Abstract
Recently, histologic subtypes of oral squamous cell carcinoma (SCC) corresponding to the human classification scheme have been proposed for dogs. A papillary squamous cell carcinoma subtype is characterized by dominant exophytic architectural growth with limited invasion, a lower metastatic rate, and better overall survival compared with conventional SCC. Whereas most canine oral conventional SCCs are easily diagnosed by histologic examination, the diagnosis of canine oral papillary squamous cell carcinoma (COPSCC) can be challenging since the exophytic portion lacks histologic features of malignancy and appears similar to oral nonviral papillomas. In contrast, the invasive portion of COPSCC has morphologic similarities to conventional SCC and canine acanthomatous ameloblastoma. The goals of this study were to immunophenotype these 3 entities and to potentially identify discriminating markers. A panel of 17 immunohistochemical markers was investigated in tissue microarrays that included 25 COPSCCs, 10 conventional SCCs, and 10 canine acanthomatous ameloblastomas. Additionally, COPSCCs were screened for papillomavirus as a potential cause using immunohistochemistry and in situ hybridization. COPSCC had immunophenotypical similarities with conventional SCC and acanthomatous ameloblastoma, but the combined differences in immunolabeling for AE1/AE3, 34βE12, p63, and calretinin discriminated between the entities. Papillomavirus was not detected in any COPSCC, making a viral pathogenesis unlikely. A better understanding of the immunophenotype of COPSCC will aid in a more accurate diagnosis and potentially improve therapeutic approaches.
Keywords
Oral squamous cell carcinoma (SCC) has long been recognized as the most common malignant epithelial neoplasm in the oral cavity of dogs. 10,14 In humans, the World Health Organization classification defines multiple subtypes of oral SCC based on histomorphology. 36 Accurate diagnosis of these different morphologic subtypes is important, since individual subtypes are associated with different prognoses. 29 Subclassification of oral SCC in dogs has historically been limited to identification of the site of origin of the neoplasm with tonsillar localization being associated with a more aggressive biological behavior. 23 Recently, subtypes of nontonsillar oral SCC have been reported in dogs that are morphologically similar to entities recognized under the World Health Organization classification scheme of human oral SCCs. 27 Such subtypes in dogs include basaloid SCC, papillary SCC, spindle cell carcinoma, adenosquamous carcinoma, and conventional SCC. Conventional SCCs are further defined as well differentiated, moderately differentiated, or poorly differentiated, or grade 1, 2, or 3, respectively. 27 Whereas the canine subtypes of oral SCC are histologically similar to their human counterparts, the biological behavior of most canine subtypes of oral SCC has not been studied in detail.
Diagnostically, differentiation of canine oral papillary squamous cell carcinoma (COPSCC) from other subtypes of oral SCC is particularly important. It has been suggested that COPSCC has a more favorable outcome of COPSCC with a lower metastatic rate and better overall survival of affected dogs, based on their exophytic character and limited invasiveness. 9,31 However, an accurate diagnosis of this subtype may be difficult as its morphologic features commonly overlap with those of both benign and malignant proliferative oral epithelial lesions.
COPSCCs present as solitary neoplasms with both exophytic superficial papillary portions that extend above the surface of the surrounding gingiva, as well as deep invasive trabeculae and islands that extend into the underlying gingival stroma and/or bone. The superficial papillary portions of these lesions consist of multiple, finger-like projections lined by neoplastic stratified squamous epithelium with minimal or no keratinization. 9,31 The deep invasive trabeculae and islands are often peripherally lined by cuboidal to columnar cells with basally orientated, palisading nuclei and are centrally composed of sheets of plump polygonal squamous cells that occasionally whorl and undergo keratinization to form keratin pearls. 26 Superficial portions of these neoplasms may be confused with oral nonviral squamous papillomas especially at low magnification, or if only the superficial portion of the mass is submitted for histopathologic assessment. The deep invasive portions may be confused with conventional SCC or canine acanthomatous ameloblastoma. Given the difficulty in differentiating COPSCC from other oral proliferative epithelial lesions (especially within incompletely excised biopsy specimens) and the prognostic significance of an accurate diagnosis, an investigation of the histogenesis of these lesions to identify potential markers of differentiation for diagnostic purposes is warranted.
In dogs, several immunohistochemical markers have been evaluated to characterize normal oral/dental tissue and a variety of oral neoplasms. 3,12,25 Distinctive immunohistochemical profiles of cytokeratins, various markers of odontogenic epithelium, and calretinin have been described in canine conventional SCC and canine acanthomatous ameloblastoma. Immunoreactivity of such markers in COPSCC has not been examined in dogs, but studies in humans have established a distinct immunophenotype for papillary SCC. 9,18,34 Similar to conventional SCCs, papillary SCCs express pancytokeratin and high-molecular-weight cytokeratins but are not immunoreactive for low-molecular-weight cytokeratins or markers of mesenchymal, neural, or muscle differentiation. 33 Overexpression of p53 protein has been uniquely observed in papillary SCC, but the role of p53 in development of these neoplasms remains unclear. 9,34
Specific risk factors have been suggested for the development of oral SCC in humans, but similar risk factors have not been identified in dogs. In humans, predisposing factors include long-term tobacco exposure and alcohol consumption, which are factors not likely to be of importance in dogs. Although few studies reported the presence of human papillomavirus (HPV) in papillary SCC, 7 a putative role of HPV in papillary SCC tumorigenesis has not been established to date. Although no such association with papillomavirus has been established in dogs either, an infection with papillomavirus was initially hypothesized as the potential cause of COPSCC in 2 reports due to their papillary appearance and their occurrence in young dogs. 28,32
The present study compared the immunophenotype of a series of COPSCCs with that of canine conventional SCCs and canine acanthomatous ameloblastomas. Characterization of the immunophenotype of COPSCCs could allow a better understanding of their histogenesis and potentially lead to the identification of a diagnostic immunohistochemical profile. In addition, to investigate a potential causative role of papillomaviruses, all COPSCCs were analyzed for papillomavirus infection using immunohistochemistry (IHC) and in situ hybridization (ISH).
Materials and Methods
Tumor Samples and Selection of Material
The study population included 50 dogs from which biopsies of oral mass lesions were submitted to the Michigan State University Veterinary Diagnostic Laboratory from 1 January 2004 to 31 December 2014 for diagnostic purposes. Cases were selected by screening the Veterinary Diagnostic Laboratory archive for cases previously diagnosed as COPSCC, conventional SCC, acanthomatous ameloblastoma, or gingival hyperplasia. Diagnoses were confirmed by histologic review of hematoxylin and eosin–stained, 4-µm sections from formalin-fixed, paraffin-embedded tissue blocks. Cases were excluded if sufficient tissue was not available.
Included cases represented 25 COPSCCs, 10 conventional SCCs, 10 canine acanthomatous ameloblastomas, and 5 cases of gingival hyperplasia. COPSCCs were defined as having superficial papillary/exophytic proliferations of well-differentiated, neoplastic epithelial cells supported by fibrovascular stalks along with invasion of the deep stroma and/or bone by nests and trabeculae of neoplastic cells. In contrast, conventional SCCs were composed of nests and trabeculae of malignant stratified squamous epithelial cells that invaded though basement membranes. Grading of conventional SCCs was done based on the following previously described grading system: grade 1 (well-differentiated) conventional SCC closely resembled normal squamous epithelium showing orderly progression from nonkeratinized basal cells at the periphery to large keratinized cells at the center of the neoplastic epithelial structure with distinct keratin pearls; grade 2 (moderately differentiated) conventional SCC exhibited marked nuclear atypia and increased mitotic activity with less keratinization; grade 3 (poorly differentiated) conventional SCC was composed of immature epithelial cells with numerous mitoses, minimal keratinization, and no keratin pearls. 27 Canine acanthomatous ameloblastomas were composed of invasive neoplastic odontogenic epithelial cells that had prominent intercellular bridges and antibasal, peripheral palisading nuclei within basal cells of nests and trabeculae.
Tissue Microarray
Using hematoxylin and eosin–stained slides as a reference, tissue microarrays (TMAs) were created using 10 representative areas from each of the 25 COPSCCs (including 5 from areas predominately composed of neoplastic epithelial cells from the papillary regions and 5 areas from the deeper invasive regions) and 5 representative areas of neoplastic cells from each of the 10 conventional SCCs and 10 acanthomatous ameloblastomas. These areas were marked on the corresponding formalin-fixed, paraffin-embedded tissue blocks and then captured with a Manual Tissue Arrayer MTA-1 (Beecher Instruments, Inc., Sun Prairie, WI) per the manufacturer’s instructions. Briefly, a 1.5-mm-diameter thin-walled punch held in an X-Y precision guide was used to create holes in a recipient paraffin block, acquire tissue cores from donor blocks, and deposit harvested cores into the recipient block at defined array coordinates. Using this method, 2 TMA blocks containing a collective total of 350 cores were generated. Hematoxylin and eosin–stained 5-μm sections from the TMA blocks were examined to assess the representativeness of the cores.
Immunohistochemistry and In Situ Hybridization
For IHC, serial 4-μm sections taken from the TMA blocks and the additional cases of gingival hyperplasia mounted on positively charged slides were labeled using the pancytokeratin antibodies AE1/AE3 and MNF116, high-molecular-weight cytokeratin antibody 34βE12, and antibodies for cytokeratin 5/6, cytokeratin 7, cytokeratin 14, cytokeratin 19, CP27, amelogenin, calretinin, CDX-2, collagen IV, laminin, p53, p63, villin, vimentin, and papillomavirus antigen. IHC was performed for 34βE12, cytokeratin 7, pancytokeratin MNF116, pancytokeratin AE1/AE3, calretinin, and laminin on a BOND-MAX Automated Staining System (Leica Biosystems Inc., Bannockburn, IL) using the Bond Polymer Detection System (Leica Biosystems Inc.) with 3,3′-diaminobenzidine as chromogen; for vimentin and p53 on a Discovery Ultra automated staining system (Ventana Medical Systems, Inc., Tucson, AZ) using the UltraMap alkaline phosphatase red detection system (Ventana Medical Systems, Inc.); or for cytokeratin 5/6, cytokeratin 14, cytokeratin 19, CDX-2, collagen IV, CP27, p63, amelogenin on a Dako Autostainer Link 48 automated staining system (Agilent Technologies, Santa Clara, CA) using the Flex detection system with 3,3′-diaminobenzidine as chromogen (Agilent Technologies). For IHC, sections were deparaffinized in xylene, rehydrated through graded alcohols, and rinsed in distilled water. A detailed summary of primary antibodies, antigen retrieval methods, and concurrently labeled positive controls is shown in Table 1. Negative controls were created for each immunohistochemical marker by replacing primary antibodies with Tris buffer on additional serial sections from the TMAs. In addition, IHC for papillomavirus antigen and ISH for papillomavirus DNA were performed on the Discovery Ultra automated staining system (Ventana Medical Systems, Inc.) using the UltraMap alkaline phosphatase red detection system or the RiboMap detection system (both Ventana Medical Systems, Inc.), respectively, as previously reported. 16,20,23,30
Antibodies and Positive Control Tissue Used for Immunohistochemistry.
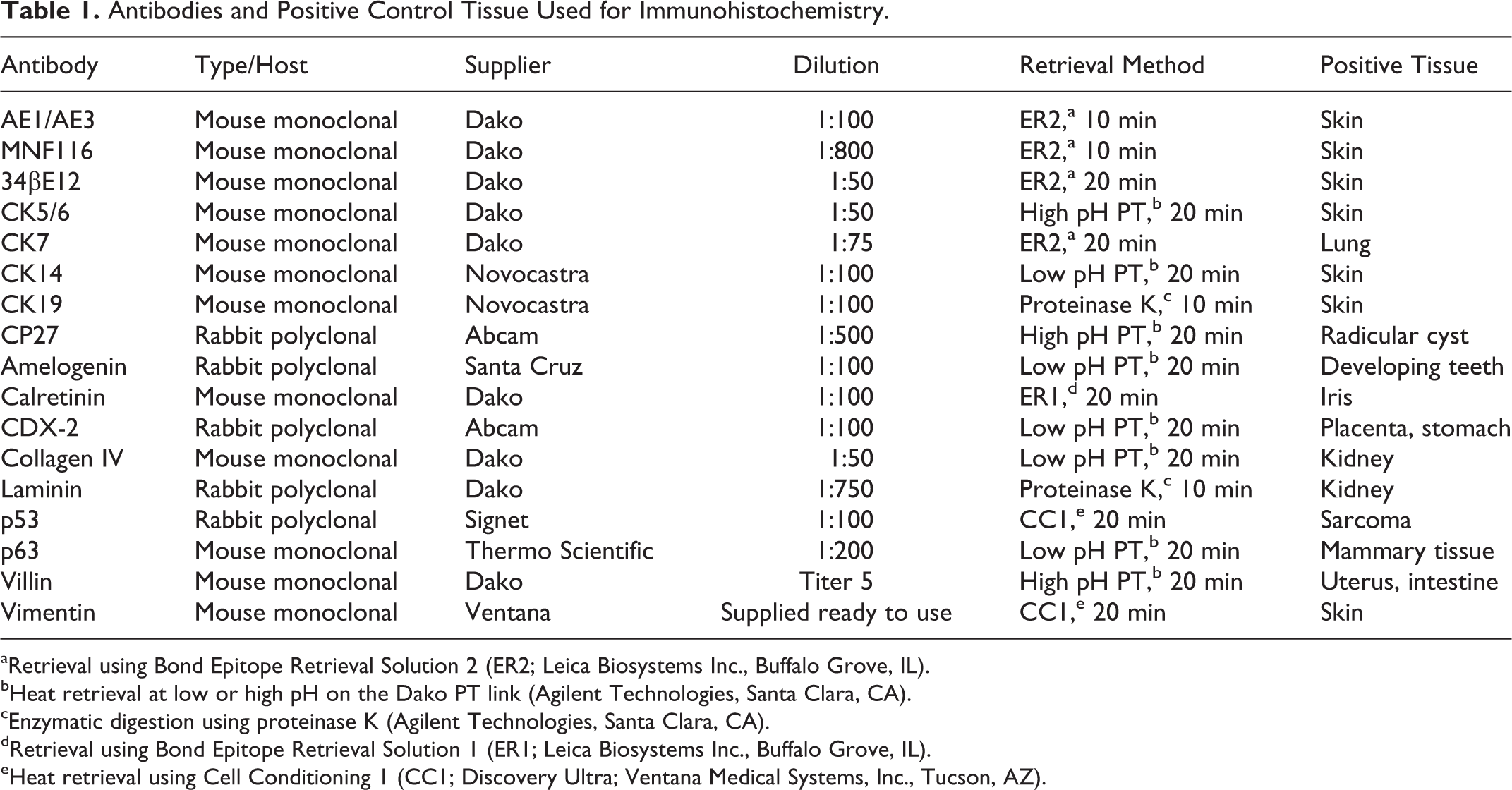
aRetrieval using Bond Epitope Retrieval Solution 2 (ER2; Leica Biosystems Inc., Buffalo Grove, IL).
bHeat retrieval at low or high pH on the Dako PT link (Agilent Technologies, Santa Clara, CA).
cEnzymatic digestion using proteinase K (Agilent Technologies, Santa Clara, CA).
dRetrieval using Bond Epitope Retrieval Solution 1 (ER1; Leica Biosystems Inc., Buffalo Grove, IL).
eHeat retrieval using Cell Conditioning 1 (CC1; Discovery Ultra; Ventana Medical Systems, Inc., Tucson, AZ).
Immunoreactivity for each antigen was evaluated by 2 independent pathologists using conventional light microscopy and categorized by the general distribution of immunoreactivity (ie, diffuse or focal), location within the neoplastic epithelium (ie, basal, suprabasal, and/or antibasal), and cellular distribution (cytoplasmic, nuclear, or both). Papillomavirus antigen or DNA was noted as detected or not detected by IHC and ISH, respectively.
Results
Clinical and Microscopic Findings
Known ages of COPSCC-affected dogs at the time of diagnosis ranged from 7 months to 13 years with the average age being 7 years and 12 of 24 dogs being 7 years old or younger. Age was unknown for 1 dog. Of the 25 dogs with COPSCC, 12 were spayed females, 1 was an intact female, 11 were castrated males, and 1 was an intact male. Ages at the time of diagnosis of dogs with conventional SCC ranged from 4 to 14 years old, and the mean was 9 years. Of the 10 dogs with conventional SCC, 5 were castrated males, 4 were spayed females, and 1 was an intact male. Ages at the time of diagnosis of dogs with acanthomatous ameloblastoma ranged from 3 to 12 years, and the mean was 8 years old. Of these 10 dogs with acanthomatous ameloblastoma, 5 were castrated males, 2 were spayed females, 2 were intact females, and 1 was an intact male.
Fourteen COPSCCs arose from the mandibular gingiva, 9 cases originated from the maxillary gingiva, and the location was unidentified in 2 dogs. Seven COPSCCs had invasion of the underlying bone. In 2 COPSCCs, basal cells of papillary fronds had nuclei arranged along the antibasal pole of the cell, thereby resembling odontogenic epithelium. One COPSCC had previously been diagnosed as a squamous papilloma due to lack of malignant features in a superficial biopsy, but the neoplasm recurred within 7 months and had bone invasion at the time of recurrence. Of the 8 conventional SCCs in which location was specified, 4 involved the maxillary gingiva, 2 involved the mandibular gingiva, and 2 were on the tongue. All cases of conventional SCC were classified as grade 2, and 2 cases invaded into bone at the time of diagnosis. Of the 10 canine acanthomatous ameloblastomas, 7 arose from the mandible, whereas 3 originated from the maxilla. Bone invasion was observed for 3 acanthomatous ameloblastomas.
Immunohistochemical Profiling
Immunoreactivity was detected in the appropriate location in suitable positive controls using all antibodies and was not detected in negative controls. Results of immunoprofiling of COPSCCs, conventional SCCs, and canine acanthomatous ameloblastomas are detailed in Table 2. In all COPSCCs, neoplastic epithelial cells were positive for the high-molecular-weight cytokeratin marker 34βE12 and the pancytokeratin markers MNF116 and AE1/AE3. All COPSCCs were also immunoreactive for p63, cytokeratin 5/6, cytokeratin 14, cytokeratin 19, CDX-2, and p53). In addition, 9 of 25 COPSCC were positive for calretinin. Neoplastic epithelial cells of COPSCC were negative for cytokeratin 7, CP27, amelogenin, villin, collagen IV, vimentin, and laminin. There were no differences in immunohistochemical findings in samples taken from the different parts of the neoplasms.
Immunohistochemical Profiling of Canine Oral Papillary Squamous Cell Carcinoma (COPSCC) and Comparison to That of Conventional Squamous Cell Carcinoma (CSCC) and Canine Acanthomatous Ameloblastoma (CAA).
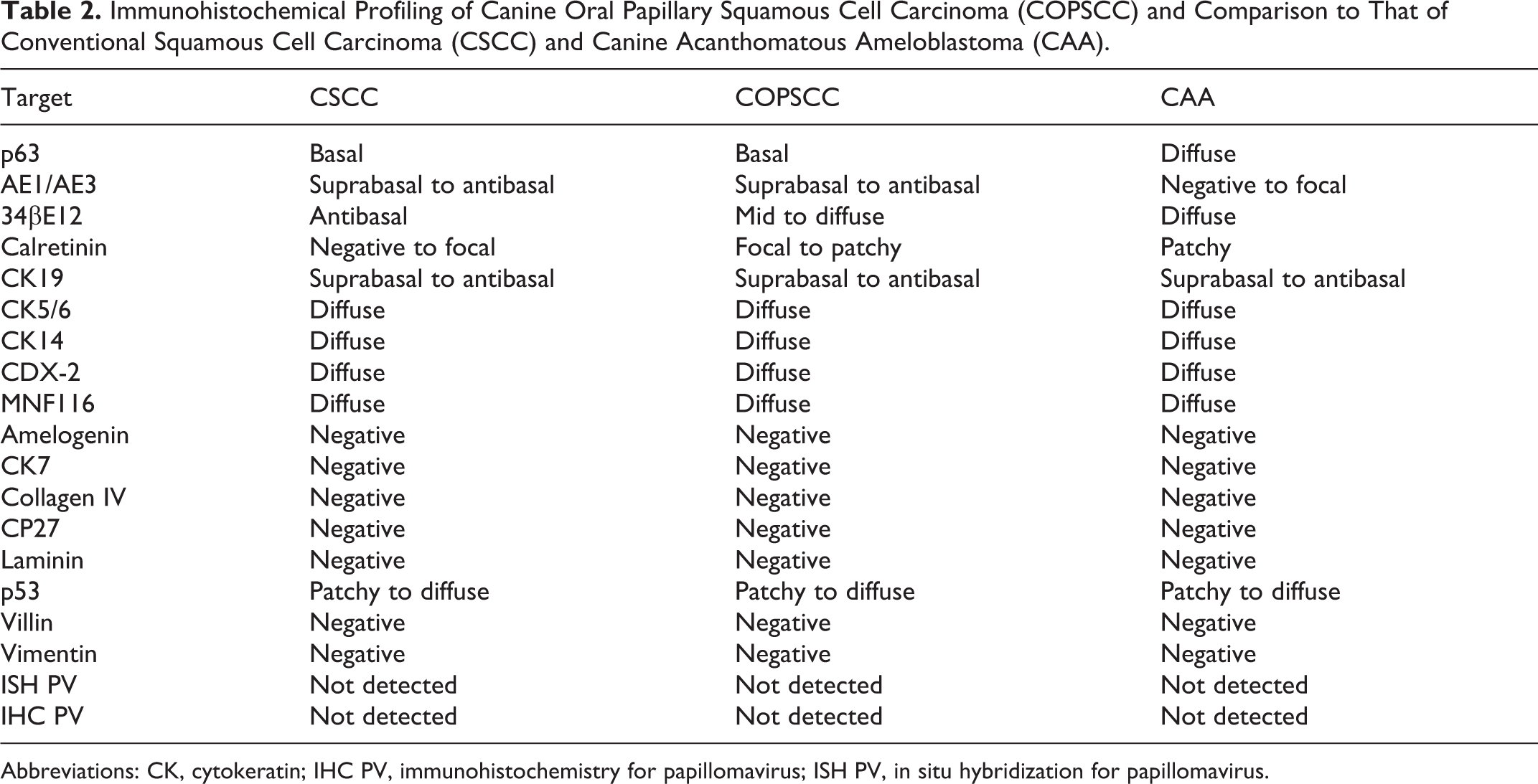
Abbreviations: CK, cytokeratin; IHC PV, immunohistochemistry for papillomavirus; ISH PV, in situ hybridization for papillomavirus.
Immunohistochemical profiling of p63, AE1/AE3, 34βE12, and calretinin discriminated among COPSCC, conventional SCC, and canine acanthomatous ameloblastoma. IHC with the high-molecular-weight cytokeratin antibody 34βE12 yielded strong perimembranous and cytoplasmic labeling within suprabasal to antibasal layers of epithelium in conventional SCC (Fig. 1). COPSCC had an intermediate labeling pattern between the conventional SCC and acanthomatous ameloblastoma, characterized by strong diffuse labeling throughout the epithelium that was less intense within the basal layer (Fig. 2). In contrast, canine acanthomatous ameloblastoma had uniformly strong and diffuse labeling throughout the neoplastic odontogenic epithelium (Fig. 3).
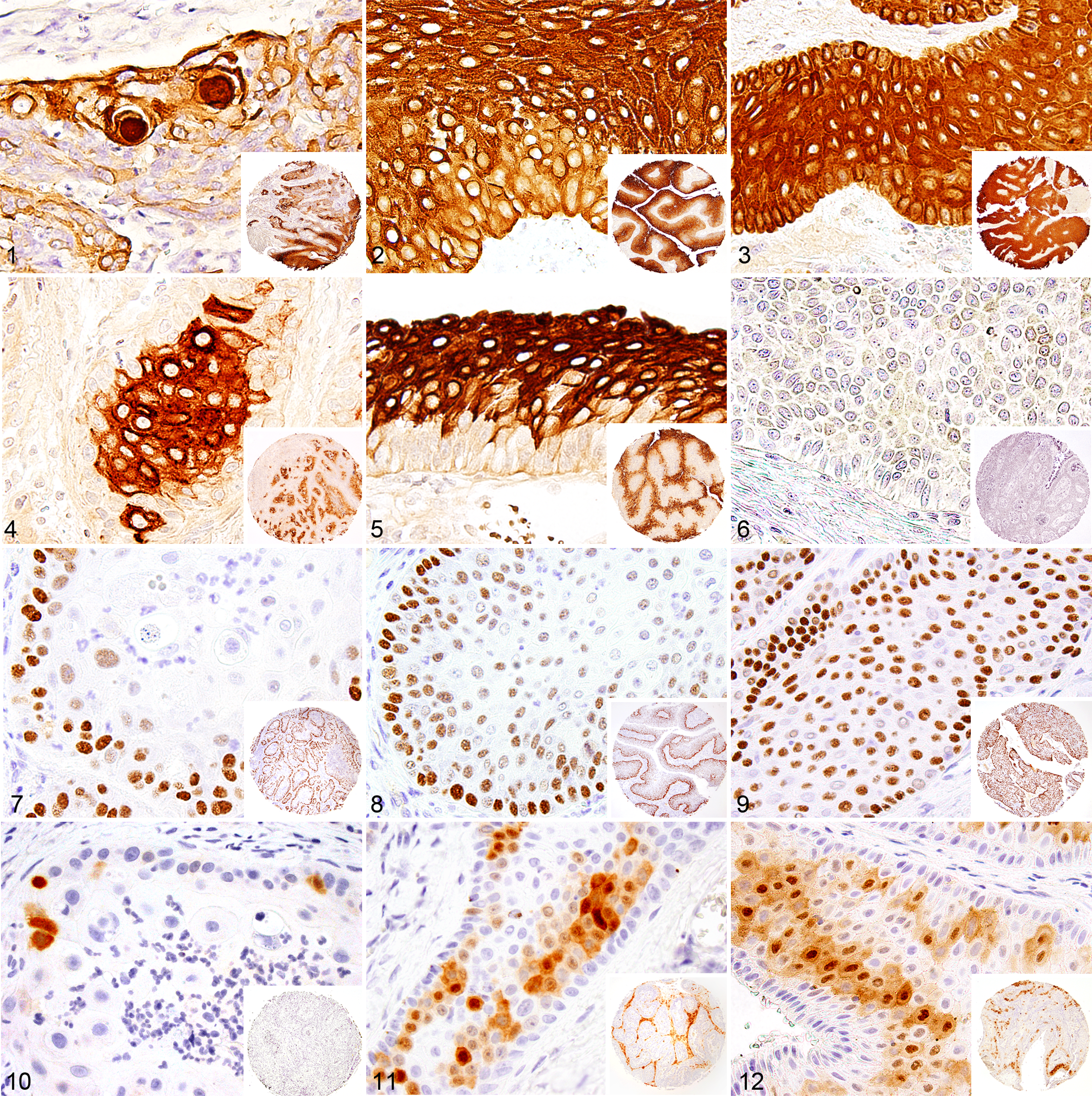
Immunolabeling for pancytokeratin AE1/AE3 in conventional SCC was strongly positive in the cytoplasm and along the membrane of all neoplastic cells except those of the basal layer (Fig. 4). COPSCC had a similar labeling pattern to conventional SCC with positive labeling in the suprabasal to antibasal layers (Fig. 5). There was no or minimal labeling for AE1/AE3 in acanthomatous ameloblastoma (Fig. 6).
There was strong nuclear labeling for p63 in conventional SCC within the basal layers and intensity of nuclear labeling gradually decreased in cells within the antibasal layers (Fig. 7). Most COPSCCs had immunoreactivity for p63 like that observed in conventional SCC, in that there was strong nuclear labeling in the basal layer and a gradual decreasing intensity of labeling within the antibasal layers (Fig. 8). There was uniformly diffuse, strong nuclear labeling of neoplastic odontogenic epithelium for p63 in acanthomatous ameloblastoma (Fig. 9). It is interesting that 2 cases of COPSCC that histologically resembled odontogenic epithelium along the base of their papillary fronds had diffuse, strong nuclear labeling for p63, which is similar to that seen in acanthomatous ameloblastoma.
Immunoreactivity for calretinin in conventional SCC was weak and limited to few cells within antibasal regions (Fig. 10). Two COPSCCs had strong nuclear and cytoplasmic labeling for calretinin; these both had more pronounced odontogenic epithelium-like morphology in the deep portions of the neoplasms (Fig. 11). There was mild to moderate cytoplasmic and nuclear immunoreactivity for calretinin in 7 other COPSCCs. In contrast, acanthomatous ameloblastomas had strong immunoreactivity in multifocal aggregates of neoplastic odontogenic epithelial cells (Fig. 12).
There was no significant difference in immunolabeling between COPSCC, acanthomatous ameloblastoma, and conventional SCC for MNF116, cytokeratin 5/6, cytokeratin 7, cytokeratin 14, cytokeratin 19, CP27, amelogenin, CDX-2, collagen IV, laminin, p53, villin, and vimentin (Table 2).
The mucosal epithelium in cases of gingival hyperplasia had a strong, diffuse immunolabeling for 34βE12 throughout all layers, which was similar to that of acanthomatous ameloblastomas. In contrast, COPSCCs had decreased immunolabeling for 34BE12 in the basal-most layer. Immunolabeling for AE1/AE3 and p63 in the hyperplastic gingival epithelium was similar to both COPSCCs and grade 2 conventional SCCs. The most discriminating antigen between hyperplastic gingival epithelium and COPSCC was calretinin: epithelial cells of the hyperplastic gingiva samples had weak nuclear labeling only within the basal layer, whereas 9 of 25 COPSCCs had moderate to strong cytoplasmic and nuclear immunoreactivity for calretinin focally or multifocally throughout all layers.
Neither immunoreactivity for papillomavirus antigen nor labeling for papillomavirus DNA was detected in COPSCC, conventional SCC, or canine acanthomatous ameloblastoma specimens using IHC and ISH, respectively.
Discussion
Based on our results, COPSCCs are histomorphologically and immunophenotypically distinct from conventional SCCs and canine acanthomatous ameloblastomas, but there are some similar features among these neoplasms. COPSCCs were immunoreactive for the 2 pancytokeratins (MNF116 and AE1/AE3), cytokeratin 5/6, cytokeratin 14, cytokeratin 19, high-molecular-weight cytokeratin, CDX-2, p53, calretinin, and p63 and had no immunoreactivity for cytokeratin 7, mesenchymal markers, amelogenin, laminin, villin, collagen IV, and CP27. This immunophenotype is similar to that reported for human papillary SCC, suggesting that COPSCC has a similar histogenesis to human papillary SCC. 9 Of the antibody targets evaluated in this study, 34βE12, p63, AE1/AE3, and calretinin were discriminatory between COPSCC, canine acanthomatous ameloblastoma, conventional SCC, and hyperplastic gingival epithelium.
Cytokeratins are intermediate filament proteins expressed by epithelial cells. Immunoreactivity for cytokeratins is conserved in a tissue-specific manner in normal organs and carcinomas. As such, the subtypes of cytokeratins expressed by neoplastic cells can be used for determining the primary histogenesis of neoplasms and their metastases. 5 In the current study, the immunohistochemical patterns of pancytokeratin AE1/AE3 and high-molecular-weight cytokeratins in the normal gingiva, COPSCC, and conventional SCC correlated with that of previously reported findings in dogs. 3,12 COPSCC exhibited immunoreactivity for the pancytokeratin marker, AE1/AE3, in the suprabasal to antibasal layers similar to the labeling pattern observed in conventional SCC as well as hyperplastic gingival epithelium, whereas acanthomatous ameloblastoma had no or minimal labeling. In contrast, COPSCC had strong diffuse immunoreactivity for the high-molecular-weight cytokeratin marker 34βE12 throughout the epithelium that was less intense within the basal layer. Comparatively, there was strong and diffuse labeling observed in the cytoplasm of all neoplastic cells of acanthomatous ameloblastoma and in hyperplastic gingival epithelium. Conventional SCC had only weak scattered immunoreactivity within suprabasal to antibasal layers of their epithelium. Therefore, positive labeling for AE1/AE3 discriminates COPSCC and conventional SCC from acanthomatous ameloblastoma, which is negative. COPSCC differs from conventional SCC by having strong diffuse immunoreactivity for the high-molecular-weight cytokeratin 34βE12 compared with the weak immunoreactivity within the superficial epithelial layers of conventional SCC. COPSCC and hyperplastic gingival epithelium had a similar immunophenotype, except in the immunoreactivity pattern for the high-molecular-weight cytokeratin marker 34βE12 and calretinin. Cytokeratin 34βE12 labeled less intense within the basal layers of the epithelium of COPSCC when compared with gingival epithelium. In addition, the hyperplastic gingiva had weak nuclear labeling for calretinin limited to the basal layer, whereas 9 of 25 COPSCCs had moderate to strong cytoplasmic and nuclear immunoreactivity randomly scattered in all layers.
The transcription factor p63, a homologue of the tumor suppressor p53, plays an important role in maintaining the progenitor basal cell populations in stratified epithelium. 6 As such, p63 is commonly used as a marker of basal and myoepithelial cells, but p63 overexpression has also been associated with keratinocyte proliferation and metastatic capacity. 15,19 In our study, strong nuclear labeling for p63 in the basal layer of the stratified squamous epithelium and weaker immunoreactivity in antibasal layers of most COPSCCs was similar to the immunoreactivity for p63 observed in the grade 2 canine conventional SCC and normal gingival epithelium, as has been previously reported. 21 In contrast, nuclear expression of p63 has been reported in all layers of grade 3 canine conventional SCC. 21 In a previous study, the diffuse pattern of p63 immunolabeling was associated with more aggressive biological behavior. 21 Although no follow-up data were available for the majority of cases in the current study, COPSCC is generally considered to be a less aggressive neoplasm, which would explain a p63 labeling pattern similar to low-grade conventional SCCs. Moreover, 1 of 2 COPSCCs with odontogenic-like morphology had diffuse immunolabeling similar to canine acanthomatous ameloblastoma. It is interesting that diffuse expression of p63 has also been reported in all types of odontogenic lesions, as p63 is important in differentiation and proliferation of odontogenic epithelial cells. 4 It has further been shown that immunoreactivity for p63 tends to be more intense in epithelial odontogenic neoplasms than in normal tooth germ cells, suggesting that p63 might be involved in oncogenesis or malignant transformation of odontogenic epithelium. 17 Consistent with these reports, diffuse labeling of nuclei for p63 was observed in canine acanthomatous ameloblastoma.
Calretinin is a 29-kDa calcium-binding protein expressed by various types of epithelia as well as by mesothelium and retina. 8 Immunoreactivity for calretinin has been reported to be significantly more common in canine oral SCCs compared with acanthomatous ameloblastomas, and the differences in expression pattern and labeling intensity have been recommended as a method to distinguish these 2 neoplasms. 12 In contrast to the previous study that identified immunolabeling for calretinin in 10 of 15 oral SCCs, but only 2 of 15 canine acanthomatous ameloblastomas, the current study found that canine acanthomatous ameloblastoma had strong immunoreactivity in multifocal aggregates of neoplastic odontogenic epithelial cells, whereas oral conventional SCC had no to rare immunoreactivity for calretinin. Although our results are in strong contrast to this single previous study in dogs, our results correlate with multiple reports that studied conventional SCC and acanthomatous ameloblastoma in humans and observed calretinin immunoreactivity exclusively in ameloblastomas. 1,2,8,12 This discrepancy may be due to differences in the epitope recognized by differing antibodies, other effects of using different antibodies for IHC, and/or differences in the retrieval conditions. Regardless, the observed immunoreactivity for calretinin in canine acanthomatous ameloblastoma is consistent with the hypothesis that these neoplasms arise from odontogenic epithelium or dental lamina. Conventional SCCs in contrast likely originate from gingival epithelium that lacks calretinin immunoreactivity. 12 In COPSCCs, there was variable immunoreactivity for calretinin; whereas many cases lacked immunoreactivity, seven cases had focal to patchy cytoplasmic and nuclear immunoreactivity and two cases had strong immunolabeling similar to that observed in canine acanthomatous ameloblastoma.
Whereas immunoreactivity for p63 and AE1/AE3 in COPSCC was similar to grade 2 conventional SCC, the immunoreactivity for 34βe12 and calretinin in COPSCC tended to resemble that of acanthomatous ameloblastoma. The overlap in immunophenotype of COPSCC with that of conventional SCC and acanthomatous ameloblastoma highlights the necessity of IHC panels rather than a single IHC marker to accurately differentiate these neoplasms, especially when dealing with small punch biopsies.
The origin of the precursor cell that gives rise to oral SCC is uncertain. Whether these neoplasms arise from cancer stem cells or dedifferentiate from mature keratinocytes is also unclear. 11 It has been suggested that oral SCCs in humans most likely arise from basal and suprabasal cells of the oral epithelium. 35 In contrast, proposed origins of canine acanthomatous ameloblastoma include epithelial nests representing remnants of dental lamina 22 or gingival epithelium, 13 or from within alveolar bone. 13 The exact histogenesis of COPSCC is also unclear. Given the differences in p63, AE1/AE3, 34βE12, and calretinin immunoreactivity between COPSCC, canine acanthomatous ameloblastoma, and conventional SCC, these lesions represent distinct neoplasms with different histogenesis and most likely different biological behavior. However, commonalities among COPSCC, canine acanthomatous ameloblastoma, and conventional SCC support the possibility that COPSCCs are derived from oral stem cells that can undergo multidirectional differentiation toward gingival and odontogenic epithelium. Alternatively, COPSCC may originate from junctional epithelium, a segment of gingival epithelium that attaches to the tooth surface like a collar and that is derived from both odontogenic epithelium and oral gingival epithelium. 24 Unfortunately, junctional epithelium has not been well characterized or immunophenotyped in dogs, thus precluding more definitive conclusions.
HPV has been detected in 50% of human patients with papillary SCCs 7,18 ; however, viral detection was attempted only when viral infection was suspected, and the role of HPV in papillary SCC oncogenesis remains unclear. In the current study, neither HPV antigen nor DNA was detected in COPSCC, conventional SCC, or canine acanthomatous ameloblastoma. Such results are consistent with prior reports and do not support papillomavirus playing a role in the development of such lesions in dogs. 26,28,32
COPSCCs can be misdiagnosed as benign squamous papillomas if only the superficial portion of the papillary mass is submitted for histopathologic examination. Since squamous papillomas rarely arise from the gingival epithelium, any papillomatous gingival mass in dogs should be considered as a potential COPSCC and a biopsy should include deeper tissue rather than only superficial portions of the mass. Conventional SCCs can be distinguished from COPSCCs by their invasiveness and only superficial cytokeratin 34βE12 immunolabeling. Lack of labeling or focal to patchy labeling for calretinin within the antibasal layers may be helpful to differentiate COPSCC from hyperplastic gingiva, which has predominantly basal labeling. In addition, fading of cytokeratin 34βE12 immunoreactivity in the basal layers of COPSCC will also help to differentiate these lesions from hyperplastic gingival epithelium, which has strong diffuse labeling throughout all layers. In some instances, COPSCC can mimic canine acanthomatous ameloblastoma when the basal cells in the invasive component of COPSCC have prominent intercellular bridges and antibasal nuclear arrangement. Immunolabeling for p63 along the basal layer of COPCC and suprabasal to antibasal immunoreactivity for AE1/AE3 are useful to differentiate COPSCC from canine acanthomatous ameloblastoma. As there is significant overlap among the different immunohistochemical markers, a panel of cytokeratin 34βE12, pancytokeratin AE1/AE3, p63, and calretinin may be required for an accurate diagnosis. Irrespective of these markers, the importance of identifying invasion of neoplastic cells into the subjacent stroma to accurately diagnose COPSCC cannot be overstated.
In conclusion, our study confirms that COPSCC represents a distinct neoplasm that exhibits both similarities and differences in histomorphology and immunophenotype compared with conventional SCC and canine acanthomatous ameloblastoma. Consistent with prior reports, the immunophenotyping of conventional SCC and canine acanthomatous ameloblastoma supports the theory that they arise from gingival epithelium and odontogenic epithelium, respectively. We speculate that COPSCCs originate from oral stem cells that can undergo multidirectional differentiation or arise from junctional epithelium that originates from both gingival and odontogenic epithelium. Regardless of the histogenesis, using an immunohistochemical panel of p63, AE1/AE3, 34βE12, and calretinin allows accurate differentiation of COPSCC from conventional oral SCC and canine acanthomatous ameloblastoma.
Footnotes
Acknowledgements
We would like to thank the histopathology section at the Michigan State University Veterinary Diagnostic Laboratory for assistance with generation of the tissue microarrays and immunohistochemical staining and the 2013 Merial-NIH Veterinary Scholars Summer Research Program, College of Veterinary Medicine, Michigan State University for funding the summer research training of Alexandra Collins-Webb.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
