69 while PR expression was associated with increased overall survival in a retrospective univariate study. 18 The value of ER and PR labeling as a predictive marker of favorable response to endocrine therapy has not been evaluated in CMT except for a single study. 47 Therefore, ER and PR analysis is not routinely performed in animals with CMT, which contrasts sharply with the situation in women with breast cancer, in whom ER and PR are routinely analyzed as prognostic and predictive markers of response to endocrine treatment. 49
All authors agree that ER and PR are decreased in malignant CMTs. 18,69,74,79,82,133 Nevertheless, the percentages of positive tumors vary greatly depending on the study, which can be attributed to differences in the method of detection and the different scoring systems used. Benign CMTs have been reported to be ER positive in 17.02%, 65 49%, 69 65.22%, 79 95.5%, 74 95.8%, 18 and 100% 133 of cases, and malignant CMTs have been reported to be ER positive in 10%, 57 12.96%, 65 22%, 69 46.52%, 79 50.6%, 18 58.3%, 37 67%, 133 and 92.3% 74 of cases. Similarly, different results have also been obtained for the percentage of PR-positive benign (50%, 74 96%, 69 and 100% 18 ) and malignant (23.80%, 74 66%, 69 and 71.9% 18 ) CMTs.
Many factors may be responsible for these significant differences among studies, including the use of different monoclonal antibodies raised against human ER and PR followed by different heat antigen retrieval protocols. These include anti-ER clone CC4-5,6, 30, 31, 36, 37, 41, 57, 64, 65, 82, 87, 89, 133 anti-ER clone 1D5, 7,8,10,42,69,79,81,111 anti-ER clone ER88, 58,59 anti-PR clone 10A9, 42,47,58,69 anti-PR clone 1A6, 6,87 anti-PR clone PR4-12, 74–76,101 anti-PR clone SP2, 81 and anti-PR clone hPRa2. 30,31 Furthermore, results may have been affected by factors such as delayed or inadequate fixation, nonoptimized antigen retrieval, and nonoptimized internal and external controls. 32 According to human data, 49 all ER false-negative tumors have 1 or more of the following 3 characteristics: poor fixation, negative internal controls, and absent internal controls. However, the most important reason for the nonstandardized interpretation of findings is the lack of a biological threshold of positive labeling in CMTs. 32 In human breast cancer, the positive threshold is defined by the percentage of ER and PR that ensures the effectiveness of hormone therapy, and it has been recently standardized at 1% because patients with such low levels of hormone receptor expression may still respond to hormonal therapy. 32,49 In the dog, this threshold has not been established, and the few reports on the prognostic value of ER and PR IHC detection are still contradictory. 18,69,74,82 These disagreements can be attributed to differences in obtaining the clinical data and the follow-up information, but also to the variability of IHC methods and the evaluation systems employed. The only 2 evaluation systems of ER 82 and PR 18 that were found to be prognostic measured both the percentage of positive cells and the intensity of immunolabeling by 2 different methods.
In human breast cancer pathology, the methods recommended for evaluating the proportion and intensity of positive cells are the following:
The Allred score (total score) is the sum of the score for the percentage of cells labeling positive (0 = no labeling, 1 = labeling in <1% of cells, 2 = 1% to 10%, 3 = 10% to 33%, 4 = 33% to 66%, and 5 = 66% to 100%) and a score for labeling intensity (absent = 0, weak = 1, moderate = 2, and strong = 3). The possible scores range from 0 or 2 to 8.
The H score is the sum of the percentage of weakly positive cells, the percentage of moderately positive cells multiplied by 2, and the percentage of strongly positive cells multiplied by 3. This gives a range of scores from 0 to 300.
The Quick score is the sum of the score for the percentage of cells labeled (no labeling = 0, labeling in 1% to 25% of cells = 1, 26% to 50% = 2, 51% to 75% = 3, and 76% to 100% = 4) and the score for intensity (absent = 0, weak = 1, moderate = 2, and strong = 3).
The diverse evaluation systems of ER and PR labeling that have been employed in CMTs are shown in Table 4. Some were based on preexisting methods used in human breast cancer. 4,5,73,102,106,107,110 However, although the percentage of immunolabeled cells in combination with the intensity of immunolabeling was recorded, different scoring systems were applied such as the ER content score, 45 the ERi score, 82 the H score, 42,81 and the total score (TS; a sum of the proportion score [PS] and intensity score [IS], also known as the Allred method 4 ). 7,8,18,31,69,83,119 Other evaluation systems analyzed the percentage of immunolabeled cells as a continuous variable 6,25,82,133 or as a categorical variable with different categories among studies. 30,79,111,133 The intensity of immunolabeling has also been accounted for separately as a categorical variable (generally categorized as 0, +, ++, +++), 6,57,87,133 and only 1 author provided the maximum labeling intensity of the control tissue (canine uterus). 45 The cutoff for considering ER- or PR-positive tumors also varied depending on the number of positive cells: any positive cell, 45 a cutoff of 5%, 10,30,41,65,74–76,101 a cutoff of 10%, 11,12,47,57,87 H score >20 (ranging from 0–300), 42 PS ≥1 and TS ≥2, 7,8,69,83,119 and TS ≥3. 18,31 Finally, the number of fields (named as representative) or the estimated number of cells evaluated was provided only in a few studies. Furthermore, none of the published studies on ER or PR detection in CMTs indicated the use of a computer-assisted image analyzer, so the measurements are subjective and approximate.
Scoring Systems Used for Immunohistochemistry of ER and PR in Canine Mammary Tumors.

All scoring systems indicate semiquantitative measurements. ER, estrogen receptor; IHC, immunohistochemistry; IS, intensity score; PR, progesterone receptor; PS, proportion score; TS, total score.
aBased on Andersen et al. 5
cBased on Schmitt. 102
dBased on Allred et al. 4
eBased on Shick et al. 106
fBased on Simpson et al. 107
Reproducibility of the Evaluation Methods
The low number of prognostic studies on CMTs makes it impossible to select an evaluation system based on prognostic assessments and, therefore, the intention is to provide an accurate and reproducible system to be used in future studies. Also, a unified system for possible future hormonal treatments should be considered on the basis of preliminary reports. 47
To find a more reproducible evaluation system of ER and PR labeling of CMTs that would serve as the basis for future studies looking for a biological threshold, the results of an IHC study performed in a limited number of CMTs were evaluated using all of the evaluation methods published up to date (Table 4).6, 10, 25, 30, 31, 36, 37, 41, 42, 45, 47, 57–59, 65, 69, 74–76, 79, 81–83, 87, 101, 111, 119, 133 To find the most highly agreed on and feasible method, 3 independent observers (L.P., J.M.M., and Y.M.) performed a semiquantitative evaluation. Two observers (L.P. and J.M.M.) are experienced CMTs histopathologists. As a control, a fourth observer (L.D.) measured the labeling of the 10 cases using a computer-assisted image analyzer. For this study, FFPE tissue samples of 10 CMTs (5 labeled for ER and 5 labeled for PR) were used (Figs. 21–24). These samples belonged to a previous prospective study with a controlled fixation time for 24 to 48 hours in 10% buffered formalin and were stored as paraffin blocks at 4°C between 0.5 and 2 years before use. Histological types according to the recent classification of CMTs 44 and histological grades 86 were determined. There were 3 carcinomas arising in benign mixed tumors (2 for ER and 1 for PR), 2 benign mixed tumors (ER and PR), 2 complex carcinomas grade II (PR), 1 tubulopapillary carcinoma grade I (ER), 1 solid carcinoma grade III (ER), and 1 comedocarcinoma grade III (PR). Sections of the tumors to be labeled were selected because nonneoplastic mammary gland tissue was present adjacent to each neoplasm. IHC assays were performed on 3-μm sections of FFPE tissue samples. For ER, a polyclonal rabbit anti-ER alpha (Zymed Laboratories, South San Francisco, CA) diluted 1:100 was used with a streptavidin-biotin-peroxidase complex technique (LSAB Peroxidase Universal Kit; Dakocytomation S.A., Glostrup, Denmark). Dewaxed and rehydrated sections were subjected to high-temperature antigen retrieval by incubation with 0.01 M citrate buffer, pH 6.0, at 95°C for 40 minutes in a water bath. For PR, a monoclonal mouse anti–human P clone, PRA109 (Immunotech, Marseille, France), diluted 1:500, was used with an avidin-biotin-peroxidase complex (ABC) technique (Vectastain, ABC Kit Elite; Vector Corporation, Burlingame, CA). Dewaxed and rehydrated sections were subjected to high-temperature antigen retrieval by incubation with 0.01 M citrate buffer, pH 6.0, at 95°C for 25 minutes in a water bath. All further IHC procedures were according to the instructions in the test kits. Tissue sections were counterstained with Mayer’s hematoxylin. Tissue sections of FFPE canine uterus as positive external controls and corresponding negative controls were included in every assay. These samples also had a controlled fixation time for 24 to 48 hours in 10% buffered formalin. To limit as much as possible variability of interpretation of published evaluation methods, the following criteria were adopted by all observers when analyzing the results of this study: (1) the term representative field was interpreted as representative of histological type and grade; 86 (2) if only the number of fields (or number of cells) was indicated, the observers selected fields representative of histological type and grade; (3) if the number of fields (or cells) was not indicated in the corresponding study, the observers evaluated a total of 10 high-power fields (HPF) in the tumor; and (4) in complex or mixed CMTs, myoepithelial cells were not counted.
The results of this study showed the following:
Agreement between both the semiquantitative evaluation of the 3 observers and the quantitative system was highest when evaluating the percentage of positive cells in 10 representative HPF of histological type and grade.
Semiquantitative evaluation produced higher numbers of positive cells than the quantitative evaluation.
A greater degree of agreement on the percentage of positive tumor cells was found between the 2 experienced observers (L.P. and J.M.M.) and when the number of positively labeled cells could be subdivided into smaller groups (ie, 4 groups instead of 3).
The method with the greatest agreement among observers was the Allred scoring system 4 used previously in CMTs. 18,31
Greater disagreement among observers occurred when tumors were classified exclusively as positive or negative according to the different methods.
Disagreement on the intensity of immunolabeling was high and was related to the familiarity of the observer with that particular IHC test for ER and PR and the comparison of maximum immunolabeling either with the uterus or the adjacent nonneoplastic mammary gland.
Recommended Guidelines
There are important unsolved technical issues on the assessment of ER and PR in FFPE CMT samples. The variability in methodology (including the use of different primary antibodies) and the interpretation of the IHC in various studies constitute major problems when attempting to compare the results because of differences among investigations.18, 37, 57, 65, 69, 74, 79, 82, 133
In our opinion, the standardization of the ER and PR detection technique and its evaluation in CMTs is possible and desirable (ie, definition of optimal fixation, internal and external controls, unmasking protocol, and positive threshold). The general recommendations for ER and PR IHC in CMTs presented here are based on the American Society of Clinical Oncology/College of American Pathologists Guideline Recommendations for Immunohistochemical Testing of Estrogen and Progesterone Receptors in Breast Cancer, 49 with some specific variations for the canine species, and are summarized in Table 5.
Guidelines for Estrogen (ER) and Progesterone receptor (PR) Immunohistochemistry and Scoring in Canine Mammary Tumors.a

CMT, canine mammary tumor; ER, estrogen receptor; HPF, high-power field; IHC, immunohistochemistry; IS, intensity score; PR, progesterone receptor; PS, proportion score; TS, total score.
aBased on the American Society of Clinical Oncology/College of American Pathologists Guideline Recommendations for Immunohistochemical Testing of Estrogen and Progesterone Receptors in Breast Cancer. 49
bThe percentage obtained either by estimation or counting cells by image analysis (recommended). The intensity is the average labeling intensity of the positive tumor cells relative to the intensity of the myometrium (maximum intensity).
cIn case of tumors with epithelial and myoepithelial proliferations.
The purpose of these guidelines is to improve the accuracy of ER and PR testing when assessing CMTs.
The time from tumor removal to fixation should be kept to a minimum (preferably ≤1 hour). Underfixation and overfixation of mammary tissue may lead to false-negative hormonal receptor results, although the effect of overfixation can be minimized with antigen retrieval.
The analytical variables include the primary antibody used and the selection of positive and negative controls. Positive (external and internal) and negative controls should be included with each ER and PR IHC run. The positive external control should be canine myometrium with known receptor content. Internal positive controls should consist of adjacent normal mammary gland or mammary hyperplasia, without premalignant changes. These control tissues must display a heterogeneous labeling pattern, with a mixture of cells exhibiting weak, moderate, and intense immunoreactivity. If only a few cells with a homogeneous labeling pattern are detected in the internal control, then the immunoreaction was insufficient and tumor cells with a weak to moderate immunoreactivity will not be detected, potentially causing false-negative assessment. 49 If no nuclear immunolabeling is found in the internal positive control, the sample is considered uninterpretable and the test should be repeated using another tumor block or another tumor specimen. Uninterpretable results can be attributed to samples fixed in alcohol or fixatives other than 10% neutral buffered formalin, fixation for less than 6 hours or longer than 72 hours, fixation delayed for more than 1 hour postsurgery, and samples with prior decalcification. 49 For immunohistochemical labeling, the use of automated devices is preferable to manual techniques.
The postanalytic recommendations are the interpretation of IHC assays for ER and PR. Besides the different IHC techniques employed, the major issues in ER and PR immunolabeling of CMTs are the different evaluation methods employed and the lack of specification of selected fields to evaluate. The method proposed is the Allred scoring system that assesses the proportion of cells with positive nuclear labeling plus the intensity of immunolabeling. When using this scoring system in humans, breast cancers that score <3 are considered negative. We cannot establish the same standard in CMTs since no biological/clinical data support this positive/negative threshold; therefore, the numerical value (0 and 2–8) should be indicated when reporting analytical results. The percentage of positive cells should be counted, preferably using an image analyzer. When evaluating the intensity of immunolabeling, we propose the canine myometrium, with a known positive reaction to ER and PR and with the highest level of intensity (score 3), to be used for comparison with the positive reaction obtained in each set of labeled samples. This will avoid the heterogeneity of IHC labeling that exists in the adjacent nonneoplastic mammary gland. The areas of the tumor to be evaluated must be representative of the histological type and grade in at least 10 HPF. The types of cells evaluated should include both epithelial and myoepithelial cells, and the Allred score should be applied separately for each cell type in cases of complex and mixed tumors. This recommendation is based on (a) the lack of agreement on the expression of hormone receptors in canine mammary myoepithelial cells as positive 10,74,82 or negative 69 and (b) the participation and relevance of myoepithelial cell proliferations in many CMTs, which differs from the pathology of human breast cancer. Stromal cells, cartilage cells, and bone cells in mixed tumors are generally negative 18,69,82 and should not be evaluated.
Molecular Classification of Canine Mammary Neoplasms
Gene expression complementary DNA microarray studies in human breast cancer has resulted in the molecular classification of these neoplasms. 88 Based on these molecular studies, 5 breast cancer subtypes were identified: luminal A, luminal B, normal breast-like, HER2, and basal-like, with basal-like carcinomas having the worst prognosis. Different immunohistochemical panels have been used to identify these subgroups, including the evaluation of hormone receptors, HER2, and basal cell differentiation proteins. There is still no internationally accepted definition for basal-like breast cancers. 9 In dogs, few studies have been undertaken using the above immunohistochemical classification, and the results are not consensual. 37,63,101
Recommendation Guidelines
These recommendation guidelines are intended to be useful in clarifying the contradictory results on the immunohistochemical classification of CMTs. When evaluating basal-like carcinomas in the dog, a different panel of basal markers and different criteria to define what constituted positive immunolabeling were used in the different studies, which may explain the discrepancies in the results. We must keep in mind that this immunohistochemical classification was originally created to investigate human breast carcinomas, which rarely show myoepithelial cell proliferation. 113 As pointed out by others, 113 and in our opinion, this fact may influence tumor basal marker expression and consequently hamper the identification of the corresponding canine counterpart of human basal-like breast carcinomas. Caution should be used when applying the human classification system to CMTs as further studies are needed to establish the appropriate molecular-based taxonomy, and a reliable immunohistochemical panel is needed. 113
Conclusions
This article provides a framework for the IHC evaluation of CMTs. Standardization of the evaluation systems will allow future studies to be more easily evaluated and compared and will allow for the establishment of thresholds of positive immunoreactivity that are both biologically and clinically significant in the dog.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been partially supported by grants AGL2011-025553 (Ministry of Science and Innovation) and BIO-287 (Junta de Andalucía, Spain).
