Abstract
The inaugural issue of Pathologia Veterinaria in 1964 contained the first detailed account of lesions in aborted fetuses following natural, experimental, and postvaccinal infection with bovine herpesvirus 1 (BoHV-1). The article, written by pathologists Kennedy and Richards, described diagnostic gross and histologic features in 13 bovine fetuses. The authors provided clinical and epidemiologic features of 1 postvaccination outbreak, including the absence of clinical signs in infected dams and the propensity for abortions to occur after 6 months’ gestation. Subsequent field and experimental studies corroborated and expanded these observations. As a result of this and later reports, veterinarians became alert to the association between infectious bovine rhinotracheitis and abortion, including the risks of exposing pregnant cattle to live vaccinal BoHV-1. Methods were developed to corroborate a morphologic diagnosis of herpetic abortion in cattle, including immunofluorescence, immunohistochemistry, and polymerase chain reaction methods. Outbreaks of postvaccinal BoHV-1 abortion in the United States began to be reported with apparently increased frequency in the early 2000s. This coincided with licensure in 2003 of modified live BoHV-1 vaccines intended for use in pregnant cattle, which are now sold by 3 manufacturers. Ten recent herd episodes of postvaccinal BoHV-1 abortion are reported. All 10 BoHV-1 isolates had single-nucleotide polymorphism (SNPs) profiles previously identified in a group of BoHV-1 isolates that contains vaccine strains, based on a BoHV-1 SNP classification system. They lacked SNP features typical of those in characterized field-type strains of BoHV-1.
Keywords
It was difficult for the Angel of Death to kill everybody in the whole world, so he appointed doctors to assist him.—Rabbi Nachman of Breslow (1772–1810), Sichot HaRan, no. 50
Fear not, gentle pathologist. Even Veterinary Pathology’s editor-in-chief from ultima Thule is unlikely to publish a piece on his southern neighbors’ odd propensity to change their chief executive with a high-velocity round. Our misleading title is an effort to tempt you to read a rumination on an article published 50 years ago in the inaugural issue of Pathologia Veterinaria by another Kennedy. 39 Along with colleague W. P. C. Richards, Dr Peter Kennedy published the first morphologic account of the diagnostic features of abortion due to bovine herpesvirus 1 (BoHV-1). Researchers in Colorado and New York reported similar findings in 1964 and 1965. 15,54,78 Kennedy’s study was based on examining tissues from 13 aborted fetuses following infection of their dams with BoHV-1. We revisit Kennedy’s article here, but not because we are sentimental about one of the original coauthors of Pathology of Domestic Animals. He wrote it in the context of suspicions that field strains of BoHV-1 were abortifacient and a report published 3 years earlier that exonerated a role for vaccinal BoHV-1 in causing abortion. 85 Kennedy’s Pathologia Veterinaria article helped establish beyond doubt the abortifacient properties of field and vaccinal strains of BoHV-1. This is pertinent because, 50 years after the risks of exposing cattle to vaccinal BoHV-1 were documented, such cases continue to occur. Postvaccinal BoHV-1 abortion began to be reported with apparently increased frequency after 2003, when the United States Department of Agriculture (USDA) licensed live BoHV-1 vaccines for use in pregnant cattle. We present evidence that 10 recent episodes of postvaccinal abortion were most likely due to vaccinal BoHV-1.
Prelude to Kennedy’s 1964 Article
The clinical syndrome of infectious bovine rhinotracheitis (IBR) was recognized independently by 2 groups in the early 1950s, one in California dairies and the other in Colorado feedlots. 30,65,90 The virus was promptly isolated, which allowed cross-protection studies demonstrating that isolates from California, Colorado, and elsewhere were identical. 48,51 Subsequent studies in the 1950s and early 1960s documented the importance of BoHV-1 in feedlot and dairy cattle. 1,7,15 It was suggested on the basis of serologic evidence that BoHV-1–like strains were in circulation as early as 1941. 25 The presumed antecedent was the agent of infectious pustular vulvovaginitis (IPV) and infectious pustular balanoposthitis (IPB). IPV (coital vesicular exanthema; Bläschenausschlag) was first reported in central Europe in 1880s.25,52,53,71 Veterinarians reported an IPV-like syndrome in the United States in the 1890s, with scattered reports in the first half of the 20th century.3,32 By 1958, it was clear that the agents involved in IPV-IPB and IBR were similar.25,28,34,53 Currently BoHV-1 comprises respiratory subtype 1.1 and genital subtypes1.2a and 1.2b. What were once assumed to be neuropathogenic variants of BoHV-1.3a and 1.3b are now reclassified as BoHV-5. 87 IBR spread to Europe and beyond, presumably via exported livestock, following its apparent origin in high-density cattle operations in North America. It is currently the focus of successful efforts in the European Union to eradicate the disease in individual countries. 43
The cause of IBR-IPV was identified as a member of the family Herpesviridae. 6 BoHV-1 was soon associated with multiple additional syndromes: abortion, conjunctivitis, gastroenteritis, encephalitis, and generalized fatal systemic infection of young calves. 1,7,11,51,83 When Kennedy wrote his article, it was thought that encephalitis in calves was due to the IBR agent alone. 22,23,31,54 It was later recognized that most neurologic cases were due to BoHV-5. BoHV-5 cases continue to occur sporadically in the United States and more commonly in Latin America and the Antipodes. 21
Veterinary biologics companies promptly recognized the importance of IBR and a need for effective vaccines. Details of the first attenuated live vaccine was published in 1956, the same year that isolation of BoHV-1 was reported. 38,48 Pitman-Moore soon announced the development of an attenuated strain of BoHV-1, based on the Colorado 1 isolate (“Cooper strain”). 91 The isolate was considered attenuated following rapid passage in bovine renal cells. 51 It induced no clinical signs after it was injected intramuscularly into cattle, other than a transient rise in body temperature. No clinical signs occurred when low doses of virus were inoculated intranasally. Higher doses (106.5 and 106.8 TCID50) induced fever and mild rhinitis. 91
Live BoHV-1 vaccines were released on the US market in 1957. 54 Initial studies indicated they were safe for use in pregnant cattle. 85 Postvaccination abortions began to be recognized by clinical veterinarians in 1961 or earlier. 74 An investigation of the purported association between vaccination and abortion concluded there was none. 85 The conclusion was based on several factors: the absence of detectable BoHV-1 in fetal tissue, no reports of clinical signs of IBR in pregnant dams as a prelude to abortion, the presence of multifocal hepatic necrosis considered at the time to be inconsistent with BoHV-1, and abortion as late as 120 days after vaccination. It was assumed herpetic abortion could occur no later than 7 days after respiratory symptoms were manifest in pregnant cattle, presumably due to BoHV-1’s short viremia. Virologists concurred, noting that there were no reports of abortion during IBR outbreaks in the first decade the respiratory syndrome was recognized. 54 Given the 1950s’ focus on bacterial abortion, especially brucellosis, the long interval between infection with BoHV-1 and fetal expulsion, and the role latent infection may play in triggering abortion, it is also possible this association existed but was missed.
The Kennedy Article
The concise 10-page account enumerated major morphologic features of BoHV-1 abortion, many reported for the first time. 39 The description was based on gross and microscopic examination of 13 bovine fetuses aborted in the wake of natural, experimental, and iatrogenic exposure (Fig. 1). Clinical, serologic, and virologic details of some of these affected calves and dams were reported in more detail elsewhere. 54,74 The naturally infected group was derived from a single outbreak in a commercial dairy in which 61 heifers aborted in a group of 238 animals (25.6% abortion rate). 74
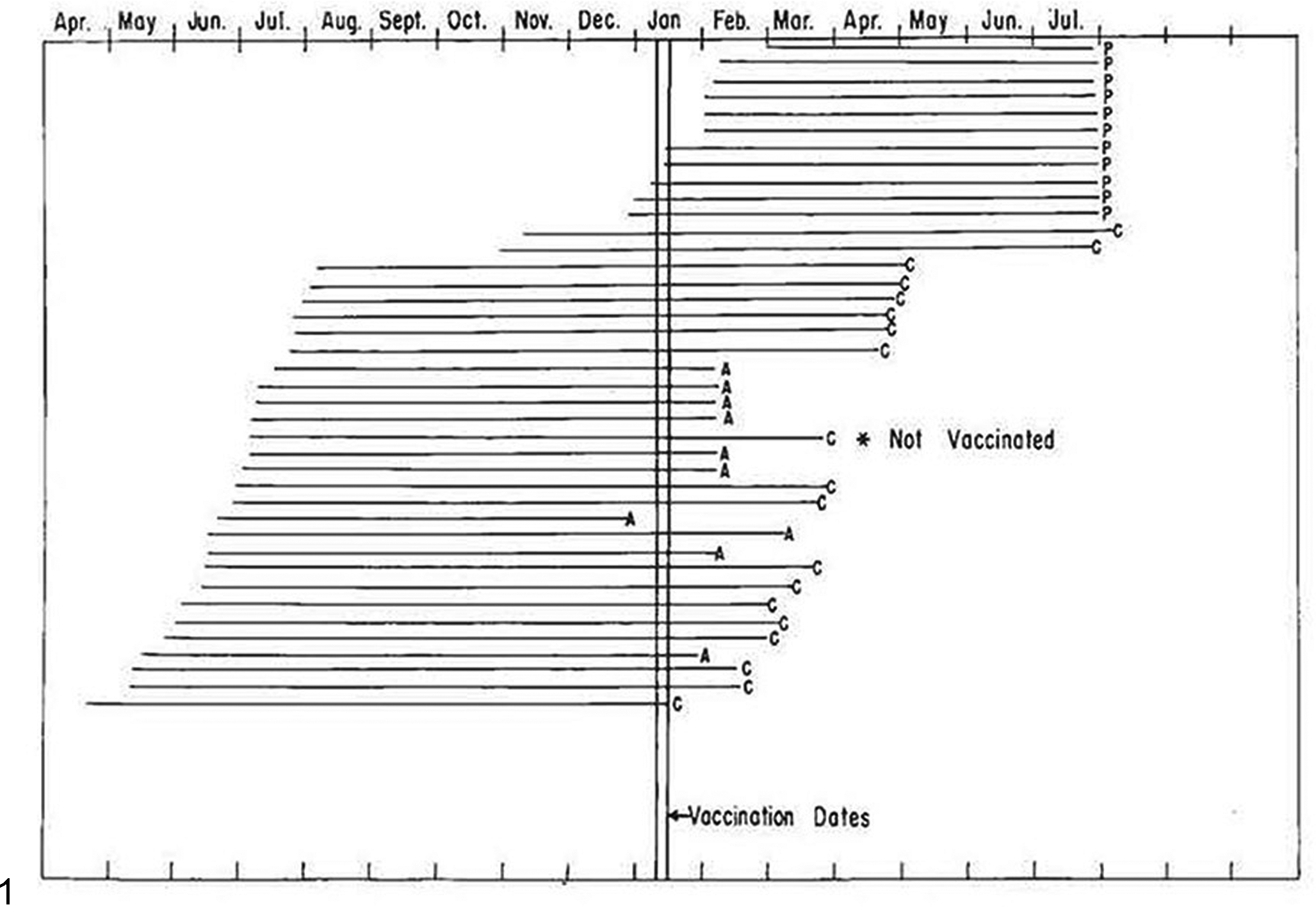
Reproduction of Fig. 1 from Pathol Vet
Lesions in infected fetuses were identical, regardless of how exposure occurred. Calves were aborted 18 to 64 and 23 to 52 days after experimental and iatrogenic challenge, respectively. Affected dams lacked detectable clinical signs. An outbreak of conjunctivitis and respiratory disease affected young calves at the commercial dairy 2 months before abortions began, raising suspicions at the time of IBR. 74 No gross fetal lesions were identifiable, apart from questionable edema in placental membranes and bloody effusion in body cavities that was difficult to distinguish from postmortem change (Figs. 2–4). Fetuses were autolytic, a process the authors estimated indicated death some 1 to 2 days before expulsion. The major histologic lesion was in the liver, comprising disseminated 200- to 500-μm foci of coagulative necrosis (Fig. 5). Hepatic lesions are sufficiently characteristic that they have since became the morphologic hallmark of BoHV-1 abortion, particularly in outbreaks where all fetuses exhibit marked autolysis. 2,12,16,27,43,86 Indeed, for more than a decade, and until a reliable specific fluorescent antibody test was deployed in the early 1970s, laboratory personnel diagnosed BoHV-1 abortion on the basis of histopathology alone. 42,84 It was for that reason that Kennedy emphasized that calcified necrotic hepatocytes, resulting in basophilic debris at the margins of necrotic foci, should not be confused with bacteria, particularly Listeria species.
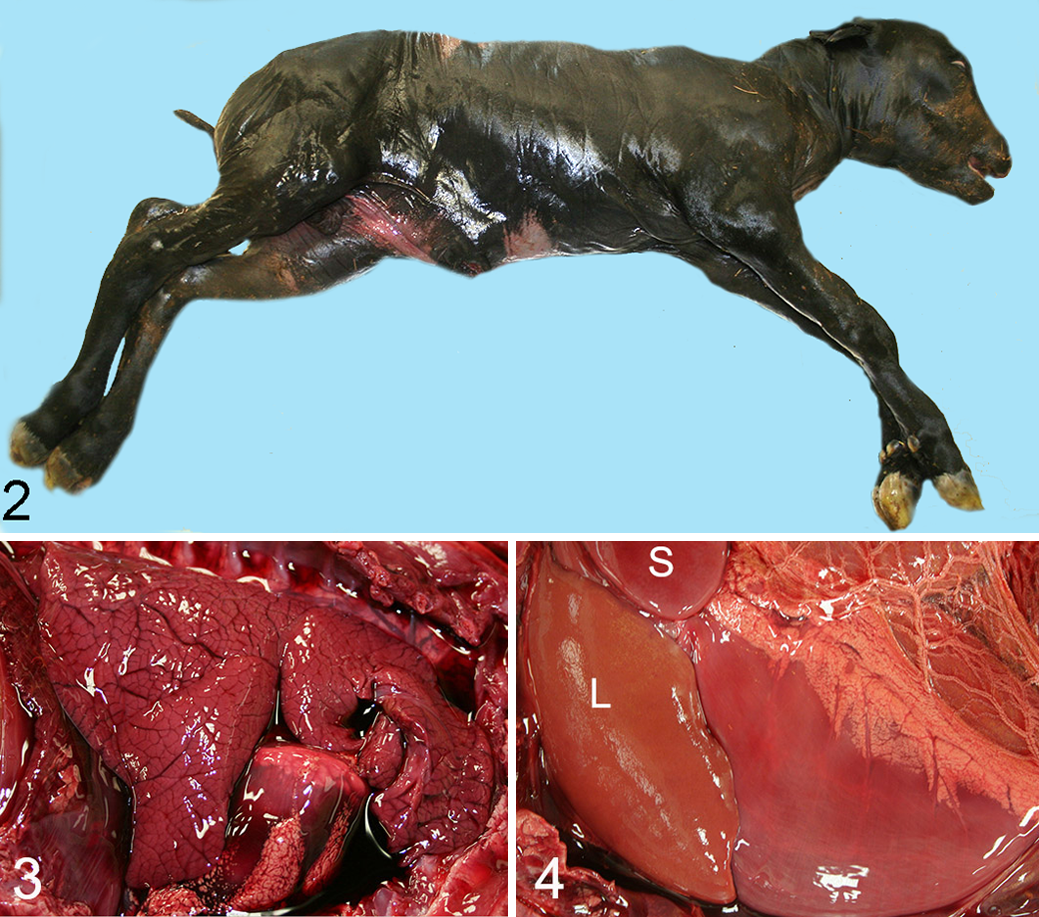
Herpetic abortion, carcass, bovine. Typical gross appearance of bovine fetus due to bovine herpesvirus 1 (BoHV-1). The fetus (15.9 kg; 217 days’ gestation) displays autolytic change. Off-label postvaccination abortion; episode 10 in Table 1.
Herpetic abortion, liver, bovine. Kennedy considered sharply defined, circular foci of hepatic necrosis to be “of considerable specificity” for bovine herpesvirus 1 (BoHV-1) abortion. The only syndrome he thought might cause diagnostic confusion was listeriosis. He remarked that mineralized hepatocytes (arrow) could be confused with bacterial aggregates. He considered infiltration by mononuclear cells in portal areas characteristic of herpetic abortion (top left). Hematoxylin and eosin (HE).
Similar necrotic lesions involved lung, spleen, thymus, adrenal cortex, skin, heart, placenta, and endometrium. The authors found no lesion in the brain, trigeminal ganglia, pituitary gland, skeletal muscle, bone, intestine, trachea, or eye. Kennedy predicted that examination of more affected calves might reveal lesions in these tissues. Attempts to identify typical herpetic intranuclear inclusions were unavailing, despite the use of Bouin’s fixative and an assumption that such inclusions probably developed but were unrecognizable due to autolysis. The admission of an unsuccessful search for inclusions is refreshingly direct. We wonder how many pathologists would pen such a statement today, for fear of being considered incompetent by reviewers. In an epidemiologic foray, the authors noted that fetuses were most likely to be aborted when exposed to live vaccinal virus in the sixth, seventh, or eighth month of gestation. By contrast, no abortions occurred among exposed cows carrying fetuses younger than 5.5 months (Fig. 1). Kennedy inferred that abortion was most likely when cattle were challenged after 6.5 months’ gestation.
Diagnosis was confirmed in affected calves by virus isolation. A commendable positive isolation rate of 20% (7/35 fetuses) was achieved in the affected dairy herd. Successful isolation of BoHV-1 from autolytic aborted fetuses and their placentas is difficult, presumably due to inactivation of viruses or inhibitors in rotten fetal tissue. 45,50,99 Until molecular diagnostic methods became available in the 1990s, diagnosis of herpetic abortion in cattle relied more on finding lesions as described by Kennedy, supplemented by immunofluorescence and other indirect forms of diagnosis, including serology. 42,80,84
Kennedy’s article, as well as a report published almost simultaneously by virologist D. G. McKercher, a colleague at the University of California in Davis, helped lay to rest any doubts that field and live vaccinal strains of BoHV-1 caused abortion. 54 Contemporary and later studies corroborated the conclusion. 7,15,46,50 McKercher speculated that the absence of reports of iatrogenic abortion between 1957 and 1960, following the release of live vaccine and recognition of the abortion syndrome in the early 1960s, was due to the evolution of abortifacient strain(s). 54 It is now clear that BoHV-1 and 2a, including live vaccinal strains, retain the ability to cause abortion; important determinants include the challenge dose of the virus and route of challenge. 64
The Sequel: 1964–2003
Subsequent studies corroborated and extended the observations. BoHV-1 is now recognized as a cause of bovine abortion worldwide. 2 The frequency of large abortion episodes in the United States appeared to peak in the early 1970s. For many years, Dr Clyde Kirkbride at the diagnostic laboratory at South Dakota State University provided the most authoritative snapshot of the incidence of bovine abortion in the United States. 40 –42 BoHV-1 was diagnosed as the cause of 5.41% of all abortions in 1980–1989, which was substantially less than its peak of 24.4% in 1970–1971. 40,42 This accorded with the impression from western Canada at the same time. 89 Kirkbride noted that many abortion episodes investigated by his laboratory in 1968–1972 involved loss of ≥50% of fetal calves. High-morbidity episodes became less common in the 1980s, a phenomenon Kirkbride attributed to widespread vaccination of cows prebreeding and better quality vaccines.
Abortion due to BoHV-1 may be sporadic or manifest as large outbreaks among naive unvaccinated cattle. At the high end, losses may be 25% to 60%; exceptionally, losses of 76.6% and 78.5% are reported. 2,66,99 The results of most published studies accord with Kennedy’s conclusion that fetal death and abortion occur as late as 2 months after dams are infected. 14 Longer intervals (up to 4 months after natural exposure, experimental challenge, or vaccination) are documented. 36,50,60,66 This can make diagnostic confirmation difficult or impossible when investigation is initiated late in the course of outbreaks. There was general agreement that pregnant cattle evince few or no clinical signs before abortion and that chronic reproductive sequelae do not occur. 14,50,74 There are exceptions to the absence of clinical signs, particularly when experimental challenge is involved. 14,16,54,77,97
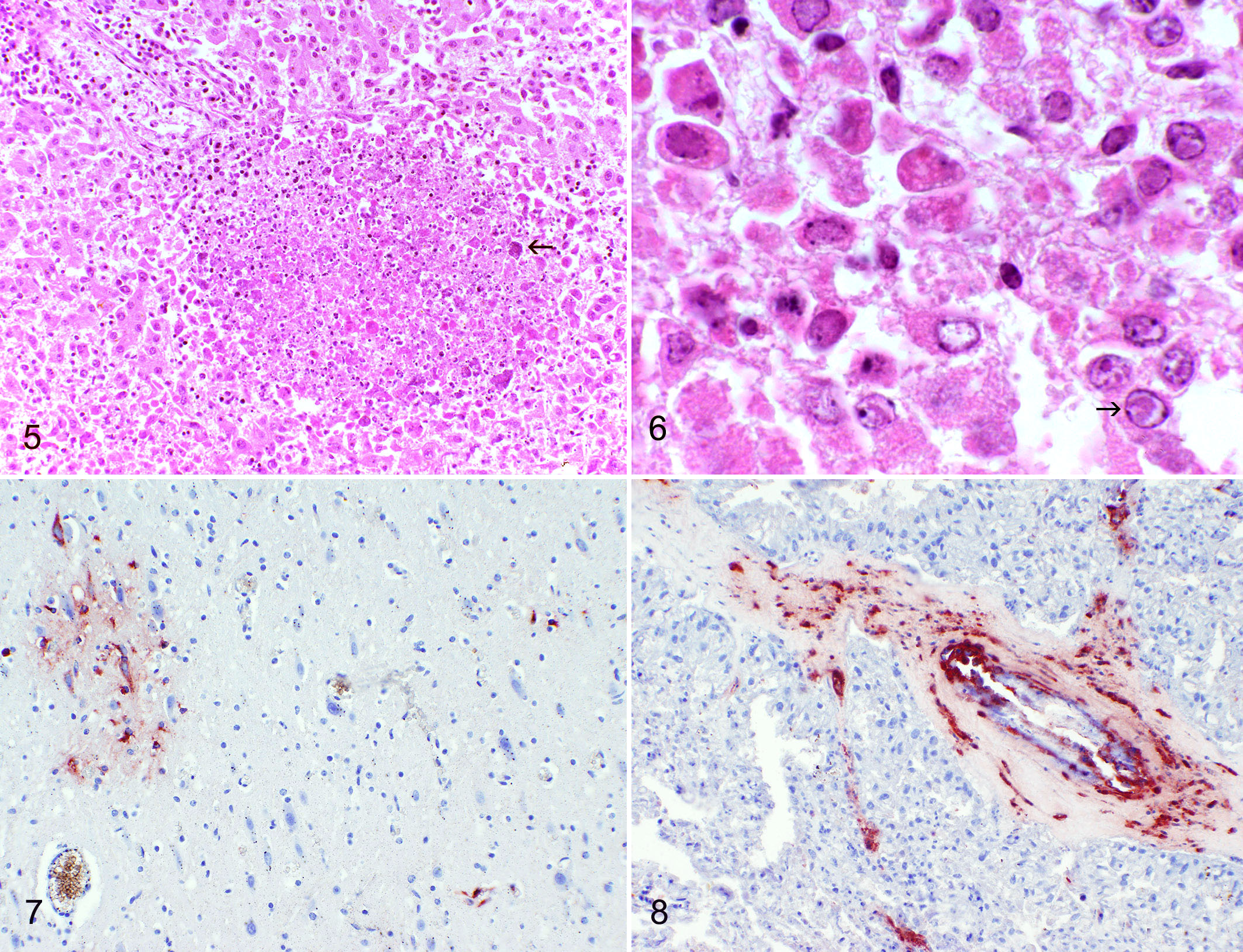
As Kennedy demonstrated and later reports corroborated, in a typical IBR abortion outbreak there are no gross lesions, fetuses are autolytic, and viral inclusions are difficult or impossible to find in conventional histologic sections. 50,78,99 Most diagnostic pathologists can testify to the rarity with which convincing intranuclear inclusions occur in BoHV-1–infected fetuses (Fig. 6). Kennedy’s assumption that lesions would be recognized in other tissues and organs as more fetuses were examined was correct. Remarkably, although it was well known that BoHV-1 could be isolated from fetal and neonatal brains, it was not until 2007–2008 that neurologic lesions were characterized in aborted fetuses. 10,86 Earlier reports noted mononuclear infiltrates or necrosis in fetal brain, and encephalitis was a recognized feature of systemic infection with BoHV-1 in neonatal calves (Fig. 7). 18,29,37,78,83 Cardiac lesions were due to the direct effects of BoHV-1 infection, as demonstrated by co-localized antigen. They were not, as Kennedy thought, the result of fetal anoxia. It is likely he underestimated the length of time for which fetuses were dead in utero. Autolysis is typical in BoHV-1 abortion, and its consistent severity is more suggestive of death 4 to 7 days before expulsion. 33
Several of Kennedy’s contemporaries disputed the presence of BoHV-1–induced necrotizing placentitis, presumably because the lesion is modest and difficult to recognize in the face of autolysis. 14,33,36 Experimental studies and the advent of immunohistochemistry helped establish the presence of necrosis of fetal villi and its association with BoHV-1 antigen. 86 Vascular injury probably accounted for the placental edema noted by Kennedy. 68 Viral antigen occurs in mesenchyme of fetal villi, in endothelial cells, and in tunica media of small- to medium-caliber villous blood vessels (Fig. 8). 86 Kennedy examined only 3 placentas for his study and did not address vascular lesions. The recognition of placentitis is clinically relevant, since placental retention is relatively consistent in cattle that abort due to BoHV-1 in the second half of pregnancy. 99 Postabortion placental retention is less common or absent when BoHV-1 causes abortion earlier in gestation. 77 It is unclear whether placental infection is a primary event leading to abortion or, as seems likely, a sequel to fetal infection via the umbilical vein. 16,36,86 Placenta is the best sample for attempted virus isolation. 41,79,89,95 An unresolved issue in BoHV-1 abortion is the site of viral persistence preceding abortion, particularly given the brief detectable viremia. 73,96 It has long been suspected that the most likely site is in placenta, specifically cotyledons. 14 It remains unclear how often fetuses mount a successful immunologic response to BoHV-1. 35
The rarity with which inclusions are identified in fetal tissues, notwithstanding the presence of widespread necrosis, is generally acknowledged. Convincing inclusions are most readily found in adrenal gland, typically at the interface of necrotic and viable tissue; adrenal gland ranks with placenta as the best candidate tissue for attempting isolation of BoHV-1. In young postnatal calves, it may be the only tissue in which lesions due to BoHV-1 are found (Fig. 6). 2,56,59,60,67 Not addressed in Kennedy’s article, but pertinent to some outbreaks of BoHV-1 abortion, are obstetrical complications such as secondary bacterial metritis affecting individual dams that abort in late gestation. 50,66,76,99
A series of articles in the 1980s by Dr Janice Miller and colleagues at the National Animal Disease Center at Ames, Iowa, added greatly to our understanding of the reproductive effects of BoHV-1, including those in nonpregnant cattle. 55 –64,100,101 Kennedy wrote in early editions of Pathology of Domestic Animals that adverse effects of BoHV-1 were restricted to cattle at 6 months’ gestation or later. Miller’s group and others documented the development of bilateral oophoritis, particularly in the first 3 to 4 days after ovulation, and severe localized necrotizing endometritis. 57,94 While corroborating Kennedy’s conclusion that abortions are most likely later in gestation, Miller documented that abortions can occur at any time during pregnancy. Given that bovine fetuses aged 5 months old are the size of a domestic cat and easy to miss on pastures, it is not surprising that Kennedy’s report emphasized late-stage abortion. Studies by Miller and others reported the effects of different strains of BoHV-1, including those in commercial vaccines, early in gestation and in cycling cattle.* An important observation was that BoHV-1 did not persist in the uterus, making recurrent reproductive failure unlikely. 59 Miller’s group reported the occurrence of systemic lesions in calves infected in late gestation. 56 Such calves can be born alive and survive for 1 or more weeks. Other experimental studies reported features that are either sporadic or difficult to identify in natural outbreaks, such as infertility, embryonic death, mummification, premature birth, and perinatal weakness with no detectable virus in tissue. 55,56,58,66,88,93,97 Miller’s group concluded that vaccinal strains of BoHV-1 were less virulent to fetuses than field strains. 55 But with the possible exception of temperature-sensitive mutants, she thought it unwise to give live vaccinal virus to cattle at any stage of pregnancy. 55,98 A later study performed by her group demonstrated that a thymidine kinase-deletion strain of the virus could induce abortion. 63
It was some years before the association between recent use of modified live BoHV-1 vaccines and abortion was widely accepted. The risk was emphasized in texts aimed at clinical veterinarians. 45 Kirkbride’s impression was that the problem began to decline in the 1980s. 41 Our impression as working diagnosticians is that BoHV-1 abortion remained sporadic during the 1990s. A recent study of the prevalence of BoHV-1 abortion in 5 US-based veterinary diagnostic laboratories (2000–2011) concluded it is now rare (1.4% of aborted fetuses). 27 Sporadic outbreaks continued, such as one episode where a Kansan producer administered live viral vaccine off label to pregnant cattle and lost 50% of the fetal calf crop as a result. 72
Convenience Kills: 2003–2014
Vaccination of cattle against BoHV-1 is beneficial, particularly in feedlots where IBR is common and a risk factor for bovine respiratory disease complex. Vaccination of nonpregnant cows was initially considered the most effective way to prevent abortion. 14,54 Modified live vaccines generally prevent severe clinical signs and reduce shedding of virus following natural infection. 103 Killed products are considered safe albeit less effective due to loss of antigenicity during inactivation. 45,103,105
In 2003, the USDA licensed the first modified live BoHV-1 product designed for use in pregnant cattle. This was done based on a safety study (79 heifers) and field trials using 3 commercial herds (1422 cows and heifers). 20 Two other manufacturers followed suit and received similar USDA licenses.
The attraction of the products is obvious. Modified live vaccines induce stronger, more prolonged immunity than inactivated counterparts and are broadly cross-protective to current BoHV-1 strains. 19 The ability to give modified live BoHV-1 to pregnant cattle is convenient, since vaccines can be used year-round and regardless of the breeding status of animals. Prelicensing studies demonstrated that the products appeared to be safe, with the major proviso that cattle be vaccinated either once or twice (depending on product) before vaccination during pregnancy. The prebreeding injection must be given within 12 months of exposing pregnant cattle to live vaccinal virus. Directions to producers and veterinarians are clear enough. In bold letters, the manufacturers warn, “Do not use in pregnant cows (abortions can result) unless they were vaccinated, according to label directions, with any [product name] vaccine within the past 12 months. Do not use in calves nursing pregnant cows unless their dams were vaccinated within the past 12 months as described above. To help ensure safety in pregnant cattle, heifers must receive at least 2 doses of any [name] product with the second dose administered approximately 30 days pre-breeding.”
Such warnings have limitations. There is no statement of the frequency with which abortion due to BoHV-1 can be expected to occur naturally. There is no acknowledgement that vaccinal BoHV-1 infects ovarian tissue, including corpora lutea, and induces necrotizing oophoritis with a return to estrus (Figs. 9–11). There is no estimate of the proportion of abortions that may occur in appropriately vaccinated cattle when natural challenge occurs, assuming manufacturers’ directions are followed. No advice is given to producers or veterinarians as to how they might distinguish iatrogenic from natural abortion, should there be a suspicion it occurred. Since no marker was put in vaccinal virus licensed for use in pregnant cattle, it was left to others to devise ways to distinguish field from vaccinal strains when abortion occurred and BoHV-1 was found in tissue. 24
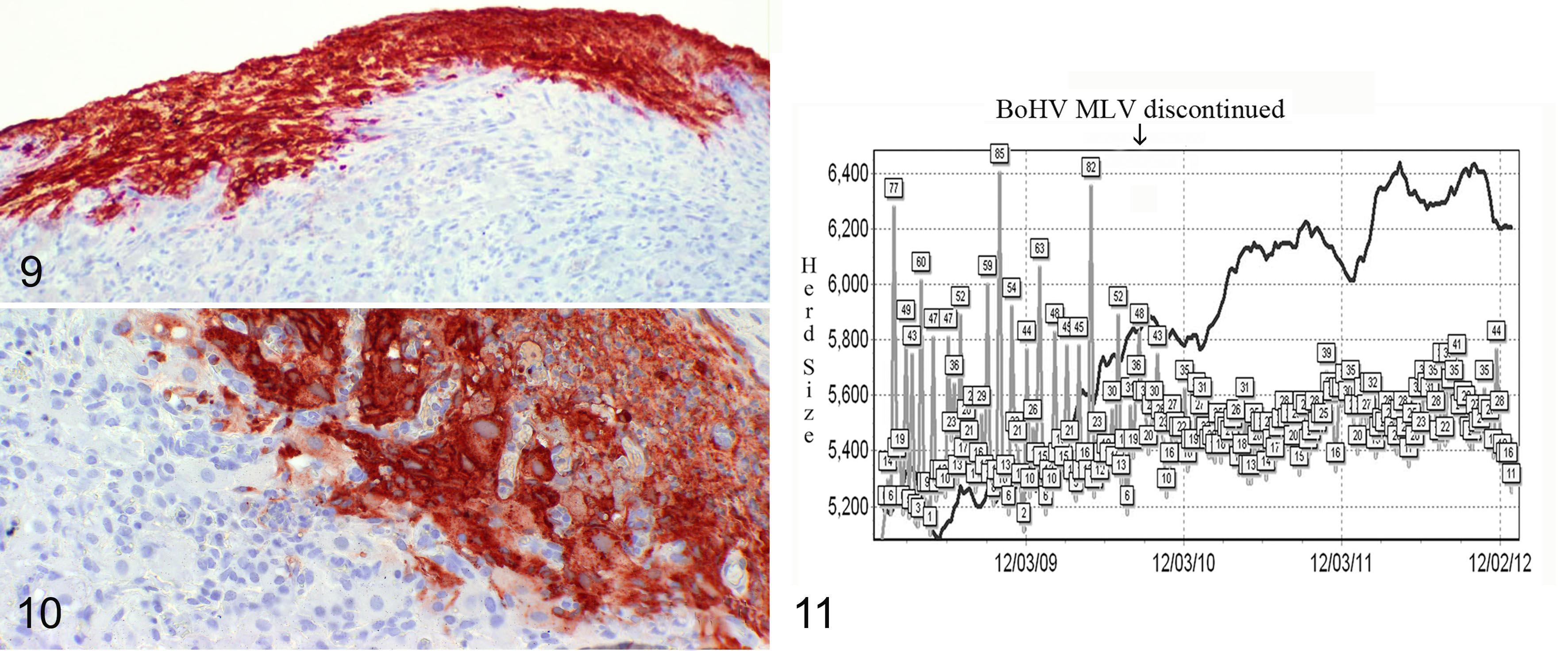
Bovine herpesvirus 1 (BoHV-1) infection (experimental), ovary, bovine. Kennedy and Richards did not address the early reproductive effects of BoHV-1 infection or herpetic oophoritis. Subsequent studies by Miller and colleagues demonstrated that BoHV-1 caused adverse reproductive effects at all stages of pregnancy and in cattle during estrus. Viral antigen in the ovarian cortex of a heifer shortly after vaccination with modified live BoHV-1.
81
Immunohistochemistry; BoHV-1 antigen.
For more than 20 years, it has been possible to create vaccinal strains that are distinguishable from field strains on the basis of serologic assays (DIVA strategy). Such DIVA BoHV-1 products are widely used in Europe. 10,49,102,103 If it is considered necessary to vaccinate pregnant cattle with a known abortifacient, the use of a marker vaccine strain might have been prudent. 8 It is common practice for veterinary biologics companies to recommend annual booster vaccinations for most products. 5 This increases the cumulative risk of adverse effects when abortifacients are used. A cynic might say that a culture of minimizing unnecessary vaccination, specifically by studies to establish the average duration of protective immunity, is not yet fully endemic in the US animal biologics industry.
Various diagnostic laboratories, including those in Wyoming, South Dakota, and Colorado, have recognized an apparent increase in postvaccinal abortion episodes during the past 10 years. Lesions were typical of BoHV-1 fetal infection, as described by Kennedy. Table 1 documents 10 such episodes of BoHV-1 abortion, one of which was reported recently. 76 In many instances, it is not possible to establish on the basis of a clinical history whether vaccines were given to pregnant cattle in accordance with manufacturers’ directions. This is because many producers keep insufficiently detailed records of the exact BoHV-1–containing product given to prebreeding animals in the previous year. This is compounded by the large number of USDA-licensed vaccines containing BoHV-1—there are 97 in all—and a bramble thicket of similar-sounding names. Prithee, ACVP-eligible pathologist, how well you would do on a multiple-choice question about which of the following must be given prebreeding so that pregnant cattle can be safely boosted with live vaccinal virus: Pyramid IBR, Pyramid 2 + type II BVD, Pyramid 2 + type II BVD LPH, Pyramid 4, Pyramid 4 + Presponse SQ, Pyramid 5, Pyramid 5 + Presponse SQ, or Pyramid 10? The correct answer (Pyramid 2 + type II BVD, Pyramid 5, or Pyramid 10) is important. If you used the “wrong” prebreeding product and abortion occurs subsequently, recompense from the manufacturer—difficult at the best of times due to preemption (see below)—is even more unlikely.
Ten Outbreaks of Abortion Following Use of Licensed Vaccine Products Containing Modified Live BoHV-1.
BoHV-1, bovine herpesvirus 1; SNP, single-nucleotide polymorphism.
aSee Fulton et al; 24 this group of BoHV-1 isolates includes 2 closely related vaccine strains, 5 isolates identified in aborted fetuses (4 known or suspected to be postvaccination), and 1 isolate from a feedlot calf.
bDetails of abortion episode reported previously. 76
Comparison of BoHV-1 DNA from diagnostic samples to characterized isolates is possible by analysis of single-nucleotide polymorphisms (SNPs), based on a recent study of field, vaccinal, and reference strains of BoHV-1. 17,24 Although genome sequencing of the BoHV-1’s ∼135-Kb genome would be optimal to establish complete identify, SNP classification provides a way to group isolates and provide guidance about their identity in diagnostic samples, especially when there is a history of vaccinating in the previous 3 months using live BoHV-1. Regardless of whether the product was given on or off label, viral DNA in all 10 episodes had SNP patterns identified in a group containing vaccinal strains (“group 3 viruses”), based on the BHV-1 SNP classification system. 24 An example of SNP patterns of BoHV-1 in tissue from an aborted fetus in a postvaccination episode is illustrated (Fig. 12); similar patterns were present in viral DNA in fetuses from the other 9 episodes. This concurs with the earlier identification of BoHV-1 of presumed vaccinal origin in aborted fetuses examined in laboratories in Colorado, Oregon, and Nebraska 24 and in similar episodes in Texas. This does not prove that vaccinal virus was causative, establish the extent of the problem in the cattle industry, or define co-factors predisposing particular herds to abort. But it does suggest a problem. A retrospective study of abortions submitted to multiple US laboratories concluded that fewer BoHV-1–positive submissions were associated with vaccination of any kind, relative to BoHV-1–negative submissions. 27 Such “Big Picture” approaches are useful when confronting an obvious cause-and-effect shortly after commercial products are used and a problem develops. They are less helpful in abortion-stillbirth episodes where many variables, particularly stage of gestation and the nutritional status of dams, 44 are at play. The interval between vaccination and the onset of abortions (20–40 days), as is the case with BoHV-1, tends to mask a relationship with recent vaccine use. This is challenging for an agent like BoHV-1 in extensive cow-calf operations and in a context where only a fraction of aborted fetuses is submitted for laboratory testing. 104 It is common for producers to be unaware of a herd’s current conception failure/abortion rate until calving is under way. By then it is too late to assess causality.

Bovine herpesvirus 1 (BoHV-1) sequences from postvaccination aborted fetus. Sequences were compared with those in published polymerase chain reaction regions after amplification using primer sets 4, 6, and 7, as described. 24 The isolate of BoHV-1 from this aborted fetus contained single-nucleotide polymorphisms corresponding to those in the vaccine-related group of BoHV-1 stains (“group 3 viruses”). 24 Sequence designations: WT, wild type, NVSL reference strain (GenBank JX898220); Vx, vaccinal virus; P-Vx, postvaccination isolate of BoHV-1 from aborted fetus in episode 3, Table 1. Sequence comparison from primer set 4 region (UL41, host shutoff factor) (A); middle row: primer set 6 region (UL19, major capsid protein) (B); and primer set 7 region (UL6, UL6 protein) (C).
Postmarketing surveillance is an effective mechanism to help ensure that licensed products are safe. 92 Despite moves in that direction, and due to some mixture of limited funding, regulatory underreach, and institutional acedia, no meaningful transparent postmarketing surveillance mechanism for veterinary vaccines exists in the United States. In its place is a largely voluntary reporting system that has yet to generate annual summary data available to consumers and veterinarians. 4 This is unfortunate, since many food animal vaccines are purchased directly by owners and then used with limited veterinary input. The bar for USDA licensure is low, as exemplified by the extraordinary number of licensed products designed to control individual diseases. It is inevitable that some adverse reactions will be detected only after products are federally licensed. This is particularly true for live vaccines that are sold for use in pregnant animals. 5 Indeed, some manufacturers of live BoHV-1 vaccines warn that “fetal health risks associated with vaccination of pregnant animals with modified live vaccines cannot be unequivocally determined by clinical trials conducted for licensure.” Under the courts’ current interpretation of the Virus-Serum-Toxin Act (VSTA) (21 U.S.C. §§151–159 [1994]), vaccine manufacturers are largely insulated from liability when products cause disease or fail to confer protection. 47,70 The negative consequences of preemption, particularly of the type embedded in VSTA, are well recognized by the public health community. 26,69,82 It may be time for public discussion about the positive and negative aspects of preemption in the case of veterinary vaccines, involving but not restricted to the USDA and vaccine manufacturers.
Each year, we are contacted by producers concerned about abortion episodes following use of live vaccinal BoHV-1 in their herds. They are surprised to find that the side effect was documented 50 years ago in the article under review. 39 There is little we can offer, other than sympathy, a little advice, and a personal view that such products should either not be sold or that use be restricted to veterinarians until the extent of the problem is defined independently by postmarketing epidemiologic investigation. We alert callers to the loss of their rights as consumers, given the past success of vaccine companies to persuade the courts to invoke blanket (“ceiling”) preemption. The best we can do as diagnosticians, unfortunately, is write what you have just read.
Footnotes
Acknowledgements
Declaration of Conflicting Interests
CCL Chase reports research support and/or speaker fees Boehringer Ingelheim Animal Health, Merck Animal Health, Zoetis, Novartis Animal Health US Inc., Elanco, Bayer, Merial, CEVA, HIPRA and Agrilabs. D O'Toole reports speaker fees from Pfizer (now Zoetis) and Novartis Animal Health US Inc. MM Miller and H Van Campen report no potential conflicts of interest.
Funding
SNP sequencing was performed with support from Center for Biological Control and Analysis by Applied Photonics in the Department of Chemistry and Biochemistry, South Dakota State University; and by the Animal Disease Research and Diagnostic Laboratory, South Dakota State University.
*References 13, 33, 58, 77, 81, 94, 100, 101.
