Abstract
Egyptian rousette bats (ERBs; Rousettus aegyptiacus; family Pteropodidae) are associated with a growing number of bunyaviruses of public health importance, including Kasokero virus (KASV), which was first identified as a zoonosis in Uganda in 1977. In this study, formalin-fixed paraffin-embedded tissues from a previous experiment in which KASV infection was confirmed in 18 experimentally infected ERBs were used for an in-depth analysis using histopathology, in situ hybridization (ISH) for detection of viral RNA, immunohistochemistry (IHC) to assess the mononuclear phagocyte system response, and quantitative digital image analysis to investigate virus clearance from the liver and spleen within a spatial context. Significant gross and histological lesions were limited to the liver, where KASV-infected bats developed mild to moderate, acute viral hepatitis, which was first observed at 3 days postinfection (DPI), peaked at 6 DPI, and was resolved by 20 DPI. A subset of bats had glycogen depletion (n = 10) and hepatic necrosis (n = 3), rarely with intralesional bacteria (n = 1). Virus replication was confirmed by ISH in the liver, spleen, lymph nodes, and tongue. In the liver, KASV replicated in the cytoplasm of hepatocytes, to a lesser extent in mononuclear phagocytes, and rarely in presumptive endothelial cells. Most KASV RNA, as detected by ISH, was cleared from the spleen and liver by 6 DPI. It is concluded that ERBs have effective mechanisms to respond to this virus, clearing it without evidence of clinical disease.
Bats (order Chiroptera) are the natural reservoir hosts for several high-consequence viral pathogens of humans, including Nipah, Hendra, and Marburg viruses.23,25,27,32,33 In addition, a growing number of arboviruses have been associated with bats, including viruses of the families Nairoviridae, Flaviviridae, Reoviridae, and Togaviridae. 11 Within the family Nairoviridae, the genus Orthonairovirus contains tick-borne viruses such as the human-pathogenic Crimean-Congo hemorrhagic fever virus (CCHFV), the ovine-pathogenic Nairobi sheep disease orthonairovirus (NSDV), along with 8 other known orthonairoviruses, including Ahun, Kasokero, and Issyk-Kul viruses, which have been associated with bats based on molecular detection or virus isolation from bats and/or their ectoparasites.1,10,11,17,20,22,34,35 Collectively, orthonairoviruses constitute an emerging group of viruses of public health importance, several of which cause severe systemic disease in humans, whereas it is unclear if bats develop orthonairovirus-associated disease. The initial description of Ahun virus, which was identified by RNA sequencing from the lungs of a whiskered bat (Myotis mystacinus) and common pipistrelle (Pipistrellus pipistrellus) in France, indicates that the 2 infected bats were moribund prior to tissue collection; however, histopathology was not included in that report to confirm or refute virus-associated pathology or other underlying disease(s) or trauma. 10 Tissue-based assessments of bats infected with orthonairoviruses are critical to begin to understand how bats respond to these viruses, if and how they avoid clinical disease, and to inform viral countermeasure development in anticipation of spillover events.
Ecological and experimental studies have linked the Egyptian rousette bat (ERB; Rousettus aegyptiacus), a common, cave-dwelling African bat, with 2 closely related orthonairoviruses, Kasokero and Yogue viruses (KASV and YOGV), as well as the human-pathogenic Marburg and Ravn (MARV and RAVV; family Filoviridae), and Sosuga viruses (SOSV; family Paramyxoviridae).2,4,3,5,19,20,30,32,33 KASV was first isolated from the sera of ERBs at Kasokero Cave, Uganda, in 1977. 20 Since its initial discovery, KASV has been collected from argasid ticks (Ornithodoros [Reticulinasus] faini) in the rock crevices of Lannar Gorge Cave, South Africa, and Python Cave, Uganda, where large colonies of ERB are present.8,20 The fact that KASV has been isolated from O. (R.) faini ticks and from ERBs on which they feed suggests that this virus is naturally maintained in an ERB-O. (R.) faini enzootic life cycle. An outbreak of KASV infection in humans occurred following the occupational exposure of 4 individuals who were either working with the virus or had access to the laboratory where virus characterization assays were performed at the Uganda Virus Research Institute. 20 During the outbreak, signs of KASV infection ranged from a mild, febrile illness to severe, systemic disease with fever, headache, abdominal pain, diarrhea, and muscle and joint pain; all individuals made a full recovery. 20
Orthonairovirus-associated pathology in vertebrate hosts is understudied, and descriptions have thus far been limited to humans naturally infected with CCHFV, murine and nonhuman primate models of CCHF, sheep experimentally and naturally infected with NSDV, and mice experimentally infected with the 11SB23 strain of Leopards Hill virus (LPHV), a virus isolated from a giant leaf-nosed bats (Hipposideros gigas) in Zambia.9,13,16,17 Gross and/or histological lesions in severe or fatal orthonairovirus infections include hepatopathy or hepatic necrosis, often with minimal inflammation, enteritis and/or gastrointestinal hemorrhage, and splenic lymphoid depletion and/or necrosis.9,16,17 Severe systemic orthonairoviral disease can progress to widespread hemorrhaging, typified by CCHF in humans, and is hypothesized to be driven by a dysregulated immune response and strong tropism of these viruses for endothelium and cells of the mononuclear phagocyte system. 9 In contrast, very little is known about how bat-derived orthonairoviruses interact with their suspected natural reservoir hosts, including KASV in ERBs.
ERBs experimentally infected with KASV did not show any overt signs of illness despite developing transient viremias (peak viral loads = 5.1–5.9 log10TCID50eq/ml) with virus replication in multiple tissues, including liver, spleen, lymph nodes, and skin at the inoculation site, and shedding of virus into the oral secretions, feces, and urine. 29 The present study aimed to build on these findings by using formalin-fixed paraffin-embedded (FFPE) ERB tissues derived from the previous experiment to conduct a comprehensive tissue-based analysis of KASV-ERB interactions within a spatial context. To accomplish this goal, we used histopathology, morphometry, in situ hybridization (ISH) for detection of viral RNA, immunohistochemistry (IHC) for colocalization of KASV RNA with cells of the mononuclear phagocytic system, and quantitative image analysis to investigate virus clearance from the spleen and liver. To our knowledge, this is the first study to investigate the pathogenesis of an orthonairovirus in a bat host. Understanding more about how these viruses circulate within their bat hosts could help inform prevention and control strategies for other emerging viral zoonosis of bats.
Methods
Tissue Sample Collection, Processing, and Virus Origin
The ERB tissues used in the present study were obtained from an experimental infection study which was performed under biosafety level 4 (BSL-4) conditions in accordance with safety regulations at the Centers for Disease Control and Prevention (CDC, Atlanta, GA). 29 The experimental infections were conducted with approval from the CDC Institutional Animal Care and Use Committee (IACUC), and in accordance with the animal welfare guidelines set forth in the Guide for the Care and Use of Laboratory Animals (Committee for the Update of the Guide for the Care and Use of Laboratory Animals, National Research Council of the National Academies 2011). Husbandry, housing conditions, and diet for the duration of the experiment were previously described. 4 Briefly, food consisted of a variety of fresh fruit dusted with a protein vitamin supplement (Lubee Bat Conservancy, Gainesville, FL). Water was provided ad libitum.
The KASV isolate was derived from an argasid tick (O. [R.] faini) collected from an ERB roost, Python Cave, Uganda, and passaged twice in Vero E6 cells. 30 To mimic a tick bite, as described in the original study, all bats were inoculated intradermally in the ventral abdominal region with 4 log10 TCID50 KASV diluted in phosphate-buffered saline (PBS); 3 control bats were mock inoculated the same volume of PBS. Bats were euthanized at 3, 6, 9, 12, 18, and 20 days postinfection (DPI), along with 3 controls (n = 3 for each time point and controls). Blood and selected tissue samples were collected for viral load determination by reverse-transcriptase quantitative polymerase chain reaction (qRT-PCR) at the time of euthanasia, with multiple tissues testing positive for KASV by qRT-PCR. Following tissue collection, all bat carcasses were immersed in 10% neutral buffered formalin (10% NBF) for a minimum of 7 days, with a complete change of formalin after day 3, prior to removing the carcasses from the laboratory. Carcasses were transferred to 70% ethanol within 2 weeks and further dissected for histological analyses as detailed below.
RNA Extraction and qRT-PCR
RNA extraction and qRT-PCR were performed as previously described. 29 KASV qRT-PCR cycle threshold (CT) values were normalized using ERB beta-2-microglobulin (B2M) qRT-PCR CT values to account for any differences in tissue specimen weights.
Gross Pathology and Histopathology
The carcasses, once removed from ethanol, had major organs in situ, with some pieces removed for qRT-PCR testing. A more rigorous examination of the tissues was done at this time, with removal and extensive slicing of various organs for observation of lesions which might be only visible on cut section. A full set of tissues from each carcass was collected, including representative sections from the brain; eye; heart; lungs; trachea; thymus; esophagus; thyroid glands; liver (4 noncontiguous sections including gall bladder); stomach; small intestines (duodenum with pancreas, jejunum, and ileum); large intestines; spleen; kidney; adrenal glands; salivary gland; tongue; submandibular, axillary, inguinal, sublumbar, and mesenteric lymph nodes; skin from the inoculation site; patagium; skin from the chest region; and pectoral muscle. Tissue sections were placed into cassettes, immersed in 70% ethanol, and routinely processed for histological analyses at the University of Georgia (UGA) Histology Laboratory. Slides were reviewed in a blinded manner without knowledge of time point or infection status. A subset of livers from 6, 9, 12, and 18 DPI, and control bats were stained with Perl’s Prussian blue and Periodic acid-Schiff reaction stains at the UGA Histology Laboratory to identify the accumulated pigment in macrophages within the foci of liver inflammation.
Morphometric Analysis of Hematoxylin-Eosin-Stained Liver Sections
Liver slides were scanned at 200× using an Aperio Slide Scanner (Aperio ScanScope XT, Aperio Technologies, Inc., Vista, CA) and loaded into the open-source bioimage analysis software QuPath (ver 0.3.2.). 7 Foci of mononuclear cells, defined as clusters of at least 3 mononuclear cells either located within the sinusoids, distorting the hepatic cords, or in the perivascular space, were manually annotated. The total liver area (mm2) was measured by tracing each section of liver using the wand tool and summing the individual annotations. The sum of the area for all foci was digitally quantified and divided by the total liver area to obtain the percentage of liver tissue affected (% liver affected).
Assessment of Hepatocellular Glycogenosis
Hepatocellular glycogenosis is a normal finding in frugivorous bats and in our colony of ERBs. 12 Semiquantitative hepatocellular vacuolation scores were determined for all bat livers. At least 4 noncontiguous hematoxylin-eosin (H&E)-stained liver sections were reviewed and assigned a score from 1 to 4 according to the following criteria: 0 = normal hepatocellular glycogenosis; 1 = minimal, focal depletion or condensation of hepatocellular cytoplasmic vacuoles, typically restricted to hepatocytes surrounding the foci of inflammation; 2 = mild, zonal depletion of vacuoles; 3 = moderate, occasionally coalescing and/or bridging depletion of vacuoles; 4 = moderate to marked, diffuse depletion of vacuoles.
RNAScope ISH
To localize KASV RNA in tissues, we performed ISH using the RNAscope 2.5 HD Reagent Kit—RED (Advanced Cell Diagnostics, Newark, CA) and followed the manufacturer’s instructions to visualize the signal in FFPE liver, spleen, and lymph nodes for bats euthanized a 3, 6, 9, 12, and 18 DPI, and in the 3 control bat tissues. 36 A custom antisense probe targeting the complementary RNA sequence of the nucleoprotein gene of KASV obtained from the published genome was designed (GenBank Accession # MT309090). 30 To confirm adequate RNA integrity of the FFPE samples, a medium-expressing housekeeping gene (peptidylprolyl isomerase B, PPIB) probe was applied to a subset of samples (3 infected and 2 control bats) as a positive control (R. aegyptiacus GenBank Accession # XM_016141088.1). A probe targeting the dapB gene (Advanced Cell Diagnostics, Newark, CA) was used as a negative control. For each bat, 4 sections of liver, 2 sections of spleen, and 1 cross section of inguinal lymph node were evaluated on a single slide. A subset of other tissues from one 3 and one 6 DPI bat with histological lesions that were suggestive of virus-induced tissue injury and/or with high tissue viral loads by qRT-PCR were subjected to ISH to investigate potential sources of shedding and replication in the subcutis at the inoculation site. These tissues included small intestines, kidneys, tongue, salivary gland, and skin at the inoculation site. IHC for detection of KASV proteins in tissues was not performed.
Co-detection of KASV With Mononuclear Phagocytes
Following the identification of KASV in cells morphologically consistent with mononuclear phagocytes in the primary target tissues (liver, spleen, and lymph nodes), a combination ISH-IHC protocol was developed to investigate KASV replication and virus interactions with cells of the mononuclear phagocyte system using the pan-macrophage/monocyte/dendritic cell marker, ionized calcium binding adapter molecule (Iba1). RNAScope was performed first according to the manufacturer’s instructions. Subsequently, the IHC protocol was initiated by applying a universal blocking reagent (Background Punisher, Biocare Medical, Pacheco, CA) and incubating at room temperature for 10 minutes, then washing the slides (1X TBST, 2 × 5 minutes). A solution containing the anti-Iba1 antibody (FujiFilm, catalog # 019-19741) diluted in Co-Detection antibody Diluent (Advanced Cell Diagnostics, Newark, CA, catalog # 323160; final antibody dilution = 1:8000) was applied to each slide, and the slides were incubated overnight at 4°C. The next day, slides were washed (1X TBST, 3 × 5 minutes), incubated for 15 minutes at room temperature with the MACH 3 Rabbit Probe (Biocare Medical, Pacheco, CA), and washed again (1X TBST, 2 × 5 minutes). Slides were then incubated for 15 minutes with the MACH 3 Horseradish-Perioxidase (HRP) Polymer (Biocare Medical, Pacheco, CA) and washed (1X TBST, 3 × 5 minutes). Finally, 1 drop of the HRP Chromogen (Biocare Medical, Pacheco, CA) was applied and incubated for 5 to 7 minutes at room temperature, and the slides were washed (1X TBST, 3 × 5 minutes), counterstained with hematoxylin (Leica, Richmond, IL), and coverslipped with Permount for a permanent record. Appropriate immunolabeling was verified using internal controls (tissue macrophages).
Semiquantitative and Quantitative Analysis of KASV Positive Hybridization Signal
ISH slides containing 4 noncontiguous sections of liver tissue, 2 spleen sections, and 1 section of lymph node were scanned at 400× using an Aperio Slide Scanner (Aperio ScanScope XT, Aperio Technologies, Inc., Vista, CA) and imported into the open-source bioimage analysis software QuPath ver. 0.3.2. 7 Positive KASV hybridization was characterized by punctate to saturated, bright red labeling within the cytoplasm of target cells. The liver, spleen, and inguinal lymph nodes were assigned semiquantitative scores based on the degree of ISH signal observed as follows: + = focal signal, less than 2 to 3 cells per 40× high-powered field (hpf; hpf = 0.086 mm2); ++ = scattered signal in multiple foci throughout section, less than 2 to 3 cells per hpf; +++ = multifocal signal throughout section; 3 to 5 positive cells per hpf; ++++ = multifocal signal throughout section, 5 to 10 cells per hpf; and +++++ = multifocal signal throughout section, frequent saturated signal, 5 to 10 cells per hpf. KASV was detected in the axillary, but not the inguinal lymph node of 1 bat; the results are presented for the former tissue in that case.
The liver and spleen sections were further analyzed to quantify the KASV signal within spatial and temporal contexts. Tissue areas of at least 95 mm2 (range = 95–231 mm2) and 5 mm2 (range = 5–22 mm2) were analyzed for liver and spleen, respectively. Positive pixels were segmented from negative regions and background staining by thresholding; the threshold was customized for each slide (range = 0.15–0.18). The other default parameters in the Create Threshold tool (Gaussian sigma = 1.5 mm and background radius = 15 mm) were used. False positives based on morphology and location were removed, as was background staining and artifact. The measurements were expressed as percent positive pixels (%PP).
Statistical Analyses
Statistical analyses were performed using Prism 9.3.1 (GraphPad Software, LLC, La Jolla, CA). For the % liver affected values obtained via digital image analysis, from KASV-infected bats at each time point (n = 3) were compared with those of the control bats (n = 3) using a 1-way analysis of variance (ANOVA) test, followed by Dunnet’s multiple comparison test if the ANOVA yielded a statistically significant result (P < .05). Correlations between the viral loads and the %PP in the liver and spleen at 3 and 6 DPI were analyzed using the Spearman rank test.
Data Availability
The data analyzed in this study are available as Supplemental Materials.
Results
Gross and Histological Lesions in the Livers of KASV-Infected ERBs
Gross lesions were observed in a subset of KASV-infected bats. The most frequently observed lesion was hemorrhage in the liver (n = 4). Hemorrhage was observed in the liver of one 9 DPI bat at the time of tissue collection. 29 Variable pinpoint hepatic hemorrhages were identified during dissection of 3 additional formalin-fixed carcasses (two 6 DPI bats and one 9 DPI bat). Hepatic hemorrhages were pinpoint and generally totaled less than 10, except for one 6 DPI bat which had numerous pinpoint hemorrhages distributed throughout all liver lobes (Fig. 1b, c). The formalin-fixed carcass of the 9 DPI bat for which hemorrhages were observed at the time of tissue collection had a regionally extensive focus of pallor, which extended into the hepatic parenchyma on cut section (Fig. 1d). All animals had moderate to abundant abdominal and subcutaneous adipose tissue stores. There were no other significant gross lesions in any animal.
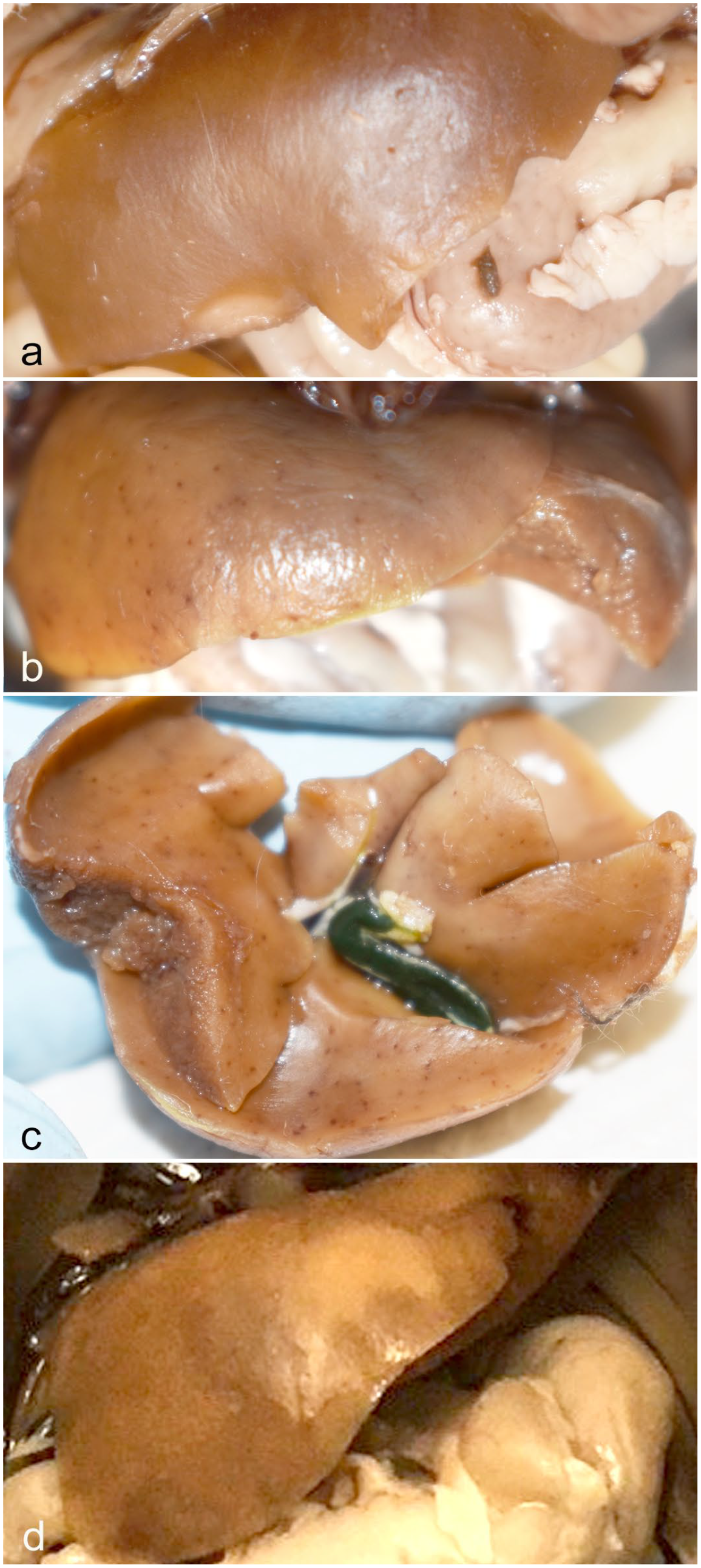
Kasokero virus (KASV) infection, liver, Egyptian fruit bat (ERB), 6 to 9 days postinfection (DPI). (a) Formalin-fixed liver of a control bat demonstrating normal color of the liver in this species. Liver color is variable, depending on the glycogen content of hepatocytes. Hepatic glycogenosis is a normal finding in ERBs, and in fruit bats in general. (b) Formalin-fixed liver of an ERB experimentally infected with KASV and euthanized at 6 DPI. The liver contains numerous pinpoint hemorrhages distributed throughout all lobes. (c) Liver from the same 6 DPI bat as in (b), removed to demonstrate the widespread distribution of pinpoint hemorrhages on the ventral surface. The gall bladder is grossly normal. (d) Formalin-fixed liver of a KASV-infected ERB euthanized at 9 DPI. There is a focally extensive region of pallor (necrosis).
The livers of all KASV-infected bats (n = 18), but not control bats, contained multifocal to coalescing, randomly distributed, variably sized inflammatory cell foci containing mononuclear inflammatory cells and rare granulocytes (Fig. 2). At 3 and 6 DPI, foci frequently contained degenerating, apoptotic, or necrotic hepatocytes, especially at the junction with the normal parenchyma, and hemorrhage (Fig. 2a–c). Hepatocytes immediately adjacent to inflammatory foci in one 3 DPI bat contained numerous eosinophilic to amphophilic, round, variably sized cytoplasmic inclusions (Fig. 2b). That same 3 DPI bat had a focally extensive area of capsular to subcapsular fibrin exudation and hemorrhage, and a small focus of hepatic necrosis with low numbers of mononuclear inflammatory cells and apoptotic cells (Supplemental Figure S1a).
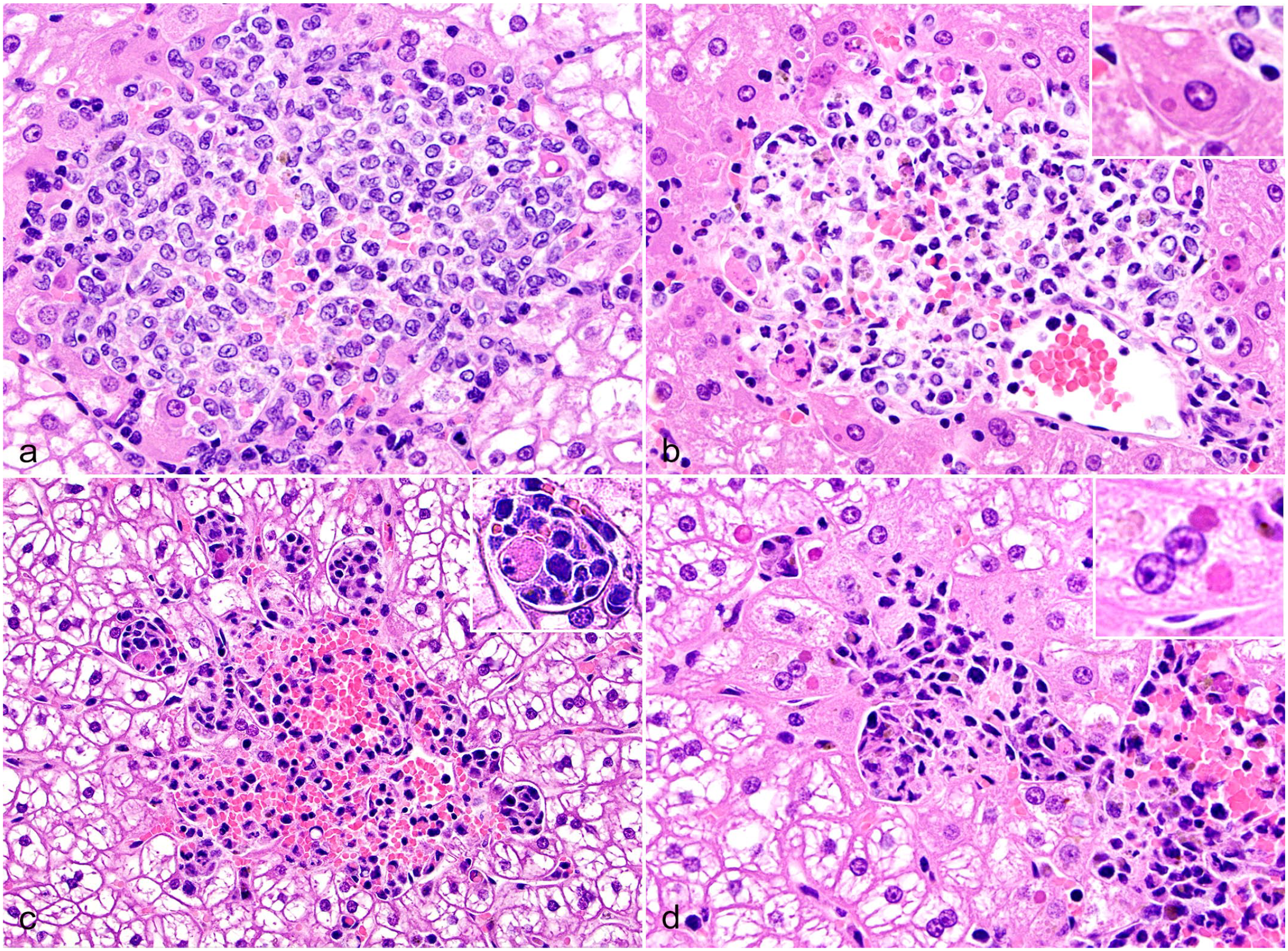
Kasokero virus infection, liver, Egyptian rousette bats, 3 and 6 days postinfection (DPI), hematoxylin and eosin. (a) A large aggregate of mononuclear inflammatory cells is poorly demarcated from the surrounding hepatic parenchyma and contains a mild amount of hemorrhage, 3 DPI bat. (b) In another 3 DPI bat liver, hepatocytes along the periphery of an inflammatory cell focus contain 1 or more irregularly round, 2–4 µm diameter, eosinophilic cytoplasmic inclusions (inset), and rare apoptotic or necrotic cells are present. (c) A large mononuclear inflammatory cell focus is well demarcated from surrounding hepatocytes and contains abundant hemorrhage and rare apoptotic hepatocytes (inset), 6 DPI bat. (d) Along the periphery of mononuclear inflammatory cell focus, low numbers of hepatocytes contain 2–4 µm diameter, eosinophilic cytoplasmic inclusions (inset), 6 DPI bat.
In the 6 DPI bats, the inflammatory cell foci frequently coalesced tended to have more hemorrhage compared with 3 DPI, and often contained hemosiderin-laden macrophages (Fig. 2c). Round, variably sized, eosinophilic cytoplasmic inclusions were rarely observed in hepatocytes along the periphery of inflammatory foci in two 6 DPI bats (Fig. 2d). Foci contained increased numbers of small mononuclear cells morphologically consistent with lymphocytes compared with the 3 DPI bats and were more clearly delineated from the surrounding hepatic cords (Fig. 2c, d). Hepatocytes with shrunken and rounded cell borders, and pyknotic nuclei (morphologic features consistent with apoptosis) were present within and along the periphery of some foci (Fig. 2c).
At 9 DPI, inflammatory foci were less numerous and comprised primarily of brown pigment-laden macrophages with fewer lymphocytes and rare apoptotic hepatocytes (Supplemental Figure S1b). The cytoplasmic pigment was interpreted to be a mixture of hemosiderin and lipofuscin based on the mixed staining on Perl’s Prussian blue and Periodic acid-Schiff reaction stains. In addition to foci of mononuclear cells, 2 of the 9 DPI bats had foci of hepatic necrosis with sinusoidal fibrin accumulation, necrotic and apoptotic cells debris, and hemorrhage (Supplemental Figure S1b). In one 9 DPI bat, at least two 1 to 2 µm bacilli were evident in an area of necrosis, suggestive of sepsis. Foci continued to decrease in size and frequency at 12 DPI, and by 18 and 20 DPI, only few small foci, consisting predominantly of hemosiderin-laden macrophages were present, either in the sinusoids or in the periportal or centrilobular connective tissues (Supplemental Figure S1c).
In addition to mononuclear inflammatory cell infiltrates and occasional hepatic necrosis, most KASV-infected bats (10/18; 56%) exhibited some degree of hepatocellular glycogen depletion that was not restricted to hepatocytes immediately surrounding the foci of mononuclear inflammatory cells. The distribution of glycogen depletion varied among bats from mild centrilobular or portal depletion, to bridging swaths of depletion, and rarely to diffuse depletion. In addition to hepatocellular glycogen depletion, one 12 DPI bat exhibited multifocal hepatocellular degeneration, characterized by hepatocellular hypereosinophilia and the accumulation of 1 to 2 round cytoplasmic vacuoles (Supplemental Figure S1d); the distribution of degenerating hepatocytes was random.
Morphometric Liver Analyses and Hepatic Viral Loads
At 3 and 6 DPI, there were statistically significant increases in the % of liver tissue affected compared with the negative control bats (Dunnet’s multiple comparison test; P = .0419 and .0148, respectively). From 6 to 12 DPI, the mean % liver affected decreased over time, and by 18 DPI, inflammatory cell foci were rare.
The detailed qRT-PCR results for blood, oral, rectal, and urine swabs, and tissues collected at the time of euthanasia were previously reported. 29 Viral loads obtained from the liver are summarized here for correlation with the hepatic morphometrics (Fig. 3). Viral loads in the liver peaked at 3 DPI (mean = 4.3 log10TCID50 eq/g) and KASV RNA was detected in all liver samples but one at 20 DPI.
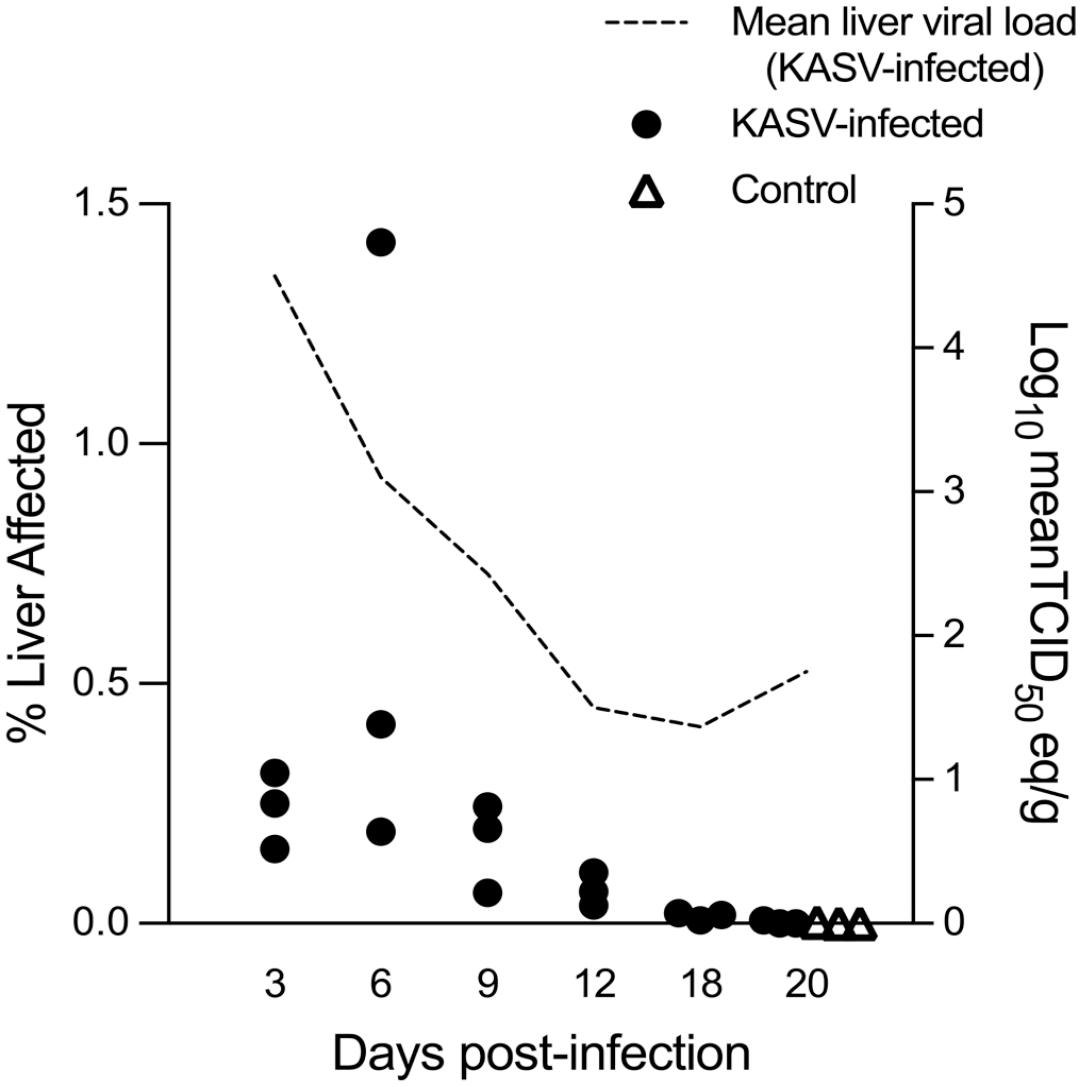
Morphometric analysis of hepatic mononuclear inflammatory cell infiltrates in KASV-infected Egyptian rousette bats and mean liver viral loads for each time point. Each dot represents the percentage of liver area occupied by mononuclear inflammatory cells obtained by whole-slide imaging and morphometric analysis for a single bat. Negative control bats were euthanized at 20 days postinfection (DPI). There was a statistically significant increase in the % liver affected at 3 and 6 DPI compared with controls. The dashed line represents the mean liver viral load for each time point (Log10 meanTCID50 eq/g) obtained at necropsy. KASV, Kasokero virus.
Hepatocellular Glycogen Scoring
The average glycogen depletion scores were highest in the 9 and 12 DPI bats (2.3 average score at both time points), but there were no statistically significant differences when comparing these scores with the control bat liver scores (Fig. 4). Control bats had diffuse hepatocellular glycogenosis, which is considered normal for this species and for fruit bats in general.12,18
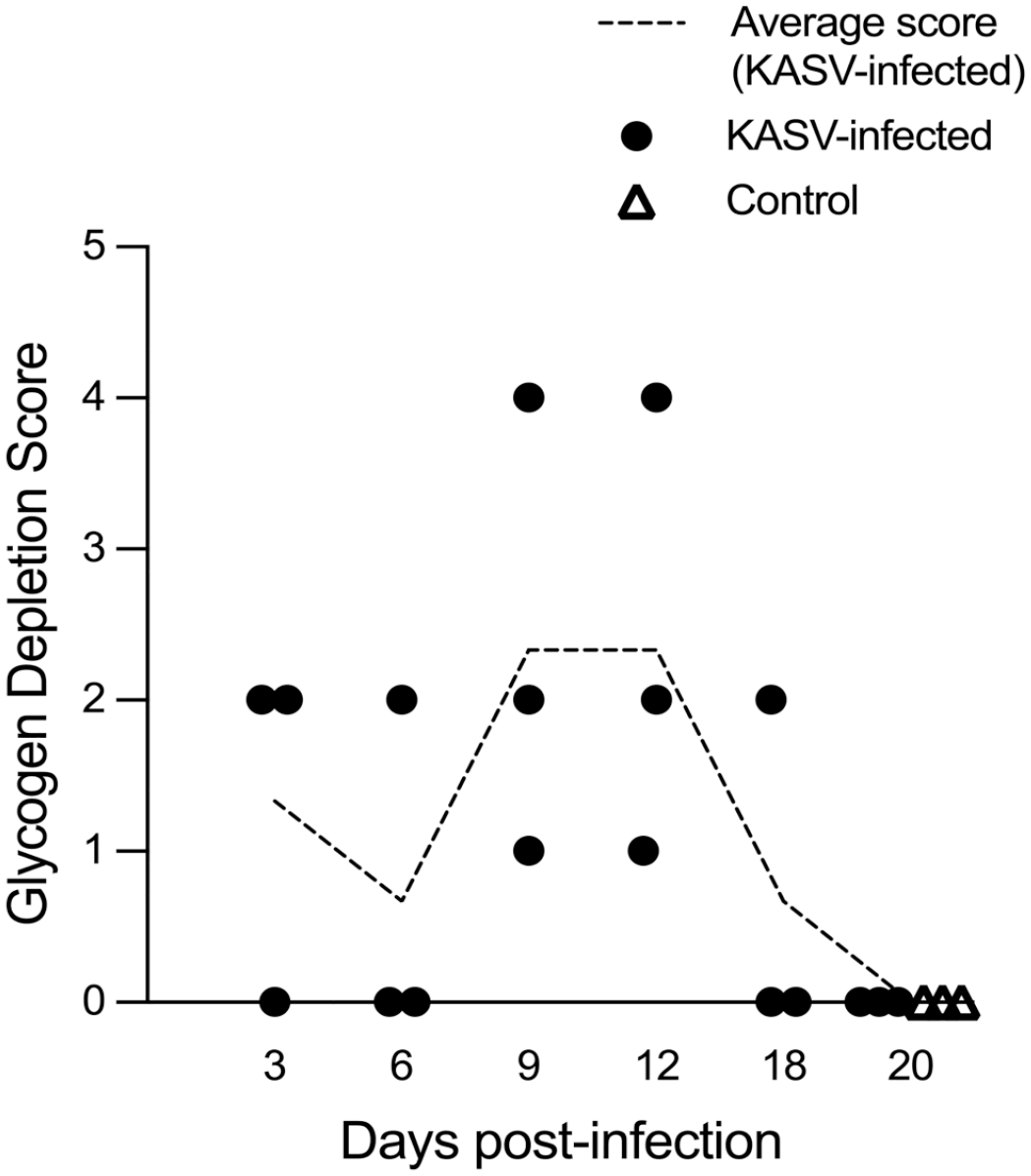
Hepatic glycogen depletion scores in KASV-infected Egyptian rousette bats. Each dot represents the score for a single bat. Negative control bats were euthanized at 20 days postinfection. Scoring criteria were as follows: 0 = no hepatocellular glycogen depletion; 1 = minimal, focal depletion, typically restricted to hepatocytes surrounding the foci of inflammation; 2 = mild, zonal depletion; 3 = moderate, occasionally coalescing and/or bridging depletion; 4 = moderate to marked, diffuse depletion. Dashed line represents the average glycogen depletion score for each time point. KASV, Kasokero virus.
Lesions in Other Tissues
The subcutaneous tissue at the site of inoculation of a subset of KASV-infected bats (n = 4) and 1 control bat contained loose aggregates of macrophages with fewer neutrophils and occasional foci of adipose tissue saponification. The macrophagic inflammation was generally mild and tended to decrease in cell density from 3 to 20 DPI. Moderate histiocytic inflammation was present in the subcutis and panniculus of one 6 DPI bat at the inoculation site (data not shown). In 1 section of duodenum from a single bat at 6 DPI, there was multifocal degeneration of villus tip enterocytes with focal ulceration of a villus tip and mild accumulation of eosinophilic fluid in the lamina propria with few neutrophils (data not shown). One 6 DPI and two 9 DPI bats had multifocal, random, small intestinal villus fusion, and 2 of these bats had mild, multifocal accumulations of eosinophilic fluid in the villus lamina propria (data not shown). No significant lesions were observed in any other tissues.
KASV Tissue and Cellular Tropism
The KASV nucleoprotein transcript was detected by ISH in the liver and secondary lymphoid tissues (spleen and lymph nodes) using a custom antisense RNA probe to detect replicating virus (Figs. 5, 6). In the liver of the 3 DPI bats, much of the positive hybridization signal was localized to discrete, randomly distributed foci of infection throughout all liver lobes. Within these foci, viral RNA was present within the cytoplasm of hepatocytes, mononuclear phagocytes, and rarely in endothelial cells (Fig. 5a–f). Foci of hepatocytes containing KASV RNA were often adjacent to the portal veins found in the portal triads. Occasionally, elongate cells (presumptive endothelial cells) lining central veins (Fig. 5c) and Iba1+ macrophages within the vessel walls contained viral RNA (Fig. 5d). Within individual hepatocytes, the extent of the hybridization signal varied from low (single or few dots) to high with filling of the entire cytoplasmic space and saturation of the ISH signal (Fig. 5a–f). In one 3 DPI bat, infection of contiguous hepatocytes along individual or adjacent hepatic cords was observed (Fig. 5e). Extracellular, intravascular signal was occasionally observed in the sinusoids adjacent to foci of infected hepatocytes (Fig. 5e). Hepatocytes along the periphery of the foci of mononuclear inflammatory cells contained abundant KASV RNA, as did fragmented cells, numerous Iba1+ mononuclear phagocytes within the cellular foci, and occasional Kupffer cells (Fig. 5f).
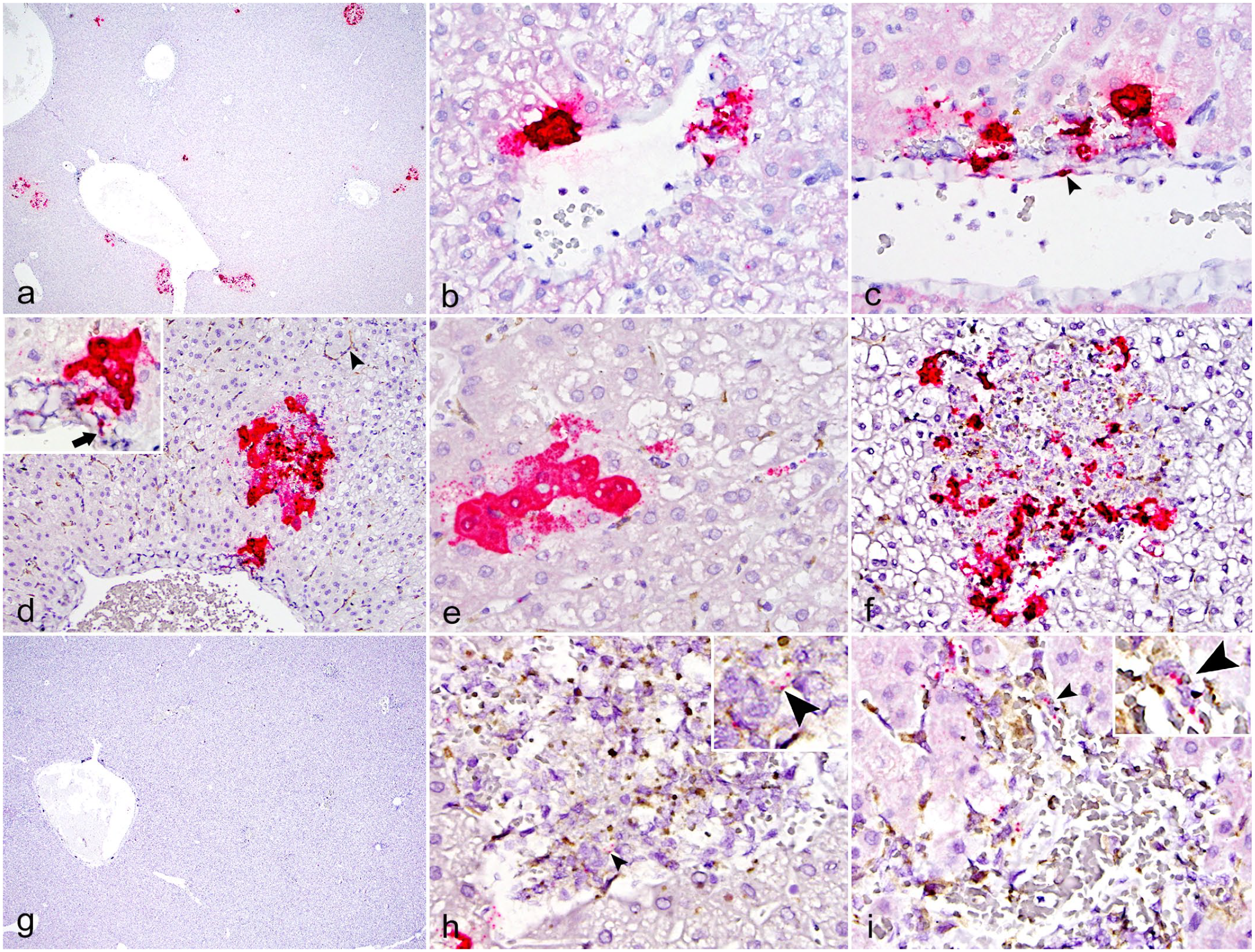
Kasokero virus (KASV) infection, liver, Egyptian rousette bats, 3, 6, and 9 days postinfection (DPI). RNAScope in situ hybridization (ISH) assay designed with an antisense probe targeting the KASV nucleoprotein gene. (a) Randomly distributed foci with intense KASV ISH signal are present in the liver, 3 DPI bat. (b) Intense KASV ISH signal within hepatocytes surrounding a small blood vessel, 3 DPI bat. (c) Hepatocytes and presumptive endothelial cells (arrowhead) surrounding a blood vessel contain KASV RNA, 3 DPI bat. (d) Dual KASV ISH (red) and Iba1 (brown) immunohistochemistry (IHC) demonstrating focal colocalization of KASV with Iba1+ cells in the wall of a large artery (inset, arrow) and in a focus of infected hepatocytes, 3 DPI bat. Iba1 highlights normal Kupffer cells throughout the section (arrowhead). (e) Dual KASV ISH (red) and Iba1 (brown) IHC demonstrating KASV RNA in several contiguous hepatocytes and in the sinusoids, 3 DPI bat. (f) Dual KASV ISH (red) and Iba1 (brown) IHC demonstrating KASV within Iba1+ cells in a focus of mononuclear cell inflammation, 3 DPI bat. (g) Low magnification image demonstrating the scarcity of KASV RNA in a 6 DPI bat. KASV ISH. (h) Dual KASV ISH (red) and Iba1 (brown) IHC demonstrating rare KASV RNA within Iba1+ cells in the focus of inflammation (arrowheads) and in a hepatocyte along the periphery of the focus, 6 DPI bat. (i) Dual KASV ISH (red) and Iba1 (brown) IHC demonstrating rare KASV RNA within Iba1+ cells and along the periphery of a focus of inflammation (arrowheads), 9 DPI bat. KASV ISH with red chromogen and hematoxylin counterstain (a, b, c, and g). Dual ISH-IHC for KASV nucleoprotein ISH (red) and Iba1 IHC (brown) with hematoxylin counterstain (d, e, f, h, and i).
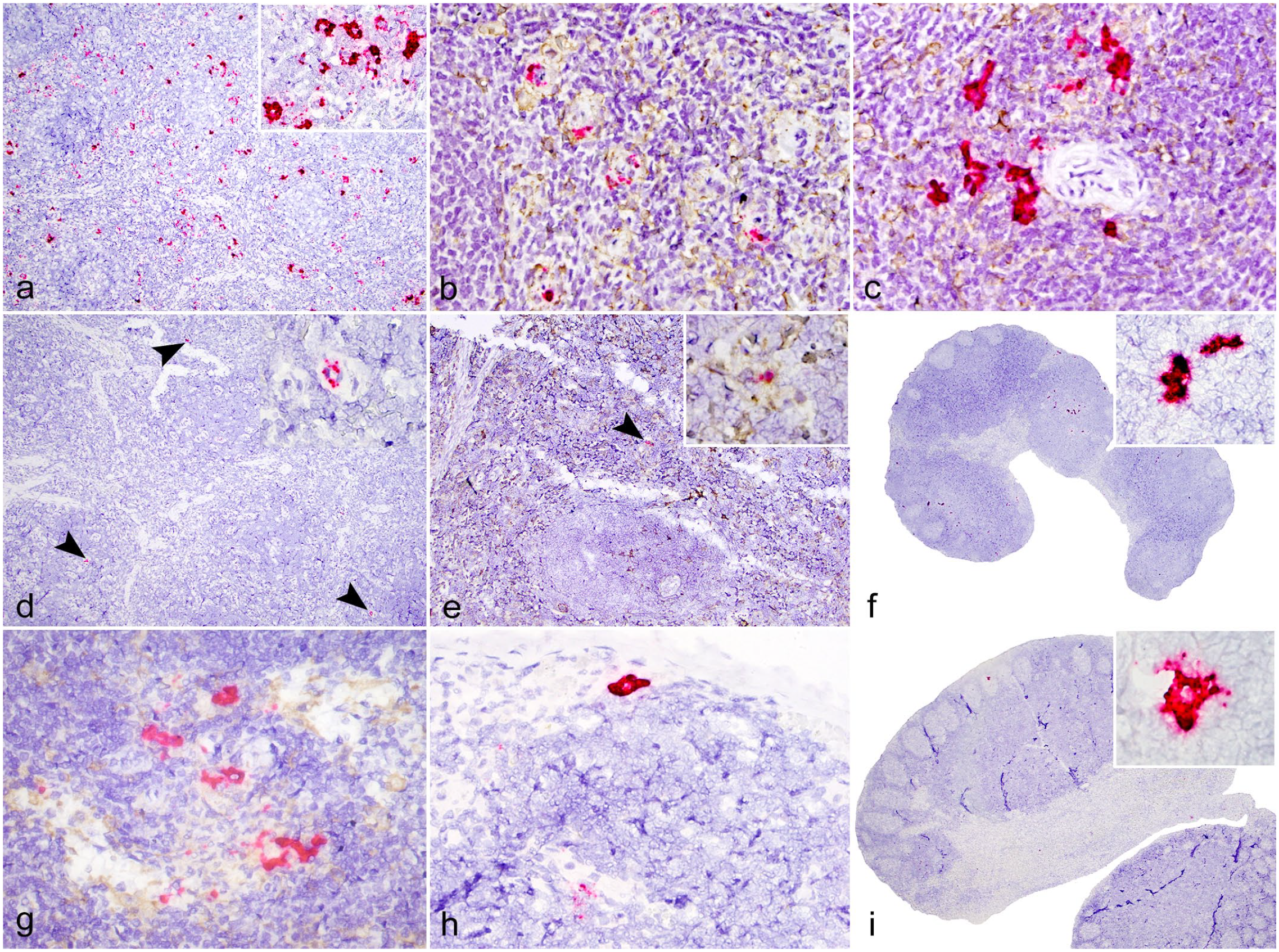
Kasokero virus (KASV) infection, secondary lymphoid tissues, Egyptian rousette bats, 3 and 6 days postinfection (DPI). RNAScope in situ hybridization (ISH) assay designed with an antisense probe targeting the KASV nucleoprotein gene. (a) Extensive positive ISH signal is distributed throughout the red pulp, spleen, 3 DPI bat. (b) Dual KASV ISH (red) and Iba1 (brown) immunohistochemistry (IHC) demonstrating multifocal colocalization of KASV with cells of the mononuclear phagocyte system and within endothelial cells of red pulp capillaries, spleen, 3 DPI bat. (c) Dual KASV ISH (red) and Iba1 (brown) IHC demonstrating multifocal colocalization of KASV with dendritiform Iba1+ cells in the white pulp, spleen, 3 DPI bat. (d) Rare KASV ISH signal is localized to the walls of scattered red pulp capillaries (arrowheads and inset), spleen, 6 DPI bat. (e) Dual KASV ISH (red) and Iba1 (brown) IHC demonstrating focal colocalization of KASV with an Iba1+ cell in the red pulp (arrowhead and inset), spleen, 6 DPI bat. (f) Multifocal KASV ISH signal throughout the paracortex and sinuses, inguinal lymph node, 6 DPI bat. (g) Multifocal positive KASV ISH signal (red) within the paracortex and medullary sinuses within Iba1+ macrophages (brown) adjacent to high endothelial venules, inguinal lymph node, 6 DPI bat. (h) Within the subcapsular sinus of an inguinal lymph node, the cytoplasm of a large macrophage or dendritic cell is saturated with KASV ISH signal (red), inguinal lymph node, 6 DPI bat. (i) Within a germinal center, the cytoplasm of a cell with follicular dendritic cell morphology is saturated with KASV ISH signal, axillary lymph node, 6 DPI bat. KASV ISH with red chromogen and hematoxylin counterstain (a, d, f, h, and i). Dual ISH-IHC for KASV nucleoprotein ISH (red) and Iba1 IHC (brown) with hematoxylin counterstain (b, c, e, and g).
Positive hybridization signal was sparse in the liver of bats euthanized at 6 DPI (Fig. 5g), where it was localized to the cytoplasm of few hepatocytes along the periphery of inflammatory foci and in the cytoplasm of scattered mononuclear phagocytes within the foci (Fig. 5h). Rare KASV RNA was detected by ISH in 2 of the 9 DPI bat livers, where it was localized to the cytoplasm of mononuclear phagocytes within the inflammatory cell foci (Fig. 5i), and rarely, within hepatocytes. No KASV RNA was detected by ISH in any of the 12 DPI bats. Of the two 18 DPI bats tested by ISH, one bat had focal positive hybridization signal in the cytoplasm of a single hepatocyte. Foci of necrosis in the two 9 DPI bats did not contain KASV RNA on the ISH assays.
Abundant KASV hybridization signal was detected in the spleen of the 3 DPI bats by ISH. Most of the ISH signal was localized to the walls of red pulp capillaries (ellipsoids). The signal varied among individual capillaries with some capillaries having 1 to 3 dots and others having saturated ISH signal with partial to complete circumferential staining of the capillary wall (Fig. 6a). Using the Iba1+ antibody to co-detect replicating KASV and mononuclear phagocytes, positive hybridization signal was observed within the cytoplasm of presumptive endothelial cells and within Iba1+ macrophages that interdigitate and cuff the endothelial cells (Fig. 6b). Viral RNA was also multifocally present within the white pulp, in the cytoplasm and branching cytoplasmic processes of Iba1+ cells morphologically consistent with macrophages or dendritic cells (Fig. 6c). KASV signal was not associated with central arterioles in the spleen. At 6 DPI, scattered Iba1+ cells containing minimal positive hybridization signal were observed in the red pulp (Fig. 6d, e). No KASV RNA was detected in the splenic white pulp of the 6 DPI bats by ISH. At 9 and 12 DPI, only 1 bat from each time point had focal positive hybridization in the red pulp. At 18 DPI, KASV RNA was not detectable in the spleens by ISH.
KASV RNA was detected by ISH in the lymph nodes for most of the KASV-infected bats at 3 and 6 DPI, and in two 12 DPI bats and one 18 DPI bat. At 3 DPI, most KASV RNA was present within Iba1+ macrophages or dendritic cells in the subcapsular, paracortical, and medullary sinuses (Fig. 6f). A smaller amount of KASV RNA was present within the paracortex and deep cortex, also in cells morphologically consistent with macrophages that were occasionally adjacent to high endothelial venules (Fig. 6g). At 6 DPI, KASV RNA was present in large mononuclear cells (macrophages and/or dendritic cells) within subcapsular sinuses (Fig. 6h) and occasionally in the germinal centers. In 1 bat at 6 DPI, the cytoplasm of a cell morphologically compatible with a follicular dendritic cell (FDC) was saturated with positive hybridization signal (Fig. 6i). Rare signal was also present within macrophages in the deep cortex and medullary sinuses. No KASV RNA was detected in any of the 9 DPI bat lymph nodes tested by ISH. Two out of 3 of the 12 DPI bats had focal positive hybridization in the paracortex. At 18 DPI, 1 bat had scattered positive signal in the germinal centers. Semiquantitative ISH scores for the liver, spleen, and lymph nodes are presented in Table 1.
Semiquantitative analysis of KASV nucleoprotein RNA, as detected by RNAScope ISH in the liver, spleen, and lymph nodes of KASV-infected Egyptian rousette bats.

Abbreviations: KASV, Kasokero virus; DPI, days postinfection; NP, not performed.
ISH was performed on inguinal lymph nodes for all bats, except for bat No. 9; multiple peripheral lymph nodes were analyzed for this individual, and ISH signal was detected in the axillary, but not in the inguinal lymph node; ISH staining was scored as follows: + = focal signal, less than 2–3 cells per 40× high-powered field (hpf; = 0.086 mm2); ++ = scattered signal in multiple foci throughout section, less than 2–3 cells per hpf; +++ = multifocal signal throughout section; 3–5 positive cells per hpf; ++++ = multifocal signal throughout section, 5–10 cells per hpf; and +++++ = multifocal signal throughout section, frequent saturated signal, 5–10 cells per hpf.
In the oral tissues, KASV RNA was detected deep within the skeletal muscle region of the body of the tongue, within the cytoplasm of few elongate interstitial cells morphologically consistent with macrophages (data not shown). Skin sections from the inoculation site of one 3 DPI and one 6 DPI bat, both of which had prominent histiocytic inflammatory cell infiltrates, were negative for KASV by ISH.
Quantitative Analysis of KASV Hybridization Signal in the Liver and Spleen
The average KASV nucleoprotein gene signal peaked in the liver at 3 DPI (%PP = 0.269%) and decreased 15-fold from 3 to 6 DPI (Fig. 7). The 3 and 6 DPI %PP values correlated with the viral loads obtained via qRT-PCR for these time points (Spearman’s r = 0.81; P = .036). After 6 DPI, KASV RNA hybridization signal was minimally detectable by ISH (<0.01 %) in a subset of the 9, 12, and 18 DPI bat livers (2/3, 1/3, and 1/2, respectively), and was undetectable by ISH/digital image analysis in the remaining 18 DPI bat. KASV RNA was not detected in any of the control bat livers (n = 3).
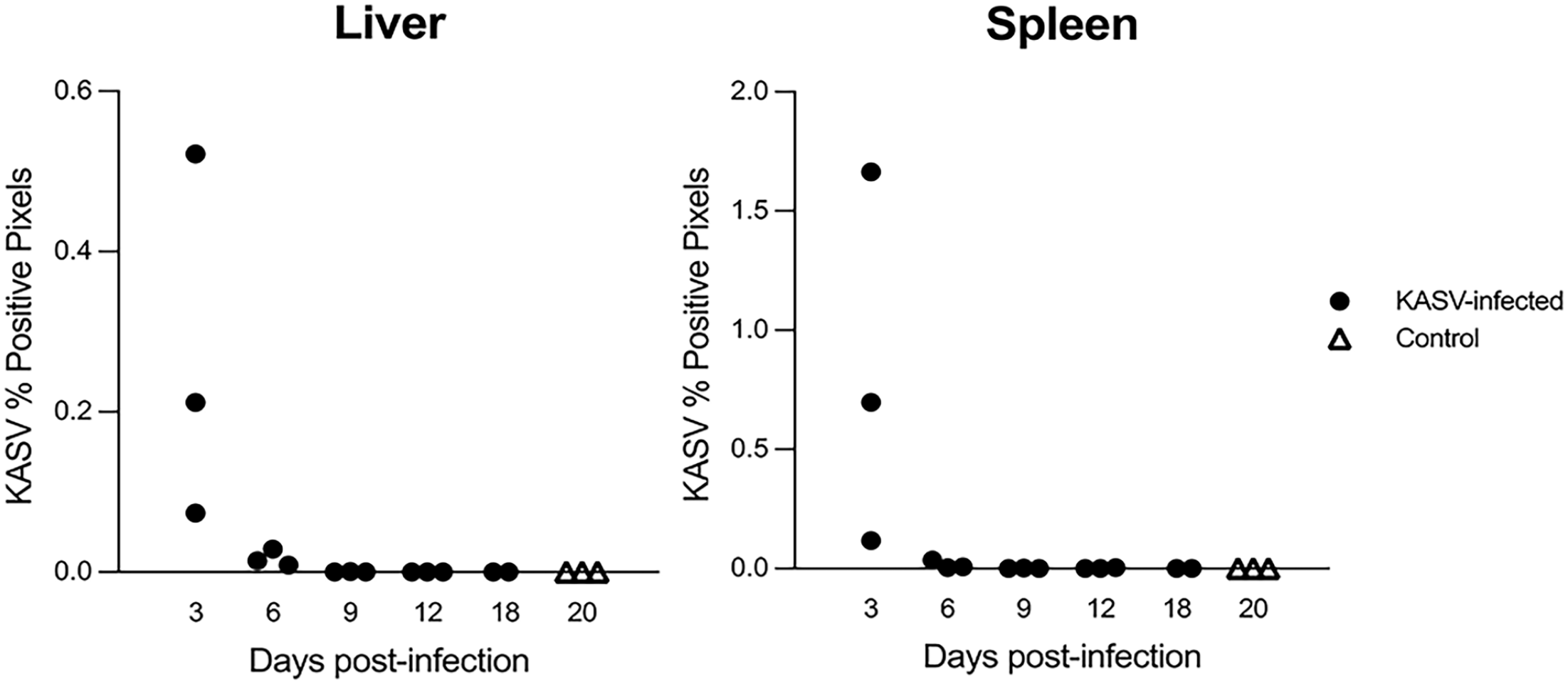
Quantification of KASV in situ hybridization signal in the liver and spleen of KASV-infected Egyptian rousette bats at 3, 6, 9, 12, and 18 days postinfection (DPI) as a percentage of total tissue area using digitized slides. Negative control bats were euthanized at 20 DPI. Each dot represents an individual bat (n = 3 per time point except 18 DPI [n = 2]). KASV = Kasokero virus.
In the spleen, the average %PP also peaked at 3 DPI with an average %PP of 0.826% (Fig. 7). The average %PP decreased 52-fold from 3 to 6 DPI. At 3 and 6 DPI, the %PP values correlated with the qRT-PCR values previously reported for each bat (Spearman’s r = 0.78; P = .032). 29 KASV RNA signal was minimally detectable by ISH at 6 DPI, with all bats having %PP values < 0.03 (2of 3 bats were < 0.001). KASV RNA was undetectable by ISH in 2 of 3 of the 9 DPI bats, and in the 18 DPI bats tested (2 of 2). KASV RNA was not detected in the control bat spleens (n = 3) by ISH.
Discussion
KASV is an emerging zoonotic bunyavirus capable of causing severe systemic disease in humans, which is thought to be maintained in an enzootic transmission cycle involving ERBs and the argasid tick O. (R.) faini.20,29,30 Experimentally infected ERBs remained asymptomatic, and KASV-induced tissue damage was limited to the liver, where lesions were consistent with acute viral hepatitis. 29 Hepatic and splenic viral replication, as detected by ISH, peaked at 3 DPI, and the vast majority of KASV RNA was cleared from both tissues by 6 DPI. In the liver at 3 DPI, hepatocellular KASV replication was associated with cytopathic effects, cell death, mononuclear inflammatory cell recruitment, hemorrhage, and necrosis in a subset of infected bats. At 6 DPI, the lymphohistiocytic hepatitis peaked, yet little KASV RNA remained, indicating that the immune response and tissue damage may contribute to hepatocellular cytopathic features (cytoplasmic inclusions and cell death). Hepatocellular cytoplasmic inclusions in one 3 DPI and two 6 DPI bats likely reflect degenerative changes in hepatocytes, as opposed to viral inclusions. Remarkably, the liver lesions were largely resolved by 20 DPI, highlighting the ability of this bat species to mount a rapid and localized immune response in this tissue following experimental KASV infection.
In comparison with studies in which ERBs were infected with MARV using very similar protocols, the liver damage and immune cell response was similar in character (ie, mononuclear immune cells), but of increased severity for KASV.18,19 The ISH data demonstrate that KASV is hepatotropic, a feature shared by several bunyaviruses of public health and veterinary importance including CCHFV, NSDV, and Rift Valley fever virus.9,16,28,31 The observed minor tropism for presumptive endothelial cells and Kupffer cells may promote more advanced liver disease due to direct and/or indirect effects of viral infection. Endotheliotropism and subsequent damage to the hepatic vasculature may also predispose to ascending bacterial infection from the gastrointestinal tract, as rare bacilli were observed in a 9 DPI bat with hepatic necrosis. Hepatocellular glycogen depletion suggests that KASV infection may be associated with increased metabolic disturbances compared with MARV. 18 Whether, and to what extent, the 3 KASV-infected bats with hepatic necrosis would have proceeded to develop clinically significant liver disease at later postinfection time points remains uncertain.
Quantitative analysis of the ISH slides permitted evaluation of virus clearance from the liver and spleen within a spatial context. The scarcity of viral RNA at 6 DPI indicates that the host responses are effective at controlling and clearing KASV early in the course of infection. Replicating KASV frequently co-localized with cells of the mononuclear phagocyte lineage in the liver, spleen, and lymph nodes, suggesting a prominent role for this diverse cell lineage in the dissemination of virus, in driving the innate and adaptive immune response(s), in the clearance of infected cells from the blood and liver, and in resolution of the liver lesions. KASV RNA was detected in the cytoplasm of a cell with FDC morphology in the axillary lymph node of 1 bat at 6 DPI. FDCs are long-lived, mesenchymal-origin cells that shape the antibody response and have been shown to harbor antigens for long periods of time.6,21,37
In ERBs, splenic red pulp endothelial cells and macrophages form small caliber, thick-walled capillaries, termed ellipsoids. 15 Extensive hybridization signal within and surrounding the capillary walls, coupled with the lack of corresponding cytopathic effects, suggests that these structures contribute significantly to the clearance of viral RNA from the blood between 3 and 6 DPI, thereby limiting spread to other tissues.
Our spatial, quantitative, and temporal analyses underscore the importance of the liver and spleen in the early host responses to KASV infection and shed light on the variety of mechanisms by which these tissues may function to control viral replication and clear viral RNA. ERB host responses to KASV may, in part, be generalized responses that are effective at controlling other bunyaviruses or RNA viruses with shared tissue tropisms such as MARV. The increased pathogenicity of this virus in ERBs compared with MARV, a host-virus relationship for which mechanisms of disease tolerance are hypothesized to limit tissue damage caused by both host and pathogen, make it an attractive experimental model for studying ERB immunology and virus-bat host interactions.14,24,26 The data presented herein suggest that early and efficient viral clearance may be a critical antiviral defense mechanism in some bats such as ERBs, which host several high-consequence zoonotic viruses, but do not themselves become sick from these viruses.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231158076 – Supplemental material for Pathogenesis of Kasokero virus in experimentally infected Egyptian rousette bats (Rousettus aegyptiacus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231158076 for Pathogenesis of Kasokero virus in experimentally infected Egyptian rousette bats (Rousettus aegyptiacus) by Shannon G. M. Kirejczyk, Amy J. Schuh, Jian Zhang, Brian R. Amman, Jonathan C. Guito, Tara K. Sealy, James C. Graziano, Corrie C. Brown and Jonathan S. Towner in Veterinary Pathology
Footnotes
Acknowledgements
We thank Jennifer Kempf, Craig Dixon, Nicole Young, and Amanda Peppers of the UGA Histology laboratory for the beautiful H&E sections and Nicole Nemeth and Nicole Gottdenker for critical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the University of Georgia, the Centers for Disease Control and Prevention, and in part by DTRA grant HDTRA1033037. The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention or the Department of Health and Human Services.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
