Abstract
Survivin is reexpressed in most human breast cancers, where its expression has been associated with tumor aggressiveness, poor prognosis, and poor response to therapy. Survivin expression was evaluated in 41 malignant canine mammary tumors (CMTs) by immunohistochemistry, in relation to histological grade and stage, and correlated with that of some related molecules (β-catenin, caspase 3, heat shock proteins) to understand their possible role in canine mammary tumorigenesis. An increase in nuclear survivin expression, compared with healthy mammary glands, was observed in CMTs, where nuclear immunolabeling was related to the presence of necrosis. No statistically significant relation was found between the expression of the investigated molecules and the histological grade or stage. The present study may suggest an important involvement of survivin in CMT tumorigenesis. Its overexpression in most of the cases evaluated might suggest that targeting survivin in CMTs may be a valid anticancer therapy.
Even though the molecular events that precede the development of canine mammary tumors (CMTs) are still poorly understood, a growing body of data exists on the molecules and the pathways that seem to be involved in their pathogenesis and are suggested as targets for anticancer therapy. Survivin is a unique member of the inhibitor of apoptosis protein (IAP) family. It is ubiquitously expressed during tissue development but absent or expressed at low levels in most normal tissues. 1 Survivin has been demonstrated to be reexpressed in most human cancers, including human breast cancers, 13 and its expression has been associated with tumor aggressiveness, poor prognosis, and poor response to therapy. 25 However, contrasting results regarding the correlation between its subcellular localization and a favorable or poor prognosis have been observed. 13,21 Such incongruity is probably related to the existence of different isoforms of survivin with different, even opposite, functions 24 or because of different methods to evaluate survivin. 30 In human breast cancer, an interesting new cascade axis from ErbB2 to survivin has been proposed via β-catenin/TCF4, clearly indicating survivin as a target gene of the β-catenin pathway. 31 Survivin is known to be a client protein of heat shock protein (Hsp) 90, physically interacting with Hsp90, forming the “survivin-Hsp90 complex.” 1 Treatments with Hsp90 inhibitors are able to reduce survivin expression, interfering with proper binding of survivin to Hsp90 and leading to its misfolding, ubiquitination, and proteasome degradation. 6 Furthermore, inhibition of Hsp72 has been shown to downregulate survivin and suppress tumorigenesis of Her2-mediated breast cancer. 18
To our knowledge, no data have been published on survivin expression in CMTs. The present study aims at evaluating survivin expression in a series of malignant CMTs, compared with normal mammary tissue, and correlating its expression with some important histological parameters, including grade and stage. Then, we investigated the possible existence of the relation between the expression of survivin and that of some related molecules (β-catenin, caspase 3, heat shock proteins), to understand their mutual interaction as well as their possible role in canine mammary tumorigenesis.
Materials and Methods
The study was conducted with 45 tissue samples, 41 of which were CMTs submitted after surgical excision. Fresh samples were fixed in 10% neutral buffered formalin and embedded in paraffin wax. Sections were stained by hematoxylin and eosin (HE). The CMTs were classified according to the criteria of the World Health Organization 19 and Goldschmidt et al 11 by three pathologists (M.R., L.B., and L.D.S.). Four samples of healthy canine mammary tissue were collected immediately after death from bitches submitted for necropsy examination that did not show any mammary gland or reproductive tract abnormality (patient information, along with histotype, grade, stage, and histological features, is summarized in Suppl. Table S1).
Histological Examination
Each tumor was evaluated on the basis of the presence of necrosis and mode of growth (expansile vs infiltrative). Grade was assessed according to Misdorp et al 19 and considering the invasion score (histologic staging), based on the classification proposed by Gilbertson et al. 10 Mitotic index (MI) was evaluated morphologically in HE-stained sections by counting the number of mitoses in 10 high-power fields (HPF, 40×) shifted randomly. The number of mitoses was scored as follows: 0, no mitotic cells; 1, 0 to 1 per 10 HPF; 2, 1 to 2 per 10 HPF; 3, 2 to 3 per 10 HPF; 4, 3 to 4 per 10 HPF; 5, 4 to 5 per 10 HPF; 6, 5 to 6 per 10 HPF; 7, 6 to 7 per 10 HPF; and 8, 7 to 8 per 10 HPF.
Immunohistochemistry
Dewaxed and rehydrated tissue sections were immunostained by means of the streptavidin-biotin peroxidase complex (SAB) method, using specific primary antibodies (Abs) (Table 1), incubated overnight in a humidified chamber at 4°C. Immunohistochemical analysis followed such procedure as reported in previous studies. 4 Endogenous peroxidase was blocked with 3% hydrogen peroxide in absolute methanol for 45 minutes. Antigen retrieval for inactive (CPP32) and active caspase 3, β-catenin, and Hsp27, 72, 73, and 90 was undertaken by heat-treating sections in citrate buffer at pH 6 in a microwave oven for 5 minutes (3 cycles) and in a pressure cooker for 20 minutes for survivin. To reduce nonspecific binding, slides were incubated in normal goat serum (Biospa, Milan, Italy) for 10 minutes at room temperature before overnight incubation with the primary Abs in a humidified chamber at 4°C. Slides were treated with secondary biotinylated goat anti–mouse + rabbit (ready to use; Biospa) or rabbit anti–rat (1:100; DAKO, Copenhagen, Denmark) antibody and detected with streptavidin-peroxidase (Biospa), incubated at room temperature for 10 minutes. Sections were visualized with 3,3′-diaminobenzidine (DAB, D5905; Sigma, St Louis, MO) solution, which was applied for 5 minutes, and finally lightly counterstained with Mayer’s hematoxylin for 2 minutes. A negative control was performed in all instances by omitting the primary antibody and incubating tissue sections with Tris-buffered saline (TBS) or with an irrelevant antibody directed against unrelated antigens such as rabbit anti–human von Willebrand factor polyclonal Ab or mouse anti–human desmin monoclonal Ab (DAKO, Glostrup, Denmark). For each antibody, sections of a tissue with known antigen expression were used as a positive control (Table 1). The quantification of immunostaining was performed by 2 pathologists who scored samples as follow. Active caspase 3 expression was evaluated by counting the number of positive nuclei in 10 HPF at 400×, counting approximately 1000 cells, and expressed as a percentage. β-Catenin expression was classified as membrane (localized at cell-cell boundaries), cytoplasmic (uniformly distributed throughout the cytoplasm), and nuclear. Survivin and heat shock protein expression was evaluated as cytoplasmic and/or nuclear. A semiquantitative immunohistochemical assessment was performed for β-catenin; pro–caspase 3; Hsp90, 27, 72, and 73; and survivin. Samples were subdivided in accordance with the protein expression levels in 5 ranges (absent, no positive cells; low, >0% to <10% positive cells; moderate, ≥10% to <50% positive cells; high, ≥50% to <75% positive cells; very high, ≥75% positive cells). A statistical analysis was performed using a Fisher exact test and Cramer’s V to evaluate the association between the expression levels of the investigated molecules and the clinicopathological parameters (grade, stage, necrosis, type of growth, and mitoses). For this purpose, all cases were grouped according to the expression score as follows: <10% positive cells (absent + low semiquantitative evaluation) vs ≥10% positive cells (moderate + high/very high semiquantitative evaluation) for nuclear survivin, Hsp27, and Hsp72; <25% positive cells (absent + low semiquantitative evaluation) vs ≥25% positive cells (moderate + high/very high semiquantitative evaluation) for pro–caspase 3, active caspase 3, and membrane and cytoplasmic β-catenin; and <50% positive cells (absent/low + moderate semiquantitative evaluation) vs ≥50% positive cells (high/very high semiquantitative evaluation) for Hsp90 and Hsp73. Analyses were performed using SPSS version 19 statistical software (SPSS, Inc, an IBM Company, Chicago, IL), and the conventional 5% levels were used to define statistical significance.
Primary Antibodies Used in the Immunohistochemical Analysis.
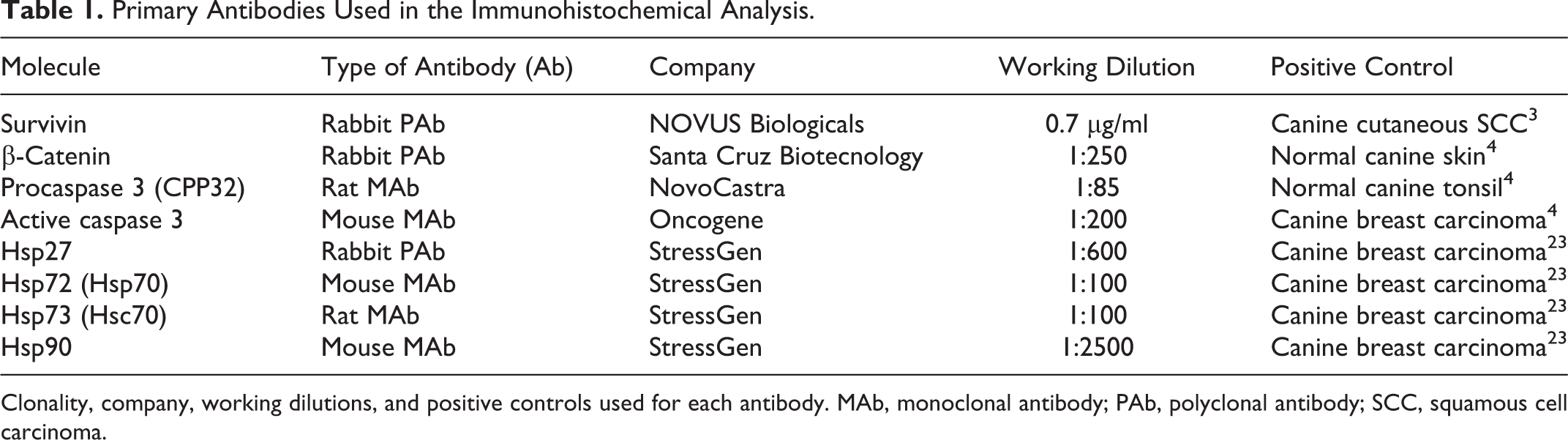
Clonality, company, working dilutions, and positive controls used for each antibody. MAb, monoclonal antibody; PAb, polyclonal antibody; SCC, squamous cell carcinoma.
Results
Forty-one CMTs were examined and classified as follows: 16 tubulopapillary carcinomas, 11 complex carcinomas, 6 solid carcinomas, 5 carcinomas in a benign (mixed) tumor, 1 inflammatory carcinoma, 1 spindle cell carcinoma, and 1 carcinosarcoma. CMTs were classified by grade and histological stage as reported in Supplemental Table S1. Necrosis was observed in 27 of 41 cases (65.9%), while infiltrative growth was present in approximately half of the cases (21/41 [51.2%]). Mitotic index ranged from 0 to 8, but most of the CMTs evaluated (21/41 [52.5%]) showed a low mitotic index (0–1) (the results are summarized in Suppl. Table S1).
Immunohistochemical Evaluation
The most important immunohistochemical results are summarized in Supplemental Table S2. In healthy mammary gland, survivin cytoplasmic staining was absent, while nuclear survivin expression was absent or low (<10%). Positive cells (10%–25%) were observed within the hyperplastic foci found in a gland from healthy dog, as well as surrounding tumors. Myoepithelial cells were always negative. An increase of survivin immunolabeling was evident in most of the primary CMTs (Fig. 1 and Suppl. Table S2), showing a mainly nuclear localization (increased nuclear survivin was observed in 69.2% of cases), with positive mitotic figures particularly evident among infiltrating cells. Positive cells were found in lymphatic neoplastic emboli (Fig. 2). Proliferating myoepithelial cells of complex carcinomas were negative.
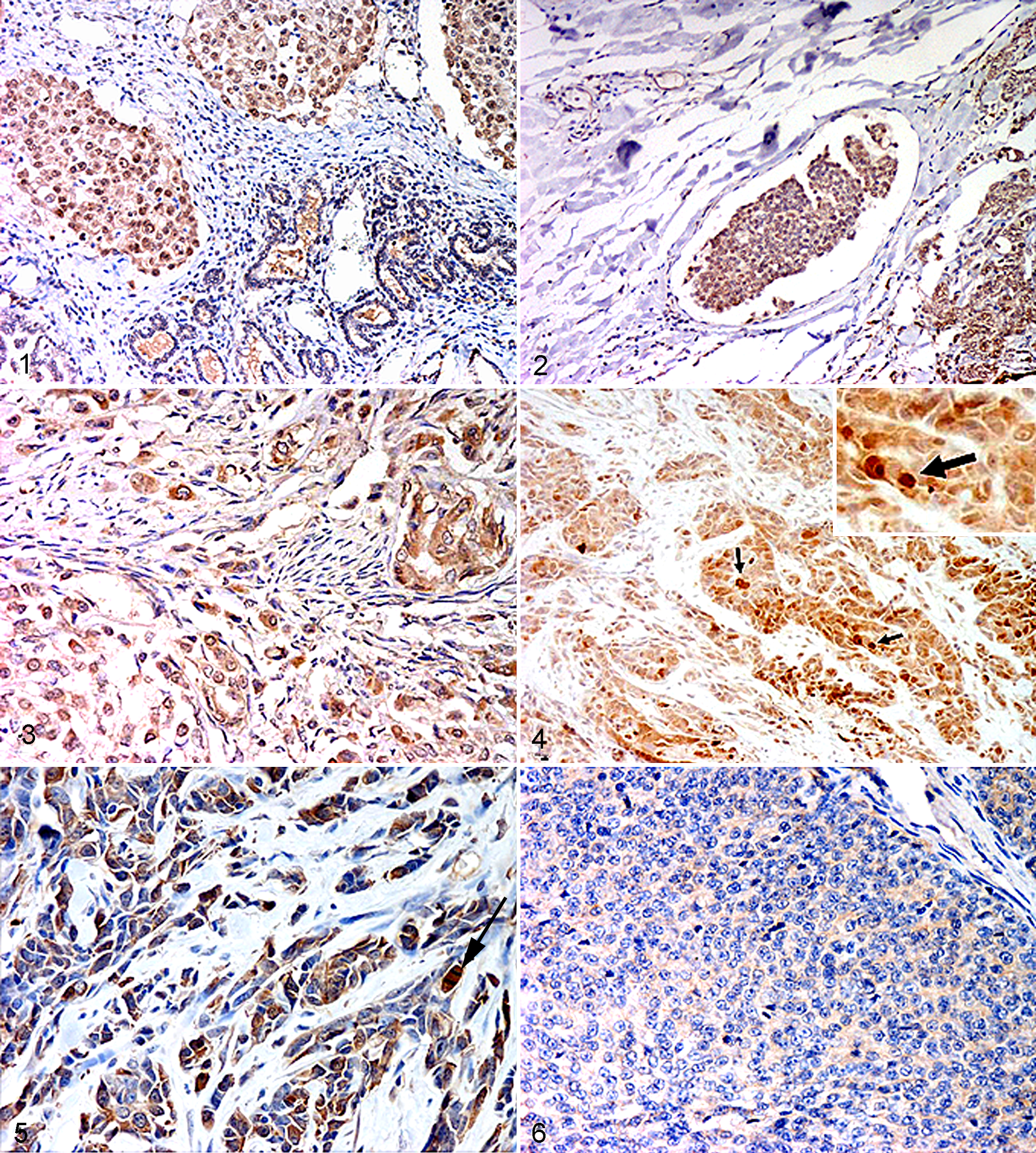
No immunostaining for Hsp27 was observed in 75% of normal mammary gland tissue samples. In the remaining 25% (1 sample), the immunolabeling was faint, cytoplasmic, and in acinar cells but never in ductular structures and myoepithelial cells. Expression was faint or moderate in most cases analyzed (87.5%) but intense in single or small clusters of neoplastic cells infiltrating surrounding tissues (Fig. 3) and in neoplastic emboli found within lymphatic vessels. Cytoplasmic staining was observed in proliferating myoepithelial cells in complex carcinoma cases as well as in metaplastic chondrocytes of carcinomas in benign (mixed) tumors.
Immunohistochemical expression of Hsp72 was faint or moderate and inconsistent, multifocal, mainly cytoplasmic, and occasionally present in the nucleus of epithelial cells of alveolar structures. No immunostaining was observed in ducts and myoepithelial cells. Increase of the expression levels was observed in 67.5% of the primary CMTs, compared to normal glands, with both cytoplasmic and nuclear staining. Nuclear staining was observed mainly in the neoplastic cells close to necrotic areas, as well as in infiltrating tumor cells (Fig. 4). Intense and constant immunoreactivity was observed in the lymphatic neoplastic emboli.
In normal mammary gland, intense and diffuse Hsp73 immunostaining was found, mainly in the acinar component of the gland, and was inconstant in ductular structures and negative in myoepithelial cells. In 95% of the primary CMTs, a reduction of Hsp73 expression was observed compared with normal tissue. However, the highest cytoplasmic and nuclear immunohistochemical expression was observed in infiltrating tumor cells. Proliferating myoepithelial cells of complex carcinomas were negative or faintly positive. Lymphatic neoplastic emboli showed a constant and intense immunoreactivity.
In healthy mammary gland, Hsp90 immunoreactivity was cytoplasmic, intense, and diffuse in lobular structures while moderate and inconstant among the ductular component and myoepithelial cells. Most of the primary CMTs (95%) showed similar or increased nuclear and cytoplasmic immunolabeling compared with normal tissue, with positive mitotic figures (Fig. 5). In complex carcinomas, proliferating myoepithelial cells showed diffuse, moderate to intense staining. In lymphatic neoplastic emboli, intense and constant immunoreactivity was observed.
Intense membrane and cytoplasmic but not nuclear β-catenin immunolabeling was observed in normal mammary gland, in both acinar and ductular structures. Reduced expression of cytoplasmic and membranous β-catenin was found in most of the primary CMTs (83.7% and 91.9%, respectively) (Fig. 6). Nuclear localization of the molecule was observed in only 3 CMTs (1 grade II and 2 grade III). In complex carcinomas, proliferating myoepithelial cells showed cytoplasmic staining.
Pro–caspase 3 was expressed in all cases examined, both in healthy mammary gland and CMTs (comprising neoplastic emboli), except for a tubulopapillary carcinoma, grade II, stage I. No statistically significant relations were observed among pro–caspase 3 expression and the clinicopathological parameters. The active form of the caspase 3 was expressed in 1 case of normal mammary gland. All the CMTs showed caspase 3 nuclear immunolabeling in neoplastic cells and in neoplastic emboli, as well as among inflammatory and mesenchymal cells. Proliferating myoepithelial cells of complex carcinomas were always negative.
Statistical Analysis
An increase of nuclear survivin expression was observed by comparing CMTs with healthy mammary glands, although it appeared not significant, most probably due to the small sample of healthy dogs. The significant results of the statistical analysis are summarized in Table 2.
Most Important Results of the Statistical Analysis.
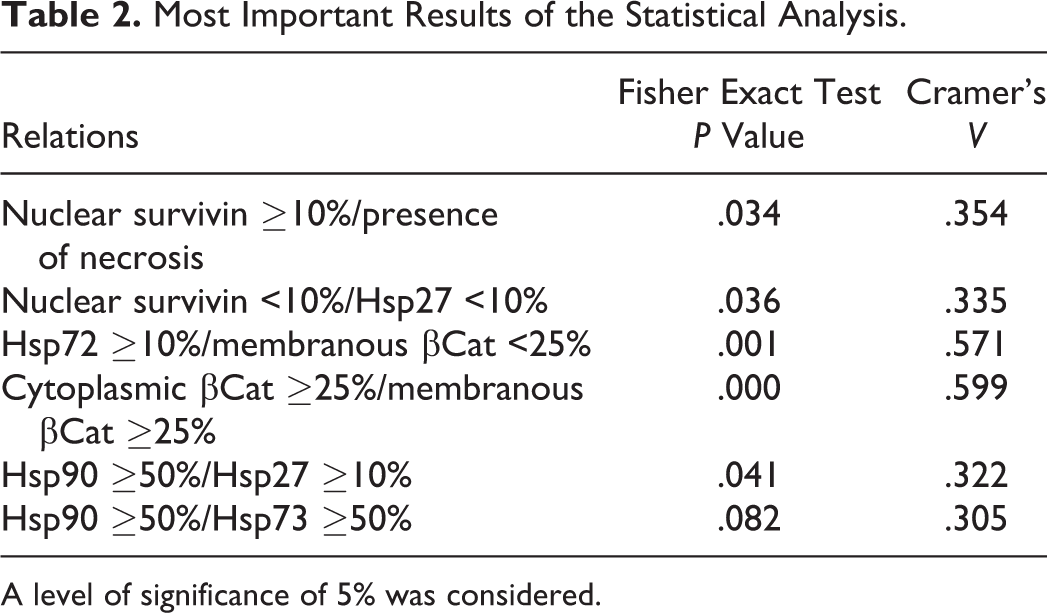
A level of significance of 5% was considered.
Discussion
We did not observe any cells expressing survivin among normal mammary gland cells. This is similar to previous results reported in human breast samples. 26 Survivin overexpression observed in most malignant mammary tumors compared with normal tissue clearly indicates the involvement of the molecules in mammary gland neoplastic transformation in dog, as proposed in human breast cancer cases. 13 However, survivin expression failed to correlate with tumor grade or stage, even if a statistically significant correlation was found between a higher nuclear survivin expression and the presence of necrosis, which is considered a histologic feature of malignancy in CMTs. 11,20 The cytoplasmic immunostaining of the molecule was diffusely expressed in most cases evaluated. Interestingly, contrasting results have been observed in human breast cancer concerning the relation between survivin subcellular localization and a favorable or poor prognosis. 13,21 A high cytoplasmic-to-nuclear ratio of survivin has been proposed as well as an indicator of improved prognosis, 22 further validating the hypothesis that the nuclear and cytoplasmic fractions of survivin have different biological roles. 1 Our data could agree with the contrasting results published in human breast cancer concerning the prognostic significance of survivin, 13 which could be related, besides the use of different methods of survivin evaluation, 30 to the existence of different isoforms of survivin with different, even opposite functions as well. 24 Although survivin is mainly an antiapoptotic and cell cycle regulator molecule, 1 the existence of different survivin isoforms (which were not distinguished in the present study) may also explain, at least in part, the lack of detection of a correlation between survivin immunolabeling and apoptotic molecules (caspase 3, inactive and active forms) or mitotic index in our cases. A statistically significant correlation was found between nuclear survivin and Hsp27 expression. Even if a direct link between survivin and Hsp27 has not been demonstrated so far, the correlation between Hsp27 and nuclear–cell cycle regulator survivin is not surprising. In fact, phosphorylation of Hsp27, which could take place as a consequence of different stimuli in cancer cells, may play multiple roles, inducing neoplastic cells through the checkpoints (cell survival pathway) and proliferation. These data seem also to agree with a previous study where the Hsp27 overexpressed, observed in CMT samples compared with normal tissue, was related to a shorter survival time. 23 Infiltrating neoplastic cells often showed an intense immunoreactivity for Hsp27, as well as for survivin. Hsp27 overexpression has been strictly correlated with the invasiveness of human breast cancer cells, and a possible influence of the molecule in the metastatic potential of these cells has been demonstrated. 15 A role of Hsp27 recently has been proposed in breast cancer stem cells, where Hsp27 functions seem to be Akt independent: it could regulate the maintenance and tumorigenicity of these cells through the epithelial-mesenchymal transition (EMT), and nuclear factor–κB 29 and may play a role in the drug-resistant character of breast cancer stem cells. 14 Similarly, a further interesting role of survivin in tumors has been recently speculated on the potential role it plays in EMT, as well as in cancer stem cells, thus contributing to tumor spread (metastasis), resistance to treatment, and disease relapse. 3 Its expression by infiltrating tumor cells and metastatic neoplastic cells within vessels observed in the cases evaluated herein appeared to be consistent with these functions.
The complexity of survivin regulation in cancer cells, both at transcriptional and posttranscriptional levels, could explain the lack of correlation we found between its expression levels and some known survivin-related molecules, such as Hsp90 and β-catenin, in CMTs. Survivin binds to Hsp90 in cells and is therefore considered an Hsp90 client protein. 1 However, survivin is only one of the Hsp90 client proteins involved in mammary tumorigenesis, and thus it is not surprising that its expression may not be correlated with survivin immunoreactivity. On the other hand, the significant relation between Hsp90, Hsp27, and Hsp73 expression found in the CMTs evaluated appears to be interesting, in light of the results reported in a recent study demonstrating that a combination of inhibition of Hsp90, Hsp27, and Hsp70 in human breast cancer cells is able to inhibit cell proliferation and migration. 14 No correlation was observed between nuclear survivin and β-catenin immunolabeling. Indeed, nuclear β-catenin immunostaining, indicative of the activation of the Wnt/β-catenin pathway, 7 was observed only in 3 cases (1 grade II and 2 grade III), thus confirming previous results reported in human breast cancers. 8 On the other hand, a significant relation between cytoplasmic and membranous β-catenin expression was observed, both of which were reduced in most of the CMTs evaluated. This could be due to E-cadherin or β-catenin mutations, but also to a loss of other Wnt components, such as Wnt-5a (noncanonical Wnt pathway), as reported in human invasive breast cancers. 17 Reduction or loss of membranous and cytoplasmic β-catenin expression has been demonstrated to be important in tumor progression in human patients affected by breast cancer, where low membrane expression of β-catenin was shown to be associated with poor outcome, 9 in agreement with studies previously conducted on CMTs, where reduced E-cadherin/β-catenin expression influenced invasion of CMTs but not their proliferation or survival. 5 Furthermore, even though the relationship between Hsp70 and β-catenin expression has not been investigated in mammary tumors so far, the statistically significant inverse correlation between Hsp72 and the membranous β-catenin immunosignal observed in our cases appears to be interesting in light of the predominant membranous expression of β-catenin detected in colon cancers from Hsp70 knockout mice, 28 thus suggesting a regulation of β-catenin localization mediated by Hsp70.
Recent works have pointed out the importance of survivin, demonstrating that its upregulation represents one of the most important mechanisms responsible for drug resistance in several types of tumor cells. 12 In breast cancer cells, survivin transcription has recently been proposed to be associated with P-glycoprotein/MDR1 overexpression. PI3k/Akt pathway is involved in P-glycoprotein/MDR1 associated survivin transcription activity in the multidrug resistant MCF-7 breast cancer cells. 16 The present data add further evidence of survivin involvement in mammary gland carcinogenesis and to the theory that inhibition of the molecule may be a viable therapeutic option. Furthermore, the immunogenic proprieties of survivin, demonstrated by the presence of autoantibodies and specific T cells against survivin in the blood of patients with breast cancer, 2 as well as of autoantibodies in dogs with mammary adenocarcinoma, 27 allow us to consider survivin a valid therapeutic target for anticancer immunotherapy.
The present study suggests a possible simultaneous involvement of survivin and Hsp27 in CMTs, which could play a role in tumor infiltration and invasion, as well as in the maintenance of the so-called cancer stem cells. Further studies are needed to better understand their exact function and possible interaction. This aspect, along with the overexpression of nuclear survivin in most CMT cases evaluated, might indicate survivin as a possible valid therapeutic target in the anticancer therapy of CMTs.
Footnotes
Acknowledgement
We thank Prof. Francesca Rosati, University of Teramo, for her support in editing the English text.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
