Abstract
High survivin expression has been correlated with poor outcomes in several canine tumors but not in soft tissue tumors (STTs). Survivin is a target gene of the Wnt/β-catenin pathway, which is involved in human STT oncogenesis. Immunohistochemistry for survivin, β-catenin, and Ki-67 was performed on 41 canine perivascular wall tumors (cPWTs), and statistical associations of protein expression and histopathologic and clinical variables with clinical outcomes were investigated. Immunohistochemically, there was nuclear positivity (0.9%–12.2% of tumor cells) for survivin in 41/41 (100%), cytoplasmic positivity (0 to > 75% of tumor cells) for survivin in 31/41 (76%), nuclear positivity (2.9%–67.2% of tumor cells) for β-catenin in 24/41 (59%), and cytoplasmic positivity (0% to > 75% of tumor cells) for β-catenin in 23/41 (56%) of cPWTs. All tumors expressed nuclear Ki-67 (2.2%–23.5%). In univariate analysis and multivariate analysis (UA and MA, respectively), every 1% increase of nuclear survivin was associated with an increase of the instantaneous death risk by a factor of 1.15 [hazard ratio (HR) = 1.15; P = .007]. Higher nuclear survivin was associated with grade II/III neoplasms (P = .043). Expression of cytoplasmic survivin, nuclear and cytoplasmic β-catenin, and nuclear Ki-67 were not significantly associated with prognosis in UA nor MA. Tumor size was a significant prognostic factor for local recurrence in UA [subdistribution HR (SDHR) = 1.19; P = .02] and for reduced overall survival time in MA. According to UA and MA, a unitary increase of mitotic count was associated with an increase of the instantaneous death risk by a factor of 1.05 (HR = 1.05; P = .014). Nuclear survivin, mitotic count, and tumor size seem to be potential prognostic factors for cPWTs. In addition, survivin and β-catenin may represent promising therapeutic targets for cPWTs.
Keywords
Canine soft tissue tumors (STTs) are a heterogeneous group of neoplasms that are named according to their histogenesis, which include fibrous, adipocytic, vascular and perivascular, skeletal and smooth muscle, mesothelial, synovial, and nerve sheath tumors (NSTs). 78 However, many canine STTs share morphological features and growth patterns. 78 Because of this, and to increase the number of enrolled cases, most prognostic studies of canine STTs have been influenced by a “one size fits all” approach, which has led researcher to merge different STTs subtypes in the same study population, resulting in a potential bias in the assessment of biological behavior and prognostic factors.8,18,27,30,49,57,58,83 Unfortunately, studies addressing the biological behavior of each STT subtype by determining the probability of recurrence and/or metastasis, disease-free intervals, and survival times in relation to homogeneous tumor stages and treatment groups are rare.9,41 This information is necessary to correlate specific tumor subtypes and their biomarkers with tumor behavior, more accurately reflect their clinical outcomes, and establish a specific “standard of care” therapy. Thus, there is the need for studies on specific subtypes of canine STTs. STTs characterized by whorling and nonspecific fasciculated growth patterns are common in dogs and were once diagnosed as hemangiopericytomas (HEPs). This misnomer was attributed to these entities because those histologic features were described in human HEPs, even though they were not the main or the most specific features of HEP. 7 Subsequently, human HEP was reclassified, and currently, this diagnosis in dogs is limited to rare tumors with a staghorn vascular growth pattern. 76 In veterinary medicine, the cell of origin of the “old HEP” has been under debate for a long time: some authors have suggested a perivascular origin, while others a perineural one. 7 The latter hypothesis derived from difficulties in differentiating the “old HEP” from NSTs. 7 However, the perivascular nature of these tumors was clarified by documenting the specific perivascular growth patterns, including whorls around capillaries by histology and electron microscopy, and by their myoid phenotype demonstrated by immunohistochemistry (IHC), features not shared by NSTs.7,68,89 In order to better reflect the origin of these tumors, the term perivascular wall tumor (PWT) was considered most suitable and replaced the term HEP. 7
Several studies have demonstrated that canine perivascular wall tumors (cPWTs) represent a form of canine STT that can recur locally and rarely metastasizes.4,7,21,92 In a recent investigation performed on 102 cPWT cases (the largest study available in the current literature), the risk of developing local recurrence (LR) was 27% at 2 years, and metastases were reported in 10/102 dogs (9.8%), between 60 and 1060 days. 22 However, most pulmonary metastases in this study were suspected based on diagnostic imaging and not confirmed by cytology or histology. cPWTs represent the most common subtype of canine STTs. 35 Indeed, in a tumor survey from a total of 11,740 skin tumors collected in 5 years, there were 1278 STTs, and of these, cPWTs represented the most frequent tumor type, totaling 536 (42% of all STTs) followed by not-otherwise-specified sarcomas (484 cases, 37.8%), and fibrosarcomas (160 cases, 12.5%), while there were only 10 cases of NST (0.8%). 35 Because cPWT is a common subtype of canine STT, 35 it is critical to better predict those dogs with cPWT that are at risk for LR, as well as identify those rare cPWTs that may develop metastatic disease.
Several studies have investigated potential clinical and histopathological prognostic factors for cPWTs.4,22,92 One study reported a high level of expression of molecules of the vascular endothelial growth factor, platelet-derived growth factor B, and basic fibroblast growth factor pathways, suggesting that inhibiting tyrosine kinase receptors could represent a possible target for postsurgical adjunctive chemotherapy for some cPWTs, 6 although no results on clinical data are currently available.
Survivin is the smallest member of the inhibitor of apoptosis protein family and acts as an apoptosis inhibitor and cell cycle regulator.70,102 Cytosolic survivin inhibits extrinsic and intrinsic apoptotic pathways through various caspase-dependent and caspase-independent mechanisms.70,102 Nuclear survivin regulates the cell cycle, including mitosis, and enhances cell proliferation.32,40,70,93,102 In addition, survivin triggers and supports angiogenesis; 31 promotes cellular migration; 29 and enhances neoplastic cells’ metastatic potential, 87 chemoresistance, 37 and radioresistance. 45 Data also suggest that survivin can inhibit autophagic death in neoplastic cells. 21
Survivin expression is negligible in most mature normal human tissues,70,102 while it is highly expressed in fetal and neoplastic tissues, being one of the most upregulated mRNAs in the human cancer transcriptome.70,99,102 Due to its overexpression in neoplastic cells, survivin has been proposed as a prognostic marker, and its expression often correlates with aggressive disease and poor clinical outcomes in several human cancers, including STTs.43,44,94 High survivin expression has been demonstrated in several malignancies in dogs,12 –16,26,64,71,74,81,86,90,91,103 but only a few studies have investigated its correlation with prognosis,14,15,73,81,86 and no studies have been conducted on canine STTs. Survivin expression and functions depend on numerous signaling pathways, and derangements in any of these can determine its aberrant expression in malignant cells.11,20,70,97,102 Therefore, survivin has been investigated in canine tumors in relation to other molecules, such as p53,15,16 BCL-2, 64 caspase-3,15,16,90 and β-catenin.14 –16 The latter is the key transcription factor of the canonical Wnt signaling pathway.25,28 Aberrant β-catenin regulation is involved in the development and progression of human colorectal carcinoma 10 and human and canine melanocytic tumors.23,33,38 However, evidence indicates that this pathway may play a role also in STT oncogenesis. 55 During Wnt/β-catenin signaling pathway upregulation in tumorigenesis, β-catenin accumulates in the cytoplasm and translocates to the nucleus, initiating transcription of target genes including c-MYC, cyclin D1, and survivin.20,46,53 In veterinary medicine, few studies have analyzed survivin and β-catenin reciprocal expression in canine tumors.14 –16
Ki-67 has been suggested as a negative prognostic factor in canine STTs since it is associated with decreased survival times in univariate analysis (UA). 30 However, no studies have demonstrated a correlation between Ki-67 immunohistochemical expression and prognosis in cPWTs, or an advantage of using this marker over the evaluation of mitotic count.
Based on the paucity of data regarding cPWTs prognostic marker expression, this study assessed the immunohistochemical expression of survivin, β-catenin, and Ki-67 in a homogenous and mono-institutional cPWT caseload, providing preliminary data on their prognostic impact. In addition, this study aimed to evaluate and confirm the prognostic role of specific histopathological and clinical variables that have already been analyzed in other cPWT studies.4,22,92
Material and Methods
Case Selection and Clinical Data
Formalin-fixed, paraffin-embedded tissue samples of cPWTs submitted for diagnostic purposes to the Pathology Service of the Veterinary Teaching Hospital of the Department of Veterinary Medicine and Animal Sciences of the University of Milan between 2001 and 2020 were retrospectively selected. cPWTs were diagnosed by histomorphology alone or by histomorphology and IHC in doubtful cases. To diagnose a cPWT, the following microscopical patterns were specifically identified in at least 40% of the analyzed tumor surface, including vascular pericapillary to periadventitial whorls, staghorn (antler-like) ramified vessels, bundles originating directly from the vascular wall, often in the subadventitial space, and placentoid patterns.5,76 When these patterns were less prevalent and associated with nonspecific growth patterns, such as bundles and epithelioid areas, tumors were diagnosed as cPWTs when alpha-smooth muscle actin (α-SMA) and/or desmin were immunolabeled (see the “Immunohistochemistry” section). 4 Cases with specific NST growth patterns, including palisades, Antoni A and B, Verocay bodies, or whorls around axons and nerve rootlet-like structures, or specific smooth muscle tumor growth patterns, including bundles radiating at irregular 90-degree angles and herringbone patterns, were excluded from the study.75,77 Other inclusion criteria applied were: (1) tumors at first presentation, (2) no lymphadenomegaly, (3) complete clinical staging (preoperative whole-body contrast-enhanced computed tomography or thoracic X-rays and abdominal ultrasound), (4) absence of distant metastatic disease (lymph nodes and distant organs) at the time of surgery, and (5) curative-intent surgery with the widest feasible excision (1–3 cm of normal tissue laterally around the grossly visible mass, and at least 1 deep fascial plane that was macroscopically not infiltrated by the tumor). In addition, exclusion criteria included administration of any neoadjuvant chemotherapy and/or any neoadjuvant or adjuvant radiotherapy. Recorded clinical data were tumor location (classified as head/neck, thorax, abdomen, proximal limb, and distant limb, with distal limb defined as below the elbow or stifle joint), presence of cutaneous ulceration, and whether the dog had undergone any adjuvant treatment after surgery.
Histopathology
All selected formalin-fixed, paraffin-embedded tissue samples were trimmed with tangential and/or cross-sectioning technique for microscopic margin assessment. During trimming, the maximum tumor diameter was measured in centimeters; 4 μm-thick sections were cut and routinely stained with hematoxylin and eosin. Cases were examined and graded independently following previously published criteria 27 by 1 board-certified pathologist (PR) and by 2 residents in training (FG and FA), and then reviewed conjunctively to reach agreement. Histologic margins were classified as tumor-free (ie, for cross-sections, all histologic tumor-free margins > 0 mm; and for tangential sections, all margin sections did not contain neoplastic cells) or infiltrated (ie, for cross-sections, at least 1 histologic tumor-free margins = 0 mm; and for tangential sections, at least 1 margin section contained neoplastic cells). The mitotic count was assessed in 2.37 mm2.60,59 Mitotic count assessment was performed in viable regions of the tumor, with greatest mitotic activity, avoiding poorly cellular, hemorrhagic, edematous, necrotic, and inflamed areas. 59 Tumor necrosis was evaluated histologically and classified as absent, ≤ 50% of the tumor, or > 50% of the tumor (microscopically assessing all available sections). Areas of necrosis were differentiated from mucinous or hyaline change, hemorrhage, and surgery-associated trauma (if any). Other recorded pathological parameters were type of growth (expansive, satellite nodules, and/or infiltrative), 4 lymphovascular invasion (present or absent), and infiltration of the underlying fascia and/or muscular layer (infiltrated or not infiltrated).
Immunohistochemistry
Information regarding antibodies and positive controls are listed in Table 1. For cases characterized by sparse cPWT-specific growth patterns, α-SMA and/or desmin immunohistochemical expression by neoplastic cells was used to confirm the diagnosis of cPWT. α-SMA, desmin, and β-catenin IHC was performed as previously described in canine (α-SMA and desmin) 4 and equine (β-catenin) 1 tissues. To support cross-reactivity of the β-catenin antibody in the dog, amino acid sequence of human β-catenin (UniProtKB-P35222) and canine β-catenin (UniProtKB-B6V8E6) were aligned on UniProt demonstrating 99.87% identity. Furthermore, the antihuman β-catenin antibody was tested on normal canine skin for cross-reactivity. The epidermal keratinocytes displayed a brown, clean signal at the level of the intercellular junctions with a membranous pattern. Antisurvivin12–16,86,103 and anti-Ki-6769,88 antibodies were previously validated in canine tissues. For these last 2 antibodies, endogenous peroxidase was quenched by incubating sections in 3% hydrogen peroxide for 10 minutes. Nonspecific binding was blocked using appropriate diluted serum for 30 minutes at room temperature. Sections were incubated with primary antibodies for 60 minutes at room temperature. Specific biotinylated secondary antibodies were subsequently added for 30 minutes at room temperature. Slides were then incubated for 30 minutes at room temperature with an avidin-biotin complex kit, and the reaction was developed using 3,3′-diaminobenzidine (DAB) chromogen. Sections were counterstained with Mayer’s hematoxylin and mounted. Appropriate positive controls were used (Table 1). Negative controls for each sample were prepared by omitting the primary antibody.
Details of primary antibodies, antigen retrieval, and positive controls used for immunohistochemistry.
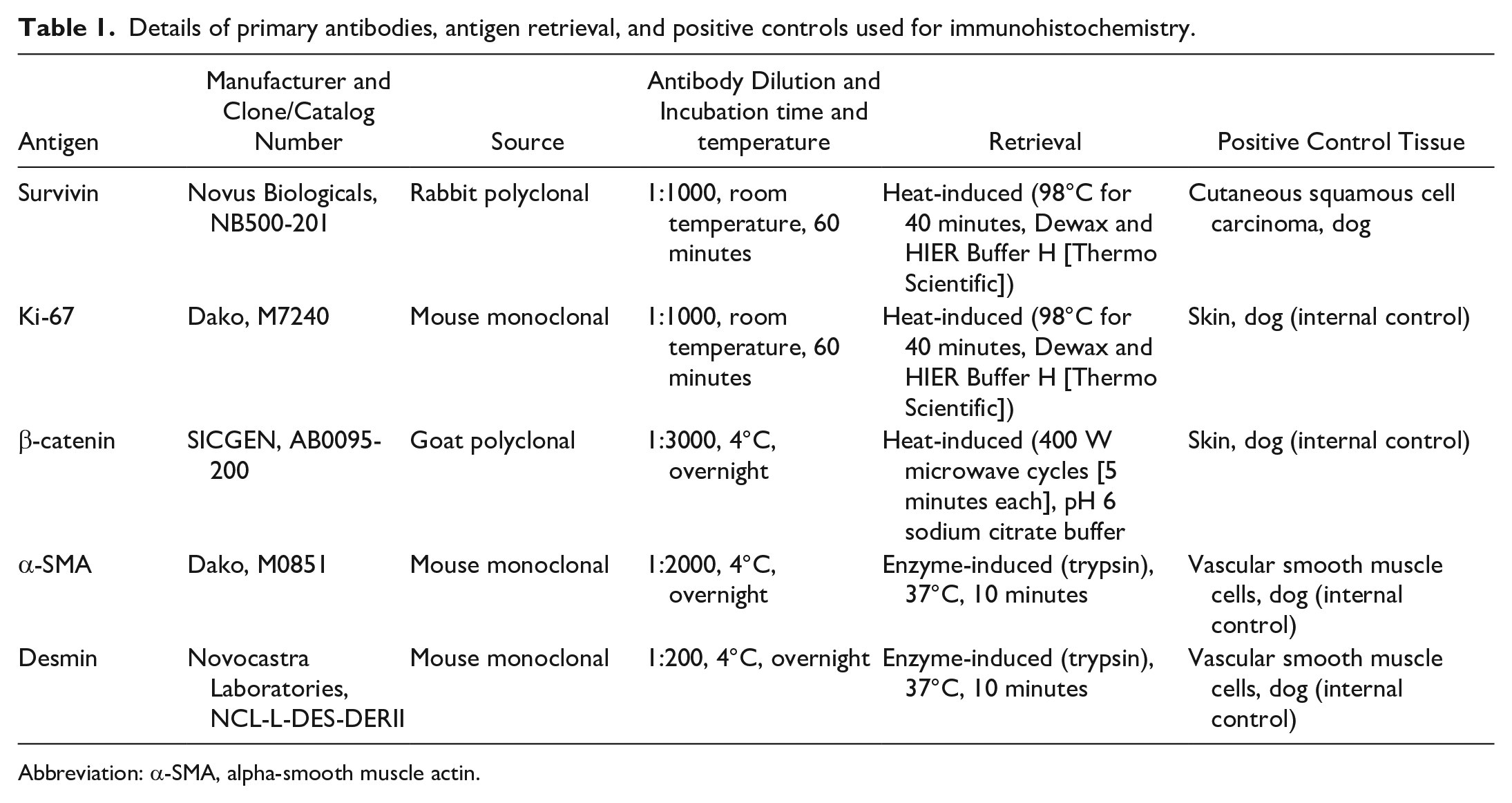
Abbreviation: α-SMA, alpha-smooth muscle actin.
Evaluation of IHC
For α-SMA and desmin cytoplasmic immunolabeling, tumors were classified as positive (from 10 to% 100% of neoplastic cells with granular to homogeneous positive immunolabeling uniformly distributed throughout the cytoplasm) or negative (< 10% of neoplastic cells with granular to homogeneous positive immunolabeling uniformly distributed throughout the cytoplasm). Immunolabellng of survivin and β-catenin was classified according to the subcellular localization of the positive signal as cytoplasmic or nuclear (no membranous β-catenin expression was observed). For cytoplasmic survivin and β-catenin, percentage of positive neoplastic cells (cells with granular to homogeneous positive immunolabeling uniformly distributed throughout the cytoplasm) was semiquantitatively assessed as previously reported 15 and classified as: 0%, 0%–10%, 11%–50%, 51%–75%, and > 75%. Nuclear survivin, β-catenin, and Ki-67 labeling indexes (LIs) were calculated as the percentage of positive neoplastic nuclei in 1000 tumor cells. Nuclear counts were performed on microscopic digital images acquired by photographic microscope in hot-spot areas at 400X. Hot-spot areas were defined as areas in which the number of positive neoplastic cells nuclei was particularly high relative to the adjacent areas. Nuclear counts were performed in viable regions of the tumor, avoiding poorly cellular, hemorrhagic, edematous, necrotic, and inflamed areas. Image analysis was performed using the manual cell-counting tool of ImageJ 1.53 Software.
Follow-Up Information
Information about the patient’s survival, status of regional lymph nodes, LR, distant metastasis, and death was obtained from the clinical records. Lymph nodes were examined clinically by inspection and palpation to exclude possible lymphadenomegaly. Follow-up information was collected by serial clinical examinations (approximately every 3 months) for the first 2 years after surgery, and by telephone calls to the owner or referring veterinarian thereafter. Time to local recurrence (TLR) was defined as the interval between the date of surgery and the date of cytological/histological confirmed diagnosis of the recurrent cPWT within 2 cm of the scar of the previous excision. Time to regional nodal recurrence was defined as the interval between the date of surgery and the date of cytological/histological confirmed diagnosis of regional lymph node recurrence of the cPWT. Time to distant metastasis was defined as the interval between the date of surgery and the date of confirmed metastatic cPWT to distant lymph nodes (nonregional) or internal organs, obtained by cytological examination or necropsy, and/or presumptive metastasis, based on radiographs or computed tomography, in absence of history of any other malignant neoplasm. Overall survival time (OST) was calculated from the date of surgery to the date of death. Dogs lost to follow-up were censored at the date of the last follow-up. Cause of death was further classified as tumor-related, if spontaneous death occurred or euthanasia was elected due to tumor progression, or tumor-unrelated.
Statistical Analysis
Variables measured on categorical scales were presented as absolute frequencies and percentages. Variables measured on continuous scales were summarized as median, mean, and range. Median follow-up was calculated by the reverse Kaplan-Meier method. 82 The median survival time was calculated by the Kaplan-Meier method. Analysis for LR was performed using a method for competing risks because the occurrence of death without a prior record of LR precludes the observation of the TLR. LR incidence curves were estimated as previously reported, 17 and the Fine and Gray regression model on the subdistribution hazard of LR was used to assess the prognostic role of the variables on the incidence of LR. Survival probabilities were estimated by the Kaplan-Meier method, and the Cox regression model for time until death was used to evaluate the prognostic role of the variables on the instantaneous death risk. Categorical variables with k categories have been included in regression models creating a set of k–1 indicator variables (dummy coding). 39 For the dummy coding, one of the categories had to be defined as a reference. Thus, each of the k–1 indicator variables allows a comparison between the hazard of the event (for the Cox model) or the subdistribution hazard of the event (for the Fine and Gray model) in a category and the hazard of the event (or the subdistribution hazard of the event) in the reference category. The hazard is the “instantaneous” rate of event occurrence at a given time, and it is different from the cumulative incidence of the event at a given time (ie, risk: the probability of the occurrence of the even within the time “t”). The latter is a common clinical measure to evaluate the impact of putative prognostic factors in longitudinal cohort studies. In particular in the presence of competing risks subdistribution hazard is generally considered a measure difficult to be interpreted from a clinical viewpoint. 3 Given the relationship between hazard and cumulative incidence, when hazard ratios (HRs; subdistribution hazard ratio [SDHR]) are significantly different form 1, this implies the ratio between the cumulative incidences (relative risk [RR]) is different from 1.0, nevertheless the magnitude of the RR is different from that of HR. The former is “time dependent” also when the ratio between hazards is considered constant in time. Model results are reported as SDHR for the Fine and Gray model and as HR for the Cox model, with 95% confidence intervals (CIs). To facilitate clinical interpretation of the variables’ prognostic effect on LR and death, the ratio of the model-estimated cumulative incidence of LR at 12 and 24 months, and the ratio of the model-estimated death risk at 12, 24, and 60 months of follow-up (RR) have been added. Continuous variables were included in their original measurement scale. Restricted cubic splines were used to investigate the putative nonlinear effect on log(SDHR) and log(HR). 34 If the contribution of the nonlinear effect was not statistically significant, the results of the model with only the linear term are reported as HR (or SDHR) for a unit increase of the variable. Moreover, model-estimated HRs (SDHRs) were also reported for selected increases of the variables: 3 vs 0, 5 vs 3, 7 vs 5, and 10 vs 7 for mitotic count; 5 vs 2 cm, 7 vs 5 cm, and 10 vs 7 cm for tumor size; 3% vs 0%, 5% vs 3%, and 10% vs 5% for nuclear survivin and Ki-67; and 3% vs 0%, 5% vs 3%, 10% vs 5%, and 20% vs 10% for nuclear β-catenin. All of the considered values were chosen in the observed range of the variables; 95% CIs were reported for all the estimated SDHRs. The corresponding model-based estimates of the RRs at the previously mentioned follow-up times were added. If the contribution of the nonlinear effect was statistically significant, the estimated cumulative incidence of the event (LR or death) as a function of the variable was also plotted, as the regression coefficients could not be interpreted easily in terms of HR or SDHR, and the RR was not constant for a unit increase in the variable. For each estimated SDHR and HR, the null hypothesis of the regression coefficient equal to zero (lack of evidence for prognostic effect of the variable) was evaluated by Wald test. The Wald test was used also to evaluate the contribution of nonlinear effects for continuous variables. As the Fine and Gray model provides reliable results on the prognostic effect of categorical variables when at least 1 event is observed in each of the variable’s categories, the incidence curves of the variable’s categories were compared with Gray’s test in the absence of this condition. In accordance with suggestions for reliable results in time-to-event regression models,2,17,68 only UA for LR and multivariate analysis (MA) with 3 variables for OST were performed. The variables considered for the multivariate models were nuclear survivin, nuclear β-catenin, Ki-67 LI, mitotic count, necrosis, histological grade, histologic margins, type of growth, and tumor size, which were analyzed by separate models in which different combinations of 3 of them were included. For each model, a backward selection procedure was adopted to evaluate which variables were retained as significant prognostic factors (P value for retaining variables in the final model was 0.10). The relationship between nuclear survivin and nuclear β-catenin, Ki-67 LI, histological grade, and mitotic count was evaluated by quantile regression. 101 The model allowed to estimate the median of the distribution of nuclear survivin, conditional to the above-mentioned variables. Histological grade was included in the model by dummy coding. For the variables measured on continuous scale, their association with nuclear survivin was first examined by scatterplots. This was done to evaluate whether a linear term was appropriate to be included in the model. When a complex pattern of association was observed, the range of the variable was divided into 3 classes and included by dummy coding to facilitate the interpretation of model results. The statistical test on the regression coefficients was performed by bootstrap methods (5000 bootstrap samples). Statistical analysis was performed using R-software 72 with appropriate R packages.36,47,96 The significance level was set at 5%.
Results
A complete summary of the results is reported in Supplemental Table S1.
Case Selection, Clinical Data, and Histopathology
From a total of 126 cases of cPWTs retrieved, 41 cases fulfilled the inclusion criteria and were included in the study. Several cases were included in previous cPWT prognostic studies: 26 cases in the work of Chiti et al, 22 25 cases in the work of Avallone et al, 4 and 25 cases in the work of Stefanello et al. 92 Six of 41 of the cases included in this study were not previously included in any other investigation on cPWTs. According to the histomorphologic features listed in the “Material and Methods” section, 29 cases were diagnosed as cPWT.5,76 Although growth patterns typical of NST were not observed in any case included in the study, 12 cases had smaller areas with growth patterns specific for cPWT, and IHC was performed to confirm their diagnosis (see below “Evaluation of IHC”).
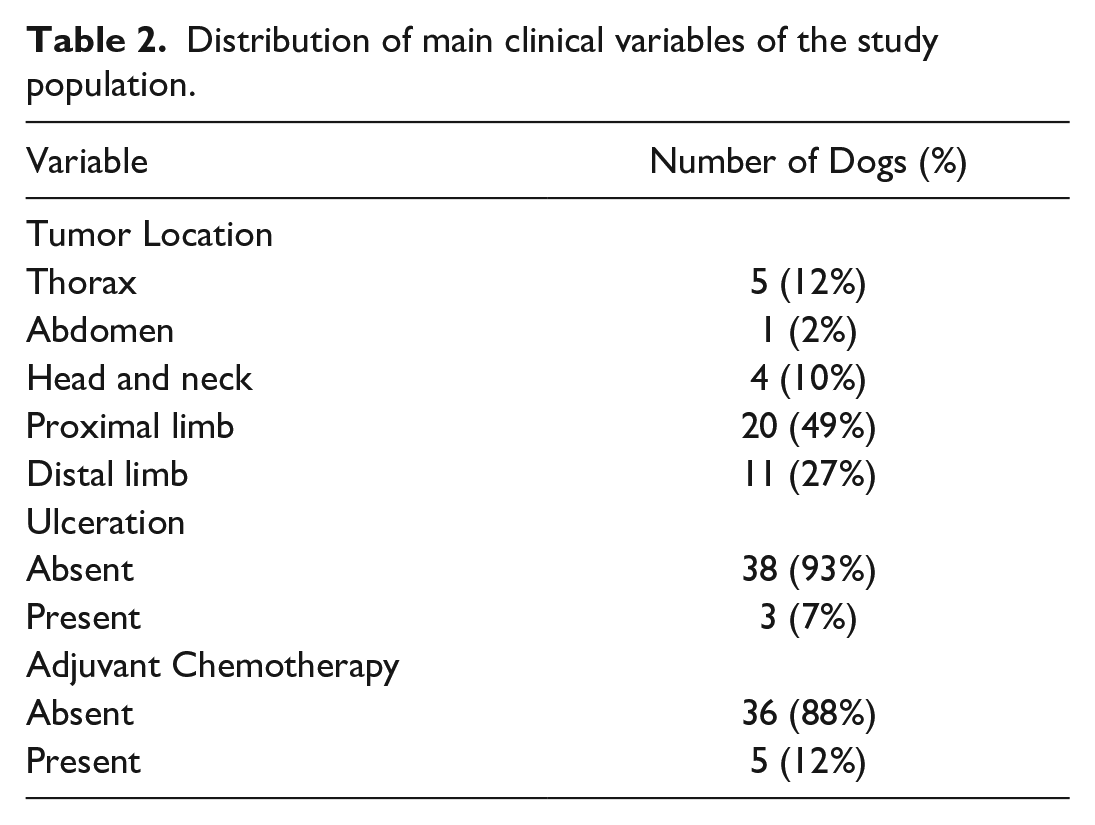
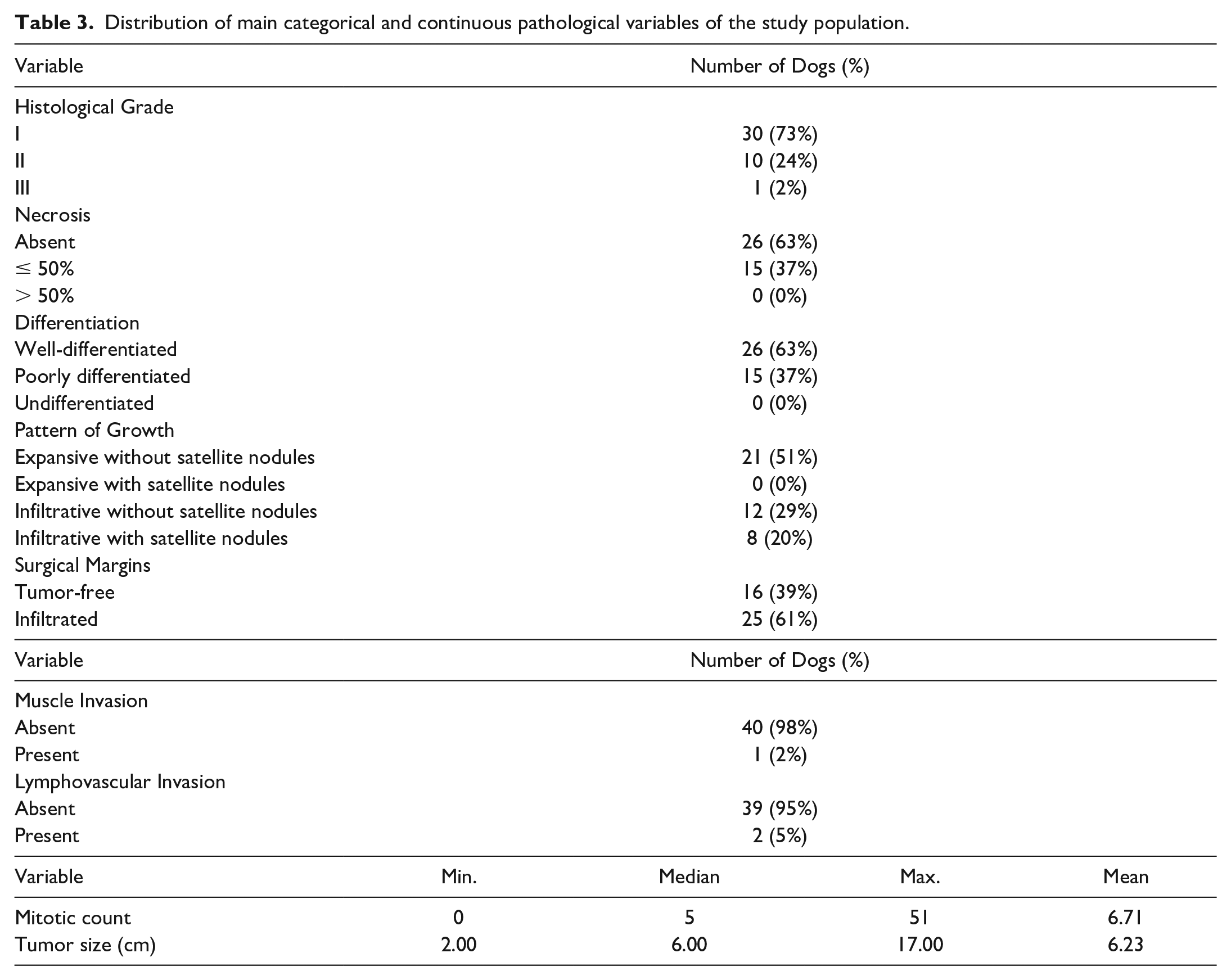
The main clinical and histopathological parameters are summarized in Tables 2 and 3, respectively. At surgery, regional lymph nodes were clinically normal or nonpalpable in all dogs and were not sampled. For statistical purposes, grade II tumors were grouped together with grade III tumor, cases with infiltrative growth were grouped together with cases with infiltrative growth and satellite nodules, and cases located on the abdomen, thorax, head and/or neck, and proximal limb were grouped together and analyzed against cases located on the distal limb. The last grouping was performed based on previous research demonstrating that PWTs located at the extremities (and infiltrating the muscular layer) have the highest hazard of recurrence.4,22
Distribution of main clinical variables of the study population.
Distribution of main categorical and continuous pathological variables of the study population.
Evaluation of IHC
The 12 cases with smaller areas of perivascular growth patterns expressed at least 1 muscular marker and lacked the typical smooth muscle cell morphology and growth patterns (bundles radiating at irregular to 90-degree angles and herringbone patterns),
77
confirming the diagnosis of cPWT. Specifically, 2 cases expressed α-SMA only, 7 cases expressed desmin only, and the remaining 3 cases expressed both α-SMA and desmin. Survivin, β-catenin, and Ki-67 LI immunohistochemical results are summarized in Table 4. Survivin was variably expressed in the nucleus and/or in the cytoplasm of neoplastic cells (Fig. 1a–c). All cases had nuclear expression of survivin ranging from 0.9% to 12.2%. In neoplastic cells undergoing mitosis, the mitotic apparatus labeled positively (Fig. 1b, inset). Cytoplasmic expression of survivin was detected in 31 cases. Nuclear β-catenin expression was observed in 24/41 cases, ranging from 2.9% to 67.2% of neoplastic cells (Fig. 1d). Cytoplasmic β-catenin was expressed in 23 cases (Fig. 1e). No membranous β-catenin expression was observed in any case. Cases with 11%–50%, 51%–75%, and > 75% of neoplastic cells positive for cytoplasmic β-catenin were grouped together (cytoplasmic β-catenin
Immunohistochemical results. Nuclear expression (% of cells) of survivin, β-catenin, and Ki-67 and cytoplasmic expression of survivin and β-catenin.
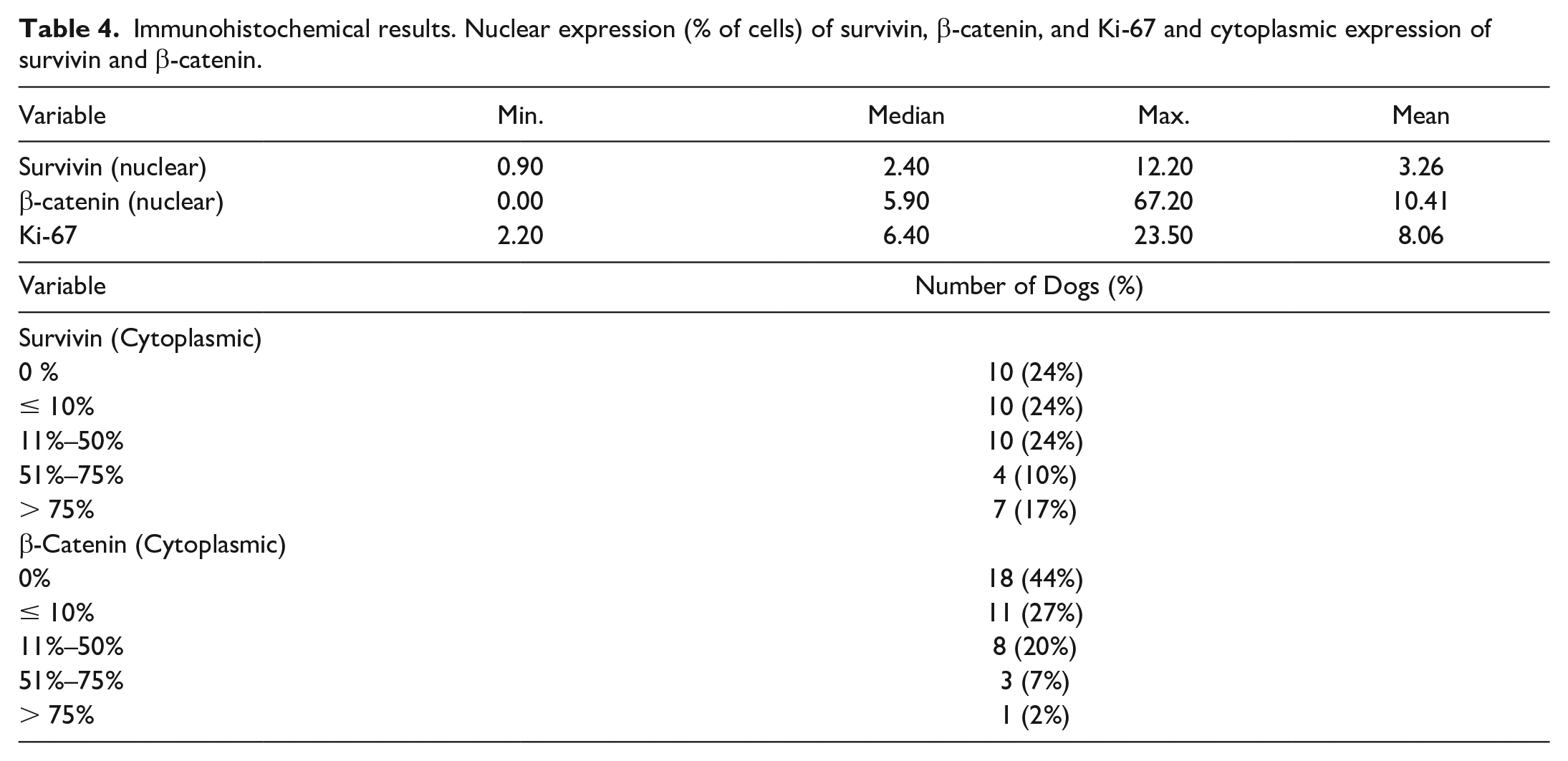
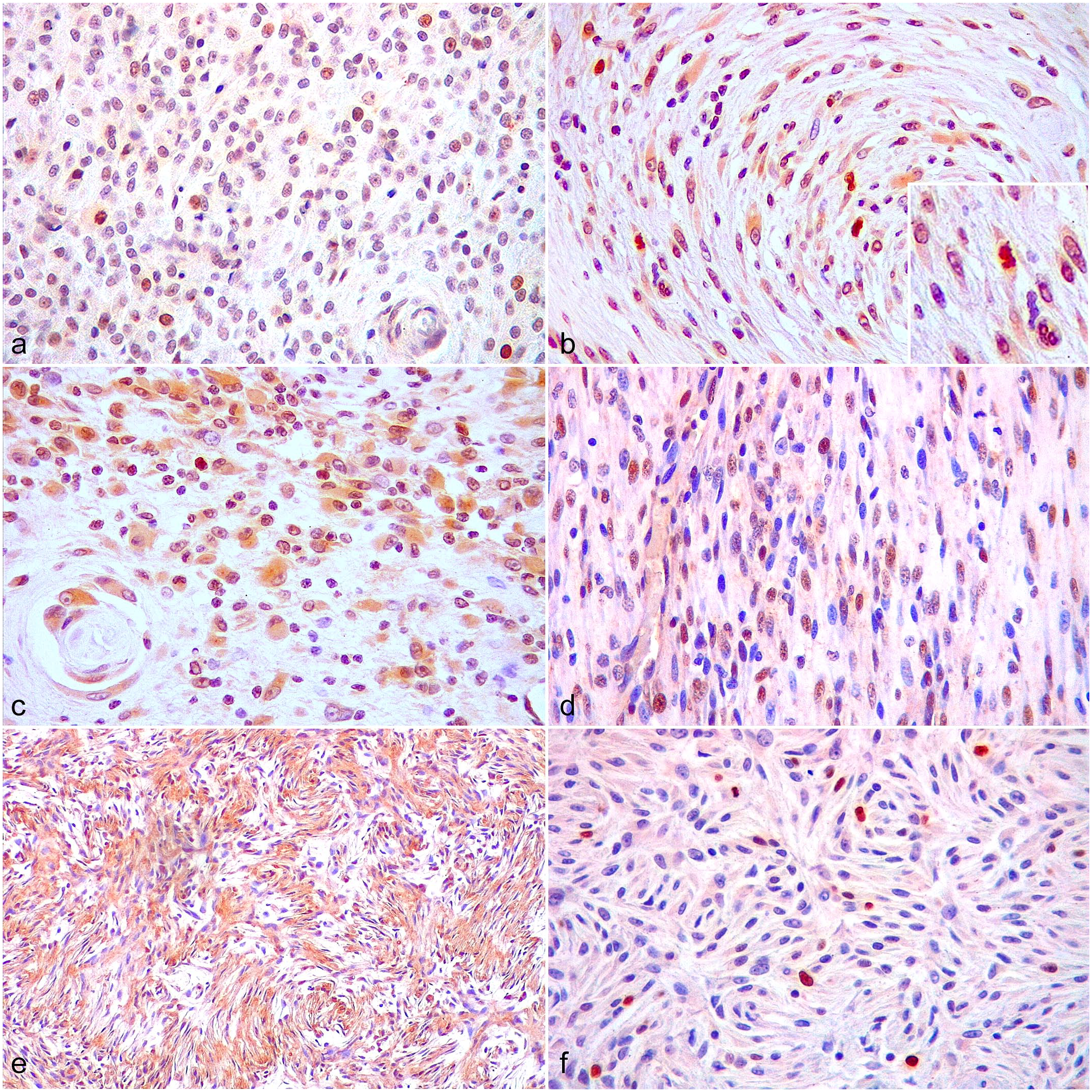
Canine perivascular wall tumors (cPWTs). Immunohistochemistry (IHC). (a) IHC for survivin in a grade II cPWT. There are high numbers of neoplastic cells with nuclear immunolabeling. (b) IHC for survivin in a grade I cPWT. There are scattered neoplastic cells with nuclear immunolabeling including an immunolabeled mitotic figure (inset). (c) IHC for survivin in a grade I cPWT. There are high numbers of neoplastic cells with cytoplasmic immunolabeling. (d) IHC for β-catenin in a grade II cPWT. There are high numbers of neoplastic cells with nuclear immunolabeling. (e) IHC for β-catenin in a grade I cPWT. There are high numbers of neoplastic cells with mainly cytoplasmic immunolabeling. (f) IHC for Ki-67 in a grade I cPWT. There are scattered neoplastic cells with nuclear immunolabeling.
Follow-Up Information
Median follow-up time could not be estimated. The last follow-up for surviving dogs was 30 months (thereafter, times were related to deceased dogs) and the probability for a dog to have follow-up at 30 months was 53%. The median survival time was 24 months (95% CI: 18–48 months). At the end of the study, 30 dogs had died, 7 were alive, and 4 were alive when lost to follow-up at 15, 330, 365, and 393 days. Survival probability at 12 months was 70% (95% CI: 57%–86%), at 24 months was 48% (95% CI: 34%–68%), and at 80 months was 5% (95% CI: 0.7%–30.7%). Twenty-one dogs died without developing LR and for these patients the cumulative incidence of death at 84 months was 70%. For these 21 dogs, death prevented the observation of TLR; thus, death was considered as a competing risk for the estimated cumulative incidence of LR.
LR was recorded in 11 cases. The first LR was observed at 25 days, and the last at 870 days. The cumulative incidence of LR at 12 months was 15% (95% CI: 3.8%–26.2%), at 24 months was 26% (95% CI: 12%–41%), and at 80 months was 30% (95% CI: 14%–45%) (Fig. 2). None of the dogs had regional lymph node involvement. Suspected distant metastases were observed in 4 dogs between 170 and 512 days postoperatively and were all located in the lungs (all detected by thoracic radiographs as a multiple nodular pattern). Of a total of 30 deceased dogs, cause of death was considered tumor-related in 7 dogs. Of these, 3 had LR, 3 developed suspected distant metastasis (assessed by radiographs), and 1 dog had concomitant LR and suspected distant metastasis. Twenty-three dogs died of nontumor-related causes, of which the most common were nephropathy and heart disease. Three dogs developed other neoplasms (lymphoma, oral melanoma, and appendicular osteosarcoma) and, according to the owners, were euthanized because of the low quality of life due to end-stage oncological disease. All cases that developed suspected metastases were euthanized in accordance with the owner. Of the cases that developed LR (11/41), 1 dog was alive at the end of the study, 1 dog was lost to follow-up at 393 days, 3 dogs died due to LR, 1 dog died due to suspected multiple lung metastases, and 4 dogs died from tumor-unrelated causes (1 nephropathy, 3 heart diseases, and 1 undetermined cause).
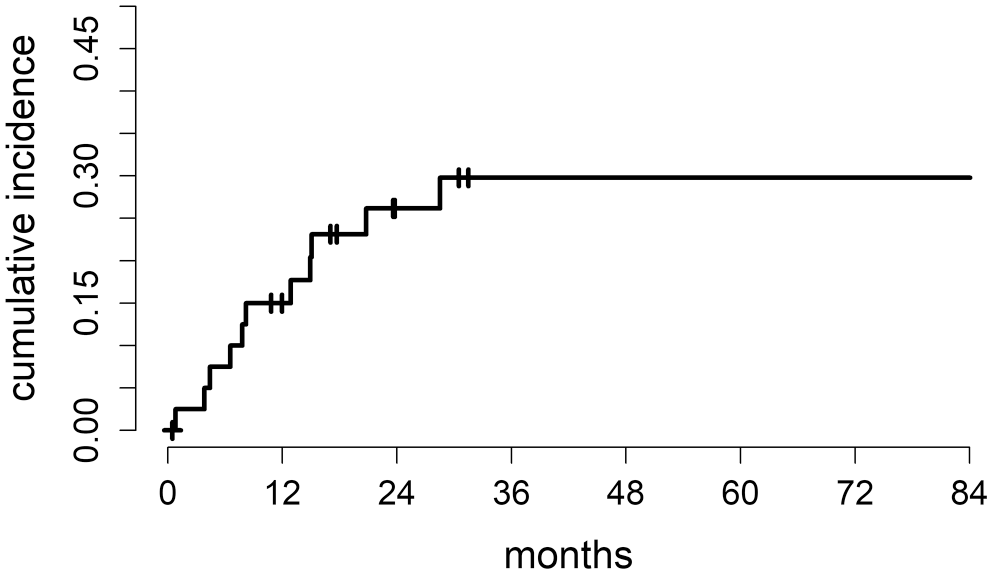
Cumulative incidence of local recurrence estimated by the method for competing risks. Continuous line represents the cumulative incidence of local recurrence. Tick marks indicate censored patients.
Association Between Nuclear Survivin and Other Variables
The association between nuclear survivin and histologic grade was statistically significant. The estimated median survivin for grade I tumors was 2.4 and increased to 7.6 for grade II/III neoplasms (P = .043). The association between nuclear survivin, nuclear β-catenin, Ki-67 LI, and mitotic count was not statistically significant. The relationship between nuclear survivin and nuclear β-catenin was complex, and a specific trend was not evident; thus, nuclear β-catenin was partitioned in 3 classes. The estimated median of nuclear survivin was 2.8 for β-catenin = 0 (reference class); it decreased to 2.2 (P = .418) for the class of 0 < β-catenin ≤ 15 and to 2.0 (P = .315) for the class of β-catenin > 15. The estimated increase of median nuclear survivin was 0.105 for each unit increase of Ki-67 LI (P = .418). For each unit increase of mitotic count, the estimated increase of median nuclear survivin was 0.083 (P = .369).
Prognostic Impact of Variables on TLR
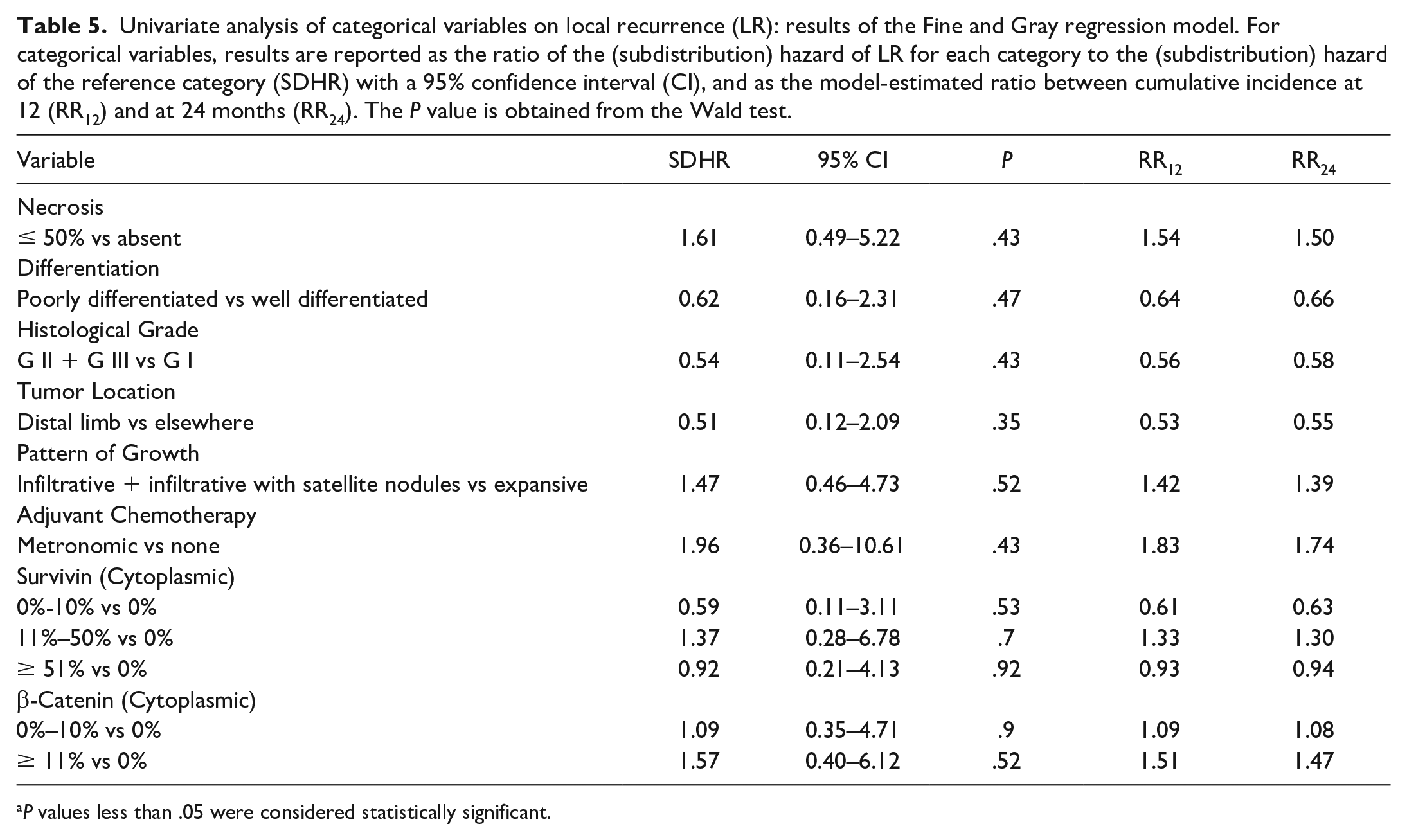
Since the small number of LRs precluded a MA, only a UA was conducted. Results of UA are reported in Tables 5 and 6. Tumor size was a significant prognostic factor for LR. The cumulative incidence of LR increased with increasing tumor size: for each cm increase, SDHR was 1.19 (P = .02). Considering a 3 cm increase in tumor size, at 12 months postdiagnosis, the RR between tumors that measured 2 and 5 cm in greatest dimension was 1.65, and between tumors that measured 7 and 10 cm in greatest dimension was 1.60. The RR was slightly lower at 24 months.
Univariate analysis of categorical variables on local recurrence (LR): results of the Fine and Gray regression model. For categorical variables, results are reported as the ratio of the (subdistribution) hazard of LR for each category to the (subdistribution) hazard of the reference category (SDHR) with a 95% confidence interval (CI), and as the model-estimated ratio between cumulative incidence at 12 (RR12) and at 24 months (RR24). The P value is obtained from the Wald test.
P values less than .05 were considered statistically significant.
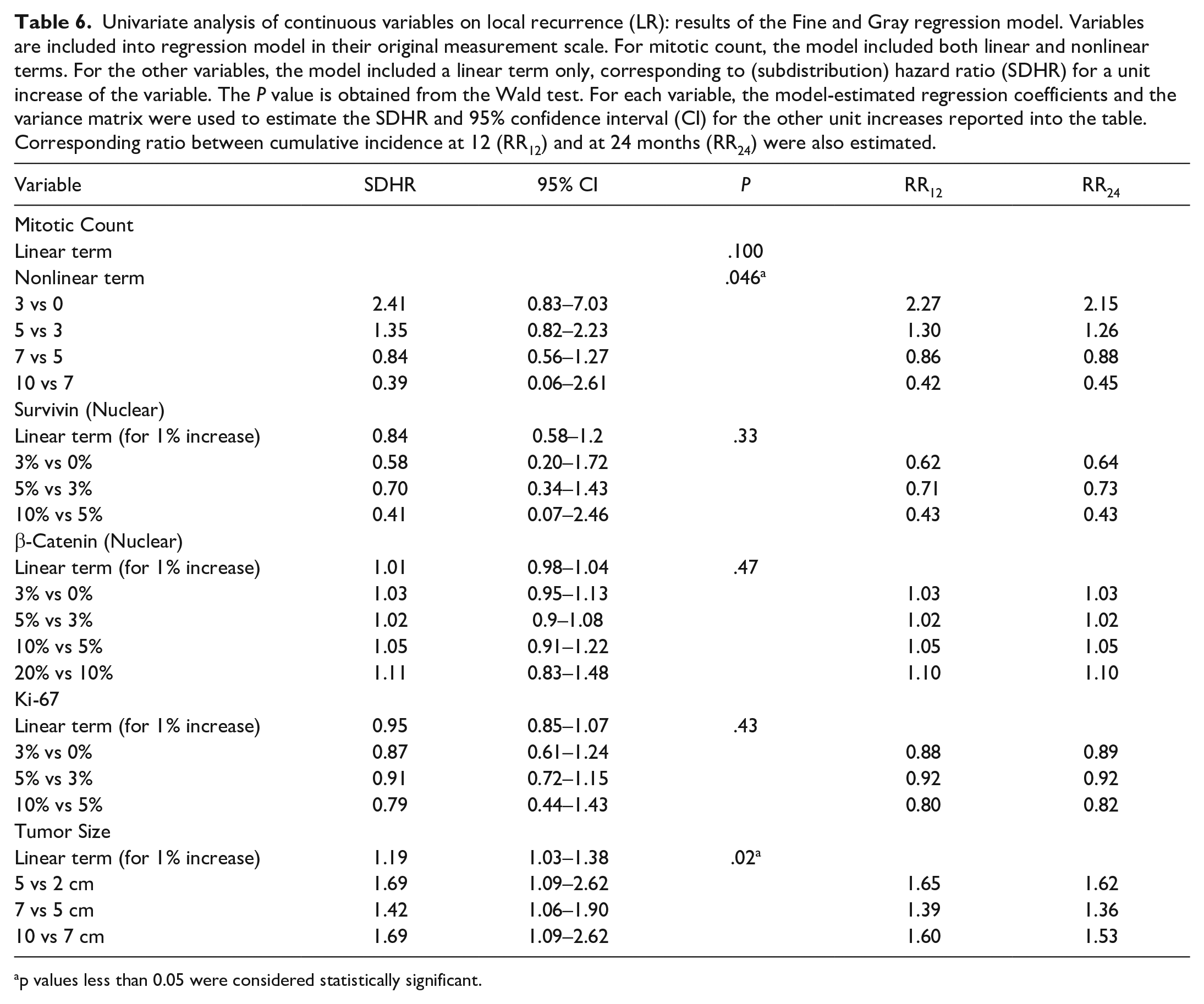
Univariate analysis of continuous variables on local recurrence (LR): results of the Fine and Gray regression model. Variables are included into regression model in their original measurement scale. For mitotic count, the model included both linear and nonlinear terms. For the other variables, the model included a linear term only, corresponding to (subdistribution) hazard ratio (SDHR) for a unit increase of the variable. The P value is obtained from the Wald test. For each variable, the model-estimated regression coefficients and the variance matrix were used to estimate the SDHR and 95% confidence interval (CI) for the other unit increases reported into the table. Corresponding ratio between cumulative incidence at 12 (RR12) and at 24 months (RR24) were also estimated.
p values less than 0.05 were considered statistically significant.
The relationship between LR subdistribution hazard and mitotic count was complex, and the contribution of the nonlinear term was statistically significant (P = .046). The incidence of LR increased up to 5 mitoses and then began to decrease as mitoses increased over 5 (Supplemental Figure S1). Considering an increase of 3 mitoses, the estimated cumulative incidence of LR for a tumor with a mitotic count of 3 was 2.27 and 2.15 times higher than a tumor with a mitotic count of 0 at 12 and 24 months, respectively, and the estimated cumulative incidence of LR for a tumor with a mitotic count of 10 was 0.42 and 0.45 times lower than a tumor with a mitotic count of 7 at 12 and 24 months, respectively. None of the cases with histologic tumor-free margins experienced LR, and all the cases that had LR had infiltrated margins. For this reason, the univariate regression model could not be used. Of 25 cases with infiltrated histologic margins, 11 cases developed LR while 14 cases did not. The comparison between LR incidence of infiltrated and tumor-free margins was performed by nonparametric Gray’s test (χ2 = 9, P = .003).
The number of ulcerated tumors, tumors invading the underlying musculature, and tumors with lymphovascular invasion were just 3, 1, and 2, respectively; thus, the statistical model was not applied. The increases of nuclear survivin and nuclear Ki-67 LI were associated with a nonstatistically significant lower incidence of LR. The association of cytoplasmic survivin and cytoplasmic β-catenin with the incidence of LR was not statistically significant. Regarding nuclear β-catenin, even considering increments of 10 units, the RR was always about 1; thus, the estimated prognostic contribution of the variable was negligible.
Prognostic Impact of Variables on OST
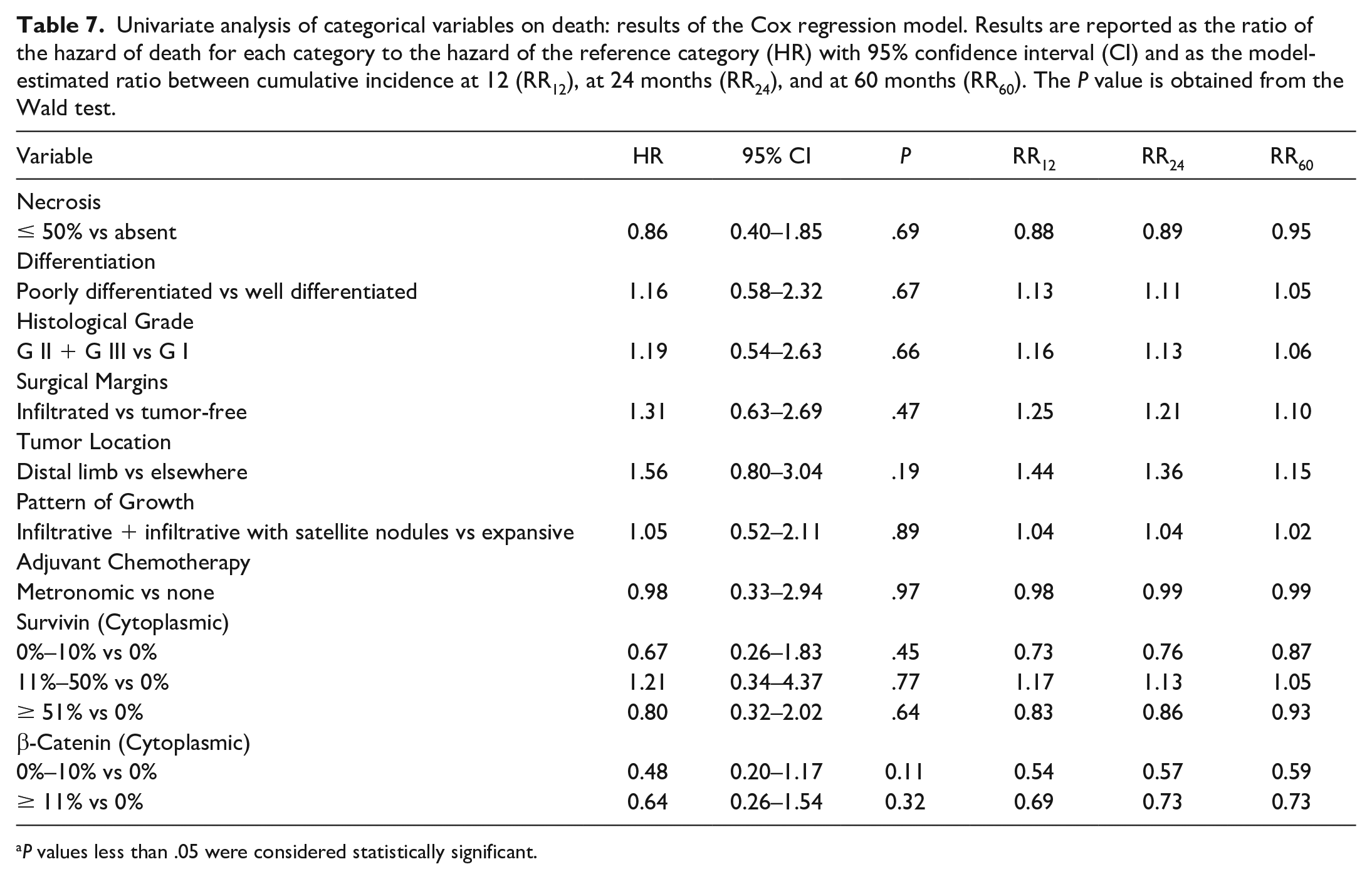
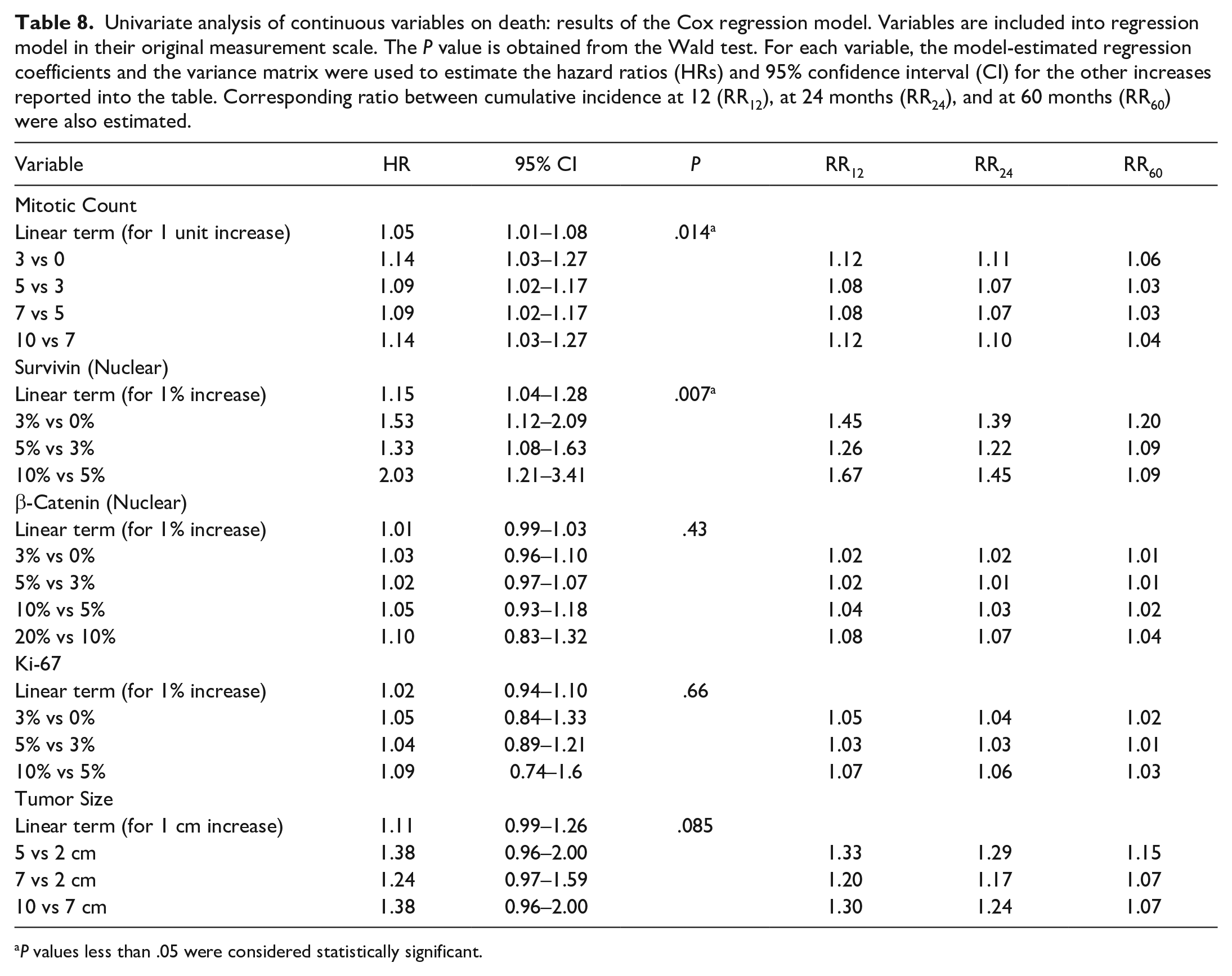
Results of UA are reported in Tables 7 and 8. Results of MA are reported in Supplemental Tables S2 and S3. Mitotic count was a significant prognostic factor. The model contribution of nonlinear term was not statistically significant (P = .29), thus a model with linear term only was reported. A unitary increase of mitotic count was associated with an increase of the instantaneous death risk by a factor of 1.05 (HR = 1.05; P = .014). Considering an increase of 3 mitoses, the RR of death was 1.12 at 12 months, both for the comparison between 3 and 0 mitoses and between 10 and 7 mitoses. For the above-mentioned comparison, RRs were slightly lower at 24 and 60 months. Mitotic count maintained its prognostic role when adjusted for nuclear survivin, nuclear β-catenin, Ki-67 LI, tumor size, status of histologic margins, type of growth, histological grade, and necrosis score in MA. In all considered multivariable models with 3 variables, mitotic count was always selected in the final model by backward selection procedure. In UA, although not statistically significant, the incidence of death increased with the increase of tumor size. Considering an increase of 3 cm, the estimated RRs at 12 months were higher than those at 24 and 60 months. At 12 months, the estimated cumulative incidence of death for a 5 cm tumor was 1.33 times higher than for a 2 cm tumor, and the estimated cumulative incidence of death for a 10 cm tumor was 1.30 times higher than for a 7 cm tumor. In MA, a statistically significant increase in the incidence of death was observed with increased tumor size when adjusted for nuclear survivin, nuclear β-catenin, Ki-67 LI, mitotic count, status of histologic margins, type of growth, histologic grade, and necrosis score. In all considered multivariable models with 3 variables, tumor size was always selected in the final model by backward selection procedure. Grade II/III tumors, tumors with infiltrated margins, tumors characterized by infiltrative and/or satellite neoplastic nodular growth, and the presence of tumor necrosis had a nonstatistically significant prognostic effect both in UA and MA. Tumor location, tumor differentiation and administration of adjuvant chemotherapy (metronomic) were evaluated in UA only and their association with prognosis was not statistically significant. According to the UA, the increase of nuclear survivin was associated with an increase of the instantaneous death risk (HR = 1.15, P = .007). In particular, the estimated cumulative incidence of death for a dog with a tumor that had 3% positive nuclei was 1.45, 1.39, and 1.2 times higher than for a dog with a tumor that had 0% positive nuclei at 12, 24, and 60 months, respectively. The estimated cumulative incidence of death for a dog with a tumor that had 10% positive nuclei was 1.67, 1.45, and 1.09 times higher than of a dog with a tumor that had 5% positive nuclei at 12, 24, and 60 months, respectively. Nuclear survivin maintained its prognostic role when adjusted for nuclear β-catenin, Ki-67 LI, mitotic count, tumor size, status of histologic margins, type of growth, histologic grade, and necrosis score in MA. Nuclear survivin was selected by backward procedure in all models with 3 variables in which it was included. Nuclear β-catenin and Ki-67 LI did not have a significant prognostic role in both UA and MA. Cytoplasmic survivin and β-catenin were evaluated in UA only, and no significant prognostic role was found.
Univariate analysis of categorical variables on death: results of the Cox regression model. Results are reported as the ratio of the hazard of death for each category to the hazard of the reference category (HR) with 95% confidence interval (CI) and as the model-estimated ratio between cumulative incidence at 12 (RR12), at 24 months (RR24), and at 60 months (RR60). The P value is obtained from the Wald test.
P values less than .05 were considered statistically significant.
Univariate analysis of continuous variables on death: results of the Cox regression model. Variables are included into regression model in their original measurement scale. The P value is obtained from the Wald test. For each variable, the model-estimated regression coefficients and the variance matrix were used to estimate the hazard ratios (HRs) and 95% confidence interval (CI) for the other increases reported into the table. Corresponding ratio between cumulative incidence at 12 (RR12), at 24 months (RR24), and at 60 months (RR60) were also estimated.
P values less than .05 were considered statistically significant.
Discussion
This study investigated the potential prognostic impact of the immunohistochemical expression of survivin, β-catenin, and Ki-67 and other histopathological and clinical variables in a series of 41 cPWTs. The main limitations of the study were the relatively small sample size (41 cases) and the relatively low number of events recorded for LR (11/41), suspected metastasis (4/41), and tumor-related death (7/30), despite a long-term postsurgical follow-up. This point hampered the power of UA and MA, with the latter being impossible for LR analysis. Because of these limitations, the results presented here should be regarded as preliminary and need to be validated by further studies on a larger caseload with a larger number of recorded events. The low number of events seems to be an intrinsic limit of studies on cPWTs, which are characterized by rarely reported pulmonary metastases and relatively low rates of LR.4,7,22,92 One study estimated the probabilities of being free from LR of 98% at 6 months, 92% at 1 year, 80% at 2 years, and 76% at 3 years postsurgery, indicating that LRs in cPWTs are more likely to develop many years after surgery. 92 As a consequence, it is very likely that some of the patients in our study died of unrelated conditions before developing LR. In addition, since cPWTs are characterized by infrequent tumor-related deaths, the analysis of OST should be cautiously interpreted. Other factors that may have contributed to the low number of cases included in this investigation and the subsequent low number of events are certainly the stringent inclusion and exclusion criteria applied, and the mono-institutional nature of the study. Nevertheless, despite all the aforementioned limitations, this study has the merit of being based on a very homogeneous caseload with accurate, complete, and very long follow-ups, which are rare in veterinary literature.
One of the main endpoints of the analysis in this study was TLR, which is an interval-censored variable, meaning that the exact date of LR cannot be detected with adequate precision as it is known to lie within the interval between the last medical examination without LR and its diagnosis. The presence of variable intervals between follow-up visits for different patients is a common situation in longitudinal studies, especially in observational ones. If all patients had the same follow-up visits, using the intervals’ midpoints or the date of event detection would not matter, and the conclusions obtained with methods for right-censored data would be (nearly) the same as those obtained with methods for interval-censored data. However, even in clinical trials where follow-up visits are planned to be at fixed intervals, not all patients stick to the plan. Interval censored analysis should always be applied, as it would give more accurate results, but this method is not commonly used. In this investigation, the standard method was applied, which is a limitation common to other observational studies.
In this study, cPWTs variably expressed survivin in the nucleus and/or cytoplasm, suggesting a potential role of the molecule in cPWT neoplastic transformation and/or progression. Increased expression of nuclear survivin was significantly associated with decreased OST by UA and MA, suggesting that survivin is an independent predictor of survival. This result parallels reports in humans, in which nuclear and/or cytoplasmic survivin expression has been frequently correlated with reduced OST or disease-free survival, LR, and metastasis for many tumors,70,102 including STTs.43,44,94,98 Increased survivin measured by ELISA and western blot and increased survivin mRNA expression have been associated with decreased OST and tumor-related death in human STTs.43,44 In dogs, survivin expression has been previously demonstrated in epithelial,12,13,16,71,74 melanocytic, 14 mesenchymal,15,64,86 and round cell tumors81,90,91,103 but has not been thoroughly investigated in STTs in general or in cPWTs in particular. In dogs, an increase in nuclear survivin immunohistochemical labeling has been statistically correlated with histological features of malignancy, presence of metastasis, and tumor-related death in cutaneous melanocytic tumors, 14 while an association between nuclear and/or cytoplasmic survivin evaluated by IHC and survival has not been proven in canine mast cell tumors. 81 Contradictory results have been reported for canine osteosarcoma, as one study identified a correlation between high survivin score (calculated combining IHC labeling intensity and percentage of positive cells with nuclear and/or cytoplasmic labeling) and a decreased disease-free interval, 86 while another publication reported that cases with moderate/high nuclear survivin immunohistochemical expression had a tendency toward a longer OST, suggesting survivin as a potential positive prognostic indicator. 15
Increased nuclear and/or cytoplasmic survivin immunolabeling has been frequently associated with LR in humans.84,107 Thus, the finding that increased nuclear survivin in cPWTs had a trend toward a decreased risk of developing LR was unexpected. The same trend was also observed for increased mitotic count and Ki-67 LI. However, these results should be carefully interpreted, considering the competing effect of death without previous observation of LR, for which a reversed prognostic trend was observed. In this study, the high number of dogs that died without LR may be explained by death from nontumor-related causes impeding time for LR development. In addition, LR in cPWTs is influenced by many variables including tumor size, histological grade, necrosis, mitotic count, histologic tumor-free margins, tumor location, and ulceration.4,22,89 Since MA was not possible in our study, it is not possible to draw definitive conclusions on the role of survivin, Ki-67, and mitotic count on the risk of LR in cPWTs.
Survivin can enhance cancer cell metastatic potential, 87 and its overexpression has been associated with development of metastasis in human and canine cancers.14,24,61 Unfortunately, even though the 4 cases in our caseload that developed suspected metastasis after surgery expressed nuclear (> 7.5%) and cytoplasmic (> 50%) survivin, the overall number of suspected metastatic cases was too low to perform a statistical analysis.
The expression of both nuclear and cytoplasmic survivin by neoplastic cells suggests that survivin may have a dual role in cPWT tumorigenesis, both as an inhibitor of apoptosis and as a cell cycle regulator. Cytoplasmic survivin showed no association with LR and OST. The finding of a significant role of nuclear survivin in predicting a shorter OST and the lack of correlation between cytoplasmic survivin and any considered endpoint parallels the contrasting results previously reported in human and veterinary medicine.14,15,52,81,86 Specifically, differing correlations between survivin subcellular localization (cytoplasmic or nuclear) predominantly detected by IHC and favorable or poor prognosis have been reported.14,15,52,81,86 Such differences could be related to different methods in assessing survivin expression and possibly to the presence of splice variants with different functions.80,81 Each of these variants has different intracellular localizations, expression patterns, and can even form heterodimers with “wild-type” survivin exerting a pro-apoptotic function.79,80 Furthermore, the most utilized antibody against canine survivin does not discriminate the splice variants of the protein,12–16,86,103 although this may not bear much relevance, as the function of the corresponding proteins has not been fully investigated.
Last but not least, survivin has been extensively studied as a potential therapeutic target in humans.51,56 Several studies have also been conducted on canine tumors, in vitro,62,63,66,85,104,105 on xenograft models,66,85,106 and in dogs with spontaneous lymphoma 95 with promising results. Given that high survivin expression in cPWTs was associated with a shorter OST, canine patients with aggressive cPWTs may be candidates for survivin-targeted therapies in the future.
Survivin is a target gene of the Wnt/β-catenin signaling pathway, 46 whose aberrant regulation has been recently implicated in oncogenesis of several human soft tissue sarcomas, including fibrosarcoma, leiomyosarcoma, liposarcoma, and synovial sarcoma.19,54,55,100 In this study, nuclear and cytoplasmic β-catenin were variably expressed in cPWTs, suggesting aberrant activation of this pathway, possibly leading to transcription of target genes involved in cellular proliferation, including survivin.28,46 In human medicine, despite the fact that several studies have demonstrated a strong expression of β-catenin in different STTs,19,48,50,54,55,100 its prognostic role has not yet been clearly recognized. 48 Although there have been few clinical trials with Wnt/β-catenin signaling pathway inhibitors in human STT patients, it has been hypothesized that patients with an over-activated pathway may benefit from Wnt/β-catenin inhibitor therapy. 55 β-catenin has been reported as being consistently expressed in canine fibrosarcomas. 65 However, the subcellular localization and the association with prognosis were not reported. 65 In our cPWT caseload, nuclear and cytoplasmic β-catenin expression did not have a statistically significant prognostic impact on LR or OST. Additional studies on a larger number of cases and events are needed to further investigate the prognostic role of this molecule in cPWTs. Analysis of survivin and β-catenin reciprocal immunohistochemical expression in canine malignancies has failed to identify an association between these 2 molecules.14–16 In this study, the exploratory statistics found a complex, noninterpretable relationship between the 2 molecules’ nuclear expression, possibly suggesting that survivin gene transcription is not regulated by β-catenin in cPWTs.In addition, survivin expression has been reported to be associated with tumor grade,42,44 mitotic count, 32 and Ki-67 LI. 91 Our exploratory analysis identified a statistically significant relationship only between nuclear survivin and tumor grade. The lack of statistical significance and the weak associations found between nuclear survivin and mitotic count or Ki-67 LI may be partially explained by the small sample size and the high variability of mitotic counts and Ki-67 LI due to tumor heterogeneity.
One study has evaluated Ki-67 immunohistochemical expression in cPWTs but has not investigated the correlation between its expression and prognosis. 6 High Ki-67 LI has been considered as a negative prognostic factor in canine STTs, as it is associated with decreased OST in UA. 30 Similarly, in this study, increased nuclear Ki-67 LI has a nonstatistically significant trend toward a shorter OST in UA but was not prognostic in any multivariate model.
Our results confirmed the association between increasing mitotic count and shorter OST, as previously reported, 22 on both UA and MA.
Tumor size was confirmed to be prognostic by predicting potential LR in UA and showed a tendency toward a shorter OST.4,22,92 The latter finding was supported by MA, where tumor size was prognostic for reduced OST when adjusted for nuclear survivin, nuclear β-catenin, Ki-67 LI, mitotic count, status of histologic margins, type of growth, histologic grade, and necrosis score. Noteworthy and to the author’s knowledge, this is the first study providing evidence that tumor size is prognostic for a shorter OST in cPWTs.
In conclusion, this is the first study exploring the prognostic value of survivin, β-catenin, and Ki-67 immunohistochemical expression in cPWTs. Immunohistochemical results suggest that survivin and β-catenin are involved in cPWTs tumorigenesis and that nuclear survivin evaluated by IHC may be a useful prognostic marker in predicting a shorter OST in cPWTs. Further studies on the expression of survivin and β-catenin, at both the mRNA and protein levels, on a larger number of cases with a higher number of events (LR, metastasis, and tumor-related deaths) are required in order to validate these results. Specifically, after the establishment of validated cut-offs, inclusion of survivin and/or β-catenin expression assessment in cPWTs may allow for better definitions of patient prognosis and eventually help in planning adjuvant treatments in selected dogs. In addition, given their role in cPWTs pathogenesis, survivin and β-catenin may be potential therapeutic targets for more aggressive phenotypes of these tumors.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241246981 – Supplemental material for Survivin, β-catenin, and ki-67 immunohistochemical expression in canine perivascular wall tumors: Preliminary assessment of prognostic significance
Supplemental material, sj-pdf-1-vet-10.1177_03009858241246981 for Survivin, β-catenin, and ki-67 immunohistochemical expression in canine perivascular wall tumors: Preliminary assessment of prognostic significance by Francesco Godizzi, Federico Armando, Patrizia Boracchi, Giancarlo Avallone, Damiano Stefanello, Roberta Ferrari, Lavinia E. Chiti, Andrea Cappelleri, Clarissa Zamboni, Silvia Dell�Aere, Attilio Corradi and Paola Roccabianca in Veterinary Pathology
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858241246981 – Supplemental material for Survivin, β-catenin, and ki-67 immunohistochemical expression in canine perivascular wall tumors: Preliminary assessment of prognostic significance
Supplemental material, sj-xlsx-1-vet-10.1177_03009858241246981 for Survivin, β-catenin, and ki-67 immunohistochemical expression in canine perivascular wall tumors: Preliminary assessment of prognostic significance by Francesco Godizzi, Federico Armando, Patrizia Boracchi, Giancarlo Avallone, Damiano Stefanello, Roberta Ferrari, Lavinia E. Chiti, Andrea Cappelleri, Clarissa Zamboni, Silvia Dell�Aere, Attilio Corradi and Paola Roccabianca in Veterinary Pathology
Footnotes
Author Contributions
FG and FA planned the study.
DS, RF and LC collected the clinical data and follow-ups.
FG, FA, and AC (Andrea Cappelleri) performed the experiments.
PR and AC (Attilio Corradi) supervised the experiments.
PB performed the statistical analysis.
FG, FA, PB, GA and PR analyzed and interpreted the data.
FG and FA prepared the article with contribution from the other authors.
PB, GA, DS, RF, LC, AC (Andrea Cappelleri), CZ, SD, AC (Attilio Corradi), and PR revised the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material for this article is available online.
Data Availability
All paper-related data are available upon reasonable request to the corresponding author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
