Abstract
Diagnostic and investigative molecular pathology frequently has to resort to extraction of DNA from formalin-fixed and paraffin-embedded tissue samples. Although many different protocols are reported for this type of material, extraction of sufficient amounts of intact DNA is still challenging. Here, the authors report a reproducible, simple, cost-effective, and efficient protocol that yields up to 140 μg of DNA from approximately 10 to 15 mg of formalin-fixed and paraffin-embedded tissue samples and compare it to available protocols. The protocol allows stable amplification of DNA fragments up to 600 bp in length in a wide variety of tissues.
Keywords
Introduction
Fresh or fresh frozen tissue samples are the best material for isolation DNA of high quality and quantity. However, storage of frozen tissue samples is expensive and time-consuming. For many retrospective studies, formalin-fixed and paraffin-embedded (FFPE) material is, therefore, the only available tissue for DNA analysis.
Isolation of sufficient amounts of intact DNA from FFPE tissue samples is challenging. One major problem is DNA-protein cross-linking caused by formalin fixation that must be broken up during the extraction process. 3,8 Furthermore, the low pH in unbuffered fixatives leads to degradation of most DNA molecules into fragments of 200 bp or less. 3 A number of approaches have been published to deal with these problems that hinder the use of FFPE for genomic analysis. 1,2,5 –7,9 These protocols use expensive DNA extraction kits, 2,5,7,9 have low DNA yield, 2,6 have suboptimal purity of DNA, 1 or fail to allow for effective amplification of DNA fragments beyond 300 bp. 1,2,5,6
Here, we describe an efficient and cost-effective protocol for the extraction of genomic DNA from archival FFPE tissue samples that yields DNA amounts between 1 and 9 μg/mg of tissue and allows stable amplification of PCR products larger than 600 bp.
Material and Methods
Case Selection
Random archival tissue samples of feline lymphomas (n = 60), canine mast cell tumors (n = 42), canine cutaneous histiocytomas (n = 29), and feline myocardium (n = 42) submitted as biopsy material to the Department of Veterinary Pathology of the Freie Universität Berlin by different referring veterinarians were used in the study. No information on time span between surgical excision and formalin fixation, duration of formalin fixation, and the amount or concentration of fixative was available for the tissues.
Nucleic Acid Extraction
For genomic DNA extraction, 20 to 40 paraffin sections of 2 μm (amounts to approximately 10 to 15 mg of deparafinized tissue) or thinner were collected into a microcentrifuge tube. To remove paraffin, samples were incubated for 10 minutes at room temperature with 800-μl xylene and pelletized at 14,000 × g. This step was repeated if macroscopically visible paraffin remnants were present and the supernatant was still turbid. In most samples, 1 rinsing step was sufficient; some samples required 2. Pellets were then resuspended and washed twice with 500 μl 99% ethanol.
After resuspending in 300 μl Cell Lysis Solution (Gentra® Puregene® Tissue Kit, Qiagen, Hilden, Germany) and thorough homogenization by pipetting up and down, the sample was heated to 65°C for 45 minutes followed by 98°C for 15 minutes. After cooling and addition of 1.5 μl Proteinase K Solution, samples were incubated for 48 hours at 56°C. During this time, 1.5 or 3.0 μl Proteinase K Solution was supplemented depending on the amount of tissue. RNA was then digested by addition of 1.5 μl RNase A Solution and incubation for 15 minutes at 37°C.
Samples were placed on ice for 5 minutes after adding 100 μl Protein Precipitation Solution (Gentra® Puregene® Tissue Kit) and centrifuged at 14.000 × g for 3 minutes. DNA in the supernatant was then precipitated by addition of 300 μl of 100% isopropanol. Samples were incubated for 5 minutes on ice and centrifuged at 14,000 × g for 3 minutes.
DNA pellets were washed twice in 300 μl 70% ethanol, air dried for 10 minutes, and resuspended in 25 μl DNA Hydratation Solution (Gentra® Puregene® Tissue Kit). DNA concentration was determined and adjusted to 100 ng/μl by adding 5 mM Tris buffer, pH 7.4.
In a preliminary step, several modifications of this protocol were tested (see Table 1). Quality of genomic DNA samples was tested by gel electrophoresis on 1% agarose gels.
Comparison of Mean DNA Yields of 3 Lymphoma Samples Extracted With Several Modifications of the Protocol

a This was repeated once with very similar results.
PCR Amplification
The influence of PCR master mix compositions on effectiveness of DNA amplification was tested regarding concentration of MgCl2 (1.5–3.0 mM), dNTPs (0.1–0.4 mM), primers (0.1–0.4 μM), and Taq DNA polymerase (0.02–0.04 U/μl) (GoTaq®, Promega, Mannheim, Germany). No effect of different master mix composition was observed, and a standard master mix was used with all the experiments. It consisted of 2.0 mM MgCl2, 0.2 mM of each dNTP, 0.2 μM of each primer, and 0.024 U/μl of Taq DNA polymerase.
Then, 80 bp and 161 bp of the gene coding for glyceraldehyde-3-phosphate-dehydrogenase (GAPDH) 4 as well as 245 and 606 bp of the feline androgen receptor (FAR) were amplified from DNA extracted from feline lymphomas and feline myocardium (primer sequences are listed in the supplemental material).
Two PCR products of the canine androgen receptor (CAR) measuring 245 and 628 bp in length were amplified from DNA extracted out of canine mast cell tumors and canine cutaneous histiocytomas. Additionally, 80 bp and 161 bp of the gene coding for GAPDH were amplified (primer sequences are listed in the supplemental material).
Cycling conditions included an initial denaturation at 94°C for 5 minutes, melting of DNA at 92°C for 30 seconds, annealing for 10 seconds, amplification at 72°C, and a final amplification at 72°C for 5 minutes. For GAPDH, the annealing temperature was 53°C, and amplification time was 15 seconds. For AR, the annealing temperature was 58°C, and amplification time was 80 seconds. Fifteen randomly selected PCR products of the 245-bp fragment of the CAR from canine cutaneous histiocytomas were submitted to a commercial sequencing service (Seqlab, Göttingen, Germany).
Results and Discussion
Satisfactory amounts of DNA were extracted from all tissues (Tables 2 and 3). However, the DNA yield was significantly dependent on the tissue analyzed. In particular, myocardium yielded less DNA (5 μg) when compared to tumors (>26 μg). This is most probably due to the fact that tumors have a smaller nucleus/cytoplasm ratio when compared to nonneoplastic tissues. However, despite lower DNA yield of feline myocardium, PCR amplification results of feline lymphomas, feline myocardium, and canine cutaneous histiocytomas were comparable. All amplifications up to 245 bp yielded equally strong bands. However, the intensity of bands with PCR products larger than 600 bp was variable. Furthermore, all samples submitted for sequencing yielded the expected sequence.
High DNA Yields Obtained From Feline Lymphomas, Feline Myocardium, and Canine Cutaneous Histiocytomas, Lower Yields From Canine Mast Cell Tumors
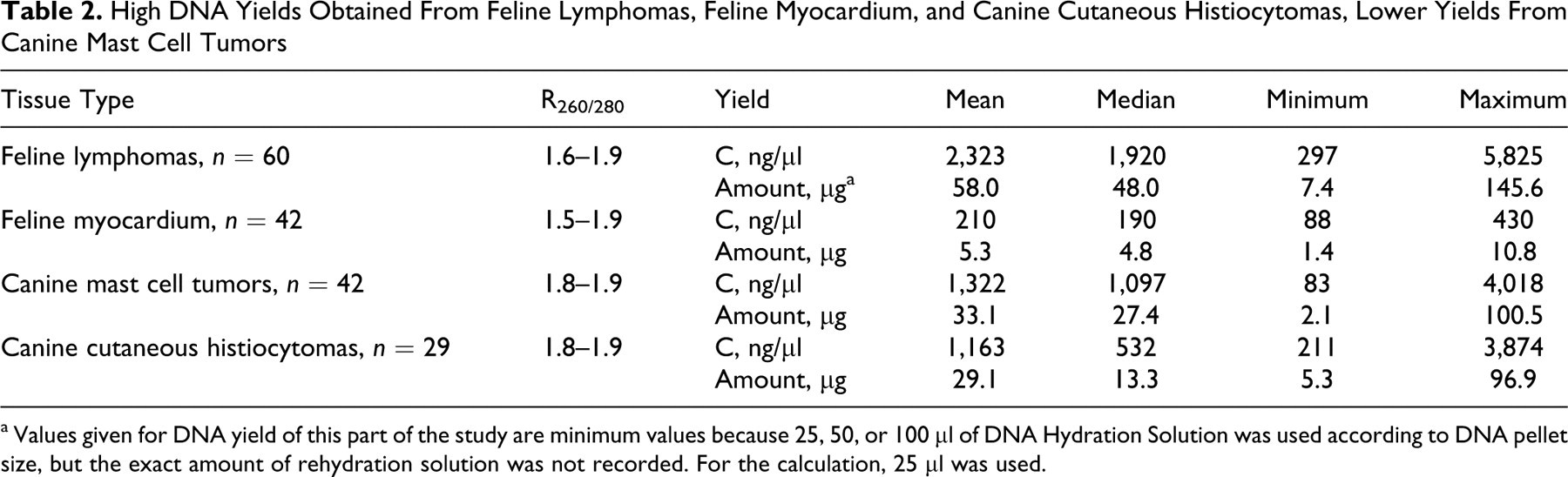
a Values given for DNA yield of this part of the study are minimum values because 25, 50, or 100 μl of DNA Hydration Solution was used according to DNA pellet size, but the exact amount of rehydration solution was not recorded. For the calculation, 25 μl was used.
Number and Percentage of Amplifiable DNA Samples Extracted From Feline Lymphomas, Feline Myocardium, Canine Mast Cell Tumors, and Canine Cutaneous Histiocytomas
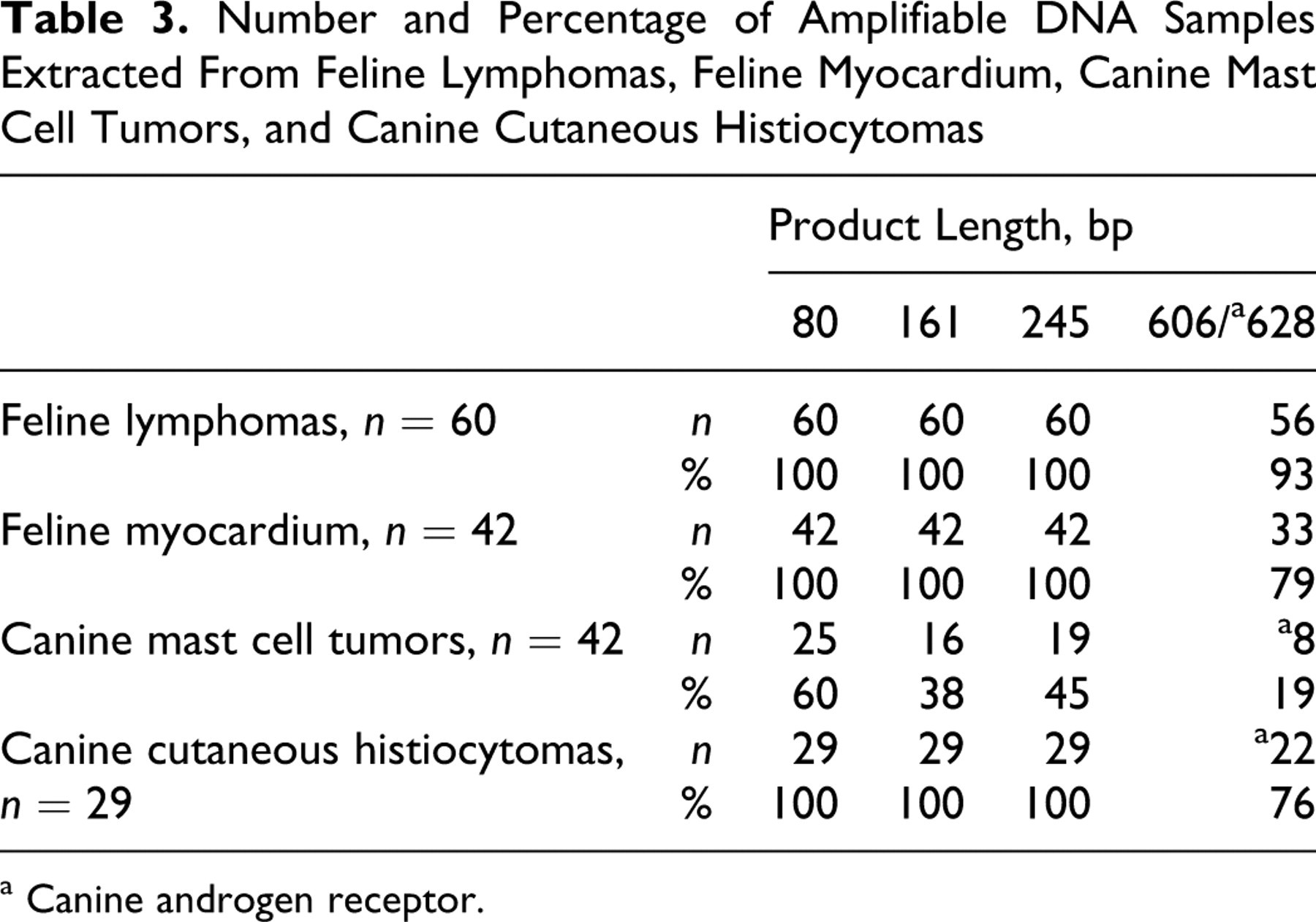
a Canine androgen receptor.
Gel electrophoresis revealed a size range of DNA fragments retrieved from FFPE samples between 0.5 and 5 kbp (Fig. 1), and fresh-frozen tissues allowed isolation of fragments larger than 20 kbp. Although all samples were of comparable size, PCR amplification results varied.
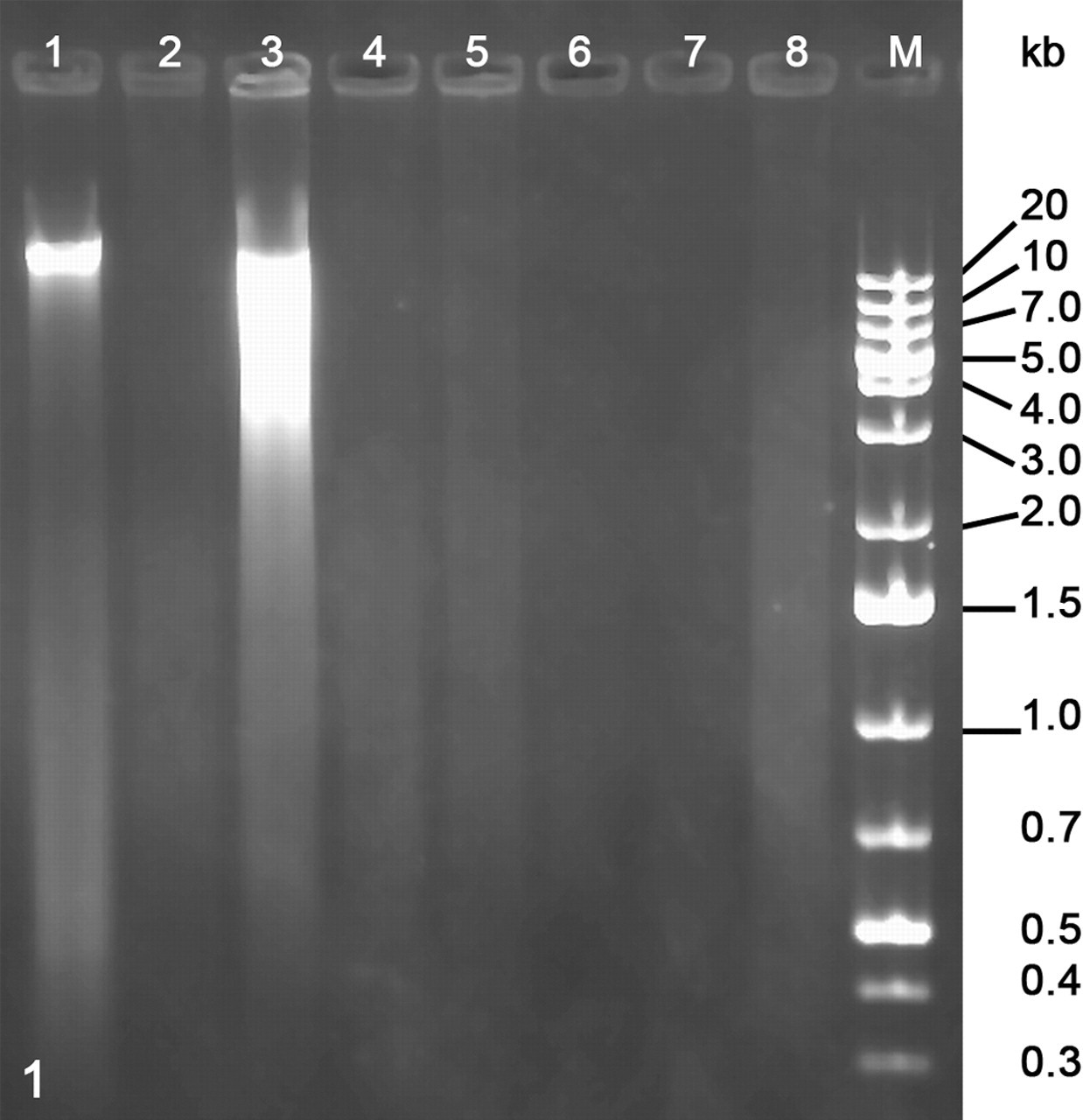
Electrophoresis of genomic DNA extracted from several canine mast cell tumors. 1 and 2 as well as 3 and 4 are paired samples of fresh-frozen (1, 3) or formalin-fixed and paraffin-embedded (FFPE) tissues (2, 4), respectively. 5 to 8 are FFPE samples of different tumors. From the first tumor (1 and 2), we obtained all PCR products; from the second (3 and 4), none. The tumor in lane 5 yielded also all bands, lane 6 yielded bands up to the 245-bp fragment, lane 7 yielded just glyceraldehyde-3-phosphate-dehydrogenase (GAPDH; 161 bp), and lane 8 yielded no bands. Size range of DNA fragments from FFPE material is between 0.5 and 5 kbp.
The heat treatment, thickness of paraffin sections, and extensive digestion time were identified as crucial factors for the satisfactory DNA yields and the most crucial differences to previously reported protocols (Tables 1, 4, and 5). Paraffin sections larger than 2 μm in thickness and a digestion time less than 48 hours markedly reduced DNA yield. Fine sectioning most probably enables a more thorough homogenization and easier digestion of protein-DNA cross-links and thereby increases DNA yield. In a diagnostic setting, the increased time for incubation seems to be a drawback of our protocol, but DNA yield often is a crucial point in research settings.
Comparison Technical Aspects of Previous Studies
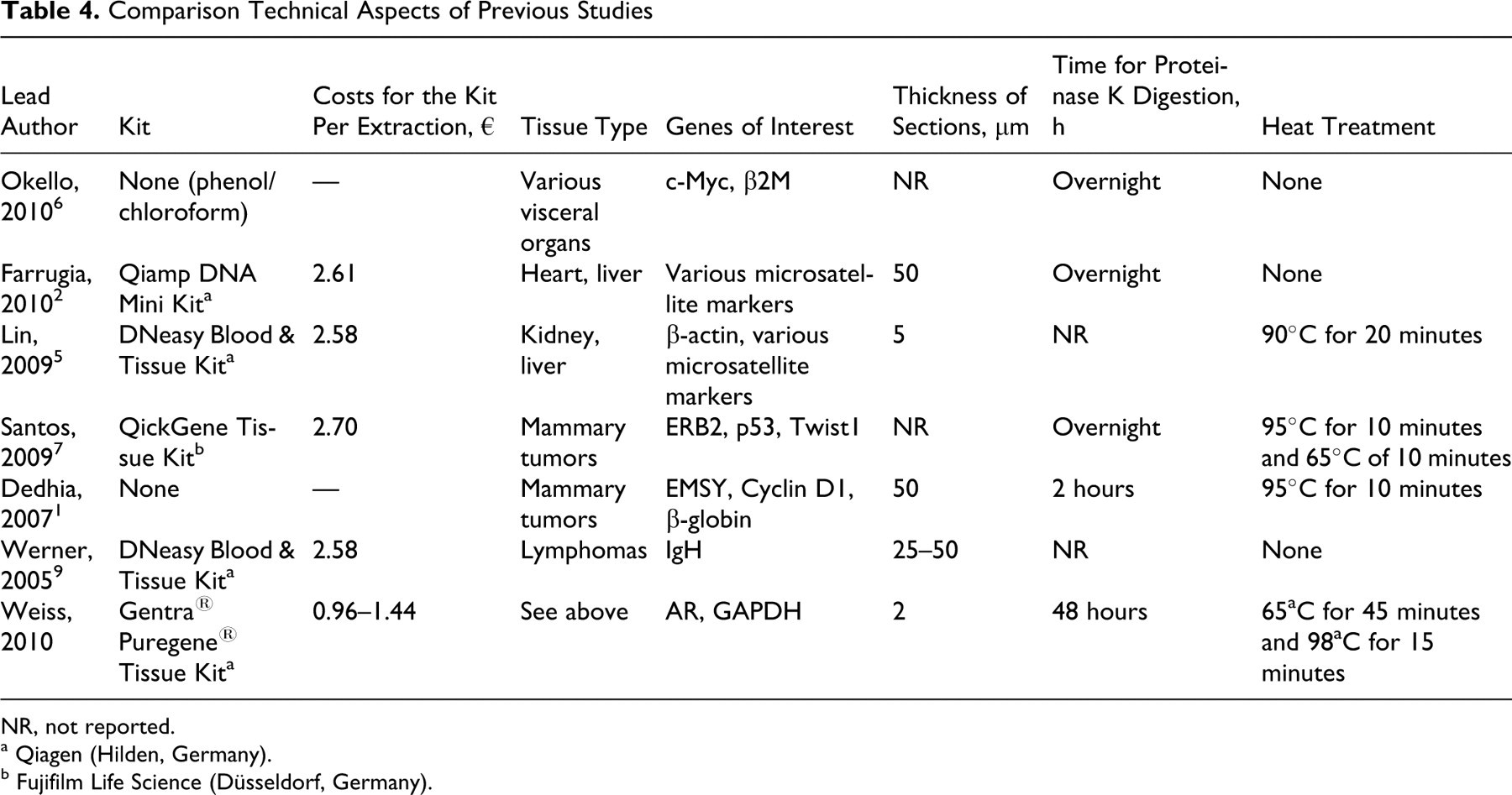
NR, not reported.
a Qiagen (Hilden, Germany).
b Fujifilm Life Science (Düsseldorf, Germany).
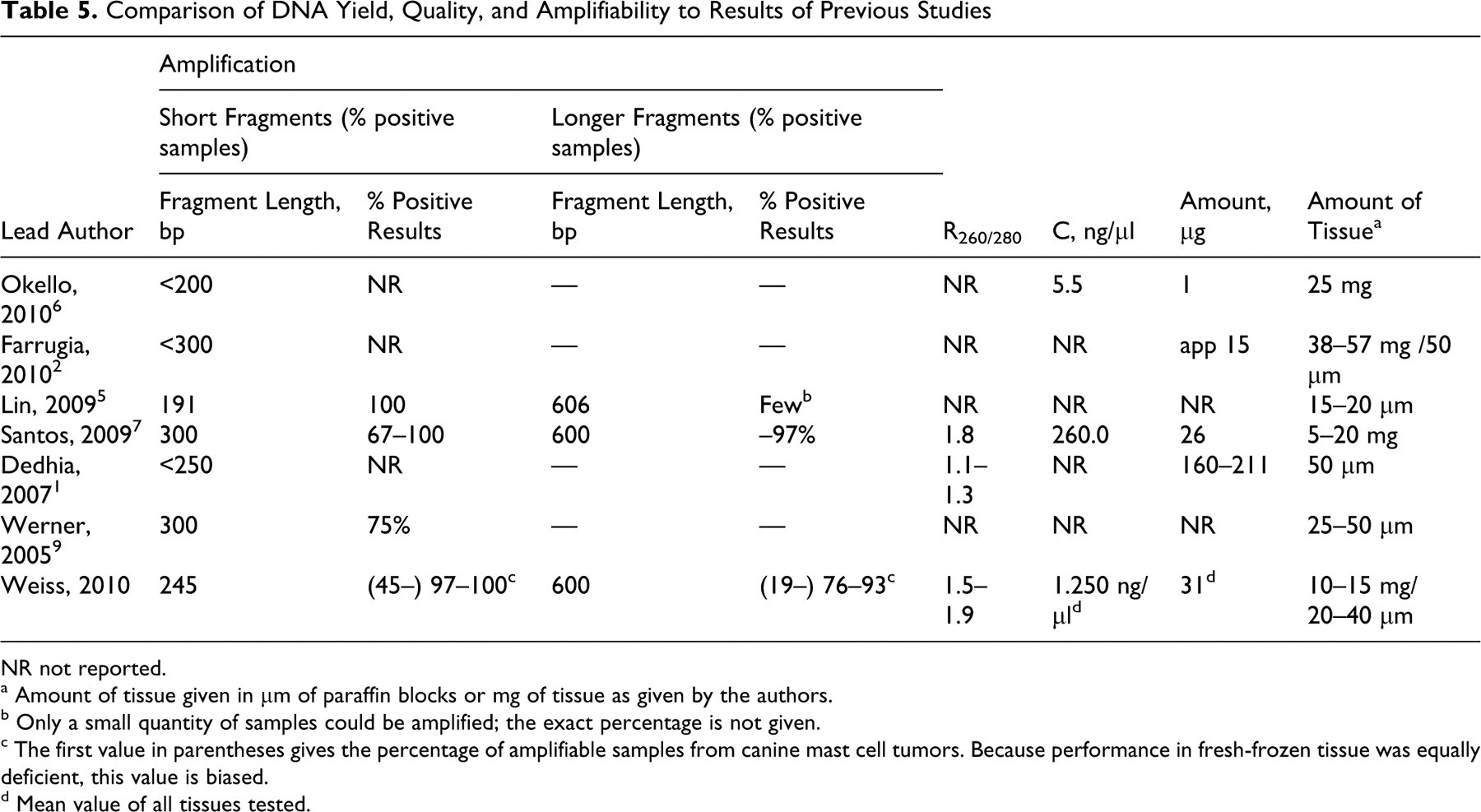
Comparison of DNA Yield, Quality, and Amplifiability to Results of Previous Studies
NR not reported.
a Amount of tissue given in μm of paraffin blocks or mg of tissue as given by the authors.
b Only a small quantity of samples could be amplified; the exact percentage is not given.
c The first value in parentheses gives the percentage of amplifiable samples from canine mast cell tumors. Because performance in fresh-frozen tissue was equally deficient, this value is biased.
d Mean value of all tissues tested.
In contrast to an earlier study that investigated clonal recombination of T cell receptor γ variants in feline lymphomas from FFPE tissues (data not shown), varying compositions of PCR master mix had no effect on amplification efficiency of the androgen receptor PCR.
The cause for less successful DNA amplification from canine mast cell tumors is not known. Interestingly, PCR amplification of genomic DNA isolated from fresh-frozen mast cell tumors failed completely in 15 of 35 cases (43%) where frozen material was available. Possible reasons for these findings may be mast cell tumor–derived DNases or retention of PCR inhibitors during DNA extraction from this tissue type. From the gel electrophoresis (Fig. 1), it seems obvious that factors other than DNA fragmentation must play a role. The fresh-frozen sample that yielded no bands is fragmented more than the other one but not so much that it should not yield bands of up to 600 bp.
The overall amplification efficiency in tissue other than mast cell tumors was comparable to a recent report of a DNA extraction protocol for FFPE tissue 7 but superior to other studies where efficient amplification of DNA fragments larger than 300 bp was unsuccessful. 1,2,5,6 A comparative overview of the different protocols for DNA isolation of FFPE is given in Tables 4 and 5. A direct comparison of our results with the literature was hampered due to different study protocols in the published data.
In summary, FFPE tissues processed with the protocol herein described yield high-quantity and high-quality genomic DNA. However, increased time for incubation could be a limiting factor.
Footnotes
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
