Abstract
Seventeen lesions diagnosed as teat sinus and duct adenomatous hyperplasia were identified in 10 dogs. All of the dogs were small breeds. Six were spayed female and 4 were male, 3 castrated and 1 intact. In 5 cases, the lesions involved multiple teats. They were pink to black, flattened to round, and sometimes crusted. Histologically, the lesions were usually pigmented (16/17), plaque-like to nodular masses composed of polygonal cells arranged in anastomosing trabeculae and bilayered ducts and/or cysts, with a fibrous to mucinous (Alcian blue-positive) stroma and squamous cysts (12/17). Scattered epithelial cells contained single, discrete, clear cytoplasmic vacuoles. Atypia was mild, and the mitotic count per 2.37 mm2 varied from 0 to 15 (average 2.7). Immunohistochemistry was performed on 14 of the lesions from 8 dogs. Epithelial cells were 100% panCK+ and included basally located CK14+/CK5_6+/p63+/calponin− cells and nonbasal CK19+/CK7+ cells. Cells manifesting squamous differentiation were usually panCK+/CK14+/CK5_6+/CK19−/CK7−/p63±/calponin−. In addition to fibroblasts, vimentin positivity was found in disseminated, round to stellate stromal and intraepithelial cells that often had black, granular, cytoplasmic pigment (consistent with dendritic/phagocytic cells and/or melanocytes). Of the 8 dogs for which clinical follow-up information was available, all were still alive and well, with no significant teat changes, development of mammary lesions or other masses 4 to 22 months (median 12.5) after biopsy. The histologic, immunohistochemical, and clinical findings were consistent with teat duct and sinus adenomatous hyperplasia. This is an uncommon, benign proliferative lesion that can involve multiple teats of female and male, small breed dogs.
Mammary glands are present only in mammals, and their function is to produce and deliver milk to the neonate via a series of ducts that terminate in the teat (nipple). The teat is the only part of the mammary tissue that is fully developed at birth; much of the ductal tissue develops at puberty, secondary to hormonal stimulation, whereas the secretory tissue only develops during pregnancy.
In dogs, the normal ventral abdominal skin consists of a thin epidermis, a few hair follicles with associated sebaceous glands and apocrine glands, a thin dermis, and a prominent panniculus adiposus. On the ventral thorax, the epidermis and dermis are thicker than on the ventral abdomen, and there are more numerous adnexa. The epidermis of the teat is thicker than the adjacent abdominal or thoracic skin, with mild rete ridge formation. Melanocytes may be present in the basal layer of the epidermis, with melanin transfer to the keratinocytes of the overlying epidermis. Although there are no hair follicles in the teat, occasional sebaceous glands and sebaceous gland ducts may be present. The dermis often has a more prominent superficial (papillary) dermis consisting of fine collagen fibers, whereas the deep (reticular) dermis consists of larger collagen fibers interspersed with numerous smooth muscle trabeculae and elastin fibers. The smooth muscle fibers that encircle the upper teat duct form a sphincter. The teat has no panniculus adiposus.
Opening onto the slightly blunted surface of the canine teat are 6 to 22 teat ducts (papillary ducts, collecting ducts) lined by a stratified squamous epithelium. The teat duct lumina may contain compact keratin, forming a keratin plug, and opening into some of the teat ducts are sebaceous gland ducts. At its base, the teat duct opens into the teat sinus (lactiferous sinus); the sinuses are arranged in parallel within the teat and are lined by a bilayered epithelium that consists of an inner layer of cuboidal to columnar luminal epithelial cells and an outer layer of cuboidal to fusiform basal cells, some of which differentiate into myoepithelial cells. On the external aspect of the basal/myoepithelial cells is the basement membrane. Emptying into the base of the teat sinuses are the interlobular ducts, and at the junction of the teat sinuses and interlobular ducts are large saccules that expand to store secreted milk. In the underlying mammary tissue are the larger interlobular ducts and the lobules formed by the smaller intralobular ducts and the acini. 18
The literature on proliferative lesions of the canine teat is sparse. Reported lesions include melanosis of the skin of the teat, teat hyperplasia, ductal adenoma, intraductal papillary adenoma, ductal carcinoma, intraductal papillary carcinoma, carcinoma with epidermal infiltration (Paget-like disease), 5,13,18 and a single case of smooth muscle hamartoma of the nipple. 3 To the best of our knowledge, ostensibly non-neoplastic epithelial proliferative lesions of the teat ducts and sinuses have not been reported. This report documents 17 such lesions in 10 dogs.
Materials and Methods
Two cases of an unusual canine teat lesion were identified by 2 of the authors (FYS and MH) during routine diagnostic biopsy service. These cases were submitted to a third author (MHG) for consultation. Once the lesion was characterized, additional cases were identified and collected through the authors’ diagnostic caseloads and via consultation from other diagnostic pathologists.
Biopsy submission forms, initial biopsy reports, and histologic sections of each lesion were collected and reviewed to characterize the spectrum of the histomorphology and to extract clinical information.
Paraffin-embedded formalin-fixed tissue from 8 cases were available for immunohistochemistry (IHC). IHC was performed on serial, 4-µm unbleached sections mounted on Superfrost Plus glass slides (Menzel GmbH). A panel of antibodies was applied to all cases and included antibodies raised against human antigens that had previously been tested in the dog. 11,14 The following antigens were evaluated using an automated immunostainer (BenchMark, Ventana Medical System, applying the UltraView Universal DAB reagent, Ventana Medical Systems). The protocols were performed as previously described 2 : panCK (AE1/AE3, 1 in 100 dilution, Dakocytomation); CK14 (NCL-LL 002, 1 in 20 dilution, Novocastra), CK5_6 (D5/16 B4, 1 in 100 dilution, Dakocytomation), calponin (CALP, 1 in 200 dilution, Dakocytomation), vimentin (V9, 1 in 150 dilution, Dakocytomation), p63 (4A4, 1 in 200 dilution, Santa Cruz Biotechnology). The expression of CK19 and CK7 was analyzed by B. Brunetti in the Department of Veterinary Medical Sciences of the University of Bologna (Italy) and by M. Rondena at the S. Marco Veterinary Clinic of Padua (Italy), respectively. For CK19 (BA 17, 1 in 400 dilution, Histo-line Laboratories), a manual IHC protocol was applied as described previously. 9 For CK7 (OV-TL 12/30, 1 in 200 dilution, Agilent), IHC was performed using an automated immunostainer (BOND RX Leica). Briefly, after automated dewaxing and rehydration, antigen retrieval was obtained by heating the sections at 97 °C for 15 minutes in a pH 6.0 buffer solution (Epitope Retrieval Solution 1 BOND). Primary antibody was applied for 15 minutes at room temperature followed by a polymer detection system (Bond Refine Detection). The primary antibody was omitted in the negative control, and the adjacent skin with residual adnexa was used as positive internal controls for all of the antibodies.
Additionally, periodic acid–Schiff (PAS), PAS with diastase, and Alcian blue pH 2.6 histochemical staining were performed according to routine protocols to evaluate the extracellular matrix and epithelial intracytoplasmic vacuoles or granules.
The submitting clinicians were contacted by phone for follow-up clinical information. Information requested included any clinical history missing from the submission form and/or biopsy report regarding signalment, number and sites of teats involved, duration of lesions prior to biopsy, age at time of neuter, whether there was any evidence of recurrence, development of additional teat or mammary gland lesions and/or metastasis, and whether the dog was still alive.
Results
Seventeen lesions from 10 dogs were evaluated (Table 1). Five of the 10 dogs had involvement of multiple teats reported, but only a single lesion was submitted in 6 dogs. In the other 4 dogs, one had 4 lesions submitted, one had 3, and the last two had 2. All of the dogs were small breed: 2 Maltese dogs, 2 Havanese, 1 toy poodle, 1 miniature poodle mix, 1 miniature poodle-miniature schnauzer cross, a Yorkshire terrier-poodle cross, a Shih Tzu mix, and a Dachshund. There were 6 spayed females, 3 castrated males, and 1 intact male. The age range was from 7 to 15 years (average = 10.5).
Signalment and outcome information in dogs with adenomatous hyperplasia of the teat sinus and duct.
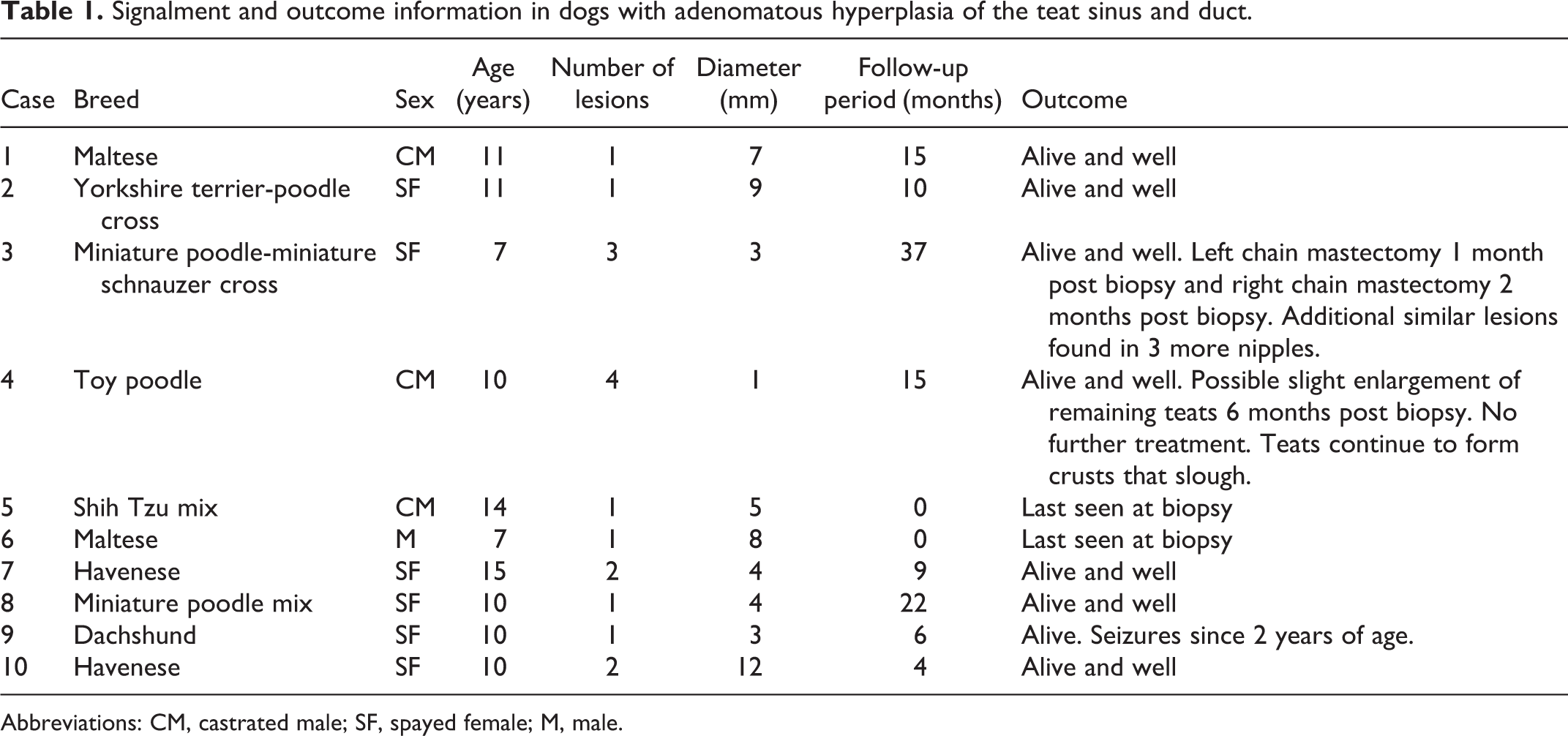
Abbreviations: CM, castrated male; SF, spayed female; M, male.
Clinically, the lesions were variably described as listed in Supplemental Table S1. Grossly, the lesions were reported to be pink to black, flattened to round and sometimes crusted (Fig. 1). The lesions ranged from 1 to 12 mm in greatest dimension. A change in shape and/or color was noted in 3 cases. The lesions were first noted in the clinical records from the day of removal to 5 months prior to biopsy.
All of the lesions were similar histologically. Continuous with the mildly to moderately acanthotic epidermis of the teat, and extending into the subjacent dermis, there was a plaque-like to nodular mass composed of polygonal to cuboidal cells arranged in anastomosing trabeculae and bilayered ducts, supported by fibrous to myxomatous stroma (Figs. 2, 3). Some of the ducts were cystic (Fig. 4), and foci of squamous differentiation with keratinization and occasionally keratin-filled cysts were present (Fig. 2). The squamous cysts were present in 12 of the 17 masses. The cells had irregularly oval nuclei, stippled chromatin, small nucleoli, small to moderate amounts of eosinophilic cytoplasm, and indistinct cell margins. The cells lining the bilayered ducts were cuboidal and often had relatively small amounts of cytoplasm. Multifocally, small ducts lined by a cuboidal epithelium were present within the squamous epithelium. Scattered cells contained a single, variably sized, clear, discrete cytoplasmic vacuole (Figs. 3, 4); some had a signet-ring morphology, characterized by a large cytoplasmic vacuole that peripheralized a crescent-shaped nucleus. The mitotic count was generally low, ranging from 0 to 15 (average = 2.7) per 2.37 mm2. Sixteen of the 17 lesions were pigmented. The black pigmentation varied from multifocal and light to diffuse and heavy. Pigment was present, both in the epithelium and in cells disseminated within the stroma of the lesion and the adjacent dermis. One of the lesions was focally ulcerated, and 3 had cellular crusts. A few lymphocytes and plasma cells were present in 9 lesions; mild, neutrophilic and lymphoplasmacytic inflammation were in 3 lesions; mild to moderate, neutrophilic, and lymphoplasmacytic inflammation in 1 lesion; and no appreciable inflammation in 4 lesions.
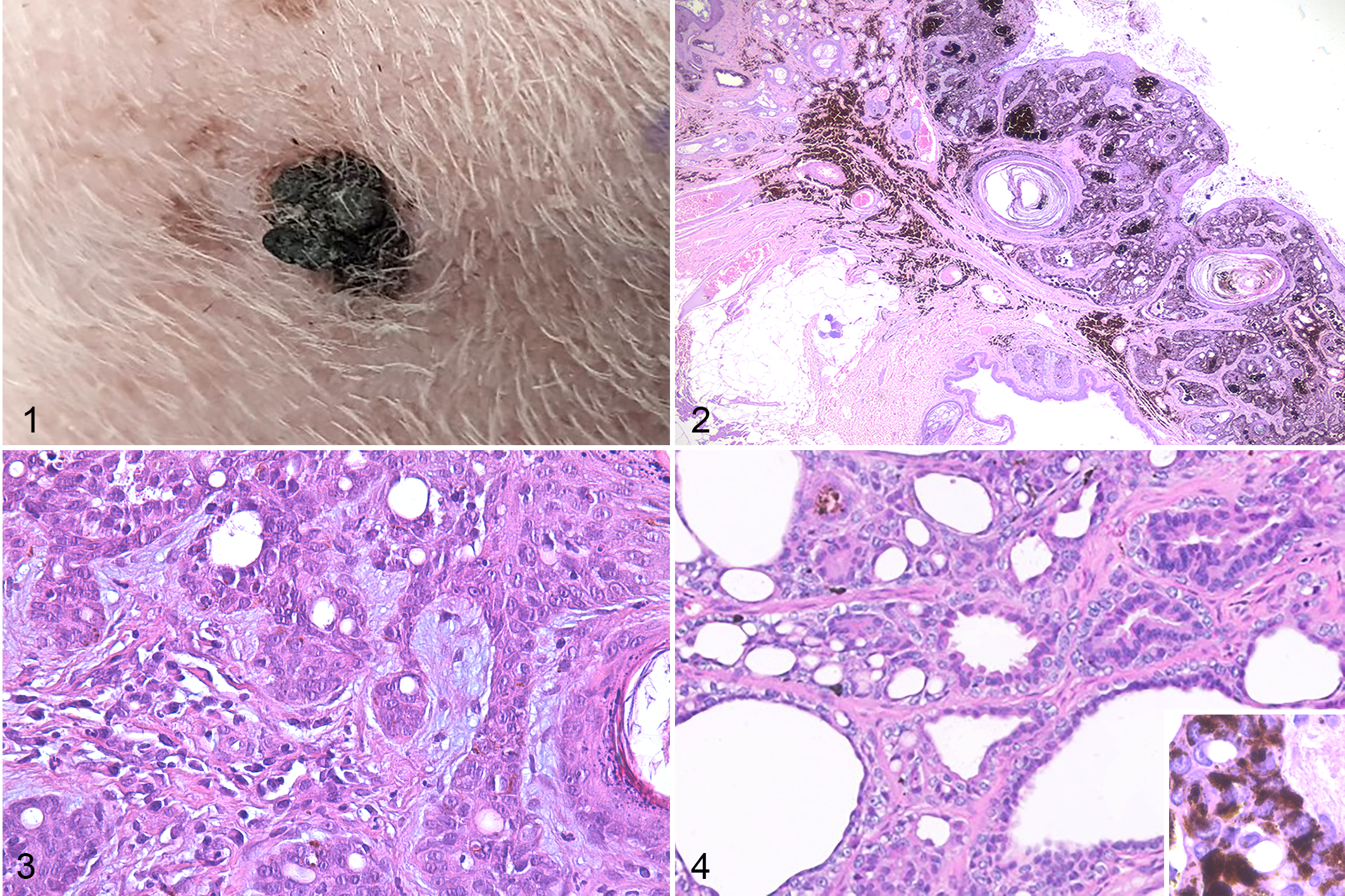
Teat sinus and duct adenomatous hyperplasia, dog. Hematoxylin and eosin.
In 3 of the cases, Paget-like disease was initially considered by the pathologists, but after consultation with the authors were diagnosed as teat duct and sinus adenomatous hyperplasia.
Histochemical staining revealed occasional PAS-positive diastase-labile cytoplasmic granules within the cuboidal cells lining ducts and trabeculae, consistent with glycogen. The intralesional, myxomatous extracellular matrix was slightly Alcian blue positive. The single, clear, discrete, intracytoplasmic vacuoles present in scattered individual epithelial cells were PAS-negative and Alcian blue-negative.
All 14 lesions on which IHC was performed had similar results (Figs. 5, S1, S2). The IHC labeling of the normal canine teat is shown in Figure 6. The basal and suprabasal keratinocytes of the normal teat epidermis labeled positively for panCK, CK14, CK5_6, and p63 but did not label for CK19, CK7, calponin, and vimentin. The normal upper teat epidermis showed identical labeling except for p63, which was negative. The IHC labeling of the squamous epithelium of the teat duct was identical to the teat epidermis. The luminal epithelial cells of the normal teat sinus labeled positively for panCK and CK19, while there was variable labeling for CK7 (±), but these luminal cells did not label for CK14, CK5_6, p63, calponin, or vimentin. The basal epithelial cell of the teat sinus labeled positively for panCK, CK14, CK5_6, and p63, and were negative for CK19, CK7, calponin, and vimentin. Myoepithelial cells in the basal layer of the teat sinus, which were dispersed near the teat duct and become more constant closer to the interlobular ducts, labeled for panCK, CK14, CK5_6, p63, calponin, and vimentin but did not label for CK7 and CK19. In the basal and suprabasal layers of the normal teat epidermis, occasional individual cells labeled positively for CK7 and CK19. No dual labeling was performed to assess colocalization of these markers.
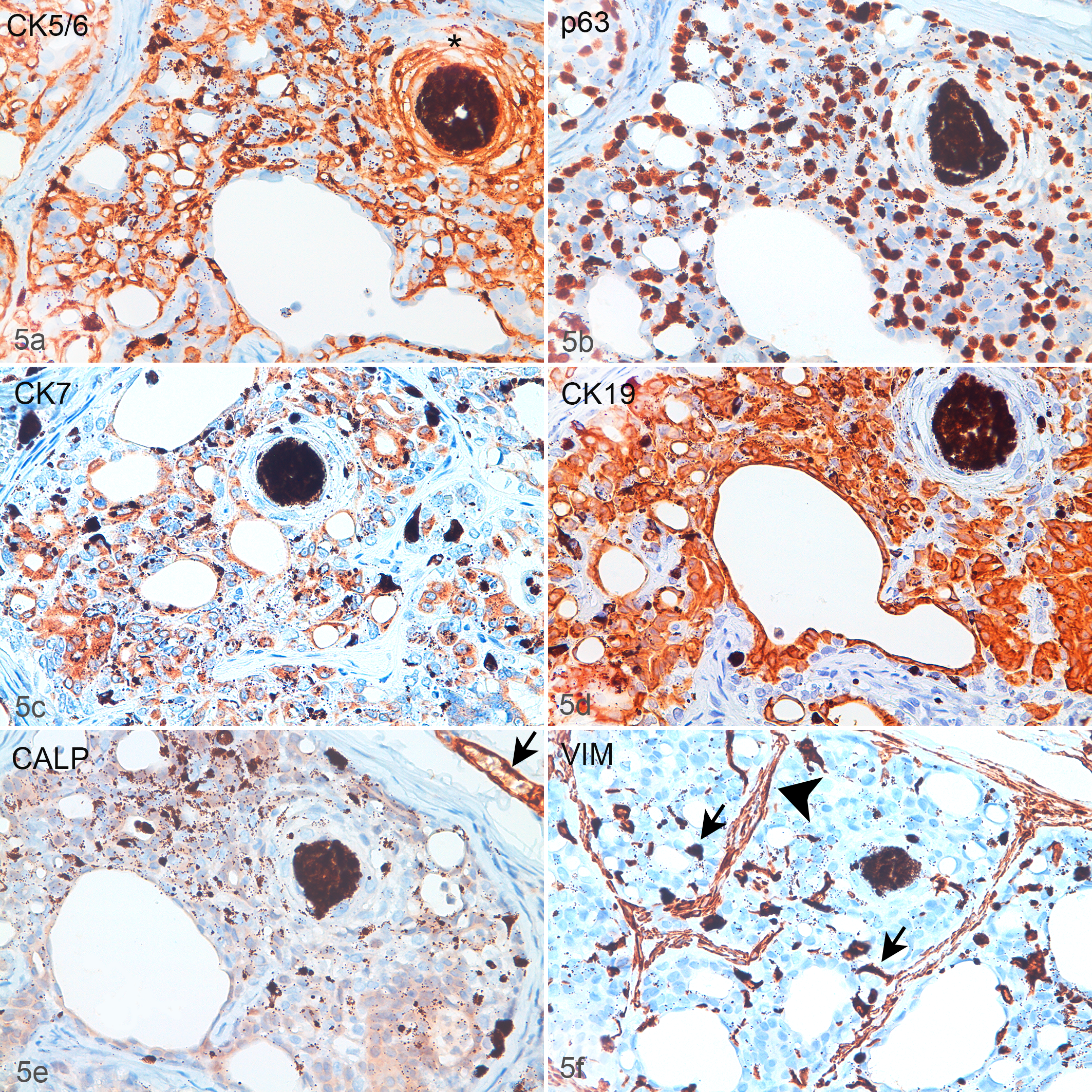
Teat sinus and duct adenomatous hyperplasia, dog, case 8. There are ducts, cells with well-defined, clear vacuoles, and an area of squamous differentiation (asterisk, panel a). Basal cells, vacuolated cells, and the external wall of the squamous cyst are labeled for CK5_6 (a) and p63 (b). The luminal and the vacuolated cells are labeled for CK7 (c) and CK19 (d). (e) There is minimal nonspecific staining with the anti-calponin antibody (CALP), but no positive myoepithelial cells are present. Note the CALP-labeled vessel (arrow). (f) Vimentin (VIM) is expressed in the stroma (arrowhead) and in round to irregularly stellate disseminated intraepithelial cells (arrows). Immunohistochemistry.
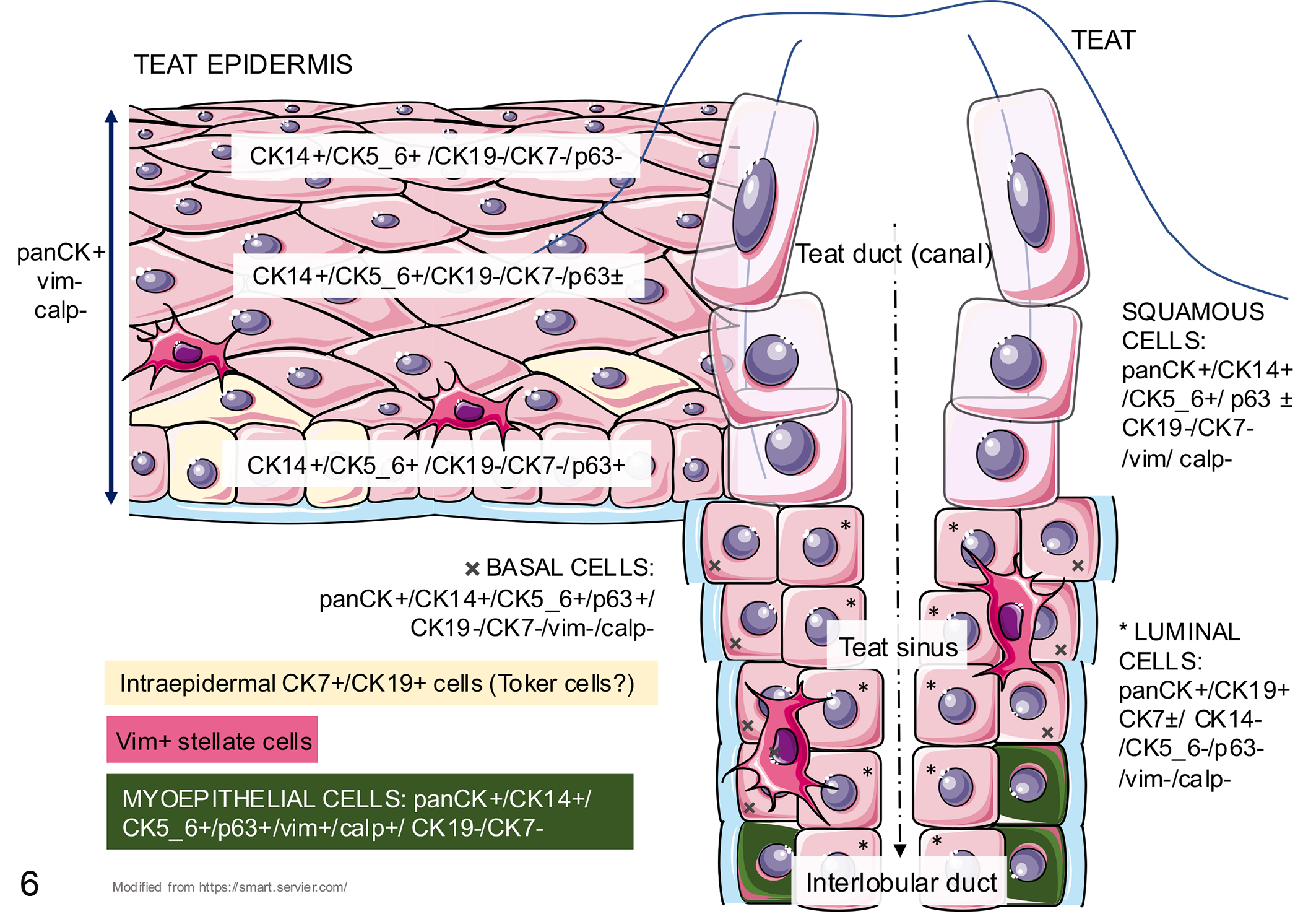
Immunohistochemical phenotype of the normal canine teat. The teat epidermis manifests a progressive superficial loss of p63, disseminated vimentin-positive cells with cytoplasmic processes, and occasional basal or suprabasal CK7+/CK19+ cells. The teat duct is surrounded by squamous cells, whereas the teat sinus has a bilayered epithelium with basal (adjacent to the basement membrane) and luminal cells (adjacent to the lumen). The basal cells of the sinus progressively become calponin-positive myoepithelial cells as they near the interlobular duct. calp, calponin; CK, cytokeratin; vim, vimentin.
PanCK labeling confirmed the lesion was continuous with the overlying epidermis. Within the lesion a basally located cell population showed diffuse (100%) and intense positivity for CK14, CK5_6, and p63, but was negative for CK7, CK19, calponin, and vimentin (Figs. 5, S2). In areas of squamous differentiation, the cells labeled positively with panCK, CK14, CK5_6, and p63 but did not label for CK19, CK7, calponin, or vimentin, while p63 was progressively lost with an increasing degree of differentiation (Figs. 5, S2). Additionally, in larger trabeculae, there was a mixture of cells showing positivity for panCK, CK5_6, CK14, CK19, CK7, and p63 that were negative for vimentin and calponin (Figs. 5, S2). The same profile was detected in the scattered intralesional cells containing a single, clear, intracytoplasmic vacuole (Figs. 5, S2). Calponin was expressed in the blood vessels and the smooth muscle trabeculae (Fig. S1). Within the lesions, a mild, multifocal, nonspecific cytoplasmic labeling for calponin was observed, and very rarely, calponin-positive basal (myoepithelial) cells encircled the teat ducts (Fig. S2). In addition to internal controls (fibroblasts, vessels, etc), vimentin marked disseminated round to stellate stromal and intraepithelial cells, that often contained pigment and were interpreted as melanocytes, dendritic cells, and phagocytic cells (Figs. 5, S1, S2). The CK7+ and CK19+ cells found in the basal and suprabasal normal teat epidermis could not be identified in the epidermis overlying the lesion.
Follow-up information was available in 8 of the 10 cases and ranged from 4 to 37 months post biopsy (median = 12.5). All of the dogs were alive at the end of the follow-up period. One dog had subsequent bilateral removal of the mammary chain. The left chain was removed a month after the initial biopsy and the right chain 2 months after the initial biopsy. Three of the teats in the right chain had similar lesions. Another dog was reported to have possible, slight progression of nonexcised teat lesions 6 months after the removal of 3 teats, with scabs that formed and fell off. No further treatment was pursued. None of the other dogs were reported to develop additional teat or mammary lesions or other masses. The age of neuter was only recorded in 4 cases: 3.5 months, 7 months, less than 1 year, and 2 years. In the cases in which the lesions were noted in the clinical records prior to biopsy (5 cases), the duration ranged from less than a week to 5 months.
Discussion
Proliferative lesions of the canine teat are uncommon, and the differential diagnosis for epithelial proliferations of the teat includes ductal adenoma, intraductal papillary adenoma, ductal carcinoma, intraductal papillary carcinoma, and carcinoma with epidermal infiltration (Paget-like disease). 5,18 Ductal adenomas or carcinomas and intraductal papillary adenomas or carcinomas are present within one or more ectatic ducts 5,18 and are generally not pigmented or continuous with the teat epidermis, unlike the hyperplastic lesions of this case series. In three of the cases in this study, a diagnosis of Paget-like disease was initially considered by the pathologist. The gross lesions had features that could be associated with Paget-like disease (ie, crusting and pigmentary changes) and scattered cells with clear cytoplasmic vacuoles (“signet ring” cells) can be a feature of Paget-like disease. 13 However, other features of Paget-like disease were not present in this study, including individual and small clusters of carcinoma cells in the epidermis of the teat 13 and underlying mammary carcinoma. 13 The presence of multiple teat lesions in some cases also helped to rule out Paget-like disease.
IHC and histochemical findings indicated that the vacuolized cells (“signet ring” cells) were panCK+/CK5_6+/CK14+/CK7+/CK19+/p63+ epithelial cells with well-defined, clear, PAS-negative, and Alcian blue-negative, intracytoplasmic vacuoles. As already reported, 9 neoplastic epithelial cells of the mammary gland can produce lipid vacuoles. Although this feature has not been described in hyperplastic or dysplastic changes of the mammary gland or teat, lipid accumulation is a possible cause of the “signet ring” cell morphology observed in the lesions of this study. Lipid staining or electron microscopy on fresh, non-paraffinized samples is needed to address this possibility.
Alternatively, differentiation to CK7-positive epithelial cells with clear cytoplasm that are normally found in small numbers in the suprabasal layers of the teat epidermis (ie, Toker cells) may account for the “signet ring” cells in these lesions. Additionally, vacuolated cells, similar to those in these lesions, have been reported in a human Merkel cell tumor as intracellular ductal differentiation (intracytoplasmic canaliculi formation) and are thought to originate from a pluripotent epidermal stem cell. 7 Electron microscopy would allow better clarification of the nature and content of the intracellular vacuoles of these canine lesions.
Another interesting histologic feature of these lesions was the presence of cuboidal-lined ducts extending into trabeculae of squamous epithelium that were continuous with the epidermis. This was interpreted as part of the hyperplastic or dysplastic process, in which the teat sinus lining epithelium grew over the squamous epithelium of the teat duct toward or into the epidermis (“teat sinus epithelial creep”).
Most of the keratin-filled, squamous-lined cysts in these lesions were consistent with teat duct (squamous) cysts characterized by elongation of the squamous epithelium-lined teat duct with intraluminal keratin accumulation. Alternatively, some may represent ectatic teat sinuses that underwent squamous metaplasia (“teat sinus cysts with squamous metaplasia”).
The IHC profiles provided evidence of an overlapping phenotype between basal and squamous cells (panCK+, CK5_6+/CK14+/p63+) of the teat epidermis and basal and squamous cells of the lesions, consistent with the teat duct component. This further identified a glandular phenotype (panCK+/CK19+/CK7+) for the luminal, non-squamous cells of the bilayered ducts within the lesions, consistent with teat sinus differentiation at the transition zone from teat duct to teat sinus with few calponin-positive, well-differentiated myoepithelial cells. p63, which can also be used as a marker for myoepithelial cells, was diffusely expressed within the lesions, but was also diffusely present in the basal and suprabasal layers of the teat epidermis and teat duct. Areas of cells with a phenotype (panCK+, CK5_6+/CK14+/CK7+/CK19+/p63+) that is a combination of the basal and squamous cell phenotype (panCK+, CK5_6+/CK14+/p63+) and the luminal phenotype (panCK+, CK7+/CK19+) further supports intermediate differentiation in the transition from teat ducts to teat sinuses.
Interestingly, the immunohistochemical labeling for CK7 and CK19 were slightly different, with less positivity for CK7. Both CK7 and CK19 have been used to identify glandular epithelial cells in both healthy and neoplastic tissue. 4 However, CK7 is not 100% sensitive; for example, glandular cells of PD can be negative. 10 Additionally, CK7 can also be present in more basally located cells, such as tactile epithelial (Merkel) cells. 8 These cells are occasionally found in the teat epidermis and may account for some of the CK7+ cells in our cases.
While involvement of multiple teats raises the possibility of hormonal stimulation in the pathogenesis of these lesions, none of the cases involved intact females, arguing against a role for estradiol and estrogen receptors. Men and male dogs rarely develop mammary gland tumors, 1,15 and in dogs, the tumors can involve multiple glands. 1 Hormonal abnormalities have been associated with mammary tumors in men, and documented risk factors include undescended testes, orchiectomy, testicular injury or inflammation, hormonal therapy, high blood cholesterol, rapid weight gain, obesity, amphetamine use, diabetes, and cigar smoking. 16 While some cases of mammary tumors in male dogs have been associated with testicular tumors, 6,12,17 many cases have no known hormonal abnormality. 1 None of the dogs in this study were reported to have testicular neoplasia, other testicular abnormalities, diabetes, or obesity. Hypersecretion of androgens from the adrenal gland is another possible source of hormonal stimulation that should be considered. Interestingly, all of these lesions affected small breed dogs.
In summary, the lesions of canine teat sinus and duct adenomatous hyperplasia had a unique histomorphology, supported by immunohistochemical findings, compatible with proliferating teat ducts and progressive differentiation into teat sinuses. The bland morphology of the cells, absence of invasive growth, and their clinical behavior were consistent with a benign lesion. The plaque-like to nodular lesions that generally did not form a discrete, expansile mass, and the involvement of multiple teats in some cases, were more consistent with adenomatous hyperplasia than adenoma. These lesions occured in both female and male dogs and affected small breed dogs. The histologic features (ie, anastomosing trabeculae of polygonal cells that were continuous with the teat epidermis, lack of significant atypia, formation of bilayered ducts, and squamous differentiation) should help differentiate this lesion from other proliferative epithelial lesions of the teat.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211058829 - Teat sinus and duct adenomatous hyperplasia in dogs
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211058829 for Teat sinus and duct adenomatous hyperplasia in dogs by F. Yvonne Schulman, Michael H. Goldschmidt, Michael Hardcastle and Valentina E. G. Zappulli in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank B. Brunetti (Department of Veterinary Medical Sciences, University of Bologna, Italy) and M. Rondena (S. Marco Veterinary Clinic of Padua, Italy) for kindly performing the immunohistochemistry for CK19 and CK7, respectively. The authors also thank S. Ferro (Department of Comparative Biomedicine and Food Science, University of Padua, Italy) for technical advice with the images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
