Abstract
Fifty years ago, bacteria in the genus Brucella were known to cause infertility and reproductive losses. At that time, the genus was considered to contain only 3 species: Brucella abortus, Brucella melitensis, and Brucella suis. Since the early 1960s, at least 7 new species have been identified as belonging to the Brucella genus (Brucella canis, Brucella ceti, Brucella inopinata, Brucella microti, Brucella neotomae, Brucella ovis, and Brucella pinnipedialis) with several additional new species under consideration for inclusion. Although molecular studies have found such high homology that some authors have proposed that all Brucella are actually 1 species, the epidemiologic and diagnostic benefits for separating the genus based on phenotypic characteristics are more compelling. Although pathogenic Brucella spp have preferred reservoir hosts, their ability to infect numerous mammalian hosts has been increasingly documented. The maintenance of infection in new reservoir hosts, such as wildlife, has become an issue for both public health and animal health regulatory personnel. Since the 1960s, new information on how Brucella enters host cells and modifies their intracellular environment has been gained. Although the pathogenesis and histologic lesions of B. abortus, B. melitensis, and B. suis in their preferred hosts have not changed, additional knowledge on the pathology of these brucellae in new hosts, or of new species of Brucella in their preferred hosts, has been obtained. To this day, brucellosis remains a significant human zoonosis that is emerging or reemerging in many parts of the world.
In 1966, the first issue of Pathologia Veterinaria—now known as Veterinary Pathology—published an article on the characteristics of Brucella granulomas by K. L. Jacob, and the third issue included a manuscript by B. I. Osburn and P. C. Kennedy describing pathologic and immunologic responses of fetal lambs after infection with Brucella ovis. As the journal embarks on its second half-century of publication, it seems appropriate to assess the advancement of knowledge on this topic since these articles were published in the first issues of the journal.
Fifty years ago, bacteria in the genus Brucella were known to cause infertility and reproductive losses with predilection for causing placentitis, fetal pneumonia, and mastitis. At that time, the genus contained only Brucella abortus, Brucella melitensis, and Brucella suis, the 3 classic species of brucellae, and these were generally thought to be somewhat host-specific pathogens of cattle, sheep and goats, and swine, respectively. They were also known to be zoonotic, with the capability for causing clinical disease in humans. At that time, taxonomy placed the 3 classic species in the family Brucellaceae, with other bacterial genera such as Bordetella, Pasteurella and so on. Although scientific and practical knowledge of brucellosis has increased dramatically, in many countries, brucellosis continues to be a continuing or reemerging zoonosis, causing significant economic losses, not only from costs associated with clinical treatment and lost productivity in human infections, but also by economic costs associated with reproductive losses in livestock.
Although a number of studies demonstrated that Brucella could be recovered for a period of time from environments associated with infected animals, knowledge at that time indicated that maintenance in a susceptible host was critical for disease transmission. The literature of 50 years ago would suggest that environmental persistence was of little epidemiologic importance. Data gathered since that time have supported the requirement for maintenance in susceptible hosts.
Addition of New Species to the Brucella Genus
Since the early 1960s, at least 7 new species have been identified as belonging to the Brucella genus. The first new Brucella species added in the late 1960s was Brucella ovis. Isolated in 1952 from a ram in New Zealand, B. ovis was believed to be the cause of epididymitis, ewe abortion, and lamb neonatal mortality. 80,115 It took 18 years after the first report before B. ovis was recognized as a member of the genus. 81 The controversy regarding its inclusion was partly due to the fact that it differed from the existing criteria for recognition of brucellae and because the manifestations of infection in sheep did not fit the classic disease pattern associated with brucellosis. 37 Also, B. ovis was a rough strain, lacking expression of the O side chain on its lipopolysaccharide (LPS). At that time, it was believed that only smooth strains, such as the classic species, which expressed the LPS O side chain, were virulent and could maintain themselves within populations of reservoir hosts. 37
Beginning in 1966, an increased incidence of abortion, epididymitis, and reproductive failure was reported in dogs in the United States primarily from large commercial breeding kennels. 26 A Gram-negative bacterium, similar to members of the genus Brucella, was isolated from placental and fetal tissues of aborted pups. Although first thought to be a biotype of B. suis because of its biochemical properties, the bacterium was later named Brucella canis. 25,84 Like B. ovis, B. canis is a rough strain that lacks expression of the O side chain on its LPS.
One of the least controversial of the new strains was Brucella neotomae. This strain was first isolated in 1957 from the desert wood rat (Neotoma lepida), 116 a rodent that inhabits the western regions of the United States. It was readily accepted as a member of the genus due to its smooth colony morphology and because it fit criteria used to identify Brucella organisms. 66 Only about 25 cultures of B. neotomae have been isolated, of which none have been recovered from domestic livestock or humans. 5
In 1994, it was reported that a Gram-negative bacterium isolated from an aborted bottlenose dolphin fetus had metabolic, biochemical, and bacteriologic characteristics that placed it in the Brucella genus. 43 In following years, numerous isolates of marine mammal brucellae have been recovered from the Mysticeti and Odontoceti suborders of cetaceans. Further studies have led to taxonomic classification of these isolates into Brucella ceti (predominantly from porpoises and dolphins) and Brucella pinnipedialis (predominantly from seals) strains, each of which has several subgroups. 46 Bacteriologic studies have indicated that these strains are phenotypically smooth, like the classic Brucella strains. Seropositive responses to marine brucellae have been observed in 53 cetaceans, with isolation or identification of the bacteria having occurred in samples from 18 marine mammal species. 59 It is suspected that cetacean brucellosis may be distributed worldwide in the oceans. The ST27 genotype in Pacific B. ceti and B. pinnipedialis strains, characterized by a specific genetic location for the mobile genetic element IS711, has been associated with zoonotic infections in Peru and New Zealand. 31
Common voles (Microtus arvalis) were found to be a host for another new species, Brucella microti, which has also been recovered from wild red foxes (Vulpes vulpes). 60,111 Data suggest potential long-time survival of B. microti in soil, as this species was isolated from soil samples after storage for 6 months at 4°C. 112 Prolonged survivability in soil suggests B. microti may have a reservoir outside mammalian hosts. Experimental studies show close similarity of this Brucella sp to B. suis. To date, there is no evidence of domestic animal or human infection with B. microti. 93
Another new strain, Brucella inopinata, was isolated from an infected human breast implant. 113 Phylogenetic studies most closely link this strain to B. ovis. 93 Additional isolates recovered from wild Australian rodents and from a human patient with chronic destructive pneumonia have been proposed to be novel lineages of B. inopinata. 120,121
Several other bacterial isolates have been identified that may eventually be classified in the Brucella genus. These include 2 atypical Brucella strains isolated in 2008 from 2 foxes from eastern Austria that had oxidative, bacteriologic, and molecular characteristics that placed them in the Brucella genus, but data indicated they represented a novel species separate from other Brucella, including B. microti. 61 Other potential new strains, with characteristics typical of Brucella, have been recently recovered from 2 stillborn baboons (Papio spp) 109 and African bullfrogs (Pyxicephalus edulis). 40
Molecular Characterization of the Brucella Genus
Molecular studies have found that most Brucella strains have 2 circular chromosomes encoding approximately 3.2 kb with 2 replicons. Bacteriophages have been isolated from Brucella strains, 32 but plasmids have not. The extent to which Brucella are able to undergo exchange among themselves or with other bacteria is unknown. It has been proposed that intracellular niches inhabited by Brucella during residence in the host limit their opportunities for genetic exchange with other bacteria. It should be emphasized that Brucella infections are polyclonal rather than clonal infections, and clinical infection is generally associated with multiple bacteria crossing mucosal barriers and colonizing tissues under in vivo conditions. Cumulative data suggest dosages of virulent Brucella species required for 50% infection across mucosal surfaces of hosts other than guinea pigs or mice are most likely in the range of 10 3 to 10 4 colony-forming units (CFU). 78,119
Development of new molecular techniques, such as DNA-DNA hybridization, G+C base ratio determinations, and DNA–ribosomal RNA reciprocal hybridizations, demonstrated that the genus Brucella was more closely related to plant pathogens in the Agrobacterium-Rhizobium complex of organisms. 24 Studies have found such high homology within the genus that some authors have proposed that all Brucella were actually 1 species. 62,123 On average, the genome sequences of 6 species of Brucella (B. abortus, B. suis, B. melitensis, B. ovis, B. canis, and B. neotomae) display an average of >94% identity at the nucleotide level, 29,124 with B. abortus and B. melitensis being the most closely related. A close relationship has also been detected between B. canis and B. suis, whereas B. neotomae and B. ovis demonstrate greater divergence levels from other Brucella species. With the exception of biovar 5, B. suis isolates cluster together. 126
Although genomic techniques have suggested that Brucella strains may comprise a single genetic “species,” the epidemiologic and diagnostic benefits obtained by assigning Brucella strains to separate “nomenspecies“ based on their distinctive phenotypic characteristics are more compelling than data gained through genomic analysis. Information gathered over the years has demonstrated that most pathogenic species are capable of infecting numerous mammalian hosts even though most species of Brucella have preferred reservoir hosts. However, over the past 50 years, the epidemiologic importance of virulent Brucella species in hosts other than the “traditional” preferred host has increased. For example, cattle or camels in Central Asia or the Middle East have become established as reservoir hosts for B. melitensis with zoonotic implications, and increasing isolations of B. suis in cattle in the southeastern United States have public health significance due to the shedding of high numbers of bacteria within milk of infected cattle.
Impact of Brucellosis in Humans
Brucellosis remains one of the most important zoonotic diseases worldwide and is reemerging in some countries. The highest prevalence of human disease is currently found in areas of Africa, Asia, Latin America, and the Middle East. Mortality is uncommon; symptoms include fever, night sweats, anorexia, polyarthritis, meningitis, and pneumonia. Endocarditis, primarily associated with B. melitensis infection, is the principal cause of human mortality. The most common manifestations of localized disease are osteoarticular (ie, peripheral arthritis, sacroiliitis, spondylitis). 10 Incubation periods can be long (up to 6 months), and symptoms may persist for years in the absence of treatment. Brucellosis in humans almost always originates from an animal reservoir. 55 Infection most commonly occurs through consumption of nonpasteurized dairy products from Brucella-infected animals but can also occur by direct contact with infected animals or tissues or fluids associated with abortion. Human infection occurs across mucosal surfaces: by aerosolization into respiratory tissues, by oral consumption, or by penetration through breaks in the epidermis. Inadvertent exposure to live vaccine strains, most commonly via needle sticks, has also been a frequent source of human infections, especially in the veterinary profession. 15 While rare, human-to-human dissemination of brucellosis through breast milk or venereal transmission has been reported. 27,81
More than half a million cases are reported worldwide, although due to the wide-ranging and nonspecific nature of clinical signs, the true number may be much higher. 55 In endemic countries, prevalence rates exceed 10 per 100 000. Multiple studies have demonstrated that addressing brucellosis in animal reservoirs is the most cost-efficient mechanism for controlling human brucellosis. 16,71,100 As such, most control strategies involve control of livestock brucellosis through vaccination and use of test and removal strategies. Livestock vaccination has proved to successfully decrease human brucellosis and can be especially helpful in developing nations. One example is illustrated in the vaccination of more than 33 million sheep and goats over an 11-year period in Mongolia. 73 The incidence rate of human brucellosis declined from 4.8 per 10 000 to 0.23 per 100 000. Concurrently, abortions in small ruminants dropped to 20% of prevaccination levels.
Although still considered uncommon, increasing numbers of reports over the past decade have documented human infection with B. canis causing endocarditis, peritonitis, and other clinical symptoms. 52,128 The most common routes of transmission to humans are through contact with infected dogs or their secretions, fetal membranes, or aborted puppies. Diagnosis of B. canis infection in humans is likely underdiagnosed due to a low index of suspicion, nonspecific symptoms (eg, fever and fatigue), and lack of cross-reaction on common serologic tests using smooth Brucella antigens. 88 Human zoonosis with B. canis appears to be an emerging problem primarily in people of low socioeconomic status living in urban slums where there are high numbers of free-roaming dogs. 79 Epidemiologic investigations of a human B. canis infection in a slum environment in Argentina revealed 29 of 97 (29.9%) neighborhood dogs and 13 of 69 (18.6%) neighbors of the index case were serologically positive for B. canis. 79 Likewise, a serologic survey in Turkey documented the highest prevalence (7.8%) in patients from an urban area where stray dogs outnumbered pet dogs. 107 Some reports suggest there may be a link between B. canis infection and patients with immunosuppressive, inflammatory, or metabolic disorders, such as endocarditis, Gaucher disease, human immunodeficiency virus (HIV), and Guillain-Barré syndrome. 76,79,128
Currently in the United States, brucellosis is predominantly a disease associated with international travel or as a food-borne disease caused by ingestion of nonpasteurized dairy products originating from Mexico. 39,122
Role of Wildlife Reservoirs
Fifty years ago, only domestic livestock were considered of significant epidemiologic importance with regard to public health or regulatory programs. In the United States, although the spillover of B. abortus into elk (Cervus elaphus) and plains bison (Bison bison) in the Greater Yellowstone Area (Yellowstone National Park and surrounding areas) had been identified decades previously, 106 brucellosis in these hosts was not considered of epidemiologic importance partly due to the high prevalence of disease in domestic livestock. In subarctic areas, the prevalence of B. suis biovar 4 in reindeer and caribou (Rangifer tarandus) was known and suspected to be of public health importance. 64 Serologic surveys had identified brucellosis in African wildlife, 38,104,108 but the epidemiologic significance of disease in these reservoir hosts and the significance to public health and livestock industries were unknown. In Europe, B. abortus had been detected by culture in Swiss chamois (Rupicapra rupicapra) 18 and B. suis by culture or serology in red foxes in Russia, Bulgaria, and Wales. 101 In general, it may be concluded that 50 years ago, the long-term health impacts of brucellosis on these wildlife populations were thought to be limited, and their role in transmission of disease to humans or domestic livestock was considered minimal.
Since that time, numerous wildlife reservoirs of both classic and new species of Brucella have been identified. As brucellosis has been eradicated from domestic livestock in the United States and other countries, persistence in wildlife reservoirs has become of greater concern. This is demonstrated by the controversy regarding B. abortus in elk and bison in the Greater Yellowstone Area and the capability of these species to transmit brucellosis to cattle under field and/or experimental conditions. B. melitensis has been isolated from wild bovids such as ibex (Capra ibex) and chamois in the European Union 51 and from Nile catfish (Clarias gariepinus). 41 The infection of B. suis in wild swine populations in the United States, Europe, Australia, and other countries not only has led to zoonotic infections but has caused regulatory issues by inducing positive responses on serologic tests when B. suis is transmitted to domestic livestock. The infection of wild boar (Sus scrofa) and European brown hares (Lepus europaeus) in Europe with B. suis biovar 2 has also confounded diagnostics due to positive serology when domestic livestock, particularly swine, are infected.
Although Brucella infections in wildlife reservoirs may have minimal impact on the viability of free-ranging populations, data demonstrating disease transmission to domestic livestock and humans under field conditions convey their epidemiologic importance. The difficulties in controlling disease in free-ranging wildlife make the problem very complex. In addition, spillover of Brucella species into other hosts has occurred under field and experimental conditions. For example, B. abortus infection led to high mortality in bighorn sheep (Ovis canadensis) after inadvertent transmission from aborting elk. 74 Wild red foxes have been found to be reservoirs for both B. suis biovar 2 and B. microti most likely due to eating diseased hares and rodents, respectively. Historical experiences suggest that spillover of disease to other hosts could become important and should not be dismissed as insignificant.
Knowledge on Cellular Entry and Intracellular Activities of Brucella
Fifty years ago, it was known that Brucella penetrate the mucosal epithelium and are transported as free bacteria, or within phagocytic cells, to regional lymph nodes. Localization within regional lymph nodes results in lymph node hypertrophy, lymphatic and reticuloendothelial hyperplasia, and inflammation. If bacteria are not localized and killed within regional lymph nodes draining the site of infection, they replicate and spread via blood or lymph to other lymphoreticular tissues and organs such as the spleen, reproductive tract, and/or mammary gland. It was also known that the bacteremia associated with most Brucella species is short, and live bacteria are not readily isolated from blood samples.
Cellular Entry
Over time, a great deal of knowledge has been gained on the interactions of Brucella with host phagocytic cells. Entry of nonopsonized smooth Brucella relies on the cytoskeleton of the host cell for internalization. Smooth strains of Brucella interact with cholesterol-rich microdomains (lipid rafts) within the plasma membrane that facilitate contact with the host cell and mediate internalization into phagocytic cells. Lipid rafts contain glycosphingolipids, cholesterol, and glycosyl-phosphatidylinositol anchored proteins 34 and facilitate membrane-associated sorting events, such as the formation of multi-subunit membrane complexes and signaling across membranes and membrane fusion. Besides the plasma membrane, lipid rafts are also found in intracellular organelles and vesicles. The Brucella LPS O-polysaccharide appears to be a key molecule for interaction with lipid rafts on host cells but also prevents complement-mediated bacterial lysis and host cell apoptosis. 9 Opsonization of smooth strains of Brucella increases entry 10-fold and occurs through IgG (Fc) and complement (C3b and 4b) receptors on the surface of phagocytes, which diverts smooth bacteria from lipid rafts and targets entry to the phagolysosomal compartment. Receptor-mediated phagocytosis leads to greater killing of internalized Brucella by monocytes.
Entry of rough strains of Brucella differs from smooth strains as rough strains are unable to sustain interactions with lipid rafts and are readily phagocytosed following either Toll-like receptor 4 (TLR4) or mannose receptor recognition of the LPS-deficient bacterial surface. As a result, rough strains demonstrate elevated invasion possibly due to exposure of ligands that are normally hidden by the O side chain and may have increased capability to adhere to macrophages. 62 Consequently, nonopsonized rough Brucella are internalized as efficiently as opsonized Brucella, are rapidly targeted to the phagolysosomal compartment, and are generally unable to replicate. 103 Entry of smooth and rough Brucella strains into the cells through different pathways may also involve receptors with a distinct ability to regulate the level of phagocytosis. In contrast to rough strains that are defective at intracellular replication, the intracellular smooth Brucella that survive after opsonin-mediated phagocytosis are capable of significant intracellular replication. 14 Genes necessary for O side chain synthesis (ie, manB, wboA) play a significant role in establishing the intracellular replicative compartment for smooth strains of Brucella.
Intracellular Trafficking and Survival
Smooth Brucella that enter via lipid rafts quickly traffic through the early endosomal compartment and depart the phagosome to form the modified phagosome (termed brucellosome) by acquiring components of endoplasmic reticulum in a manner similar to autophagosome biogenesis. Brucella initially localize within acidified phagosomes, 102 where they are exposed to free oxygen radicals generated by the respiratory burst. Brucella require acidification of the phagosomal compartment to a pH <4.5 before they display wild-type intracellular replication. The requirement for low pH is transient and extends only through the initial stages of intracellular infection. Localization in an acidified environment induces expression of the VirB operon (virB 1–10), which controls expression of genes associated with a type IV secretion system. The VirB operon interacts with the endoplasmic reticulum to neutralize the pH of the phagosome. 28 The Brucella-induced modifications of the phagosome prevent fusion with the lysosome. The brucellosome environment provides Brucella with conditions of nutrient depletion and limited oxygen availability. It should be noted that under in vitro conditions, up to 90% of virulent Brucella and 99% of nonvirulent Brucella may be killed following intracellular entry. 21,98
Another mechanism used by Brucella for intracellular survival involves modification of the lipid content of the phagosome limiting membrane. Virulent Brucella strains express a cyclic glucan synthase (cgs) that produces and secretes low-molecular-weight cyclic glucans. These molecules disrupt the lipid raft microdomain structures within intracellular membranes surrounding the bacteria. This modification of lipid raft distribution in phagosomal membranes inhibits phagosome maturation, prevents fusion with lysosomes, and is independent from VirB and type IV secretion systems. 8
Greater understanding of the mechanisms that Brucella use to survive under intracellular conditions continues to occur. Since oxidative killing is the primary mechanism employed by host phagocytes to control replication of intracellular pathogens, it has been found that Brucella have multiple mechanisms to detoxify free radicals. Brucella expresses 2 superoxide dismutases, SodA and SodC, with SodC believed to be most important for detoxification of superoxide anions generated by the respiratory burst of phagocytes. The Brucella gene ahpC (alkyl hydroperoxide reductase C) may also protect against oxidative killing; in other bacteria, it protects against low levels of hydrogen peroxide and can detoxify the oxidizing compound peroxynitrite (ONOO–). The dps gene, which plays a critical role in stationary-phase resistance to oxidative killing and acidic pH in other bacteria, has also been identified in Brucella. The O side chain also appears to protect against cellular cationic peptides and oxygen metabolites.
Brucella probably use both stationary and exponential stages for intracellular survival, with stationary-phase physiology providing Brucella with benefits for adapting to the harsh conditions encountered in the phagosome and exponential stages associated with replication under favorable conditions. Molecular mechanisms may regulate adaptation. For example, VirB has been found to be maximally expressed during exponential growth, but expression is repressed upon entry into the stationary phase. 105 Like other intracellular pathogens, Brucella have adapted to their intracellular lifestyle and no longer require the accumulation of energy-storing molecules. 29 The presence of cytochromes (cytochrome bc1 complex or quinol oxidase) with a high oxygen affinity may represent an important adaptation of Brucella to their intracellular survival. Several Brucella have been shown to use heme as an iron source in vitro and have a critical need for heme during residence in the phagosomal compartment and during replication in trophoblasts. 34 Macrophages, the preferred host cell for Brucella, are important for heme recycling in mammals. Brucella scavenge iron through siderophores such as 2,3 dihydoroxybenzoic acid or brucebactin, 14 which are regulated by iron response regulators and AraC-like transcriptional activators. 7 Mutation of iron regulatory genes has been demonstrated to attenuate virulent Brucella strains in mammalian hosts and make Brucella more sensitive to oxidative killing.
Avoidance of Innate Immunity
In accordance with its stealthy nature, Brucella has developed mechanisms to minimize stimulation of pattern recognition receptors (PRRs) by the innate immune system of the host. The Brucella cell envelope has high hydrophobicity and its LPS has a noncanonical structure that elicits a reduced and delayed inflammatory response compared with other Gram-negative bacteria 102 and has lower stimulatory activity on TLR4 receptors. 103 The O side chain on the LPS can form complexes with the major histocompatibility complex class II molecules that interfere with the ability of macrophages to present exogenous proteins. Brucella ornithine-containing lipids and lipoproteins in the outer membrane are poor activators of innate immunity. Brucella bacteria are also devoid of many classic structures involved in virulence such as pili, fimbriae, capsules, and plasmids that stimulate PRRs. The ability of Brucella to prevent phagosome maturation and fusion with lysosomes may interfere with other innate and adaptive immune processes. As proteins have been identified in Brucella that demonstrate significant homology with TLR adaptor molecules, these peptides may be a mechanism to interfere with or subvert TLR signaling. 90 In addition, the ability of smooth Brucella strains, but not rough strains, to inhibit macrophage apoptosis is believed to enhance bacterial survival in the host by maintaining the favorable environment of the phagocyte, preventing release of the bacteria into the extracellular environment, where it exhibits reduced replication, and contributing to avoidance of antibody and complement inactivation. 95 Compared with other Gram-negative bacteria, Brucella induces a reduced innate immune response and a lower rate of maturation and activation of dendritic cells, which may impair development of adaptive immune responses.
Although it was known that latent infections of Brucella could occur, the mechanisms for long-term in vivo survival without immune recognition remain unknown. It is known that Brucella may reside in a dormant, nonreplicative state in phagocytic cells in calves infected with B. abortus at a young age, and recrudescence of clinical brucellosis is also a problem in human patients. The epidemiologic significance of latency with Brucella spp in other reservoir hosts has not been well characterized. Currently, the molecular mechanisms controlling recrudescence or host physiology influencing in vitro replication of Brucella remain unidentified in human and animal hosts. Although the significance of erythriol metabolism remains controversial, it has been hypothesized that the preference for Brucella to metabolize erythritol as an energy source, as well as the presence of erythritol within placental tissues, plays a role in the tropism of B. abortus for the pregnant reproductive tract. 33 However, this postulated relationship between erythritol metabolism and Brucella virulence in ruminants has not been directly tested under experimental conditions.
Pathogenesis of Brucella Species in Preferred Hosts
Brucella abortus
First described by veterinarian Bernard Bang 11 as early as 1906, B. abortus remains an important zoonotic disease in ruminants, causing economic losses due to fetal wastage and infertility.
Regardless of the route of infection, adherence to mucosal epithelium is a requisite. With the exception of intestinal mucosa, it is not entirely clear how Brucella, once adhered to mucosal epithelium, translocate across the epithelial barrier. Oral exposure to aborted fetuses or placental membranes and ingestion of contaminated milk by calves are considered primary routes of infection. Using ligated ileal loops, B. abortus S19 was seen inside ileal lymphoepithelial cells and inside submucosal lymphatics, suggesting mucosal translocation via lymphoepithelial cells or M cells. 1 As Brucella pass to draining lymph nodes, acute lymphadenitis results. In experimental infection studies, acute lymph node changes have been characterized as hyperplastic with either neutrophilic or eosinophilic lymphadenitis. 2 Chronic lymph node changes are characterized by granulomatous lymphadenitis with deep cortical and paracortical histiocytosis and germinal center expansion. 30,110,127 Other lymph node lesions described include multifocal hemorrhage and extramedullary hematopoiesis. 42
Brucella can be found in parotid lymph nodes and placenta of cattle as early as 2 days and 14 days, respectively, after conjunctival exposure. 42,94 Extension of disease from the regional lymph nodes is chiefly hematogenous. Bacteremia may last several months and, with time, ceases or becomes intermittent, recurring in 5% to 10% of animals. 127 Bacteremia is followed by seeding of infection in preferred sites such as gravid uterus, placenta, spleen, mammary gland, and lymph nodes. In males, testes and accessory sex glands (ie, epididymis, ampulla, seminal vesicles) are preferentially colonized. 19,42,110 Arthritis, tenosynovitis, and bursitis have also been reported. 110 Once infection is established in sexually mature animals, it often persists indefinitely. 110
Cattle, goats, and sheep have cotyledonary, villous, epitheliochorial, nondeciduate placentas, and there is similarity in the pathogenesis and morphology of placentitis in cattle infected with B. abortus and goats and sheep infected with B. melitensis. 6,26,82,83,85 That is, all are characterized by maternal bacteremia, massive intracellular replication of Brucella in chorioallantoic trophoblastic cells, peri- and interplacentomal exudate, necrosis, high numbers of Brucella in tissues and exudates, and abortion.
In cattle, the disease course is long, being several months from the time of placental infection to abortion, 108 and the development of disease does not progress at the same rate for all animals. 92 Late-term (ie, 7–9 months of gestation) abortion and birth of a nonviable calf are the most obvious and often the only clinical signs. Gross lesions of the placenta vary in severity and are similar to those of placentitis of other bacterial or fungal origin 108 but generally focus on periplacentomal and intercotyledonary placenta. Most commonly, there is a necrotizing placentitis with viscous, brown exudate that may contain blood, fibrin, and flocculent detritus. 85,108,125 Not all placentomes are equally affected, and some placentomes appear grossly normal. The post-abortion uterus may be characterized by diffuse suppurative metritis with greenish-hemorrhagic exudate covering the endometrium and caruncles. 6 Cows may present with hemorrhagic and suppurative vaginal discharge for several days after abortion. 6
Placental entry, localization, and proliferation of Brucella have been described in experimental infection studies of goats inoculated with B. abortus. 6 Using this model, it was shown that Brucella were first present in hematomas present at the junction of the maternal septal tip and chorionic villi base, a region containing erythrophagocytic trophoblastic cells. Infection extends to adjacent chorioallantoic trophoblastic cells. 6 Massive intracellular replication of Brucella in chorioallantoic trophoblastic cells precedes fetal infection. This occurs as Brucella-laden trophoblastic cells die and are shed into the uterine lumen. Trophoblastic cell death results in erosion and ulceration of the chorioallantoic membrane, allowing for widespread hematogenous dissemination of Brucella to other parts of the placenta and fetus. 6
Microscopically, there is superficial to deep caruncular necrosis and hemorrhage with neutrophilic and mononuclear exudates; however, a characteristic finding is the presence of numerous coccobacilli within both intact and desquamated trophoblastic epithelial cells and macrophages (Figs. 1, 2). Organisms are also found free in the exudates that fill the uterochorionic space. Maternal portions of the placenta are variably affected, ranging from superficial necrosis and multifocal neutrophilic and lymphohistiocytic endometritis to severe and extensive ulcerative endometritis (Fig. 3). 92 Once placentitis has progressed to the point of inevitable abortion, endometritis is diffuse, severe, and necrotizing, with endometrial mucosa replaced by inflammatory granulation tissue. 94,110 There may also be severe neutrophilic vasculitis within endometrium and caruncular peduncles. 125 Mastitis is characterized as multifocal, interstitial, and lymphosuppurative or lymphohistiocytic. 127
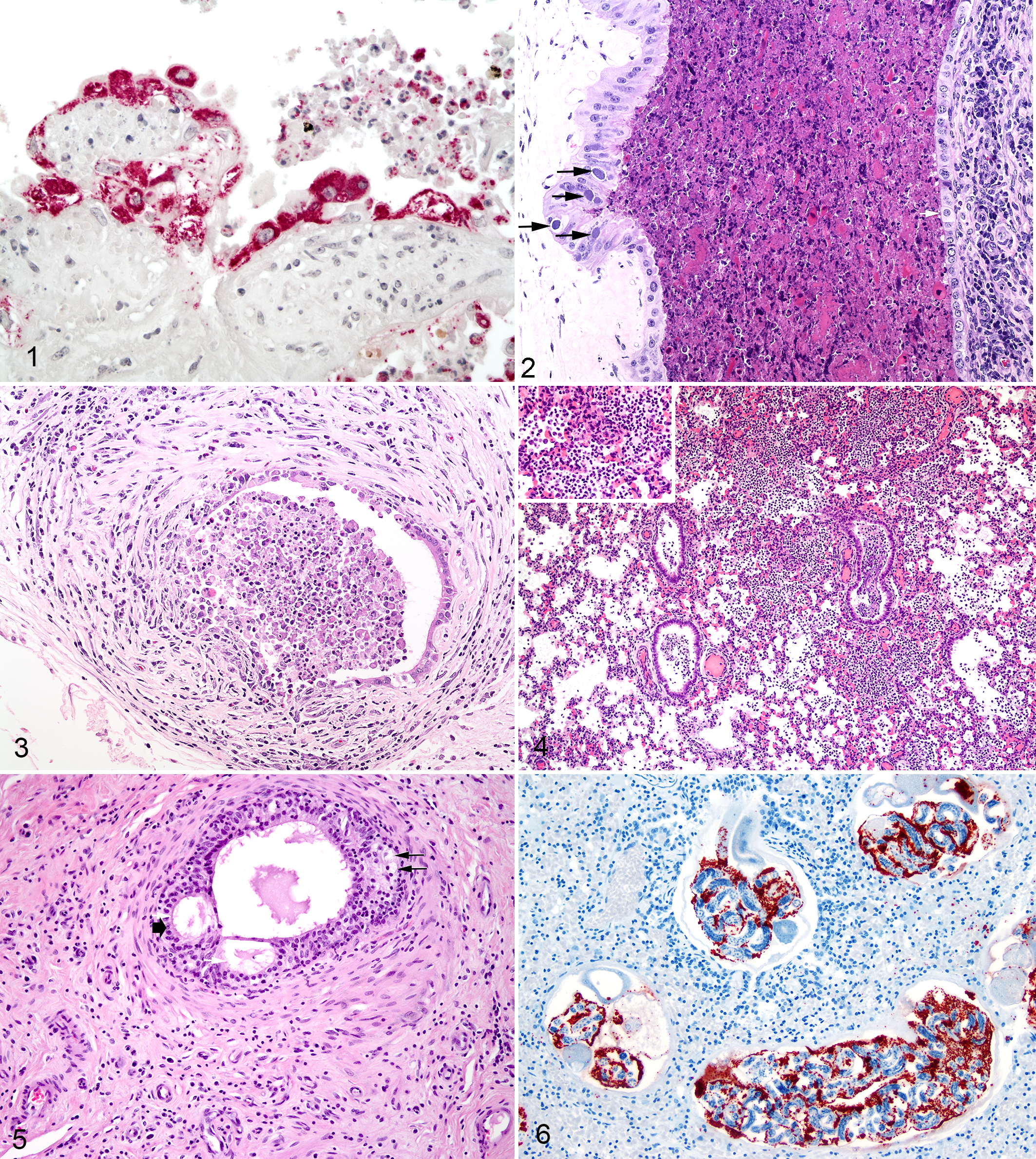
Brucellosis; placenta, cow.
It is believed that abortion is due to compromised fetal-maternal metabolic exchange. 94 Aborted fetuses may be autolytic. The most characteristic fetal lesions are those of bronchitis and bronchopneumonia (Fig. 4); however, experimental infection studies show fibrinous pleuritis, peritonitis, and pericarditis may also be seen. 127 The most commonly infected fetal organ is the spleen, thought to be infected as a result of bacteremia. 94
Swelling of the testis, within the confines of the tunica albuginea, results in changes ranging from testicular atrophy to vascular compromise and infarction. Gross lesions are generally multifocal, containing liquefied necrotic material. Lesions are often chronic, with fibrosis and dense adhesions between parietal and visceral tunics. Lesions may resemble abscesses surrounded by a fibrous capsule. Microscopically, the granulomatous to pyogranulomatous infiltrate is centered on tubules. Intracellular Brucella may be seen within macrophages. More chronic lesions exhibit degeneration and necrosis of the tubular epithelium, decreased mitotic activity, and decreased spermatogenesis. Lesions may extend along the epididymis and involve the seminal vesicles and ampullae.
Brucella melitensis
Widely considered the most virulent, this species has proved difficult to eliminate from domestic livestock. After more than a century, no major country has been able to eradicate the disease following its widespread establishment. 87 Pathogenesis and lesion development in goats infected with B. melitensis are similar to that of cattle infected with B. abortus. Goats are very susceptible to infection with B. melitensis and are the primary source of B. melitensis infection for humans. 97 Although sheep are also susceptible to B. melitensis, the disease is more variable and often self-limiting. 3
In goats, early infection is subclinical; however, bacteremia may produce severe clinical signs and even death in some animals. Abortion occurs in late gestation, with or without retention of fetal membranes, and these may be the only clinical signs. 3 The gross lesions of placentitis in the goat are similar to those seen in cattle aborting due to B. abortus infection. Entry, localization, and proliferation of Brucella in the goat placenta have been described in experimental infections of goats with B. abortus. 6
A common site of infection in goats and sheep is the mammary gland. Mastitis is a more common feature of caprine brucellosis compared with bovine brucellosis. The affected mammary gland may be characterized by multinodular firmness with watery, clotted milk. 34 Excretion of organisms in milk may be protracted in goats but less so in sheep. 3,97 Although late-gestation abortion is generally the rule, it can occur at other stages. Aborted fetuses often appear grossly normal; however, bronchopneumonia, hemorrhagic pleural fluid, and enlargement of lymph nodes, liver, and spleen are common. 3,97 In male goats, infection of the testis, epididymis, seminal vesicle, and deferent ducts can occur. Infection of male genitalia often results in decreased fertility.
Brucella suis
First described in 1914 from an aborted pig fetus in Indiana, this species has multiple biovars that vary in host specificity and phenotype. 4 Biovars 1, 2, and 3 infect primarily domestic and feral swine and wild boar. Biovar 1 is most often found in South America and Asia. Both biovars 1 and 3 have been reported in the United States, Australia, and China. 35 Biovar 2 is the most common strain in Europe. 50,118 Biovars 1, 2, and 4 can be transmitted from swine to cattle, inducing transient seroconversion, which can confound B. abortus diagnostic assays. 85 Biovar 2 differs from the other biovars. It was isolated from European hares (Lepus capensis) in Germany, Switzerland, France, and Denmark. 34 It was first thought to be a mutated B. abortus or B. melitensis since at that time, swine brucellosis did not exist in France and B. suis had not been isolated from domestic animals. We now know that biovar 2 can establish a reservoir of infection in hares and can be horizontally transmitted to swine and cattle. Although pathogenic in cattle and swine, biovar 2 is not zoonotic.
Abortion in swine infected with B. suis is much less common than abortion in cattle infected with B. abortus. However, abortion may be the only clinical sign observed in female swine infected with B. suis. Early fetal loss may go unnoticed and appear as infertility. 87,97 Abortion generally occurs during the second or third month of gestation, and stillborn or weak full-term piglets are common. 65,110 Placentas from aborting sows have been described as hyperemic and edematous, with yellowish purulent cheese-like miliary nodules. 35
Orchitis in boars is generally characterized by unilateral or bilateral enlargement that may be uneven and bulging. 65,72 In chronic cases, the affected testes may be small and atrophic. On cut surface, the interlobular septa are thickened, and the tubules may appear as sharply rounded, granular areas. 65 Abscesses of the head of the epididymis have also been described. 65 Arthritis with lameness and occasional posterior paralysis can be seen in both males and females. 35
Histologic lesions of orchitis vary in severity but are generally characterized by multifocal to coalescent degeneration and necrosis of seminiferous tubules. 65,72 In some cases, there may be extensive liquefactive necrosis with abscess formation. Intertubular tissues may be thickened with infiltrates of variable numbers of lymphocytes and macrophages. Langhans-type multinucleated giant cells may also be present. 65 Within the epididymis, there may be infiltrates of lymphocytes and macrophages with abscess formation. Azoospermia may be present.
In reindeer, B. suis biovar 4 causes abortions, stillbirths, weak calves, retained placenta, orchitis, epididymitis, metritis, mastitis, arthritis, hygromas, lymphadenitis, and nephritis. 44 However, experimental infection of cattle with biovar 4 results in no clinical signs of disease or lesion formation but can cause seropositive responses on standard brucellosis assays. 45
Brucella canis
First recognized in 1966, B. canis caused widespread abortions in Beagles. 22,61,121 Although B. abortus and B. melitensis can transiently infect dogs, only B. canis infection results in clinical disease. The host range of B. canis is limited as experimental infections of pregnant and nonpregnant cattle, sheep, and swine have been unable to induce clinical signs. 96 However, there is a high zoonotic risk for persons who handle breeding dogs in kennels or are exposed to infected animals. 77,84
Transmitted both venereally and orally, lesions of B. canis infection in males include epididymitis, prostatitis, and orchitis. In females, lesions include endometritis, placentitis, and late-term abortion. Infection can result in infertility in either males or females. Ocular and skeletal lesions (diskospondylitis) may occur but are less common. Clinical infection is usually afebrile, and signs are generally limited to spontaneous abortion or failure to conceive. 26 Some dogs remain asymptomatic despite active infection. 125 Puppies may be born dead or weak, with some litters containing both live and dead puppies. After abortion, there may be prolonged vaginal discharge.
Infectious materials include fetuses, placenta, lochia, semen, and urine. Lochia may contain as many as 10 10 bacteria/ml. Male urine generally contains more Brucella than does female urine, reaching 10 3 to 10 6 bacteria/ml. 23 Bacteremia is an important feature of the disease and can be prolonged (ie, 2 years or more) and intermittent. Transmission usually occurs at the time of mating or abortion.
Gross lesions of lymphadenopathy and splenomegaly can be seen in adults and puppies of both sexes. Males may have acute epididymal swelling as fluid accumulates between tunics. Testicular atrophy can result from chronic infection. Females often have few gross lesions Aborted fetuses are often partially autolyzed, but fetal lesions may include bronchopneumonia, myocarditis, multifocal renal hemorrhage, lymphadenitis, and hepatitis.
Lymphadenopathy is the result of diffuse lymphoid hyperplasia with increased perifollicular lymphoblasts and infiltration of numerous plasma cells into subcapsular and medullary sinuses. Macrophages with intracellular bacteria may be seen in lymph nodes and spleen if bacteremia is present. In males, necrotizing vasculitis with infiltrates of lymphocytes, plasma cells, and neutrophils may be seen in the prostate gland, epididymis, and testes. Spermatogenesis is decreased and eventually ceases as the disease progresses. In females, there may be subacute to chronic endometritis. Endometrial and myometrial lesions have been described as lymphoplasmacytic with endometrial gland hyperplasia. Hepatic necrosis, myocarditis, meningoencephalitis, iridocyclitis, and retinitis have also been described. 57
As epididymitis, orchitis, and infertility are common features of the disease, seminal fluid cytology may be an important part of the diagnostic workup. Sperm abnormalities are common and include immature sperm, deformed acrosomes, bent tails, swollen midpieces, double tails, heads lacking tails, retained protoplasmic droplets, head-to-head agglutination, and decreased motility. 23,57 As early as 2 weeks after experimental infection, abnormal sperm are noted; by 20 weeks, 90% of sperm may be abnormal. Neutrophils and macrophages with phagocytized sperm are often increased in number. When chronic infection has progressed to bilateral testicular atrophy, azoospermia is seen.
Brucella ovis
B. ovis has a predilection for the genital tract of sheep. Most important, it is a cause of epididymitis. It is contagious and progressive, resulting in testicular atrophy and infertility. Lesions may be unilateral or bilateral, and bilaterally infected rams are usually sterile. Clinical signs are not obvious and may go unrecognized until fertility problems are noted; 117 moreover, Brucella can be found in the semen of infected rams with and without epididymitis. 58 In general, transmission occurs between rams mating the same ewe during the same estrous cycle. Clinically normal rams at the beginning of breeding season have been shown to have palpable epididymitis and Brucella-containing semen by the end of breeding season. 20,58 Ram-to-ram transmission may also occur from rams mounting other rams. 58
Disease progression is slow. The organism can persist on mucous membranes (eg, prepuce) for 1 to 2 months. Acute infection lasting 2 to 5 weeks may go unnoticed, especially when occurring during breeding season. In the acute phase, the testis, particularly the epididymis, is hot, swollen, and doughy. 70 Lesions may be unilateral or bilateral. 17 Grossly, serofibrinous exudate and edema are present in the loose scrotal fascia and between the parietal and visceral tunics. Chronic infection focuses on the epididymis, specifically the tail, which becomes enlarged and firm.
Although relatively avirulent for the nongravid uterus, B. ovis induces placentitis and abortion in pregnant ewes. 110 A yellowish purulent exudate may be seen, particularly in intercotyledonary areas. 47,92 The placenta is edematous, gelatinous, and thickened with adherence of amnion to chorioallantois. Intercotyledonary placenta is thickened with multifocal to coalescent areas of plaque-like thickening resembling yellow-white chamois leather. 92,110 Cotyledons may show various stages of necrosis and detachment.
As with macroscopic lesions, microscopic lesions focus on the tail of the epididymis; however, lesions can also be seen in the ampulla ductus deferens, seminal vesicle, and testis. Lesions in these glands are usually secondary to epididymitis. 47 As such, in contrast to bovine brucellosis, there is no primary orchitis in B. ovis infection. 48 The epididymal epithelium is hyperplastic with hydropic degeneration and intraepithelial lumina or cyst formation (Fig. 5). 48 Epithelial hyperplasia combined with fibrosis results in obstruction and spermiostasis. Exacerbation of lesions is dependent on spermatozoa extravasation and the formation of sperm granulomas. 48,70 Much of the resulting pathology during the chronic disease phase is not the result of the virulence of B. ovis per se but rather the host reaction to extravasated spermatozoa. Free spermatozoa entering the cavity of the tunica vaginalis incite granuloma formation that contributes to testicular atrophy. 48
In the uterus, periarteritis and arteritis, with or without fibrinous thrombi, are distinctive features, 92,110 as is necrosis of the intercotyledonary and chorionic epithelium. As with Brucella-induced placentitis in other species, trophoblastic epithelial cells can be seen to contain numerous intracellular Brucella. Lymph nodes and spleen show signs of activation and hyperplasia. Acute interstitial nephritis focused on the corticomedullary junction and hepatitis of the portal triads are also described. 92
Marine Brucella spp: B. ceti and B. pinnipedialis
Marine Brucella spp have been isolated from numerous cetaceans with no evidence of Brucella-induced pathology. 43 Those lesions that have been associated with Brucella infection include blubber and sub-blubber abscesses; meningoencephalomyelitis; hepatic, splenic, or lymph node necrosis; pneumonia; myocarditis; osteoarthritis; orchitis; mastitis; endometritis; placentitis; and abortion. 13,49,53,56,89
Cetaceans from which B. ceti has been isolated are often stranded and in poor condition. 49,56 Many emaciated dolphins are heavily parasitized by a lung nematode. Within reproductive and intestinal organs of these nematodes are found numerous coccobacilli, identified by immunohistochemistry and polymerase chain reaction (PCR) as Brucella sp. 36,75 Brucella-laden nematodes have been seen in the lungs of both cetaceans and pinnipeds (Fig. 6). Nematode-associated lesions are characterized as multifocal pulmonary granulomas containing degenerate nematodes and nematode larvae, macrophages, lymphocytes, multinucleated giant cells, and abundant immunohistochemically positive Brucella. Ultrastructural studies show Brucella organisms in close association with developing larvae. The species identity of the pulmonary nematodes is unclear. Nematode morphology suggests Parafilaroides sp 36 or Otostrongylus circumlitus. 75 Parafilaroides decorum is a common parasite of sea lions; however, Brucella has also been isolated from another lungworm of harbor porpoises with similar morphology, Pseudalius inflexus. 99 Although the role of nematodes in the pathogenesis of disease in cetaceans is unclear, the primary localization of B. ceti in parasites raises the intriguing possibility that they may serve as a vector for transmission of this Brucella spp.
In contrast to B. ceti, most isolations of B. pinnipedialis have been from clinically normal animals, with no Brucella-associated pathology. 56 However, there are cases in which B. pinnipedialis was isolated from emaciated seals, particularly from pups in rehabilitation centers. 49 The relative paucity of gross lesions in pinnipeds infected with B. pinnipedialis suggests either a difference in virulence between the 2 Brucella spp or a difference in susceptibility to brucellosis between pinnipeds and cetaceans.
Brucella neotomae
First isolated in 1957 from a desert wood rat (N. lepida), B. neotomae is not pathogenic for domestic animals or humans. 54,117 Infection was rapidly cleared with minimal lesion development in experimentally inoculated, guinea pigs, wood rats, and mice, despite dosages as high as 8 × 10 9 bacteria/mouse. 13,54 Swine inoculated intravenously with 1 to 50 × 10 9 bacteria developed bacteremia of up to 5 weeks duration and agglutinating antibodies but did not show lesions or persistent colonization of tissues. 13
Brucella microti
First identified in 1999, B. microti was implicated in an epizootic infection in common voles in the Czech Republic. 63 Affected voles displayed edematous extremities, lymphadenomegaly, draining subcutaneous abscesses, orchitis, splenomegaly, hepatomegaly, and hepatic granulomas. 63 It has also been isolated from mandibular lymph nodes of red foxes in lower Austria; however, it is not clear if lesions were present in affected lymph nodes. Although most closely related to B. suis, B. microti replicates more rapidly in human-derived monocytes and is more virulent in experimentally inoculated mice compared with B. suis.
Vaccines
Vaccination is a valuable tool for helping to control brucellosis. Current vaccines with widespread use under field conditions are attenuated live strains that are highly effective in preventing the clinical effects of brucellosis (abortion and/or infected offspring), which lead to transmission of disease. Currently available vaccines are less effective at preventing transient infection or seroconversion after exposure to virulent field strains. In the early 1960s, live vaccines containing B. abortus strain 19 and B. melitensis strain Rev1, as well as the killed B. abortus strain 45/20 vaccine, were being used under field conditions around the world. 91 A the present time, use of the killed 45/20 vaccine has ceased. Except for the introduction of B. abortus strain RB51 in the United States in 1996 91 and the implementation of the use of B. abortus strain 82 in the Russian Federation in 1974, 69 there has been a paucity of vaccines developed over the past 50 years that have progressed to significant use under field conditions in natural hosts. Similarly, a brucellosis vaccine with acceptable safety and efficacy in humans remains elusive.
Although data from new vaccine candidates continue to be published, most work has been conducted in murine models. At the present time, none of the new vaccine candidates have progressed to significant field use. Due to safety issues associated with current live vaccines, technologic advances in in vivo antigen expression, adjuvants, and delivery methods are attractive for development of nonliving Brucella vaccines with equivalent or greater efficacy. Although some genes have been identified as playing a role in protection, progress in the development of subunit vaccines is impaired by the difficulties in fully characterizing which Brucella antigens mediate protective immunity. The possibility cannot be excluded that protective antigens may differ by Brucella spp and reservoir host. Although it is anticipated that developmental work will continue, it is impossible to predict when a new vaccine will be developed that demonstrates properties that are an improvement over currently available vaccines.
Laboratory Models for Studying Brucella Pathogenesis and Vaccine Efficacy
Inbred mice, particularly BALB/c and C57BL strains, have been used most frequently as laboratory models for studying the pathogenesis of brucellosis. 114 Most studies have used intraperitoneal infection, but aerosol, oral, and intranasal routes have also been used. Some strains such as the C57BL/10 have been described as being more resistant to Brucella colonization due to T-helper 1 polarization of the immune response. Bacterial loads in the spleen and/or liver are most commonly used to assess pathogenesis. Murine models may more closely relate to human infection where splenomegaly is a common clinical observation. However, in preferred hosts, Brucella spp localize more frequently in lymphatic tissues, mammary glands, and reproductive organs, and replication of clinical signs in preferred hosts (ie, abortion) in murine models is rare. The fact that natural hosts are genetically outbred compared with inbred strains of mice may also be responsible for differences in pathogenesis and immunologic responses between laboratory models and reservoir hosts. Although murine models are popular due to lower cost and reduced requirements for animal facilities, the authors’ opinion is that murine models have significant limitations with regard to evaluating vaccine efficacy and understanding the pathogenesis of brucellosis in natural hosts. It should be noted that with the exception of abortion, natural hosts generally do not demonstrate clinical disease. In comparison, humans appear to be aberrant hosts for Brucella and often have significant symptoms of clinical disease that are frequently chronic.
Other models that have been used include guinea pigs, rabbits, rats, and nonhuman primates. The guinea pig is probably the laboratory model with the greatest susceptibility to Brucella infection. 114 It has been used to facilitate isolation of classic species of Brucella, evaluate effectiveness of antibiotic treatments, and also assess the efficacy of vaccines and vaccine-adjuvant combinations. Lesions of brucellosis are observed in the liver, spleen, lungs, and lymph nodes. 67 Rats have also seen limited use as laboratory models of brucellosis. Brucella-induced abortion is rare, but rats can venereally transmit B. abortus, and offspring can be latently infected. 67,68 Lesions of necrosis in periplacentomal chorionic epithelium and metritis have been observed in the uterus of pregnant rats infected with B. abortus. Nevertheless, the rat model appears to have similar limitations to those of murine models. Rabbits are partially susceptible to Brucella, with susceptibility increasing with pregnancy. However, rabbits are not considered a model of choice for studying the pathogenesis of brucellosis. Nonhuman primate models (Macaca arctoides and Macaca mulatta) have been infected through oral, subcutaneous, and aerosol routes with virulent strains of Brucella. Bacteremia was demonstrated for up to 8 weeks, and lesions of focal granulomatous hepatitis, splenitis, lymphadenitis, and occasionally orchitis, epididymitis, and endometritis were reported, similar to lesions in humans infected with Brucella. 114 Current knowledge would suggest that primate models most closely mimic pathogenesis of disease in humans but are probably of limited value for understanding the disease in preferred hosts.
The validity of murine models of brucellosis has been questioned due to differences between natural transmission across mucosal surfaces and intraperitoneal experimental infection, as well as differences between natural reservoirs and murine hosts in tissue localization and clinical signs. Key differences between human and mice immune systems have been noted. 12 These concerns are also supported by observations that efficacy of brucellosis vaccines in the murine models does not always correlate with protection observed after experimental vaccination and infection of natural hosts. These observations support the value of studying the pathogen in its preferred hosts, particularly when the goal is resolution of disease issues in preferred hosts. By studying the pathogen in its natural host, we may be able to devise efficient control measures in that host, thereby disrupting transmission of the pathogen to humans. 12 Although laboratory models continue to have value for basic research, they cannot circumvent the need for studies in natural hosts of Brucella.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
