Abstract
The expression of p63 and E-cadherin was studied in 22 oral squamous cell carcinomas in the dog according to immunohistochemical techniques. The association between these markers and clinicopathologic parameters was assessed. All tumor cells studied showed enhanced p63 expression. Regarding E-cadherin expression, 17 of 22 cases (77.3%) showed decreased immunoreactivity, and in 13 of 22 cases (59.1%), its expression was cytoplasmic. Neither p63 nor E-cadherin expression patterns were associated with tumor size, bone invasion, or lymph node metastasis. p63 score was related to proliferating cell nuclear antigen proliferative index (P = .020). A statistically significant correlation between the expression patterns of these 2 markers was noted (P = .026). Furthermore, they were related with tumor grade. An atypical p63 labeling and a cytoplasmic E-cadherin staining were statistically related with a higher tumor grade (P = .022 and P = .017, respectively). These findings suggest that changes in p63 and E-cadherin expression are frequent events in oral squamous cell carcinoma in dogs.
Squamous cell carcinoma (SCC) is the second-most frequent oral cancer in dogs 23 and one of the most common head and neck neoplasias in humans. 2,42 In spite of its low metastatic rate, this tumor is locally aggressive to the bone and other surrounding soft tissues. 23 Regardless of location, stage, and treatment approaches, local recurrence is very common, especially when clean surgical margins cannot be achieved.
The role of p63 and E-cadherin expression in human head and neck SCC has been extensively studied, mainly with reference to their implications for prognosis. 4,12 However, there is a lack of understanding of the molecular pathogenesis underlying this cancer. 37 Substantial data indicate that both proteins play a relevant role in SCC initiation and progression. 16,29 Abnormal expression patterns of these 2 proteins have been associated with the decrease of epithelial characteristics of squamous cells and the acquisition of a mesenchymal-like phenotype. 18,31,39,40
The tumor protein p63 is a member of the p53 protein family, essential for the development of stratified epithelia, playing a pivotal role in the regulation of cellular adhesion and survival of epithelial cells. 7,43 In normal stratified squamous epithelia, p63 expression shows a restricted pattern of nuclear immunoreactivity, manifested predominantly in basal cell layers with a gradual down expression in more differentiated layers and an absence of expression in more superficial ones. 10 It is highly expressed oral SCC, with nuclear expression disseminated throughout the tumor, in all layers, with different intensities. 27 The p63 gene is transcribed from 2 alternative promoters, TAp63 and ΔNp63, producing 6 isoforms: α, β, γ. 8,32 TAp63 isoforms regulate mainly the expression of epithelial differentiation genes, 6,20,32 and ΔNp63 is involved in the pathways of cell adhesion, differentiation, and cell proliferation, being predominantly expressed in human head and neck SCC. 6,30,32,41 Expression of p63 was evaluated in myoepithelial cells of mammary tissues in the dog and in primary cutaneous glandular carcinomas. 34,38 Also, both p63 and ΔNp63 expression was studied in canine mammary mixed tumors. 3
E-cadherin is a calcium-dependent cell-to-cell adhesion surface glycoprotein with a relevant role in epithelial cell behavior, tissue formation, and cancer progression. 39,40 Progression of oral SCC has been associated with low E-cadherin expression, 16 which is more commonly observed in poorly differentiated carcinomas and lymph node metastasis. 2,36 E-cadherin expression is considered an independent marker for prediction of survival time in cases of human oral SCC. 12,22 In dogs, this marker has mostly been studied in mammary tumors, specifically with the reduction of its expression associated with tumor size, histologic type, invasion, lymph node metastasis, grade, and mitotic index. 5,14,25
Proliferating cell nuclear antigen (PCNA) is a 36-kDa nonhistonic nuclear peptide necessary in DNA replication and cell proliferation, and it has been used as a proliferation marker by different researchers in various canine tumors 15,21,24 and in oral SCC in humans. 28
In the present study, we evaluated the expression of p63 and E-cadherin in 22 canine oral SCCs by immunohistochemistry, relating these markers with clinicopathologic parameters, including the proliferative index revealed by the expression of PCNA.
Materials and Methods
Cases
From January 2008 to January 2012, 22 cases of canine oral tumors were obtained from various surgery services and retrospectively evaluated. Inclusion criteria comprised availability of records, histologic diagnosis of SCC, and the existence of surgical specimens properly embedded in paraffin for immunohistochemistry. All dogs underwent en block surgery for the removal of the tumor. Complete records were reviewed, including age, breed, sex, body weight, tumor location and size, bone invasion, and lymph node metastasis at the time of presentation. Staging was determined by computed tomography, based on the TNM classification of tumors in domestic animals. Tumors were classified as T1 (<1 cm in diameter), T2 (1–3 cm) or T3 (>3 cm or the presence of ulceration); as N0 (absence of node invasion) or N1 (node invasion); and as M0 (absence of distant metastasis) or M1 (distant metastasis), according to established procedures. 9 Lymph node metastasis was evaluated by computed tomography and fine-needle aspiration.
Histopathologic and Immunohistochemistry Analysis
Tumor samples were fixed in 10% buffered formalin for 2 days and routinely processed for histopathologic diagnosis. Sections of 3 μm were cut for routine histologic examination and stained with hematoxylin and eosin. Tumors were graded on a scale of I to III. 17 Grade I, or well-differentiated SCC, corresponded to well-differentiated SCC resembling normal epithelium: central accumulations of keratin pearls are surrounded by different cell layers with decreasing keratinization, from the center to periphery, being basal cells nonkeratinized. In grade II, or moderately differentiated SCC, less keratinization, more nuclear pleomorphism, and mitotic figures were observed. In grade III, or poorly differentiated SCC, there is no sign of keratin pearl formation; mitotic activity is higher; cellular pleomorphism and immature cells are present; and general architecture is distant from the tissue of origin.
Immunostaining was performed as follows: antigen retrieval in citrate buffer (pH = 6.2) in a microwave for 15 minutes at 600 W; incubation with primary antibodies listed in Table 1; and the use of the Novolink Max Polymer Detection System (Novocastra).
Antibodies Used and Technical Details.a

aAntigen retrieval for each antigen: Microwave 15 minutes in citrate buffer (pH = 6.2). Detection system/source for each antigen: Polymer System/Novocastra.
Negative controls for immunohistochemistry were achieved by replacing the primary antibody with buffered saline. Normal gingiva in each section was used as internal positive controls for p63 and E-cadherin. Positive controls for PCNA were from canine tonsil. For PCNA analyses, the cutoff value was 10%. 21
Two veterinary pathologists performed the microscopy assessment of the immunostaining, including a diplomat of the European College of Veterinary Pathology (M.C.P.). In cases where a different classification was made, an agreement was reached. All samples were photographed, and the software AnalySIS Image Processing 2.0.0.6 (SMC Networks, Inc.) was used for cell count.
The percentage of p63 was quantitatively scored as follows: 0, absence of staining; 1, <50% nuclear staining; 2, >50% and <70%; 3, >70%, as previously described. 4 The expression of p63 was classified as typical or atypical. Typical expression was considered when it was similar to that observed in normal epithelium—that is, an intense nuclear staining in basal and suprabasal layers while no staining in the most superficial. Atypical patterns were considered when intense nuclear staining was observed in all layers, including superficial layers.
E-cadherin-positive tumor cells were semiquantitatively calculated and scored for each sample from 0 (absence of staining) to 4 (maximum staining intensity), as previously published. 33 Tumors were also classified according to membrane or cytoplasmic immunoreactivity. 14 The technique was tested and refined before any attempted for classification. To reduce variability, all samples were processed in the same day.
Proliferative Index
A proliferative index was calculated as the percentage of positive cells for PCNA in a total of 1000 neoplastic cells, counted in the most representative areas.
Statistical Analysis
p63 and PCNA were evaluated with analysis of variance and Student t test for independent samples to access the differences of means among factors, such as tumor size, bone invasion, lymph node metastasis, and grade. When the assumption of normality was not verified, an alternative nonparametric approach was used—namely, Kruskal-Wallis and Mann-Whitney. Associations between continuous variables were assessed with the Pearson correlation or, if normality or linearity failed, the Spearman correlation.
Associations between expression patterns of either p63 or E-cadherin and clinicopathologic variables were evaluated with chi-square tests and Fisher exact tests.
All statistical analyses were performed with SPSS 20.0. P values less than .05 were considered statistically significant.
Results
The mean age of the study sample was 9.9 years (range, 6.0–13.0), and weight was 22.0 kg (range, 4.0–35.0). There were 13 males and 9 females. Five animals were mixed breed, 4 Standard Poodle, 2 Doberman Pincher, 2 Labrador Retriever, 2 German Shepard, and 7 from other breeds.
Regarding size, 8 tumors were registered as T1 (<1 cm in diameter), another 8 as T2 (1–3 cm), and 5 as T3 (>3 cm). With respect to differentiation, 68.2% of the tumors (n = 15 of 22) were classified as grade II and 31.8% (n = 7) as grade III. Bone invasion was verified in 59.1% of the cases (n = 13) and lymph node metastasis in 18.2% (n = 4).
Clinicopathologic and immunohistochemical features of the tumors studied for p63 expression are summarized in Table 2.
Association Between p63 Expression and Clinicopathologic Parametersa
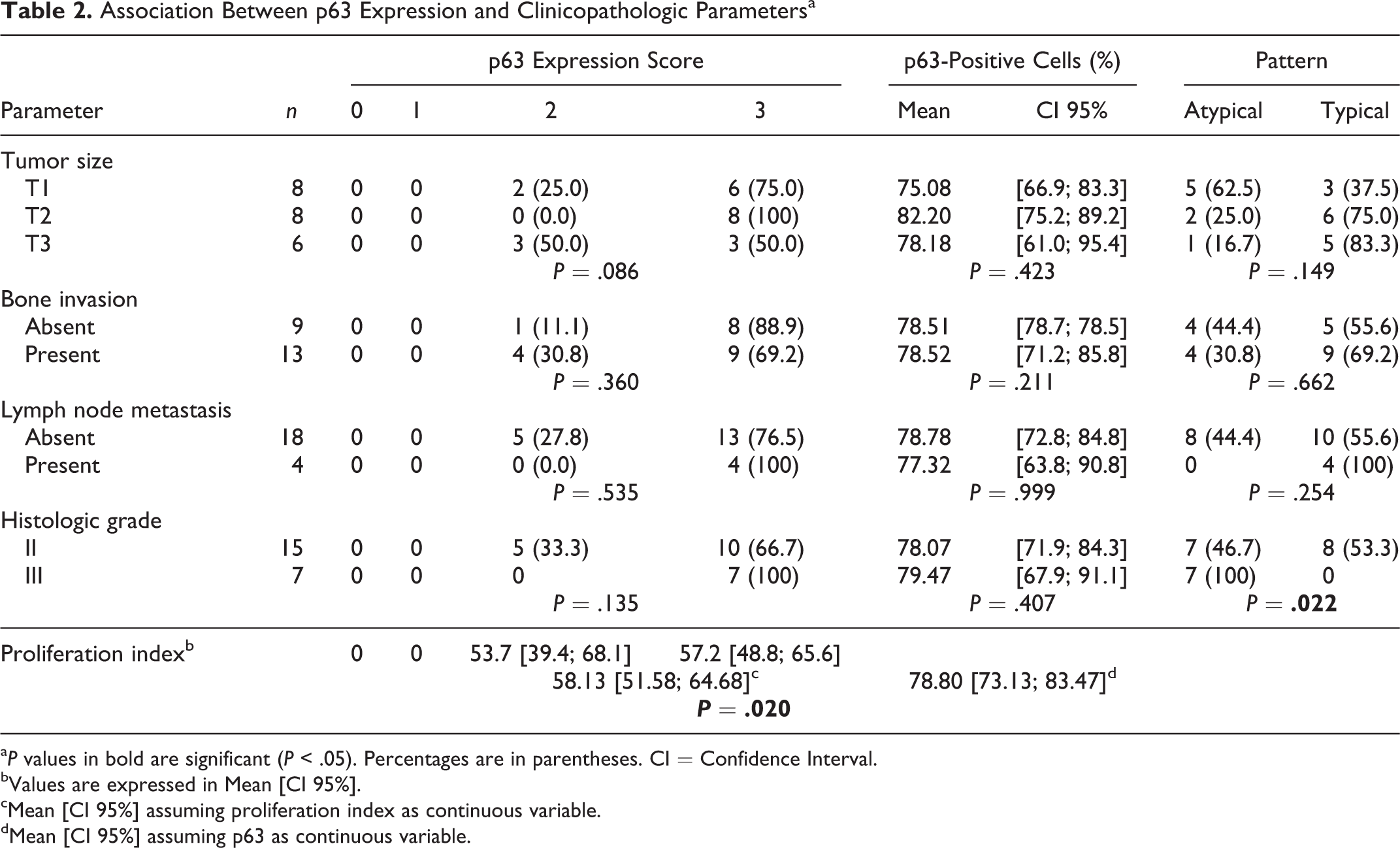
a P values in bold are significant (P < .05). Percentages are in parentheses. CI = Confidence Interval.
bValues are expressed in Mean [CI 95%].
cMean [CI 95%] assuming proliferation index as continuous variable.
dMean [CI 95%] assuming p63 as continuous variable.
Regarding p63 staining, 77.3% (n = 17) of the tumors in this study were classified as score 3, 22.7% (n = 5) as score 2, and none as score 1 or 0. Scores were not obviously related with the grade of the tumor. p63 staining patterns of tumor cells were found to be variable. In grade II SCC, the labelling was typical (Fig. 1), in contrast to grade III, where all tumors showed atypical labeling with strong positive staining of all epithelial cell layers (Fig. 2). Atypical labeling was significantly correlated to the grade of the tumor (P = .022). PCNA labeling was related to p63 score (P = .020). Size, bone invasion, and lymph node metastasis were not significantly correlated to p63 score or pattern of expression.
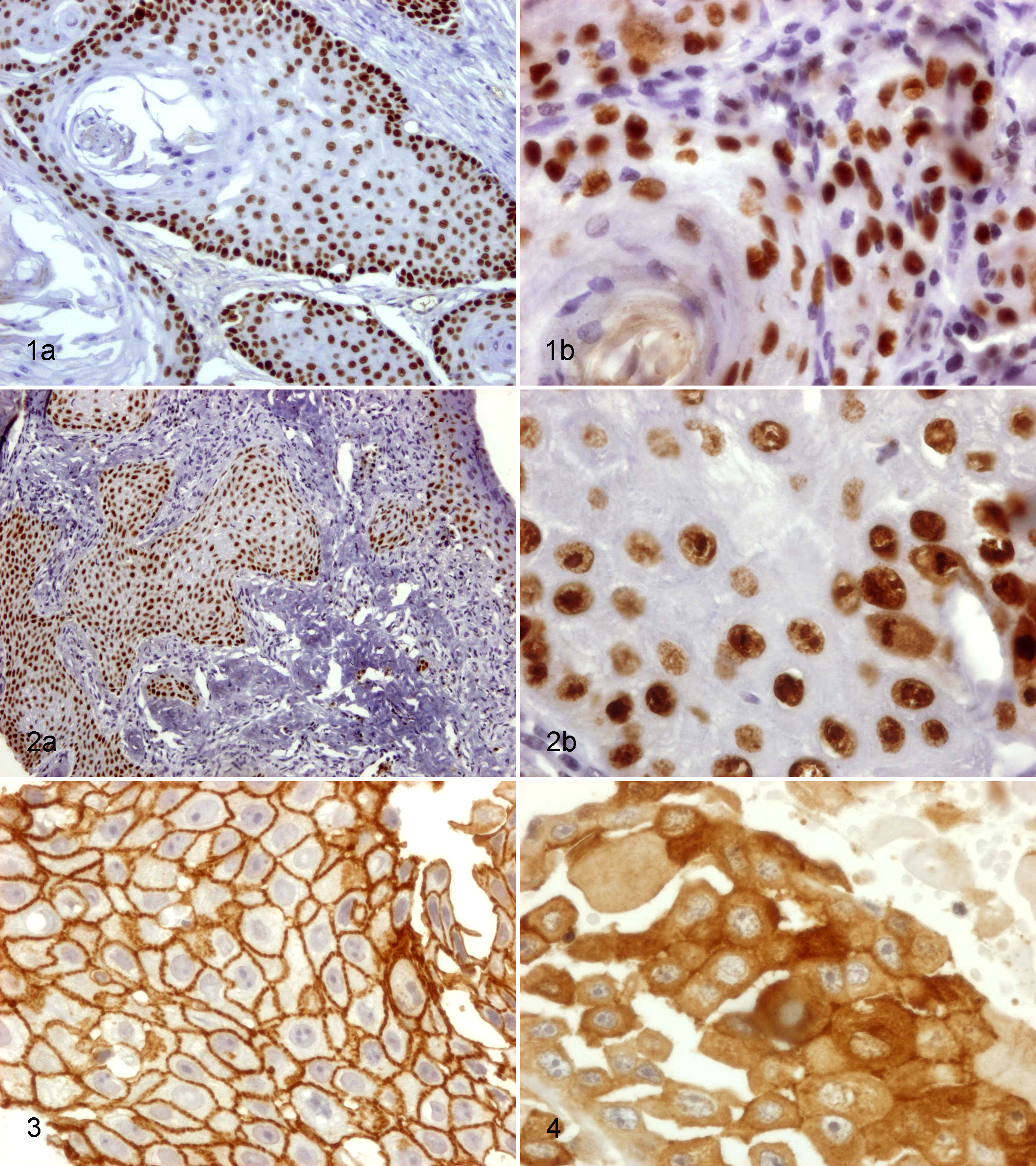
Grade II squamous cell carcinoma (SCC), oral cavity, dog. Typical expression pattern for p63: Nuclear staining is strongly positive in basal and parabasal layers, being faint and absent in the more superficial layers. Immunohistochemistry (IHC) for p63. (a) Original magnification. (b) Higher magnification.
E-cadherin expression was found in all tumors (Figs. 3, 4). The association between E-cadherin expression and SCC clinicopathologic features is summarized in Table 3: 77.3% (n = 17 of 22) of observed tumors scored less than 4. In 59.1% (n = 13) of the cases, E-cadherin expression was cytoplasmic (Fig. 4). This expression pattern was correlated with histologic grade (P = .017). None of the examined parameters, such as size, bone invasion, lymph node metastasis, or proliferative index, were significantly related to E-cadherin score or expression pattern.
Association Between E-Cadherin Expression and Clinicopathologic Parameters.a
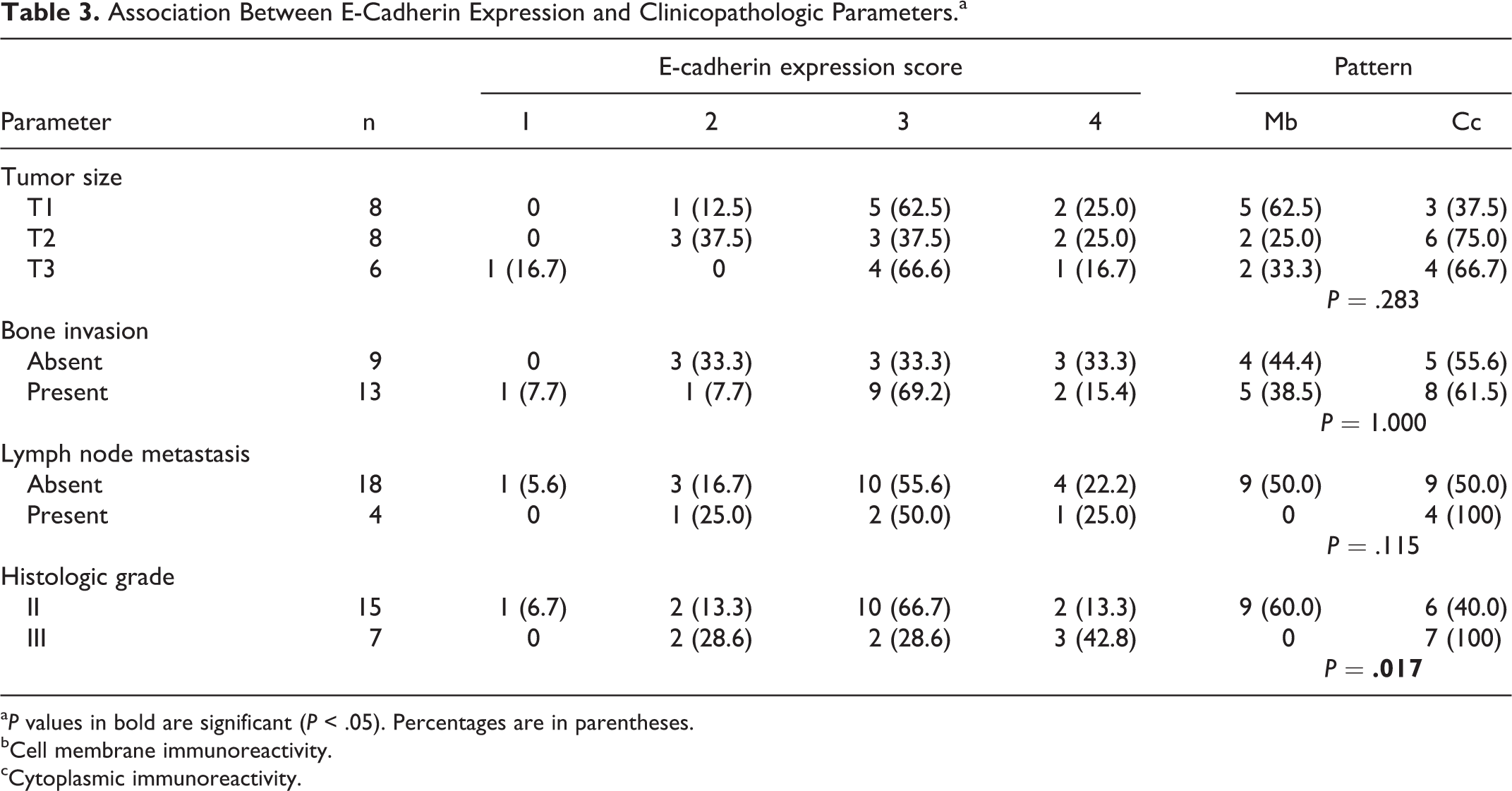
a P values in bold are significant (P < .05). Percentages are in parentheses.
bCell membrane immunoreactivity.
cCytoplasmic immunoreactivity.
A cytoplasmic E-cadherin expression was observed in 76.9% (n = 10 of 13) of the tumors with an atypical p63 expression, while membranous expression was observed in 75% (n = 6 of 8) of those with a typical p63 expression. An association was observed between cytoplasmic expression pattern of E-cadherin and an atypical labelling of p63 (P = .026).
Discussion
Tumor protein 63 is known to play an oncogenic role by promoting cancer stem cell survival, differentiation, and proliferation. 7,10,42,43 In this study, all oral SCCs showed positive p63 expression, with 77.3% of the tumors showing more than 70% of stained cells. These observations are suggestive of a reactivation of this protein, secondary to gene mutation and/or overexpression. The p63 score was found to be significantly related to the PCNA index, probably due to the role of this protein in epithelial cell proliferation. p63 overexpression per se seems to favor keratinocyte proliferation and, indirectly, enhancement of metastasizing capacity. 19,20 In humans, it was reported that a high labeling index of PCNA in oral SCC is related to p63 expression. 19,20 To our knowledge, this relationship has not yet been established in oral SCC in dog.
Different labeling patterns for p63 were observed in the tumors studied. Typical labelling was seen mainly in grade II tumors (53.3%). In contrast, all SCC grade III tumors showed an atypical labeling, with all nuclei in all layers positive, albeit with different intensities. This pattern was significantly related to tumor grade, which can be explained by the fact that p63 acts as an “epithelial organizer,” with a key role in stemness, senescence, cell death, and cell cycle arrest—all determinant features in cancer. 26 In this study, there were no well-differentiated tumors diagnosed; therefore, no conclusions can be drawn on staining patterns in low-grade tumors.
Ventana’s clone 4A4 is a mouse anti-human monoclonal antibody, which reacts with the first 205 amino acids of the p63 protein. It showed optimal labeling with a strong, distinct nuclear staining in all tests used, which included normal skin and oral mucosa. This antibody indiscriminately recognizes all p63 isoforms and was previously used successfully in the dog. 3,34,38 The lack of specificity in the evaluation of the various isoforms was one of the limitations in the present study. ΔNp63 isoforms are expressed at consistently higher intracellular levels compared with TA isoforms. 32 Possibly in dogs, in a similar way as in humans, 6,30,32 the ΔNp63 isoforms are predominant in tumors, and TAp63 isoforms are almost undetectable. 6,32 In a study performed in canine mammary tumors, the antibodies labeling all p63 isoforms or just the ΔNp63 group provided similar results in most of the samples studied, suggesting that ΔNp63 was the predominant isoform. 3
Expression of E-cadherin was evident in all tumors. In most of the samples studied, cell membrane E-cadherin expression was decreased or changed to cytoplasmic staining. This expression varied with histologic grade. Although without statistical significance, all cases with lymph node metastasis showed cytoplasmic expression of E-cadherin. These findings are consistent with a loss of cell-to-cell adhesion, secondary to a downregulation of this protein with the loss of tumor differentiation. Some authors propose that a low E-cadherin expression could predict lymph node metastasis in cases of human oral SCC, while others relate it to tumor differentiation. 11,13,16 Similar results were obtained in canine mammary tumors. 14,25,35
Our findings suggest that there is an association between p63 and E-cadherin expression and tumor grade. Furthermore, cytoplasmic E-cadherin expression was related with an atypical labeling of p63 expression. Taken together, these results suggest that a change in the expression of these 2 molecules could be a common event in oral SCC in dogs, similar to what occurs in humans. 7,12
Epithelial-to-mesenchymal transition is an essential process in tumor progression, being implicated in gain of invasiveness and capacity to escape from the primary site and metastasize. 40 E-cadherin is a hallmark in this process, being involved in the epithelial-to-mesenchymal transition mechanism also in SCC, whereas a downregulation of this cell adhesion molecule is associated with a poor prognosis in terms of recurrence and mortality. 12,18,40 Contrarily, recent reports showed that p63 expression reintroduces epithelial characteristics in mesenchymal type cells and therefore reverses the mesenchymal tendency of epithelial tumors. 18,31 These 2 markers seem to act as regulatory molecules in the epithelial-to-mesenchymal transition equilibrium. 6,27,44 Advanced knowledge on tumor behavior regarding these and other markers would be very important to assess response to treatment and would lead to novel therapies with higher efficacy and less toxicity.
Footnotes
Acknowledgments
The authors thank John M. Archibald for final reading and language correction and Sandra Carvalho for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
